1 West China School of Stomatology, Sichuan University, 610041 Chengdu, Sichuan, China
2 State Key Laboratory of Oral Diseases, National Clinical Research Center for Oral Diseases, Department of Prosthodontics, West China Hospital of Stomatology, Sichuan University, 610041 Chengdu, Sichuan, China
3 State Key Laboratory of Oral Diseases, National Clinical Research Center for Oral Diseases, West China Hospital of Stomatology, Sichuan University, 610041 Chengdu, Sichuan, China
4 State Key Laboratory of Oral Diseases, National Clinical Research Center for Oral Diseases, Department of Cariology and Endodontics, West China Hospital of Stomatology, Sichuan University, 610041 Chengdu, Sichuan, China
†These authors contributed equally.
Abstract
Dental pulp stem cells (DPSCs) are a type of mesenchymal stem cells derived from dental pulp that serves as an important model for investigating biological regeneration. DPSCs have a multipotent differentiation capacity and can promote different biological processes, including osteogenesis, odontogenesis, chondrogenesis, and angiogenesis. These biological processes are regulated by an extensive range of intra- and extra-cellular factors. Further, biomechanical cues, such as substrate stiffness, physical stress, and cell spreading, have been highlighted as particularly important modulators of DPSC function. This review sought to discuss various related signaling components involved in biomechanical cues and their respective roles in cellular and tissue responses in DPSCs, summarize current findings, and provide an outlook on the potential applications of biomechanics in regenerative medicine and tissue engineering.
Keywords
- dental pulp stem cells
- mechanical signaling
- cell fate determination
Dental pulp stem cells (DPSCs), a type of mesenchymal stem cells derived from dental pulp [1], can differentiate into a variety of cells, such as dental pulp, dentin, and osteoblasts, and serve as important models for investigating biological regeneration [2, 3, 4]. Biomechanics, an interdisciplinary field that investigates the effects of forces and motion on biological systems, focuses on mechanical stimuli and their impact on cell behavior [5, 6, 7]. Over the past decades, mechanical stimuli, such as tissue stiffness, physical forces, and cell spreading, have led to significant advances in the fields of regenerative medicine and tissue engineering [8, 9, 10]. Understanding the underlying mechanisms of DPSC response to mechanical stimuli can provide multiple ideas for the regeneration and immune regulation of various cells or tissues, such as dentin, bone, and cartilage. Here, we summarize these findings and provide a perspective on the prospects and potential applications of DPSCs in bioengineering and regenerative medicine.
Mesenchymal stem cells (MSCs) are heterogeneous stem cells that exhibit self-renewal and multilineage differentiation ability [11, 12]. MSCs can be isolated from different tissues, such as bone marrow, adipose tissue, and umbilical cord tissue [13, 14, 15]. In the oral cavity, MSCs can be classified into several distinct subtypes, including DPSCs [1, 16], stem cells from human exfoliated deciduous teeth (SHEDs) [17], stem cells from apical papilla (SCAPs) [18, 19], periodontal ligament stem cells (PDLSCs) [20], dental follicle stem cells (DFSCs) [21], gingival-derived mesenchymal stem cells (GMSCs) [22], and buccal fat pad-derived stem cells (BFPSCs) [23]. DPSCs, which were first isolated from the dental pulp of third molars [1, 24, 25], are characterized by high self-renewal and differentiation capacities [1, 26, 27]. Similar to MSCs, DPSCs express stem cell markers, such as CD29, CD44, CD73, CD90, CD105, CD146, CD271, and STRO-1 [1, 28], whereas the hematopoietic markers including CD14, CD34, CD45, and CD117, are absent or expressed at very low levels [29, 30]. DPSCs also express the vascular endothelial growth factors, TLR4 and TLR5 [31, 32, 33]. Therefore, owing to their heterogeneity, DPSCs could be a great choice for regenerative medicine and clinical therapy.
Several studies have revealed that DPSCs can differentiate into odontoblasts,
osteoblasts, neural cells, chondrocytes, and adipocytes [3, 4]. Odontoblasts are
specialized cells that produce the dentin matrix and are responsible for mineral
deposition [34]. Similarly, osteoblasts are specialized cells that produce the
bone matrix and regulate bone metabolism [35]. Odonto/osteogenesis is the most
important differentiation potential of DPSCs. Many studies have explored the
effects of different culture conditions [1, 36, 37], signaling molecules [38, 39, 40, 41],
and chemicals [42, 43, 44, 45] on the odonto/osteogenic differentiation of DPSCs. For
instance, BMP and TGF-
Owing to the multilineage differentiation potential and availability of DPSCs, they are attractive candidates for tissue engineering and regenerative medicine. DPSCs have been demonstrated to regenerate the pulp-dentin complex [1], which provides an immense step toward endodontic treatment and enables dentin-pulp regeneration. DPSCs can also stimulate endothelial [60] and immune cells [61], indicating a potential role for DPSCs in angiogenesis, wound healing, and immune regulation. As DPSCs originate from the neural crest, they exhibit neuroregenerative properties. Although emerging evidence has revealed that DPSC-based therapy is promising, more studies on the biological attributes of DPSCs are warranted.
The complex process of mechanical signal perception in cells is primarily mediated by specialized mechanosensors that convert these signals into biochemical signals, triggering a cascade of biological effects. Mechanosensors consist of various molecules, including cadherin-catenin junctions and cell adhesions complexes, which mediate cell-cell adhesion and cell-extracellular matrix (ECM) adhesion, respectively, and then transduce mechanical forces into intracellular signaling (mechanotransduction) and mediate cellular processes, such as gene expression, cell proliferation, and differentiation [62, 63, 64, 65] (Fig. 1). Below, we opted to discuss various signaling components involved in biomechanical stress and their respective roles in cellular and tissue responses, with a particular focus on stem cells.
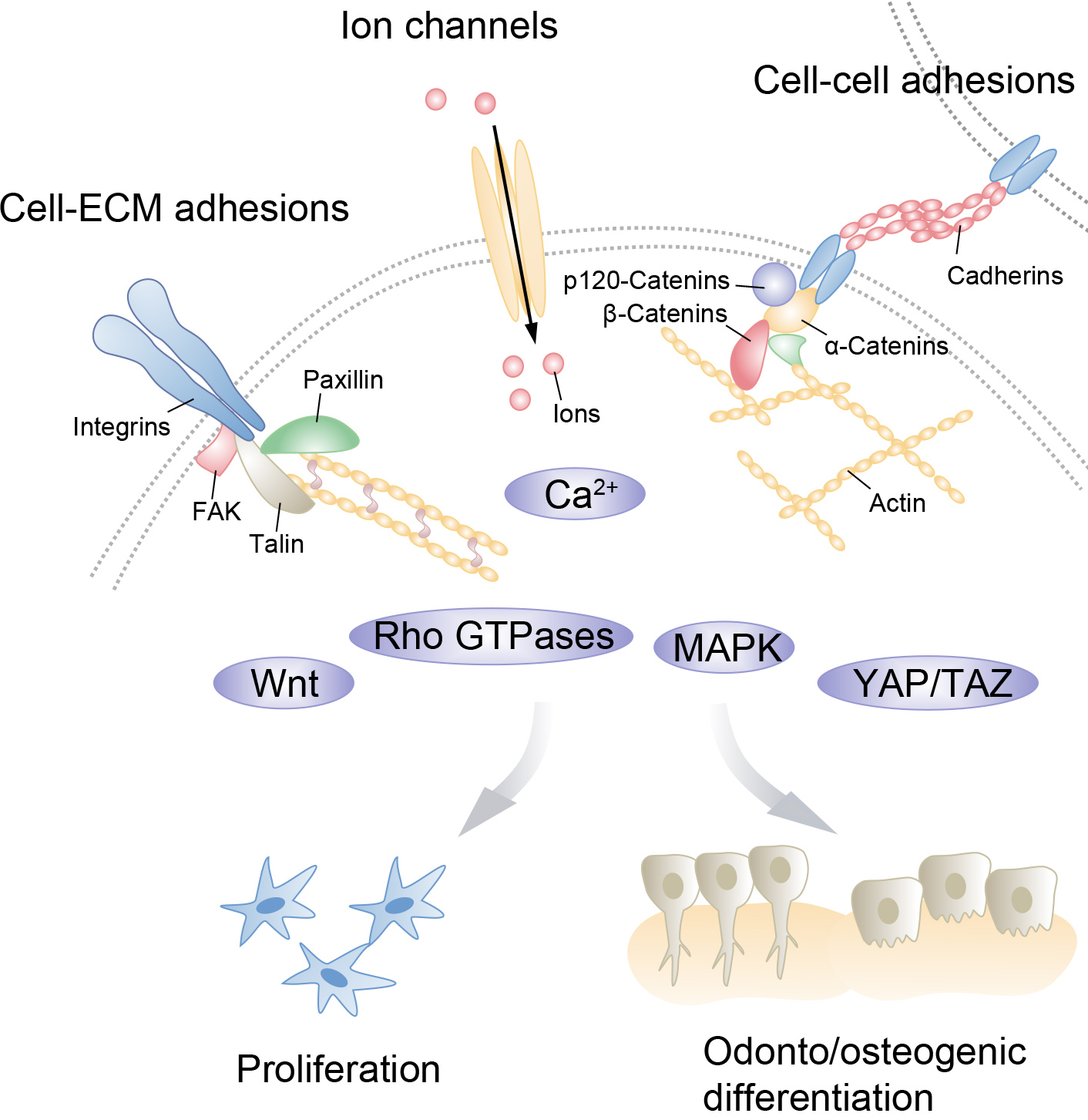 Fig. 1.
Fig. 1.Interconnected mechanical network in dental pulp stem cells (DPSCs). Various mechanical cues from the cell and the extracellular environment activate mechano-sensing factors on the cell surface. These factors, including cell-extracellular matrix (ECM) adhesion complexes such as focal adhesions and cell-cell adhesion such as cadherin–catenin junctions and ion channels, regulate specific mechanotransduction pathways, which affect DPSC proliferation and odonto/osteogenesis.
Adherens junctions (AJs), one of the major mechanosensory cell-cell junction structures, allow cells to be precisely interconnected, ensuring the structural stability of the multicellular layer of specific tissues, such as the epithelium and pulp [66, 67, 68]. The transmission of mechanical signals between cells varies with cell density [69]. Cells can sense compressive or tensile forces at intercellular contacts and respond by modifying the biomedical properties of adhesion receptors, constituting mechanochemical feedback loops that eventually affect biological behaviors, including cell proliferation [70]. Proper intercellular mechanical signal exchanges are critical for tissue homeostasis.
The most widely studied adhesion receptors for modifying cell-cell junctions and
mediating mechanotransduction are cadherins [71, 72]. Various cadherin members
are specifically expressed in tissues. E- and P-cadherin are most prevalent in
the epithelia, whereas N-cadherin is more frequently present in non-epithelial
cells. Further, only blood vessels express VE-cadherin. Notably, these molecules
are structure equivalent [73]. Classic cadherins phenotypically interact with
adjacent cell surfaces and contribute to AJ formation. Classic cadherins consist
of a single-pass transmembrane domain, an N-terminal extracellular domain with
five cadherin domains that create adhesive contacts, and a cytoplasmic domain
associated with actin-binding proteins, including
Endogenous or exogenous stimuli can be converted into biochemical signals by
AJs. However, the cadherin-catenin core complex, which links cadherin to the
actin cytoskeleton, regulates the cytoplasmic mechanical signaling chain and
directly coordinates the dynamic organization of the cytoskeleton [77].
p120-catenin (which binds to the membrane-proximal region of the cytoplasmic
domain of cadherins) can modify cell cohesion by decreasing cadherin renewal in
the membrane [78].
Cell-ECM interactions are generally mediated by focal adhesions (FAs), which have been demonstrated to physically connect the ECM to the cytoskeleton [84]. Cells are enclosed in their local ECM, and can detect mechanical changes caused by varying ECM components [85]. Mechanochemical feedback loops also exist, whereby cells release synthesizing or degrading enzymes that eventually modify the mechanical properties of the ECM [86]. Cell morphology and motility respond quickly to mechanical variations, influencing tissue and organ development, wound repair, and immunological response [87, 88, 89].
The ECM, which mainly consists of collagen fibers, elastin, proteoglycans, and
glycoproteins, contains a large amount of biological information [90]. Integrin,
a key mechanoreceptor or FA, may identify certain ECM ligands and transmit
mechanical signals intracellularly by binding to kindlin, talin, fibronectin, or
vinculin, which are eventually linked to the actomyosin cytoskeleton [91]. The
quantity and intensity of FAs vary in mechanically induced conformations of
integrin-ligand complexes [92]. Talin and vinculin are F-actin-binding proteins.
The head and tail domains of vinculin bind talin and actin, respectively.
Vinculin enhances Talin-F-actin links by recruiting more F-actin [93]. Focal
adhesion kinase (FAK), paxillin, Arp2/3, and
As FAs and AJs are intracellularly related to the actin cytoskeleton, they share a common collection of receptor proteins and signaling molecules, including the Rho family of GTPases and vinculin, forming a cross-regulation network [98, 99]. FAs and AJs communicate and collaborate to maintain the mechanochemical signal ecology and achieve tensional homeostasis within tissues.
Mechanosensitive ion channels, including Piezo and transient receptor potential
(TRP) channels, are vital clusters of operative proteins that convert physical
stimuli into intracellular biological signals [100]. These channels, located in
the cell membrane, open and close in response to mechanical tension, voltage, and
ligand binding, thereby mediating the influx of cations, such as Ca
Similar to Piezo channels, TRP channels serve as signal transducers by modifying
the intracellular Ca
Rho GTPases are a family of small G proteins [116] that have been found to
regulate a variety of fundamental cellular processes, including morphogenesis,
cell migration, cell division, and gene expression [117]. The link between
mechanical cues and Rho GTPases has been widely recognized [118, 119, 120]. One of the
major mechanical cues regulating Rho GTPases is the ECM, where integrins and
fatty acids are the main mediators. For instance, ECM stiffness modulates the
activity of Rho GTPases through focal adhesion kinases and integrins, leading to
actin cytoskeleton assembly via the LIMK-cofilin pathway [121, 122]. Integrin
Ion channel activation stimulates Rho GTPases by inducing Ca
YAP/TAZ is a transcriptional co-activator initially discovered downstream of the Hippo signaling pathway. YAP/TAZ comprises a cascade signaling module of two pairs of kinases: MST1/2 and LATS1/2 [123]. MST1/2 and LATS1/2 function as core protein kinases that phosphorylate YAP/TAZ, leading to the inhibition of nuclear localization and transcriptional coactivation [124]. Although the cytoplasmic restriction of YAP/TAZ promotes their degradation or regulates other signaling pathways, the accumulation of YAP/TAZ in the nucleus drives their interaction with DNA-binding transcription factors, such as TEADs, thereby regulating cell proliferation and differentiation [125].
The ability of YAP and TAZ to respond to diverse mechanical inputs underscores
their importance in the regulation of mechanotransduction. The status of YAP/TAZ
activity is based on their cellular localization [126, 127, 128, 129]. For example,
different ECM elasticities and cell spreading can modulate YAP/TAZ localization.
While stiff substrates and large cell spreading promote YAP/TAZ nuclear shift,
contact inhibition, which involves cell geometry remodeling, inhibits YAP/TAZ
activity. Thus, changes in cell-ECM contacts, which are typically affected by
FAs, and cell-cell contacts, which are mediated by cadherin adhesion sites, are
assumed to be strongly related to YAP/TAZ activity [130, 131]. The angiomotin
(AMOT) complex at tight junctions directly restricts YAP/TAZ in the cytoplasm
and/or induces LATS1/2-mediated YAP/TAZ phosphorylation. E-cadherin and
The Wnt/
There are four main MAPK signaling pathways in mammalian cells: extracellular signal-regulated protein kinase (ERK)1/2, c-Jun amino-terminal kinase (JNK), p38 MAPK, and ERK5. Therefore, the MAPK signaling pathway plays a crucial role in cytoskeletal regulation [139, 140]. ERK1/2 modulates cytoskeletal signaling by activating myosin light chain kinase (MLCK), which phosphorylates the light chain regulatory sequence of myosin and induces microfilament contraction [141, 142]. ERK1/2 can affect cytoskeletal signaling by phosphorylating calpain, a calcium-dependent protease that cleaves structural proteins, leading to the breakage of cell adhesion sites. ERK1/2 can also phosphorylate kinases at the local adhesion site, which regulates the activity of integral proteins and prevent their polymerization with piled proteins, thereby facilitating the depolymerization of integral proteins from the ECM. These pathways, which are regulated by ERK1/2 signaling, are critical for maintaining the balance of cytoskeletal dynamics.
The p38 MAPK pathway is considered a stress-activated signaling pathway (SAPK) as it is activated by various environmental stresses [143, 144]. The p38 MAPK signaling pathway is related to anti-proliferation and apoptosis [145], which differs from the function of ERK1/2; thus, an interaction occurs between these two pathways. Understanding the intricate interplay between different MAPK signaling pathways is essential for gaining insights into the mechanisms underlying cell fate determination and tissue homeostasis.
The effects of mechanical stimulation on DPSCs are multifaceted. Multiple factors that control cell fate determination of DPSCs have been identified. Below, we focused on the role of mechanical signaling components in the proliferation and differentiation of DPSCs.
Integrin is crucial for DPSC proliferation in the cell-ECM interplay. For
instance, the inhibition of integrin-
The role of ion channels in DPSC proliferation has been extensively studied. The
activation of Piezo1 by Yoda1 induces ATP release, which promotes DPSC migration,
subsequently activating the P2 receptor purinergic signaling pathway and the
downstream PYK2 and MEK/ERK signaling pathways [149]. The proliferation of DPSCs
is affected by the activation of MAPK/ERK1/2 signaling after 24 h of
low-intensity pulsed ultrasound (LIPUS) stimulation [150]. In contrast, the
presence of the piezo blocker, ruthenium red (RR), inhibits the proliferation of
LIPUS-stimulated DPSCs [151]. These results demonstrate that Piezo positively
modulates DPSC proliferation. The TRPC1 channel inhibitor, SKF96365, inhibits the
proliferation of DPSCs in a dose-dependent manner [152], whereas TRPM4 is
essential for the proliferation and survival of DPSCs by mediating Ca
Rho GTPases, particularly Rac1, have been shown to participate in the regulation of DPSC proliferation. Rac1 silencing suppresses the pro-apoptotic effect of miR-224 in DPSCs. However, whether other Rho GTPases are involved in the regulation of DPSC proliferation remains to be investigated [155].
Based on growing evidence, YAP/TAZ plays a role in the proliferation process.
DPSCs seeded in a static magnetic field accumulate nuclear YAP/TAZ has been
demonstrated to promote cell proliferation [156]. In addition, the inhibition of
TAZ in DPSCs downregulates the regulation of CTGF and Cyr6, and suppresses
cellular proliferation and migration through the TGF-
Wnt signaling is another signaling pathway that is essential for the growth of
DPSCs. The promotion of DPSC stemness by the Wnt signaling pathway is related to
its effects on oxidative metabolism upon activation. In particular, metabolic
remodeling is accompanied by enhanced glycolysis and mitochondrial tricarboxylic
acid cycle (TCA) activity [161]. When Wnt/
Increasing evidence highlights the role of the MAPK signaling pathway in the mechanically stimulated proliferation of DPSCs. When uniaxial stretching is applied to DPSCs, the expression of phosphorylated Akt, ERK1/2, and p38 MAPK is induced, which promotes DPSC proliferation. In contrast, stretch-induced proliferation of DPSCs is abolished after inhibition of the MAPK/ERK pathway. Interestingly, osteocalcin and osteopontin are significantly inhibited by stretching, indicating that stretching inhibits osteogenic differentiation of DPSCs [164]. As previously mentioned, LIPUS stimulation activates MAPK/ERK1/2 signaling in DPSCs to increase their proliferation. Using selective ERK1/2 inhibitors before ultrasound exposure abolished the stimulatory effect on DPSC proliferation, whereas the inhibition of p38 and JNK had no effect [148]. Further, vibrations induce G0/G1 arrest in DPSCs, inhibiting their proliferation [165]. These data emphasize the unique role of ERK1/2 in the MAPK pathway in the presence of vibrations during DPSC proliferation [150]. Atypical physical stimuli may contribute to the proliferation of DPSCs via MAPK signaling. For example, the application of 0.4-Telsa static magnetic fields (SMFs) on DPSCs significantly triggers p38 MAPK and promotes the proliferation of DPSCs. During this process, the cytoskeleton and cell morphology are reorganized [166]. Cells treated with a 0.4-Telsa SMF also exhibit a higher regeneration potential capacity for pulp repair by regulating MAPK signaling. Furthermore, the promotion of proliferation and migration is inhibited by the p38 inhibitor, SB203580 [167].
The role of mechanical ion channels in the process of odonto/osteogenic differentiation of DPSCs has been demonstrated. TRPM7 in DPSCs reduces the expression of specific odontoblast markers, such as ALP, DSPP, BSP, RUNX2, and OSX, indicating that TRPM7 plays an important role in the osteogenic differentiation of DPSCs [154]. Odonto/osteogenic differentiation induced by low-level light-emitting diodes has been shown to be mediated by TRPV1. Specifically, capsazepine, a selective TRPV1 inhibitor, inhibits odonto/osteogenesis in DPSCs [168]. However, the piezo-dependent mechanism mediating DPSC differentiation remains unclear.
Rho GTPases and the downstream effector protein, ROCK, promote the differentiation of DPSCs. Treatment with the C3 exoenzyme, a RhoA/ROCK signaling pathway inhibitor, was found to suppress the expression of RUNX2. In particular, the presence of the C3 exoenzyme has been demonstrated to significantly promote odontoblast differentiation of DPSCs in the late stage without affecting the early stage. An interesting research direction would be to determine whether other Rho GTPases can mediate mechanical signals to regulate DPSC differentiation [169].
The mechanoregulation of YAP/TAZ during DPSC differentiation has been reported
previously. SMFs rearrange the cytoskeleton of DPSCs and recruit YAP/TAZ to the
nucleus, which upregulates the corresponding genes, CTGF and
ANKRD1, finally promoting DPSC mineralization [156]. Consistently, the
roughness and pore sizes associated with the scaffold topographic cues affect the
nuclear localization of YAP by altering the arrangement and morphology of
cellular F-actin, thereby promoting the odonto/osteogenic differentiation of
DPSCs [170]. The topographical factors of PGLA membranes promote the nuclear
translocation of
Emerging evidence connects DPSC differentiation to mechanically induced Wnt
signaling. Pulsed electromagnetic fields (PEMFs), such as DSPP, DMP1, and RUNX2,
induce a significant increase in odontogenic markers, and
GSK-3
Several studies have supported the role of the MAPK pathway in DPSC differentiation. Exposure to medium-magnitude sonic vibration was found to enhance the odontogenic differentiation of DPSCs, which was accompanied by an increase in the expression of osteogenic markers (osteocalcin, BMP-2, and ALP). These studies suggest that mechanical vibrations are related to MAPK signaling during DPSC differentiation. Nevertheless, 0.4-Telsa SMF exposure promoted a significant increase in the expression of DSPP and DMP-1, whereas DPSC differentiation was significantly reduced in the presence of the p38 inhibitors, SB203580 and SMF [167]. Another study revealed that the odontoblastic differentiation of DPSCs under mechanical compression is mediated via the MAPK pathway by ERK1/2 and p38 rather than by JNK, as the phosphorylation level of JNK remains the same. In addition, coordination between the MAPK and Wnt signaling pathways has been demonstrated to play a role in promoting DPSC differentiation [179].
Recently, there has been a significant interest in the role of mechanical stimuli in tissue homeostasis. DPSCs, which exhibit self-renewal and multilineage potential, play a critical role in pulp homeostasis and restorative dentin formation. This review provides further insights into the biomechanical properties of DPSCs. Notably, the diversity of mechanical stimuli increases the potential intricate interaction in DPSCs, thereby providing a greater opportunity to explore the interplay between each mechanical stimulation factor in determining DPSC fate. Thus, mechanical cues may offer innovative biomaterial platforms or biochemical-mechanical strategies that regulate the differentiation and proliferation of DPSCs by interconnecting cell microenvironments, biomaterials, and cell behaviors. Overall, this study provides a theoretical basis for DPSC applications in tissue engineering and regenerative medicine.
JZ, WenD and WeiD conceived the study conception, design and manuscript preparation. JZ and SW preformed literature review. DG contributed to figure preparation. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
Not applicable.
Not applicable.
This work was supported by NSFC grant 82201003, Natural Science Foundation of Sichuan (No. 2022NSFSC0612), and the Research Funding from West China Hospital of Stomatology Sichuan University (RCDWJS2023-11).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

