1 Laboratory of Applied Biochemistry, Faculté des sciences de la nature et de la vie, Université Frères Mentouri Constantine 1, 25000 Constantine, Algeria
2 Unité de Valorisation des Ressources Naturelles, Molécules Bioactives et Analyses Physicochimiques et Biologiques, Université des Frères MentouriConstantine, 25000 Constantine, Algeria
3 Département de Chimie, Faculté des Sciences, Université Mohammed Boudiaf-M’sila, 28000 M’Sila, Algeria
4 Department of Biomedical Sciences, University of Sassari, 07100 Sassari, Italy
5 Department of Biomedical Sciences, College of Health Sciences, QU Health, Qatar University, 2713 Doha, Qatar
6 Department of Basic Medical Sciences, College of Medicine, QU Health, Qatar University, 2713 Doha, Qatar
7 Department of Medical Laboratory Sciences, College of Health Sciences and Sharjah Institute for Medical Research, University of Sharjah, 27272 Sharjah, United Arab Emirates
Abstract
Pulmonary fibrosis (PF) is a feared component in over 200 interstitial pulmonary diseases, which are characterized by increased alveolar wall thickness, excessive scarring, and aberrant extracellular matrix restructuring that, ultimately, affect lung compliance and capacity. As a result of its broad range of biological activities, including antioxidant, anti-inflammatory, antiapoptotic, and many others, resveratrol has been shown to be an effective treatment for respiratory system diseases, including interstitial lung disease, infectious diseases, and lung cancer. This work reviews the known molecular therapeutic targets of resveratrol and its potential mechanisms of action in attenuating PF in respiratory diseases, including cancer, COVID-19, interstitial lung diseases (ILDs) of known etiologies, idiopathic interstitial pneumonia, and ILDs associated with systemic disorders, such as rheumatoid arthritis, systemic sclerosis, Schrödinger’s syndrome, systemic lupus erythematosus, and pulmonary hypertension. The current issues and controversies related to the possible use of resveratrol as a pharmaceutical drug or supplement are also discussed.
Keywords
- resveratrol
- pulmonary fibrosis
- interstitial lung diseases
- SIRT1
- TGF-β1
- Nrf2
Due to the associated perception of safety, natural compounds instead of synthetic ones are emerging as more desirable therapeutic tools [1, 2, 3, 4, 5]. In fact, over the past two decades, natural supplements have massively conquered the market, while scientific research associated with natural compounds has also extensively increased [6, 7, 8, 9, 10, 11]. Nonetheless, despite the multitude of studies conducted on plant extracts and individual components, the final therapeutic constituent, whether a single molecule or the synergy of several constituents, remains to be clarified.
Among the current natural compounds being analyzed, resveratrol is a naturally occurring polyphenolic phytoalexin, which is present in multiple plants and aliments that possesses anti-inflammatory, antioxidant, vasculoprotective, neuroprotective, and anticancer activities [12, 13, 14, 15]. Due to its extensive range of biological effects, resveratrol has become a widely studied molecule in the treatment of many pathologies, including respiratory system diseases [4, 14, 16, 17, 18].
Pulmonary fibrosis (PF) is the end stage of many diffuse respiratory diseases characterized by excessive extracellular matrix deposition, destruction of pulmonary parenchymal architecture, and loss of lung function, which in most cases leads to the patient’s death [19]. Interstitial lung disease (ILD) refers to a diverse group of disorders that are characterized by varying degrees of inflammation and/or fibrosis in the lung parenchyma and interstitium [20], which includes idiopathic pulmonary fibrosis (IPF) [21]. PF can also be a shared component in connective tissue diseases (CTD), which are a heterogeneous group of inflammatory autoimmune disorders that affect different tissues and organs, including systemic lupus erythematosus (SLE), rheumatoid arthritis (RA), Sjogren’s syndrome (SS), polymyositis (PM)/dermatomyositis (DM), systemic sclerosis (SSc), and mixed connective tissue disease (MCTD), which are commonly associated with ILDs [22]. Conversely, the development of PF can also occur secondary to viral pneumonia, especially during COVID-19 infection, and also in lung cancer [23]. Indeed, lung tumors in IPF patients develop preferentially in the peripheral adjacent zones rather than in the fibrotic areas, thereby showing different histologic distributions and immunohistochemical characteristics compared to non-IPF-associated lung tumors [24, 25].
Furthermore, there is an ever-increasing body of evidence emphasizing the protective role of resveratrol in respiratory system diseases [18]. Additionally, the vast amount of current literature indicates that the therapeutic effects of resveratrol against PF-related pathologies are predominantly linked to its ability to induce sirtuin-1 (SIRT1) expression during oxidative stress, and inflammatory and apoptotic events, which ultimately suggests that it possesses a critical role in attenuating lung fibrosis development [26, 27, 28, 29, 30]. Our work provides an update on the current literature and focuses on both PF and the classical resveratrol-exerted effects, such as antioxidant, antifibrotic, and anticancer [18], alongside systematically and comprehensively reviewing and analyzing the common molecular mechanisms that inter-relate PF to both ILD and CTD as potential resveratrol targets. Here, we schematically describe the classical and specific therapeutic molecular targets of resveratrol that are potentially suggested to attenuate PF as a common component in both ILDs, as primary disorders, and in CTD related to ILD, as secondary disorders. Then, we highlight the molecular mechanisms in ILDs therapeutically targeted by resveratrol, and mechanisms believed to be dependently targeted in ILDs-related to CTD, to provide a new comprehensive panorama that will enhance the way for how resveratrol dosage and administration are visualized and understood in future clinical studies. In this context, we also provide an overview of the aspects regarding the current controversies on the use of resveratrol as a pharmaceutical drug or supplement.
Resveratrol is a natural stilbene monomer, which is chemically known as
3,5,4
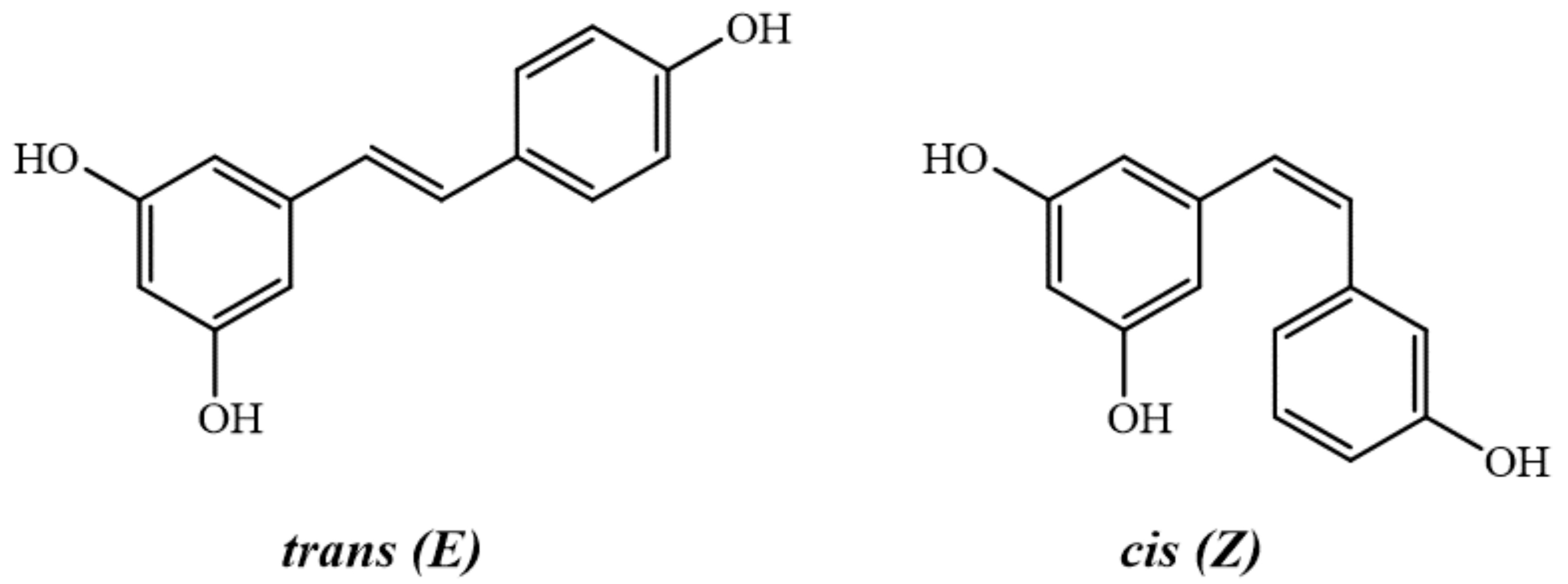 Fig. 1.
Fig. 1.Structures of trans- and cis- resveratrol.
The first report on resveratrol was published by Dr. Michio Takaoka, who studied a poisonous medicinal herb named Veratrum grandiflorum and led to the isolation and identification of this bioactive compound [31]. Plants produce resveratrol in response to external stimuli, such as mechanical harm, ultraviolet radiations, drastic environmental changes, microbial infection, and fungicides [32]. Resveratrol synthesis starts with a non-oxidative deamination reaction of two aromatic amino acids, named L-phenylalanine and L-tyrosine, which leads to the generation of cinnamic acid and 4-coumaric acid, respectively. The hydroxylation reaction catalyzed by cinnamate-4-hydroxylase converts the obtained cinnamic acid into 4-coumaric acid. Next, the transformation of 4-coumaric acid into 4-coumaroyl-CoA by the enzyme 4-coumaroyl CoA ligase generates an active intermediate, which is used by plants in normal growth conditions. Conversely, in stress conditions, the enzyme stilbene synthase (STS) works to condense a portion of the 4-coumaroyl-CoA to 3 molecules of malonyl-CoA through repeated decarboxylating reactions, to ultimately produce one molecule of resveratrol [33, 34, 35, 36, 37]. Resveratrol is abundantly present in many foods and food products, such as grapes, peanuts, plums, blueberries, olive oil, and hops [38, 39] (Table 1).
| Source | Scientific name | Parts | Content (µg/g) | References |
| Grapes | Vitis amurensis | Leaves, stems, and roots | 1.5–7.3 | [40, 41, 42] |
| V. betulifolia | Stems | [43, 44] | ||
| V. chunganensis | Whole plant | [45] | ||
| V. coignetiae | Berries, stems, and leaves | [46, 47] | ||
| V. davidii | Stems | [48] | ||
| V. labrusca | Berries, leaves, and stems | [49, 50, 51, 52, 53] | ||
| V. pentagona | Stems | [48, 54] | ||
| V. riparia | Leaves | [54] | ||
| V. riparia x, V. | Roots | [54] | ||
| berlandieri | ||||
| V. rotundifolia | Berries | [50, 54, 55, 56, 57, 58] | ||
| V. thunbergii | Stems | [59] | ||
| V. vinifera | Berries, cell suspension cultures, stems, and leaves | [35, 60, 61, 62, 63] | ||
| Grape juice | 0.12–0.26 | [64] | ||
| Peanuts | Arachis hypogaea L. | Peanut kernels: raw and roasted | 0.10–2.99 | [65] |
| Skins | [66] | |||
| Roots | [67] | |||
| Blueberries | Vaccinium corymbosum L. | Fruit | 86–170 | [68, 69] |
| Cranberries | V. macrocarpon | Fruit | 90 | [59] |
| Bilberries | V. myrtillus L. | Fruit | 77 | [70] |
| Pistachios | Pistacia vera | Kernel | 6–69.7 | [71] |
| Cocoa | Theobroma cacao L. | Powder | 19–34 | [64] |
| Dark chocolate | 3.8–6.5 | [72] | ||
| Milk chocolate | 0.8–2.6 | [64] | ||
| Plums | Prunus domestica (Arandana) | Skin and pulp | 0.3–2.8 | [73] |
| P. domestica (Laetitia) | Skin and pulp | 1.2–6.2 | ||
| P. domestica (Red beauty) | Skin and pulp | 0.3–0.9 | ||
| P. salicina (Damask) | Skin and pulp | 0.1–0.9 | ||
| P. salicina (Golden Japan) | Skin and pulp | 1.3–1.6 | ||
| P. salicina (Metley) | Skin and pulp | 0.2–0.4 | ||
| Dates | Phoenix dactylifera L. | Fruit | 3.0 | [74] |
| Tomato | Solanum lycopersicum L. | Fruit | 0–2.1 | |
| Itadori | Reynoutria japonica | Roots | [75] | |
| Tea | [76] |
Since oxidative stress and inflammation cause DNA damage and alveolar cell
injuries, they play a crucial role in the pathogenesis and progression of lung
diseases. Moreover, additional lifelong illnesses, such as cancer, metabolic
diseases, and neurological and cardiovascular complications are also associated
with oxidative stress [12]. It is also widely recognized, for example, that
crosstalk between oxidative stress and inflammation in lung tissue can cause lung
fibrosis and associated pulmonary diseases [21, 77, 78, 79, 80, 81, 82, 83]. Resveratrol has been
demonstrated to have free radical scavenging ability [84], antioxidant properties
[85], and antioxidant enzyme expression modulatory activity [84], all of which
can eventually attenuate oxidative stress-related diseases [86]. Indeed,
resveratrol can exert its antioxidant ability by directly scavenging free
radicals, including superoxide radical (O
Resveratrol has also been shown to indirectly scavenge free radicals through its
modulatory effect on the expression and activity of antioxidant enzyme genes,
such as superoxide dismutase (SOD), catalase (CAT), glutathione
(GSH), and glutathione peroxidase (GPX) [96]. Moreover, other
endogenous antioxidant enzymes appear to be involved in the resveratrol free
radical scavenging mechanisms, including NADPH oxidase (O
Interestingly, other signaling pathways were also found to be implicated in
resveratrol-mediated oxidative stress attenuation. In a murine model, resveratrol
attenuates LPS-induced epithelial–mesenchymal transition (EMT) and PF by
suppressing oxidative stress and tumor growth factor
Inflammation is a localized protective response to tissue damage and/or
microbial invasion, which serves to isolate and destroy the harmful agent and
prepare the injured tissue for eventual repair and healing [106]. Chronic
inflammation leads to tissue fibrosis [106]. The fibrosis of parenchymal organs
(i.e., lung or other organs) is caused by prolonged injury, inflammatory
diseases, deregulation of the normal wound healing processes, and extensive
deposition of extracellular matrix (ECM) proteins [21, 81, 107]. In this context,
it has been demonstrated that resveratrol activates several anti-inflammatory
responses. In acute lung injuries, a fibrotic life-threatening condition, studies
on animals demonstrated that resveratrol exerts a protective effect by inhibiting
the myd88-dependent toll-like receptor 4 (TLR4) signaling pathway [108]. In lung
tissue, such an effect is mediated by SIRT1 activation, decreased inflammatory
cytokines, such as IL-1
It is widely recognized that chronic inflammatory conditions are associated with
cancer, especially in people of older ages [113]. A recent study performed in
A549 lung cancer cells showed that the NLR family pyrin domain-containing protein
3 (NLRP3) inflammasome was implicated in tumor-related inflammation and its
progression [114]. Moreover, a significant marker of NLRP3 inflammasome activity
is NF-
Andrews et al. (2016) [116] showed that resveratrol was able to alleviate non-typeable Haemophilus influenzae (NTHi)-induced airway inflammation by upregulating a negative modulator of inflammation, the myeloid differentiation factor 88 (MyD88). Such an effect appears to be driven by a resveratrol-mediated, cAMP-PKA-dependent, inhibition of NTHi-induced ERK1/2 phosphorylation and an increase in mitogen-activated protein kinase phosphatase-1 (MKP-1) expression [116].
Although the definitive molecular mechanisms are yet to be fully understood,
compelling data indicated that resveratrol may be effective in inhibiting the
occurrence and development of lung cancer [117]. Nonetheless, a wide range of
data indicated that resveratrol anticancer properties were mediated through
mechanisms such as apoptosis induction, autophagy regulation, cell cycle
inhibition, angiogenesis suppression, and anti-oxidation [118]. In one study,
resveratrol was reported to decrease cell viability, inhibit cell proliferation,
and induce cell senescence and apoptosis by disrupting the intracellular
antioxidant defense, increasing ROS production, and destroying lung cancer cell
redox homeostasis [119]. Resveratrol has the ability to bind to the pro-oncogenic
nuclear receptor 4A1 (NR4A1, Nur77), thereby hindering NR4A1-dependent
transactivation in lung cancer cells. This implies that resveratrol can act as an
NR4A1 antagonist and elicit effects similar to NR4A1 knockdown. Hence,
resveratrol might be a promising therapeutic approach for lung cancer patients
with high NR4A1 expression, which is considered a negative prognostic factor for
certain types of solid tumor-derived cancers [120]. Interestingly, resveratrol
can induce apoptosis and inhibit the viability of the small-cell lung cancer
(SCLC) cell line H446. Indeed, this cancer currently lacks effective treatments,
meaning that resveratrol can affect cell survival through a redox-dependent,
PI3K/Akt/c-Myc-mediated signaling mechanism [121]. Moreover, resveratrol
complexation with sulfobutylether-
Resveratrol was indicated to decelerate the development of inflammatory chronic diseases [128]. Additionally, it was also suggested that the anti-inflammatory and anti-fibrotic action of resveratrol may promote a potential therapeutic effect toward pulmonary diseases with fibrotic complications [129] (Fig. 2).
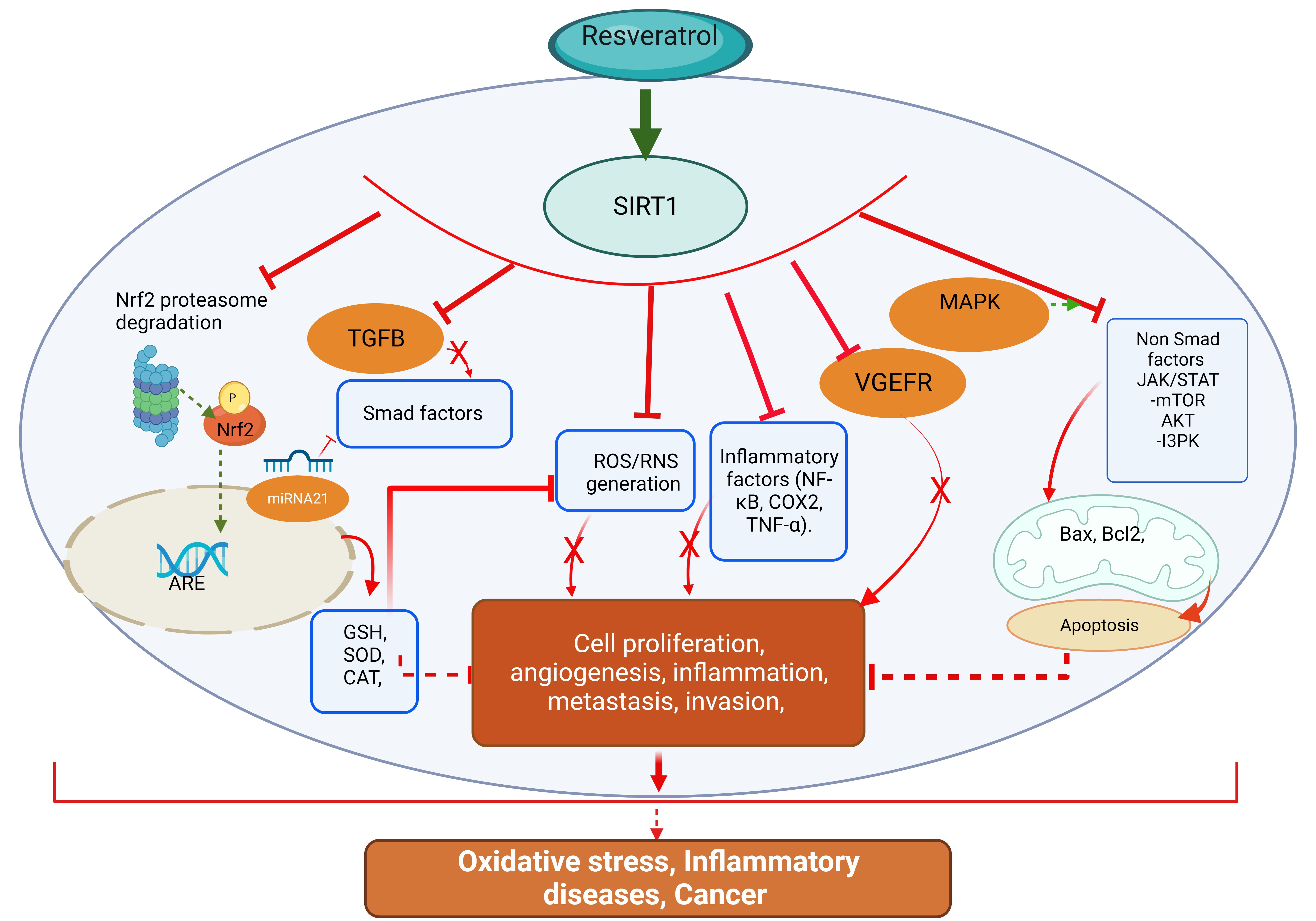 Fig. 2.
Fig. 2.Molecular pathways underpinning resveratrol antioxidant,
anti-inflammatory, and anticancer effects. ARE, Antioxidant Response Elements; GSH, glutathione; SOD, dismutase; CAT, catalase; TNF-
PF is a feared component in over 200 interstitial pulmonary diseases, which are characterized by reduced lung compliance and capacity as well as increased alveolar wall thickness [21, 23]. Some of these diseases have known etiologies; however, many of them have a complex etiology that remains obscure or not fully elucidated [20]. ILD refers to a diverse group of disorders characterized by varying degrees of inflammation and/or fibrosis in the lung parenchyma and interstitium [22]. These diseases are broadly classified into different categories, including ILDs with a known etiology, which are those that occur from occupational/environmental factors, drugs, hypersensitivity reactions, and infections; ILDs associated with systemic disorders, such as sarcoidosis and collagen vascular disorders; rare miscellaneous conditions, such as eosinophilic granuloma, and the idiopathic interstitial pneumonias (IIPs) [20]. In the latter category, IPF figures as the most common form among the 27 known classes, while other forms include nonspecific interstitial pneumonia, cryptogenic organizing pneumonia, acute interstitial pneumonia, respiratory bronchiolitis-associated interstitial lung disease, desquamative interstitial pneumonia, and lymphocytic interstitial pneumonia (Fig. 3) [21].
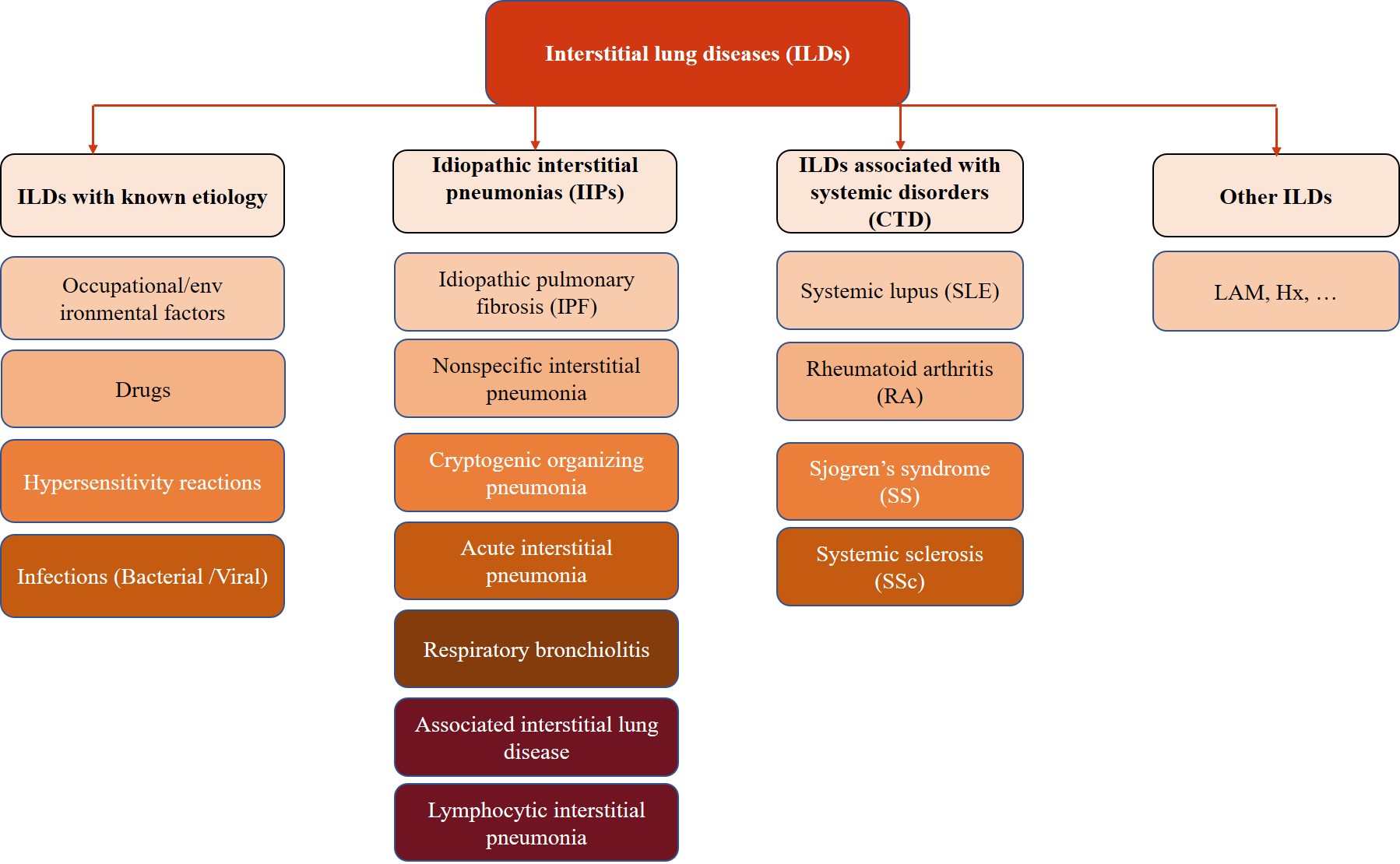 Fig. 3.
Fig. 3.Schematic summary of the different interstitial lung diseases.
IPF is a severe and progressive disease with limited treatment options, while exacerbations are associated with a high degree of morbidity and mortality [130]. Alternatively, the CTD-associated ILDs may share PF as an associated pattern. CTD is a heterogeneous group of inflammatory disorders that affect bone, cartilage, tendons, ligaments, muscle, joints, blood vessels, and specific organs [22]. Many CTDs, such as SLE, RA, SS, polymyositis (PM)/dermatomyositis (DM), SSc, and mixed connective tissue disease (MCTD), are autoimmune-mediated [22]. CTD-related interstitial lung disease (CTD-ILD) is one of the leading causes of CTD-associated morbidity and mortality. Clinically, CTD-ILD is highly heterogenous and involves rheumatic immunity and multiple manifestations of respiratory complications that affect the airways, vessels, lung parenchyma, pleura, and respiratory muscles [22]. The major CTD pathological feature is the chronic inflammation of blood vessels and connective tissues, which can progress and affect any organ, ultimately leading to multisystem damage [22]. The complex CTD-ILD etiology includes genetic risks, epigenetic changes, and dysregulated immunity, which interact and lead to disease onset and development from various ill-defined environmental triggers [22]. CTD-ILD exhibits a broad spectrum of clinical manifestations, ranging from asymptomatic to severe dyspnea and from respiratory system single-organ involvement to multi-organ involvement [22]. The disease course is also featured by remissions and relapses, stability, or slow progression over several years to rapid deterioration, and presents highly progressive clinical manifestation from the onset of the disease. Currently, the diagnosis of CTD-ILD is primarily based on a distinct pathology subtype(s) and imaging, as well as the presence of related CTD and autoantibodies profiles. Meticulous comprehensive clinical and laboratory assessments to improve diagnostic processes and management strategies are much needed [22]. In this regard, heterogeneity and the lack of preclinical ILD and CTD-ILD pathogenesis models further complicate the establishment of an accurate diagnosis [131].
A great body of research on identifying the PF pathophysiological mechanisms is still ongoing owing to the diversified etiologies of this disease, which may result in different primary mechanisms that all lead to a common fibrotic outcome. PF patients share the common characteristic of a manifestation of excessive fibrotic components, which lead to the development of PF [132]. Moreover, they can extend to IPF following the development of a severe pulmonary fibrotic disease with an unknown insult [23].
The progression of PF may be stimulated by microenvironmental insults of intrinsic (genetic, age, and gender) or extrinsic origins (viral infections such as COVID-19, cigarette smoke, or occupational exposure to irradiations) targeting the alveolar epithelial cells (AECs), which are responsible for regulating many inflammatory aspects [23]. Following the initial insult, AECs initiate an immune response to counteract the microenvironmental insult, thereby promoting the resolution and repair of the damaged tissue. Nevertheless, if this process is not correctly performed, it will result in the formation of a perpetual fibrotic scar, which is characterized by ECM constituents, such as fibronectin, interstitial collagens, hyaluronic acid, and proteoglycans [133, 134]. At the same time as the tissue injury occurs, endothelial cells and epithelial cells produce inflammatory mediators that activate antifibrinolytic-coagulation mechanisms, resulting in ECM formation and deposition [135]. Platelet accumulation and the recruitment of inflammatory cells (e.g., eosinophils, macrophages, neutrophils, and lymphocytes) to the site of injury [136] induce a cascade of chemokines and cytokines that additionally enhances the inflammatory response and stimulates fibroblast proliferation and mobilization [137, 138]. Generally, fibrosis occurs when the injury insult is aggressive or when the healing process mechanism is altered [135].
Conversely, the activation of myofibroblasts, which is responsible for collagen
synthesis, alveolar epithelial type II cells (AEC2) apoptosis, and PF formation,
is accompanied by a dysregulation in the immune response [139]. Inflammatory
cytokines, including TGF-
At a cellular level, previous studies have indicated the presence of extensive B cell infiltrations in lung tissue of SSc-ILD patients [146], while in RA-ILD, a prominent increase of follicular B cell hyperplasia and CD4+ cell number has been observed [147, 148]. In RA-ILD and SSc-ILD patients, T cells release fibrogenic mediators, which subsequently stimulate fibroblasts and prime the fibrotic response [149]. Toll-like receptors (TLRs), which are critical components of innate immunity, have multifaceted effects on ILD in patients with CTD; indeed, they have been proposed as markers of ILD progression [150]. Correlation studies showed that TLR2 [151] and TLR9 [152] are profibrotic, while TLR3 [153] is antifibrotic in PF. On the other hand, TLR4 can be either profibrotic [154] or antifibrotic [155] depending on the micro-environment. However, the contribution of other innate players on CTD-ILD remains to be explored [22].
Promoting myofibroblast development is another mechanism through which the lungs
respond to insults or injuries. Myofibroblasts are responsible for ECM secretion
within the damaged area, a phenomenon that is supposed to prompt the repair
process; however, an imbalance in this process with an altered ECM deposition
results in the formation of fibrosis [133]. Various factors, including
TGF-
Oxidative stress is another important player in PF development; indeed,
excessive amounts of ROS are generated during disease development [21]. Since a
large amount of O
Based on the previous PF presentation, the pathogenesis of diseases, such as
ILDs and CTD-ILD, is believed to be tightly linked to fibrosis. However, it is
largely accepted that the impairment of inflammatory-associated pathways is the
primary molecular mechanism that promotes profibrotic cascades and leads to
aberrant tissue remodeling and fibrogenesis [181]. Nonetheless, conventional
treatments are not always efficient in reversing PF progression and provoke
multiple side effects; hence, alternative therapies are necessary for controlling
the patient’s symptoms and ameliorating their quality of life [182]. The
promising anti-PF roles for natural compounds have increasingly attracted the
interest of scientists over the last two decades. Indeed, natural compounds
encompass multiple biological properties and potent safety standards, which are
pivotal in PF prevention and treatment [183]. Interestingly, natural
product-based treatments have demonstrated promising outcomes in PF management,
with studies demonstrating that natural products can exert antifibrotic effects
by inhibiting inflammation, oxidative stress, endothelial mesenchymal transition
(EndMT), and counteracting TGF-
In addition to its antioxidant, anti-inflammatory, antiapoptotic, and anticancer
effects, resveratrol was found to exert preventive and therapeutic effects on PF
by impacting signaling pathways specifically implicated in PF-associated
pathologies. Indeed, the inhibition of fibrogenic events such as
TGF-
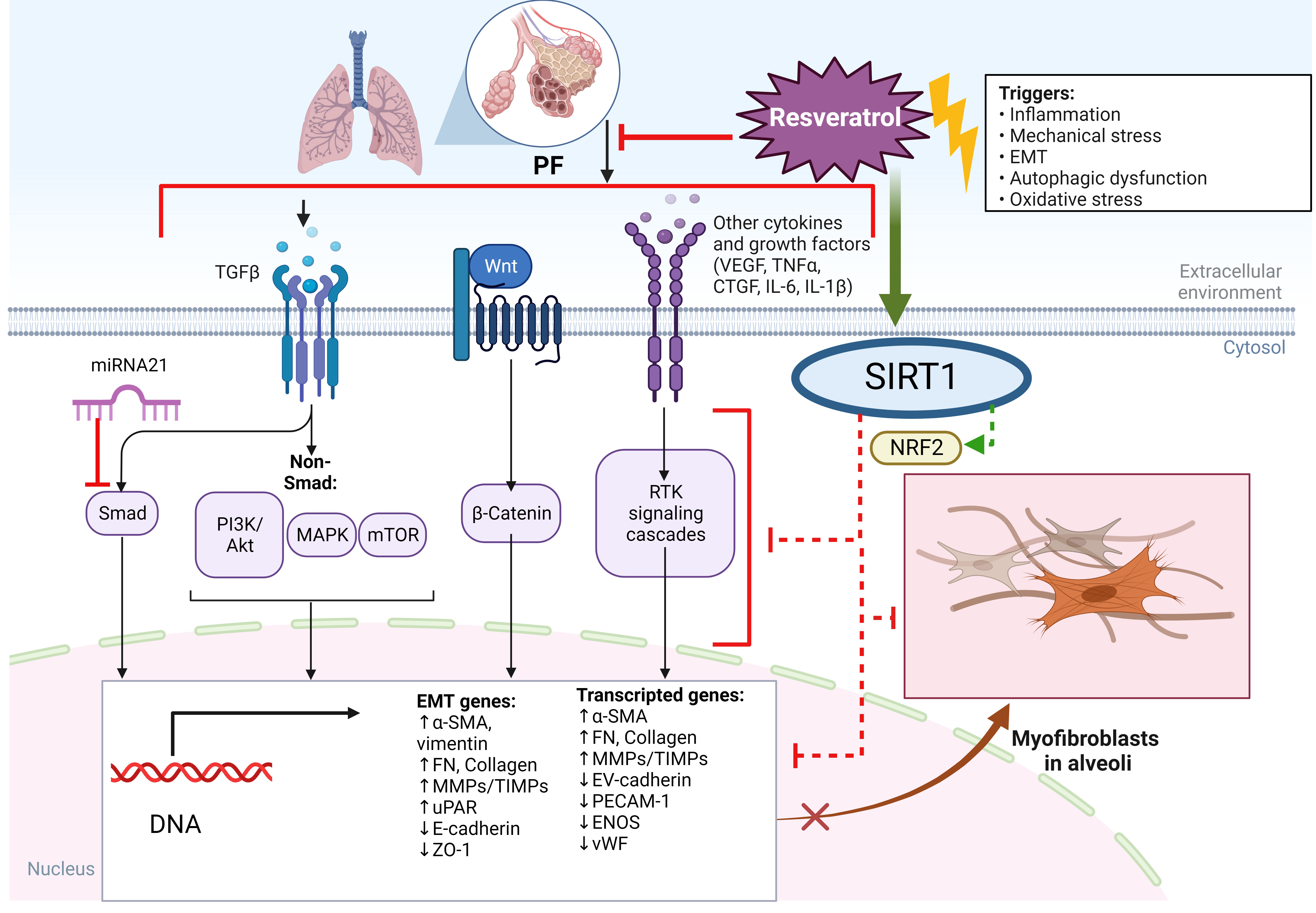 Fig. 4.
Fig. 4.Molecular targets underpinning the antifibrotic effects of resveratrol in lungs. PF, Pulmonary fibrosis; EMT, epithelial mesenchymal transition; Nrf2, the nuclear factor (erythroid-derived 2)-like 2; SIRT1, sirtuin-1; PI3K, phosphoinositide 3-kinase; VEGF, vascular endothelial growth factor; mTOR, mammalian target of rapamycin; IL-6, interleukin-6.
PF is associated with differentiating fibroblasts into myofibroblasts, aberrant ECM deposition, and parenchymal disorganization, all of which, ultimately, affect lung biomechanical parameters [185]. EMT is a process during which epithelial cells lose apical-basal polarity and adhesion and undergo a transition into mesenchymal cells, which potentially leads to PF formation [186, 187].
Resveratrol has shown antifibrotic effects in multiple tissues and organs,
including vessels, kidneys, and livers. As for the lung, resveratrol was found to
alleviate fibrotic-associated pathological changes. Interestingly, by inhibiting
the profibrogenic cytokine TGF-
TGF-
The activation of various Nrf2 downstream signaling targets, such as NAD(P)H quinone dehydrogenase 1 (HO-1/NQO1), NADPH oxidase 4 (NOX4), and GSH, via several Nrf2-induced mechanisms, has been shown to promote potent antifibrotic effects and significantly attenuate PF, both in vivo and in vitro, by counteracting inflammation, oxidative stress, fibroblast–myofibroblast differentiations, and EMT [199]. Consonantly with these findings, resveratrol was found to exert a protective effect on PF by reducing systemic oxidative and nitrosative stress through the activation of AMPK- and Nrf2-associated antioxidant defense mechanisms [200, 201].
The potential therapeutic and preventive effects of resveratrol are believed to both decelerate ILD and indirectly inhibit the progression of autoimmune-related ILD via the same molecular mechanisms and pathways [202].
RA-ILD is one of the leading causes of interstitial fibrosis and pulmonary failure and occurs from a complication of RA. RA is a common autoimmune disease that causes progressive articular damage, functional loss, and comorbidities; thus, presenting ILD as one of its most common extra-articular manifestations [203]. Autophagy is a self-digestion process that occurs in eukaryotic cells and mediates several intracellular biological functions, such as the removal of cytoplasmic material [204]. Autophagy plays a pivotal role in maintaining lung tissue metabolic homeostasis as well as in the occurrence and progression of chronic lung diseases owing to its role in the regulation of several respiratory tract biological functions, including inflammatory response, DNA damage repair, cell apoptosis, and cell proliferation and differentiation [205, 206]. Under specific circumstances, autophagy may occur in autophagosomes, which when combined with lysosomes leads to the formation of autophagolysosomes. Moreover, autophagolysosome-associated degradation of some junction proteins, such as P62, leads to the disruption of homeostasis-associated cellular signals and metabolic pathways that are involved in several metabolic disorders [207]. In this regard, evidence supporting the connection between autophagy and ILD, further, demonstrated that resveratrol can improve RA progression through autophagy regulation. Thus, resveratrol can potentially attenuate PF by ameliorating the autophagic flux and modulating the autophagy–lysosome pathways [28]. Among the signal transduction pathways implicated in RA onset and progression, IL-6 predominantly activates the Janus kinase/signal transducers and activators of the transcription (JAK/STAT) pathway [208]. JAK/STAT has been found to be involved in PF pathogenesis [209], and its downregulation is believed to prevent the development of RA [208].
The receptor activator of nuclear factor kappa-
The activation of SIRT1 and its downstream pathways has been highlighted as a
common molecular pathway that underpins the resveratrol therapeutic effects on RA
and ILD. Overall, the activation of SIRT1 and its downstream pathways can
downregulate the NF-
The Nrf2– Kelch-like ECH-associated protein 1 (Keap1) pathway is recognized as
playing a key role in resveratrol-mediated antioxidant activity [217]. Under
physiological conditions, Nrf2 is found sequestered in the cytoplasm in its
inactive form, which is bound to Keap1 [218, 219, 220]. In response to appropriate
stimuli, the conformation of Keap1 is modified, which releases Nrf2 to complex
with the small Maf (sMaf) protein. The Nrf2–sMaf protein complex, then,
translocates into the nucleus and binds to DNA-contained antioxidant response
elements (AREs), which promotes the expression of heme oxygenase-1 (HO-1), and
exerts antioxidant protective effects [221, 222, 223]. Resveratrol was found to promote
Nrf2 and HO-1 expression, while downregulating the expression of Keap1 which
reduced ROS and malonaldehyde (MDA) generation, and inhibited NF-
Systemic sclerosis (SSc), also referred to as scleroderma, is a rare autoimmune
disease that is associated with vasculopathy and characterized by microvascular
damage, innate and adaptive immune dysfunction, and fibrosis of the skin and/or
internal organs [77, 78, 79, 80]. ILD is a common complication and the leading cause of
death in SSc patients and is often linked to pulmonary hypertension (PH) [226].
Although there is a paucity of studies investigating the effect of resveratrol on
SSc-associated PF, recently, it has been reported that resveratrol could
counteract SSc-related PF by suppressing proinflammatory and profibrotic
processes, including cell proliferation and fibroblast differentiation, via a
SIRT1-mediated inhibition of the TGF-
ILD is considered the most frequent and severe pulmonary complication in primary
Sjögren’s syndrome (SS), with significant morbidity and mortality. Although
ILD was described as a late manifestation of SS, recent studies have demonstrated
that between 10 and 51% of patients might develop ILD years before the onset of
SS [229]. Resveratrol was shown to improve SS by enhancing SIRT1 activity and the
expression of IL-10. Thus, the therapeutic effect of resveratrol on SS-associated
salivary dysfunction appears to be mediated by a SIRT1-like activity, which is
known to block NF-
The development of pulmonary hypertension (PH) as a secondary ailment to PF (PF-PH) is the primary factor in the mortality and morbidity of the disease [231]. The high prevalence of PH in PF patients is very worrisome since it presents a significant predictor of mortality [231]. Until recently, PH was thought to be solely provoked by fibrosis-mediated lung parenchyma destruction, which leads to hypoxic vasoconstriction, severe pulmonary vascular remodeling, loss of vascular bed density, and elevation in pulmonary pressure [232]. The increase in pulmonary pressure is maintained, at least in part, by sustained inflammation, oxidative stress, and dysfunction in endothelial cells (ECs) proliferation and angiogenesis as well as ECs- and vascular smooth muscle cells (VSMCs)-mediated ECM remodeling [77, 78, 79, 80]. This aberrant vascular remodeling occurs in all types of vessels within the pulmonary vascular tree, with ECs and VSMCs the most prominently involved vascular cells [77, 78, 79, 80]. However, it has been recently demonstrated that there is no significant correlation between fibrosis severity and the development of PH [232]. Indeed, relevant histological and molecular differences between PF and PF-PH patients have recently been identified and have helped to deviate from this paradigm [233, 234, 235, 236]. Interestingly, the systemic and cardiac vasculature therapeutic properties of resveratrol may also target pulmonary hypertensive disease mediators, including the antifibrotic pathways [237]. In this regard, resveratrol was shown to protect against pulmonary artery hypertension (PAH), via chemoprotective [95], anti-inflammatory, antioxidant [238], antiproliferative [239], and antiapoptotic mechanisms [240]. Specifically, resveratrol alleviates hyperoxia-induced histological lung injury, regulates redox unbalance, decreases proinflammatory cytokines, and downregulates fibrosis-associated protein expression [241].
The Wnt/beta-catenin signaling pathway not only mediates cell differentiation in
the early stages of lung development, yet also participates in the organization
of the lung tissue structure. In fact, Wnt signaling expression was shown to be
impaired in the lungs of IPF patients [242, 243]. Accordingly, recent studies
demonstrated that the inhibition of glycogen synthase kinase (GSK)-3
SLE is an autoimmune disease, which is characterized by the production of
autoantibodies and presents a complex pathogenesis that is triggered by both
environmental factors and genetic predisposition [247, 248]. The early stages of
SLE are associated with the impaired clearance of apoptotic cells by macrophages,
which leads to an altered autoimmune response, characterized by the
hyperactivation of T cells, a shift from Th1 to Th2, increased IL-10 levels,
activation of B cells, and the excessive generation of autoantibodies [249, 250].
This altered response can cause damage to the peripheral tissue blood vessels,
vasculopathy, and vasculitis, which are common aspects in SLE patients that lead
to cardiovascular and renal damage [251, 252, 253]. In this context, resveratrol was
shown to attenuate proteinuria, decrease IgM/IgG kidney deposition, and reduce
kidney histological lesions [254]. Authors also reported that resveratrol
alleviated the altered autoimmune response, T cells/B cells activation, the
proliferation profile [254], Th1/Th2 ratio, and Th1 cytokine-promoted
immunoglobulins release by acting in a SIRT1-mediated fashion [254]. Conversely,
resveratrol enhanced the apoptosis of T cells, either by acting through Fas,
BCL-2, Bax, or p53-mediated pathways, or through a mechanism involving the
depolarization of mitochondrial membranes and caspase activation [29, 255, 256, 257].
Ultimately, both methods alleviated the impaired apoptosis, which characterizes
the SLE autoimmune response. Additionally, the activation of SIRT1 by resveratrol
was found to inhibit COX2 expression by suppressing both NF-kB activation and
TNF-
PF may co-occur in other pathologies than the previously mentioned autoimmune diseases. Indeed, an important number of PF and lung cancer patients were infected with COVID-19 during the virus pandemic. As featured diseases of PF, they are known to cause high morbidity and mortality rates worldwide [259]. Therefore, a significant body of data has been generated by investigating the effects of resveratrol on lung cancer and COVID-19 infections by targeting PF as a component.
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a recently
emerged viral pathogen that causes coronavirus disease-2019 (COVID-19) [260].
SARS-CoV-2 belongs to a family of coronaviruses, which are enveloped and
segmented RNA viruses [261]. The rapid rate of transmission and infectivity of
this viral disease resides in the fact that the SARS-CoV-2 protein is cleaved by
many serine proteases [23]. Interestingly, IPF patients and patients with severe
post-COVID-19 infection-PF manifest similar pathological characteristics [23].
Since AEC2 and alveolar macrophages are essential regulators in PF promotion,
their altered physiology due to COVID-19 infection may cause PF as a sequela of
the disease [23]. SARS-CoV-2 potentially infects epithelial cells, AEC2, alveolar
macrophages, intestinal enterocytes, and eventually the basal epithelial cells in
the nasal passages [23, 261, 262]. COVID-19 patients have increased levels of
IFN-
PF patients manifest chronic progressive and diffuse fibrotic lung disease. This
persistent inflammatory state, along with the repeated fibrotic scars, has been
linked to the etiologies of various neoplasms, including lung cancer [83, 113].
In this regard, studies demonstrated an increased estimated risk of lung cancer
in ILD patients of between 7 and 14 times [284]. Interestingly, lung tumors in
IPF patients develop preferentially in the peripheral adjacent zones to fibrotic
areas and present different histologic distributions and immunohistochemical
characteristics compared to non-IPF-associated lung tumors [24, 25]. Furthermore,
studies have indicated that IPF and lung cancer share many pathogenic features,
including genetic and epigenetic mechanisms [24, 25]. Indeed, genetic and
epigenetic impairments lead to the abnormal activation of common transduction
pathways, such as Wnt/
These alterations are also believed to be age-related and may result in
deregulated gene expression, increased oxidative stress, and the accumulation of
dysfunctional organelles, ultimately resulting in the development of lung
diseases, including cancer. Resveratrol has been reported to positively impact
lifespan regulations, health maintenance, and age-associated disorders [285].
Additionally, resveratrol administration appears to be a potential method for
slowing the aging-related decline in lung function and structure by maintaining
the integrity of AEC2 [195]. In lung cancer cells, resveratrol acts as an
inhibitor of the epithelial growth factor receptor EGFR [286, 287], mTOR [286, 288], and Akt [289, 290, 291]. Moreover, in addition to the GLUT1 glucose transporter,
resveratrol has been reported to inhibit the NF-
Accumulated data have also shown that resveratrol amplifies the effects of lung cancer chemotherapy drugs, by increasing their intracellular concentrations and triggering apoptosis, autophagy, and senescence [287]. Additionally, resveratrol can interact with other chemotherapeutic drugs to induce and/or potentiate their antitumor effects and, in some cases, abolish chemotherapeutic drug resistance [295, 296].
Given the diverse resveratrol effects, this compound was subjected to increasing multidisciplinary studies aiming to highlight its therapeutic prospect in ILD and CTD-ILD.
Research on the potential therapeutic effects of resveratrol in ILDs is ongoing, and although the results are still preliminary, there is evidence that suggests it may have beneficial effects. In animal studies, resveratrol has been shown to reduce the key features in ILDs, such as lung inflammation and fibrosis. Additionally, some studies have found that resveratrol can improve lung function in patients with ILDs.
Despite the compelling data highlighting the therapeutic potential of
resveratrol, in silico studies remain limited. A resveratrol-targeted
genes interaction network analysis established using both STITCH and the Kyoto
Encyclopedia of Genes and Genomes (KEGG) database has shown that 79 genes were
included in the pathways associated with the therapeutic effects of resveratrol
in SSc [227]. The study focused on the SIRT1/mTOR signaling pathway as one of the
crucial regulatory components, previously suggested to alleviate inflammation and
fibrosis [227]. Another study on Polygonum cuspidatum, a
resveratrol-contained plant, found that it exhibited excellent inhibitory
activity of TGF-
| Model | Type of cell/rodent | Dose | Major results | Reference |
| In vitro | BEAS-2B, WI38-VA13, and Raw264.7 | 30 mg/kg; 10 µM | Activation of AMPK and Nrf2-associated antioxidant defense mechanisms | [196, 197] |
| C57BL/6 female mice | 50 µM | SIRT1 activation, |
[30] | |
| A549 cells | 10 µM | Activation of Nrf2 and |
[99] | |
| HPAEpiCs | 50 μM | Activation of JNK1/2 /p38 MAPK, via the c-Src/PDGFR pathway, |
[197] | |
| U937 cells | 10–50 µM | Inhibition of PI3K and VEGF functions | [298, 299] | |
| HUVECs | 10 µM | SIRT1-mediated mTOR degradation | [300] | |
| MCF-7 cells | 50 µM | [301] | ||
| In vivo | Mice | 15–60 mg/kg | Activation of AMPK and Nrf2-associated antioxidant defense mechanisms | [196, 197] |
| Mice | [102] | |||
| C57BL/6 mice | 20 mg/kg | [302] | ||
| Wistar albinos | 10 mg/kg | [303] | ||
| Rats | 10 mg/kg | [304] | ||
| BALB/c mice | 25–50 mg/kg | [305] | ||
| Wistar male rats | 10 mg/kg/day | [306] | ||
| Wistar male rats | 50 mg/kg | SIRT1/PGC-1 |
[307] | |
| ICR male mice | 0.3 mg/kg | [100] | ||
| Sprague-Dawley rats | 60 mg/kg | [309] | ||
| Inflammatory responses and EMT inhibition | ||||
| Bleomycin-induced lung fibrosis in mice | [102] | |||
| Sprague-Dawley rats | 40 mg/kg | [308] | ||
| Bleomycin-treated mice | [310] | |||
| Bleomycin-induced PF in mice | 50 µM | [29] | ||
| C57BL/6J mice | 20 mg/kg | [111] | ||
| ICR mice | 5 mg/kg | [295] | ||
| Wistar male rats | 10 mg/kg | Inhibition of JAK/STAT/RANKL signaling pathways | [207] |
AMPK,
AMP-activated protein kinase; JNK1/2/p38
MAPK, c-Jun N-terminal protein kinase/1/2/protein38–mitogen-activated protein
kinase; a c-Src/PDGFR, proto-oncogenes tyrosine kinases/platelet-derived growth
factor receptor; VCAM-1, vascular cell adhesion molecule 1; H
Extensive data have confirmed the role of oxidative stress in the onset and
development of PF [78, 79, 80, 81, 82, 103, 311]. In particular, several studies concluded
that the SIRT1-mediated protective effect against oxidative stress and
inflammation by resveratrol played an important role in regulating several
pathways implicated in PF-associated pathologies [293]. For instance, a dose of
50 µM resveratrol activated SIRT1 and protected the lungs against
bleomycin-induced inflammation and fibrosis [30]. In A549 cells, which are used
as a model of human AEC2, treatment with resveratrol (10 µM) protected
against cigarette smoke-induced oxidative stress by activating Nrf2 and inducing
glutathione synthesis [281]. Studies conducted on the human lung epithelial cell
line (HPAEpiCs), reported that resveratrol (50 µM) could attenuate
Staphylococcus aureus-induced inflammation [298] by (a)
activating both JNK1/2 and p38 MAPK via the c-Src/PDGFR pathway, (b) upregulating
VCAM-1 expression, and (c) attenuating H
PF is characterized by continued alveolar epithelial injury and unregulated
repair, which occur through several different mechanisms, including fibroblast
accumulation and differentiation and excessive ECM deposition [303]. Bleomycin is
an antitumor drug that can induce PF during clinical treatment and, for this
reason, has been used as means to artificially induce PF in animal models [304].
Here, resveratrol was shown to counteract weight loss and reduce the mortality
rate in a bleomycin-induced mouse model of lung fibrosis [192]. Another fibrosis
hallmark is the differentiation of fibroblasts into myofibroblasts [305]. In this
context, resveratrol was shown to decrease fibroblast differentiation and ECM
deposition, thereby alleviating lung fibrosis in a mouse model of
bleomycin-induced lung fibrosis [306]. In comparison to the control group,
resveratrol (14 days treatment at 10 mg/kg) significantly decreased lung collagen
deposition, proinflammatory cytokines secretion (TNF-
In an LPS-induced PF mouse model, treatment with resveratrol (0.3 mg/kg) managed
to downregulate TGF-
Additionally, an increase in the atherosclerotic hallmark vascular cell adhesion molecule 1 (VCAM-1) protein and mRNA expression was observed in mice intratracheally administered with a heat-killed Staphylococcus aureus (HKSA) preparation. Resveratrol administration (5 mg/kg) managed to reduce the HKSA-induced increase in VCAM-1 protein levels and mRNA expression. Notably, both the VCAM-1-neutralizing antibody and resveratrol treatment were able to reduce the HKSA-induced leucocyte infiltration in the bronchoalveolar lavage (BAL) fluid and pulmonary hematoma formation, in a similar way [299]. It was also observed that resveratrol (10 mg/kg) inhibited the JAK/STAT/RANKL signaling pathway and ameliorated RA-ILD symptoms in a rat model [211]. Moreover, further studies have also demonstrated that resveratrol can improve RA progression by regulating autophagy, a process, as previously explained, which is believed to be connected to ILD (Table 2) [28].
Resveratrol is a well-known nutraceutical compound that possesses a wide range of pharmacological properties. Several clinical trials have previously highlighted this molecule as a promising therapeutic drug candidate in the treatment of many diseases [318]. In addition to its protective effects on the respiratory system, resveratrol also possesses beneficial actions against cardiovascular diseases, platelet aggregation, diabetes, and neurodegenerative diseases [319]. Indeed, resveratrol and its analogs are pharmacologically safe and can be used alongside other drugs to improve their therapeutic efficacy and reduce toxicity [318]. However, its application in the food and pharmaceutical industries is still limited, mainly due to its low bioavailability, water solubility, and chemical stability; moreover, resveratrol is easily degraded and excreted, especially in biological systems [320, 321]. In the liver and the intestine, UDP-glucuronosyltransferase (UGT) rapidly degrades resveratrol to glucuronic acid and sulfate conjugations producing resveratrol-3-O-glucuronide (R3G) as the most abundant metabolite in both animal and human models [322, 323]. This metabolic pathway appears to be the main factor in modulating resveratrol bioavailability [321, 324]. Indeed, glucuronidation was demonstrated to reduce resveratrol permeability and increase its excretion in cells [118, 322, 323], while metabolites resulting from the degradation of resveratrol can become a systemic reservoir of these compounds [325].
In line with this, resveratrol is also known to be photosensitive,
thermosensitive, and pH sensitive, and because of its low hydrosolubility, it
requires emulsifiers or stabilizers to reach a biologically effective dosage
[320, 326]. Another potential issue is whether resveratrol can accumulate in
target organs at suitable bioactive concentrations. Indeed, studies have proven
that circulating levels of plasmatic resveratrol are barely detectable [321, 327], and in most cases, resveratrol is administered in its free form, which has
low solubility and, therefore, is poorly suited for its delivery in biological
systems [328]. In this regard, pulmonary administration could be considered an
appealing alternative to the oral administration of resveratrol since the
pulmonary route may ameliorate the pharmacokinetic parameters of molecules with
extensive metabolism [329]. Several research groups have attempted to ameliorate
resveratrol bioavailability through its complexation with
Based on the data presented in this review, a large body of evidence supports the multifaceted and quite bountiful benefits of resveratrol on PF. Resveratrol can exert its lung antifibrotic action through more classical effects, such as scavenging free radicals, modulating antioxidant enzymes, and inhibiting inflammatory-associated signaling pathways, as well as by modulating closely associated PF molecular targets, including SIRT, Nrf2, and EMT. The data gathered and discussed here indicate that the reported resveratrol benefits are not only disease-preventive but also disease-ameliorative; thus, it would not be surprising if this nutraceutical becomes more adopted into the routine management of PF patients or those at high risks of similar fibrotic diseases. In fact, despite some challenges involving the adverse effects and poor bioavailability of resveratrol, various efforts have been put forward to mitigate these issues, and indeed, much improvement has been achieved during the past decade by employing new resveratrol formulations, which have been specifically adapted for lung-associated pathologies. Nonetheless, future challenges should be directed to performing long-term clinical trials on PF patients using well-determined resveratrol concentrations to possibly reinforce and prove resveratrol’s protective and/or therapeutic role. Indeed, in vitro and in vivo concentration ranges of biologically active resveratrol differ significantly; thus, based on the available data, it is difficult to extrapolate the most beneficial resveratrol dosage with which to avoid potential toxicity in humans [12]. A more critical aspect of that mentioned above is the dose-dependent hormetic behavior of resveratrol, which tightly determines whether the final biological outcome is positive or negative [337, 338, 339]. Moreover, the interaction between resveratrol and the body’s redox state also affects its final effect [340, 341, 342]. Furthermore, to improve the poor bioavailability attached to resveratrol, new lung-oriented formulations with better absorption and pharmacodynamics need to be developed and commercialized. All the aforementioned factors might explain many of the controversial results associated with resveratrol that are found in the literature; therefore, a more comprehensive and systematic investigation is needed to further define the therapeutic potential of resveratrol. In conclusion, we firmly believe resveratrol could be helpful in fighting PF and associated fibrotic conditions; therefore, more pharmacological and clinical studies should be conducted to better determine its safety, dosages, and efficacy.
IR, TC, AMP, RG, HZ, AHE, and GP participated in the manuscript conceptualization, original draft preparation, review, and editing of the final version. GP was responsible for the project administration and funding acquisition. All authors have read and agreed to the published version of the manuscript.
Not applicable.
Not applicable.
This work has been made possible thanks to grants from “Progetto Fondazione di Sardegna-Bando 2022–2023”, “FAR2020-Pintus”, and “DM 737/2021 resources 2021–2022, funded by the European Union - NextGenerationEU”.
The authors declare no conflict of interest. Given his role as Editor, Gianfranco Pintus had no involvement in the peer-review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Md Soriful Islam.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




