1 Institute of College of Pharmacy, Changchun University of Traditional Chinese Medicine, 130117 Changchun, Jilin, China
2 Ginseng Research Institute, Changchun University of Traditional Chinese Medicine, 130117 Changchun, Jilin, China
Abstract
Background: Disorders of purine metabolism are the main cause of
hyperuricemia. Current drugs for the treatment of hyperuricemia usually cause a
degree of cardiovascular damage. Methods: This study aimed to investigate
the therapeutic effects of Armillaria mellea fruiting body (AFB),
Armillaria rhizomorph (AR) and Armillaria mellea fermentation
product (after rhizomorphs removal) (AFP) on hyperuricemic mice. The
hyperuricemia mouse model was established by oral administration of potassium
oxonate 0.9 g
Keywords
- medicinal mushrooms
- Armillaria mellea
- serum uric acid
- transporter protein
Disorders of purine metabolism cause hyperuricemia, a disease of recent concern [1]. Nationally, representative National Health and Nutrition Examination Survey data showed the prevalence of hyperuricemia in the United States was 20.2% for men and 20.0% for women in 2015–2016 [2]. A survey showed the co-prevalence of hyperuricemia was 13.3% on the Chinese mainland during 2000–2014 [3]. Hyperuricemia is associated with cardiovascular disease [4], kidney damage [5], diabetes [6, 7], hypertension [8], dyslipidemia [9], rheumatism, and other diseases [10].
Uric acid is a product of the purine metabolic pathway. Uric acid is deposited in the form of sodium salts in joints, soft tissues, cartilage, and kidneys, resulting in elevated uric acid levels in the body that gradually evolves into hyperuricemia and other diseases when the human purine metabolism is disordered and serum uric acid (SUA) levels rise sharply. Research has shown that uric acid in the renal interstitium enters tubular epithelial cells in a retroelectrochemical gradient through organic anion transporter 1 (OAT1) and organic anion transporter 3 (OAT3) in the basement membrane of epithelial cells, while intracellular ketoglutaric acid is excreted into the renal interstitium, thereby promoting the reabsorption of uric acid and reducing uric acid levels [11, 12]. Current therapeutic drugs and natural alternatives for hyperuricemia have a SUA-lowering effect, but their long-term use has adverse effects on the cardiovascular system and other diseases [13]. Thus, it is necessary to develop drugs that have fewer side effects during the stabilization and effective reduction of uric acid. A number of natural alternatives help treat hyperuricemia (Table 1) [14, 15, 16, 17]. Honey mushroom, Armillaria mellea (Vahl.) P. Kummer (Physalacriaceae, Agaricomycetes) is widely distributed in tropical and temperate forests and is a representative edible and medicinal fungi in China [18, 19]. Modern pharmacological studies have demonstrated that Armillaria mellea ameliorates insulin resistance and diabetes-induced kidney damage [20, 21], reduces Lipopolysaccharides (LPS) stimulated glial cell inflammation in BV-2 mice [22], is an antioxidant [23, 24], anticancer [25, 26], relieves depression [27, 28], improves sleep [29], and other effects, but a high consumption leads to gastric upset [30]. Evidence suggests that aqueous extracts of Armillaria mellea fruiting body (AFB) have a degree of uric acid-lowering properties [31].
| Natural alternatives | Therapeutic method |
| Inonotus obliquus (Chaga) | Triterpenoid acid from Inonotus obliquus reduces SUA level by inhibiting XOD activity |
| G. applanatum | Ganoderma applanatum reduces SUA by up-regulating OAT1 protein and down-regulating GLUT9 protein expression |
| Agrocybe aegerita | Agrocybe aegerita inhibits XOD activity, increases OAT1 protein expression and reduces uric acid content |
| Caffeic acid phenethyl ester | Caffeic acid phenethyl ester in dietary plants reduces uric acid by inhibiting XOD and up-regulating OAT3 protein expression |
| Anthocyanins | Anthocyanins reduces uric acid by regulating the expression of renal urate transport proteins OAT1 and OAT3 |
SUA, serum uric acid; XOD, xanthine oxidase; OAT1, organic anion transporter 1; OAT3, organic anion transporter 3; G. applanatum, Ganoderma applanatum; GLUT9, glucose transporter 9.
Therefore, this study targets the OAT1/OAT3 membrane channel protein to explore the protective effects of Armillaria mellea and its fermentation products on mice with hyperuricemia induced by oral potassium oxonate (PO) and hypoxanthine (HX). HPLC was simultaneously employed to determine the purine content of AFB, Armillaria mellea rhizomorph (AR), and Armillaria mellea fermentation product (AFP).
Potassium oxonate (PO, 98%) was obtained from Macleans Biotechnology Corporation (Shanghai, China), HX (98.5%) was provided by Yuanye Biotechnology Co., Ltd. (Shanghai, China), and allopurinol (99.5%) supplied by Hefei Jiulian Pharmaceutical Co., Ltd (Anhui, China). SUA, serum creatinine (SCr), blood urea nitrogen (BUN) kits were provided by Nanjing Jiancheng Bioengineering Institute (Nanjing, China). Mouse anti-OAT1 and OAT3 were purchased from Affinity (San Francisco, CA, USA) and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) from Cell Signaling Technology Inc. (Beverly, MA, USA). All other reagents used in the experiment were analytically pure. Phenylmethanesulfonyl fluoride (PMSF) and radio immunoprecipitation assay were obtained from Beyotime Biotechnology (Shanghai, China). Armillaria mellea strains were high-quality Armillaria mellea (Vahl) P. Kumm. provided by Xingtianjian Pharmaceutical Group Company (Benxi, China).
Preparation of Armillaria mellea strains. The liquid shaker flask
culture medium was transferred from the Armillaria mellea slant strain,
placed in a constant temperature shaking incubator, 28 °C, 150 r/min
culture for 15 days and subcultured 1–2 times for seven days each time to obtain
stable liquid culture strains. The above liquid culture medium (Glucose 2%,
Yeast powder 1%, KH
Considering the convenience of oral administration and avoiding secondary injury to mice caused by intraperitoneal injection, oral administration was chosen. Refer to the usage and dosage of Armillaria mellea in the Encyclopedia of Chinese Medicinal Materials, take 30–60 g of decoction or grind it orally, such that 60 g is the high dose and 30 g is the low dose. The dose for mice was converted according to a body weight of 70 kg and its equivalent dose ratio was 0.0026 (converted according to the body surface area). AFB: soaked 78.0 g fruiting body in water for 30 min and decocted twice for 60 min each time, combine the two filtrates and concentrate to 100 mL. AR: Filter the rhizomorphs, dry at 65 °C at a constant temperature, pulverized, soaked 78.0 g rhizomorphs in water for 30 min and decocted twice for 60 min each time, combine the two filtrates and concentrate to 100 mL. AFP: the fermentation product (after rhizomorphs removal) was dried at a constant temperature of 105 °C, pulverized, and 39.0 g of the fermentation product powder was taken and dissolved in 100 mL of normal saline.
Preparation of purine reference substance: Precisely weigh an amount of HX,
xanthine and adenine reference substance, add water, and dissolve in a 10 mL
volumetric flask to prepare HX (0.526 mg
Waters XSelect HSS T3 column (4.6 mm
90 healthy male C57 mice (SPF, weighing 18
The 90 mice were divided into nine each groups containing 10 mice. After one
week of adaptation an animal model of hyperuricemia were established in the mice
by intragastric administration of PO (0.9 g
The serum obtained from mouse whole blood was centrifuged at 3000 rpm for 10 min at 4 °C. SUA, SCr, and BUN in mice serum were determined by commercially available kits.
Stomach tissue was fixed with 4% paraformaldehyde solution and embedded in paraffin, cut at a thickness of 5 µm, and stained with hematoxylin and eosin (H&E). Histopathological changes of the stomach were then observed by light microscope.
Following addition of 500 µL precooled protein lysate (containing PMSF) to 100 mg of kidney tissue, samples were homogenized and incubated on ice for 30 min. The protein concentration of the tissue supernatant was determined by Bicinchoninic Acid Assay (BCA) after centrifugation at 3500 rpm for 10 minutes at 4 °C. Electrophoresis was performed with equal amounts of total protein for sodium dodecyl sulfate – polyacrylamide gel electrophoresis (SDS-PAGE), then transferred to nitrocellulose membranes. Non-specific binding was prevented by incubating with 5% skimmed milk powder at room temperature for two hours. The blot was incubated with primary antibody (anti-OAT1, anti-OAT3, and anti-GAPDH) overnight at 4 °C, then incubated with the secondary antibody plus fluorescent markers for two hours at room temperature. A chemiluminescent detection system was used to detect bands and optical density analysis with ImageJ software (ImageJ 1.48v Jave1.6.0-20(64-bit), National Institutes of Health, USA) was used to measure band intensity. The ratio of OAT1 and OAT3 to the corresponding control GAPDH was calculated for each sample.
Part of the gastric tissue was fixed with 4% paraformaldehyde solution, embedded in paraffin, and sectioned into 5 µm slices. Phosphate buffered saline with Tween (PBST) washed 3 times, 3% hydrogen peroxide room temperature was closed for 20 min, PBST washed 3 times, 5% bovine serum albumin was closed at room temperature for 1 hour, primary antibody incubation overnight at 4 °C. Samples were incubated with PBST and horse radish peroxidase-labeled secondary antibody at room temperature for 45 min, PBS washed three times, the diaminobenzidine was color developed, stained under the microscope, and washed with deionized water. Hematoxyfolk was counterstained, dehydrated after transparency, and sealed with neutral gum.
Data were processed with the aid of Graph Pad Prism 8.3 (Graph Pad Software
Inc., San Diego, CA, USA), the difference between groups was analyzed by one-way
ANOVA, and purine content was analyzed by paired t-test. Experimental
data were expressed as mean
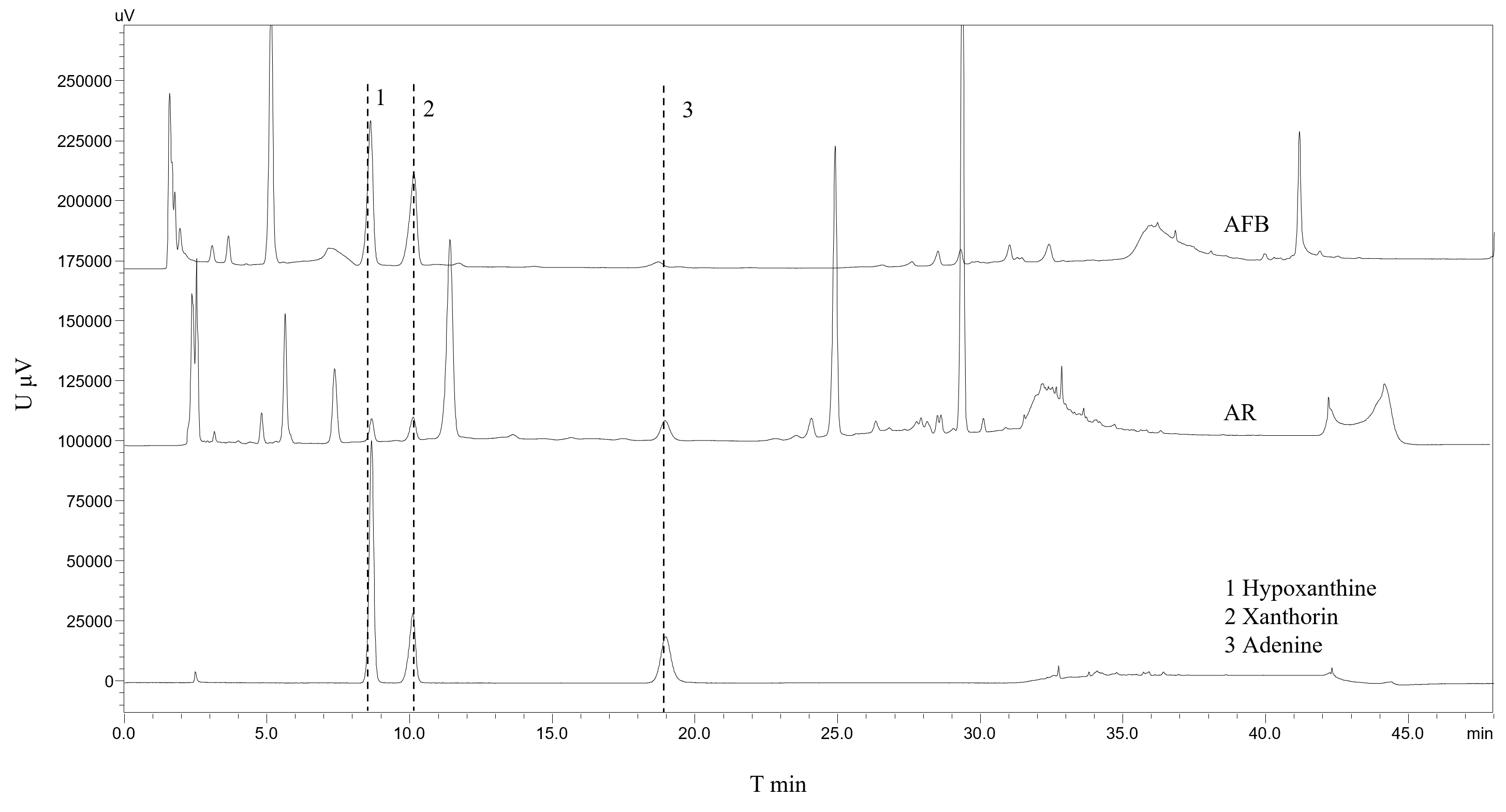
The chromatogram of sample and control products are given (Fig. 1) and show that
HX, xanthine, and adenine in both AFB and AR exhibit a good chromatographic peak
shape and that AFB contains more purine components. The content of three purines
was too low and did not reach the detection limit in AFP. Analytic results show
that the density of HX, xanthine, and adenine in AFB were 1001.87, 807.87, and
113.12 µg
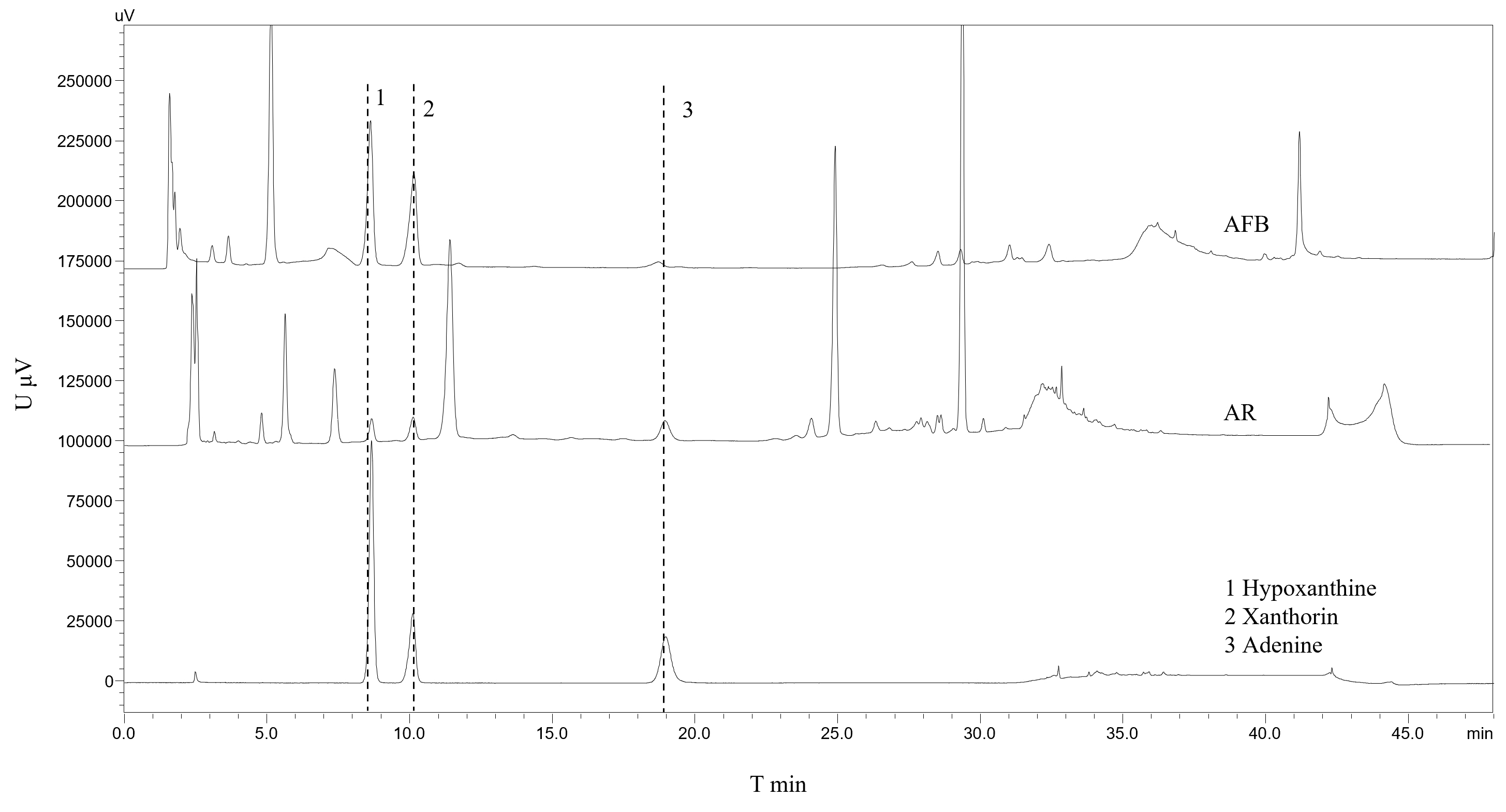 Fig. 1.
Fig. 1.Liquid Chromatogram of Samples and Standards. 1: hypoxanthine; 2: xanthine; 3: adenine.
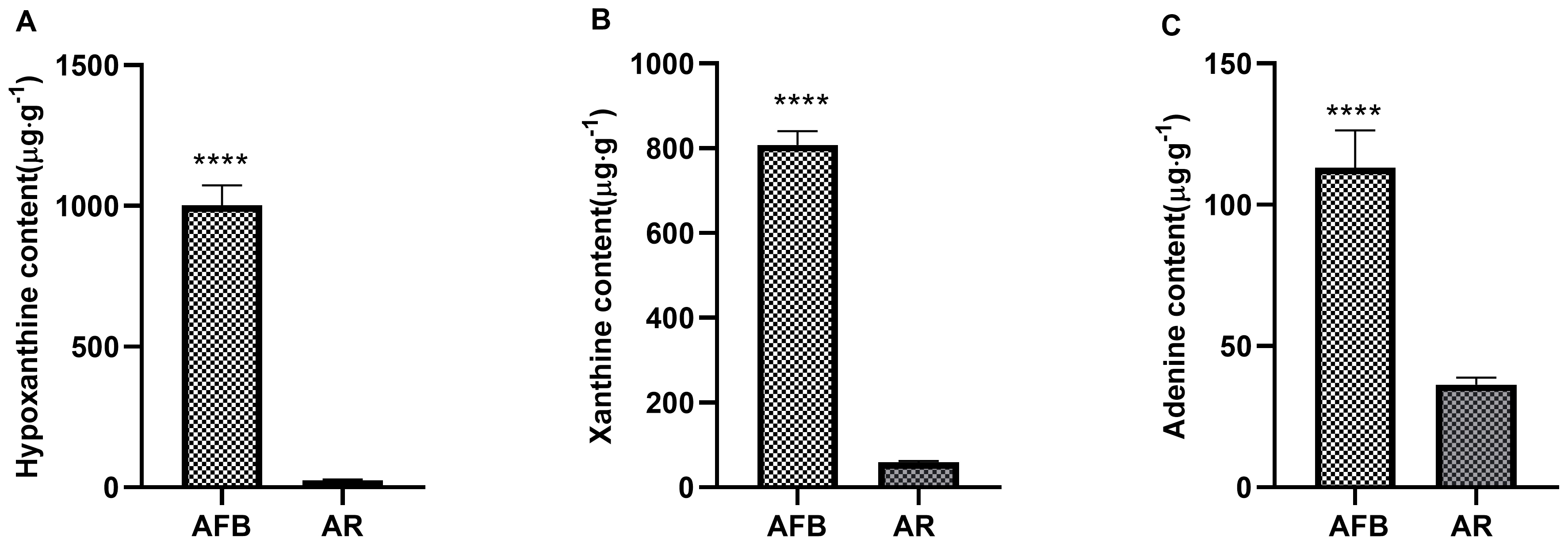
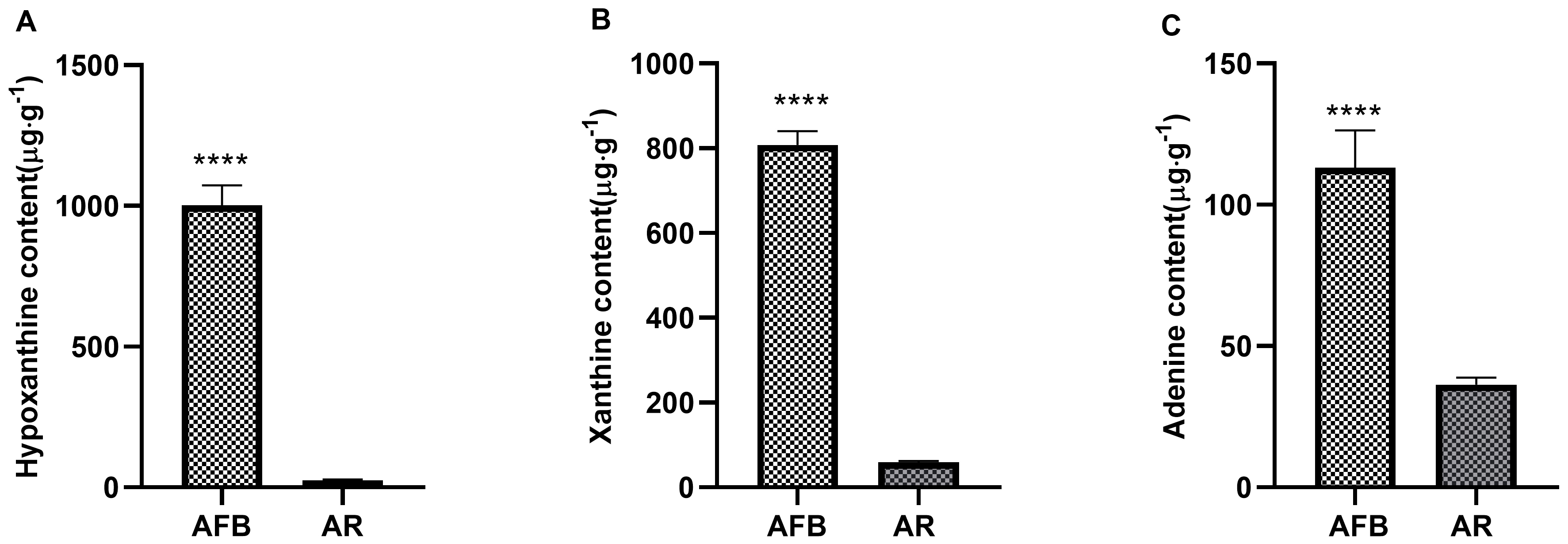 Fig. 2.
Fig. 2.Comparative data of AFB and AR in
hypoxanthine, xanthine, and adenine content. A:
hypoxanthine content; B: xanthine content; C: adenine content. Data expressed as mean
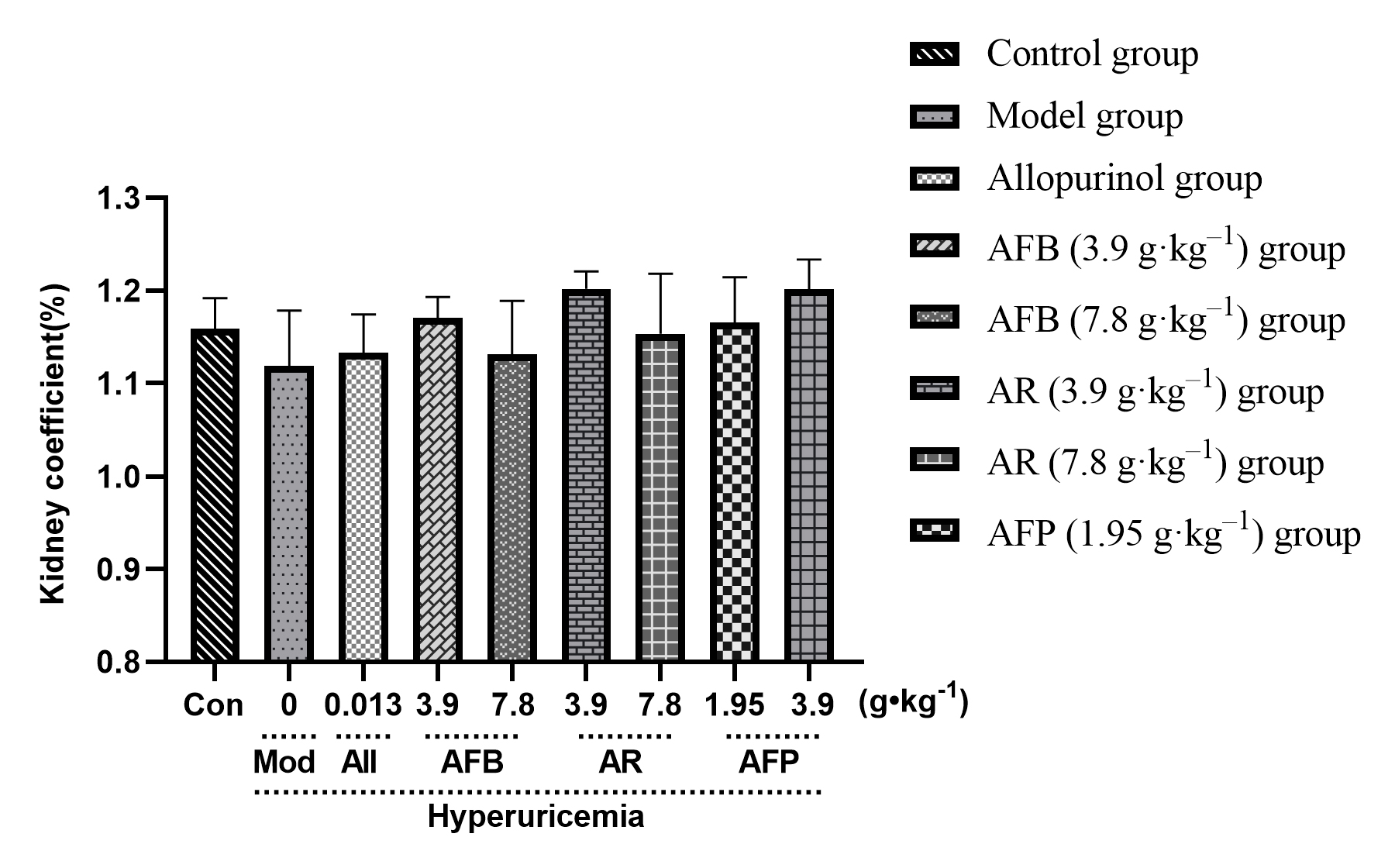
There was no significant difference in the kidney coefficient (bilateral kidney weight (mg)/body weight (g), Fig. 3) indicating that Armillaria mellea and their fermentation products had no effect on the kidney.
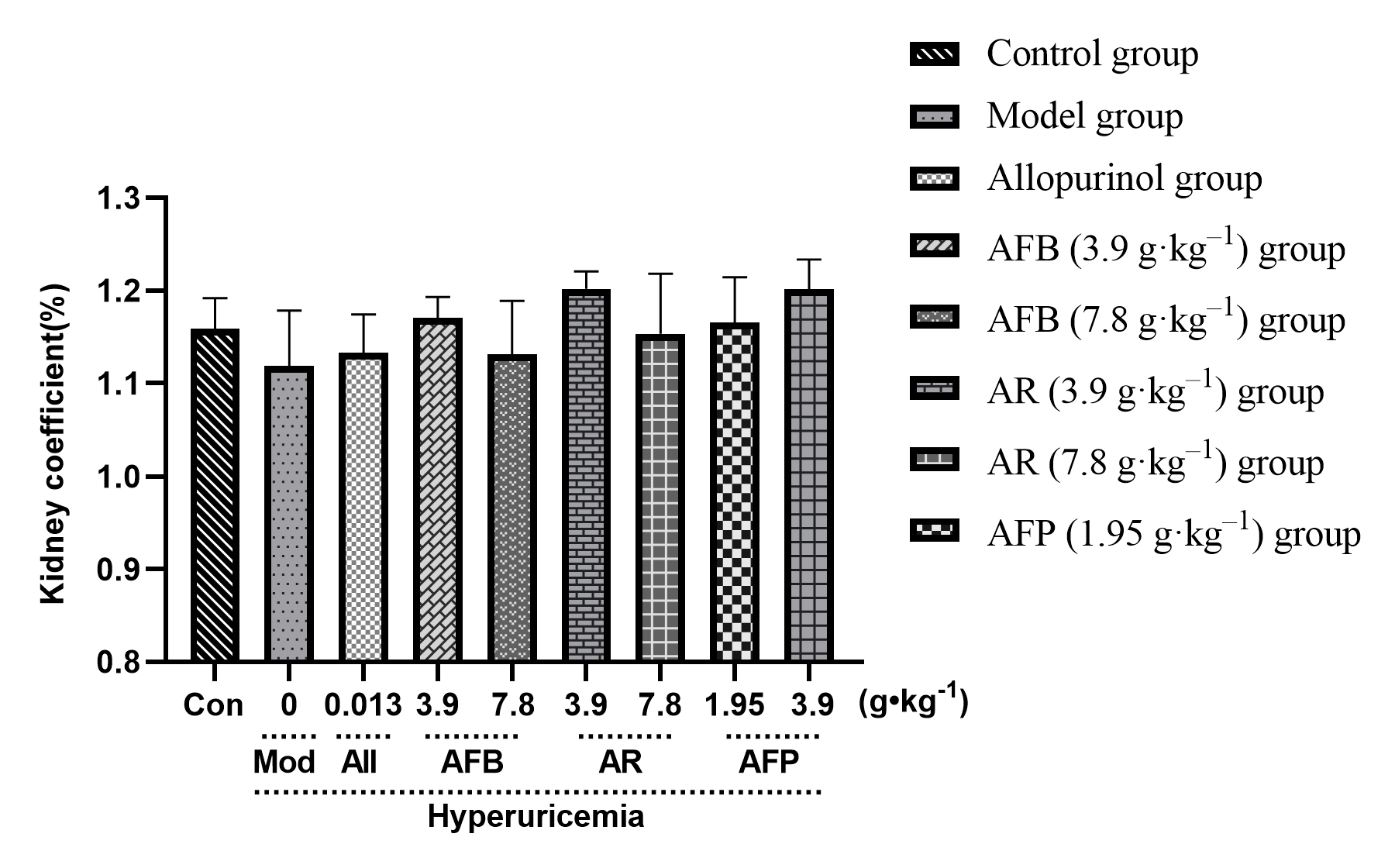 Fig. 3.
Fig. 3.Effect of Armillaria mellea
and its fermentation products on the kidney organ coefficient of mice.
Data expressed as mean
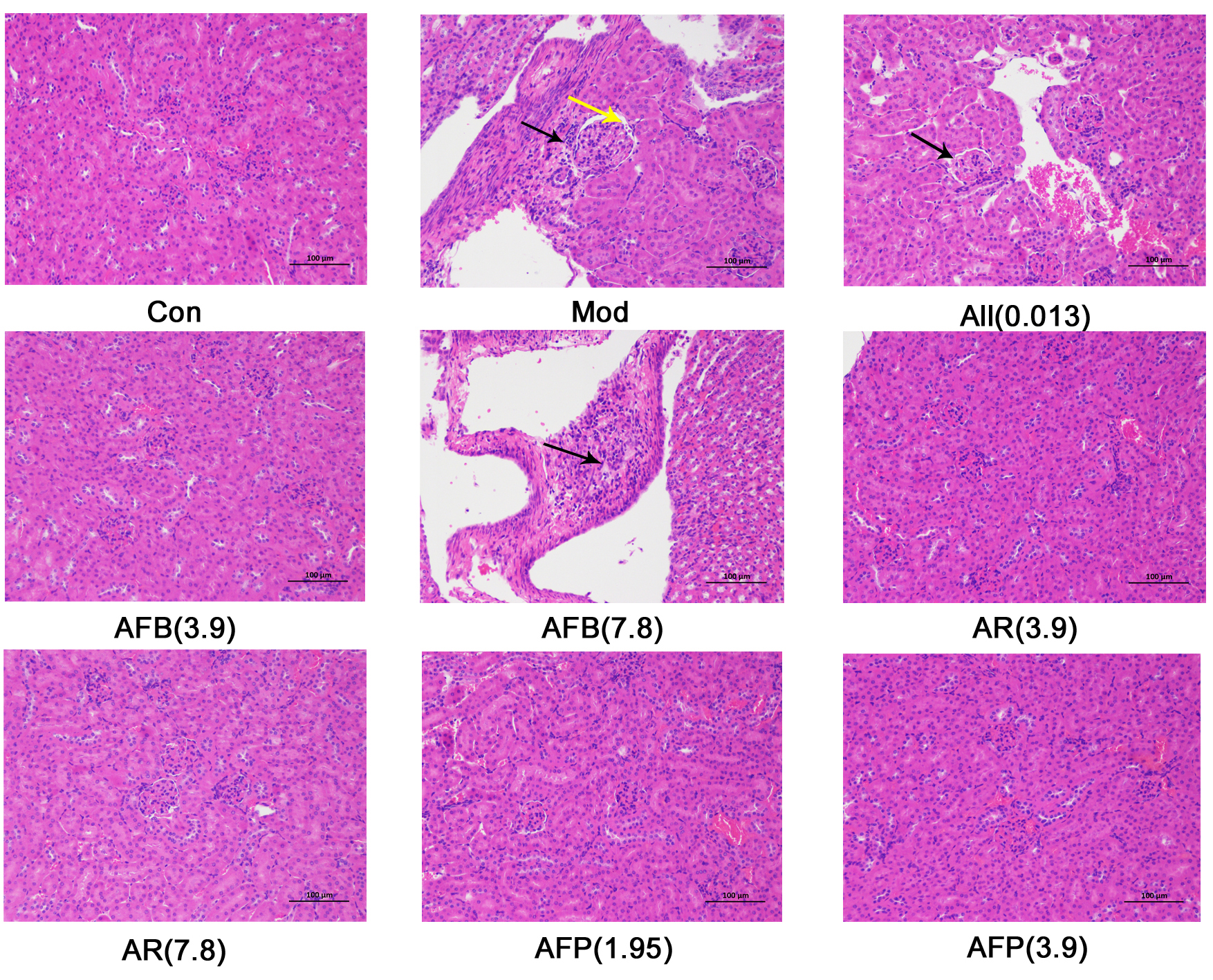
Histopathological results showed (Fig. 4) that the surface envelope of kidney
tissue in the control group was composed of dense connective tissue with uniform
thickness. No obvious abnormalities were found in the kidney parenchyma, neither
was there significant hyperplasia of the kidney interstitium, nor obvious
inflammatory changes. Compared with the control group, punctate infiltration of
lymphocytes (black arrow) and eosinophilic substances (yellow arrow) were seen in
the kidney capsule in the model group. After administration, exception
for a small amount of eosinophils in the AFB (7.8 g
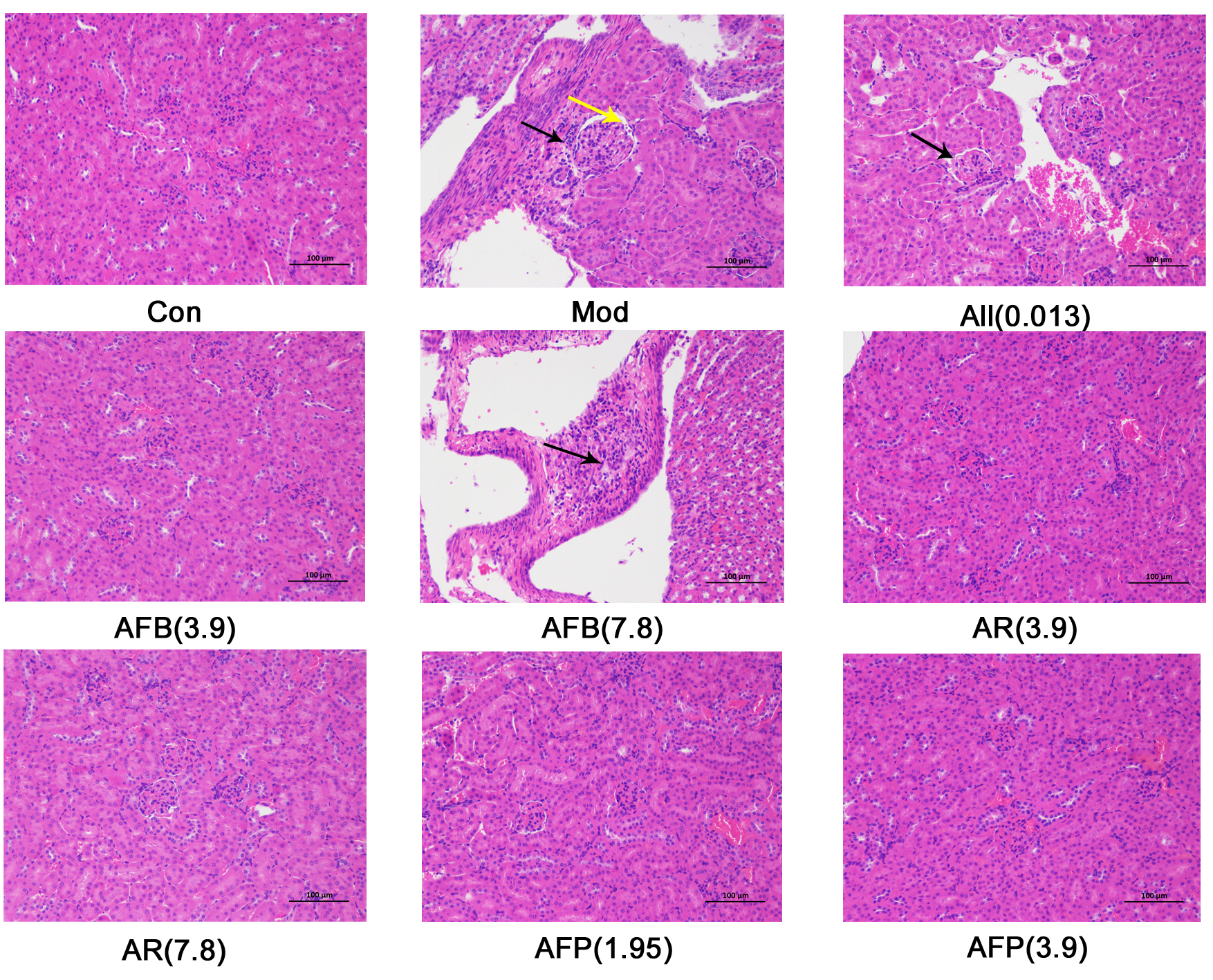 Fig. 4.
Fig. 4.Effect of Armillaria mellea
and its fermentation products on kidney tissue sections in mice kidney.
Histopathological changes are shown following H&E staining
(magnification,
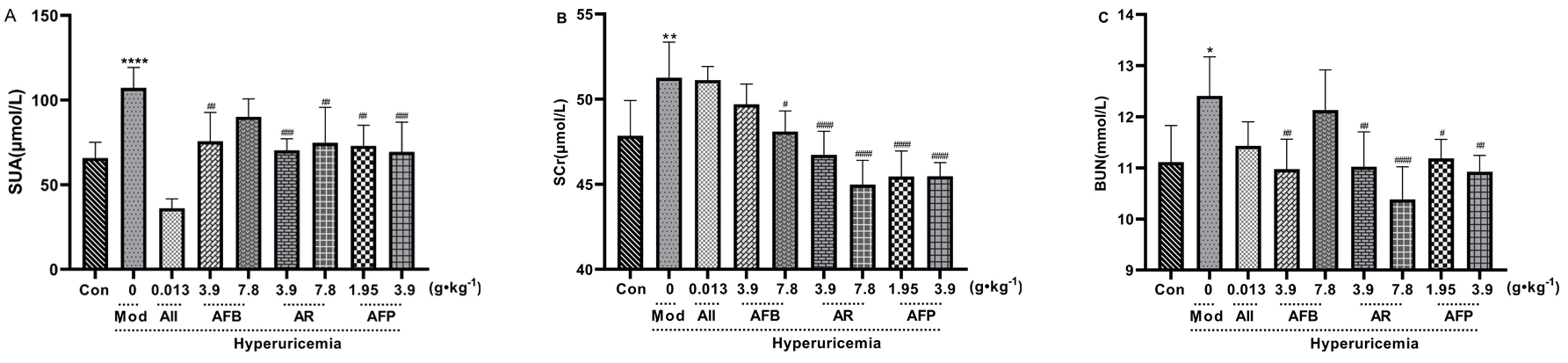
Compared with the control group, the serum levels of SUA, SCr, and BUN in the
model group were significantly improved (p
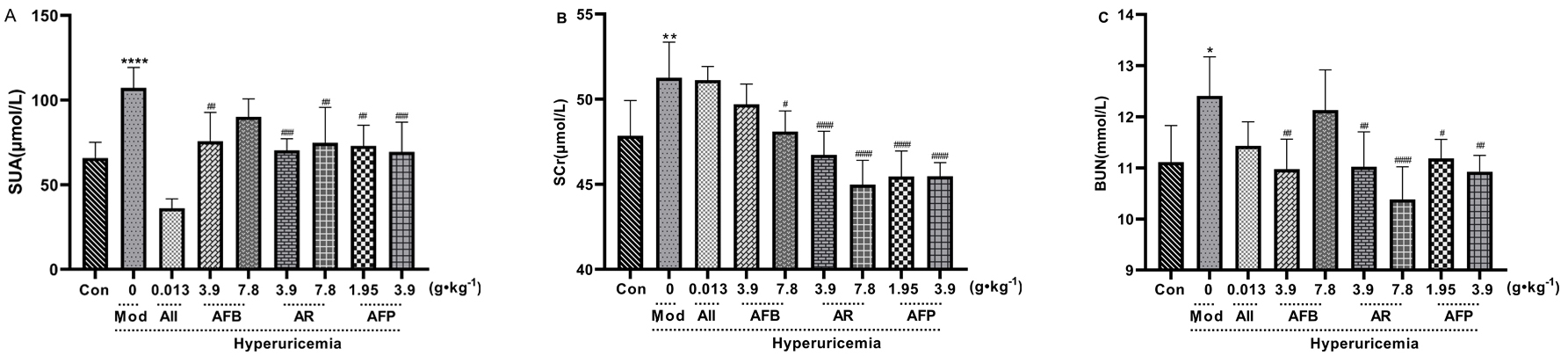 Fig. 5.
Fig. 5.Effect of Armillaria mellea
and its fermentation products on SUA, BUN, and SCr. A: SUN content; B: SCr content; C: BUN content. Data are expressed as mean
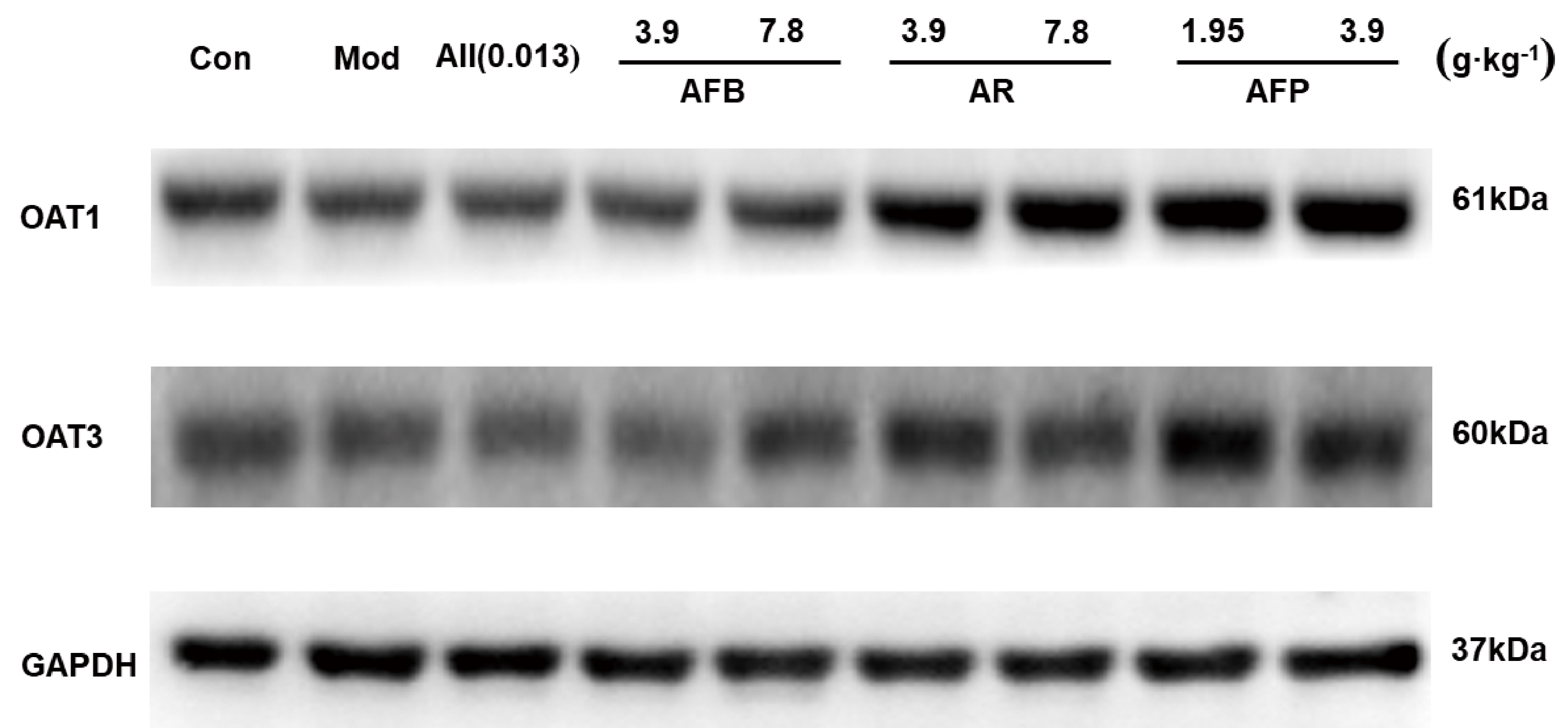
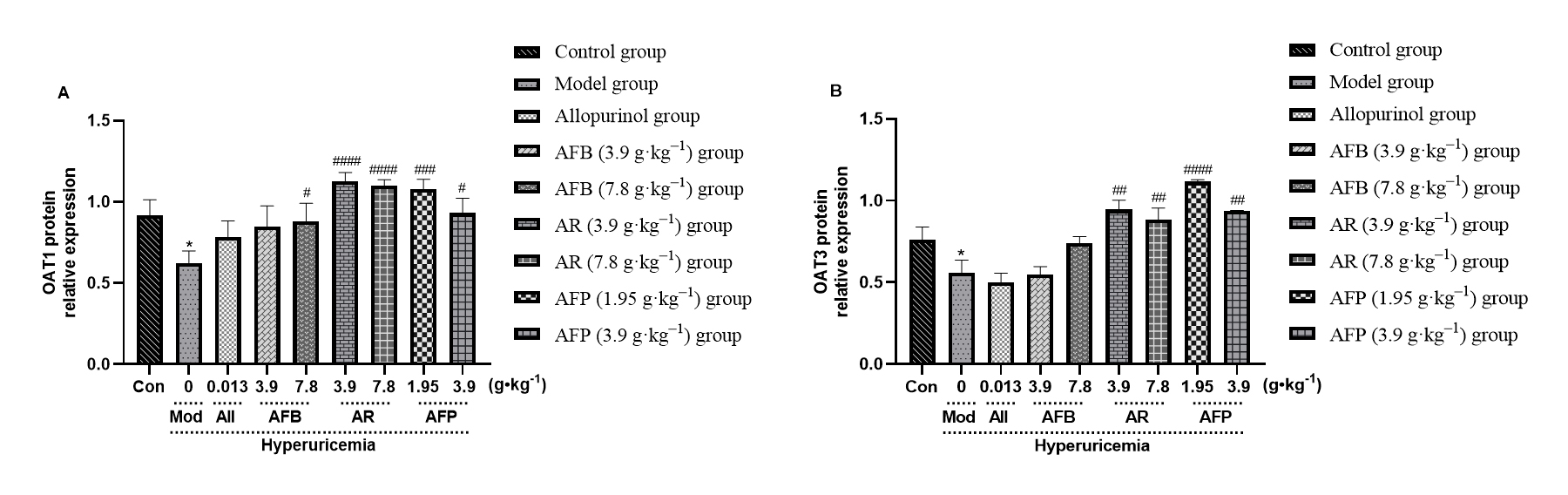
The OAT1/PAT3 protein results are shown in Fig. 6. The expression level of OAT1
and OAT3 proteins in the kidney tissue of the model group decreased significantly
compared with the control group (p
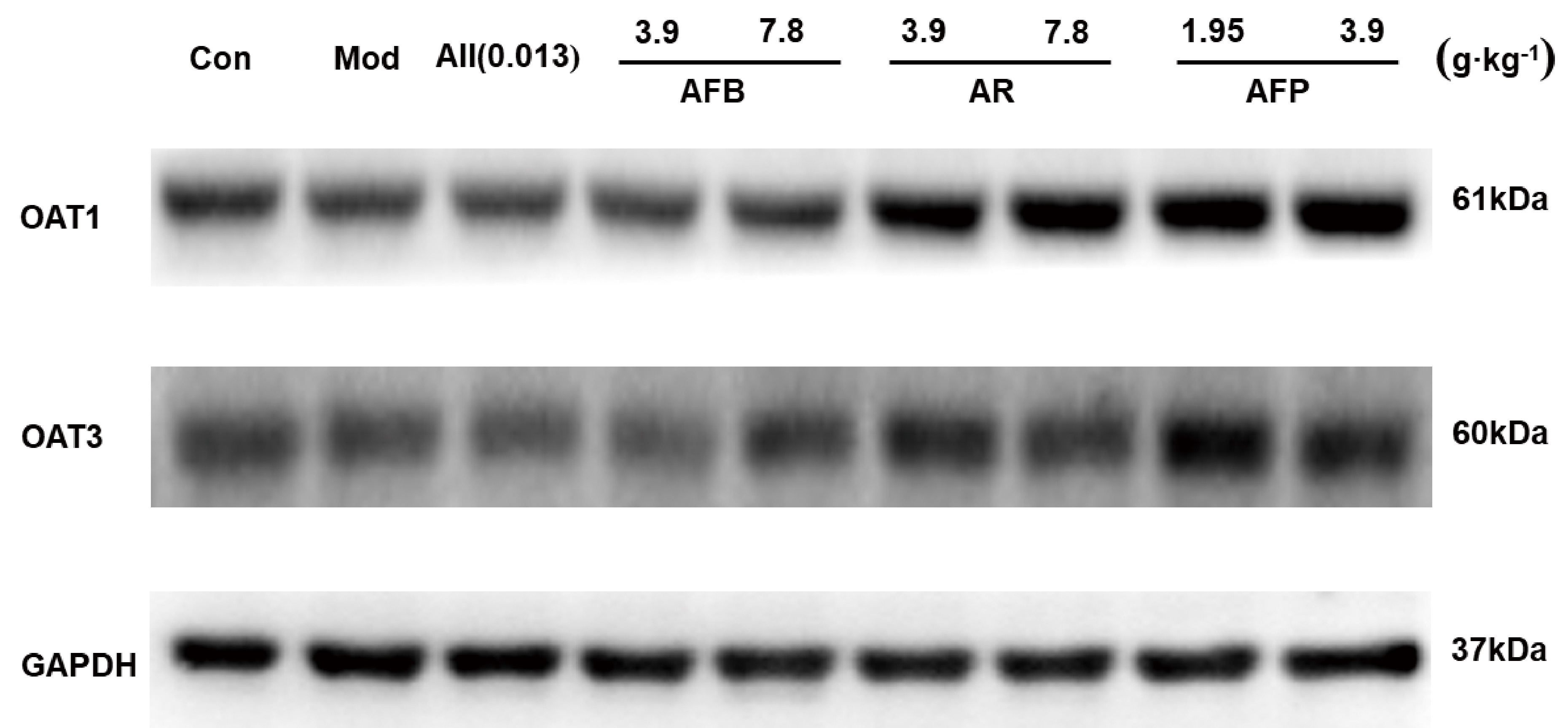 Fig. 6.
Fig. 6.OAT1/OAT3 protein expression results.
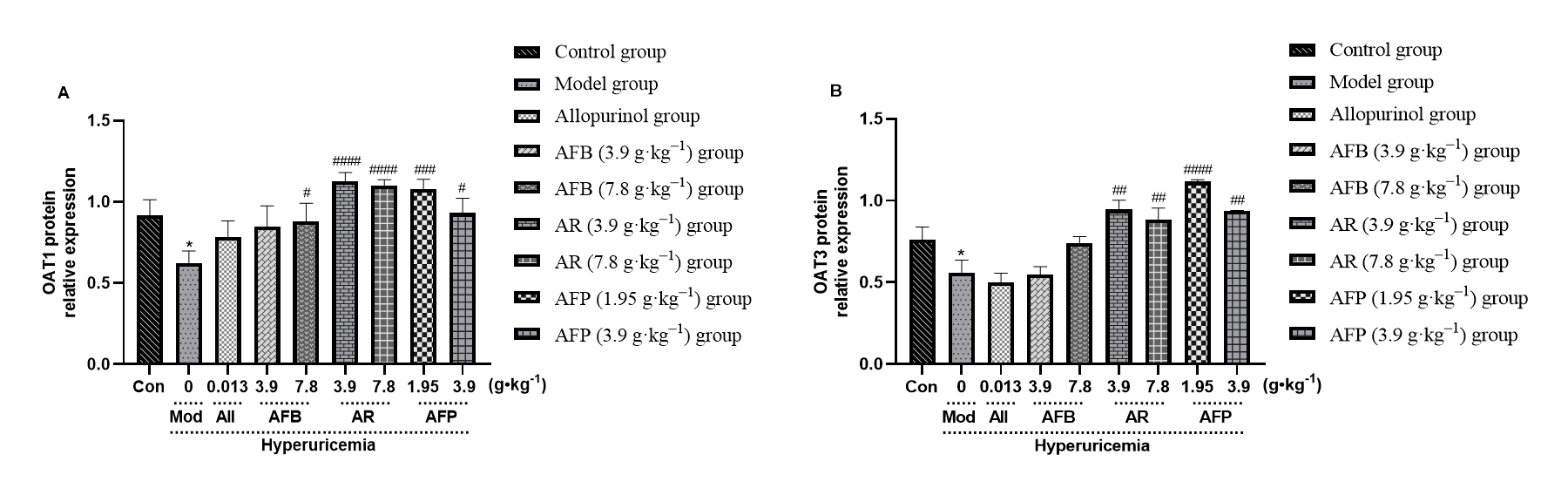 Fig. 7.
Fig. 7.Armillaria mellea and its
fermentation products increase the activation of OAT1 and OAT3 in kidney tissue.
A: OAT1 protein content; B: OAT3 protein content. Data expressed as mean
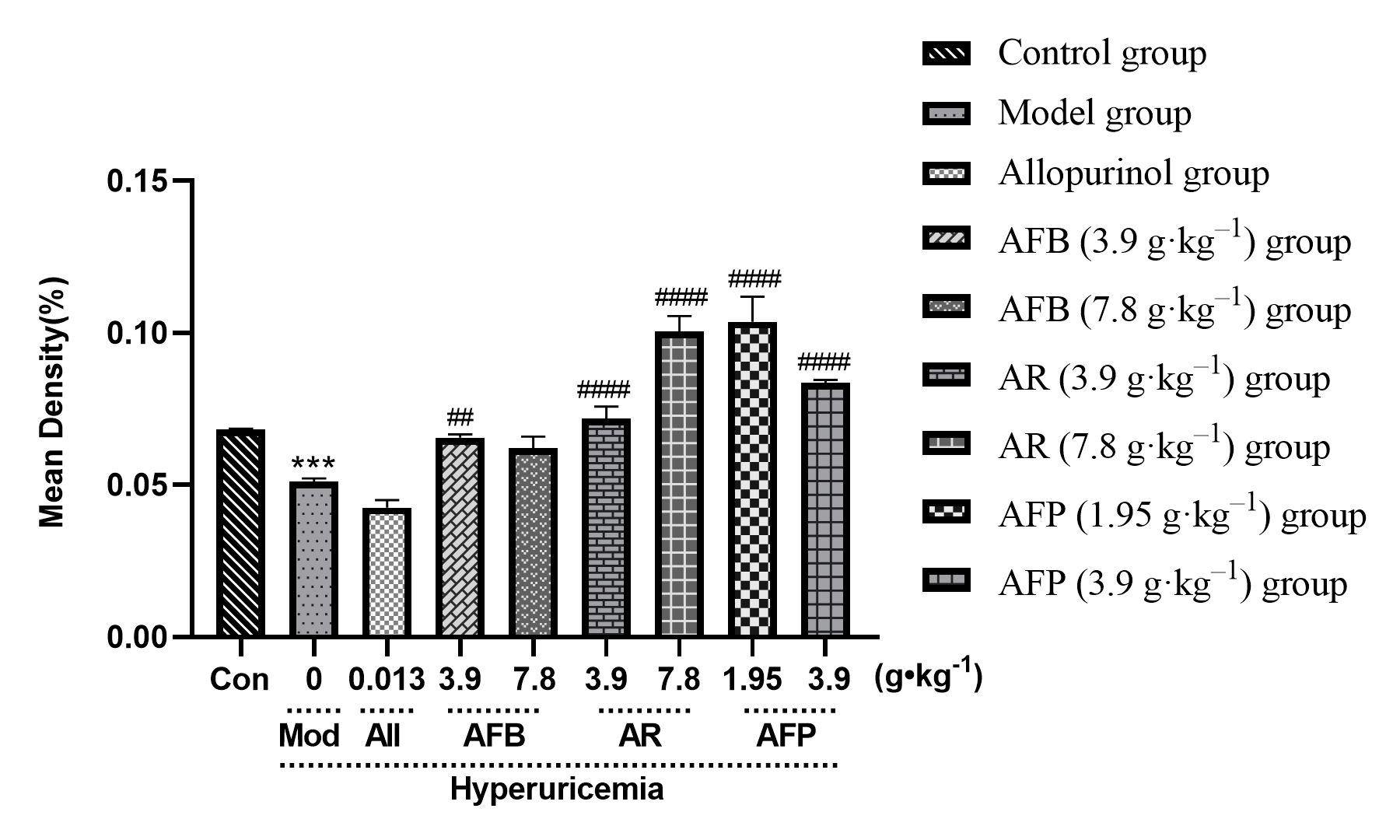
OAT1 protein levels were significantly reduced in the hyperuricemia model group
(p
 Fig. 8.
Fig. 8.OAT1 Immunohistochemical results.
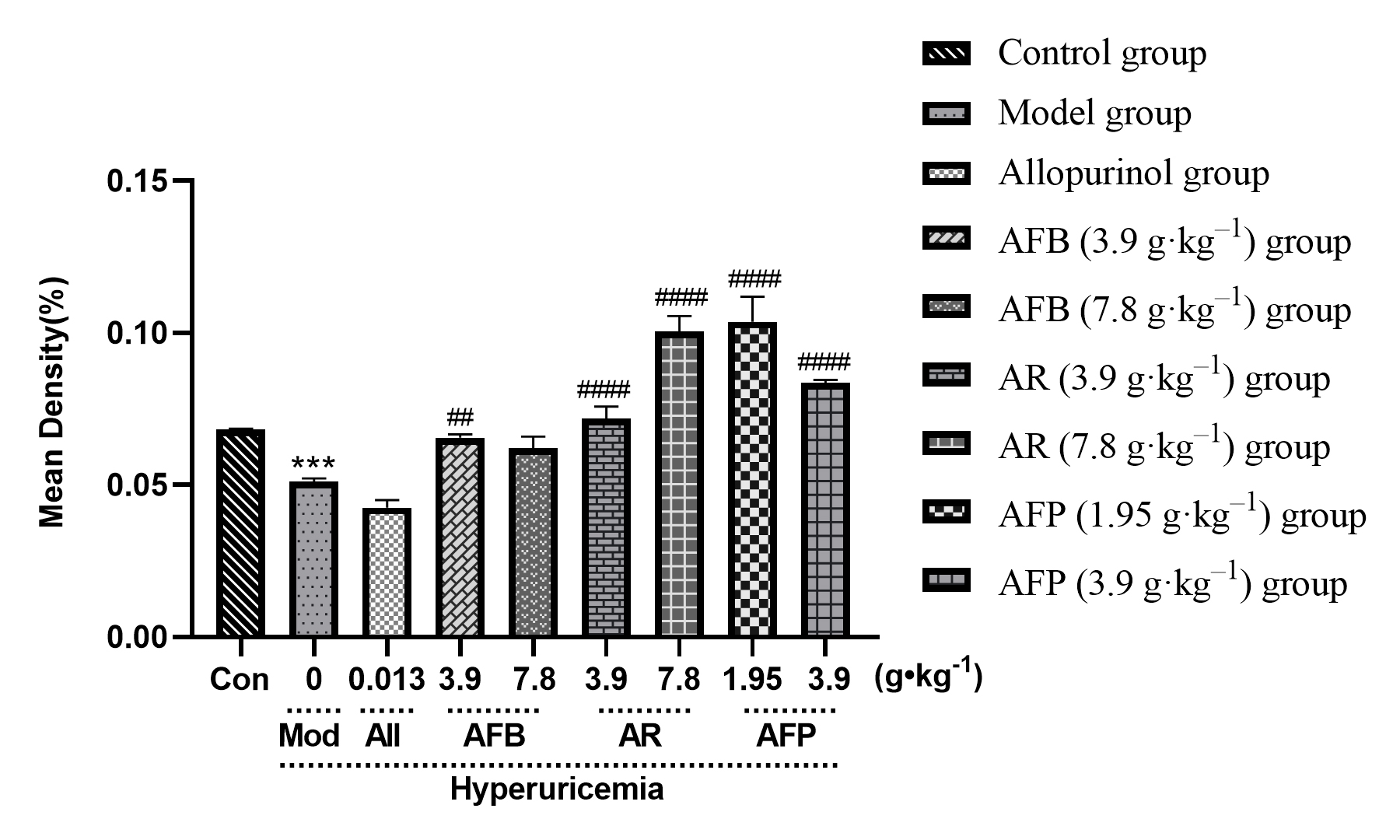 Fig. 9.
Fig. 9.Immunohistochemical analysis of
Armillaria mellea and its fermentation products on mice OAT1 protein.
Data expressed as mean
There are two major types of hyperuricemia mouse model: genetically induced and
environmentally induced. PO and HX are commonly used to establish hyperuricemia
models to study hyperuricemia-related diseases. The definition of uric acid
content for hyperuricemia is not the same in humans and mice, and urate
concentrations in mice have not been specified. Following the experiment, the
serum content of mice in the model group could reach the urate concentration
standard of some mouse models [32], even in the case of reversible hyperuricemia
[33]. This showed that the serum urate content of mice after modeling is greater
than the serum urate content of mice after the experiment. In this experiment the
model was used to study the uric acid-lowering effect of Armillaria
mellea and its fermentation products. Results showed that AFB (3.9
g
PO has traditionally been thought to seriously impair kidney function. SCr and
BUN levels are commonly used indicators to assess kidney health. In this
experiment, the model group increased the concentration of Scr, BUN, and damaged
kidney tissue. The AR (3.9 and 7.8 g
Uric acid homeostasis primarily depends on an equilibrium between liver
production, secretion, renal reabsorption, and intestinal excretion, among them,
the kidney reabsorbs about 90% of the filtered urate, so increased reabsorption
is extremely important for urination drugs [34, 35]. OAT1 was identified as a
molecular transporter regulating the secretion of uric acid in the first step,
which drives other OATs (OAT2, OAT3, OAT4) that together constitute an
amphiphilic solute transporter family (Slc22a) within the major facilitator
superfamily [36]. OAT3 is not only the most expressed protein in the kidney, but
also has the same function of exchanging substrates as OAT1, which has great
research significance. In terms of uric acid transport, the kidneys of humans are
similar to those of mice. OAT1 and OAT3 are basolateral urate transporters
encoded by SLC22A6 and SLC22A8, respectively, that are involved in urate
secretion and urate conversion in the blood, which is transported to proximal
tubule cells. This study showed that AR (3.9 and 7.8 g
Yong’s [31] experimental results showed that both alcoholic and aqueous extracts of AFB reduced the uric acid levels of hyperuricemic mice by reducing xanthine oxidase activity, while both up-regulating the expression level of OAT1 and down-regulating the expression of GLUT9 in kidney tissue and CNT2 in the gastrointestinal tract. The current study confirms that AMW reduces uric acid levels and shows that AFB also reduces uric acid levels in hyperuricemic mice by increasing the expression of OAT1 protein in kidney tissue, but that the activity of AFB is far less than that of AR and AFP. Increased HX content leads to increased SUA. When combined with the purine content experiment results, it was revealed that AFB contained much more purine than AR and AFP. Therefore, the down-regulation effect of uric acid on hyperuricemia mice is weaker than AR and AFP. Additionally, it shows that the fermentation products AR and AFP of Armillaria mellea are more effective and more meaningful in treating hyperuricemia. Armillaria mellea and is often used for research into the state of fruiting bodies [37]. Nevertheless, in this study, it was found that the fermentation products of Armillaria mellea have better effect in treating hyperuricemia, which suggests that the fermentation products of Armillaria mellea also have research significance.
Armillaria mellea and its fermentation products can treat hyperuricemia by up-regulating OAT1 protein and OAT3 protein, reducing the content of uric acid, creatinine and urea nitrogen in mice, and the fermentation products have better therapeutic effect.
AFB, Armillaria mellea fruiting body; AFP, Armillaria mellea fermentation product (after rhizomorphs removal); AR, Armillaria mellea rhizomorphs; BUN, blood urea nitrogen; GLUT9, glucose transporter 9; H&E, hematoxylin-eosin staining; HPLC, High Performance Liquid Chromatography; HX, hypoxanthine; IHC, Immunohistochemistry; OAT1, organic anion transporter 1; OAT3, organic anion transporter 3; PO, potassium oxonate; SCr, serum creatinine; SUA, serum uric acid; WB, Western blot.
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
ZL and SW conceived the study, designed the experiments, supervised all the research, analyzed the data, and revised the manuscript. HW and ZL completed the experiments and analyzed the data. ZL wrote the manuscript. ZL carried out the experiments. All authors have read and agreed to the published version of the manuscript.
All procedures and experimental methods were approved by the Animal Ethics Committee of Changchun University of Traditional Chinese Medicine (Certificate No.: 2021196).
De Hu helped the writing of the manuscript.
This research was partially supported by the Traditional Chinese Medicine Science and Technology Project of Jilin Province (2021006), the Jilin Province Science and Technology Development Plan Project Technology Innovation Guidance-Medical and Health Industry Development Special (YDZJ202301ZYTS509), and the “Xinglin Scholars Project” Young Scientist Program of Changchun University of Traditional Chinese Medicine (QNKXJ2-2021ZR03).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.