Frontiers in Bioscience-Elite (FBE) is published by IMR Press from Volume 13 Issue 2 (2021). Previous articles were published by another publisher on a subscription basis, and they are hosted by IMR Press on imrpress.com as a courtesy and upon agreement with Frontiers in Bioscience.
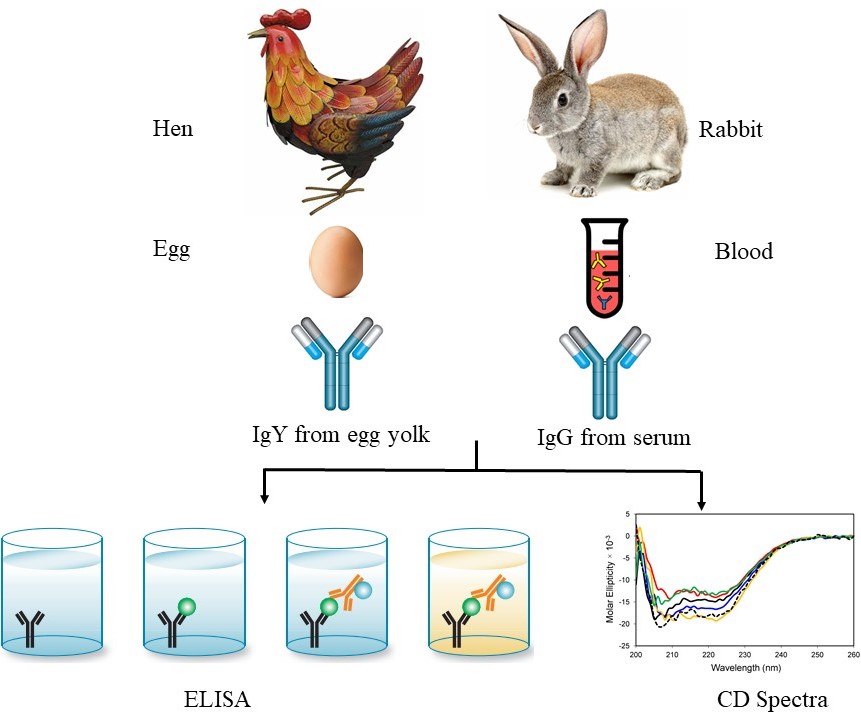
Antibodies play a vital role in a variety of applications from diagnostics, imaging, and therapeutics. The stability of antibodies is one of the most important key attributes for its prolonged activity and functionality. Here, we demonstrate a detailed comparative study of the molecular stability of the rabbit immunoglobulin G (IgG) and chicken egg yolk immunoglobulin Y (IgY) at different pH, temperatures, and time points. The molecular stability of IgG and IgY is compared on the basis of its binding activity and conformational changes. The optimum temperature and time were found to be 30 °C, and 37 °C, up to 8 h for both IgY and IgG antibodies. With increasing temperature and time, IgG antibodies were found to be significantly less stable in comparison to IgY antibodies. IgY is almost twenty-fold more stable than IgG at 60° C for up to 8 hours. The extra domain present in the heavy chain of IgY plays a significant role in providing more molecular stability as compared to IgG under the above-mentioned experimental conditions. The results show that, as compared with IgG, the IgY are more stable, are less expensive to make in high yield and exhibit minimal conformational changes and hence are cost effective for use in a diverse array of purposes.