1 Department of Endocrinology, The Second People's Hospital of Gansu Province, Northwest Minzu University, 730000 Lanzhou, Gansu, China
2 Department of Endocrinology, Second Affiliated Hospital of Harbin Medical University, 150086 Harbin, Heilongjiang, China
Abstract
Background: Lung cancer is a malignant disease with high morbidity and
mortality. Lung cancer and diabetes are closely related, and diabetic patients
with lung tumors are common in clinical practice. Liraglutide, a glucagon-like
peptide-1 receptor (GLP-1R) agonist, is commonly used in the treatment of type 2
diabetes. In this study, we examined the effect of liraglutide on lung cancer and
its potential protective effect on high glucose-induced lung aging.
Methods: Indirect mmunofluorescence was done to assess the expression levels
of p-AKT, ki67, Caspase3, Bax and PI3K. Western blotting was
conducted to determine the expression levels of BAX, BCL2, Caspase9,
E-cadherin, N-cadherin, PI3K, AKT and vimentin. Cell viability,
cell cycle and cell apoptosis were evaluated by colony formation,
CCK-8 assay and flow cytometry. Immunohistochemistry was performed
to evaluate the expression of Nf-
Keywords
- liraglutide
- lung cancer
- proliferation
- aging
- lung cells
In China, lung cancer is a malignant disease with high morbidity and mortality, which is exacerbated by the fact that making an early diagnosis is difficult [1], with about 70% of patients having advanced disease at the time of diagnosis. Lung cancer is mainly divided into two types: non-small cell lung cancer (NSCLC) and small cell lung cancer [1]. About 80% of all lung cancer patients have NSCLC, which is currently the focus of lung cancer prevention and treatment [2]. Radiotherapy and chemotherapy do not significantly improve the survival of patients with advanced lung cancer, and the lung cancer mortality rate is still very high [2]. Although platinum-based single or combined chemotherapy drugs are widely used in the treatment of lung cancer, their side effects include nephrotoxicity, bone marrow suppression, and gastrointestinal reactions [3, 4]. Therefore, the search for new lung cancer drugs is crucial.
Glucagon-like peptide-1 receptor (GLP-1R) agonist is a new type of widely used hypoglycemic drug [5]. There are two types of GLP-1R agonists commonly used in clinical practice. One is a short-acting GLP-1 receptor agonist such as exenatide and another is a long-acting GLP-1R agonist such as liraglutide [6], which is highly stable. Liraglutide is an effective hypoglycemic agent, and its safe and convenient use is increasingly favored by diabetic patients. GLP-1 is an incretin hormone secreted by the gut; the continuous use of GLP-1R agonists can induce the occurrence and development of tumors such as colorectal cancer. Koehler et al. [7] have found that the GLP-1R agonist exenatide can inhibit tumor cell proliferation and induce increased apoptosis in mouse colon cancer cells. It was also shown by Ligumsky et al. [8] to inhibit the proliferation of breast cancer cells, but it was not dependent on the expression of GLP-1R. Zhao et al. [9] found that the GLP-1 receptor agonist liraglutide could inhibit the proliferation of pancreatic cancer cells and promote cancer cell apoptosis. Similarly, Müller et al. [10] found that for human prostate cancer cells with GLP-1R expression, the GLP-1R agonist exenatide could inhibit the proliferation of human prostate cancer cells by L cells in the small intestine.
Under physiological conditions, GLP-1 is rapidly digested and degraded by DPP4
(dipeptidyl peptidase-4) within about 1–2 minutes (half-life), which results in
low plasma concentrations. GLP-1 inhibits gastric emptying, increases anorexia,
reduces body weight, and increases the sensitivity of surrounding tissues to
insulin [10]. The physiological role of GLP-1 is mainly mediated through
activation of GLP-1R. Studies have shown that GLP-1Rs are widely distributed in a
variety of cells and tissues, including islet beta cells, brain, heart, smooth
muscle, as well as others [5]. Binding of GLP-1 to GLP-1R can activate the
protein kinase A signaling pathway as well as the calmodulin pathway and the
mitogen-activated protein kinase and phosphatidylinositol 3 kinase signaling
pathways, which are also involved in Wsnt signaling. Although most clinical
research data show that GLP-1 depends on the expression of GLP-1R for its
physiological role, it has been reported that GLP-1 can induce hepatic glycogen
production when it acts on hepatocytes and skeletal muscle cells, even though
there is no GLP-1R expression on the surface of these cells [11, 12]. Furthermore,
Montrose-Rafizadeh et al. [13] found that GLP-1 can induce the physiological
effects of insulinoma when it acts on 3T3-L1 adipocytes, even in the absence of
GLP-1R expression on these cells. These experimental results show that GLP-1 can
still exert physiological effects when it acts on GLP-1R-negative cells. One
possible reason is the existence of GLP-1 alternate receptors on the surface of
these cells. Therefore, for GLP-1R-negative cells, in the presence of GLP-1
alternate receptor expression, GLP-1 will still have a physiological effect on
these cells. Previous studies have found that in patients with diabetes, the
incidence of tumors is higher, indicating that the two are related.
Perfetti et al. [14] showed that GLP-1R agonists can activate the Wnt
signaling pathway in islet
In the current study, we evaluated the effect of liraglutide on lung cancer. First, we found that liraglutide could inhibit lung cancer cell proliferation and induce lung cancer cell apoptosis. This has important significance for the treatment of patients with diabetes and lung cancer. On this basis, we further evaluated the effect of liraglutide on high glucose-induced lung aging and oxidative damage and found that liraglutide could protect the lung from aging and endoplasmic reticulum stress (ERS).
Liraglutide was from Novo Nordisk A/S (China). The Annexin V-FITC apoptosis
detection kit and RIPA lysate were purchased from Beyotime Biotechnology Co., Ltd
(Shanghai, China). The BCA protein concentration assay kit was purchased from
Shanghai Biyuntian Biotechnology Co., Ltd (Shanghai, China). Lung normal
epithelial cells (BEAS-2B, CRL-9609) were purchased from ATCC. DMEM high glucose
was purchased from Gibco (California USA). PVDF membranes were purchased from
Bio-Rad (Shanghai, China). The SDS-PAGE gel preparation kit was purchased from
China Salorbio Biotechnology Co., Ltd (Beijing, China). The hypersensitive ECL
chemiluminescence kit was purchased from Salorbio Biotechnology Co., Ltd
(Beijing, China). The penicillin-streptomycin solution (100
A549 (SCSP-503), H1299 (Catalogue number TCHu160) and BEAS-2B were purchased
from National Collection of Autheticated cell cultures (Shanghai, China). H1299
and A549 cells were cultured in DMEM+10% FBS and placed in a 37 °C, 5% CO
Lung cancer cells were collected after liraglutide treatment. Proteins in cell samples were measured using the BCA protein kit (Beyotime). The protein samples were separated using SDS-PAGE and transferred to PVDF membranes. PVDF membranes were incubated in 5% skimmed milk solution for 1 h. Membranes were washed 3 times with TBST (5 min each time) and then incubated with diluted primary antibodies at 4 °C overnight. The membrane was then washed 3 times with TBST (5 min each time) and incubated with secondary antibodies for 2 h at room temperature. After washing three times with TBST (10 min each time), ECL was added to detect the immunoprotein bands.
Cellular apoptosis was detected using an apoptosis detection kit (BD Bioscience, Bedford, MA, USA) according to the manufacturer’s instructions. Briefly, the cells were digested with trypsin, then washed twice with PBS, and subsequently mixed in the binding buffer. Then 5 µL Annexin and 5 µL propidium iodide were added to the cell suspension and incubated at room temperature for 15 min (in the dark). Cell samples were analyzed using flow cytometry.
Cells were seeded in 96-well plates at a density of 6000 cells per well. After 24 h, cells were treated with different concentrations of liraglutide for 72 h. Cell viability was determined using the MTT kit (Abcam, Shanghai, China) according to the manufacturer’s instructions.
5
Cells in logarithmic growth were trypsinized and harvested using centrifugation.
Cells were then resuspended and added to 96-well plates (2
Lung cancer cells were inoculated into 6-well culture plates, and control plates and drug-added plates were established. Then liraglutide was added (150 nmol/L), and the 6-well plates were placed in a 37 °C cell incubator for 14 days, while the culture medium was changed regularly. The culture was terminated when cell clones could be observed, and then the number of cell clones were counted from images taken after crystal violet staining.
For the long-term continuous exposure to glucose, BEAS-2B cells were cultured in
6-well plates and stimulated with 5 mM glucose (normal glucose) or 25 mM glucose
(high glucose). All BEAS-2B cells were propagated and maintained in the same
treatment conditions. During cell passage, the cells were collected for
senescence-associated
We detected the expression of lung tissue-related proteins using
immunohistochemistry. Mouse lung tissues were cut into 0.5 cm
The isolated tissues were fixed into 4% formalin for 48 h, and then the tissues were immersed in alcohol after dehydration through a graded ethanol series. After paraffin embedding and dewaxing, H&E staining was performed.
SA-
Tissue sections were blocked with 5% BSA for 30 min. The blocking solution was discarded, the primary antibody was then added and placed in a wet box overnight at 4 °C. Sections were washed three times (5 min each), secondary antibodies were added to cover the tissue and incubated for 60 min at room temperature. After washing, DAPI solution was added and incubated for 5 min at room temperature. Sections were sealed with resin. Then laser confocal microscopy was performed to observe samples using an Olympus FV-3000 confocal laser-scanning microscope (DAPI: excitation wavelength: 330–380 nm, emission wavelength 420 nm, blue; Alexa Fluor 488: excitation wavelength 495 nm, emission wavelength 519 nm, green; DyLight 594: excitation wavelength 593 nm, emission wavelength 618 nm, red).
After liraglutide treatment of the cells, 1
Statistical analysis was performed on the experimental data using SPSS21.0
software (IBM Corp., Chicago, IL, USA). The data were represented by the mean
A CCK-8 kit was used to assess the effect of liraglutide on the proliferation of
human lung tumor cells (H1299 and A549). Liraglutide, at increasing
concentrations (10 nmol/L, 50 nmol/L, 100 nmol/L, 200 nmol/L, 500 nmol/L, and
1000 nmol/L) inhibited the proliferation of human lung cancer cells (A549 and
H1299) (Fig. 1A). The IC50s of liraglutide were 146.4 nmol/L for A549 cells and
122.9 nmol/L for H1299 cells. Given these results, we selected 150 nmol/L of
liraglutide for subsequent experiments. We also assessed the inhibitory effect of
liraglutide on the proliferation of lung cancer cells at different time points
(24 h, 48 h, 72 h, and 96 h). The results showed that compared with the controls,
liraglutide was able to inhibit the proliferation of lung cancer cells at
different time points (p
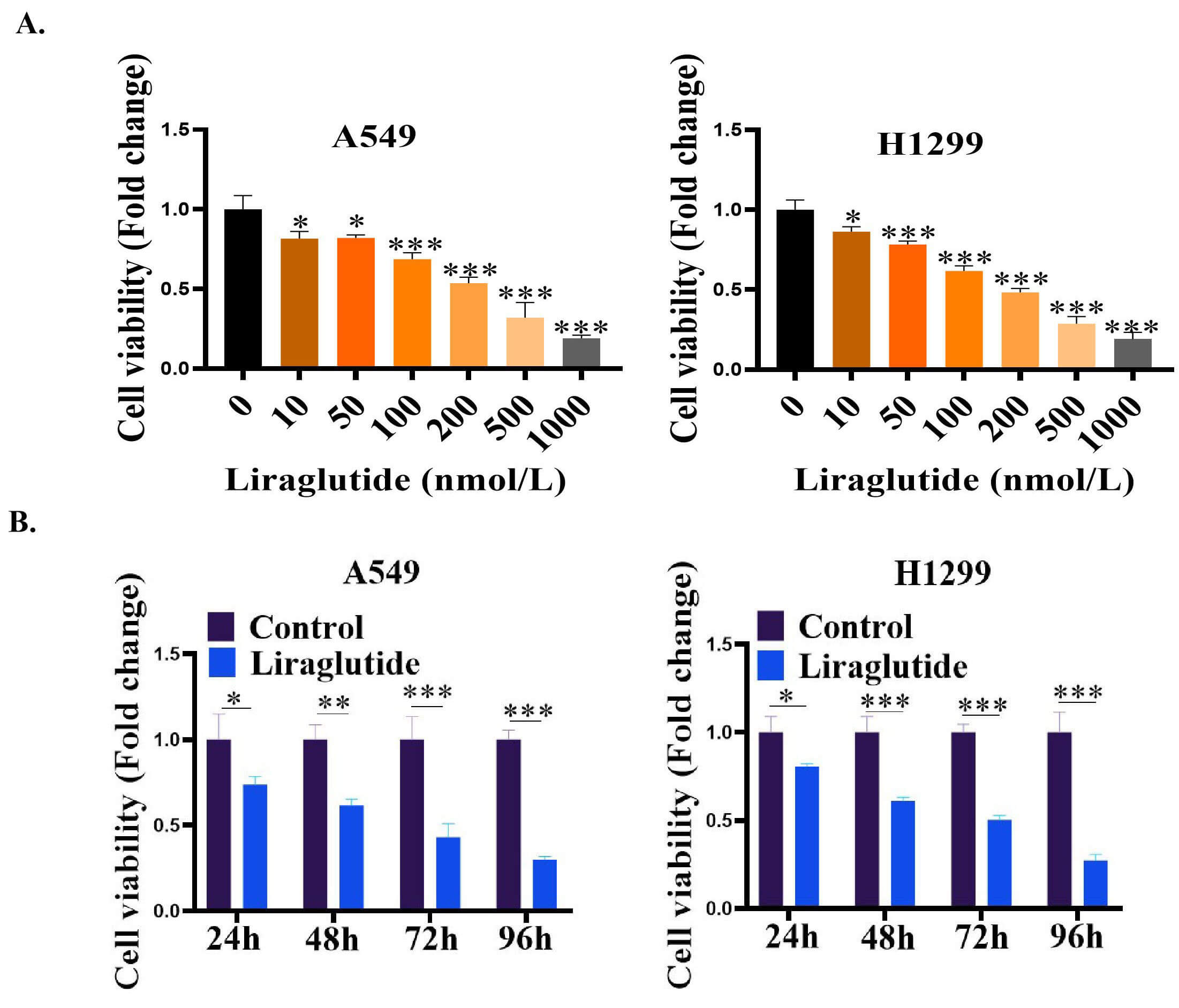 Fig. 1.
Fig. 1.Liraglutide inhibited the proliferation of lung cancer cells in
A549 and H1299 cell. (A) Effect of different concentrations of liraglutide (10
nmol/L–1000 nmol/L) on the proliferation of lung cancer cells (A549 and H1299).
Lung cancer cells were treated with liraglutide for 48 h. (B) Effect of
Liraglutide (150 nmol/L of liraglutide) on the proliferation of lung cancer
cells. The cells were treated with Liraglutide (150 nmol/L) at 24–96 h. The
proliferation of lung cancer cells was detected by CCK8 Kit according to the
manufacturer’s instructions. The absorbance value (OD value) was measured and
recorded using a microplate reader at a wavelength of 450 nm. Data were presented
as mean
The results of the colony formation assay showed that after liraglutide was
added to A549 and H1299 cells for 14 days, the number of lung cancer tumor cell
colonies in the liraglutide treatment group was significantly reduced compared
with the control group (p
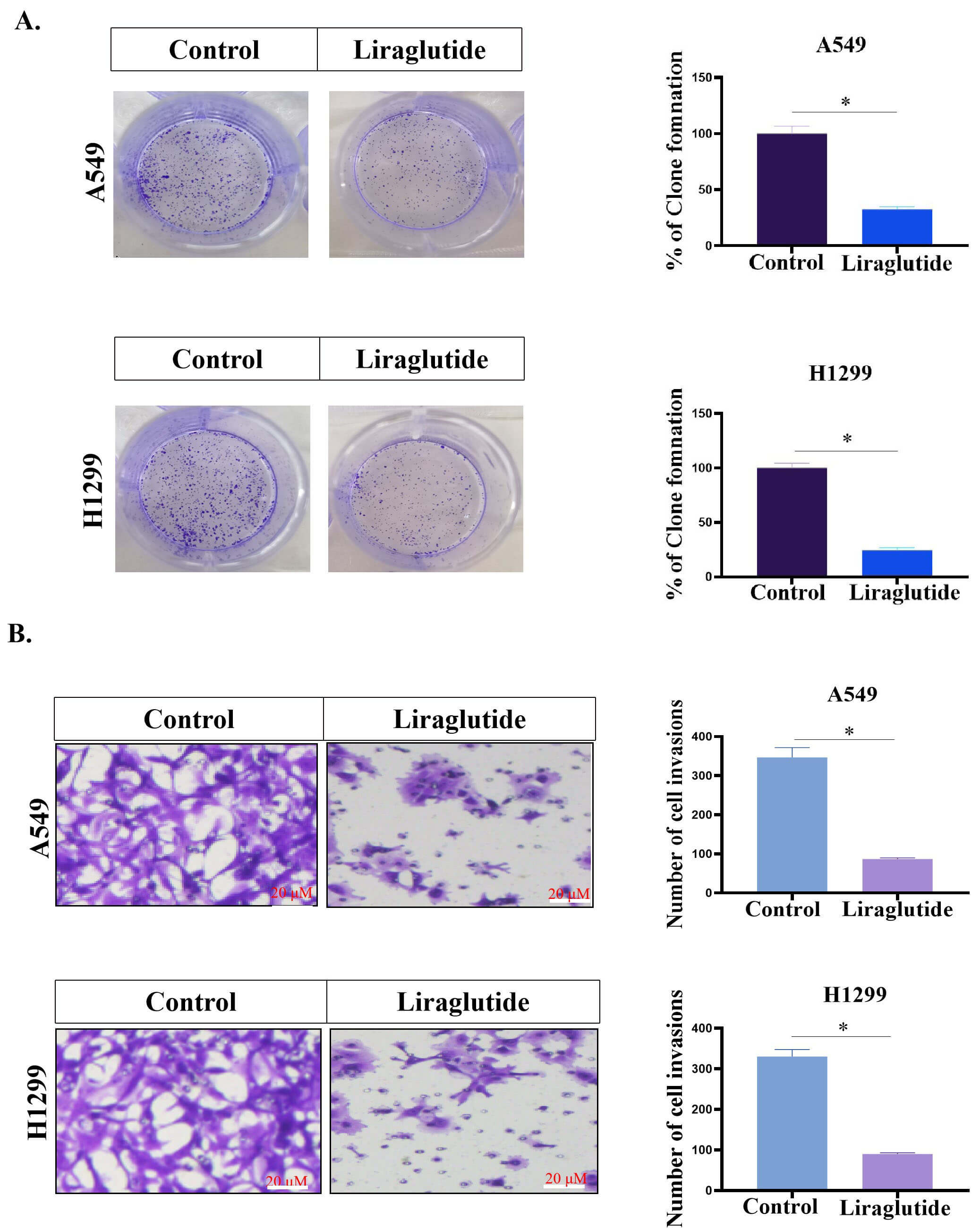 Fig. 2.
Fig. 2.Liraglutide inhibited the proliferation and migration of lung
cancer cells. (A) Effect of Liraglutide (150 nmol/L) on the colony formation of
lung cancer cells. Lung cancer cells were inoculated into 6-well culture plates.
Liraglutide was then added (150 nmol/L) and placed in a 37 °C cell
incubator for 14 days. The number of cell clones were counted from images taken
after crystal violet staining. (B) Liraglutide inhibited the migration of lung
cancer cells (A549 and H1299). Data were presented as mean
In addition, we examined the effect of liraglutide on the migration ability of
human lung tumor cells (A549 and H1299). The results showed that after
liraglutide (150 nmol/L) was added to the cells for 48 h, the migration of
liraglutide-treated tumor cells was significantly reduced compared with that of
the non-treated control cells (p
The effect of liraglutide on lung cancer cells was assessed using flow
cytometry. The results showed that liraglutide induced apoptosis in H1299 and
A549 cells (p
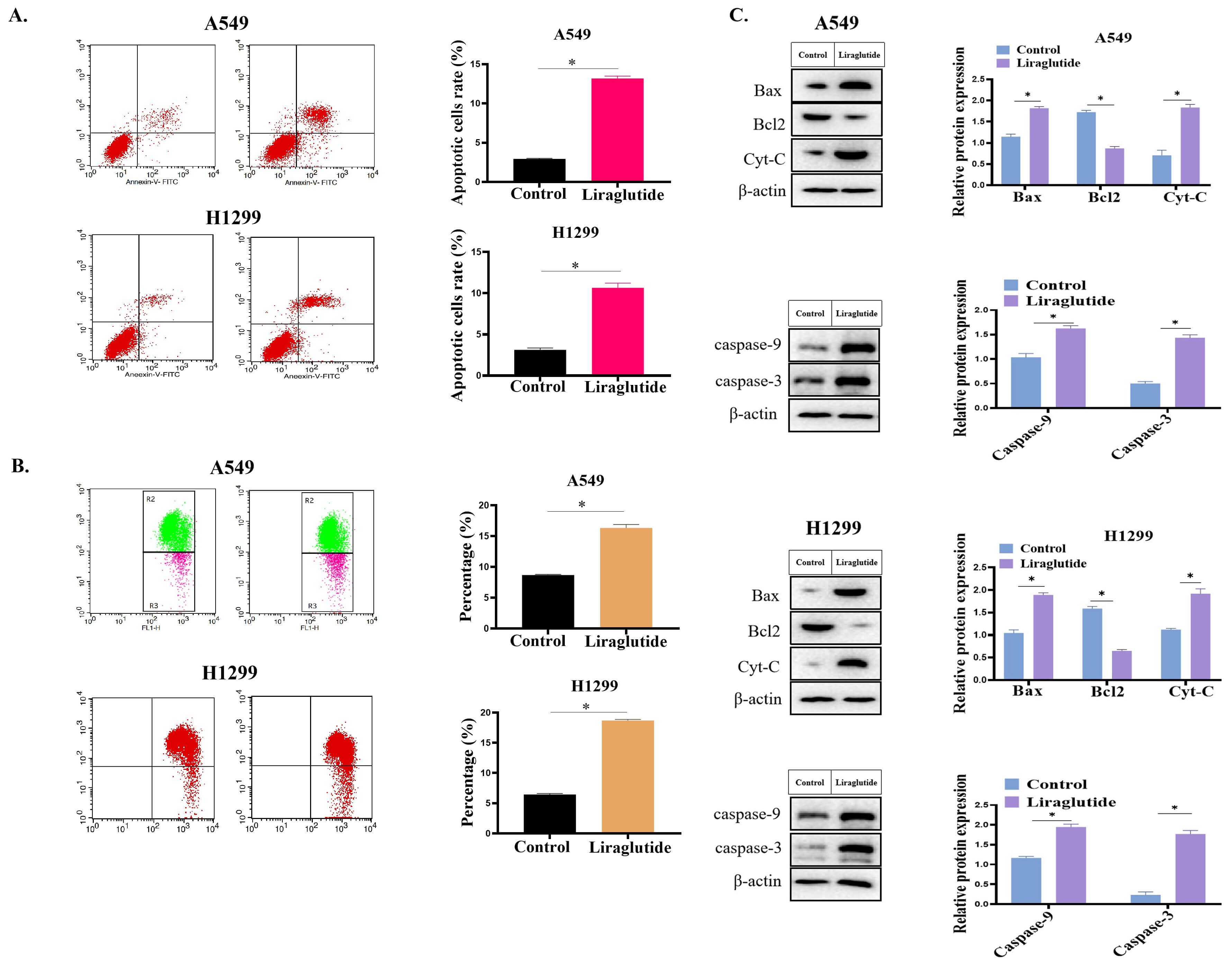 Fig. 3.
Fig. 3.Liraglutide (150 nmol/L) treatment increased the apoptosis rate
of lung cancer cells. (A) Effect of liraglutide on the apoptosis of lung cancer
cells. The cells were digested with trypsin, then washed twice with PBS. Then 5
µL Annexin and 5 µL propidium iodide were added to the cell
suspension and incubated at room temperature for 15 min (in the dark). Cell
samples were analyzed using Flow cytometry. (B) Effect of liraglutide treatment
on Mitochondrial membrane potential. (C) Effect of liraglutide on the expression of apoptosis-related molecules (Bax, Caspase-3, Caspase-9 and
Cytochrome c). Lung cancer cells were collected after liraglutide treatment. The
protein samples were separated using SDS-PAGE and transferred to PVDF membranes.
PVDF membranes were incubated in 5% skimmed milk solution for 1 h. After
washing, the membranes were incubated with diluted primary antibodies at 4 °C
overnight. The membrane was then washed 3 times with TBST and incubated with
secondary antibodies for 2 h at room temperature. After washing, ECL was added to
detect the immunoprotein bands. Data were presented as mean
We analyzed the effect of liraglutide on lung cancer cells to determine whether
the inhibitory effect of liraglutide on lung cancer cell growth was associated
with cell cycle arrest. We performed cell cycle assays on liraglutide-treated
cells using flow cytometry. Cells were stained with propidium iodide after
treatment, and cell cycle distribution was detected using flow cytometry. The
results are shown in Fig. 4. The cell cycle distribution in the A549 control
cells was: G1: 64
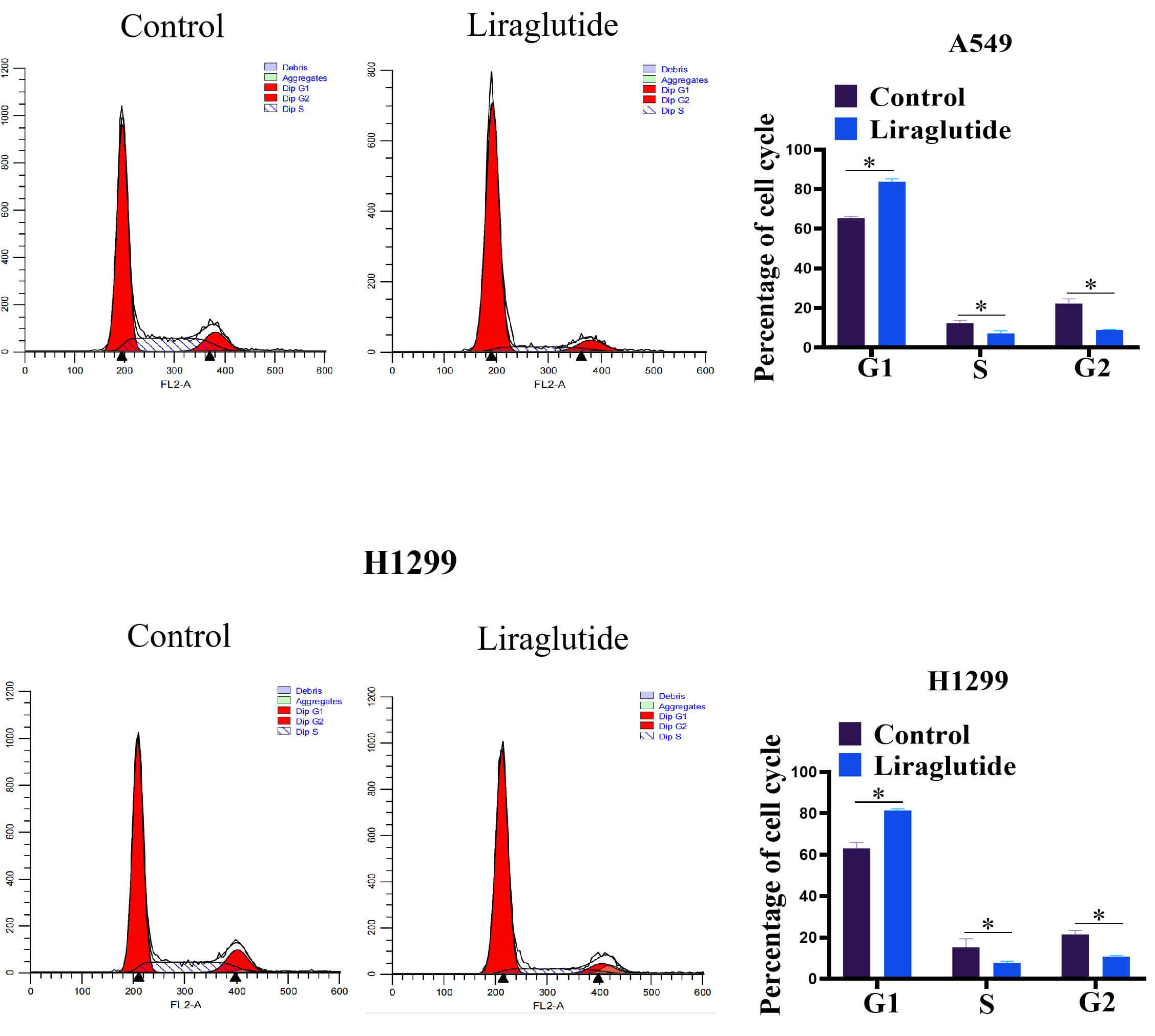 Fig. 4.
Fig. 4.The effect of liraglutide (150 nmol/L of liraglutide) on cell
cycle in the lung cancer. The cells were digested by using trypsin, the lung
cancer cells were collected by centrifugation. After washing, the cells were
treated with RNaseA solution for 20 min. The propidium iodide (PI) staining
solution was assdded, and incubated at 37 °C for 30 min. The cell
samples were analyzed by using Flow cytometry. Histogram represents the
percentage of cell cycle (G1, S and G2). Asterisks indicate significant
differences compared with control group. Data were presented as mean
EMT is generally considered to be an important factor leading to the invasion and migration of cancer cells during tumorigenesis and development [19]. Therefore, we evaluated the effect of liraglutide on the EMT process in lung cancer cells. To this end, 150 nmol/L of liraglutide was added to the medium of A549 cell cultures for 24 h, and western blots were used to detect the cellular expression of EMT-related markers. It was found that liraglutide could significantly reduce the expression of mesenchymal cell markers N-cadherin and vimentin and increase the expression of the epithelial marker E-cadherin (Fig. 5A), Similar results were obtained with the small cell lung cancer line H1299 (Fig. 5B). These results indicated that liraglutide can inhibit the EMT process of lung cancer cells. Further study found that liraglutide treatment decreased the level of PI3K/AKT phosphorylation (Fig. 5C). These results suggested that liraglutide could inhibit the PI3K/AKT signaling pathway. To further clarify whether the inhibitory effect of liraglutide on EMT was related to the PI3K/AKT signaling pathway, lung cancer cells were treated with liraglutide and 20 µM 1,3-dicaffeoylquinic acid, a PI3K agonist. Western blotting showed that the PI3K agonist could reverse the liraglutide-induced increase in E-cadherin levels and decrease N-cadherin and vimentin levels (Fig. 5D), indicating that activation of the PI3K/AKT signaling pathway is the key to the EMT process and that liraglutide suppressed the EMT process through the PI3K/AKT pathway.
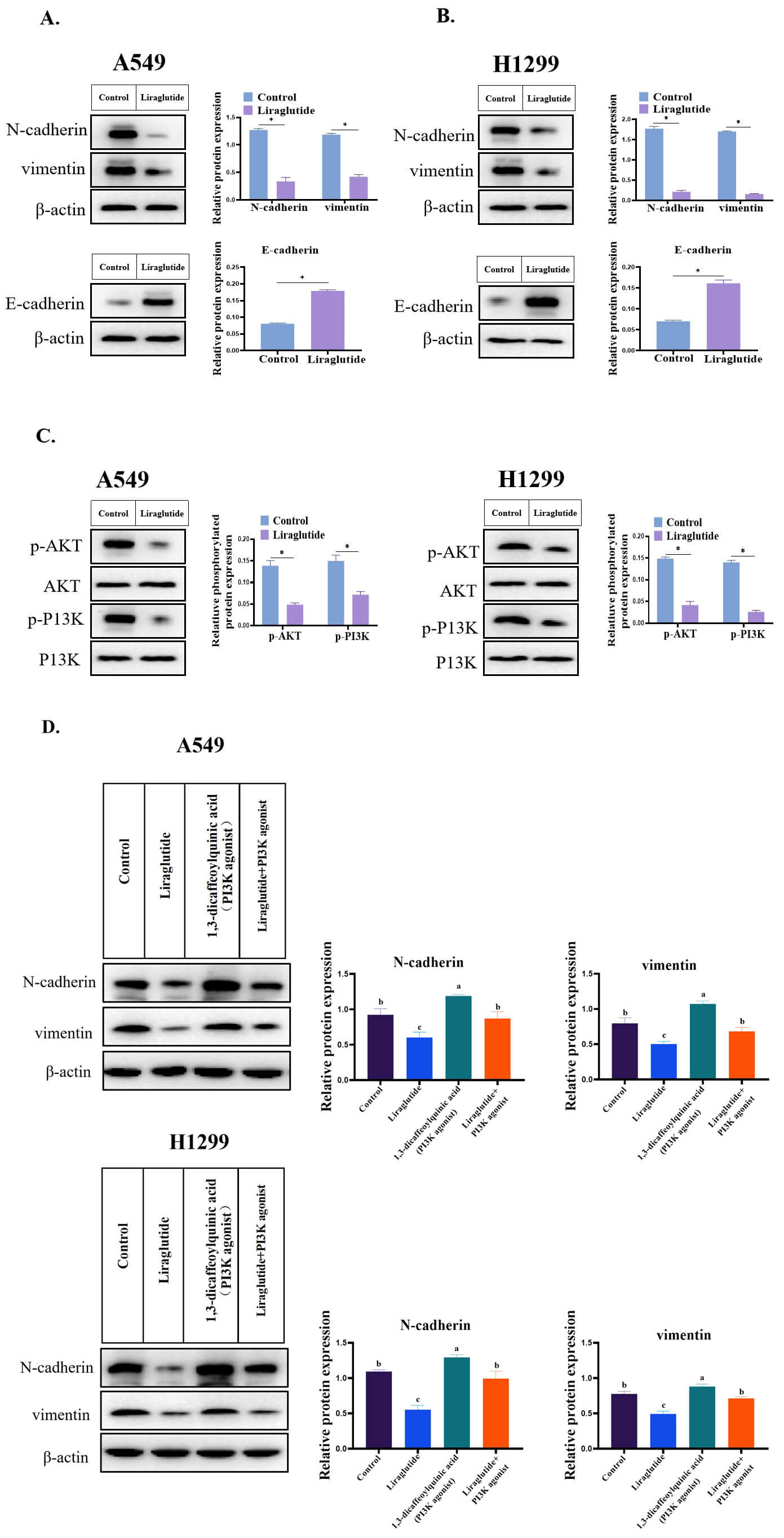 Fig. 5.
Fig. 5.Liraglutide inhibited the EMT process of lung cancer cells. (A)
Effect of liraglutide on N-cadherin, vimentin and E-cadherin in the A549 cell.
Data were presented as mean
To explore whether liraglutide could also inhibit the growth of NSCLC in
vivo, a lung cancer xenograft model in nude mice was established. BALB/c nude
mice were divided into two groups: a control group and a liraglutide-treated
group (six nude mice in each group). The mice were subcutaneously injected with
lung cancer A549 cells (1
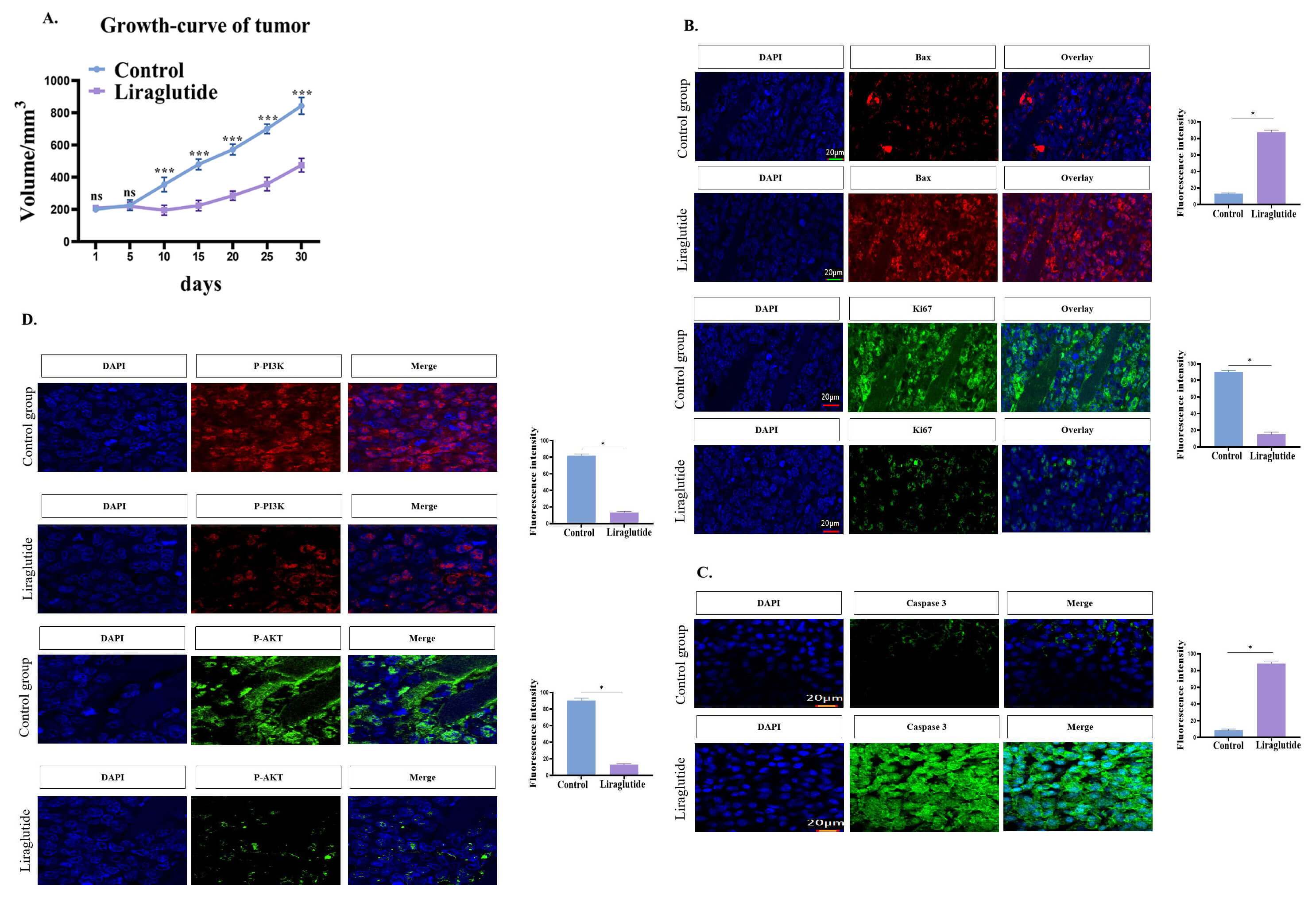 Fig. 6.
Fig. 6.The effect of liraglutide on tumor growth in vivo. (A)
Liraglutide treatment inhibited the proliferation of lung cancer in
vivo. Mice were treated with liraglutide, and tumor volume was measured for 4
weeks. (B) The effect of Liraglutide treatment on the expression of Bax and Ki67.
Tissue sections were blocked with 5% BSA for 30 min. The primary antibody was
then added and placed in a wet box overnight at 4 °C. After washing,
secondary antibodies were added to cover the tissue and incubated for 60 min at
room temperature. Then laser confocal microscopy was performed to observe samples
using an Olympus FV-3000 confocal laser-scanning microscope (DAPI: excitation
wavelength: 330–380 nm, emission wavelength 420 nm, blue; Alexa Fluor 488:
excitation wavelength 495 nm, emission wavelength 519 nm, green; DyLight 594:
excitation wavelength 593 nm, emission wavelength 618 nm, red). Mean fluorescence
intensity of Bax and Ki67 was measured using Image J software (right penal). (C)
Caspase3 was down-regulated in the liraglutide-treated group. Mean fluorescence
intensity of Caspase3 was determined using Image J software (right penal). (D)
Liraglutide treatment down-regulated p-PI3K and p-AKT expression. Mean
fluorescence intensity of P-PI3K was determined using Image J software. Data were
presented as mean
In the above studies, we found that liraglutide exhibited a certain potential
anti-lung cancer effect. In this part of the study, we evaluated the protective
effects of liraglutide on the lung. We established a high glucose-induced lung
cell injury model using BEAS-2B cells. We established three different glucose
cell groups: a control cell group, a high glucose cell group, and a high glucose
+ liraglutide cell group. The results showed that in the high-glucose group,
BEAS-2B cells were senescent using SA-
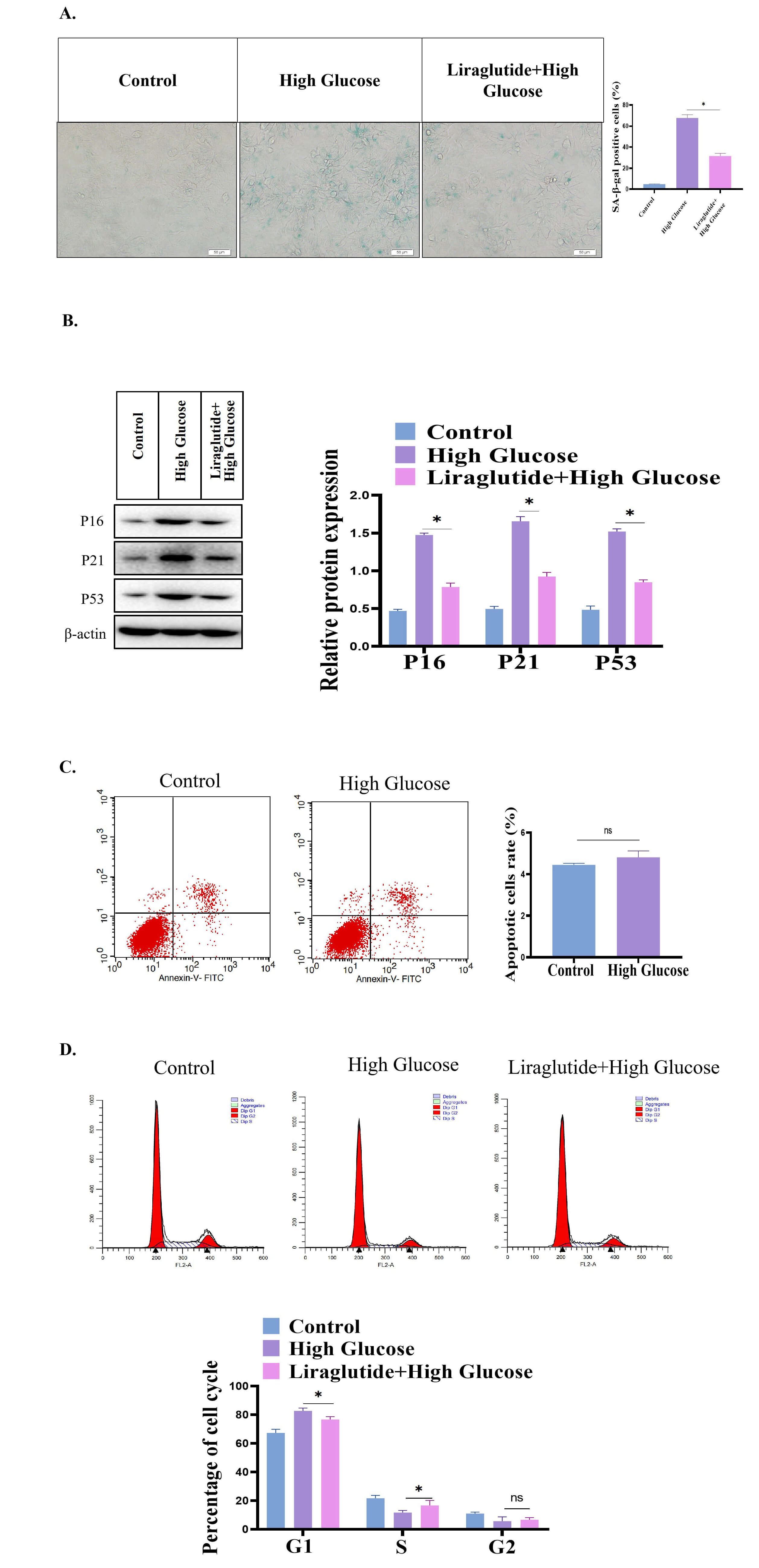 Fig. 7.
Fig. 7.Protective effect of liraglutide on high glucose-induced lung
cell damage. (A) SA-
Additionally, high glucose also caused ERS in BEAS-2B cells. As shown in Fig. 8A, the level of reactive oxygen species (ROS) was significantly increased in the high glucose group. The expression of ERS-related markers GRP78 and CHOP was significantly increased in the high glucose group, while the expression of ERS-related markers was significantly downregulated in the liraglutide treatment group (Fig. 8B). In addition, the expression of unfolded protein response-related molecules (XPB1, ATF6, and IRE1) were also significantly increased in the BEAS-2B group. However, the ubiquitin–proteasome system (UPS)-related signaling molecules (including XPB1, ATF6 and IRE1) were significantly decreased in the liraglutide treatment group (Fig. 8C). These findings showed that liraglutide can significantly alleviate the ERS caused by high glucose.
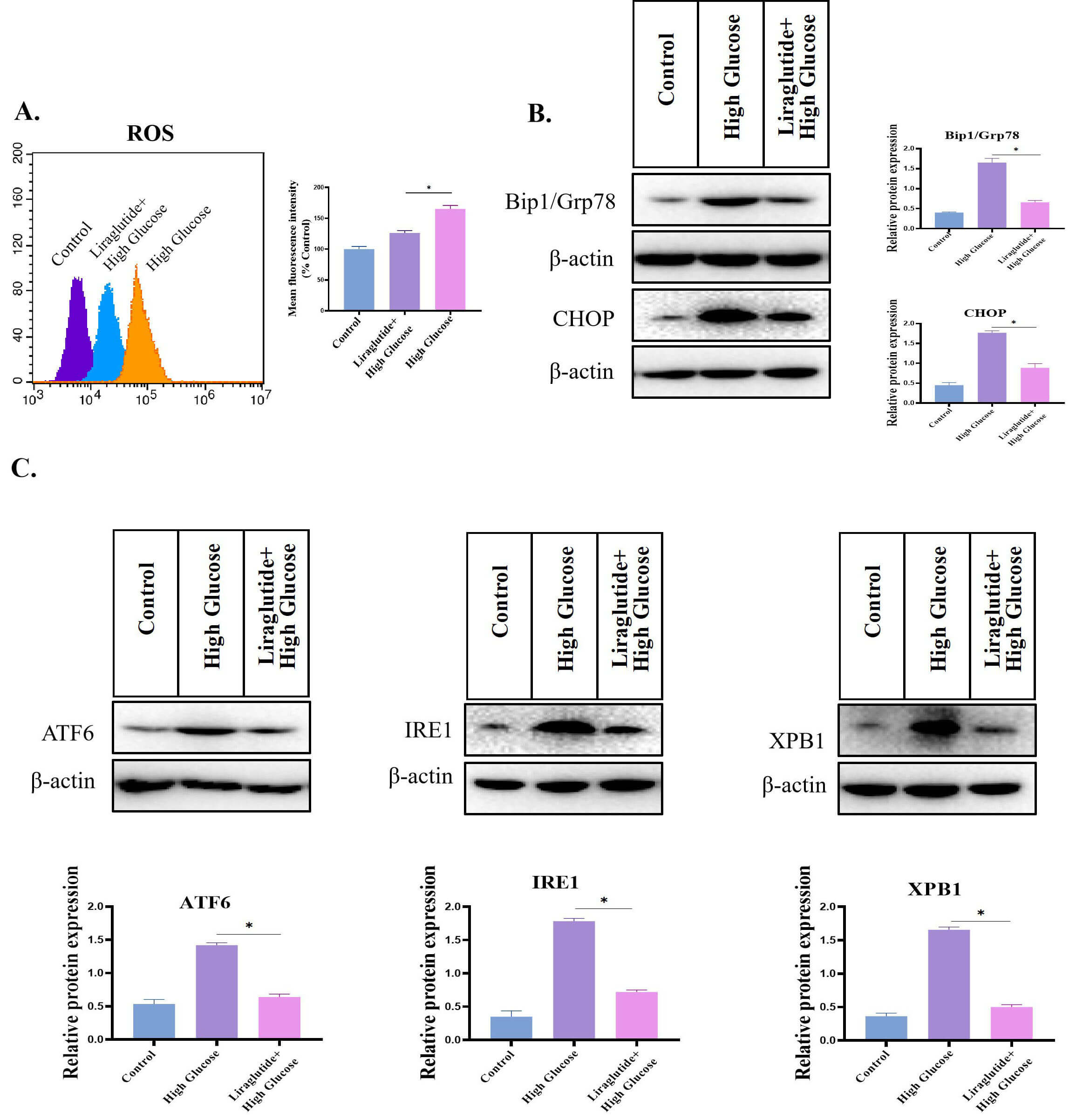 Fig. 8.
Fig. 8.High glucose caused ERS in BEAS-2B cells. (A) Effect of high
glucose on ROS level. (B) Western blotting analysis of GRP78 and CHOP in the
liraglutide treatment group. The protein samples were separated using SDS-PAGE
and transferred to PVDF membranes. PVDF membranes were incubated in 5% skimmed
milk solution for 1 h. After washing, the membranes were incubated with diluted
primary antibodies at 4 °C overnight. The membrane was then washed 3 times with
TBST and incubated with secondary antibodies for 2 h at room temperature. After
washing, ECL was added to detect the immunoprotein bands.
The above studies evaluated the effect of liraglutide on lung cell damage
in vitro. Therefore, in a diabetes mouse model, we further evaluated the
effect of liraglutide on the lungs of diabetic mice. The mice were treated with
Liraglutide (200 µg/kg s.c.) once a day (4 weeks). SA-
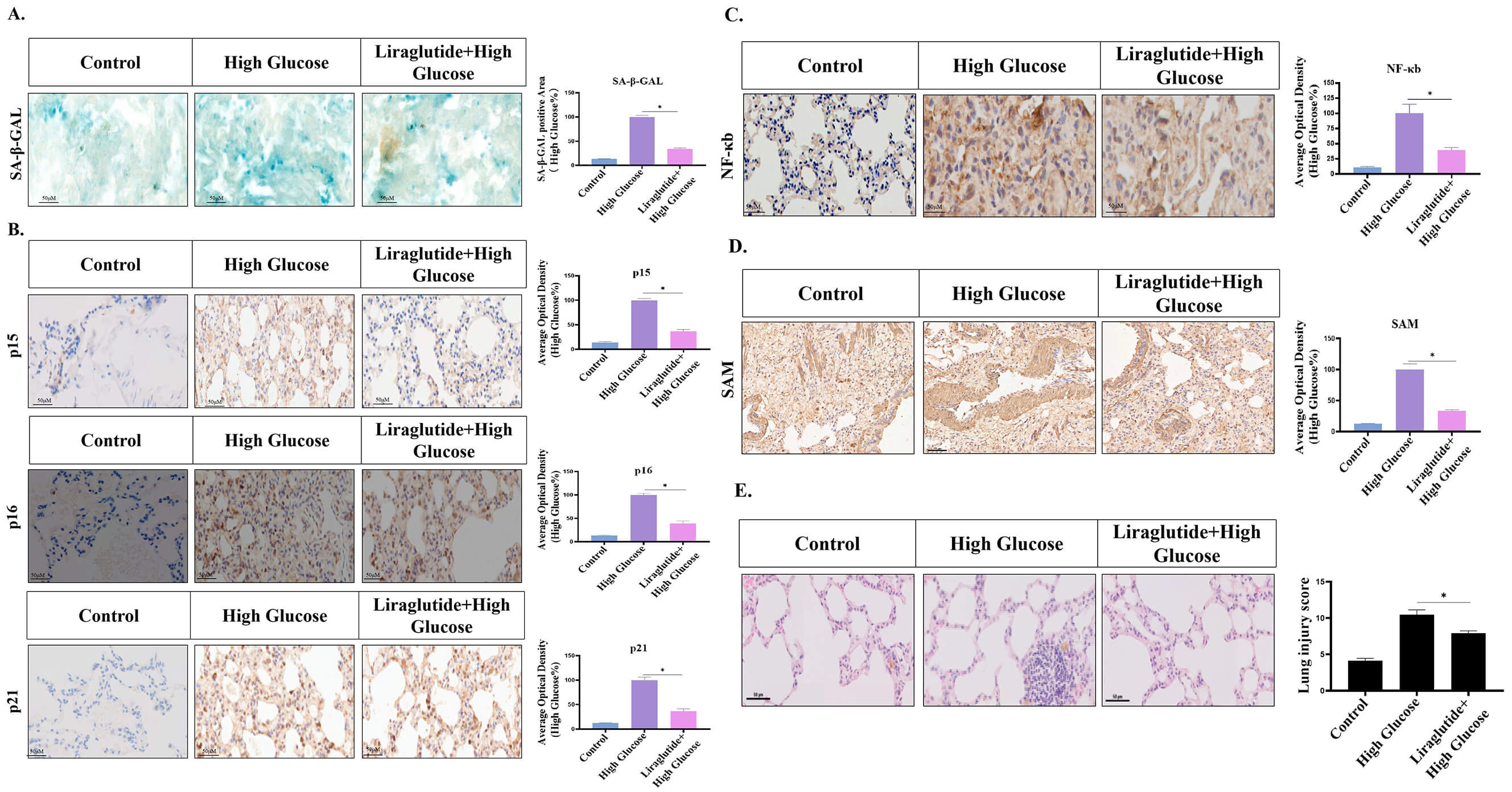 Fig. 9.
Fig. 9.Effect of liraglutide on the lung tissues of diabetic mice. (A)
SA-
Studies have shown that diabetes is closely related to the incidence of lung cancer [21] and that there is a link between diabetes and lung cancer [22]. There are many diabetic patients who present with malignant tumors in clinical practice [23]. Therefore, we examined the effect of liraglutide on lung cancer and found that it has a potential anti-lung cancer effect. Moreover, we found that liraglutide had a protective effect on lung damage.
In lung cancer patients with diabetes mellitus, an important scientific question
to address is whether glucagon analogs or agonists (liraglutide) used in the
treatment of diabetes have effects on tumors. In the current study, we found that
liraglutide exhibited partial anti-lung cancer potential. However, previous
studies on the effects of glucagon analogs or agonists on tumors have been
conflicting. Studies have shown that GLP-1R agonists can activate the Wnt
signaling pathway in islet
Although existing clinical studies have shown that exenatide can inhibit the proliferation and migration of human pancreatic cancer, prostate cancer, breast cancer, and other tumor cells, these studies were mostly limited to investigating the effect of short-acting GLP-1R agonists on the biological behavior of tumor cells, while study of the long-acting GLP-1R agonist liraglutide has been limited. In our study, we explored the biological effect of liraglutide on human lung cancer cells. The results showed that liraglutide could significantly inhibit the migration of lung cancer cells. EMT is generally considered to be an important factor leading to the invasion and migration of cancer cells during tumorigenesis and development [24]. Therefore, we evaluated the effect of liraglutide on the EMT process of lung cancer cells, and results showed that it could inhibit the EMT process.
Diabetes can cause lung damage [25]. Diabetes is a chronic metabolic disease with complications that can affect various organs throughout the body. However, most of the attention has been given to the damage that diabetes causes to the heart, kidney, and eye, while the lung has been neglected, even though it is one of the target organs [26]. Therefore, we investigated whether liraglutide had a protective effect on the lung damage caused by hyperglycemia. We established a model of high glucose-induced lung cell injury and showed that high glucose induced the senescence of BEAS-2B cells and that liraglutide could significantly inhibit high glucose-induced lung cell damage. High glucose also caused ERS in BEAS-2B cells. The expression of ERS markers was significantly increased in the control cells, while in liraglutide-treated cells, the ERS-related protein expression was significantly downregulated. In addition, the expression of UPS-related molecules was also significantly increased in BEAS-2B control cells, while in the liraglutide-treated cells, the expression of UPS-related signaling molecules was significantly decreased. These results showed that liraglutide could significantly alleviate the ERS caused by high glucose.
The dose of liraglutide used in the current study was based on the following two considerations: (1) One is based on the findings of previous studies [27, 28, 29, 30]; (2) The other one is based on our pre-experimental results. The concentration of liraglutide used in in vitro experiments varies depending on the cell type. There is no absolute standard dose. Another interesting phenomenon is that liraglutide induces apoptosis in tumor cells, but liraglutide protects normal somatic cells from senescence damage. In this work, Liraglutide indicated an anti-ageing effect on normal lung cells. In contrast, liraglutide exhibited a pro-apoptotic effect on the tumour model. They are not contradictory. Previous study has showed that Liraglutide can induce apoptosis in other types of tumours [31, 32, 33]. However, in normal somatic cells, liraglutide exhibits a protective effect [27, 28, 29, 30]. In addition, the anti-ageing effect displayed by liraglutide in lung tissue can also be explained, our finding showed that liraglutide is able to reduce oxidative stress which is considered to be one of the important factors that induce aging. Of course, the deep molecular mechanism of anti-aging of liraglutide needs to be further revealed in future research.
We evaluated the effects of liraglutide on lung cell damage in vitro and on the lungs of diabetic mice. The results showed that aging of the lungs was alleviated by liraglutide. Therefore, we have shown that liraglutide can not only inhibit lung cancer but can also significantly alleviate lung damage.
ERS, endoplasmic reticulum stress; FBS, Fetal Bovine Serum; NSCLC, non-small cell lung cancer; CLSM, Confocal Laser scanning microscopy; GLP-1R, Glucagon-like peptide-1 receptor; EMT, Epithelial-Mesenchymal Transition; PI3K, phosphatidylinositol 3 kinase; AKT, protein kinase B.
Data are available from the corresponding author on reasonable request.
Conceptualization, ZP, YY and WC; formal analysis, ZP and SQ; funding acquisition, WC; investigation, ZP and XL and CC; writing-original draft, ZP, WC and CC; writing-review and editing, ZP and WC. All authors contributed to editorial changes in the manuscript. All authors have read and agreed to the published version of the manuscript.
The experimental protocol was approved by the Institutional Animal Care and Use Committee of Northwest Minzu University (IACUC-20210215).
We thank Dr. Zhang Wei for technical assistance.
This work was supported by Gansu Province Science and Technology Program Project (2022) - Natural Science Foundation (22JR5RA738), and by Central Universities Basic Research Business Fund (2021) - Young Teachers Innovation Project (31920210047).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.









