Background: Interspecies variations in mammalian red blood cells (RBCs)
are observed in circulating RBC lifespan, cell size, fluidity, aggregation, water
permeability, metabolism, lipid composition, and the overall proteome. Bovine RBC
cell membrane is deficient in phosphatidylcholine and exhibits anomalies in the
arrangement of phosphatidylethanolamine within the lipid bilayer. However, like
human RBCs, virtually all the aminophospholipid phosphatidylserine (PS) is found
within the cytoplasmic side of the cell membrane of intact circulating bovine
RBCs. During apoptotic cell death of human and murine RBCs, PS translocates to
the outer leaflet of the cell membrane via Ca-dependent and -independent
signaling mechanisms. However, little is known about this process in bovine RBCs.
Methods: Using cytofluorometry analyses, we characterized and compared
the cell death responses in bovine and human RBCs in vitro exposed to
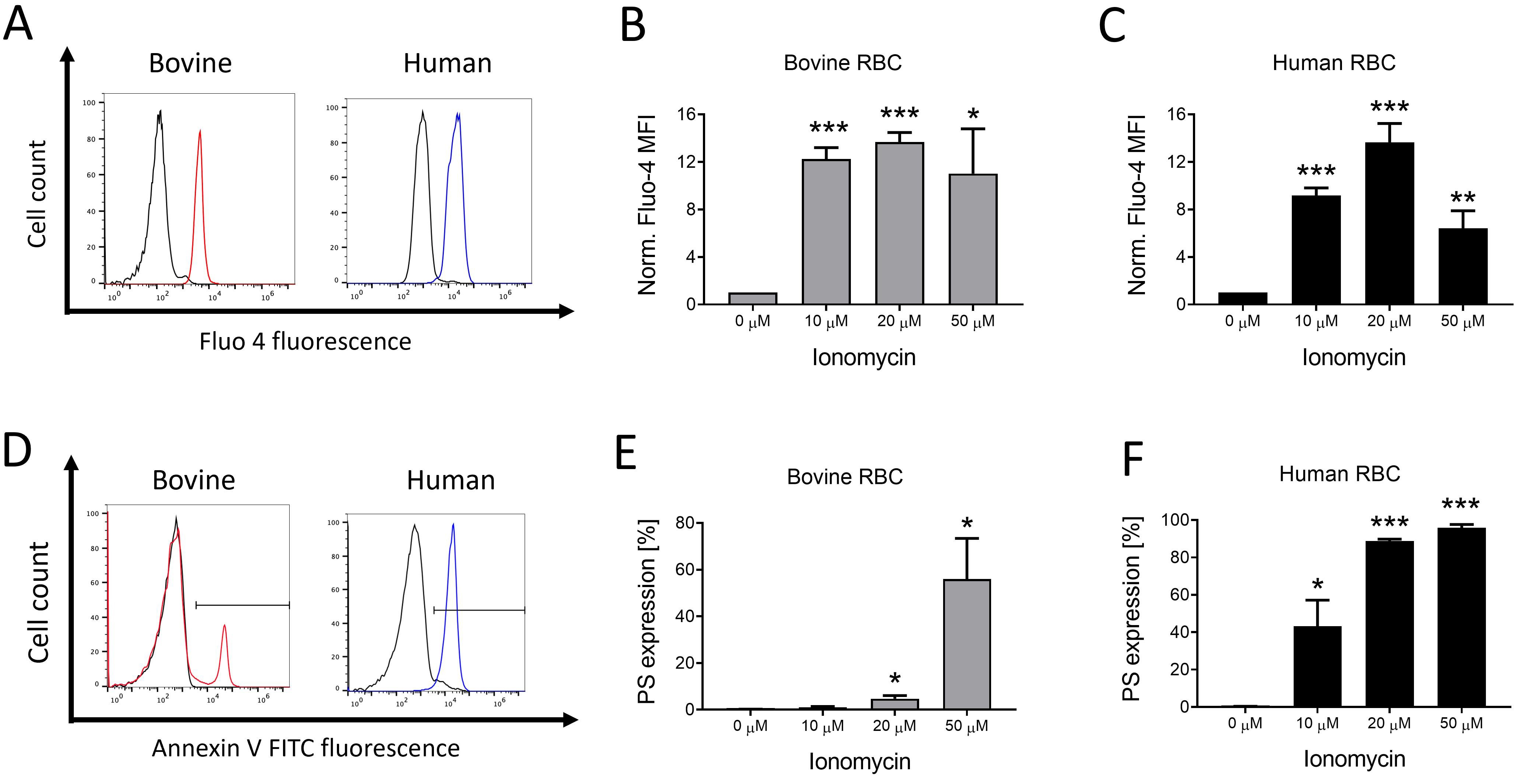
various pathophysiologic cell stressors. Results: Ionic stress, by
ionophore treatment, and oxidative stress enhanced cytoplasmic Ca levels
and cell membrane PS expression in both bovine and human RBCs. Fever-grade
hyperthermia and energy starvation promoted Ca influx and elevated
reactive oxygen species levels in both human and bovine RBCs. However, bovine
RBCs displayed minimal increases in PS expression elicited by hyperthermia,
energy starvation, and extracellular hypertonicity as compared to human RBCs. In
response to decreased extracellular osmolality, bovine RBCs exhibited
significantly enhanced fragility as compared to human RBCs. Conclusions:
Bovine RBCs display differential cell death patterns as compared to human RBCs,
only partly explained by increased Ca influx and oxidative stress.
Premature removal of circulating RBCs could potentially contribute to the
pathogenesis of anemia in cattle caused by a wide range of factors such as
systemic diseases, parasitic infections, and nutritional deficiencies.