1 Laboratory of Influenza Research, College of Veterinary Medicine, Chungnam National University, 34134 Daejeon, Republic of Korea
2 Institute of Influenza Virus, Chungnam National University, 34134 Daejeon, Republic of Korea
Abstract
Background: Highly pathogenic H5Nx viruses cause avian influenza, a
zoonotic disease that can infect humans. The vaccine can facilitate the
prevention of human infections from infected poultry. Our previous study showed
that an H5 cleavage-site peptide vaccine containing the polybasic amino acid RRRK
could protect chickens from lethal infections of the highly pathogenic H5N6 avian
influenza virus. Methods: Chickens immunized with the various polybasic
amino combinations (RRRK, RRR, RR, R, RK, and K) of H5 cleavage-site peptides
were challenged with highly pathogenic H5N6 avian influenza viruses. The
challenged chickens were monitored for survival rate, and viral titers in swabs
and tissue samples were measured in Madin-Darby canine kidney (MDCK) cells using
the median tissue culture infectious dose 50 (log
Keywords
- vaccine
- peptide
- highly pathogenic avian influenza virus
The highly pathogenic (HP) H5Nx viruses belong to the influenza A virus group of the family Orthomyxoviridae and contain enveloped RNA genomes [1]. Their genomes consist of eight segments that encode 10 proteins: polymerases—polymerase basic proteins 1 and 2, and polymerase acidic protein; two major surface proteins—hemagglutinin (HA) and neuraminidase; matrix proteins M2 and M1; non-structural proteins NS1 and NS2; and the nucleocapsid protein [1, 2, 3].
The influenza A virus has caused previous human pandemics: the H1N1, H2N2, H3N2, and H1N1 pandemics in 1918, 1957, 1968, and 2009, respectively [1]. HP H5Nx avian influenza viruses are considered potential pandemic viruses because they have infected numerous poultry flocks, including chickens and ducks in many countries, and are endemic to wild birds [4, 5, 6, 7, 8, 9, 10, 11].
To control the outbreak of HP H5Nx viruses, conventional inactivated vaccines were developed to protect poultry [12, 13]. Vaccine efficacy is related to neutralizing antibodies against the HA protein on the surface of avian influenza viruses.
Various methods were used to develop vaccines against HP H5Nx avian influenza viruses for poultry. Plant-derived H5 hemagglutinin antigens protected chickens from the lethal infections of HP H5N1 avian influenza viruses [14]. Newcastle disease virus expressing H5 hemagglutinin protein protected chickens from the HP H5N2 avian influenza viruses [15]. The H5/H7 trivalent inactivated vaccines protected chickens, ducks, and geese from the infections of HP H5N1, H5N8, and H7N9 viruses [16].
Our previous study showed that an H5 cleavage-site polybasic peptide vaccine (RRRK-containing cleavage peptide) protected chickens against the HP H5 viruses [17]. In this study, we investigated the role of each amino acid of RRRK in its action against the HP H5 avian influenza virus.
HP avian influenza virus, A/waterfowl/Korea/S57/2016 (H5N6) (clade
2.3.4.4.), containing the polybasic amino acids (RRRK) at the HA cleavage site,
was employed in this study. Madin–Darby canine kidney (MDCK) cells (American Type Culture Collection, VA, USA) were used for viral amplification.
The MDCK cell line was validated by STR profiling and tested negative for mycoplasma.
Cells were all cultured in a humidified incubator at 37 ℃ and 5% CO
Synthesized H5 cleavage-site peptides were obtained from Peptron Co. (Daejeon, South Korea) and labeled with keyhole limpet hemocyanin. The synthesized peptides are listed in Table 1.
| Peptide sequences | Peptide names |
| KLH-TGLRNSPLRERRRKR/GLFGAIAGFIEGGWQ | RRRK-containing peptide |
| KLH-TGLRNSPLRERRRR/GLFGAIAGFIEGGWQ | RRR-containing peptide |
| KLH-TGLRNSPLRERRR/GLFGAIAGFIEGGQ | RR-containing peptide |
| KLH-TGLRNSPLRERR/GLFGAIAGFIEGGWQ | R-containing peptide |
| KLH-TGLRNSPLRERKR/GLFGAIAGFIEGGWQ | RK-containing peptide |
| KLH-TGLRNSPLREKR/GLFGAIAGFIEGGWQ | K-containing peptide |
| KLH-TGLRNSPLRER/GLFGAIAGFIEGGWQ | RRRK(-) peptide |
KLH, keyhole limpet hemocyanin.
Fertilized eggs (from white leghorns) were obtained from local farms and hatched in the laboratory. The hatched chicks were grown for 2 weeks before utilization for the vaccine study. Chickens (n = 13 per group) were intramuscularly inoculated with 300 µL containing 5.0 µg of keyhole limpet hemocyanin-labeled H5 cleavage-site peptide and 30% oil (SEPPIC, Courbevoie, France). Booster doses were administered to the immunized chickens 3 weeks after the first dose.
Immunized chicken sera were collected a week after administration of the second dose to measure antibody induction.
Enzyme-linked immunosorbent assay (ELISA) was performed to determine antibody titers with bovine serum albumin (BSA)-labeled peptides: BSA-TGLRNSPLRERRRKR/GLFGAIAGFIEGGWQ, BSA-TGLRNSPLRERRRR/GLFGAIAGFIEGGWQ, BSA-TGLRNSPLRERRR/GLFGAIAGFIEGGQ, BSA-TGLRNSPLRERR/GLFGAIAGFIEGGWQ, BSA-TGLRNSPLRERKR/GLFGAIAGFIEGGWQ, BSA-TGLRNSPLREKR/GLFGAIAGFIEGGWQ, and BSA- TGLRNSPLRER/GLFGAIAGFIEGGWQ.
The wells in the immunoplate were coated with 100 µL of peptide (4
µg/mL) in coating buffer, incubated overnight at 4 °C, and then
washed three times with PBS–tween 20 (0.05%). The wells were blocked with 100
µL of 1% BSA in PBS (pH 7.4) for 2 h at room temperature and then
100 µL of diluted chicken sera in PBS (1:100) was added. The plates
were incubated for 1 h at room temperature. The wells were washed five times and
100 µL of horseradish peroxidase-conjugated rabbit anti-chicken
antibody (Sigma-Aldrich) diluted in PBS (1:5000) was added. The plates were
incubated for 1 h at room temperature. The wells were washed five times with
PBS–Tween 20 (0.05%) and 100 µL of
3,3
The vaccinated chickens were intranasally infected with 1 mL (10
Confluent MDCK cells were treated with trypsin–EDTA (Sigma-Aldrich) and seeded
in a tissue culture plate (96 wells) with MEM containing 10% fetal bovine serum
and 1
Student’s t-test using IBM SPSS Statistics version 20 software (IBM Corp., Armonk, NY, USA) was performed to analyze statistical significance. Statistical significance was based on p-values less than 0.05.
Fig. 1 shows the antibody titers measured using ELISA in sera (n = 10 per group) collected from chickens vaccinated with two doses of H5 cleavage-site peptide containing RRRK. Antibodies were similarly induced in chickens immunized with H5 cleavage-site peptides containing diverse combinations of polybasic amino acids (Table 1), including peptides containing RRRK, RRR, RR, R, RK, K, and RRRK(-), with a mean OD range of 0.51–0.54. The mean OD value for the sera from PBS-mock vaccinated chickens was 0.12.
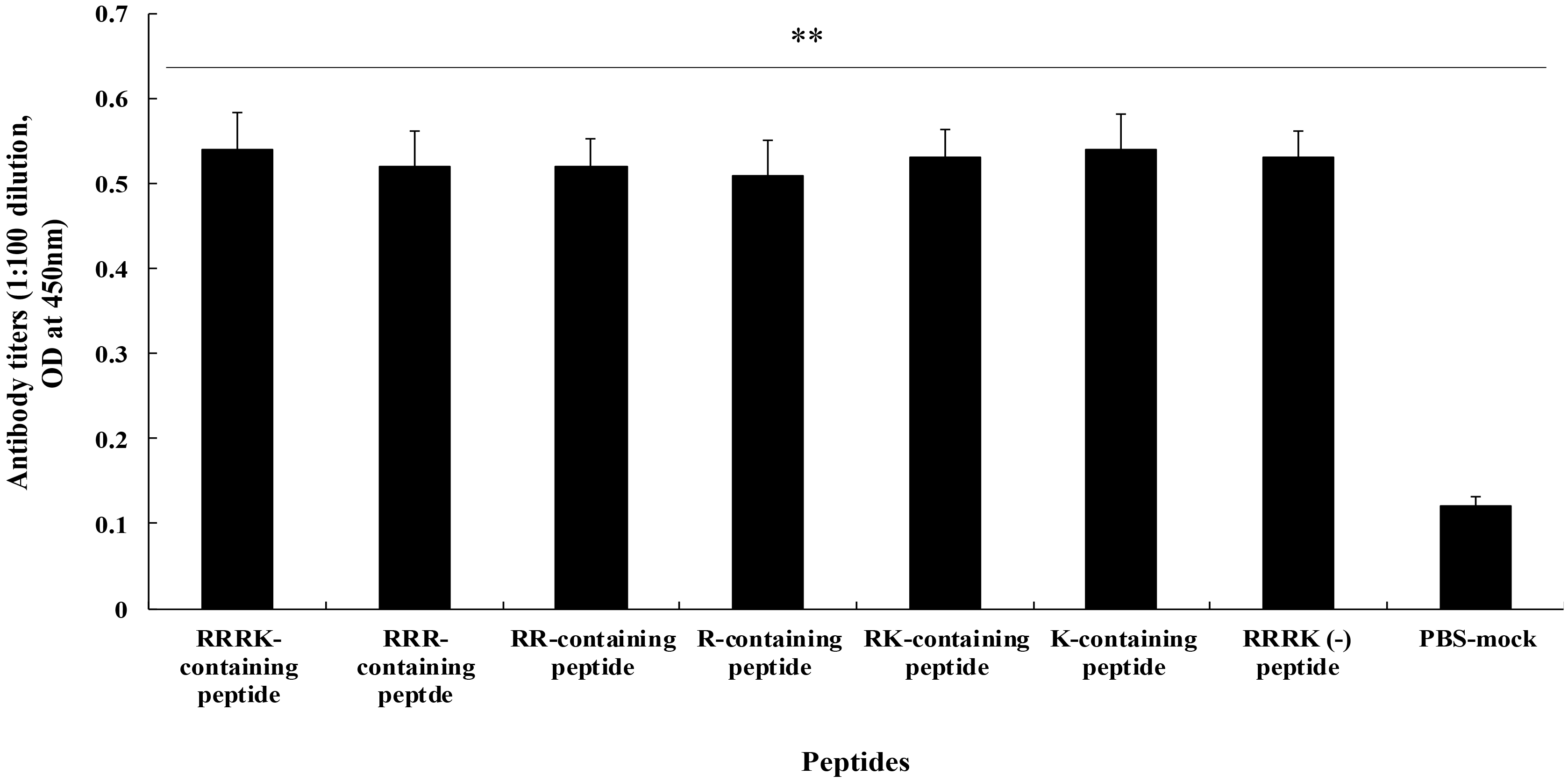 Fig. 1.
Fig. 1.Antibody titers in chickens immunized with H5 cleavage-site
peptides containing different combinations of polybasic amino acids. Sera were
collected from the intramuscularly immunized chickens (n = 13 per group)—with
two doses (5.0 µg per dose) of oil-adjuvanted HLA-labelled peptides—4 weeks
after the vaccination. Antibody titers were measured via ELISA using BSA-labelled
peptides. **p
We aimed to determine the role of individual amino acids of RRRK in protecting chickens from HP avian influenza viral infections. Chickens immunized with most combinations of polybasic amino acids survived when intranasally challenged with the HP H5N6 virus (Fig. 2). All the immunized chickens with H5 cleavage-site peptides, including RRRK-, RRR-, RR-, R-, and K-containing peptides survived, whereas all chickens immunized with RK-containing peptide died 6 days post-infection. All chickens immunized with RRRK(-) peptide and PBS-mock did not survive (Fig. 2).
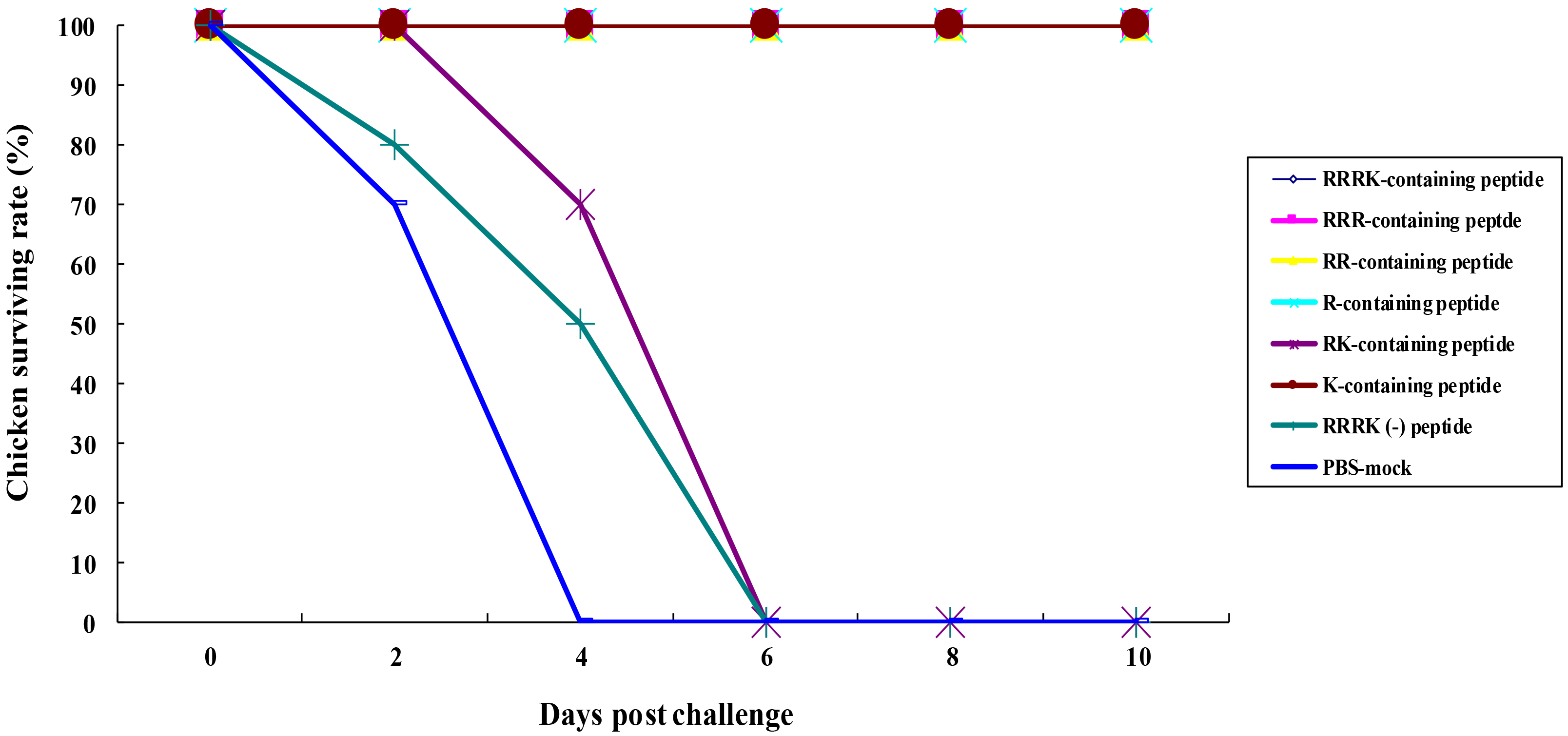 Fig. 2.
Fig. 2.Mortality of the immunized chickens with H5 cleavage-site
peptides containing different combinations of polybasic amino acids. The
immunized chickens (Fig. 1) were intranasally challenged with 1 mL (10
No viruses were detected in the tracheal (Fig. 3A) and cloacal swabs (Fig. 3B)
of chickens immunized with H5 cleavage-site peptides, including RRRK-, RRR-, RR-,
R-, and K-containing peptides. In contrast, high viral titers were detected in
tracheal and cloacal swabs in the chickens immunized with RK-containing peptide
in the range of 2.0 to 4.5 TCID
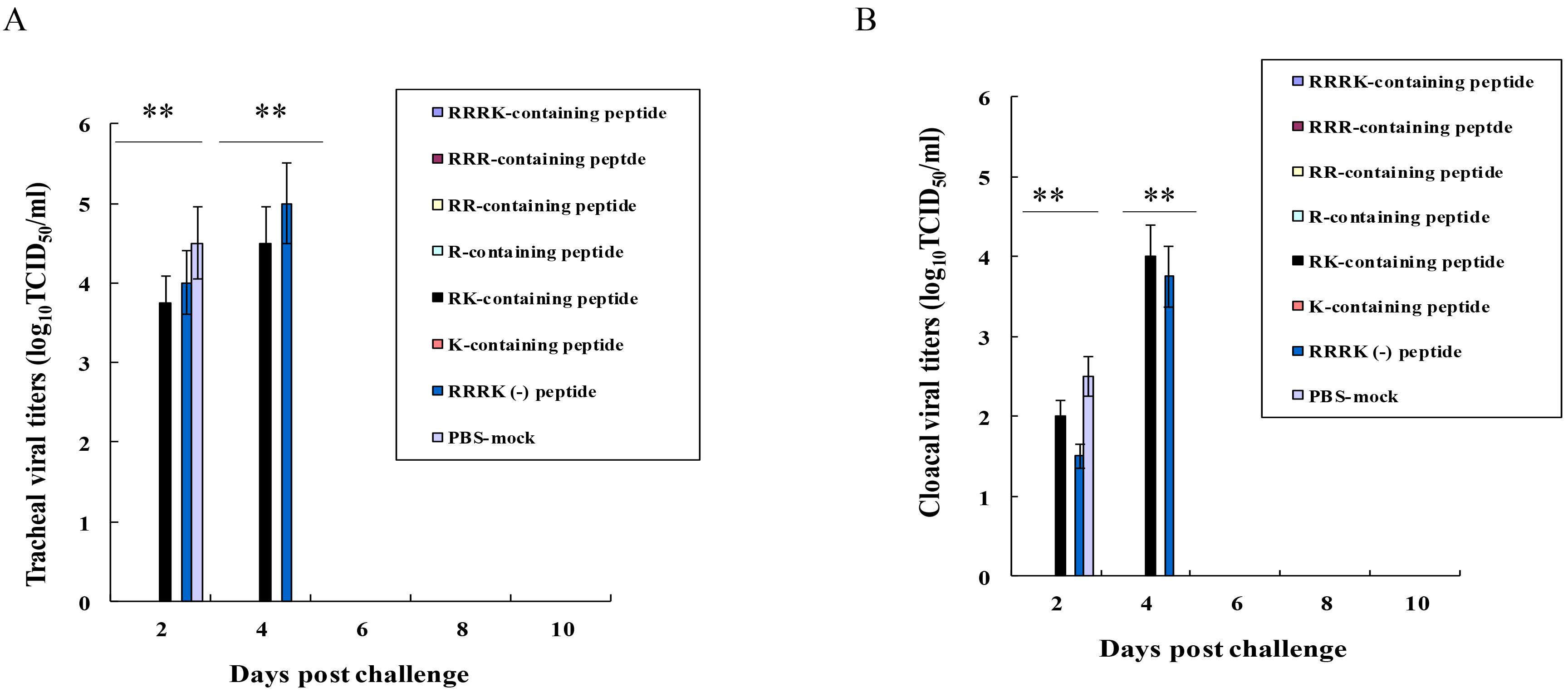 Fig. 3.
Fig. 3.Viral titers in swabbed samples in the challenged chickens.
Swabs in tracheas (A) and cloacae (B) in the challenged chickens (Fig. 2) were
performed in a 2-day interval until 10 days p.i. Viral titers in the swabbed
samples were measured using Madin-Darby Canine Kidney (MDCK) cells by log
The infected chickens (n = 3 per group) were euthanized to collect the lungs and
brains 2 days post-infection to measure tissue viral titers. No virus was
detected in either the lung or brain tissues of the chickens immunized with H5
cleavage-site peptides, including RRRK-, RRR-, RR-, R-, and K-containing peptides
(Table 2), whereas high viral titers were detected in both the lung and brain
tissues of chickens immunized with the RK-containing peptide, in a range of 3.0
to 3.5 TCID
| Vaccine antigens | Mean viral titers in lungs (log |
Mean viral titers in brains (log |
| RRRK-containing peptide | - | - |
| RRR-containing peptide | - | - |
| RR-containing peptide | - | - |
| R-containing peptide | - | - |
| RK-containing peptide | 3.0 |
3.5 |
| K-containing peptide | - | - |
| RRRK(-) peptide | 3.5 |
4.0 |
| PBS-mock | 4.0 |
4.5 |
-: under detection limit, 1 TCID
HP H5Nx avian influenza viruses continue to infect poultry flocks, resulting in significant economic losses in the poultry industry. In addition, these viruses can infect humans and are considered potential pandemic pathogens. We developed a vaccine based on HA cleavage-site peptides containing polybasic amino acids to protect poultry from infections caused by the HP H5 virus.
This study was performed to determine the potential protective role of individual polybasic amino acids in chickens immunized against the HP H5N6 virus. Our results showed that most combinations of polybasic amino acids in the cleavage-site peptide provided complete protection against the HP H5N6 virus in immunized chickens. The RK-containing H5 cleavage-site peptide vaccine did not protect immunized chickens against the virus. The development of broad-spectrum protection strategies against H5Nx infections has been attempted. The herpesvirus of turkeys was developed as a vector expressing the HA antigen of the HP H5N1 virus [19]. Layers vaccinated with this vector 2 weeks prior to infection with various HP H5N1 viruses were fully protected, with reduced shedding of the viruses. Newcastle disease virus vectors encoding H7 or H5 HA were shown to provide complete protection in chickens against HP H7N9 or H5N1 viruses [20]. A chimeric QH/KJ recombinant virus vaccine encoding the HA-1 part of the HP virus A/chicken/China/QH/2017 (H5N6) (clade 2.3.4.4.), and HA-2 part of the HP virus A/chicken/China/KJ/2017 (H5N1) (clade 2.3.2.1.) completely protected immunized chickens against HP H5N6 and HP H5N1 viruses [21].
Our study showed that the H5 cleavage-site peptide containing the RK combination of polybasic amino acids (RRRK) did not protect immunized chickens from HP H5N6 virus infection. The lack of protection of chickens from the vaccine with RK-containing peptides may be due to the absence of antibody induction, which could bind to polybasic amino acids (RRRK) of the HP H5N6 virus. However, the underlying mechanism requires further investigation.
Keyhole limpet hemocyanin (KLH) was used for stimulating the immune response of H5 cleavage-site peptide
vaccines. It is assumed that KLH could stimulate CD4
Polybasic amino acids are a determining factor for HP avian influenza viruses. The deletion of these amino acids for making infectious live attenuated vaccine is necessary to prevent reversion to HP avian influenza viruses. Our cleavage-site peptide containing polybasic amino acids could not be a source for HP reversion since the peptide could not contribute to the creation of a recombinant virus. The previous study to make fowlpox vaccine viruses co-expressing avian influenza H5 and chicken IL-15 deleted the polybasic amino acids in the H5 cleavage site [22].
We used homologous BSA-conjugated peptides to measure antibody titers. Further study may need to include heterologous BSA-conjugated peptides to find out the difference in antibody levels.
The polybasic amino acids (RRRK) of H5 cleavage cleavage-site peptide vaccines are important for protecting chickens against HP H5N6 avian influenza virus. The H5 cleavage cleavage-site peptide containing RK did not protect chickens against the virus.
The data are available from the corresponding author upon reasonable request.
SHS had the sole role in designing, data collecting, analyzing, and writing the manuscript.
The vaccine efficacy study in chickens was performed following the protocol (CNU-01191) approved by the Chungnam National University (CNU) Internal Animal Use Committee (202307A-CNU-115). All experiments were performed in accordance with the relevant guidelines and regulations of Chungnam National University, Republic of Korea.
Yunyueng Jang assisted in the preparation of experimental materials.
This work was supported by a grant by the National Research Foundation of Korea (NRF) funded by the Korean government (MSIT) (2019R1A2C2002166812).
The author declares no conflict of interest. Given his role as Guest Editor, SHS had no involvement in the peer-review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Giuseppe Murdaca.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



