1 Federal State Budgetary Scientific Institution “Research Institute of Fundamental and Clinical Immunology” (RIFCI), 630099 Novosibirsk, Russia
2 State Research Center of Virology and Biotechnology “Vector”, 630559 Koltsovo, Russia
3 V. Zelman Institute for Medicine and Psychology, Novosibirsk State University, 630090 Novosibirsk, Russia
Academic Editor: Graham Pawelec
Abstract
Background: A search for efficient graft rejection modulation
techniques for the promotion of durable engraftment remains to be a matter of
close study all over the world. Despite the variety of immunosuppressive drugs,
the schemes currently used show a lack of selectivity and have a number of side
effects. Here we investigated an approach for the induction of antigen-specific
tolerance in a human “stimulator-responder” model in vitro, using
dendritic cells (DCs) transfected with designed DNA constructs encoding the
stimulator’s major histocompatibility complex (MHC) epitopes. Methods:
The object of the study is peripheral blood mononuclear cells (PBMCs) from 10
healthy donors. To induce antigen-specific tolerance, personalized DNA constructs
were created for five responder–stimulator pairs, based on the sequences of
donors’ and recipients’ MHCs. DNA sequencing was performed to select epitopes for
incorporation into genetic constructs. A mixed lymphocyte culture assay was used
(i) to assess the proliferative response in both directions for all possible
stimulator–responder pairs (90 reactions) and (ii) to assess the tolerogenic
properties of the generated transfected DCs (5 reactions). Results:
A significant increase in the amounts of FoxP3
Keywords
- mediated immune suppression
- mixed lymphocyte culture
- T regulatory cells
- tolerogenic dendritic cells
- DNA constructs
- HLA
- MHC
- alloantigens
The issues of transplanted organ rejection and graft versus host disease (GVHD) reaction remain topical despite modern methods for antigenic matching of donor and recipient tissues and immunosuppressive drug therapy. For example, according to published data, 60 out of 315 kidney allografts developed an acute rejection reaction [1], and 1921 out of 2813 bone marrow cell transplantations were associated with an acute graft versus host disease reaction [2]. Allograft rejection is controlled by two main immunological mechanisms: innate nonspecific reactions with proinflammatory signals and donor-specific adaptive responses. The adaptive response results from the presentation of alloantigens by antigen-presenting cells, mainly dendritic cells (DCs), and their allorecognition by recipient T-cells [3, 4, 5, 6].
Currently available immunosuppressive drugs do not exhibit sufficient selectivity and their use may cause side effects including suppression of IFN-gamma production, hematopoiesis inhibition leading to the development of leukopenia, thrombocytopenia, anemia, and even pancytopenia, impairment of defense functions of the body, the activation of secondary infection, septicemia, and neoplasia [7]. Therefore, reducing the dependence on immunosuppressive drug therapy and inducing tolerance to donor antigens are the main purposes of studies on transplantation, and technologies for inducing antigen-specific tolerance are now considered innovative strategies to promote durable engraftment [8]. Various cell types are used to induce suppression, including mesenchymal stem cells in bone marrow transplantation [9], regulatory T cells [10], myeloid regulatory cells (macrophages, dendritic cells, myeloid suppressors) [11], regulatory B cells [12], and apoptotic donor cells [13]. Dendritic cells link innate and acquired immune responses and control immunity and tolerance, and therefore are key components for the modulation of graft rejection. To induce antigen-specific immunological tolerance, dendritic cells are loaded with an antigen to which tolerance is to be generated. Currently, a donor cell lysate [14] or vesicles derived from donor tissue [15] are used to load tolerogenic DCs with alloantigens. However, this method does not ensure the long-term presence of an antigen in dendritic cells; therefore, DC-induced antigen-specific tolerance may disappear when transferred to a recipient [16]. In this study, we investigated the potential of generating antigen-specific tolerance in stimulator–responder pairs using DNA constructs encoding major histocompatibility complex (MHC) epitopes of stimulators as an antigen source for dendritic cells as responder cells, as well as the induction of non-specific suppression associated with the expression of the immunoregulatory cytokine IL-10.
Peripheral blood samples from healthy donors (n = 10) were used in the study. The donor group included six females and four males with a mean age of 35.3 years. Whole blood samples were obtained from Blood Procurement Station No. 1 of the State-Government-financed Institution of Public Health of the Novosibirsk Region (Novosibirsk Blood Center). Voluntary informed consent was obtained from all donors. The study followed the principles outlined in the Declaration of Helsinki for all human and animal experimental investigations and was approved by the local Research Institute of Fundamental and Clinical Immunology (RIFCI) ethics committee.
The following plasmid DNA constructs were used in the study. (i) Five personalized DNA constructs were created for five responder–stimulator pairs, based on the sequences of donor and recipient major histocompatibility complexes. The constructs produced for each of the five donor-recipient pairs are hereinafter designated as the “pMLC”. (ii) pIL-10, a pmax-pIL-10 construct encoding human IL-10, was used to stabilize the tolerogenic phenotype of DCs. Artificial genes were designed using Gene Designer 2.0 software (DNA2.0, ATUM, Newark, CA, USA), utilizing the developed amino acid sequences of human IL-10. (iii) A control plasmid DNA construct based on the DNA-vector pDNAVACCultra5 without inserts of immunogenic peptides is hereinafter designated as the “p5”.
Peripheral blood was drawn from donors into vacuum tubes containing EDTA as an
anticoagulant (Improvacuter, China). Peripheral blood mononuclear cells (PBMCs)
were isolated from whole blood samples using a conventional Ficoll–Urografin
density gradient method [17] (Ficoll: PanEco, Moscow, Russia; Urografin: Schering
AG, Germany). Briefly, peripheral blood was diluted with an equal volume of
RPMI-1640 medium (Biolot, BerliMed, S.A., Spain), then layered on a
Ficoll-Urografin solution (
The immature dendritic cells were generated as described above. Then the DCs
were harvested, washed with PBS, and transfected with a DNA plasmid encoding
IL-10 (pIL-10) to achieve the tolerogenic phenotype using the DC electroporation
method. The transfection of dendritic cells with the DNA constructs of interest
was performed by an electroporation method using the BTX ECM 830 square-wave
electroporator (BTX, Holliston, MA, USA) with a pulse strength of 260 V; pulse
duration of 10 ms; and plasmid DNA concentration of 60
The phenotype of DCs transfected with the pIL-10 was evaluated by flow cytometry according to the expression of the surface markers CD11c (PeCy7), HLA-DR (FITC), CD83 (APC), CD86 (PE), CD80 (Brilliant Violet 421), and CCR7 (APC-Cy7) at 6, 24, and 48 h after transfection, using corresponding monoclonal anti-mouse antibodies (BioLegend, USA). Production of IL-10 by transfected DCs was determined in the supernatant of cultured cells at 1, 2, 3, 6, 24, and 48 h after transfection by ELISA using the Interleukin-10-EIA-BEST kit (Vector-Best, Novosibirk, Russia).
To evaluate the possible effects of pIL-10-transfected tolerogenic DCs on a culture of mononuclear cells, the pIL-10-transfected DCs were co-cultured with autologous mononuclear cells (non-adherent PBMC fraction) at a 1:10 ratio at 6 h after transfection.
The production of IL-10 was assessed by ELISA, using the Interleukin-10-EIA-BEST
kit. To determine the number of T-regulatory cells, a flow cytometry analysis of
the joint “MNCs+DCs” culture was performed after 48 h of co-cultivation, using
monoclonal antibodies to CD45 (PerCP), CD4 (PeCy7), CD25 (PE), and FoxP3 (Alexa
488) (BioLegend, San Diego, CA, USA) T-regulatory cells were identified as
CD4
The level of intracellular IL-10 was measured by intracellular staining of IL-10 via flow cytometry using monoclonal antibodies to human IL-10 (PE) (BioLegend, San Diego, CA, USA).
PBMCs were isolated from whole blood samples as described above. A mixed
lymphocyte culture (MLC) assay was performed to assess the proliferative response
in both directions for all possible inducer–responder pairs (90 reactions). The
MLC assay was performed in 96-well round-bottom plates (TPP, Trasadingen,
Switzerland) in triplicate for each reaction. Responder PBMCs were added at 2
Blood samples were frozen at –20 °C and transferred to the Dmitry Rogachev National Research Center of Pediatric Hematology, Oncology, and Immunology of the Ministry of Health of the Russian Federation (Moscow, Russia) for the DNA sequencing of MHC loci. DNA sequencing was performed using NGS on an Illumina MiSeq® sequencer (Illumina, San Diego, CA, USA) with a TruSight HLA v2 Sequencing Panel kit (Illumina, San Diego, CA, USA) according to the manufacturer’s instructions. The DNA sequence data were used to analyze sequences and select epitopes for incorporation into genetic constructs.
A software product was developed to generate the constructs of interest, which enabled the personalized selection of major histocompatibility complex epitopes chosen based on a complex analysis of the responder and stimulator HLA sequences. The design of artificial genes encoding target constructs, cloning of the artificial genes into the pmax-Ub vector, and production of final genetic constructs encoding target protein products were carried out. Then, genetic constructs encoding target protein products were amplified and purified from endotoxins.
Therefore, personalized DNA constructs were created for five responder–stimulator (recipient–donor) pairs, based on the sequences of donor and recipient major histocompatibility complexes. Artificial genes were designed using Gene Designer 2.0 software (DNA2.0, ATUM, Newark, CA, USA) using the developed amino acid sequences of target immunogens. Human codon frequencies were used to optimize the codon composition. The recognition sites of restriction endonucleases AgeI, BamHI, and SacI were excluded from the nucleotide gene sequences for further possibility of cloning into the pmax-Ub vector.
At the gene 5
Human dendritic cells were generated from peripheral blood mononuclear cells in
the presence of rhGM-CSF and rhIL-4 as described above. The resulting immature
dendritic cells were harvested and transfected, using electroporation, with the
DNA plasmids encoding epitopes of stimulators’ MHC molecules (the pMLC
constructs). The IL-10-encoding plasmid was also used in a part of samples to
stabilize the tolerogenic phenotype of DCs. Next, transfected dendritic cells
were co-cultured with autologous mononuclear cells for 96 h. Then the number of
T-regulatory cells and IL-10 production were determined in the joint culture
using the methods described above. After co-cultivation, the MLC assay was
performed on joint cell cultures to assess the ability of generated tolerogenic
DCs to restrict the activation of autologous MNCs in response to stimulation by
MHC alloantigens. For this, the joint cultures of transfected DCs and autologous
MNCs were seeded as respondent cells into a flat-bottomed plate at 1
Statistical analysis was performed using Prism 7.0 (GraphPad Software, La Jolla,
CA, USA). Statistical significance was determined using the nonparametric
Kruskal-Wallis test with Dunn’s multiple comparison test. Values of p
A tolerogenic response can be induced by suppressive cytokines, such as IL-10 and TGF-beta. One technique to preserve the immature DC phenotype after in vivo delivery is the genetic modification of DCs with viral vectors expressing IL-10 and TGF-beta, because the continuous expression of these cytokines prevents the maturation of DCs. In our study, the pIL-10 plasmid was used to induce a stable, nonspecific, and tolerogenic phenotype of dendritic cells. The supernatants of cultured dendritic cells were collected at 1, 2, 3, 6, 24, and 48 h after transfection to determine the content of IL-10 using an enzyme-linked immunosorbent assay. IL-10 production upon cultivation of transfected DCs increased as early as the first hours after transfection and up to 24 h. Cytokine production reached a plateau between 24 and 48 h (Fig. 1). The control plasmid p5 did not induce IL-10 production. Similarly, we previously showed that the data obtained in mouse models were consistent with previous studies using the plasmid encoding murine IL-10 [19].
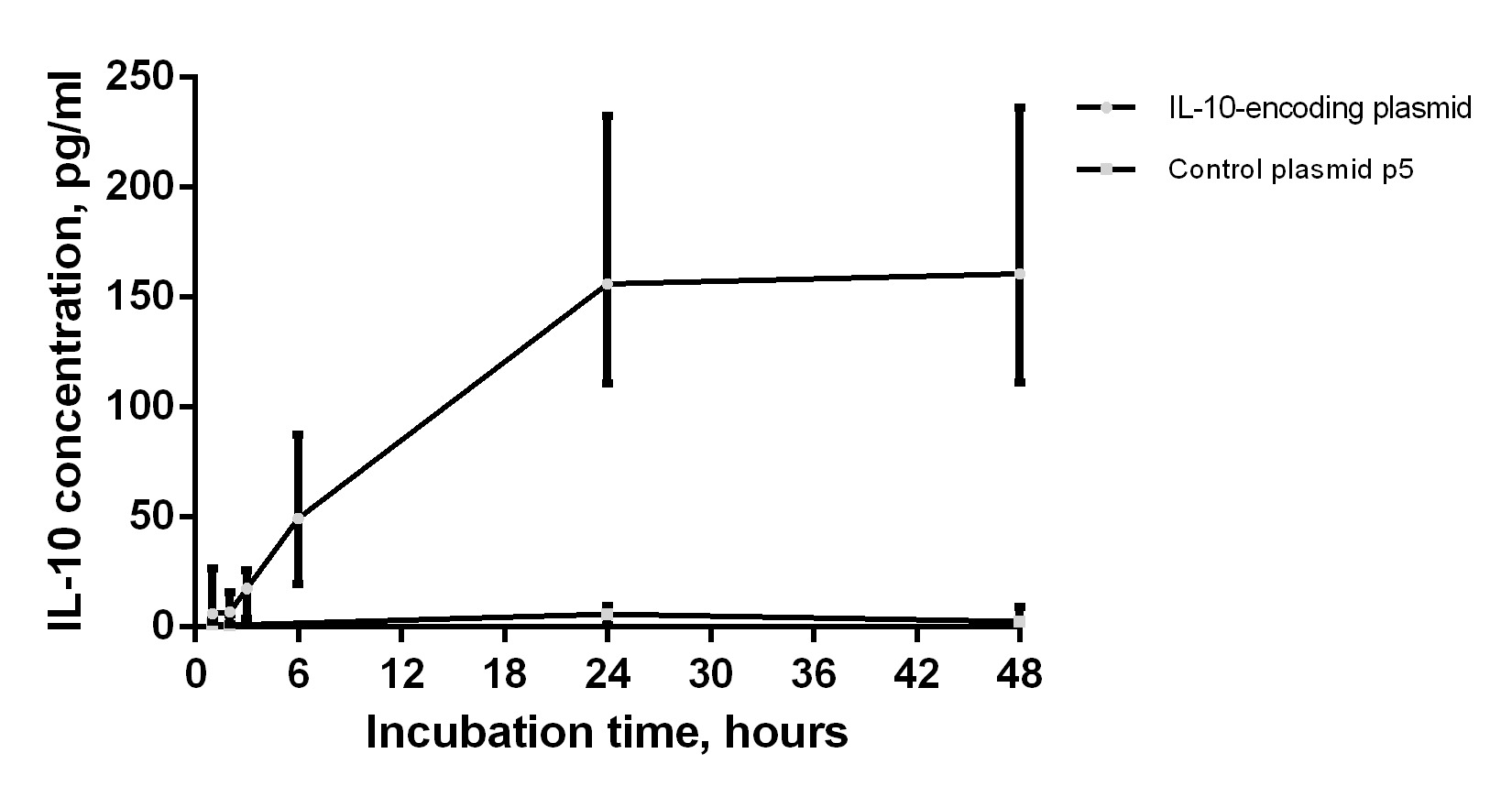 Fig. 1.
Fig. 1.Production of IL-10 by transfected dendritic cells (n = 8). The x-axis corresponds to the number of hours after the pIL-10 DC transfection. Data are presented as median and interquartile range.
Results of flow cytometry assessment of intracellular IL-10 in dendritic cells in 1, 2, 3, 6. 24 and 48 h after transduction (Fig. 2) showed that the relative number of cells expressing intracellular IL-10 increased significantly on hour 2 after transduction. The relative number of dendritic cells transfected by control p5 plasmid was under 5% during 48 h after the transfection.
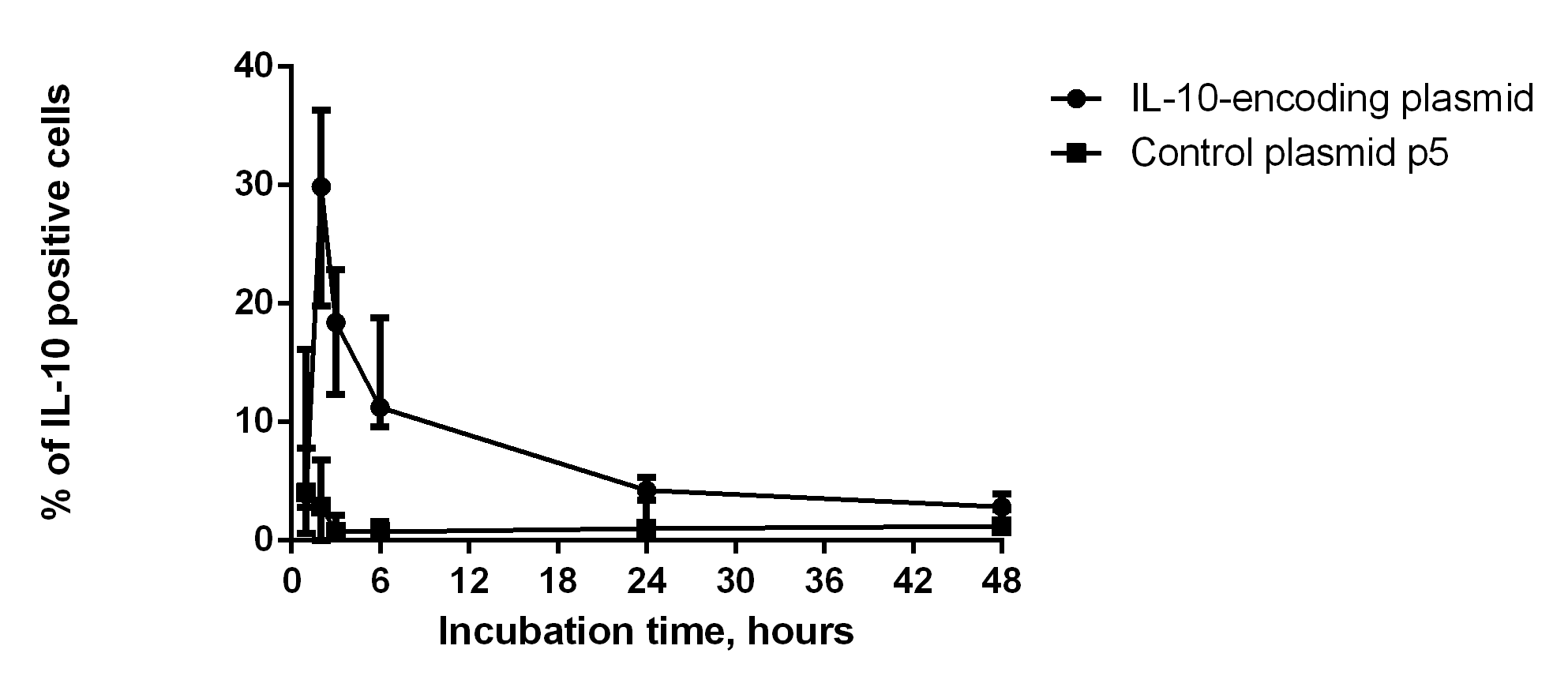 Fig. 2.
Fig. 2.Intracellular production of IL-10 by transfected dendritic cells (n = 6). The x-axis corresponds to the number of hours after the pIL-10 DC transfection. Data are presented as median and interquartile range.
The phenotype of DCs transfected with the IL-10-encoding plasmid was evaluated by the expression of surface markers including CD83, CD86, CD80, and CCR7 at 6, 24, and 48 h after transfection. CD80 marker expression was significantly increased on the surface of transfected DCs. CCR7 expression was increased on the surface of transfected DCs 6 h after transfection, after which it decreased but remained elevated compared with non-transfected DCs. Expressions of CD83 and CD86 were decreased on the surface of transfected DCs and remained stable for 48 h of cultivation. Therefore, a stable tolerogenic phenotype with preserved cell migration potential was produced in transfected dendritic cells (Fig. 3).
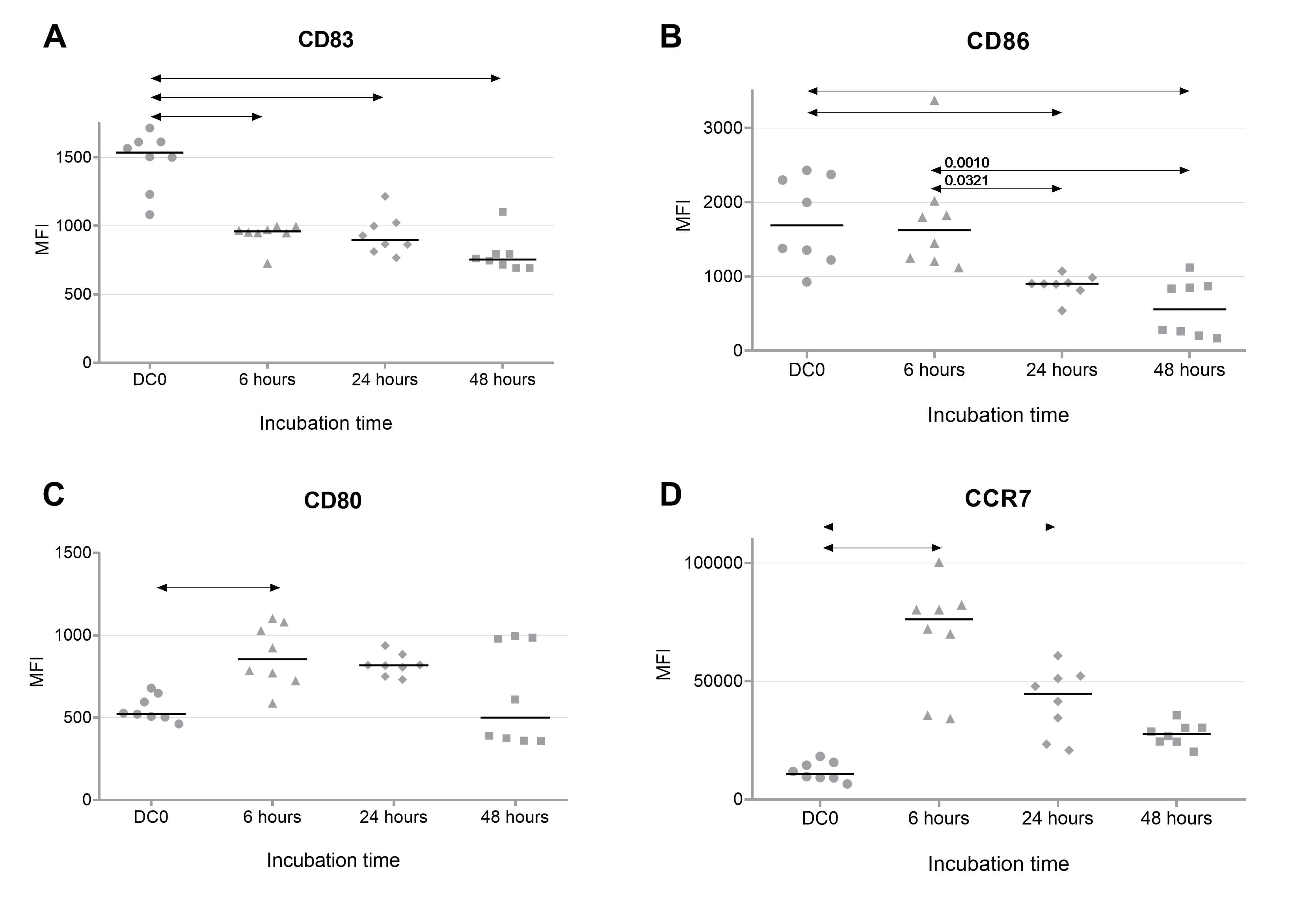 Fig. 3.
Fig. 3.Expression of DC maturation markers CD83 (A), CD86 (B), CD40 (C)
and CCR7 (D) at 6, 24, and 48 h after transfection with a plasmid encoding IL-10
(n = 8). DC (0) – non-transfected dendritic cells. Data are presented as median
and individual values. The arrows indicate statistically significant differences,
p
Transfected DCs were co-cultured with mononuclear cells at a 1:10 ratio 6 h after transfection, and the production of IL-10 and the number of T-regulatory cells were evaluated after 48 h of co-culture. Co-cultivation of MNCs and DCs transfected with the pIL-10 plasmid was accompanied by the stable production of IL-10, which promoted the maintenance of tolerogenicity. Co-cultivation of MNCs and DCs transfected with the IL-10 plasmid increased the number of T-regulatory cells, which promoted the maintenance of tolerogenicity (Fig. 4).
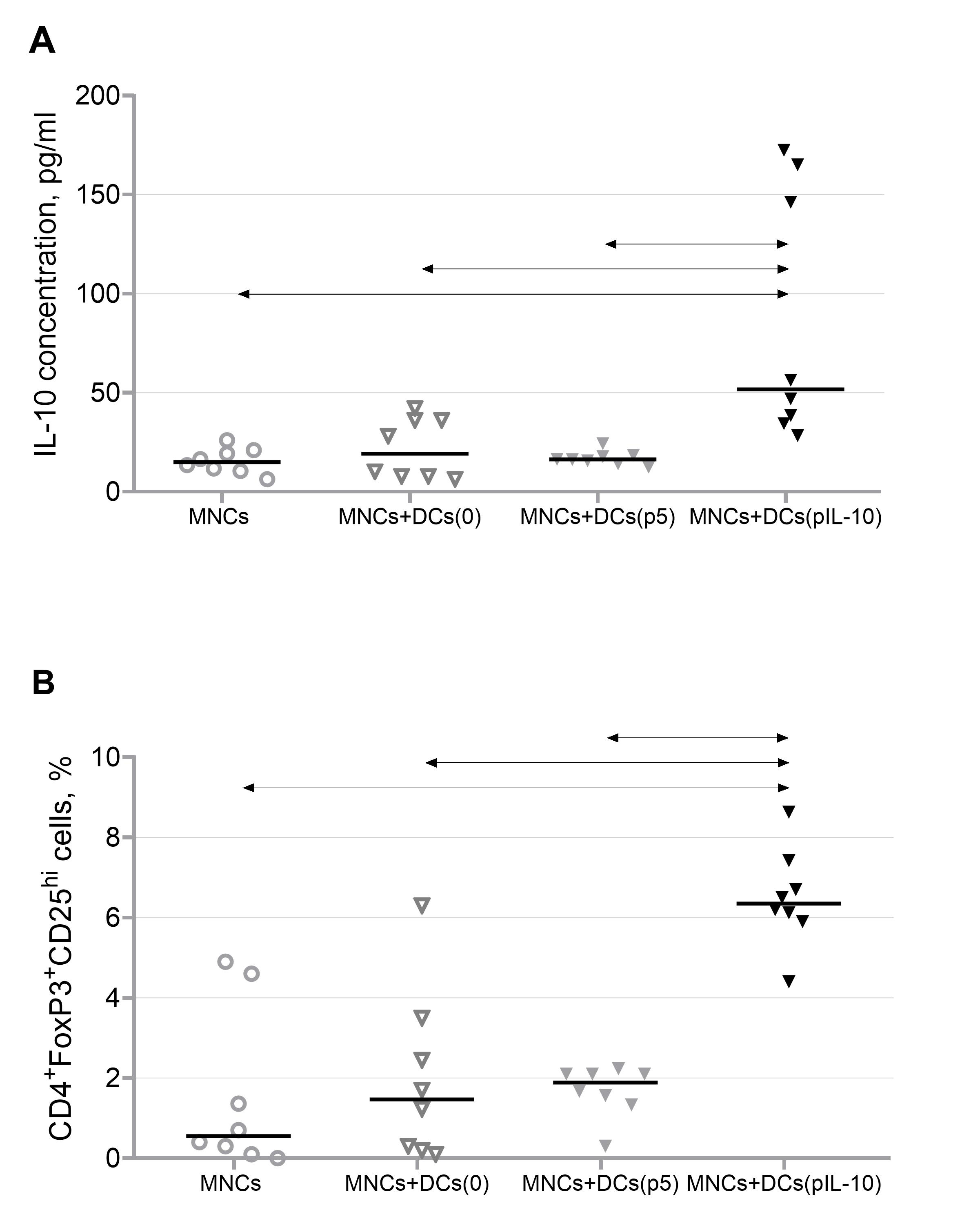 Fig. 4.
Fig. 4.Tolerogenic properties of pIL-10-transfected cells. (A)
Production of IL-10 by MNCs in the presence of DCs transfected with a plasmid
encoding IL-10 (n = 8). (B) Relative numbers of CD4
Therefore, the use of a DNA construct encoding IL-10 enabled stable cytokine production in DC cultures and the generation of DCs with a tolerogenic phenotype. To assess the maximal and minimal allogeneic responses, we conducted mixed lymphocyte cultures from 10 healthy donors and generated a panel of donor genotypes. To evaluate the level of response, stimulation index (SI) was calculated that is a result of dividing the average cpms of every donor to the average control cpms of the corresponding donor (Fig. 5). Control cpm was a result for the control probe that was lymphocytes without stimulation. A SI greater than 2 was considered as a max response. SI levels between 1.55 and 2 were considered as a medium response; SI levels below 1.55 were considered as a minimum response.
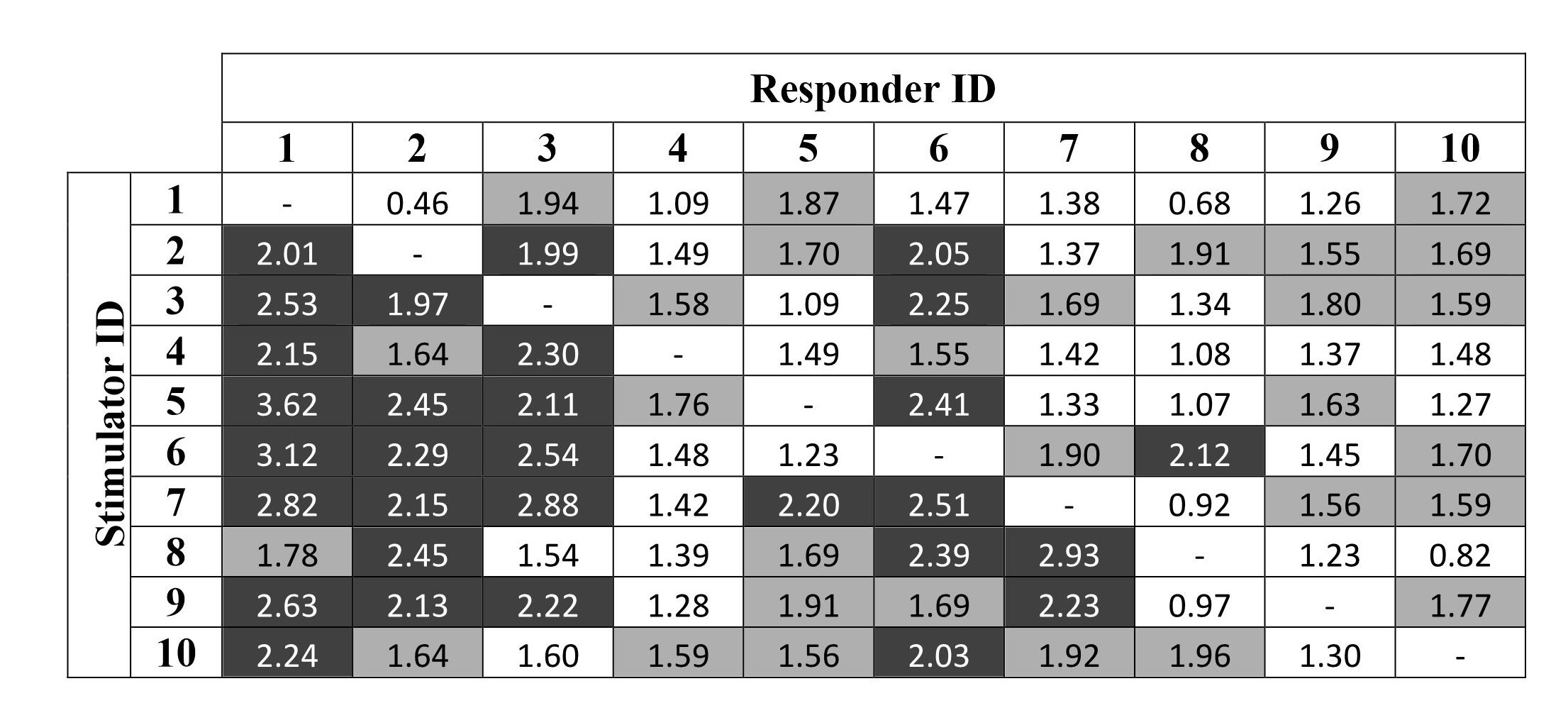 Fig. 5.
Fig. 5.Proliferative response in 90 stimulator–responder pairs (mixed
lymphocyte culture): results of H3-Thymidine incorporation assay. The data are
presented as stimulation index donor (SI = mean CPM of test wells/mean CPM of
control wells). The SIs for each pair of donors (90 pairs) were sorted in
descending order and divided into three groups: maximal (30 responses, SI
To identify which HLA genotypes were most active regarding the stimulation and response to alloantigens, genotype frequencies were determined in the first third of the values, and to determine the weakest genotypes in MLC, the genotype frequencies were determined in the last third of the values (Tables 1,2). Thus, we selected the best responder genotypes (ID 1, 2, and 3) and the best stimulator genotypes (ID 6 and 7). They formed potential responder–stimulator pairs: 1–7, 1–6, 2–7, 2–6, and 3–6, the DNA sequences of which were further used for the calculation and analysis of antigenic epitopes and subsequent synthesis of DNA constructs.
| Responder ID | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| Frequency of max. responses, counts | 8 | 6 | 6 | 0 | 1 | 6 | 2 | 1 | 0 | 0 |
| Frequency of max. responses, % | 26.7 | 20.0 | 20.0 | 0.0 | 3.3 | 20.0 | 6.7 | 3.3 | 0.0 | 0.0 |
| Frequency of min. responses, counts | 0 | 1 | 2 | 6 | 3 | 1 | 4 | 6 | 5 | 3 |
| Frequency of min. responses, % | 0.0 | 3.3 | 6.7 | 20.0 | 10.0 | 3.3 | 13.3 | 20.0 | 16.7 | 10.0 |
| Stimulator ID | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| Frequency of max. responses, counts | 0 | 3 | 3 | 2 | 4 | 4 | 5 | 3 | 4 | 2 |
| Frequency of max. responses, % | 0.0 | 10.0 | 10.0 | 6,7 | 13.3 | 13,3 | 16.7 | 10.0 | 13,3 | 6.7 |
| Frequency of min. responses, counts | 5 | 2 | 2 | 5 | 3 | 3 | 2 | 4 | 3 | 4 |
| Frequency of min. responses, % | 16,7 | 6,7 | 6,7 | 16,7 | 10.0 | 10.0 | 6,7 | 13.3 | 10.0 | 13.3 |
The results of DNA sequencing are presented as laboratory conclusions on the donor HLA genotype (summarized in Table 3) and as FASTQ data files from an Illumina MiSeq sequencer. The DNA sequence data were used to develop a software product, analyze sequences, and select epitopes for incorporation into genetic constructs.
| ID | HLA genotype | ||||||||||
| A | B | C | DPA1 | DPB1 | DQA1 | DQB 1 | DRB1 | DRB3 | DRB4 | DRB5 | |
| 1 | 01:01, 02:01 | 07:02, 18:01 | 07:01, 07:02 | 01:03 | 03:01, 04:01 (*03:01/124:01, 04:01/350:01) | 01:02, 05:05 | 03:01, 06:02 | 15:01, 11:04 | 02:02 | —– | 01:01 |
| 2 | 11:01, 30:01 | 13:02, 35:01 | 04:01, 06:02 | 01:03 | 03:01, 04:01 (*03:01/124:01, 04:01/350:01) | 01:02, 01:04 | 05:03, 06:04 | 13:02, 14:54 | 02:02, 03:01 | —– | —– |
| 3 | 02:01, 29:02 | 39:01, 45:01 | 06:02, 12:03 | 01:03 | 02:01, 04:01 | 01:02, 05:05 | 03:01, 05:02 | 16:01, 11:01 | 02:02 | —– | 02:02 |
| 4 | 23:01, 25:01 | 35:01, 41:01 | 04:01, 07:01 | 01:03, 02:01 | 04:01, 13:01 (*13:01/107:01) | 01:01, 01:02 | 05:01, 06:04 | 01:01, 13:02 | 03:01 | —– | —– |
| 5 | 02:01, 23:01 | 08:01, 49:01 | 07:01 | 01:03 | 04:01 | 01:02, 05:01 | 02:01, 06:02 | 15:01, 03:01 | 01:01 | —– | 01:01 |
| 6 | 02:01, 03:01 | 15:01, 51:01 | 03:04, 15:02 | 01:03 | 03:01 | 01:03, 03:02 | 03:03, 06:03 | 13:01, 09:01 | 02:02 | 01:03 | —– |
| 7 | 02:01, 26:01 | 35:01, 58:01 | 04:01, 07:18 | 01:03 | 04:01, 04:02 (*04:01/126:01, 04:02/105:01) | 01:01, 01:02 | 05:01, 05:02 | 01:01, 16:01 | —– | —– | 02:02 |
| 8 | 01:01, 11:01 | 08:01, 35:02 | 04:01, 07:01 | 01:03, 02:01 | 04:01, 10:01 | 05:01, 05:05 | 02:01, 03:01 | 03:01, 11:04 | 01:01, 02:02 | —– | —– |
| 9 | 24:02, 26:01 | 27:14, 35:03 (*35:03/279) | 01:02, 04:01 | 01:03, 02:02 | 03:01, 13:01 (*13:01/107:01) | 01:01, 01:03 | 05:01, 06:03 | 01:01, 13:01 | 01:01 | —– | —– |
| 10 | 03:01 | 51:01, 56:01 | 01:02 | 01:03 | 04:01 | 01:01, 01:02 | 05:01, 06:02 | 01:01, 15:01 | —– | —– | 01:01 |
| —– – not identified genotype. * – possible genotype variant. | |||||||||||
A software product was developed for the generation of constructs, which enabled the personalized selection of major histocompatibility complex epitopes chosen based on a comprehensive analysis of HLA sequencing of the patient and the donor, and the theoretical design and synthesis of personalized DNA constructs for their transfection into dendritic cells to suppress the graft rejection reaction were performed. Then, artificial genes encoding target constructs were designed, the artificial genes were cloned into the pmax-Ub vector, and genetic constructs encoding target protein products were generated, followed by the amplification of genetic constructs encoding target protein products and their purification from endotoxins. Thus, personalized DNA constructs were generated for five responder–stimulant (recipient–donor) pairs, based on these sequences of the major histocompatibility complex.
Responder dendritic cells were transfected with: (1) DNA plasmids encoding
epitopes of stimulator histocompatibility complex molecules; (2) an
IL-10-encoding plasmid to induce nonspecific tolerance; and (3) two plasmids
simultaneously. After 96 h, significantly higher FoxP3 expression in
CD4
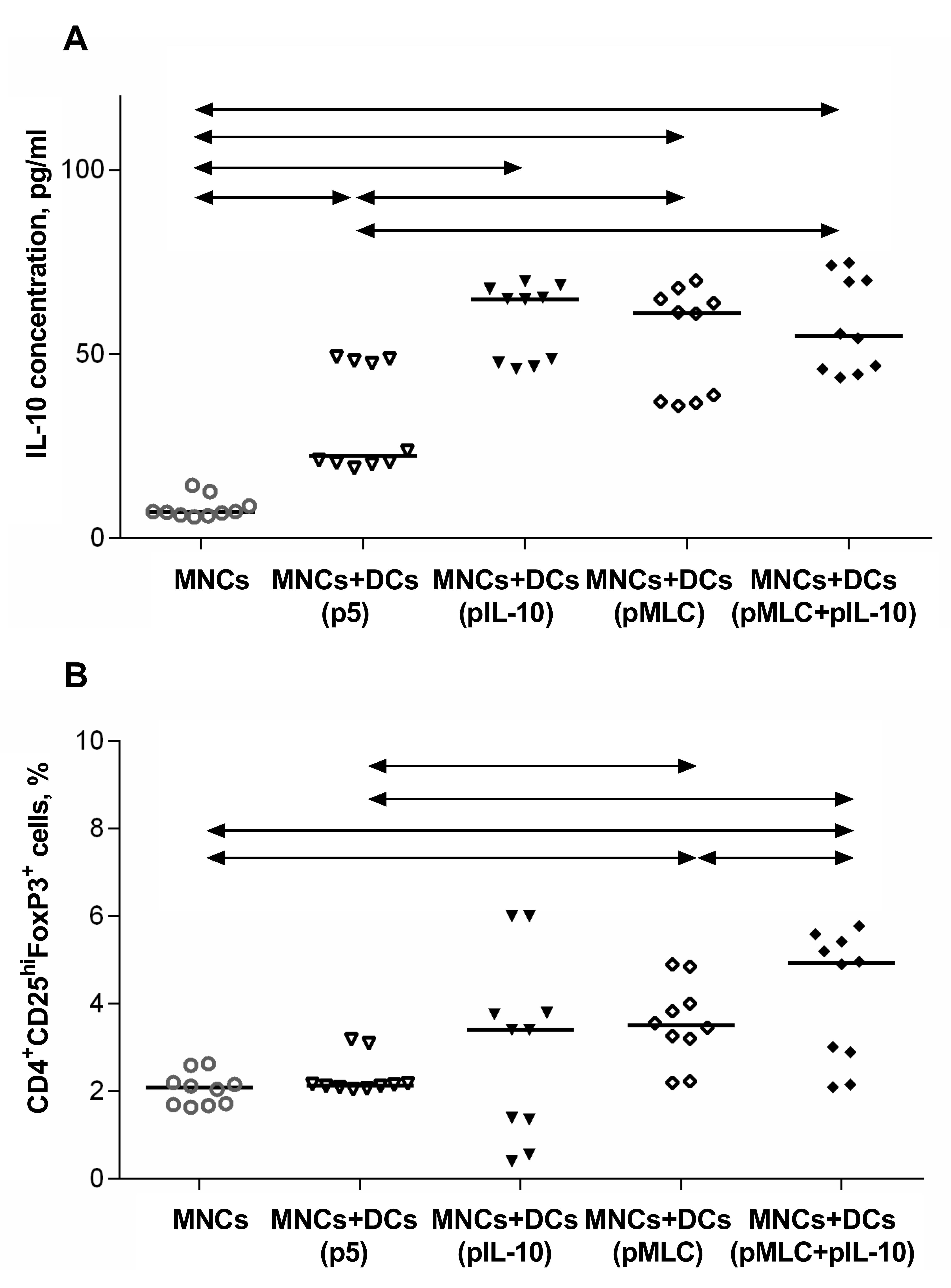 Fig. 6.
Fig. 6.Tolerogenic properties of pIL-10- and pMLC-transfected cells.
Production of IL-10 (A) and a relative number of
CD
To assess the ability of cultures of transfected DCs and MNCs (MNCs+DCs) to restrict the activation of autologous MNCs in response to stimulation by MHC alloantigens, mixed lymphocyte cultures were performed. The cultures of mononuclear cells from responder donors were co-cultured with MNCs from stimulator donors pretreated with mitomycin C. After 96 h of co-cultivation, the proliferation level in cultures was evaluated, and the proliferation suppression index was calculated ([proliferation of MNC culture without allogeneic stimulation]/[proliferation of culture upon addition of mitomycin C-treated allogeneic MNCs from stimulator donors]). Therefore, the higher the suppression index, the lower the proliferative response of the culture to stimulation with alloantigens. The results showed that cultures generated using tolerogenic DCs transfected with MHC epitopes had a significantly greater ability to inhibit the proliferation of autologous MNCs in response to an allogeneic MHC stimulus. The results are presented in Fig. 7.
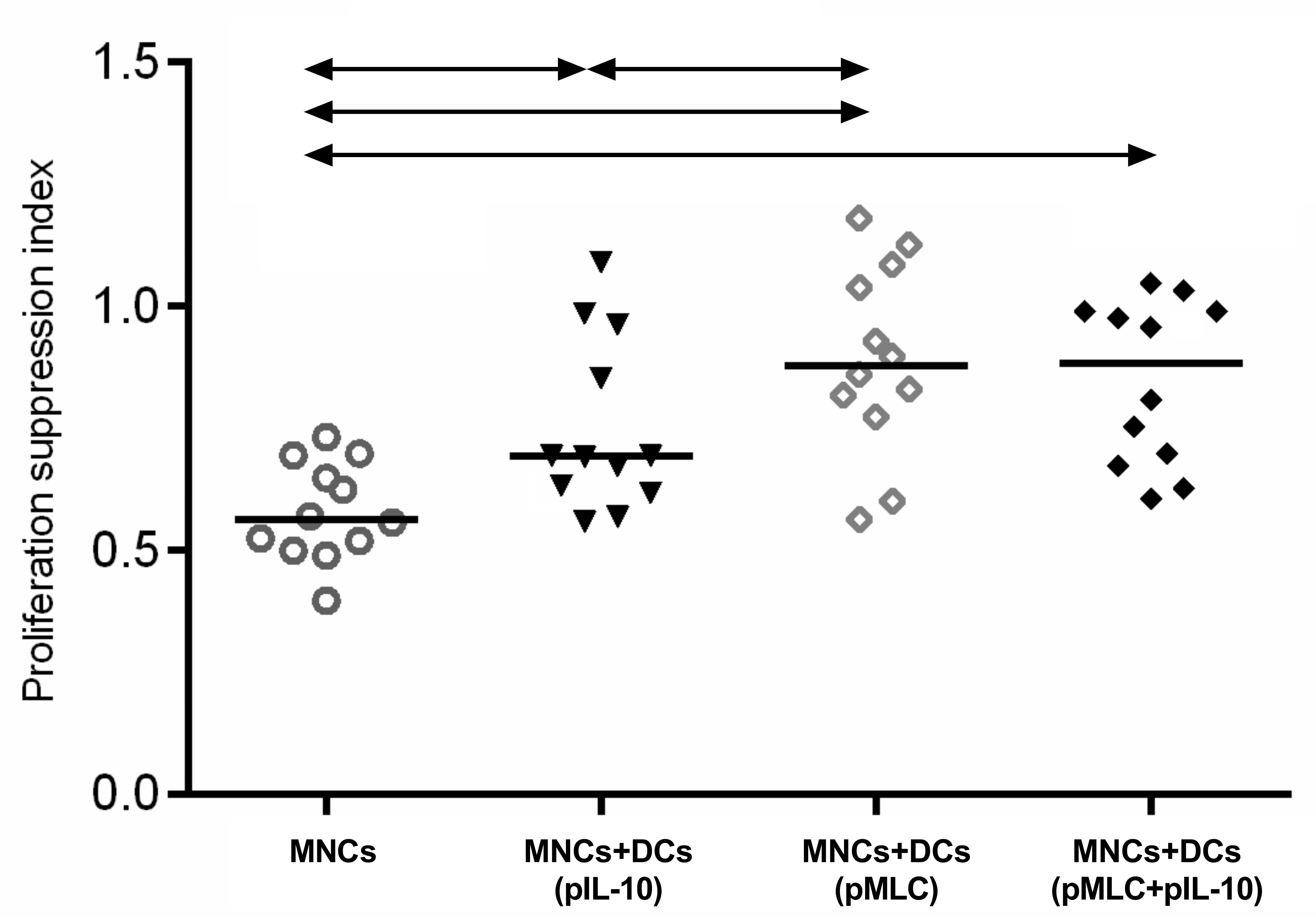 Fig. 7.
Fig. 7.Suppression of proliferation in allogeneic mixed lymphocyte
cultures (MLC) using tolerogenic human dendritic cells transfected with
personalized human DNA constructs encoding HLA epitopes of stimulators. MNCs –
responder mononuclear cells. Data are presented as median and individual values.
The arrows indicate statistically significant differences, p
Dendritic cells are a heterogeneous population classified in humans into myeloid DCs, plasmacytoid DCs, and Langerhans cells, based on ontogenesis, phenotype, and functionality [20, 21]. Mature dendritic cells mediate immune responses in inflammatory conditions, while immune tolerance is induced by tolerogenic DCs with an immature phenotype.
The development of antigen-specific tolerance, especially for donor antigens, is an urgent problem of transplantology, the solution of which might reduce the immunosuppressive drug load and toxic effects on recipients as well as increase the overall survival of patients after transplantation [22, 23].
The HLA locus is the most polymorphic region in the human genome and is associated with various diseases. HLA matching is a key factor in improving patient outcomes after transplantation. HLA matching has traditionally focused on HLA-A, -B, and -DR loci in solid organ transplantation and on HLA-A, -B, -C, and -DRB1 loci in hematopoietic cell transplantation [24]. Because of the high diversity of HLA haplotypes, the search for a suitable donor usually begins with close relatives, in particular siblings. However, ideal donors are found only for 15%–30% of recipients, and unrelated donors are often involved in the search. Previously, matching for HLA-A, -B, -C, and -DRB1 (8/8) alleles was demonstrated to correlate with better survival rates, while mismatches were associated with an increased risk of acute GVHD and patient death [24]. Single -DQ mismatches probably have no significant effect on the outcome of transplantation, whereas -DP mismatches slightly increased the risk of acute GVHD without compromising overall survival [25].
Based on the mixed lymphocyte culture, donor pairs with a maximum response to alloantigens and donors whose cells maximally stimulated responses to alloantigens, were identified. Among these donors, stimulator and responder pairs with a maximum difference in HLA loci were selected. These donor and recipient haplotypes would normally have an extremely high risk of transplant rejection in the donor during the transplantation of solid organs or hematopoietic cells. A software product for the personalized selection of HLA epitopes was developed, which provides a comprehensive analysis of the recipient and donor HLA sequences, selection and comparison of main histocompatibility antigens of the donor and recipient, and, based on the selected different antigens, the selection of epitopes of these antigens for the generation of a personalized DNA construct. The generated DNA construct should ensure the recipient’s tolerance to donor cells; in this case, there was no direct contact with allogeneic material, which reduced the risk of infection and uncontrolled composition (lysates or vesicles).
Various agents (IL-10, vitamin D3, dexamethasone and rapamycin) have been used to induce sufficient numbers of ex vivo tolerogenic DCs for the effective suppression of GVHD [26, 27]. These DCs provide tolerance in culture without reference to a specific antigen. In this study, we compared the effects of DNA constructs on the development of nonspecific immunosuppression (DNA construct encoding IL-10) and antigen-specific tolerance (DNA constructs encoding major histocompatibility complex antigens), as well as their joint action. The stage of interaction with mononuclear cells in vitro suggests that tolerogenic DCs do not become immunogenic after their introduction in vivo. Using five genetic constructs (three responders and two stimulators), respondent tolerogenic DCs transfected with epitopes of the stimulator main histocompatibility complex decreased the proliferative activity of recipient cells in response to alloantigen (by almost 50%). In this case, the use of a construct encoding only alloantigens without the use of IL-10 suppressed proliferation in stimulator cells, and the maximum suppression of proliferation in this group was observed when cells from stimulator No. 6 were used. An analysis of IL-10 production in co-cultures of MNCs and DCs transfected with plasmids encoding IL-10 and major histocompatibility complex antigens revealed a similar level of cytokine production in all groups, regardless of the antigen specificity. An analysis of the T-regulatory cell number showed that only the simultaneous application of the IL-10-encoding plasmid and the plasmid encoding major histocompatibility complex antigens led to a significant increase in target cells compared with groups where only one of the plasmids was used. Tolerogenic DCs control the differentiation of T-regulatory cells that, in turn, modulate the phenotype and function of DCs. This feedback between tolerogenic DCs and T-regulatory cells is supported by the fact that weak, if any, CD80/CD86 co-stimulation is required to induce Foxp3+ T-regulatory cells, and that IL-10 and TGF-beta secreted by T-regulatory cells inhibit DC maturation and promote the maintenance of DCs in the tolerogenic state [28]. In our study, transfection of DCs with the IL-10-encoding plasmid led to the production of the target cytokine after 3 h and an increase during the first days after transfection. In this case, there was a decrease in the expression of costimulatory molecules CD83 and CD86, an increase in the number of T-regulatory cells, and suppression of mononuclear cell proliferation. Furthermore, IL-10 production by transfected DCs slightly exceeded the reference values provided by the manufacturer of kits for PHA-stimulated cultures, which indicates the stimulation of cytokine production within physiological levels without a potential destructive effect on organs and tissues of the potential recipient.
Therefore, the produced dendritic cells exhibited tolerogenic properties and might be used to develop antigen-specific tolerance. Since the formation of tolerogenic properties is not associated with a long and high-affinity interaction between DCs and T cells and actively involves feedback mechanisms, the use of a plasmid encoding MHC antigens and the absence of maturing stimuli leads to the development of tolerogenic DCs and to the manifestation of effects at a level comparable with the use of a construct encoding IL-10. Thus, the co-application of plasmids encoding IL-10 and MHC antigens does not lead to a dramatic change in the studied effects. On the basis of this study, we think that the generation of DNA constructs enabling antigen-specific tolerance is no less effective than constructs encoding only certain immunosuppression factors. Depending on the vector and promoter used, it is possible to generate a DNA construct with a very high production of the target cytokine, cell death, and immune response suppression caused by the hyperproduction of IL-10 or TGF-beta. Therefore, DNA constructs encoding antigen-specific epitopes will be free from this disadvantage.
In our previous study [19] using an in vivo mouse model, we reported the suppression of GVHD reactions in an allogeneic transplantation model and better engraftment of a transplanted allogeneic skin flap using nonspecific and specific DNA constructs encoding IL-10 or MHC antigens, respectively. In this study, we demonstrated that the effectiveness of antigen-specific DNA constructs encoding histocompatibility complex antigens from a donor can be preliminarily assessed in vitro, which, together with the data on in vivo use suggests the potential clinical use of this method. A preparation of transfected DCs and mononuclear cells can be produced before surgery or lymphadenopathy tested, and then used during further treatment. The main application of these DNA constructs is the transplantation of bone marrow, kidneys, and liver from a living donor, i.e., in cases where the donor is known and available, and surgery is elective rather than urgent.
We demonstrated in vitro that human dendritic cells transfected with DNA constructs against HLA stimulating epitopes exhibited tolerogenic properties and may be used to suppress proliferative activity against allogeneic cells. Thus, we proposed a perspective approach to the induction of antigen-specific tolerance, which should be further studied for possible use in clinical practice to prevent GVHD reactions and graft rejection.
DC, dendritric cells; GVHD, graft versus host disease; HLA, human leukocyte antigen; MHC, major histocompatibility complex; MNC, mononuclear cells (non-adherent PBMC fraction); PBMC, peripheral blood mononuclear cells.
JAS contributed to the experimental work (MLC assays and experiments with pIL-10-transfected DCs), data analysis and interpretation, and drafting the manuscript. JAL—experimental work (MLC assay to identifying the best stimulator-responder matches), JNK—experimental work (DC transfection), ANS—interpretation of the DNA sequencing data and calculations for the determination of the most active stimulator-responder pairs. MSK—data interpretation, drafting the manuscript, figure preparation. VVK—experimental work (flow cytometry). AZM—designed and produced plasmid DNA constructs. SVS—contributed to the conception, design, revision, and final approval of the manuscript.
Voluntary informed consent was obtained from all donors participated in the research. The study followed the principles outlined in the Declaration of Helsinki for all human and animal experimental investigations and was approved by the local Research Institute of Fundamental and Clinical Immunology (RIFCI) ethics committee (protocol №127 dated Feb 17, 2020).
The authors are grateful to the staff of the Dmitry Rogachev National Research Center of Pediatric Hematology, Oncology, and Immunology of the Ministry of Health of the Russian Federation (Moscow, Russia) for performing the DNA sequencing and to the Laboratory of molecular immunology, RIFCI, for all assistance provided. We thank Edanz Group (www.edanzediting.com/ac) for editing a draft of this manuscript.
The research (MLC, culture work, functional assays) was conducted under the framework of State Assignment № 1021062512015-4 (FGMN-2021-0003). The steps of DNA transduction and HLA-A genotyping were sponsored by the grant of the Russian Science Foundation project no. 21-65-00004, https://rscf.ru/project/21-65-00004/.
The authors declare no conflict of interest. SS is serving as one of the Editorial Board members of this journal. We declare that SS had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to GP.







