1 Department of Biological Sciences, Virtual University of Pakistan, 54000 Lahore, Pakistan
Abstract
Background: The promoters of mammalian genes contain clusters of CG dinucleotides known as CpG islands. Most mammalian housekeeping genes predominantly contain CpG islands (CGIs), facilitating gene transcription. Numerous studies have explored the physiological implications of the relationship between CGIs and gene expression. However, the evolutionary implications of this relationship remain largely unexplored. Pseudogenes, in contrast, are genomic remnants that have lost their function over evolutionary time. Methods: In our current research, we employed comparative genomic techniques to demonstrate a correlation between the absence of gene expression due to a lack of CGIs in the gene promoters and pseudogenization. Results: We showed that there is a significant enrichment of tissue-specific genes in the functional orthologs of pseudogenes. We also found a significant correlation between the lack of CGIs and enriched tissue specificity in these functional orthologs of pseudogenes. Conclusions: We inferred that perhaps tissue-specific genes are more prone to the process of pseudogenization. In this way, because of their impact on gene expression, CGIs may affect the fate of a gene. To our knowledge, this is the first study to propose a connection between CGIs, gene expression, and the pseudogenization process and discuss the evolutionary implications of this potential trilogy.
Keywords
- CpG islands
- pseudogenes
- gene expression
- pseudogenization
- tissue specificity
- genome evolution
Pseudogenes are non-coding and generally non-functional copies of the genome. Despite being physiological dead ends, they are universally prevalent in all metazoan genomes. In vertebrates, three types of pseudogenes are commonly present: Duplicated pseudogenes (DPGs), which arise from the duplication of a parent gene [1]. The second type is processed pseudogenes (PPGs), which originate from the retro-transposition of a gene. Both DPGs and PPGs have functional paralogs within a given species, referred to as parent genes. The third type is a distinct type of pseudogenes, termed Unitary pseudogenes (UPGs), which emerge due to the accumulation of disabling mutations, although they have functional orthologues in other species [2]. All these pseudogenes arise from the accumulation of disabling mutations due to the relaxed functional constraints these genes face for various reasons. For instance, the presence of multiple copies in the case of DPGs and PPGs and species-specific physiological variations in the case of UPGs [2, 3, 4].
CpG islands (CGIs) are clusters of cytosine and guanine dinucleotide aggregates in the promoters of most mammals and other warm-blooded animals. In other vertebrates, the genome is generally heavily methylated. This indiscriminate methylation process leads to a general depletion of CG dinucleotides due to the mutagenicity of methylcytosine [5]. However, although the genome is heavily methylated in mammals, CGIs generally remain unmethylated compared to other vertebrates. It has long been observed that DNA methylation generally suppresses the transcription process [6, 7]. Therefore, the fact that CGIs remain unmethylated suggests that their presence promotes gene transcription. This comprehension is reinforced by the observation that nearly all housekeeping genes have CGIs in their promoters [8, 9, 10, 11]. Furthermore, the existence of CGIs in the promoters of nearly all mammalian housekeeping genes and a portion of tissue-specific genes suggests their possible involvement in regulating transcription [12]. Genome-wide in situ chromatin immunoprecipitation (ChIP) and transcriptome analysis has also shown the recruitment of RNA polymerase II (RNAPII), specificity protein 1 (SP1), nuclear respiratory factor 1 (NRF-1), E2 promoter binding factor (E2F), and other important transcription factors, to promote the transcriptional process [13, 14, 15]. This recruitment of transcription factors is also considered one of the reasons for keeping the CGIs in their unmethylated state [16, 17, 18]. However, it is noteworthy that not all CGIs remain unmethylated. CGIs are methylated in certain cases where gene methylation is indispensable, such as x-chromosomal inactivation and genetic imprinting [19, 20]. Similarly, in particular cancer tissues, CGIs are hypermethylated, leading to the transcriptional suppression of certain tumor suppressor genes [21]. The methylated CGIs recruit different proteins that methylate these sites and, ultimately, promote gene silencing. The main proteins that bind with methylated CGIs are the methyl-CGI-binding domain proteins (MDB1, MDB2, MDB3, MDB4, etc.) and another class of several structurally unrelated methyl-CpG-binding zinc-finger proteins in the Kaiso family (ZBTB33, ZBTB4, ZBTB38, etc.) [22]. In this way, proteins binding with the unmethylated CGIs differ from those binding with the methylated CGIs, where the former promotes transcription, and the latter suppresses it. Nevertheless, even though there are exceptional cases of CGI methylation, CGIs generally remain unmethylated and, consequently, are implicated in activating the transcription process.
Although much has been written on the effect of CGIs on gene expression, the evolutionary implications of this relationship have been thoroughly ignored. For instance, to our knowledge, no study has previously investigated the impact of CGI-based gene expression on gene evolution. In our previous work, we showed that there is a lack of CGIs in pseudogenes and emphasized that this lack of CGIs may have led to the pseudogenization process, promoting the formation of UPGs [23]. So, an inevitable question arises: How could the CGI profiles affect the fate of genes? The current research project is a step forward in determining this missing link between CGIs and pseudogenization. To the best of our knowledge, this is the first study that focuses on the potential way through which CGIs might be implicated in gene pseudogenization.
The identification of pseudogenes, their orthologs, and the prediction of CGIs was conducted as described by Khan et al. (2021) [23]. Briefly, pseudogenes were extracted using the Ensembl–BioMart tool [24]. Functional orthologs of the UPGs were identified using the Ensembl–BLAST tool [25, 26]. We classified genes as orthologous to pseudogenes based on three criteria: (i) they were the highest scoring results in the BLASTN search, (ii) the associated human UPGs were the top-ranked when the orthologous gene was utilized as a query sequence in a reverse-BLAST search, and (iii) the list of human orthologs in the Ensemble genome browser did not contain a functional ortholog for the mouse/primate gene. For the gene expression data, we utilized three gene expression databases: (i) gene expression data from the fantom-5 project, focusing on 35 adult mice tissues [27], (ii) gene expression data from a study by Huntley et al. [28], focusing on gene expression in 9 organs in adult mice, and (iii) gene expression data from a study by Brawand et al. [29], focusing on gene expression in 6 organs in adult mice. Using data from three different sources rather than one minimized the chances of bias and increased the coverage of our target genes. A gene was considered expressed in a given tissue if its expression level was equal to or greater than 1 read per kilobase per million mapped reads, or RPKM [30]. Genes that were expressed in two-thirds or more of the total tissues for which gene expression data was taken were considered broadly expressed, while genes below this threshold were considered tissue-specific [31]. CGIs were predicted using the CpGprod tool [32]. We selected a 1200 bp DNA segment for each gene, which included 1000 bp upstream and 200 bp downstream from the transcription start site (TSS), to identify the CGIs in the gene promoters. CpGProD employs rigorous standards for detecting CpG islands, specifically, DNA segments exceeding 500 bp with an average G + C content over 0.5 and a CpG observed/expected ratio above 0.6. The significance levels were calculated using the chi-squared test to determine the association between two categorical values [33].
Gene pseudogenization is a universal feature of all genomes. Among the three
pseudogene classes, unitary pseudogenes (UPGs) are unique since they are
non-functional in one species but functional in many others. Gene expression is a
hallmark of all protein-coding genes. Depending on the extent of the expression,
genes can be categorized as either tissue-specific or broadly expressed. In this
research project, we sought to explore if any relationship exists between gene
expression and gene pseudogenization. To this end, we compared the expression
profile of the functional orthologs of human UPGs (O-UPGs) in mice. Intriguingly,
the majority of O-UPGs were tissue-specific in all our datasets that were used
for the gene expression analysis, i.e., 46 out of 56 (82%), 41 out of 50 (82%),
and 28 out of 32 (88%) genes were tissue-specific in the studies by Huntley
et al. [28], Brawand et al. [29], and Fantom-5, respectively
(Fig. 1A–C and Supplementary file-1). All these studies showed that a
significantly dominant portion of O-UPGs was tissue-specific compared to all
other protein-coding genes, with p
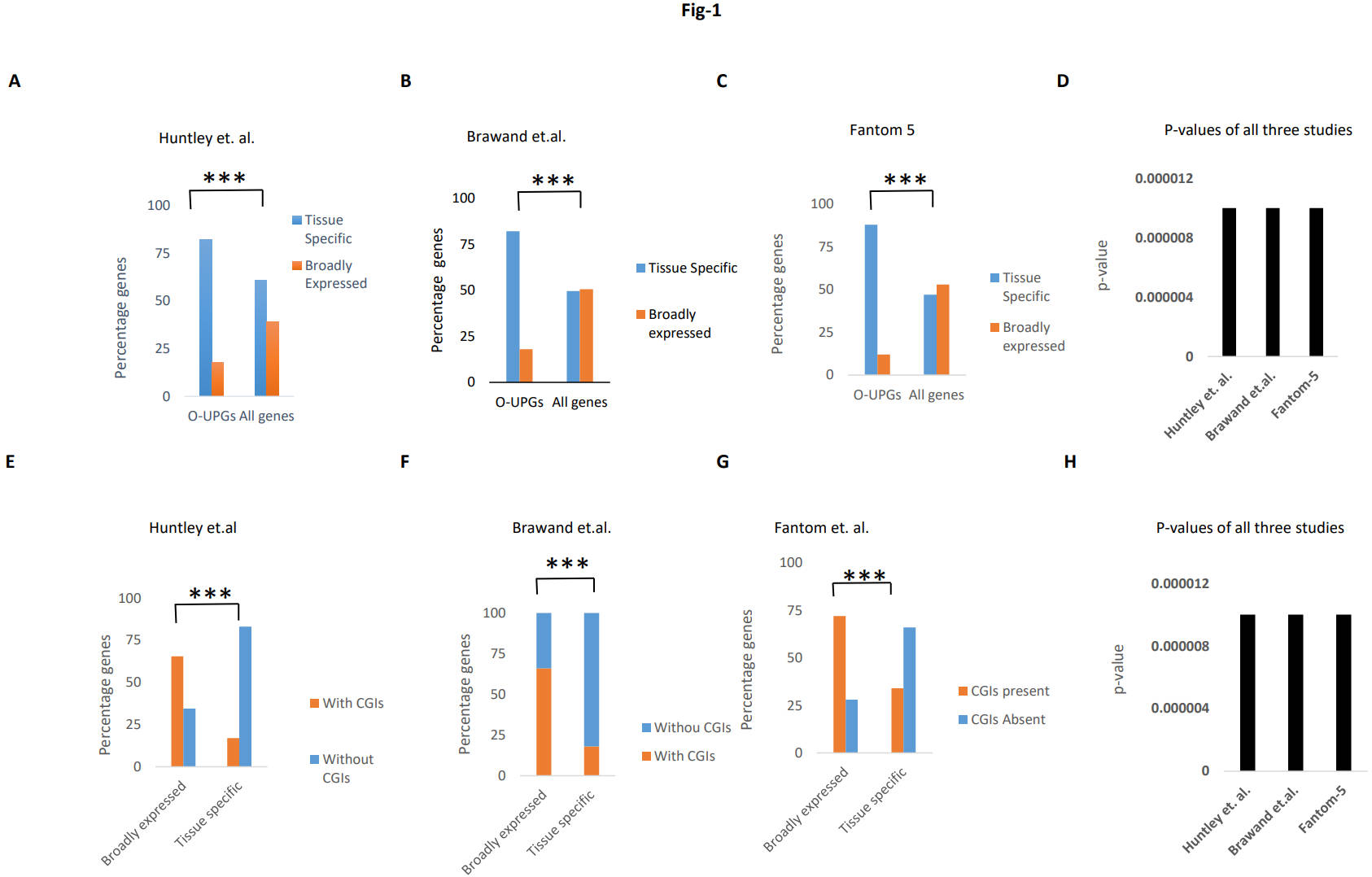
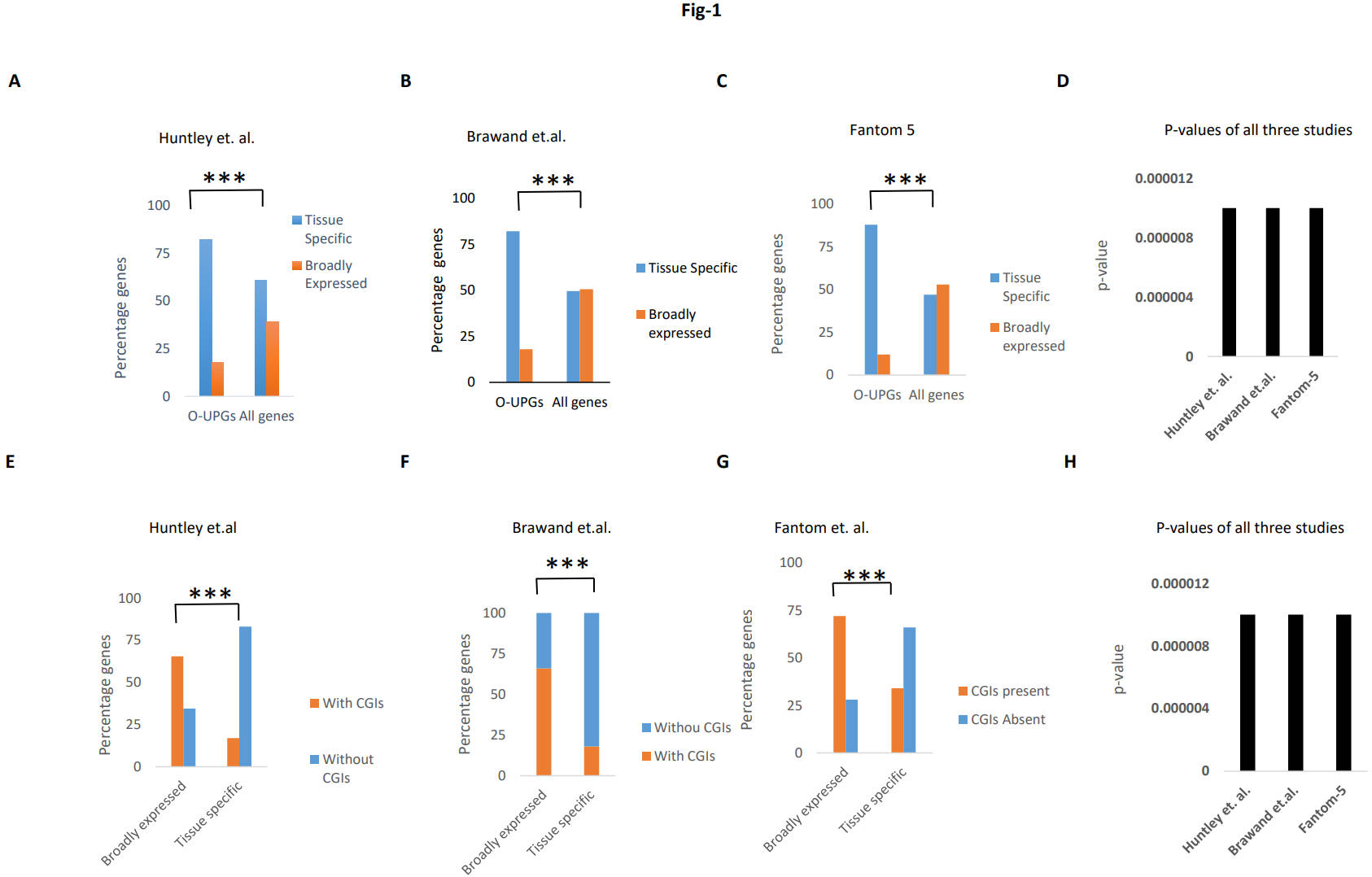 Fig. 1.
Fig. 1.Enrichment of tissue-specific genes. (A–C) The frequency of
broadly expressed and tissue-specific genes in O-UPGs and all the protein-coding
genes from three gene expression datasets. (D) Comparison of p-values
for our results in all three datasets. X
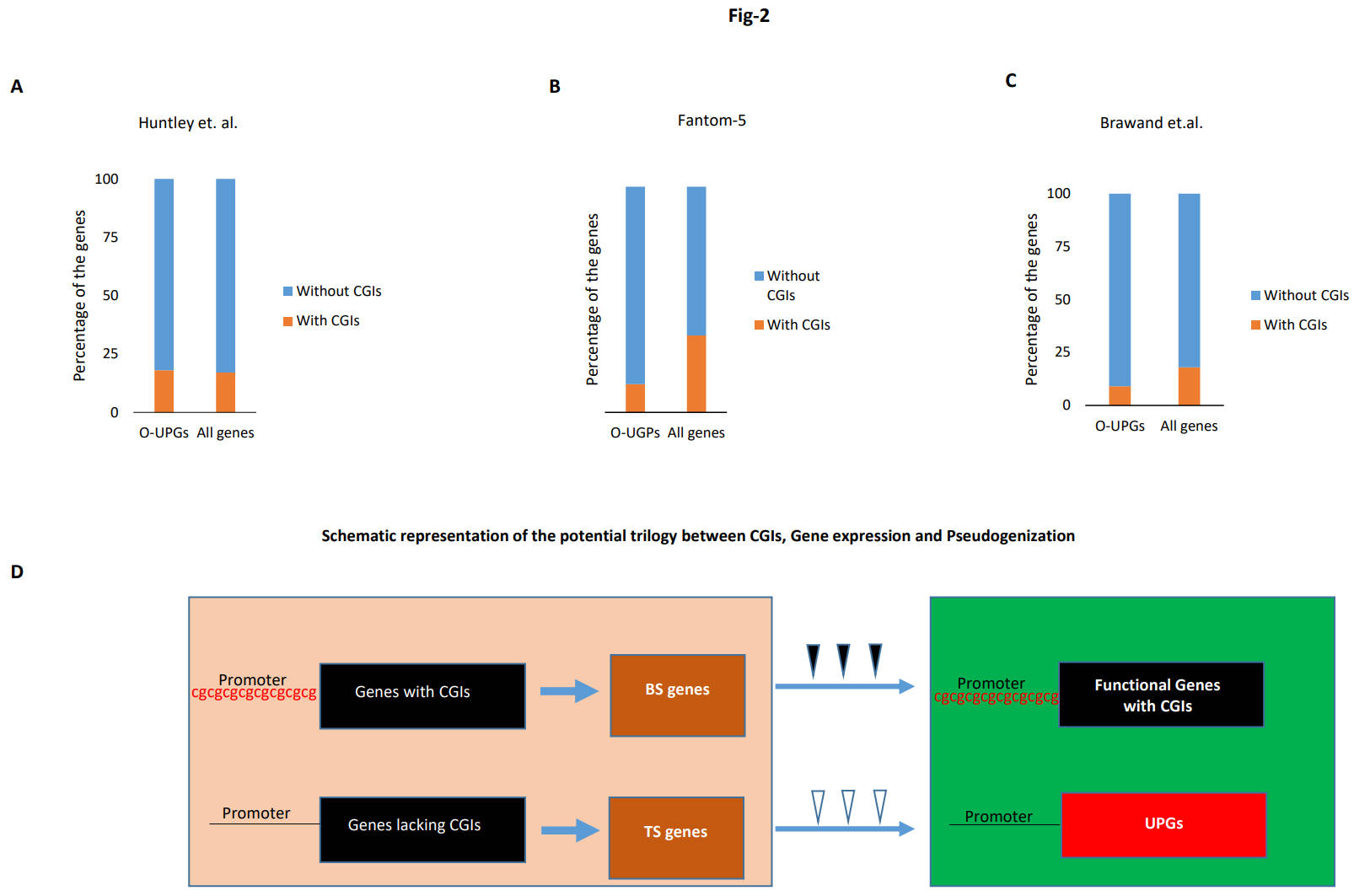
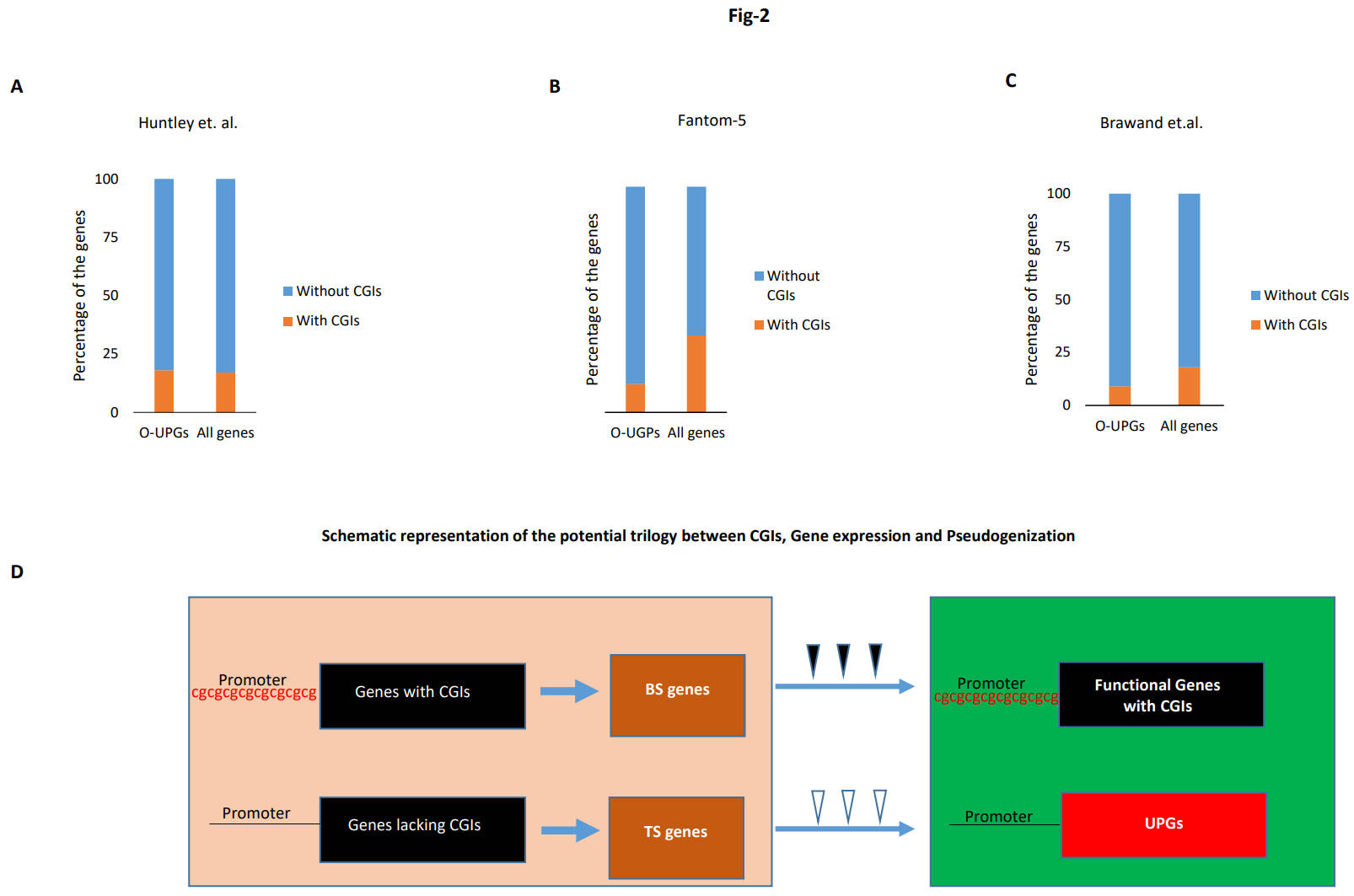 Fig. 2.
Fig. 2.Tissue specificity and pseudogenization. (A–C) Lack of CGIs in tissue-specific O-UPGs. Distribution of CGIs in the tissue-specific O-UPGs compared to their distribution in the tissue-specific gene in all the mouse protein-coding genes. The gene expression data from three different studies was used in this analysis. (D) A schematic representation of the potential trilogy. Impact of the status of the CGIs on gene expression and their fate. The left-hand side (light brown background) represents proto-pseudogene. The lack of CGIs leads to tissue specificity, which paves the way for UPGs to form in certain species, as shown on the right-hand side (green background). The triangles indicate functional constraints, with solid ones for strong and hollow ones for weak functional constraints.
Our previous work demonstrated a predominant absence of CGIs in the PGs [23]. We were particularly intrigued by the fact that this lack of CGIs was also present in the mouse and primate genes orthologous to human UPGs. This led us to hypothesize that the genes lacking CGIs are more susceptible to pseudogenization. However, how the lack of CGIs could have led to gene pseudogenization was unclear. Our findings in the current research project provide the missing link between the lack of CGIs and gene pseudogenization, i.e., perhaps the lack of CGIs leads to an absence of gene expression, which results in tissue-specific gene expression. These tissue-specific genes are more prone to gene pseudogenization than highly active, broadly expressed genes. In this way, we can clearly see a correlation between the lack of CGIs, gene expression, and the gene pseudogenization process. Subsequently, the inevitable question arises: Why does nature tend to discount broadly expressed genes from the pseudogenization process and instead prefer tissue-specific genes for this fateful decay? One possible and intuitive explanation could be that genes expressed in a broad spectrum of tissues are perhaps under more functional constraints than genes expressed in only a few tissues. A gene, which is required by more tissues, will undoubtedly be quite crucial to the functioning of different body parts and will contribute immensely to the survival of the organism. However, a tissue-specific gene may be under relatively lesser functional constraint because its pseudogenization may not be a survival issue. Thus, compared to broadly expressed genes, tissue-specific genes will harbor more mutations, meaning a relatively relaxed functional constraint will ultimately lead to an ever-increasing accumulation of disabling mutations. Furthermore, the accumulation of disabling mutations ultimately leads to pseudogenization [37, 38, 39]. Hence, there are indeed studies hinting toward a relatively more conserved evolution of broadly expressed genes compared to tissue-specific genes [31, 40]. One obvious consequence of this faster evolution of tissue-specific genes is that a possible lesser functional constraint, compared to broadly expressed genes in certain species, may lead to their relatively easier pseudogenization (Fig. 2D). In this way, our assertion that the lack of CGIs may be one of the reasons for the pseudogenization of UPGs is reinforced here; the lack of CGIs in gene promoters leads to the expression of genes in only a limited number of tissues, which in turn relaxes the functional constraint on these genes, ultimately, leading to pseudogenization in certain species.
CGIs are a significant feature of mammalian gene promoters, yet CGIs are predominantly absent from pseudogene promoters. The absence of CGIs in most O-UPGs may have contributed to these genes becoming tissue-specific. These tissue-specific genes, in turn, are under lesser functional constraints and might be an easy target for pseudogenization. Therefore, the introduction of CGIs into the mammalian genome has not only affected the gene expression profile of genes, it may also have influenced their fate, with genes lacking CGIs becoming tissue-specific and, ultimately, pseudogenized in certain species, owing to relatively reduced functional constraint. This study underscores the need to further explore the control of gene expression through CGIs and how this process influences the evolution of vertebrate genomes in general and the mammalian genome in particular. The trilogy between CGIs, gene expression, and gene pseudogenization highlights the importance of introducing CGIs into the mammalian genome and underscores the need for a deeper and more comprehensive exploration of this all-important event in the history of mammalian evolution.
All the Data is provided as supplementary material. In addition, the data utilized and/or examined in the present study can be obtained from the corresponding author upon a reasonable request.
AAK conceived the idea. AAK planned and supervised the research. AAK and AF extracted the data. AAK and AF analyzed the data. AAK wrote the manuscript. In addition, both authors have participated sufficiently in the work to take public responsibility for appropriate portions of the content and agreed to be accountable for all aspects of the work in ensuring that questions related to its accuracy or integrity. Both authors read and approved the final manuscript. Both authors contributed to editorial changes in the manuscript.
Not applicable.
We are thankful to HEC for providing the funding (HEC-NRPU-17093) for this project.
This research is funded by Higher Education Commission of Paksitan (HEC) under the project HEC-NRPU-17093.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.