1 Department of Pharmacognosy, Faculty of Pharmacy, Gazi University, 06330 Ankara, Turkey
2 Department of Biology, Faculty of Science, Gazi University, 06560 Ankara, Turkey
3 Instituto de Investigación y Postgrado, Facultad de Ciencias de la Salud, Universidad Central de Chile, 8330507 Santiago, Chile
4 Department of Organic Chemistry, Faculty of Pharmacy, University of Santiago de Compostela, 15782 Santiago de Compostela, Spain
Abstract
Background: Aerial parts of Malva nicaeensis All. are preferred in the prevention and treatment of intestinal infections and hemorrhoids in Turkish traditional medicine. This study is planned to evaluate the pharmacological activity of M. nicaeensis extracts on rats with acetic acid-induced colitis. Methods: The plant material was subsequently extracted with n-hexane, ethanol, and water, respectively. All of these extracts were tested for efficacy in the acetic acid-induced rat colitis model. The aqueous and polysaccharide extracts regulated cytokine levels and antioxidant parameters. Furthermore, the aqueous extract in particular regulated myeloperoxidase and caspase-3 levels in this rat model. In addition, the polysaccharide-rich fraction was separated from the aqueous extract. Results: The polysaccharide-rich fraction and aqueous extract regulated cytokine levels and antioxidant parameters. The aqueous extract also positively affected myeloperoxidase and caspase-3 levels. The phytochemical studies revealed that the aqueous extract had the highest phenolic content. In addition, the polysaccharide fraction was found to contain total sugars, sulfated groups, uronic acids, and total proteins in 78.4%, 0.9%, 1.5%, and 14.7%, respectively, and was rich in monosaccharide-type compounds, especially galactose (36.4%). Conclusions: M. nicaeensis was discovered to be a drug lead in the future treatment of irritable bowel diseases or as a complementary therapeutic agent that aided conventional treatments.
Keywords
- colitis
- herbal medicine
- Malva nicaeensis
- Malvaceae
- metabolic disorders
- myeloperoxidase
Idiopathic inflammatory bowel diseases (IBDs) include Crohn’s disease and ulcerative colitis (UC), which show retention in different regions of the gastrointestinal system and affect people differently. UC mostly affects the colonic mucosa and is characterized by acute, non-infectious inflammation [1, 2]. Both genetic and environmental factors are important in the onset of this damage. UC gives rise to some symptoms, including diarrhea with blood and tenesmus. UC is usually seen between the ages of 15 and 30 [3]. Side effects and inadequate treatment outcomes of the present medications used for UC encouraged scientists to examine better alternative treatments with fewer side effects [4].
In recent years, numerous plants and plant constituents have been considered potential drug leads for treating UC [5].
Members of the Malva L. genus (Malvaceae) are mainly distributed in widespread throughout the world and especially in the Mediterranean region. They are usually known as “Mallow (in English) and Ebegümeci (in Turkish)” [6, 7]. Malva sp. has been used in traditional medicines for its therapeutic properties such as mucolytic, antiseptic [8], diuretic, sedative, spasmolytic, and laxative effects [7]. Previous studies which evaluated the effects of Malva sylvestris L. on UC showed that Malva species can be worth researching alternative sources for the medication of UC [4, 5]. In traditional Lebanese medicine, whole parts of Malva nicaeensis All. are used to stop coughing, for wound healing [9], for renal infections, and for kidney stones [10]. The aerial parts of M. nicaeensis are used in Turkish folk medicine as an expectorant for coughs [11]. According to phytochemical analysis on Malva species, monoterpenes, diterpenes, coumarins, flavonoids, malvin, malvone A (a naphthoquinone), malvaline and many polyphenols have been identified [7, 12, 13, 14].
Since there are traditional records of Malva sp. especially M. nicaeensis in infectious diseases, this study was planned to investigate the effectiveness of Malva nicaeensis in an experimental colitis model in rats.
Aerial parts of M. nicaeensis were gathered from Antalya, Turkey, in June 2019. The voucher specimen (Herbarium number: GUEF3836) has been deposited at the Herbarium of the Faculty of Pharmacy of Gazi University, Ankara, Turkey.
The air-dried and powdered aerial parts of M. nicaeensis (300 g) were
extracted with n-hexane, ethanol, and water (3
Aqueous extract of M. nicaeensis was macerated with absolute ethanol, stirred vigorously for 8 hours, and kept overnight at 4 °C. After centrifugation (20 minutes at 4 °C and 16 000 G), the supernatant was removed and the remaining part was freeze-dried. The precipitated polysaccharide-rich fraction was initially applied to an ion exchange column (equilibrated Sephadex A-25, Pharmacia, Uppsala, Sweden), eluted with gradient solutions of 0.02 to 2 M NaCl, and then applied to a size exclusion column (Sepharose CL-6B, GE Healthcare, Uppsala, Sweden). The isolated polysaccharide fraction was lyophilized and kept at –20 °C until usage (Yield: 6.1%) [4].
Lowry and colleagues’ method (1951) [15] was used to estimating the protein content of the polysaccharide fraction. Total sugars, total uronic acids, and sulphated groups were determined by the methods previously published by Dubois et al. (1956) [16], Bitter and Muir (1962) [17], and Lloyd et al. (1961) [18], respectively. Monosaccharide composition was determined after hydrolyzation of the sugars followed by high-performance anion-exchange chromatography. Concentrations of the selected nine monosaccharides (glucose, galactose, fucose, xylose, mannose, rhamnose, galacturonic acid, and glucuronic acid) were calculated by using external calibration with an equimolar mixture of the standards (Sigma Chemicals, St. Louis, MO, USA).
The method previously published by Singleton and Rossi (1965) was used to
measure the phenolic content of the samples [19]. 2 g of dried extracts were
mixed with 50 mL of n-hexane. In order to extract phenolic metabolites, hexane
extract is partitioned with 60% methanol (3
After the permission of the Experimental Animal Ethics Committee of Kobay (Protocol number: 234) male Sprague-Dawley rats (180–200 g) were obtained from Kobay Laboratory (Ankara, Turkey) and kept in the working laboratory conditions for 3 days prior to the study. Rats were fed standard pellet feed and tap water during their adaptation period, housed in a 12-hour light/12-hour dark cycle, and kept at room temperature. Rats were divided into six groups, each with six rats. All procedures were carried out in accordance with international laws protecting the rights of animals used in research and biodiversity.
The extracts were suspended in 0.5% carboxymethyl cellulose (CMC). The control group of rats received only 0.5% CMC, while the reference group received the drug sulfasalazine at 100 mg/kg dose that was prepared with 0.5% CMC [20]. Meanwhile, 100 mg/kg of the extracts were administered to the rats.
Colitis was induced by injecting 2 mL of acetic acid solution (3% v/v) into the rectum through a polyurethane tube for enteral feeding (2 mm in diameter) that was inserted to a depth of 4.5 cm. To prevent solution leakage, the rats were held in the Trendelenburg position throughout rectal instillation and for 1 min following instillation [21].
The control group was assigned as the ulcerative colitis-induced rats that were orally treated with 0.5% CMC, daily for 6 days and intra-rectally injected with acetic acid (2 mL of 3% (v/v) in 0.9% NaCl) on day 4. The extract (n-hexane, ethanol, aqueous extract, polysaccharide fraction) groups were assigned as the ulcerative colitis-induced rats that were orally treated with extracts, daily for 6 days and intra-rectally injected with acetic acid (2 mL of 3% (v/v) in 0.9% NaCl) on day 4. The reference group represented the ulcerative colitis-induced rats that were orally treated with sulfasalazine as a reference drug (100 mg/kg/day) for 6 days and then injected with acetic acid (2 mL of 3% (v/v) in 0.9% NaCl) intra-rectally on day 4 [21].
On day 6, all groups received samples one last time, and rats were sacrificed on day 7. The entire colon was removed, opened longitudinally, and the fecal content was cleared with normal saline. The evaluation of biochemical parameters such as caspase-3, and myeloperoxidase (MPO) was conducted in the proximal part of the colon (6–7 cm). This part of the colon was stored in a physiological buffer at pH 7.4 until the homogenization of the samples. Further, a small part of the proximal colon was removed and kept in 10% formalin for histopathological analysis as well. The homogenization of tissue was performed in a cold environment at a concentration of 10% (w/v) in 11.5 g/L solutions of KCl, and the homogenized samples were centrifuged for 15 minutes at 10.000 rpm and 4 °C. The supernatant part was taken and separated for the biochemical investigations [21].
The MPO activity of colon tissue was estimated according to the previously published method by Kupeli Akkol et al. (2019) [21]. A total of 0.1 mL of the supernatant was mixed with 2.9 mL of 50 mM phosphate buffer (pH 6) containing 0.167 mg/mL O-dianisidine dihydrochloride and 0.0005% hydrogen peroxide. The change in absorbance at 460 nm was measured using a UV/VIS spectrophotometer (Beckman, Fullerton, CA, USA). Myeloperoxidase activity was reported as units (U) per gram (g) weight of wet colon tissue. The myeloperoxidase hyperactivity was calculated according to the following formula:
MPO activity (mU/mg) = 1000
where X = 10
Griess reagent (100 µL) was added to the 100 µL of serum and colonic tissue homogenates. Sulfanilamide was used as a standard, and the calculations were performed at 540 nm. The concentrations were determined using a standard curve of sodium nitrate and the results were expressed as ng/µg of wet tissue [21].
Enzyme-linked immunosorbent assay kits (Elabscience, Maryland, USA) were used for the
measurement of TNF-
Caspase-3 activity is assessed according to the method of Jonges et al.
(2001) [22]. The tissues were suspended in 1 mL of ice-cold PBS and washed twice.
The resulting pellet was resuspended in a lysis buffer consisting of 10 mM HEPES,
pH 7.0, 40 mM
Rat colonic tissues were removed and deposited in a 10% formaldehyde solution. Tissue processing was carried out using Thermo Scientific Excelsior (London, UK). Histocentre 2 was used to make paraffin blocks from the processed tissues. The marine glass was utilized to cut 3.5-m slices using the Leica RM2255 microtome (Texas, USA). All tissue samples were examined under a light microscope after being stained with hematoxylin-eosin (HE) in the Shandon Varistan apparatus (Ohio, USA) [21].
Statistical analyses and comparisons were performed using GraphPad Prism
(version 6.0) software (DMCA Compliance Agent, Boston, MA, USA). p values of
Normal colon homeostasis requires the equilibrium between pro-inflammatory and
anti-inflammatory cytokines in the colon mucosa. Cytokine profile disruption
causes some negative conditions, such as inflammatory bowel diseases. Further, an
increment of pro-inflammatory cytokines such as TNF-
| Groups | Serum | Colonic tissue | ||
| TNF- |
IL-6 (pg/mL) | TNF- |
IL-6 (pg/mg) | |
| Control | 16.7 |
49.3 |
307.5 |
218.9 |
| n-Hexane extract | 20.1 |
47.6 |
298.4 |
210.6 |
| Ethanol extract | 14.6 |
40.1 |
267.5 |
181.8 |
| Aqueous extract | 7.3 |
19.6 |
114.8 |
146.1 |
| Polysaccharide fraction | 8.0 |
22.7 |
136.9 |
153.2 |
| Sulfasalazine | 5.8 |
16.3 |
87.4 |
100.8 |
*: p
Additionally, oxidative stress can be one of the initial factors in UC development. Changes in the levels of oxidative parameters such as Glutathione (GSH), Myeloperoxidase (MPO) , and MDA have been seen during the inflammatory response [25]. In the current study, the control group exerted an important increase in lipid peroxides due to oxidative stress. Furthermore, the aqueous extract and polysaccharide fraction significantly increased serum nitrite levels from 0.5 to 1.9 and 1.1 µg, respectively. On the contrary, the aqueous extract and polysaccharide fraction decreased nitrite levels in colonic tissue from 2.5 to 1.0 and 1.6 ng/µg, respectively. Additionally, MDA levels were significantly diminished in both serum and colonic tissues (Table 2).
| Groups | Serum | Colonic tissue | ||
| Nitrite (µg) | MDA (nmol/mL) | Nitrite (ng/µg) | MDA (nmol/g) | |
| Control | 0.5 |
10.8 |
2.5 |
385.1 |
| n-Hexane extract | 0.3 |
11.6 |
2.7 |
403.3 |
| Ethanol extract | 0.7 |
9.7 |
2.3 |
344.2 |
| Aqueous extract | 1.9 |
4.1 |
1.0 |
196.4 |
| Polysaccharide fraction | 1.1 |
4.9 |
1.6 |
217.6 |
| Sulfasalazine | 2.1 |
2.4 |
0.4 |
147.9 |
*: p
Under various pathological conditions or by the mechanism initiated by apoptotic tissue damage, apoptosis is a process that involves the normal turnover of damaged cells. Caspases are classified as inactive procaspases that are present in the majority of cells. When activated, they can cause the activation of other procaspases, which is what leads to the breakdown of intracellular proteins and programmed rapid cell death of the cells [26]. Similar to starter caspases, executioner caspases, such as caspase-3, is in charge of causing cell death during apoptosis. In the current investigation, a significant increase in colonic caspase-3 activity was seen in the rats in the control group, but a significant decrease in the mean value of caspase-3 in the colon of the experimental group was discovered (Table 3).
| Groups | Caspase-3 level | MPO concentration |
| (pmolAMC/min/mg/protein) | (U/mg of tissue) | |
| Control | 46.2 |
25.53 |
| n-Hexane extract | 48.4 |
20.33 |
| Ethanol extract | 43.6 |
19.26 |
| Aqueous extract | 27.9 |
8.47 |
| Polysaccharide fraction | 34.2 |
10.96 |
| Sulfasalazine | 11.6 |
4.94 |
*: p
Acute inflammation is characterized by an increase in neutrophil MPO activity that is observed after the infiltration of polymorphonuclear leukocytes. One of the most crucial components of an organism’s immune system is MPO [27]. In the current study, an increment in MPO activity was detected in the inflamed colon, while a decrease in MPO levels was detected in the aqueous extract and polysaccharide fraction along with the sulfasalazine-treated group animals when compared to the control group. Thus, it was established that the rise in MPO levels and acetic acid-induced colitis were closely connected. All of these dangerous biochemical characteristics created by acetic acid treatment were reversed by treatment with aqueous extract and polysaccharide fraction (Table 3).
The colitis model triggered with acetic acid is one of the most utilized methods for evaluating the efficacy of drug leads against IBD. After administering acetic acid intrarectally, massive necrosis diffuses inflammatory leukocyte infiltration and ulcerated mucosa. Linear ulcers, inflammation, localized hyperemia, significant loss of mucosal structure, cellular infiltration, cryptic abscess, and goblet cell decline are observed in the colon tissues treated by acetic acid in the intrarectal route [28]. In the control group, there were several mucosal erosions, including the invasion of certain inflammatory cells in the lamina propria, villus flattening, and some damage to the crypts and cylindric epithelium in the mucosa in the duodenum and jejunum and it was determined that mucosal inflammation developed on the duodenum (duodenitis) and jejunum (jejunitis) (Fig. 1A). The n-hexane extract-treated group of animals exerted histopathological modifications, such as new villus formation for intestinal absorption, little collagen fiber regeneration in connective tissue, and tight regular lamina propria (Fig. 1B). The polysaccharide fraction treated group showed histopathological changes, like villus formation, and some lipid vacuole formation in connective tissue (Fig. 1E). Finally, the ethanol and aqueous extract treated groups also had some histopathological modifications, such as regeneration in the mucosa and villus degeneration as well as collagen fiber regeneration in the submucosa (Fig. 1C,D). In the positive control group namely the sulfasalazine-treated group, villus and collagen fiber regeneration in mucosa and submucosa were examined, respectively (Fig. 1F).
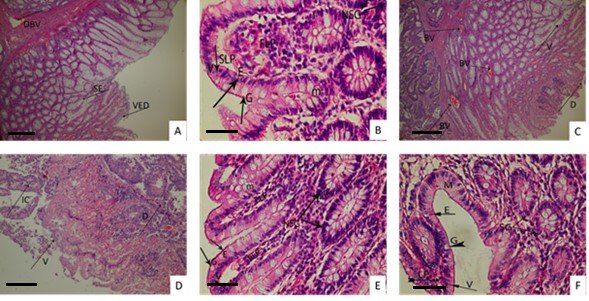 Fig. 1.
Fig. 1.Histopathological views of treated groups. The original magnification was × 100 and the scale bars represent 120 µm for figures Data are representative of 6 animal per group. [(A) Control group. (B) n-Hexane extract. (C) Ethanol extract. (D) Aqueous extract. (E) Polysaccharide fraction. (F) Sulfasalazine]. DBV, Dilated blood vessel in the submucosa; SE, Separation and degeneration of the epithelium in the mucosa; VED, Villus epithelium degeneration; BV, Blood vessels; V, Villus degeneration; D, Crypts degeneration; IC, Inflammatory cells; M, Mucosa layer; SG, Secret glands in the mucosa; G, Goblet cell; E, Normal epithelium cell; LP, Lamina propria; M. Mucosa; NSG, New developing secret glands; SLP, Small gaps in the newly formed in lamina propria structure; FLP, Formation of new lamina propria structure.
In the current study, not only the pharmacological activities of the extracts
and fraction were evaluated, but their phytochemical compositions were also
determined. According to our results, the highest phenolic content was found in
the aqueous extract, which is mainly important in terms of antioxidant activity
during inflammation. The phenolic content value was 1.2
The amounts of total sugars, sulfated groups, uronic acids, and total proteins in the polysaccharide fraction were calculated as 78.4%, 0.9%, 1.5%, and 14.7%, respectively (Table 4).
| Chemical group | % Composition |
| Total sugars | 78.4 |
| Uronic acids | 1.5 |
| Sulfated groups | 0.9 |
| Total proteins | 14.7 |
The findings demonstrated that the polysaccharide fraction is rich in monosaccharides, with galactose (36.4%) being the main monosaccharide and arabinose (0.5%) being the least prevalent (Table 5).
| Monosaccharide | % Composition |
| Galactose | 36.4 |
| Mannose | 8.7 |
| Glucose | 19.7 |
| Arabinose | 0.5 |
| Rhamnose | 4.9 |
| Glucuronic acid | 32.3 |
UC is a multifactorial disease that originates from a dysfunctional epithelial, innate, and adaptive immune response to intestinal microorganisms. Currently, the management of IBD is a challenging process for gastroenterologists; 5-aminosalicylates, immunomodulators, antibiotics, steroids, and surgery have been used to alleviate the symptoms of the disease and its remission. However, long-term use of these conventional treatments is associated with severe toxicities for patients [29]. Therefore, due to those causes, scientists are looking for new and safer treatments for UC.
In the present study, the effects of various extracts obtained from M. nicaeensis were appraised in acetic acid-induced UC in rats. Macroscopically, no defect was seen in the colon of the rats treated with the aqueous extract and polysaccharide fraction of M. nicaeensis. The effects of the tested extracts against UC seem to be related to their polysaccharide and polyphenol content. Polyphenols such as anthocyanins, flavonoids, and tannins are secondary metabolites with different biological activities such as anticarcinogenic, antimicrobial, vasodilatory, wound healing [30, 31, 32], antioxidant, and anti-inflammatory properties [33, 34, 35, 36]. These data were in parallel with the results of previous publications on M. sylvestris polysaccharides [37, 38, 39].
Previous reports showed that the aqueous extract prepared from different parts of M. sylvestris was known to be rich in mucilaginous polysaccharides with rhamnose, trehalose, galacturonic acid, glucuronic acid, galactose, glucose, fructose, and sucrose [38, 39, 40, 41]. The polysaccharides of M. sylvestris were stated to have anti-complementary activity, which may be one of the possible mechanisms for the experimental results. Furthermore, in the study conducted by Classen and Blaschek, high amounts of arabinogalactans were extracted from cultures of M. sylvestris [42]. According to the study, these polysaccharides could be the main effective compounds in the aqueous fraction of the plant. These polysaccharides were found to have preventive effects against acid-induced UC. Liu et al. [43] exhibited that the application of the polysaccharides significantly decreased ulcers, mortality, and colon mass. Wong et al. [44] reported that Angelica sinensis (Oliv.) Diels and Rheum tanguticum Maxim. ex Balf. polysaccharides could be due to the prevention of the pathological process of UC. Polysaccharides from Vaccinium oxycoccos reduced neutrophil infiltration and were also reported to have significant antioxidant properties [45]. Polysaccharides from M. sylvestris were reported to have potential antioxidant activity [46].
Magro et al. [47] reported that the aqueous extract of M. sylvestris leaves displayed a remarkable antimicrobial effect against fungi, and it was reported that M. sylvestris leaves inhibited prostaglandin synthesis by the cyclooxygenase pathway. Saldanha et al. [29] reported that some polyphenols, including resveratrol, quercetin, kaempferol, ellagic acid, rutin, green tea polyphenols, and silymarin showed remarkable effects against UC in addition to the polysaccharides.
In the literature, there is no previous report on the effects of M. nicaeensis polysaccharides. The previous studies reported that the polysaccharides of V. oxycoccos, R. tanguticum, and A. sinensis showed remarkable activity against UC. Similarly, these results are parallel with our results [43, 44, 45]. The aqueous extract of M. nicaeensis displayed higher efficacy against UC than the polysaccharide fraction, suggesting that other constituents in the aqueous fraction showed synergistic effects due to the polysaccharides. Magro et al. [47] demonstrated that the aqueous extract of M. sylvestris was the only extract with significant antimicrobial effects against fungi, which was reported to inhibit prostaglandin synthesis via the cyclooxygenase pathway. The aqueous extract prepared from the leaves of M. sylvestris was reported to contain various terpenoids such as blumenol A, dehydrovomifoliol, linalool-1-oic acid, linalool, malvone A, and different megastigmene derivatives [13, 48]. There are also some glycosides, including ascorbic acid, fructose, glucose, and trehalose, along with several glycosidic phenolic compounds [33, 49]. Additionally, Geerling et al. [50] exhibited that the consumption of vitamin C, fructose, magnesium, and fruit reduced the development of UC. Moreover, these kinds of metabolites were reported for significant anti-inflammatory and antioxidant potentials, which play a principal role in the observed efficacy of this fraction. As far as we know, this is the first published report about the beneficial effects of M. sylvestris in the treatment or prophylaxis of UC, though Sleiman and Daher previously reported on the anti-ulcerogenic and anti-inflammatory properties of the aqueous extract of M. sylvestris against gastric ulcers when taken orally. Through the inhibition of elastase and trypsin by the aqueous extract of M. sylvestris, enhanced mucous production in mucosal tissue and improved elastin promoter activity improved the elasticity of mucosal tissues [36].
Other than phenolic and terpenic metabolites, previous studies also demonstrated that the n-hexane extract of M. sylvestris was reported to include carotenoids, tocopherols, and various fatty acids, with a large amount (50% to 80%) of polyunsaturated fatty acids. Although carotenoids and tocopherols have the potential for antioxidant properties and could be responsible for the effects of those fractions. In the studies of Geerling et al. [50], it was demonstrated that consuming a lot of mono- and polyunsaturated fatty acids may increase the risk of developing UC, and this finding may help to explain the lower beneficial outcomes (compared to aqueous and alcoholic fractions) observed in the present study.
In conclusion, this study suggests that M. nicaeensis might be an encouraging therapeutic option for UC through its antioxidant and anti-inflammatory properties. Findings revealed that M. nicaeensis can be used in the cell, tissue, or person-specific treatments that will be developed in the future for the treatment of IBD, or as a complementary therapeutic agent that supports conventional treatments. The activity of M. nicaeensis could be due to its polysaccharides and phenolic contents. Furthermore, this plant can be recommended to be used as a prophylactic supplement for patients with UC to improve their health complaints or reduce the risks of adverse effects from their conventional treatment. Nonetheless, especially clinical studies are required to confirm its effects on humans.
Datasets used and/or analyzed for this study are avail-able from the corresponding author upon appropriate request.
Conceptualization—EKA; Design—EKA; Supervision—EKA; Resources—EKA, HT; Materials—EKA, GT; Data Collection and/or Processing—EKA and ES-S; Analysis and/or Interpretation—EKA, GT, HT and ES-S; Writing—EKA; Critical Reviews—EKA, and ES-S. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work to take public responsibility for appropriate portions of the content and agreed to be accountable for all aspects of the work in ensuring that questions related to its accuracy or integrity.
All experiments on Sprague-Dawley rats were performed with permission of the Experimental Animal Ethics Committee of Kobay (Protocol number: 234) (Ankara, Turkey).
Not applicable.
This work was supported by the Scientific Research Project of Gazi University [Grant Number: 05/2018-11].
The authors declare no conflict of interest. EKA served as one of the Guest editors of this journal. We declare that EKA had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Luigi De Masi.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

