1 Faculty of Pharmacy, Hubei University of Chinese Medicine, 430065 Wuhan, Hubei, China
2 Modern Engineering Research Center of Traditional Chinese Medicine and Ethnic Medicine of Hubei Province, 430065 Wuhan, Hubei, China
3 Xinjiang Uygur Pharmaceutical Co., Ltd, 830026 Wulumuqi, Xinjiang, China
†These authors contributed equally.
Abstract
Background: Hugan Buzure Granule (HBG) is a traditional prescription of
Uygur nationality in China mainly used to treat liver cold, stomachache, spleen
and rib pain, arthralgia, rheumatism and urinary system diseases. Its mechanism
of action in treating acute kidney injury (AKI) continues to remain unconfirmed.
This study’s objective was to investigate the pharmacodynamics and mechanism of
HBG in the management of AKI. Methods: The damage to the kidney tissue
was examined by using H&E (Hematoxylin-eosin) staining. The BUN (Blood Urea
Nitrogen) and Cr (Creatinine) in serum were examined by biochemical kit. The
content of ROS (Reactive oxygen species) in kidney tissue was determined by ROS
frozen section staining, while the amount of MDA (Malondialdehyde), GSH
(Glutathione), and the enzymes of CAT (Catalase) and SOD (Superoxide dismutase)
were assessed by using a biochemical kit. The tissue apoptosis was seen by using
the TUNEL assay. ELISA kit was utilized to assess the content of IL-6,
TNF-
Keywords
- hugan buzure granule
- acute kidney injury
- inflammation
- apoptosis
- oxidative stress
Acute kidney injury (AKI) is a urinary system disease that can be brought on by a number of circumstances. It leads to a rapid deterioration in kidney function as well as a reduction in urine volume or nitrogen metabolism (urea, creatinine). There are approximately 13 million AKI patients worldwide each year. Hospital mortality is 8.8%, and patients with severe AKI have a greater risk of dying there [1]. Studies have shown that some patients with AKI who suffered from chronic inflammation for a long time, transformed to chronic kidney injury [2]. Clinically, the most important treatment of AKI is kidney replacement therapy, namely hemodialysis, which has infection risks in the process of treatment. Clinical use of statins, such as pravastatin alleviates the symptoms of kidney ischemia/reperfusion injury [3]. The body produces too much ROS, leading to oxidative stress, which causes inflammation and apoptosis, so antioxidants can be used to treat AKI. Inflammation is the main pathogenic factor of AKI, so anti-inflammatory drugs also are good at anti-AKI. In recent years, Chinese medicine has achieved remarkable results in the treatment of nephropathy. Cai et al. [4] found that Huangqi Danggui Mixture composed of Huangqi and Danggui has protective effect on AKI induced by ischemia/reperfusion. In the treatment of chronic glomerulonephritis, it has been illustrated that Tripterygium wilfordii has immunosuppressive and anti-inflammatory properties [5]. Kang et al. [6] discovered and summarized that many natural products can prevent and treat Chronic Kidney injury (CKI). Traditional Chinese medicine provides more clear advantages than western medicine in the treatment of nephropathy, including lower costs and fewer complications.
Studies have shown that after intraperitoneal injection of a certain amount of
cisplatin, the renal function, nitrogen metabolism and urine volume of
experimental animals continue to decline, renal tubules and glomeruli are
damaged, which is consistent with the pathological manifestations of AKI, and is
a good AKI model [7, 8]. Oxidative stress, which is intimately associated with the
occurrence and progression of AKI, is brought on by an excessive production of
ROS or for other factors. Overexpression of inflammatory factors is an important
manifestation of AKI. These inflammatory factors mainly include IL-6,
IL-1
Hugan Buzure Granule (HBG) is derived from traditional Uyghur medicine classic prescription Hugan Buzure, it is clinically used to treat liver cold, stomachache, spleen and rib pain, arthralgia, rheumatism and urinary system diseases. The Uyghur Medicine Department of the Ministry of Health of the People’s Republic of China includes its prescription formulation and indications in its drug standard (China Pharmacopoeia Commission, 1998). HBG consists of seven herbal medicines: (1) Fennel (Foeniculi Fructus, the fruit of Foeniculum vuLgare Mill.) 106 g, (2) Fennel root bark (Cortex Foeniculi, the root bark of Foeniculum vuLgare Mill.) 212 g, (3) Cuscuta chinensis (Cuscutae semen, the seed of Cuscuta chinensis Lam.) 53 g, (4) Chicory (the seed of Cichorium intybus L.) 212 g, (5) Chicory root (the root of Cichorium intybus L.) 106 g, (6) Celery seed (Fructus Apii, the seed of Apium graveolens L.) 106 g, (7) Celery root (the root of Apium graveolens L.) 212 g. These seven herbal remedies are listed in the Chinese Pharmacopoeia (China Pharmacopoeia Commission, 2015) and the Encyclopedia of Chinese Medicine Uygur Medicine. According to the literature review, the herbs in HBG have therapeutic effects on diseases of the urinary system. For example, Wu Song and other studies have shown that Cuscuta chinensis ethanol extract has protective effect on mice with AKI induced by lipopolysaccharide [12]. Fennel has some certain curative effects on nephritis and renal deficiency [13]. Chicory is mainly used for damp heat hepatitis, nephritis, urinary incontinence and systemic edema. However, there are few references on the therapeutic effects of HBG in AKI, which is our innovation. We want to put it on a solid scientific foundation so that it can be used in clinical practice. The objectives of this study are to demonstrate that HBG can ameliorate cisplatin-induced AKI in mice and to investigate its potential pharmacodynamics and mechanisms.
Animals and drugs: 72 SPF balb/c male mice, aged at 4–6 weeks, weighed at 18–20 g (Experimental Animal Center of China Three Gorges University, Yichang, China). HBG (Cat# 200568, Xinjiang Uygur Pharmaceutical Co., Ltd., Wulumuqi, China). Cisplatin (Cat# N0624A, Dalian Meilun Biotechnology Co., Ltd., Dalian, China). SKP (Cat# JP17095, Tianjin Tongrentang Group Co., Ltd., Tianjin, China). The standards of quercetin (Cat# 19082203), kaempferol (Cat# 20082105), quercitrin (Cat# 18082102), apigenin (Cat# PS011063) and chlorogenic acid (Cat# 516E023) were acquired from Shanghai Yuanye Biotechnology Co., Ltd (Shanghai, China).
Kits and instruments: Cr (Cat# C011-2-1) and BUN (Cat# C013-2-1) kit (Nanjing
Jiancheng Pharmaceuticals, Nanjing, China). MDA (Cat# A003-1-2), GSH (Cat#
A005-1-2), SOD (Cat# A001-3-2) and CAT (Cat# A007-1-1) kits (Nanjing Jiancheng
Pharmaceuticals, Nanjing, China). Mouse TNF-
Accurately weighing 1.6 g of HBG was dissolved in 80% methanol, extracted by ultrasonic wave (power: 350 W, frequency: 53 kHz) for 40 min, and fixed the volume to 10 mL. The organic microporous membrane was used for filtration, and the filtrate was the test solution.
The HPLC of Agilent 1260 was used, Eclipse XDB-C
72 Balb/c mice were caged in a climate-controlled room under specified SPF conditions. Day and night were alternated for 12 h. This research project was approved by the Ethics Committee of Hubei University of Traditional Chinese Medicine (NO. HUCMS202006011). The mice were randomly divided into six groups before the experiment. The control group received oral saline for eleven consecutive days, and a single intraperitoneal (i.p.) injection of saline was given on the seventh day. The cisplatin group received oral saline for eleven consecutive days, and a single i.p. injection of cisplatin (13 mg/kg) was also given on the seventh day. HBG groups: For eleven days straight, HBG (1.6, 3.2, or 6.4 g/kg/day) was given orally once, and on the seventh day, cisplatin was applied intraperitoneally just once. SKP group: For eleven days straight, SKP (1.2 g/kg/day) was administered orally. On the seventh day, cisplatin was delivered intraperitoneally once. The final day, all mice were gavaged, sedated with 0.3% sodium pentobarbital (80 mg/kg body weight), and then put to death. All subsequent test samples were 10. The remaining kidneys were stored in reserve at –80 °C while a portion of the kidneys were removed and fixed in 4% paraformaldehyde.
The weight of the mice in each group was recorded daily and all behavioral traits were scrutinized. The organ coefficient was computed after the liver and kidney of mice were weighed.
The blood sample was centrifuged. Upper serum was taken out and kept at –80 °C. A commercial kit was utilized to detect the contents of serum Cr and BUN in accordance with the manufacturer’s instructions.
The kidneys were taken out of 4% PFA, encased in paraffin, dehydrated with ethanol, and removed with xylene. H&E staining was applied to paraffin sections (5 µm) before being inspected under a light microscope (NIKON ECLIPSE CI, Tokyo, Japan).
The frozen sections of kidney were incubated in dark with ROS staining solution. The nuclei were stained with DAPI. A quenching agent for anti-fluorescence was employed to seal the film. Through using fluorescent microscope, the images were inspected and recorded.
The kidney and ice-cold normal saline were homogenized and centrifuged (1:10, w/v). Using commercial kits in accordance with the directions, the resulting supernatant was utilized to assess the concentrations of MDA, reduced GSH, SOD, and CAT in conjunction to their contents and activities.
The kidney tissue’s apoptosis was observed by using the TUNEL assay. After already being treated with proteinase K, kidney slices were cultured in a solution of nucleotides that contained both fluorescein dUTP and TDT. DAPI was applied to stain the nuclei. A quenching agent for anti-fluorescence was added to seal the film. Through using fluorescent microscope, the images were inspected and recorded.
TNF-
After rehydrating and eliminating the paraffin from the kidney slices, endogenous peroxidase was blocked before being coated with BSA. A specific amount of the primary antibody made with PBS was dripped on the sections, which were then incubated overnight at 4 °C. PBS was utilized to rinse the sections. The sections were then covered with a second antibody from the relevant species, which was HRP-labeled, and they were allowed to legally sit at room temperature for 50 minutes. PBS was employed to rinse the sections. To parts, DAB chromogenic solution was used to produce color. Harris hematoxylin was dyed for about 3 min and used tap water to cleanse for 10 min. After being dried and sealed with neutral gum, the pieces were sanitized with ethanol and xylene. Scrutiny under a microscope, image gathering, and evaluation.
When creating a protein sample from kidney tissue, 5% 2-ME activation buffer
and 5% loading buffer were added. The protein samples were placed onto an
electrophoresis gel made of 10% SDS-polyacrylamide. The proteins were then
transferred at 300 mA for 60 min to polyvinylidene difluoride membranes. The
membranes were submerged in 5% skim milk (Sigma) for two hours. The membrane was
in interaction with the diluted primary IL-18, IL-1
One-way ANOVA and post hoc examination were utilized to contrast the results to
the cisplatin group. Estimated to be a noteworthy value was p
As shown in Fig. 1, HPLC of HBG and reference standard (327 nm), 1 is Chlorogenic acid, 2 is Quercetin, 3 is Kaempferol, 4 is Apigenin, 5 is Quercitrin.
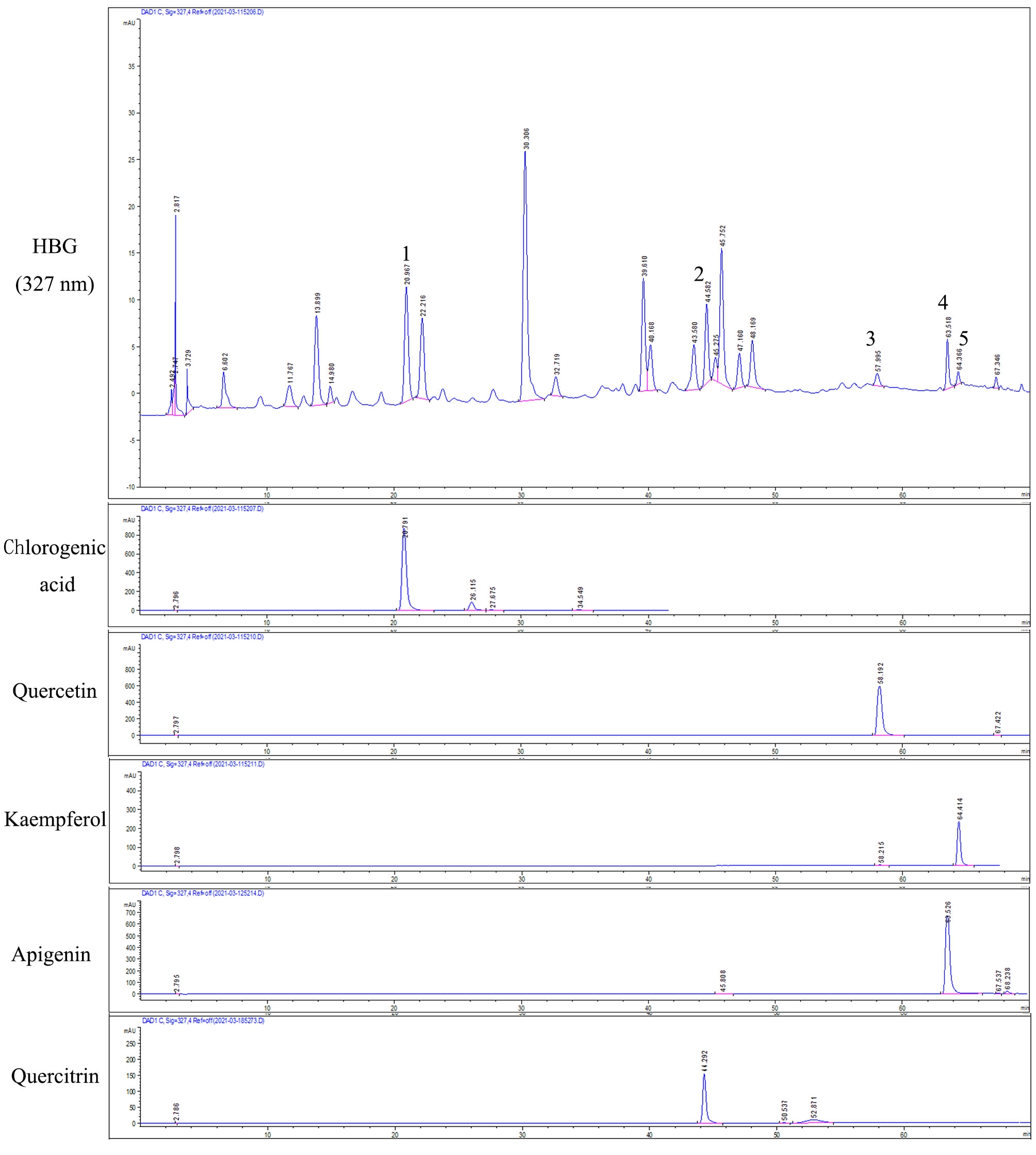 Fig. 1.
Fig. 1.High Performance Liquid Chromatography (HPLC) of Hugan Buzure Granule (HBG) and reference standard (327 nm). 1 is Chlorogenic acid, 2 is Quercetin, 3 is Kaempferol, 4 is Apigenin, 5 is Quercitrin.
The mice of Cisplatin group were dispirited, their hair color was dim and
lusterless, their weight dropped sharply, their drinking water and eating
decreased, and some mice had loose stools and their stomachs were swollen (Fig. 2A). The mental state of the mice in the HBG groups was noticeably better than
that in the Cisplatin groups, their hair was smoother and their diet and drinking
water were normal compared to the Cisplatin groups, and the phenomena of loose
stools was diminished. The recovery of mice in Cisplatin + 6.4 g/kg HBG group was
the fastest, and that in SKP group was the same as that in Cisplatin + 3.2 g/kg
HBG group. The kidneys in the Control group were slender and long, and they were
a dark red color. The kidneys in the Cisplatin group, in contrast hand, were
plainly enlarged, and they were a lighter red color than the kidneys in the
Control group (Fig. 2B). In HBG group, the enlargement of kidney was alleviated
and the color gradually became dark. In contrast to the Control group (Fig. 2C),
the Cisplatin group’s kidney coefficient increased considerably (p
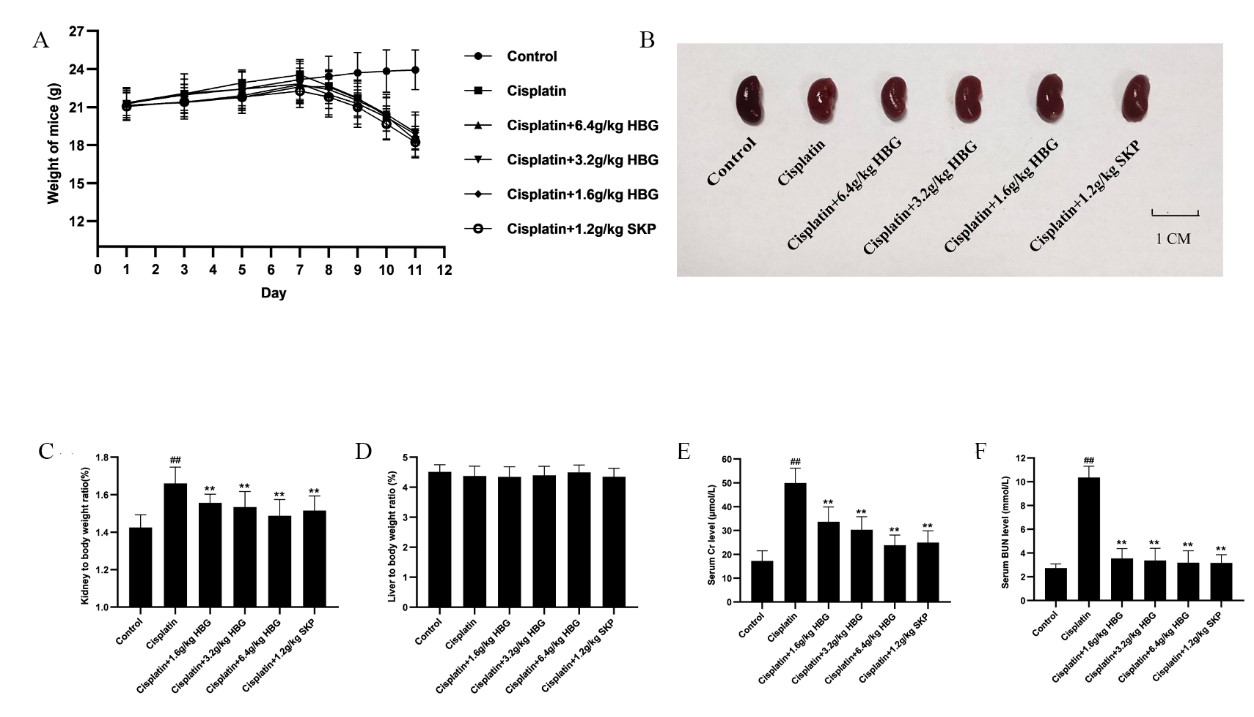 Fig. 2.
Fig. 2.The effect of HBG on organ coefficient and serum Creatinine (Cr) and Blood Urea Nitrogen (BUN) in
Chronic Kidney Injury (CKI) mice. (A) The weight of mice changed with time. (B) Appearance of kidney in
AKI mice. (C) Kidney coefficient. (D) Liver coefficient. (E) Serum Cr. (F) Serum
BUN. Data were expressed as the mean
As shown in Fig. 2E,F, the serum Cr and BUN were considerably higher in
Cisplatin group (p
The morphology of glomeruli and renal tubules was healthy, the structure of kidney tissue cells was ordinary, the arrangement of renal tubular cells was ordered and tight, and the boundary between cells was visible (Fig. 3A). The structure of kidney tissue in the Cisplatin group was obviously destroyed, many large vacuoles could be seen, the renal tubules were seriously dilated, and the arrangement of renal tubular cells was scattered. In comparison to Cisplatin group, the histological characteristics of the kidney injury in HBG groups were significantly improved, the number of vacuoles decreased, the expansion of renal tubules decreased, and the arrangement of renal tubular cells tended to be orderly. The Cisplatin + 6.4 g/kg HBG group showed more significant injury reduction. The recovery of SKP group was similar to that of Cisplatin + 3.2 g/kg HBG group.
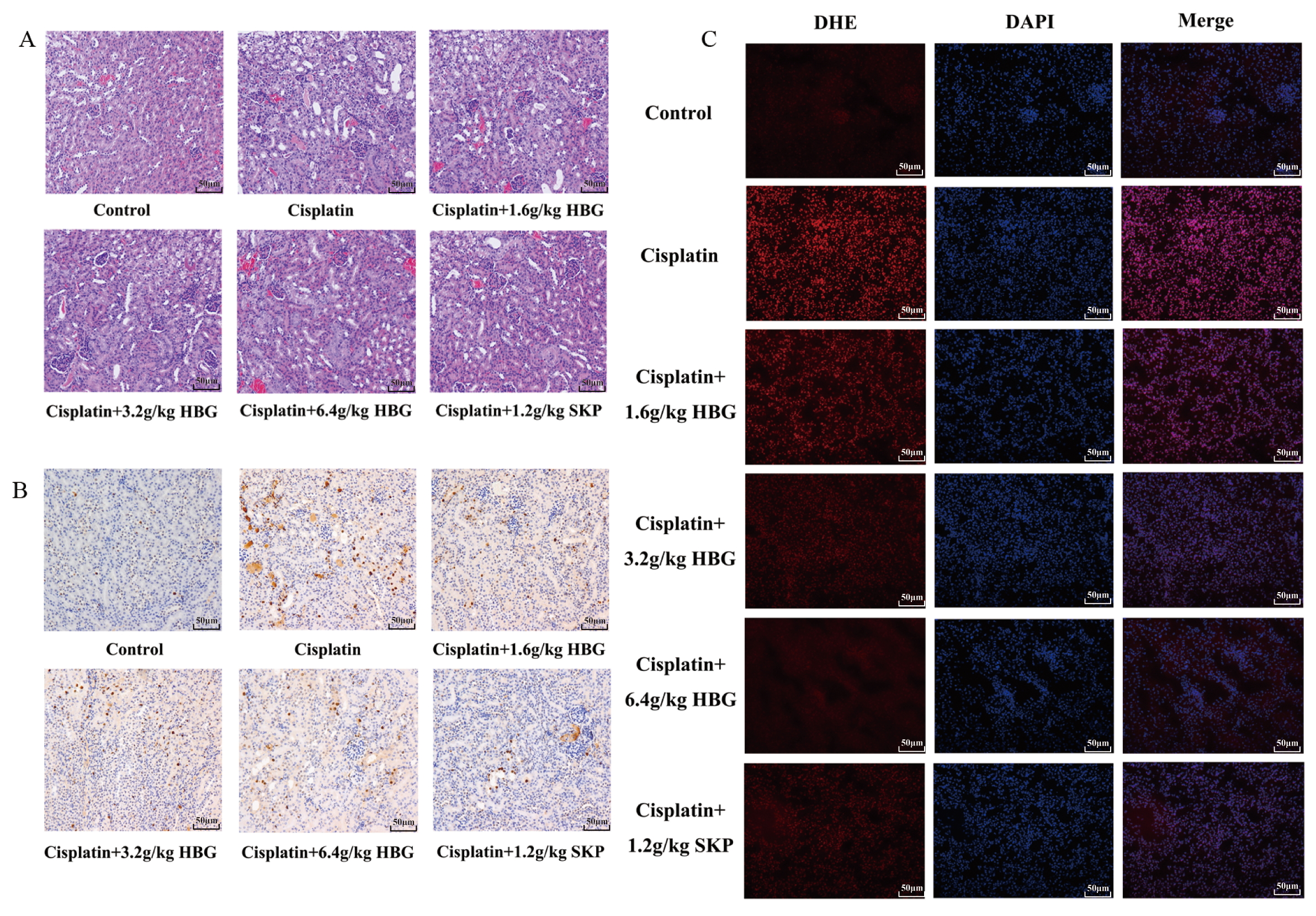 Fig. 3.
Fig. 3.HBG influences the morphological change of kidney tissues in CKI
mice. (A) H&E staining section of kidney tissues (
As shown in Fig. 3B, brown was positive expression, indicating apoptosis. The positive expression of Cisplatin group was considerably increased. The positive expression of HBG groups was noticeably reduced, while the positive expression of SKP groups was even more noticeably reduced.
In contrast to the Control group, as seen in Fig. 3C, the fluorescence intensity of the Cisplatin group was much greater, indicating that the amount of ROS was dramatically higher. Compared with Cisplatin group, the fluorescence intensity was noticeably reduced in HBG groups, and the most obvious decrease was found in Cisplatin + 6.4 g/kg HBG group, the amount of ROS in SKP group was as similar to that in cisplatin + 3.2 g/kg HBG group.
As seen in Fig. 4A–D, the Cisplatin group’s MDA content increased distinctly
(p
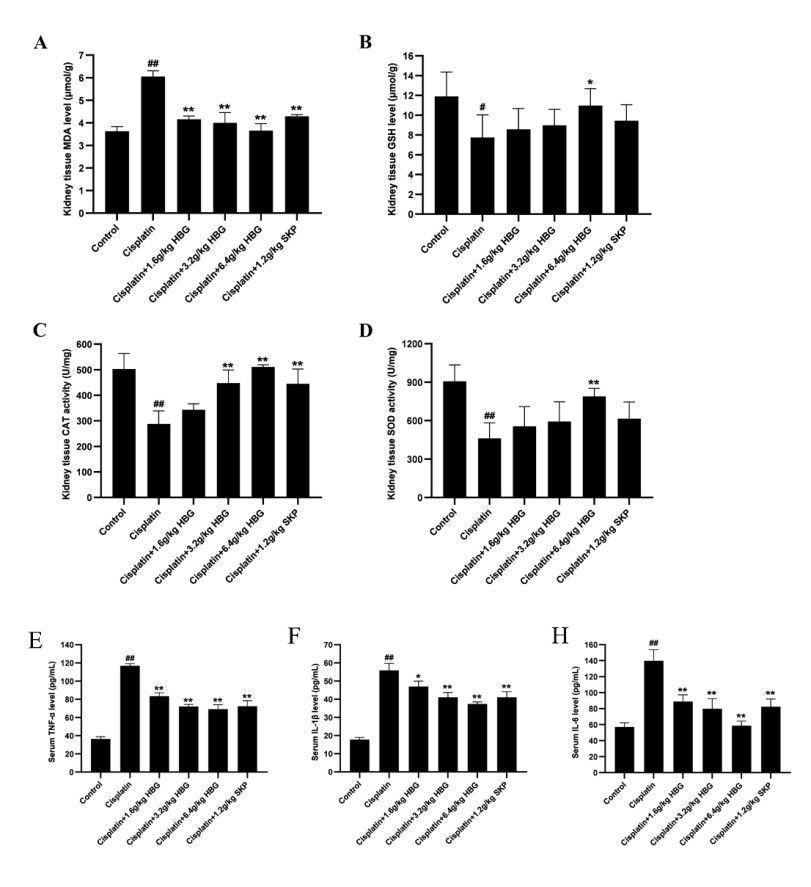 Fig. 4.
Fig. 4.The level of oxidative stress associated to indicators in AKI
mice’ kidney tissue. (A) is the MDA content, (B) is the GSH level, (C) is the
activity of CAT, (D) is the content of SOD, (E) is the serum TNF-
As depicted in Fig. 4E–H, TNF-
NLRP3 and Caspase-1 immunohistochemical staining of kidney tissues
(
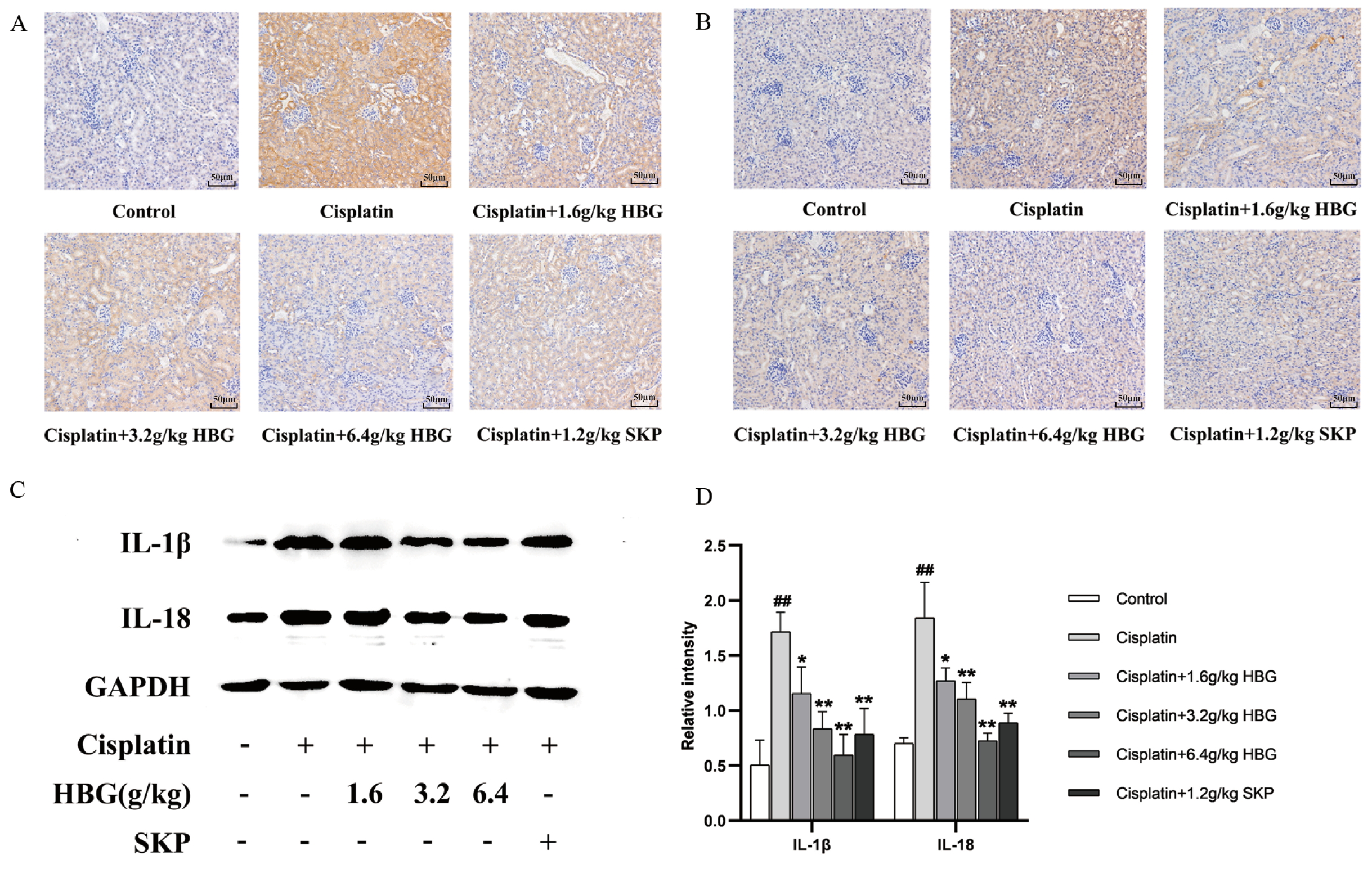 Fig. 5.
Fig. 5.Expression of key proteins in NLRP3/Caspase-1 pathway. (A)
NLRP3 immunohistochemical staining of kidney tissues (
Results of IL-1
The positive translation of TLR4 and NF-
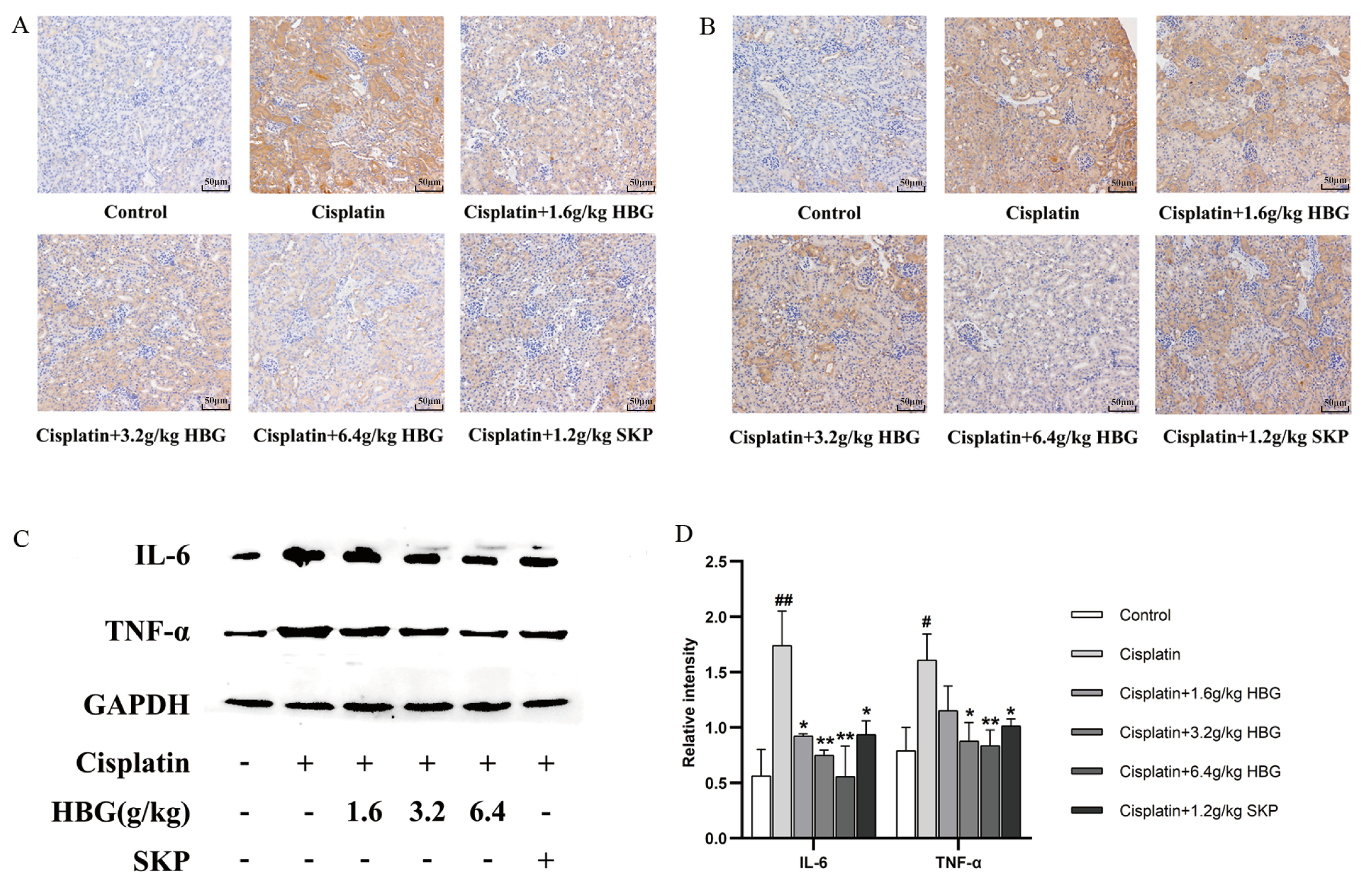 Fig. 6.
Fig. 6.Expression of key proteins in TLR4/NF-
Results of Western blot for IL-6 and TNF-
Through the analysis of HPLC, the results showed that HBG contained quercetin,
kaempferol, chlorogenic acid and so on. The effectiveness of quercetin in
treating AKI has been demonstrated [14, 15], and inhibits TLR4/NF-
Cisplatin is a commonly used anti-tumor drug, however, 20–30% of patients have AKI after receiving cisplatin treatment [19]. In order to help study the complex mechanism of the development of kidney injury, the molecular mechanism of cisplatin metabolism and cisplatin nephrotoxicity have been created and extensively studied by using the animal model of cisplatin-induced AKI [20, 21], likewise the potential medications for treating AKI. In this work, a mouse AKI model was created with once intraperitoneal dose of cisplatin (13 mg/kg). The cisplatin group were depressed, their hair color was dim and lusterless, their weight dropped sharply, and some mice had loose stools. HBG could improve the bad behavior of mice with AKI. Meanwhile, HBG group significantly reduced the kidney coefficient of mice, and improved the appearance of the kidney, indicating that HBG improved the swelling phenomenon of the kidney in mice with AKI. The H&E staining showed that HBG could improve the vacuoles, tubule dilation and scattered arrangement of renal tubular cells in mice with AKI. The serum’s Cr and BUN content was an important indicator of kidney function [22]. Mice in the HBG and SKP groups had vastly lower blood levels of Cr and BUN. It showed that HBG reduced the kidney function damage of mice with AKI.
Apoptosis of tissue cells, a rise in ROS (causing oxidative stress), and an
amplification of inflammatory markers were frequently in relation to the course
of AKI, aggravating kidney damage [23]. The apoptosis in the kidney of HBG groups
was significantly decreased, indicating that HBG inhibited the apoptosis of
kidney tissue cells in mice with AKI. Compared with Cisplatin group, the amount
of ROS in HBG groups were lower, which indicated that HBG could reduce the
content of ROS in kidney tissue. MDA is a biomarker of lipid peroxidation in
living things, and it measured the level of lipid peroxidation in living
creatures as well as the degree of cell damage in an indirect manner [24, 25]. The
biological defense system’s primary enzyme, CAT, scavenges hydrogen peroxide in
the body to shield cells from its toxicity [26]. All cells that metabolize
oxygen had SOD, which shielded them against excessive superoxide. Two superoxide
anions were converted into oxygen and hydrogen peroxide by the action of SOD
[27]. The findings revealed that the HBG group’s MDA content substantially
dropped, while GSH content increased and CAT and SOD activity significantly
enhanced. These results indicated that HBG reduced the oxidative stress in mice
with AKI by reducing the amount of ROS and raising the antioxidant enzymes’ rate
of activity. The content of TNF-
In the pathogenesis of AKI, the increase of ROS induced oxidative stress and
activated the NLRP3/Caspase-1 pathway which was related to cell death. When AKI
occured, the expression of TLR4 increases, and its further activation of
NF-
This study established a model of cisplatin-induced AKI and treated it with HBG.
It was confirmed that HBG had good anti-AKI pharmacodynamics. It was clarified
that HBG could alleviate cisplatin induced AKI in mice by anti-oxidant stress,
inhibiting the apoptosis of tissue cells, reducing the content of inflammatory
factors in serum. The mechanism involved the inhibition of NLRP3/Caspase-1
pathway and TLR4/NF-
HBG, Hugan Buzure Granule; AKI, Acute kidney injury; HPLC, High Performance
Liquid Chromatography; SKP, Shenyan Kangfu Pian; H&E, Hematoxylin-eosin; Cr,
Creatinine; ROS, Reactive oxygen species; MDA, Malondialdehyde; GSH, Glutathione;
CAT, Catalase; SOD, Superoxide dismutase; IL-6, Interleukin-6; TNF-
For reasonable requirements, the data related to this study can be requested from the corresponding author.
HZW and QY designed the research study. CWR and BY performed the research. HLY and YFY provided help and advice on the ELISA experiments. CWR analyzed the data. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This research project was approved by the Ethics Committee , Hubei University of Traditional Chinese Medicine, Wuhan, China (NO. HUCMS202006011).
Not applicable.
This work was supported by National Key Research and Development Program of China plan key project of TCM Modernization Research (2017YFC1701000).
The authors declare no conflict of interest. This research was supported by Xinjiang Uygur Pharmaceutical Co., Ltd in which I had a financial interest. I have fully disclosed these interests to Qiang Yin and have developed an approved plan to manage any potential conflicts that arise from such an arrangement.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






