1 Universidade Católica Portuguesa, CBQF - Centro de Biotecnologia e Química Fina – Laboratório Associado, Escola Superior de Biotecnologia, 4169-005 Porto, Portugal
Abstract
Oral diseases affect over three billion people worldwide, making it one of the most common infections. Recent studies show that one approach to reducing the risk of chronic infections, such as caries, gingivitis, periodontitis, and halitosis, is to control the ecology of the oral microbiome instead of completely removing both the harmful and beneficial microorganisms. This is based on the knowledge that oral diseases are not caused by a single pathogen but rather by a shift in the homeostasis of the entire microbiota, a process known as dysbiosis. Consequently, it is of the utmost importance to implement strategies that are able to prevent and control oral dysbiosis to avoid serious complications, including heart, lung, and other systemic diseases. Conventional treatments include the use of antibiotics, which further disrupt the equilibrium in the oral microbiota, together with the mechanical removal of the decayed cavity area following its formation. Therefore, it is imperative to implement alternative strategies with the potential to overcome the disadvantages of the current therapy, namely, the use of broad-spectrum antibiotics. In this sense, probiotics and postbiotics have received particular attention since they can modulate the oral microbiota and decrease the dysbiotic rate in the oral cavity. However, their mechanisms of action need to be addressed to clarify and drive their possible applications as preventive strategies. In this sense, this review provides an overview of the potential of probiotics and postbiotics, focusing on their antimicrobial and antibiofilm activities as well as their ability to modulate the inflammatory response. Finally, it also showcases the main advantages and disadvantages of orodispersible films—a promising delivery mechanism for both probiotics and postbiotics to target oral dysbiosis.
Keywords
- oral dysbiosis
- probiotics
- postbiotics
- oral health
- Lactobacillus spp.
- Streptococcus mutans
- orodispersible films
Dental caries represents the most predominant infection worldwide among oral diseases, with more than 3.5 billion people experiencing it at least once in their lifetime [1].
In addition to impacting the oral cavity, oral health is also highly related to general health [2]. Since 1989, with the study by Mattila et al. [3], it has been known that there is a link between insufficient oral health and heart problems. Moreover, complications during pregnancy have also been linked with chronic periodontitis, as well as chronic obstructive pulmonary disease and bone resorption [4]. Even gastric diseases, namely inflammatory bowel disease, are related to an unbalanced oral microbiota [2, 4]. Additionally, a potential link has been observed between oral infections, namely, periodontitis, and neurodegenerative diseases, such as Alzheimer’s disease or multiple sclerosis [5].
In the work by Thomas and collaborators, it was clear that diseases with an inflammatory base cannot be treated nor alleviated in the presence of oral dysbiosis, which is particularly relevant for patients with diabetes [2]. Another study by Lamont et al. [6] showed that an unhealthy oral cavity is related to a decline in the state of general illness, which is partially caused by the presence of lipopolysaccharides (LPSs), an endotoxin from gram-negative bacteria that can easily enter the blood circulation and reach different organs [2].
Despite being related to different systems, such as the cardiac and respiratory systems, oral health, specifically gum health, is also intrinsically connected to psychological well-being [7]. In fact, oral health highly depends on the equilibrium of the oral microbiota, also called oral biota. Such microbiota, when balanced, has the function of protecting the oral cavity [8, 9]. However, oral diseases, namely caries, appear when this community shifts and dysbiosis occurs [9, 10]. In addition to caries, periodontitis is the second most prevalent dental problem and represents an inflammatory disease that can lead to the loss of teeth and support tissues [11, 12].
The oral microbiota comprises more than seven hundred species of bacteria, making it one of the most complex populations in the human body [13]. For example, Loesche observed that the first colonizers, Streptococcus mutans and Streptococcus sobrinus, appeared as soon as the teeth started erupting [14]. Although they are highly associated with dental illness, they are also colonizers of the oral microbiota [15]. The importance of these early colonizers is not only in preventing oral diseases but also in modulating the immune system of the host [16].
However, in dysbiosis, some members of the microbiota begin to overgrow, which formulates the beginning of biofilms. This is based on the ecological plaque hypothesis, which states there are no specific microorganisms responsible for oral disease but instead follows the overgrowth of some species in the oral biota. In this case, the treatment should focus on regulating and controlling the environment that causes the microbiome shift, not on antimicrobial therapy [17, 18, 19].
The main bacteria found in dental biofilms are Streptococcus, Actinomyces, Prevotella, Porphyromonas, Tannerella, and Fusobacterium spp. [8]. However, only a few are associated with periodontitis, mainly gram-negative and anaerobic bacteria [17], such as Aggregatibacter actinomycetemcomitans, Fusobacterium nucleatum, and Porphyromonas gingivalis [20]. The bacteria in the biofilm synthesize a matrix that is rich in glucans and exopolysaccharides (EPS), thereby allowing for better adherence to both one another and the surrounding tissues [10]. Other vital compounds in biofilms are endotoxins, namely LPS, which trigger an inflammatory response in the host. The characteristics of the biofilm are also protective and do not allow chemical agents to penetrate the microbiome barrier, thus leading to higher antibiotic resistance [12]. Another crucial factor in maintaining oral health is diet [16]. Cleaver et al. [21] showed that the amount of carbohydrates ingested is related to acid production, providing an adequate environment for the growth of cariogenic microorganisms. The mechanical removal of the lesion and/or dental plaque is the standard treatment used to treat cavities and gingival inflammation. However, this means the removal of both beneficial and harmful microbiota, which increases microbial imbalance as a consequence. Furthermore, removing the entire microbiome offers dental pathogens more free colonization sites [22]. In addition, bacteria in biofilms are usually more resistant than in the planktonic state, thereby making antibiotherapy an inadequate choice [10]. Furthermore, the consumption of antibiotics can lead to an even more notable dysbiosis, to a point where it can become irreversible [19].
Recent studies have focused on preventive treatments that are capable of re-establishing the oral biota equilibrium, modulating the microbiome, and presenting a positive effect on inflammation [8, 23]. Taking this into consideration, probiotics, prebiotics, and postbiotics represent promising strategies that also consider consumer awareness of health [1, 24, 25].
The World Health Organization defines probiotics as “live microorganisms that, when administered in adequate amounts, confer a health benefit to the host” [26]. They can have a positive effect on the microflora [27], control the inflammatory response [28], modulate the innate and adaptative immune responses, and inhibit the production of cytokines by pathogens [24]. It has also been shown that probiotics stimulate immunoglobulin production and alter the DNA of the host [29]. Moreover, they do not cause any side effects experienced following antimicrobial therapy, meaning they can be considered an alternative to this treatment [30]. Thus, since probiotics are able to modulate both the inflammatory response [28, 31] and the microbiota, there has been increased attention toward their use as an adjunctive therapy because both are critical properties for the prevention of dental caries and other diseases [27, 32].
Although it is also important to remember that probiotics cannot substitute primary oral care and hygiene practices, they are now an exciting choice for a preventive approach [31]. However, this therapy will not replace conventional treatment, even though it shows better results than the preventive strategies used nowadays.
In addition to probiotics, the current knowledge concerning pre- and postbiotics has been increasing recently, with more research mainly focusing on the functions of cell-free supernatants and even non-living bacteria to circumvent the disadvantages and limitations associated with using probiotics [33, 34].
This review aimed to deeply understand the use of advanced approaches to oral health prevention, to avoid the use of antibiotics, and to control oral dysbiosis while in a reversible state, showing that novel strategies demonstrate beneficial properties with an interesting outcome in terms of health care.
The oral biota can be affected by several factors: dietary habits, type of dentition, medication, age, and general behaviors of the individual [2, 16, 35, 36]. Regarding general behaviors, it was noted that dietary habits, namely, carbohydrate intake, are of the utmost importance to define the type of bacteria that inhabit the oral cavity. The higher the ingestion of considerable amounts of carbohydrates, the more negatively affected the oral microbiota is [1].
An interesting fact about the oral cavity is that it cannot be perceived as a whole; instead, it should be viewed as a set of different microenvironments with distinctive characteristics that reach equilibrium and allow different taxa to grow and multiply. According to recent research, it was observed that each specific niche has a separate set of microorganisms [37, 38, 39]. This is presumably due to the different microenvironments that determine different conditions across the oral cavity, such as the temperature or the presence of oxygen. However, it can also relate to the tissue type and the presence of nutrients. For example, the buccal mucosa presents minimal diversity, while the tongue and the dental surfaces show elevated levels of microbial communities [16]. It should be noted that when daily oral hygiene is poor, there is an increase in the number and diversity of microorganisms present in the oral cavity [40, 41].
Despite presenting a high diversity of microorganisms, certain genera, such as Streptococcus, Prevotella, Fusobacterium, and Veillonella, are more commonly found in a healthy oral cavity. Other commensal microorganisms, such as viruses, archaea, fungi, and protozoa have also been found among the oral biota. Similar to processes in other commensal communities, all microorganisms perform distinct roles when inhabiting a host, meaning they all contribute to the organization and survival of each other [16, 42].
In a healthy environment, the commensal bacteria in the mouth symbiotically cohabitate with the host. This means that an equilibrium is reached between not only the bacteria and the individual’s immune system but also between the commensals themselves. This equilibrium is called homeostasis and forms an essential characteristic of the healthy oralome [1, 2, 18]. It is important to understand that maintaining oral homeostasis is a multifactorial process that mainly depends on the host and the microenvironments created in the different sites of the oral cavity, although it also depends on the bacteria and other microorganisms present [1].
The microorganisms in the oral cavity are acquired as soon as the individual is born and start changing over time. When the teeth erupt, new structures allow different microorganisms to inhabit the oral cavity: the enamel and the gingival sulcus. However, Kaan and collaborators demonstrated that a major microbiological shift occurs as consumed food changes from liquid to solid [16].
The acquisition of specific bacteria seems to be highly dependent on the oralome of the mother. In fact, it was noted that if children’s caretakers harbored certain species, such as S. mutans, S. sobrinus, Fusobacterium nucleatum, Prevotella intermedia, and P. gingivalis, there was a high chance that those species would also be found in the children under their care. This relation was particularly relevant at the time of the retrieval of anaerobes in the oral cavity [16]. When in homeostasis, the commensals of the oral cavity promote oral health and, as a result, general health. These microorganisms present a protective capacity against pathogens by competing for nutrients or binding sites [1, 18].
Regarding the microorganisms that inhabit the oral cavity as commensals, it can be noted that Streptococci are the main oral colonizers, namely, Streptococcus mitis, Streptococcus salivarius, and Streptococcus cristatus, among other species. The emergence of these microorganisms in the oral cavity occurs as soon as the eruption of the first teeth, mainly due to the adherence ability of Streptococci to the surfaces of the teeth, tongue, and gingiva. They also allow other bacteria to bind to them and, thus, become part of the oral microbiota, even if they do not possess the capacity to adhere to dental surfaces or other tissues themselves [1].
Due to the presence of the early colonizers, their metabolites, and excreted products, the environment becomes suitable for the growth of other bacteria. In this sense, new anaerobic sites emerge as the microorganisms start arranging themselves [16].
As the early colonizers start changing the environment, which allows other bacteria to start inhabiting the oral cavity, major changes occur regarding the present bacteria. This is called the “permanent colonization” of the mouth and is responsible for creating the “core taxa” of the oral microbiota, mainly due to the adherence ability of Streptococci, which operates as a bridge between the dental surfaces and other bacteria [42].
Similar to the processes in other areas of the human body, the commensals present beneficial properties for the host: Streptococci species, namely, S. mitis, S. sanguinis, and S. cristatus, prevent the adhesion of pathogens, such as P. gingivalis, via an enzyme called arginine deaminase, which affects the production of binding proteins. They also can counteract acid production from cariogenic bacteria, such as S. mutans, thereby increasing the salivary pH [1].
It is important to understand that commensal microorganisms can also reduce nitrate, which prevents the occurrence of caries. Nitrate-reducing bacteria present the capacity to produce ammonia, which, in turn, and similarly to other commensals mentioned above, raises the pH of the oral cavity, causing it to function as a buffer for acid production from S. mutans. Furthermore, they can decrease the number of anaerobes due to the presence of nitrate, nutrient competition, and binding site competition. This is important since anaerobes are mostly responsible for periodontal diseases and halitosis. Relevant nitrate-reducing bacteria belong to the genus Actinomyces and Kingella, among others, and are usually present in individuals who exhibit good oral health [43].
In addition to homeostasis and the action of commensals in the equilibrium of the oral cavity, another critical factor for maintaining oral health is the presence of saliva since it presents several distinct functions, namely, the removal of microorganisms, which could be potentially pathogenic, the transference of microorganisms from one location to another, facilitation of the commensal colonization, and its buffer capacity, all of which are critical for controlling against caries [2, 16, 18].
Regarding the buffering ability, it should be mentioned that saliva influences the microorganisms that can survive in the biofilm since it can uphold a pH level of 6.5–7.5, which is neutral [18, 30]. The most critical consequence of pH maintenance is the remineralization of enamel, which prevents the formation of cavities by acidogenic bacteria [1, 44]. Saliva also possesses immunoglobulins in its composition, thereby allowing it to serve as a limitation for bacterial growth since it presents an antimicrobial capacity. However, this property is not visible when homeostasis is not maintained [2, 16, 18].
Finally, it is crucial to understand that similar to other microbiotas in the human body, oral bacteria can also act systemically in the individual. For this reason, some researchers noted that it serves as an indicator of overall health, an activator of the host’s immune system, and, on the downside, a source of systemic inflammation when homeostasis is lost [2].
As mentioned above, the oral microbiota is a community of microorganisms that inhabit the oral cavity. When they co-habit in homeostasis, they can prevent disease and maintain a certain environment that is suitable for the survival of the commensals [1].
The oral microbiota presents a certain level of stability; nonetheless, certain circumstances, such as a modification in dietary habits or the host’s ability to interact with the commensals, disrupt the oral equilibrium and alter the environment, resulting in the loss of homeostasis, called dysbiosis. A relationship between oral dysbiosis and the appearance of oral diseases, such as caries, periodontitis, and gingivitis, has already been established. In addition, dysbiosis also impairs the host’s general health by affecting the cardiovascular system [19].
The loss of homeostasis is multifactorial, whereby it can be host-related, while it can also occur from the use of antibiotics. In this case, it is called chemical-induced environmental dysbiosis [18]. Another underlying cause of dysbiosis is an insufficient salivary flow, which results in the deficient removal of microorganisms, contributing to biofilm maturation [2].
Dysbiosis results in some species growing uncontrollably, causing deep
alterations in the oral microbiota, ultimately resulting in a loss of oral and
general health [1, 2, 18]. The systemic implications are mainly due to changes in
the epithelial barriers in the oral cavity, which occur in a dysbiotic
environment due to the production of proinflammatory cytokines, such as tumor
necrosis factor
This shift in the environment, i.e., the dysbiosis per se, usually improves the growth capacity of certain bacteria, namely species belonging to the genera Streptococcus, Actinomyces, and Lactobacillus, whose metabolic activity creates an anaerobic environment. Eventually, this allows anaerobic bacteria, such as P. gingivalis, Treponema denticola, and Aggregatibacter actinomycetemcomitans, which are usually pathogenic, to grow due to their ability to produce acids. The production of acid results in an enhanced demineralization process of the dental enamel, resulting in a cavity [1]. Furthermore, it also creates conditions for the overgrowth of periodontopathogens since the formation of a biofilm in the teeth can expand to subgingival areas [45].
An important aspect of dysbiosis in the oral cavity is that environmental
alterations shape the type of microorganisms that can grow due to the creation of
specific conditions [2]. One particular characteristic is the reduction in the
levels of H
The following segments provide an overview of the main mechanisms that lead to dysbiosis, its correlation with oral diseases, and the major pathobionts and conventional treatment options.
It is essential to understand that the bacteria in the oral cavity are organized in a multidimensional structure, predominantly known as a biofilm composed primarily of EPS. Since the oral cavity presents many challenging characteristics for bacteria to survive, their organization in biofilms is their most efficient survival mechanism [1, 4, 47, 48].
The biofilm allows the bacteria to interact with each other through quorum sensing and gene regulation, reaching an important level of organization and complexity by co-aggregating; thus becoming less prone to removal [2, 16, 42, 47]. As the biofilm starts to form and the environment changes, there is a loss in diversity in the microbiota, which contributes to dysbiosis. This loss in diversity is not limited to bacteria but also fungi and other microorganisms [16].
One of the reasons bacteria in biofilms present a higher resistance to antibiotics may be the physical impossibility of the antibiotic molecule reaching the deeper levels of the biofilm. In addition, differences in the environment, such as the reduced pH, can alter the actions of antibiotics. Moreover, the capacity for the horizontal transfer of genes allows the microorganisms to quickly acquire resistance genes, rapidly becoming resistant to the previously effective antibiotics [46].
The ability to adhere is the main property of bacteria found in the biofilm, although this depends on the presence of a receptor and the adhesin protein [16, 48]. The biofilm formation shows progressive development, beginning with reversible adherence, then irreversible adherence, and consequent maturation. As the biofilm reaches the mature state, an equilibrium is formed between the microorganisms involved [1, 2]. When the biofilm reaches its mature state, it is known as dental plaque [2].
However, to avoid deep dysbiosis in the oral cavity, the biofilm must be removed before maturation. Nonetheless, it should be noted that it is challenging to eliminate the biofilm after a state of irreversible attachment [49].
A couple of hours after oral hygiene, the teeth become covered in a thin pellicle mostly composed of proteins, enzymes, and lipids, called the acquired enamel pellicle or acquired exogenous pellicle (AEP) [16]. The AEP serves mostly as a protective barrier for the teeth, although it can also aid in the adherence of microorganisms to the dental surface, thus initiating biofilm formation. The first colonizers are mostly gram-positive bacteria [2, 18]. In a healthy individual, the components present in the saliva can neutralize the plaque after 30 minutes of carbohydrate consumption. For this reason, a relationship can be noted between the salivary flow and the buffer capacity in an individual [1, 44].
With the ingestion of carbohydrates, more acid metabolites are produced and expelled to the matrix. The AEP becomes more acidic and allows the growth of tolerant microorganisms, favoring its growth instead of other commensals; thus contributing to the state of oral dysbiosis [18].
Specific microorganisms, such as S. gordonii, Streptococcus oralis, Streptococcus mitis, and those belonging to the genera of Actinomycetes can bind to the proteins present in the AEP via the GtfB protein (a glucosyltransferase) and serve as a bridge on which other bacteria can bind, such as species belonging to the genera Fusobacteria, Veillonella, and Rothia [16, 47, 50].
To prevent this, the host’s saliva is rich in antibodies, namely, immunoglobulin A (IgA), which prevent bacterial adhesins from binding to the AEP. However, some bacteria, both pathogenic and commensal, have the ability to cleave IgA through the production of a specific protease, meaning it possesses a colonization advantage [16].
Another important characteristic of the biofilm is the presence of an oxygen gradient, mostly due to the tridimensional organization of the bacteria, which allows for the growth of anaerobic bacteria that, in other circumstances, would not be able to grow [1]. Of note, biofilm formation is not exclusive to a dysbiotic state; bacterial biofilms in a healthy oral cavity can occur when the alkaline compensation is balanced with the acid production [1]. As a matter of fact, dental plaque is considered by many authors as an evolutive microbial community [51]. However, in an unbalanced oral cavity, it is observed that the biofilm promotes the disease and facilitates dysbiosis. For this reason, in an attempt to prevent oral diseases, the biofilm should be one of the first targets of action, especially since the supragingival biofilm is highly correlated to the appearance of periodontitis [1, 52].
Over the last decade, our knowledge of oral diseases has improved drastically, and the idea that certain pathogens were responsible for oral diseases was abandoned. Nowadays, a vast number of researchers accept that the ecological plaque hypothesis can explain the appearance of oral diseases. This hypothesis states that dysbiosis is the main cause of pathobiont growth in the oral cavity and not a specific set of pathogens [18].
Depending on the type of dysbiosis and, consequently, the type of plaque formation, different diseases can appear in the oral cavity [2, 18]. The most common oral disease is caries, followed by gingivitis, periodontitis, peri-implantitis, halitosis, and oral cancer, the only oral disease that does not directly correlate to a dysbiotic state [12].
Regarding caries, biofilm formation tends to increase the development of gram-positive bacteria, which are usually the producers of acidic metabolites that reduce the pH and demineralize the tooth surface, resulting in the appearance of a cavity [1, 2, 53]. The pH limit before the cavity starts to form is 5.5. For this reason, the ingestion of excessive amounts of sugars is considered a risk factor for the development of caries [18, 48]. Curiously, one of the most important risk factors for developing caries is “caries re-experiencing”, which demonstrates the tendency for this disease to appear in certain individuals with a predisposition for caries [54].
When caries appear in children under the age of six, they are named early childhood caries (ECC) and represent one of the most prevalent diseases in this age group worldwide [53]. ECC are closely related to a reduced quality of life and are also considered an expensive burden for families [55]. However, in the preliminary stages, caries is a reversible process consisting solely of enamel loss, meaning it can be prevented without invasive therapies [51, 55].
Another common oral disease is gingivitis, which is called, in a more exacerbated state, periodontitis. Similar to caries, gingivitis is a multifactorial disease that begins with dysbiosis [18, 56]. It is thought that gingivitis worsens with the hormonal change accompanying puberty onset [16]. The major symptom of gingivitis is bleeding, which, in turn, increases the number of blood components, such as erythrocytes, hemin, and fibrin. These molecules represent key factors for specific groups of bacteria, usually existing in dwindling numbers, then rapidly increasing when this imbalance occurs [18]. In addition to blood components, another factor that contributes to the progression of this disease is the presence of inflammatory cytokines that are produced by the immune system of the individual [45].
It is essential to understand that gingivitis itself is a reversible condition and is not responsible for the destruction of gingival tissues. However, if left untreated, it can progress to a more severe condition called periodontitis. This condition is a major cause of tooth loss in adults [7, 35, 45]. Periodontitis is characterized by irreversible damage to the periodontium, the structure responsible for tooth attachment in the oral cavity [2, 35, 57].
The aggravation of dysbiosis and gingivitis results in an accumulation of plaque at the supragingival level. Consequently, the environment becomes deprived of oxygen, which results in the proliferation of gram-negative anaerobic microorganisms. As the pockets get deeper and the carbon source becomes depleted, the environment becomes more propitious for the growth of Treponema denticola, P. gingivalis, Fusobacterium nucleatum, and Veillonella spp., microorganisms, usually found in periodontal dysbiosis [2, 18, 57]. In addition to oxygen and carbon, the pH and temperature of the environment also change, resulting in new microorganisms having the possibility to grow, thereby increasing the diversity in the oral cavity, contrary to what happens in cariogenic dysbiosis [2].
In addition to losing teeth, periodontitis is also responsible for bone reabsorption, leading to additional tooth losses in the neighboring sites [18].
Another important consequence of dysbiosis is oral malodor, also known as halitosis, which is deeply connected to the prevalence of oral diseases, even if it is not associated with any specific infection [58]. Halitosis affects almost 50% of the population and has significant effects on the quality of life, specifically self-esteem and psychological factors [59]. The main cause of halitosis is the plaque that forms on the surfaces of the teeth and the tongue, alongside an increase in gram-negative anaerobic bacteria, which produce volatile sulfur compounds, such as P. gingivalis, Treponema denticola, Prevotella intermedia, and Fusobacterium nucleatum [58, 59, 60].
Furthermore, there is another group of oral diseases called oral mucosal diseases, which affect, as the name suggests, the mucosal tissues in the mouth. This group comprises oral infections, such as oral candidiasis, oral lichen planus, and ulcerative lesions. Despite these diseases being multifactorial, it is also well-established that they result from oral dysbiosis [2]. However, oral mucositis is prevalent in individuals undergoing radiotherapy and chemotherapy treatments, which induce dysbiosis on their own [61].
Another common disease found in the oral cavity is oral candidiasis, which is an opportunistic infection caused by yeasts that belong to the genus Candida and is especially challenging in immunocompromised individuals [62, 63]. It appears as a consequence of oral dysbiosis since this state allows the growth of certain species, such as Candida albicans. However, infections caused by other species of Candida sp. are increasing worldwide since they are more resistant to treatment than C. albicans [62]. Moreover, fungal infections have been increasing recently, possibly due to the considerable use of broad-spectrum antibiotics, among other factors [63].
Definitively, it is important to indicate that certain types of cancer, such as oral squamous cell carcinoma, have been linked to the presence of an oral dysbiotic state, even though it was not demonstrated that it could initiate this disease [19]. In some oral cancers, an imbalance in the number of Streptococci can be found in the oral cavity, whereby they are decreased [2].
Although the worldwide prevalence of oral diseases is increasing, even with more access to general care, there is an urgent need to find new and alternative strategies to prevent this imbalance in the oral cavity from occurring since it links with so many negative impacts on human health [2, 19].
It has been demonstrated that there is not an oral pathogen responsible for causing oral diseases; instead, oral diseases usually initiate after a change in the environment, and this change is responsible for modifying the oral microbiota into a dysbiotic state [2]. For this reason, it is relevant to distinguish between pathogens and pathobionts: Pathogens are microorganisms that can cause disease in any environment they inhabit, whereas pathobionts are commensals that acquire pathogenic properties after specific environmental changes that benefit their growth [18].
S. mutans is the most studied member among oral pathobionts since it is considered the primary colonizer and the microorganism responsible for increasing the adherence of other bacteria; moreover, it can adhere to teeth surfaces and also has the ability to adhere to the oral mucosae [12, 16]. Despite the difference between the pathogenic microorganisms and pathobionts, S. mutans is considered a true pathogen of the oral cavity since its presence usually indicates a positive correlation to dental caries [64, 65]. Furthermore, the virulence factors that S. mutans present are essential for caries formation, such as the ability to produce acid, survive in acidic environments, and produce EPS from sucrose. Despite that, its capacity for biofilm formation, facilitating the adherence of other microorganisms, and forming dental plaques has also been described [56].
Usually, in a state of oral disease, an increase in aciduric microorganisms, such as Streptococcus spp., Lactobacillus spp., Bifidobacterium spp., Actinomyces spp., and Veillonella spp, is particularly noticeable. Interestingly, the complexity of the biofilm is not limited to bacteria. For instance, Candida albicans, a well-known fungi member, is also more commonly found in a cariogenic biofilm [1, 2].
However, there are other relevant pathobionts, such as P. gingivalis, Tannarela forsythia, and Fusobacterium nucleatum, alongside gram-negative anaerobes and LPS producers that are being characterized as major causes of oral inflammation in cases of periodontitis and halitosis [12, 35, 66]. The bacteria known as periodontopathogens, similar to those mentioned above, and others, such as Treponema denticola, Bacteroides spp., Aggregatibacter actinomycetemcomitans, Captnocytophaga spp., and Veillonella spp., are also associated with oral lichen planus [2, 35]. Regarding periodontitis and halitosis, P. gingivalis is the most studied pathogen since it is considered a key pathogen owing to its ability to increase inflammation, worsen the dysbiotic state, and produce volatile sulfur compounds; additionally, it has the ability to evade the host’s immune system [20, 60, 67, 68, 69]. Likewise, the presence of P. gingivalis and A. actinomycetemcomitans not only increases the inflammatory state but also promotes virulence factors of the other species that inhabit the dental plaque [70].
Another relevant pathobiont is C. albicans, which, alongside other Candida species, is responsible for oral candidiasis. These microorganisms present specific adhesins that are capable of binding to dental surfaces, mucosa, and even dental appliances, such as orthodontic braces or artificial crowns. Similar to other commonly found pathobionts, those found in the Candida species can organize in biofilms, resulting in augmented virulence and therapy resistance [62].
Despite oral health being a problem in general health and a burden for public health, there are no implemented preventive strategies for this matter. However, it has been noted that the best angle of preventive approaches should be the regulation of the commensal microorganisms to prevent oral dysbiosis [57].
Regarding conventional treatment for oral diseases, the most frequent guideline is to pay regular visits to the dentist, where the focus should be the removal of dental plaque and the implementation of antibiotics where an infection has occurred [16].
The physical removal of dental plaque, either by brushing the teeth or with professional utensils, such as a scaler, is one of the best traditional strategies to prevent the aggravation of plaque and, consequently, the emergence of caries and gingivitis. However, brushing the teeth is not always effective, and it is not feasible to depend solely on professional actions to prevent oral diseases [7].
The most common treatment, outside of the dentist’s office, to alleviate dental plaque is chlorhexidine mouth rinses. Although its efficacy has been assessed toward different microorganisms, especially S. mutans, it has a few disadvantages, such as teeth staining, loss of salivary flow, and the elimination of all microbiota, which eventually worsens the dysbiotic state [64].
In order to prevent caries, sealants are usually placed to form a physical barrier on the fissures, thus stopping the microorganisms from reaching them. However, where a cavity forms in a tooth with a sealant, what is noted is that it becomes much more severe than when the sealant has not been implemented. Here, the sealant can only be applied to molar teeth since these are the only teeth to present deep sulcus and fissures; nonetheless, caries can affect all teeth in the oral cavity [18]. Another action to prevent the formation of caries is the use of fluor due to its ability to remineralize the tooth surface and prevent cavity formation. Yet, it does not control the adherence of microorganisms or biofilm formation, and dysbiosis can still occur in the oral cavity [51].
Another important disadvantage in the current treatment of oral caries and periodontitis is the use of antibiotics, particularly broad-spectrum antibiotics. The use of these drugs may not be effective due to the bacterial resistance in the biofilm structure and their use may also aggravate the resistance to antibiotics while disrupting the oralome and causing a deeper state of dysbiosis [47].
Regarding periodontitis, the conventional treatment is based on the physical removal of infra-gingival plaque with a technique called scaling and root planning. Despite being effective in the removal of plaque and lowering the levels of microorganisms in the sulcus, periodontopathogens can re-colonize the subgingival pockets and rapidly enter a dysbiotic state again [66].
The established treatment for oral candidiasis is the application of antifungal drugs, such as azoles. However, this therapy deeply affects the host since these drugs are directed to the synthesis of the eukaryotic cell wall. For this reason, common side effects, namely hepatotoxicity and nephrotoxicity, must be taken into consideration during prolonged treatment [62]. Moreover, these microorganisms have the potential to disseminate systemically, causing candidemia. Moreover, multiresistant species belonging to the Candida genus, including C. albicans, have been found, ending in ineffective biotherapy [62, 63].
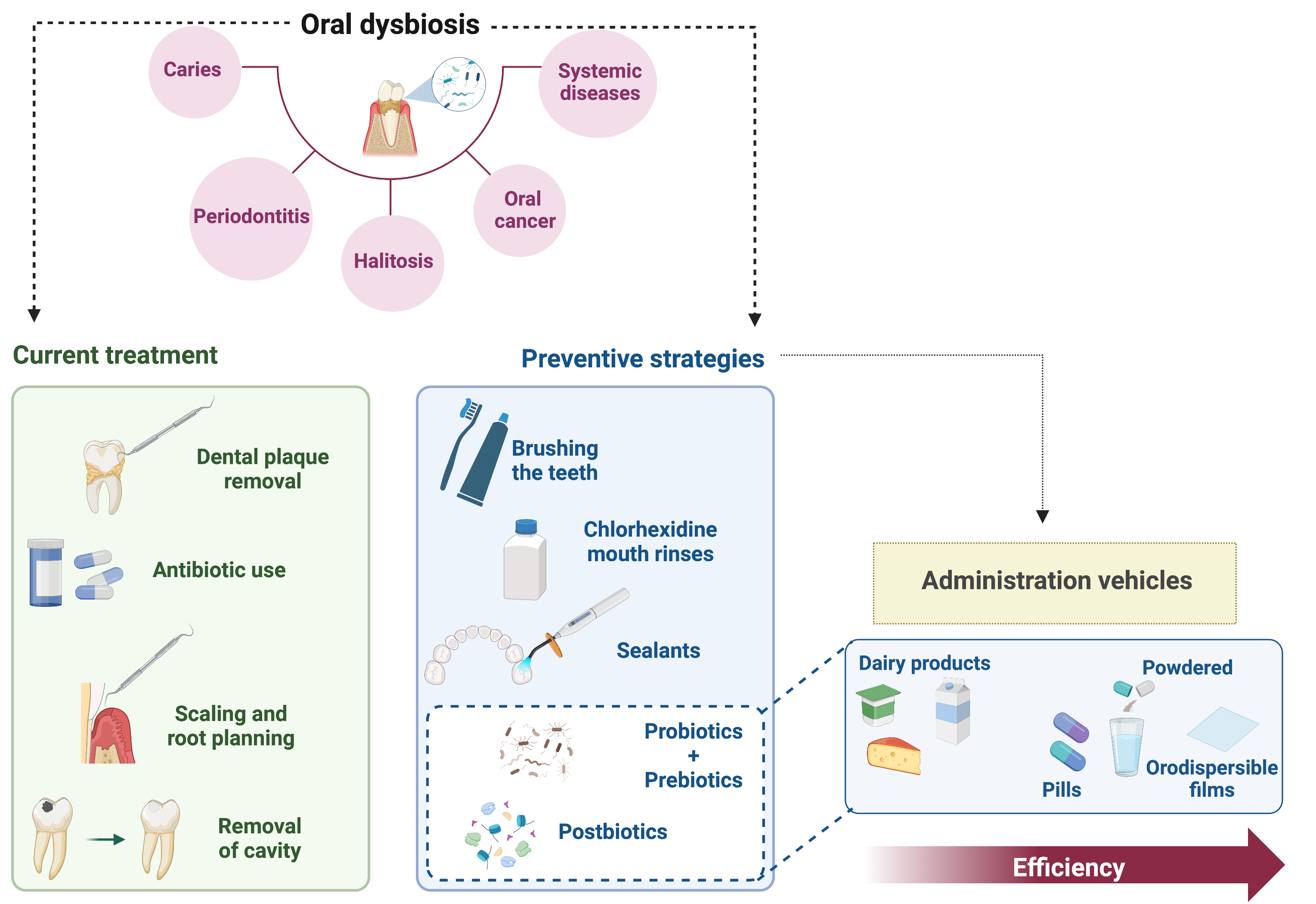
Altogether, it is clear that new strategies for preventing oral dysbiosis need to be implemented to circumvent the problem of oral diseases (Fig. 1).
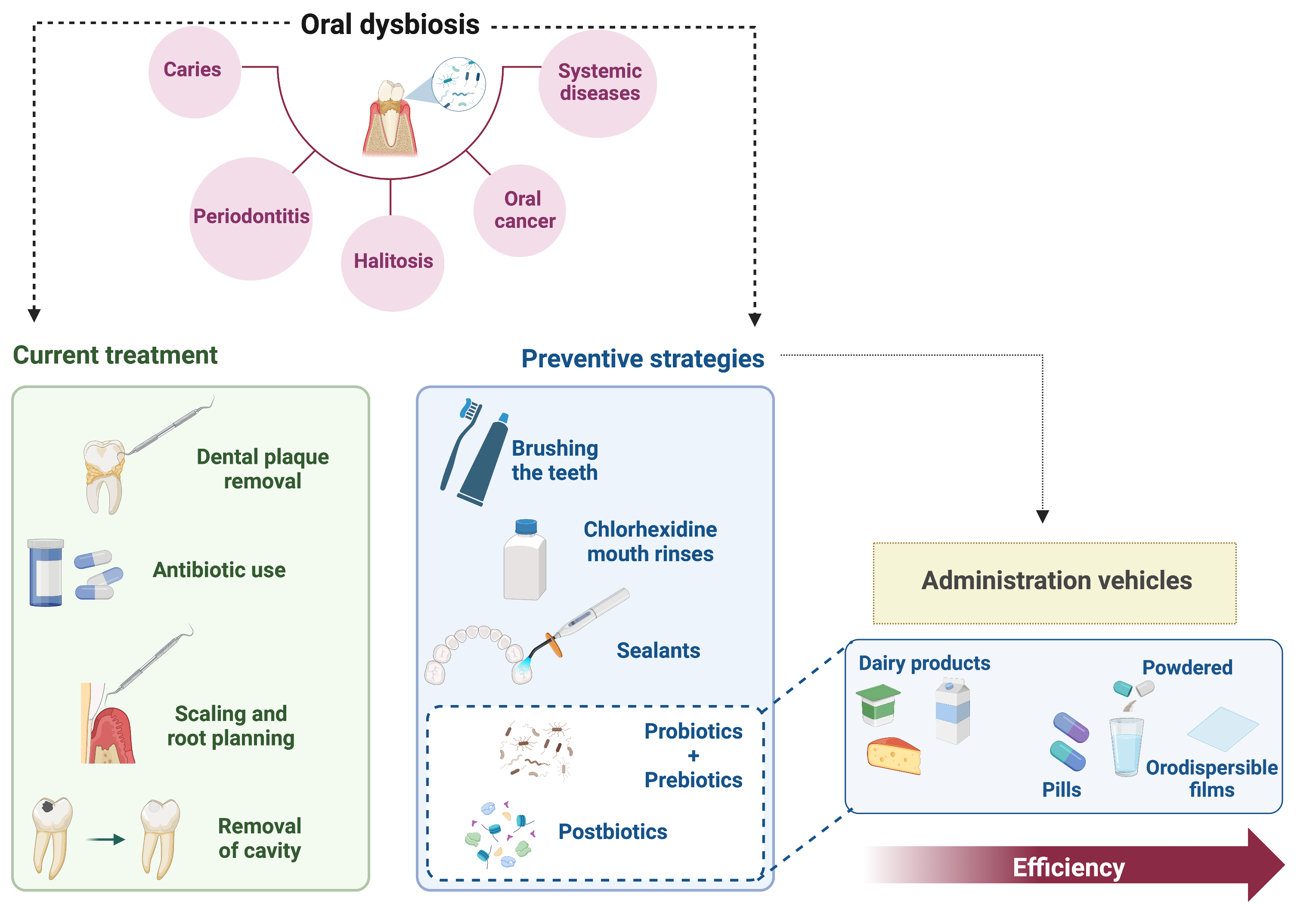 Fig. 1.
Fig. 1.Oral diseases, current treatments, and preventive strategies. Oral dysbiosis is the main factor contributing to the appearance of oral diseases, namely, caries, periodontitis, halitosis, and oral cancer. In addition, oral diseases are intimately correlated with systemic diseases, such as diabetes and heart and lung conditions. The current treatment is based on the removal of dental plaque, either physically or through the use of antibiotics; scaling and root planning for periodontitis, and cavity removal for caries. Regarding preventive techniques, it is clear that there is a lack of efficient strategies. The traditional prevention methods are teeth brushing and the use of chlorhexidine mouthwashes. Sealants can be used in molars, which are more common in young patients. Recently, novel strategies have been gaining attention: probiotics, prebiotics, and postbiotics. Several studies correlated the efficiency of these methods with the administration vehicles. Notably, dairy products are the least effective vehicles regarding oral dysbiosis control, whereas orodispersible films, which represent a promising method, were the most effective.
Oral dysbiosis culminates in oral diseases if the balance is not restored. Interestingly, even though it is one of the most prevalent infectious diseases worldwide, they are still considered a “challenge to modern dentistry” [64]. For example, it is estimated that ECC affects more than six hundred million children globally, even with the implementation of educational strategies worldwide [53].
One of the most important strategies that must be implemented to prevent oral dysbiosis is controlling biofilm formation [47]. For this reason, novel strategies should focus on the removal of mature biofilms in order to maintain oral homeostasis, especially since it has been proven that oral dysbiosis is the leading cause of the emergence of a pathogenic environment [4, 18]. Another important factor is that the microorganisms associated with the emergence of oral diseases and oral dysbiosis are mostly endogenous species and not pathogens brought from another environment. Consequently, the treatment and prevention strategies should focus on controlling the growth of these microorganisms rather than their complete removal [56].
Even with the implementation of novel techniques to prevent oral dysbiosis, oral hygiene should never be disregarded since it is one of the main factors that influence the regulation of the microbiota [16].
One aspect where current strategies have been failing in the prevention of oral diseases is the maintenance of oral homeostasis or keeping the dysbiotic rate low. This rate is defined as the relationship between the pathobiont species present in the oral environment and the species that are mostly associated with oral health [57, 67]. One approach that should be considered is the use of probiotics, prebiotics, and postbiotics to control the oral cavity, especially since they can modulate the oral microbiome and influence the dysbiotic rate by controlling the growth of disease-related microorganisms [67].
In order to fill the gap left by the lack of preventive strategies for oral diseases, a vast number of studies have been focusing on the ability of probiotics and their derivates to maintain homeostasis in the oral cavity [57, 64, 71].
Probiotics have been used to reduce gut inflammation for over two decades and have shown potential in modulating the normal microbiota in other parts of the human body [66]. Recently, it was noted that they are able to reduce the number of oral pathogens, such as S. mutans, when ingested daily for extended periods [27, 32]. Moreover, it was observed that probiotics may restore dysbiotic environments [1, 25, 45, 51]. However, to obtain the full potential of probiotics in the prevention of dysbiosis, their use should be implemented before any signs of inflammation or disease have appeared [72].
Since it is well-known that most oral diseases stem from dysbiosis, it is interesting to understand the mechanisms through which probiotics could aid. Notably, only a reduced number of genera, such as Lactobacillus spp., Bifidobacterium spp., Streptococcus spp., and Weissella sp., have been considered effective in the oral cavity because not all the bacteria that are usually administered as probiotics adhere to the oral mucosa [35]. Commonly, the species with potential probiotic effects are lactic acid bacteria (LAB) and exhibit resistance to considerably low pH levels [73]. These species are “Generally Recognized as Safe” (GRAS) by the European Food Safety Authority [74].
Several researchers have shown a positive correlation between oral health, or better yet, the reduction of pathobiont levels, following the use of probiotic species [35, 45, 66, 72, 75, 76]. In fact, Kamble and collaborators concluded that using probiotics was as effective as chlorhexidine, a common disinfectant [64].
In addition, many studies have explored how probiotics can modulate the microbial composition of the oral cavity. These mechanisms have then addressed how the probiotics (i) inhibit the adhesion of pathogens, (ii) present antimicrobial activity, (iii) affect biofilm formation, and (iv) balance the pH change in the environment [1, 50, 55, 64, 65, 77, 78]. In this regard, probiotics have the capacity to compete for nutrients directly with pathobionts, produce antimicrobial molecules, and modulate the host immune response [1, 45, 64]. Additionally, they decrease the virulence factors of common pathobionts, namely, S. mutans [1, 48, 79].
Despite lowering the levels of potentially pathogenic microorganisms through competition for binding sites and nutrients, probiotics also increase oral health due to their ability to control inflammation in the oral cavity [45, 51, 61, 72, 80]. This anti-inflammatory capacity is reached through the activation of toll-like receptors or by blocking interleukin production [1, 66].
Probiotics may also increase the salivary flow, which is directly related to the maintenance of homeostasis and the prevention of caries [36].
In addition, certain species of probiotics, such as Lactobacillus acidophilus LA5, can also downregulate certain virulence factors of important periodontopathogens, such as the adhesion of P. gingivalis and F. nucleatum [35].
Moreover, probiotics can produce peptides with important functions, such as bacteriocins, which increase the environmental pH, counteract the acid effect of S. mutans, and activate the immune system [1, 45].
Another important characteristic of some probiotic species is the production of
antimicrobial agents, which decrease the levels of pathobionts in the oral
cavity. For instance, Lactobacillus reuteri produces reuterin, a natural
antibiotic that is effective against both gram-negative and gram-positive
bacteria [35, 45, 57, 75]. Other probiotics, particularly L. lactis, are
major producers of nisin, which helps with the maintenance of oral health [1, 19]. Other LAB bacteria, frequently administered as probiotics, produce ammonia,
which prevents the growth of S. mutans in the oral cavity, while others,
usually belonging to Streptococcus spp., produce H
Babina and Kijima also demonstrated that probiotics increase the level of certain immunoglobulins (Ig) in the oral cavity, namely IgA. This property reduced the ability of pathogens to adhere to oral mucosa and dental surfaces while also controlling the inflammatory process [36, 77].
Essentially, the activity of probiotics in the oral cavity demonstrated a reduction in dental plaque and a controlled inflammatory response [35, 57, 75, 81].
Regarding periodontitis, probiotics improved clinical parameters, such as bleeding on probing and periodontal pocket depth [4, 35, 45].
Although some studies have shown that using probiotics alone as a therapeutic option is not able to reverse the disease state, Jansen concluded that if the probiotics reduce the levels of dental pathogens only minimally, it is still enough to provide the immunological system of the patient with the ability to act against the pathogens, thus, reducing the infection [20].
Probiotics from the Lactobacillus genus can be applied to treat inflammation in the oral cavity [75] and as an adjuvant in the treatment of some oral diseases, namely periodontitis. This can be correlated with the fact that the application of probiotics improves bone regeneration and repair [78].
Bizzini demonstrated that Lactobacillus fermentum inhibited the production of glucans by S. mutans; thus reducing the pathogenicity of the biofilm [17]. Additionally, in 2022, Zhang et al. [35] demonstrated that the administration of L. plantarum CCFM8724 was more effective than treatment with chlorhexidine. Accordingly, several studies have demonstrated that L. reuteri produces antimicrobial molecules (reuterin) against S. mutans [78, 82]. Moreover, it can reduce the levels of several pathobionts in the oral cavity and the release of inflammatory cytokines by the host [78].
Lin et al. [83] managed to drastically reduce the levels of S. mutans in patients with moderate and severe gingivitis. Bustamante et al. [24] showed that the daily ingestion of L. reuteri reduced the levels of caries and gingivitis during the first nine years of life. In another study, the same authors demonstrated that after ten months of ingesting L. rhamnosus SP1, the indices of caries lowered. For these reasons, some studies have emphasized that probiotic supplementation is a good strategy for preventing ECC [53].
Additionally, a recent study showed that probiotics, specifically Lactobacillus salivarius, can also favor the salivary buffer ability [56]. This probiotic is interesting due to its resistance to acidic environments [77], while L. salivarius is also important for maintaining periodontal health [75].
Another study demonstrated that Lactobacillus gasseri has probiotic activity towardcaries and periodontitis in vitro [16]. Additionally, the probiotic Lactobacillus acidophilus is able to decrease the number of S. mutans [1].
Several studies have demonstrated that L. rhamnosus could reduce the
number of inflammatory molecules in the oral cavity and the growth of
periodontopathogens, such as Fusobacterium nucleatum [35, 47, 66].
Interestingly, this probiotic also showed the capacity to reduce biofilm
formation by S. mutans [48]. One study demonstrated that the use of
probiotic L. paracasei showed a better capacity to recover from
gingivitis. In this case, this probiotic inhibited the secretion of
IL-1
Another important probiotic is L. lactis, which is usually administered to improve the homeostasis of the biofilm [19]; however, according to the study by J. Wu et al. [50], L. casei is one of the most effective probiotics for controlling caries and was the most effective at lowering the levels of S. mutans.
Recently, Streptococcus salivarius and Weisella cibaria have been gaining attention due to their display of a high number of probiotic properties in the oral cavity [35, 47, 51, 56, 71]. These species are usually chosen based on their enhanced adherence ability, representing an advantage compared to traditional probiotics, such as Lactobacillus spp. and Bifidobacterium spp [36]. In fact, probiotics, such as Streptococcus salivarius and W. cibaria, which are present in caries-free individuals, are thought to improve oral health and modulate the immune system [35, 36, 51, 54, 66, 85]. S. salivarius shares multiple characteristics with S. mutans and S. sobrinus, two important pathobionts in the oral cavity. Consequently, it competes for nutrients and bonding sites, decreasing the number of pathobionts in the microbiome [36]. Moreover, probiotics belonging to the Streptococcus genus may negatively influence the release of proinflammatory cytokines, thus controlling the inflammatory response [1]. Furthermore, S. salivarius exhibits antibiotic action by releasing bacteriocins into the environment, which are particularly active against S. mutans [51, 80], or as a strategy to decrease halitosis [59].
It is important to state that several Lactobacillus species have shown interesting properties against fungal infections, namely those caused by the Candida species. Interestingly, when probiotics are applied, the symptoms of the infection appear alleviated [62]. Curiously, when probiotic therapy is used concomitantly with fluconazole, the antibiofilm activity and the restoration of the oral equilibrium are reached more quickly [63].
Conversely, W. cibaria, a gram-positive bacterium [85], has demonstrated antibacterial and antifungal activity toward oral pathobionts, and its presence is associated with healthy individuals [86]. Moreover, it actively inhibits the growth of periodontopathogens, such as F. nucleatum and P. gingivalis, presents an anti-inflammatory capacity, and reduces the formation of dental plaque [35, 85]. Notably, some studies suggest that W. cibaria can effectively reduce halitosis [59, 85]. This species also produces bacteriocins and is resistant to a wide range of pHs and temperatures, thereby making it an interesting approach for preventive strategies [86].
A key advantage of probiotic therapy is that it can be used for long periods without the risk of side effects [45, 53].
However, it is necessary to remember that probiotics must be taken continuously to be effective [1]. Therefore, to enhance probiotic activity, the use of prebiotics, which are ingredients that improve probiotic activity, is highly recommended by several authors [1, 4, 23, 48, 51, 56, 65].
Despite some promising results, some studies failed to effectively identify probiotics as a tool for controlling dysbiosis. Actually, some studies have demonstrated that probiotics belonging to the genus Lactobacillus do not present a proper ability to adhere to dental surfaces, and for that reason, they cannot appropriately prevent S. mutans colonization [48]. This elucidates the need for an adjuvant therapy to be used concomitantly with probiotics or the need to develop a better strategy.
Chuang et al. [87] found nonces in the levels of S. mutans during their study on the effect of L. paracasei tablets on controlling cariogenic bacteria. Since there are few long-term studies, there is still no data on how long it takes for S. mutans to re-populate and colonize the oral cavity after preventive procedures, such as probiotic intake, have ended [8]. A different study showed no increase in immunoglobulin production after a four-week intake [36], while a meta-analysis showed that the use of probiotics did not improve the plaque index [75].
An important detail to keep in mind is that probiotic functions are specific to each strain, and they cannot be inferred from one study to another. Notably, Gao et al. [4] could not find a reduction in periodontopathogens, such as P. gingivalis and F. nucleatum, after applying L. rhamnosus GG, one of the best-studied probiotics.
Despite all the efforts to understand the mechanism of action behind probiotic efficacy, a considerable amount of research still needs to be performed to fill the gaps in the current knowledge [35].
Moreover, a reasonable number of studies showed a slight decrease in the number of pathogens, although not to the levels of statistical significance. This could be due to the wrong delivery mechanism, dose, or even probiotic strain being applied.
Despite that, because probiotics are live bacteria, they could be weakened or damaged during several steps in their preparation and storage [76]. To overcome these limitations, recent studies have now focused on the use of postbiotics.
Postbiotics are described as inactivated microorganisms or their cell components and metabolites, which promote health when administered [33, 88]. They can be grouped into two different categories: cell-free and cell-derived probiotics, which are usually obtained from fermentation processes [33, 34], are composed of metabolites, and are either secreted by live bacteria or acquired after the lysis of the bacterial membrane, or from fragments of bacterial cells. These components may have positive effects on commensal microorganisms and the immune system of the host [88].
The most common metabolites found in postbiotics are organic acids, namely acetic acid, lactic acid, and propionic acid; antimicrobial molecules, such as bacteriocins [34, 89]; amino-acids, such as alanine and leucine [90]; EPS; and varied enzymes [34].
Recent studies conducted using postbiotics have demonstrated their ability to affect commensal microorganisms in the oral cavity by leading them out of a dysbiotic environment [33, 52]. Additionally, postbiotics have shown the ability to promote the function of the epithelial barrier by improving the protection of the host, as well as the same immunomodulation capacity as probiotics [52]. They have also demonstrated an anti-inflammatory capacity, which is related to the postbiotic [33, 52].
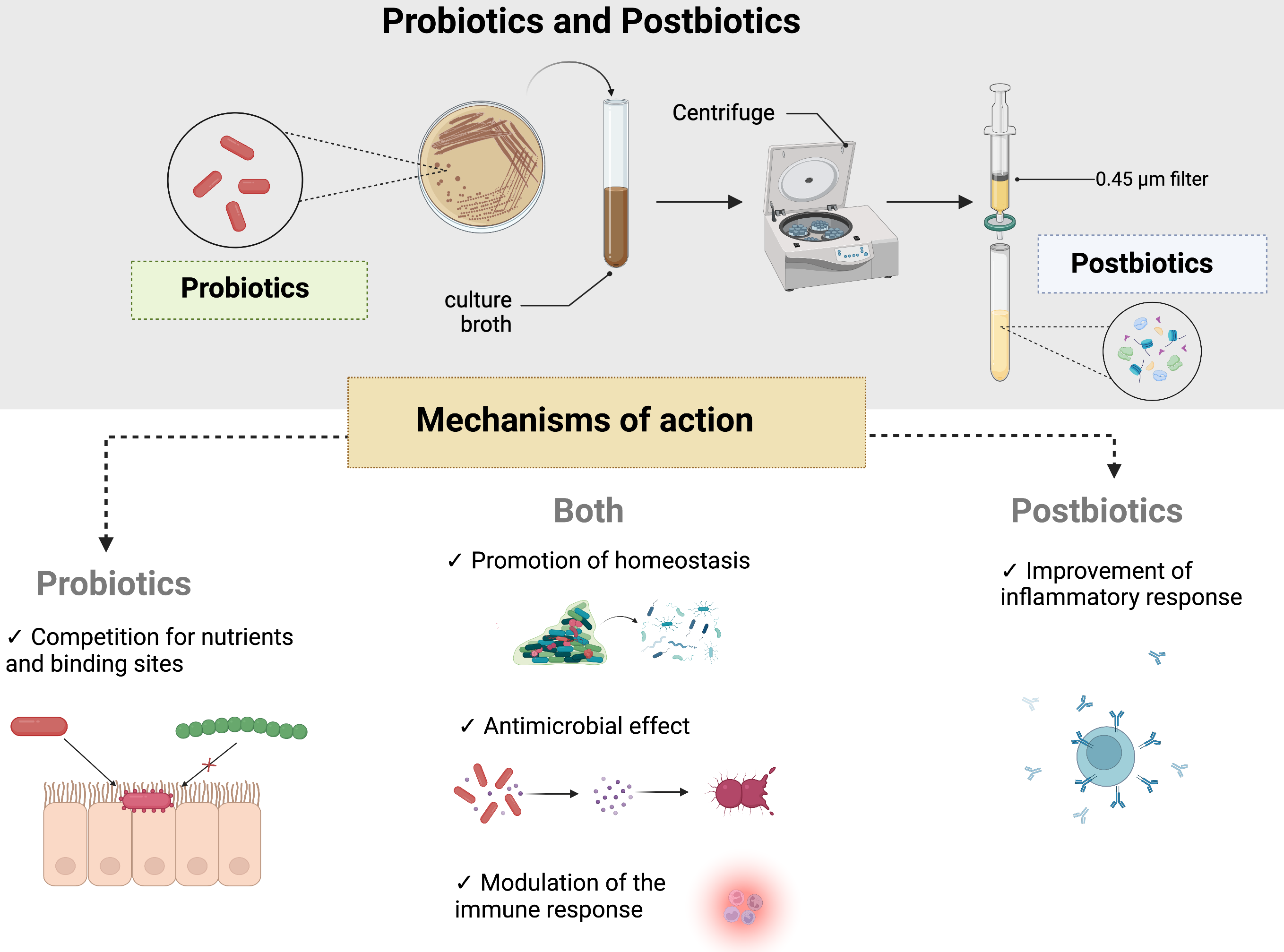
Furthermore, postbiotics present an interesting ability to reduce the levels of oral pathobionts in the oral cavity. In this case, short-chain fatty acids, such as acetate, propionate, and butyrate [91], are indicated as a possible mechanism of growth inhibition since they disrupt the bacterial membrane [12]. Another characteristic of postbiotics is the antioxidant effects on the EPS of certain bacteria, such as L. plantarum, which can reduce the level of reactive oxygen species [34] (Fig. 2).
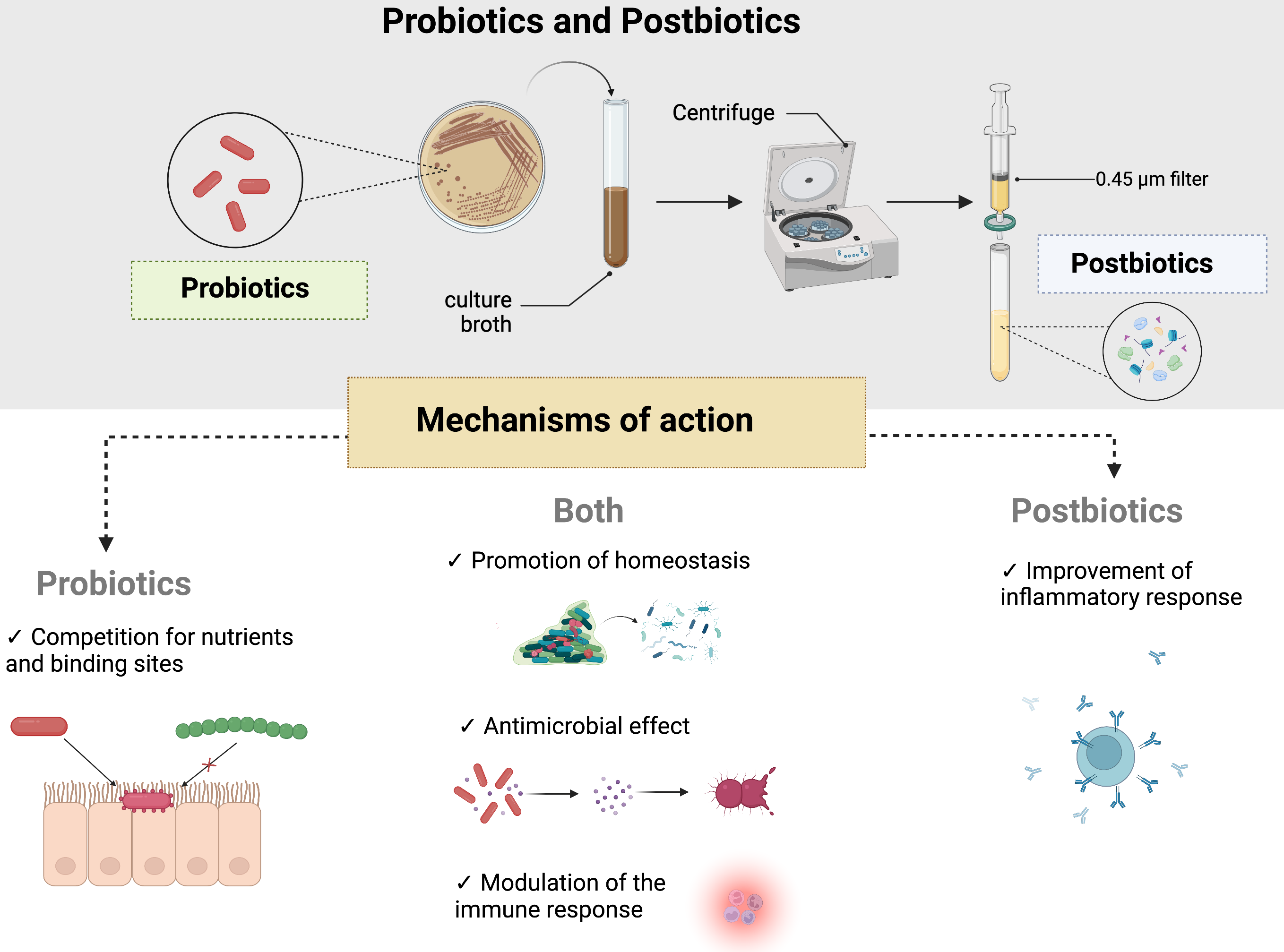 Fig. 2.
Fig. 2.Mechanisms of action for probiotics and postbiotics. As described in the main text, postbiotics are produced by metabolites of probiotics and are obtained after a cycle of centrifugation and a filtration step with a membrane of 0.45 µm. They exert their functions as adjuvants of human health through different mechanisms, such as promoting homeostasis of oral microbiota, disrupting biofilm formation, and modulating immune and inflammatory responses. Probiotics also compete for nutrients and binding sites, which helps decrease pathobiont levels and produce molecules with antimicrobial effects. Notably, postbiotics also show antimicrobial effects since they disrupt bacterial cell walls.
In fact, several studies have shown promising results against pathogenic biofilms in the oral cavity [92, 93, 94]. However, the efficacy of the postbiotics against pathobionts is always related to their concentration [12].
Furthermore, postbiotics have recently been related to improved oral health following periodontal issues due to their ability to reduce periodontal pathogens, such as P. gingivalis [12]. Moreover, another study demonstrated that postbiotic extracts from Lactobacillus spp. have antimicrobial activity against gram-negative pathogens [52]. Further, the administration of postbiotics also presented an antibiofilm capacity against S. mutans, a desirable characteristic for the control of dysbiosis. This effect can be correlated with the presence of teichoic acids produced by Lactobacillus spp. Despite that, another interesting study demonstrated that the application of postbiotics did not result in cytotoxic activity against the cells [89].
The most important advantage of postbiotic administration is that there are no living microorganisms present; this means there is no risk of the transmission of resistance genes, and there is no risk of causing infection in immunosuppressed individuals or high-risk groups (elderly, children, and pregnant women) [52].
Regarding the limitations of postbiotics, there have been a few studies where their use did not show intense immunomodulation activity [52]. Additionally, there is still a lack of information about the identification of the present compounds, which hinders the scaling-up process [33].
Moreover, the efficacy on the oral health of both postbiotics and probiotics is intrinsically correlated to the form of administration because they determine the time of contact with the oral cavity and how well they are released in the mouth to provide the benefits of their biological properties. As such, the form of administration significantly impacts the effectiveness of the postbiotics. One form of administration might be to vehicle the postbiotics through orodispersible films.
Therefore, the following section presents the main advantages and disadvantages of using oral films as a promising delivery mechanism for both probiotics and postbiotics.
Several different vehicles can be used to administer probiotics and postbiotics to the oral cavity. The most common are tablets and lozenges, although there are studies that have reported on the use of milk, powders, and even ice creams as vehicles of administration to the oral cavity [95].
However, the efficacy of the treatment is intimately related to the chosen vehicle [96]. Thus, it should be improved regarding pro- and postbiotic efficiency [97]. These common administration vehicles are effective when the goal is to improve intestinal health. However, when it comes to improving oral health, dairy options should not be considered [53]. Furthermore, to allow the probiotics and postbiotics to act on the colonization sites, the administration vehicles should not be intended for swallowing but rather focused on in-mouth disintegration [25, 53, 55, 97, 98].
Orodispersible films (ODF) are not a new strategy for the delivery of drugs to the oral cavity; they have been explored for more than 40 years [99]. They are described as solid, thin, polymeric preparations with the ability to deliver the active principle quickly and easily. ODF must be non-toxic and biocompatible, which is why no adverse effects are expected after its use [25, 68, 71, 95, 100, 101]. Another interesting property of ODF is their solubility and quick disintegration, which usually occurs in under five minutes [102] and without the need for water [101]. Hence, no choking risk is associated with its intake [76, 99]. This translates to an advantage in its application since it broadens its use to pediatrics and geriatrics and individuals with dysphasia or bedridden patients [101].
An important characteristic that should be reached is mucoadherence because it allows the active principle to be present in the oral cavity for enough time to be released and to colonize different areas of the mouth [66, 103]. One of the most important advantages of ODF is the elevated tolerability by several different groups of individuals [71]. For instance, several studies have demonstrated the lack of compliance with traditional oral hygiene methods in both children and adults [55]. Moreover, the manufacturing of ODF is simple and brief, and the process is not expensive [101].
Alternatively, it is important to understand that, due to its characteristics,
ODF show a loss in effectiveness throughout time. Therefore, particular attention
should be given to packaging [101]. In addition, there must be extra care in
achieving high concentrations of probiotics and postbiotics since several studies
demonstrate that at least 10
Regarding its composition, it is observed that ODF are mostly composed of film-forming polymers, plasticizers, sweetening agents, and saliva stimulators [25, 41, 101, 104]. However, the properties of each ingredient and its quantity are key to establishing the most favorable preparation. It should be noted that oral dispersible films should be applied at least twice a day in order to obtain maximum results, with more than considered inconvenient for daily use [84].
Polymers, such as hydroxypropyl methylcellulose, maltodextrin, or chitosan, comprise nearly 50% of the ODF [25, 104, 105]. Interestingly, it has been shown that polymers positively affect oral health, even without the administration of pro- or postbiotics [66].
Plasticizers, the second most important excipient of ODF, optimize the plasticity and handling of the films [41, 47, 99, 101, 104, 105]. The most used plasticizers are glycerol, sorbitol, and mannitol, among others [104].
It is relevant to add a saliva stimulant, namely organic acids, to improve the characteristics of the ODF and the convenience of its use [99, 104]. The importance of using citric acid as a stimulator of saliva production is based on the buffering capacity of saliva and its potential to eliminate microorganisms using the salivary flow. Moreover, it has been demonstrated that saliva stimulators aided in the increase of the oral pH [18, 35].
Other ingredients can be used for specific purposes. For example, Mentha sp. is used to reduce the S. mutans biofilm and can be applied to postbiotic ODF, while sugars have been used for their antimicrobial properties [1, 64].
Finally, it is useful to include excipients relevant to caries prevention in ODF formulations, such as fluoride and arginine. When arginine is present, some bacteria can produce ammonia and, consequently, raise the oral pH, thereby preventing tooth demineralization [44, 51]. Nitrate is another common prebiotic that is added to the ODF preparation owing to its ability to shift oral communities toward a healthier environment [106].
Although ODF are a promising strategy for administering pro- and postbiotics, presenting attractive advantages and satisfying the conditions are needed to release these molecules in the oral cavity effectively. Moreover, their actual impact on oral dysbiosis needs to be addressed and clarified.
In summary, oral diseases are deeply related to an unbalanced oral cavity microbiome called oral dysbiosis. Since this lack of homeostasis forms the basis for most oral diseases, preventive strategies need to focus on re-establishing commensal microorganisms rather than removing the entire microbiota or, in other words, re-establishing homeostasis.
For this, novel strategies have been gaining attention, such as the use of probiotics, prebiotics, and postbiotics. Probiotics and prebiotics are well-established and accepted in society. While probiotics are a group of bacteria that provide specific benefits to the human body when taken in considerable amounts, prebiotics are a group of ingredients that enhance the growth and activity of probiotics. Additionally, postbiotics are considered the metabolites of probiotics, as well as fragments of bacteria itself, which provide benefits to the host.
Both probiotics and postbiotics have been widely studied, and their efficacy has been proven in many studies, either by decreasing the numbers of oral pathobionts, controlling their growth, or simply modulating the inflammatory and immune response of the host. In such cases, their efficacy has been deeply associated with how they are employed, namely the administration vehicle used.
Orodispersible films are a promising strategy owing to their characteristics and lack of toxicity. ODF composition is simple, with most being composed of polymers and plasticizers, which dissolve easily without water. Further, they can adhere to the mucosal membranes, thereby allowing the probiotics or postbiotics to act as desired. Moreover, they can be easily administered to patients without any risk of choking, they are not expensive to manufacture, and they have scale-up potential.
Therefore, the impregnation of ODF with probiotics or postbiotics should be considered as a potential alternative with which to target oral dysbiosis and aim to prevent oral diseases, such as caries or periodontitis, as well as its antibiofilm capacity, which reduces not only the virulence of common oral pathobionts but also the risk of acquiring antibiotic resistance genes. However, detailed studies on the physical characteristics of ODF with and without pro- and postbiotics should be performed to fully understand the mechanisms through which it delivers the active compounds to the oral cavity, as well as the need for special storage conditions and careful handling.
Of note, some barriers remain that need to be overcome to start using these strategies as preventive approaches for oral dysbiosis.
Firstly, intensive investigation should be performed to evaluate and clarify the mechanisms of action of both probiotics and prebiotics. In fact, a more detailed understanding of their mechanisms of action could result in the best option being used as a preventive strategy to avoid oral diseases. Furthermore, regarding the viability of the probiotics in oral films, more studies are required to understand the survival rate since some of the used probiotics were not usual commensals in the oral cavity. Regarding postbiotic use, the ability to produce its effects locally should also be studied since deglutition possibly removes them from the mouth.
Secondly, studies that establish and define the actual composition of postbiotics are crucial to understanding the components that confer antimicrobial activity to postbiotics and to determine the ideal conditions and parameters for large-scale production, as well as the identification of specific molecules with antibiotic properties and other critical health-related characteristics.
In addition, it is essential to remember that the in vitro characteristics are significantly different from the in vivo behaviors since the effect of the environment is not achieved in a Petri dish. The salivary flow and deglutition, mastication and food debris, and the 3D structure of the biofilm are important factors that change the way probiotics perform in the oral cavity. Therefore, it is important to conduct clinical tests to determine the actual benefits of using ODF in the future.
LPS, lipopolysaccharides; EPS, exopolysaccharides; IL, interleukin; AEP, acquired exogenous pellicle; ECC, early childhood caries; LAB, lactic acid bacteria; GRAS, generally recognized as safe; ODF, orodispersible films.
FKT contributed to the conception of the present work and provided supervision on data acquisition and interpretation. MBR and CSO designed the research study. MBR performed the research and wrote the manuscript. CSO and FKT provided oversight and critical revision of the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
Not applicable.
All figures were created using Biorender.com. The authors acknowledge Biorender’s support. The authors would like to thank the staff of the CBQF for their assistance in this project.
This work was supported by National Funds from Fundação para a Ciência e a Tecnologia (FCT) through the project [UIDB/50016/2020]. CSO was supported by the Be@t – Textile Bioeconomy Project (Investment TC-C12-i01 – Sustainable Bioeconomy [02/C12-i01/202]) through an individual Junior Researcher contract.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.