1 Geriatric Medicine Center, Department of Endocrinology, Zhejiang Provincial People’s Hospital, Affiliated People’s Hospital, Hangzhou Medical College, 310014 Hangzhou, Zhejiang, China
2 Key Laboratory of Endocrine Gland Diseases of Zhejiang Province, 310014 Hangzhou, Zhejiang, China
3 Jinzhou Medical University, 121001 Jinzhou, Liaoning, China
4 Laboratory Medicine Center, Department of Clinical Laboratory, Zhejiang Provincial People’s Hospital, Affiliated People’s Hospital, Hangzhou Medical College, 310014 Hangzhou, Zhejiang, China
Abstract
Background: Phosphodiesterase-5 (PDE-5) inhibitors have been found to play an important cardio-protective role. This study aimed to clarify the inhibitory effects of PDE-5-silenced bone marrow mesenchymal stem cells (BMSCs) on high glucose-induced myocardial fibrosis and cardiomyocyte apoptosis. Methods: Cardiomyocytes and fibroblasts of neonatal rats were treated with high glucose (HG), and co-cultured with PDE-5-overexpressed or -knocked down BMSCs. The viability and apoptosis as well as the levels of cytokines, Cardiac troponin I and Vimentin of cardiomyocytes and fibroblasts were studied. The expressions of PDE-5, cyclic guanosine monophosphate (cGMP) and protein kinase G (PKG), in both cells were evaluated. Results: BMSCs that silenced PDE-5 facilitated the viability of cardiomyocytes, decreased the viability of fibroblasts, and inhibited the apoptosis of cardiomyocytes and fibroblasts. The contents of collagen-I, collagen-III, tissue inhibitor of metalloproteinase (TIMP)-1 and Dermin in fibroblasts were decreased by the PDE-5 inhibitor, but the levels of matrix metalloproteinase (MMP)-1 in fibroblasts and troponin-I in cardiomyocytes were increased by the PDE-5 inhibitor. PDE-5 inhibitor also suppressed the expression of PDE-5 but up-regulated cGMP and PKG expression in cardiomyocytes and fibroblasts. Conclusions: PDE-5-inhibited BMSCs can decrease HG-induced myocardial fibrosis and cardiomyocyte apoptosis by activating the cGMP/PKG pathway, and may play a role in the prevention and treatment of diabetic cardiomyopathy.
Keywords
- phosphodiesterase-5
- BMSCs
- diabetic cardiomyopathy
- cGMP/PKG pathway
- myocardial fibrosis
- cardiomyocytes apoptosis
The morbidity and mortality of Diabetes Mellitus (DM) are increasing annually [1]. DM is an important risk factor for the development of cardiovascular diseases. Epidemiological studies have found that the incidence of cardiovascular diseases in DM patients is 2–3 times higher than that in non-DM patients [2]. The structure and function of myocardial tissue in DM patients is affected by hyperglycemia, which can lead to diabetic cardiomyopathy (DC) [3]. The pathological characteristics of DC include myocardial hypertrophy, myocardial collagen proliferation, increased glycogen and lipid droplet deposition, and myocardial interstitial fibrosis. These pathological changes will lead to ventricular remodeling and ultimately decreased cardiac function [4]. Myocardial fibrosis is considered to be one of the main causes of cardiac dysfunction in DC patients [5]. Therefore, inhibition of the development of myocardial fibrosis plays an important role in preventing DC in DM patients.
Studies have found that the cyclic guanosine monophosphate (cGMP)/protein kinase G (PKG) pathway is an important mediator of cardiac protection [6]. cGMP is a ubiquitous second messenger in cells, and its role is mainly mediated by PKG. The activated PKG can act on its downstream targets and is involved in the regulation of vascular tension, myocardial contractility, myocardial hypertrophy, interstitial fibrosis and cardiac remodeling [7]. Phosphodiesterase-5 (PDE-5) is the key enzyme to degrade intracellular cGMP, and PDE-5 inhibitors have been found to play a cardioprotective role by increasing cGMP activity and activating PKG [8]. Therefore, we hypothesized that PDE-5 inhibitors may improve myocardial fibrosis through the cGMP/PKG pathway, and prevent DC.
Mesenchymal stem cells (MSCs) are ideal target cells for cell replacement therapy and gene therapy, especially bone marrow-derived MSCs (BMSCs). BMSCs have many advantages, including easy access to materials, the absence of ethical issues, easy transfection and stable expression of exogenous genes, and the potential for repair of myocardial damage [9, 10]. Several studies have shown that transplanted BMSCs can protect the myocardium and improve cardiac function through the mechanisms of antioxidation, antifibrosis, antiapoptosis, and promoting angiogenesis and repairing cardiomyocytes [10, 11, 12]. Therefore, using, BMSCs as the cell carrier of PDE-5 inhibitors to limit collagen proliferation and interstitial fibrosis of myocardial cells may be an effective method for the treatment of DC.
In this study, we investigated the effects of BMSCs that overexpressed or knocked down PDE-5 on the biological function of cardiomyocytes and fibroblasts induced by high glucose (HG) and the cGMP/PKG pathway, in order to prevent and potentially treat DC.
Sprague-Dawley (SD) rats (1-3-day-old, n = 3) supplied by Shanghai SLAC
Laboratory Animal Co., Ltd (Certificate No. SCXK (Hu) 2017-0005, Shanghai, China)
were raised under a 12-hour-light/-dark cycle with 22
The rats were euthanized using excessive pentobarbital sodium, and then the
ventricular tissues of rats were collected, chopped on ice and digested with type
II collagenase (1148090, Merck, Darmstadt, Germany) to obtain a cell suspension.
High-and low-density Percoll (P4937, Merck, Darmstadt, Germany) were prepared in
advance. Low-density Percoll (0.407 g/mL) was added slowly over high-density
Percoll (1.017 g/mL), and the cell suspension was added over Percoll and
centrifuged at 1700 g for 30 minutes. The cells on the upper layer of the
centrifuge tube were fibroblasts, and the cells on the lower layer were
cardiomyocytes. The cardiomyocytes were cultured in rat cardiomyocytes complete
medium (CM-R073, Procell, Wuhan, Hubei, China), and the fibroblasts were cultured
in rat myocardial fibroblast complete medium (CM-R074, Procell, Wuhan, Hubei,
China). All cells were maintained in a humidified atmosphere at 37 °C
with 5% CO
The second generation of cardiomyocytes and fibroblasts were inoculated into a 6-well plate with cell slides. After 48 hours of incubation, the cells were fixed with 4% paraformaldehyde (abs9179, absin, Shanghai, China) and then reacted with 0.3% Triton-X 100 (A1009, Applygen, Beijing, China) for 3 minutes. Cells were blocked with 3% BSA (abs9157, absin, Shanghai, China) and incubated with primary antibody overnight at 4 °C and with the secondary antibody at room temperature for 1 hour the following day. Cells were then sealed with antifade mounting solution with DAPI (C1211, Applygen, Beijing, China). The final results were observed under a fluorescence microscope (Ts2-FC, Nikon, Tokyo, Japan). Information on the antibodies is shown in Table 1.
| Name | Catalog | Manufacturer |
| Cardiac troponin I | sc-365446 | Santa cruz, Dallas, TX, USA |
| Vimentin | ab8069 | abcam, Cambridge, UK |
| α-SMA Antibody | AF1032 | Affinity, Chicago, IL, USA |
| Nkx2.5 Antibody | DF2699 | Affinity, Chicago, IL, USA |
| Goat anti rabbit (Alexa Fluor |
ab150077 | abcam, Cambridge, UK |
| PDE-5 | ab259945 | abcam, Cambridge, UK |
| cGMP | sc-21727 | Santa cruz, Dallas, TX, USA |
| PKG | 13511S | Cell signaling technology, Danvers, MA, USA |
| AF7018 | Affinity, Chicago, IL, USA | |
| Goat anti rabbit | ab205718 | abcam, Cambridge, UK |
| Goat anti mouse | ab6789 | abcam, Cambridge, UK |
Rat BMSCs were purchased from Procell Life Science&Technology Co.,Ltd (CP-R131,
Wuhan, Hubei, China). Rat BMSCs were cultured in a complete medium (CM-R131,
Procell, Wuhan, Hubei, China) and maintained in a humidified atmosphere at 37
°C with 5% CO
The gene sequence of rat PDE-5 was obtained from the NCBI database and then
amplified by polymerase chain reaction (PCR). The amplified gene sequence was
inserted into the pRRL WS1.6 GFP vector (K4820-01, Invitrogen, Waltham, MA, USA),
named overexpression (oe)-PED-5, and the empty vector was regarded as a negative
control (NC). The PDE-5 specific short hairpin RNA (shRNA) was synthesized by
VectorBuilder (Guangzhou, Guangdong, China) with the target sequence
(5
Cardiomyocytes or fibroblasts, were divided into 4 groups (control group, model
group, PDE-5 OE group and PDE-5 shRNA group). Cardiomyocytes or fibroblasts (1
Cell counting kit 8 (CCK8, C0005, Topscience, Shanghai, China) was used to
detect the cell viability. Cardiomyocytes and fibroblasts in logarithmic phase
were taken and prepared into a 3
Rat collagen (col)-I (ml058800, Mlbio, Shanghai, China), col-III (MM-0765R2, Meimian, Yancheng, Jiangsu, China), tissue inhibitor of metalloproteinase (TIMP)-1 (ml003040, Mlbio, Shanghai, China), desmin (MM-70789R2, Meimian, Yancheng, Jiangsu, China), Matrix metalloproteinase (MMP)-1 (ml002968, Mlbio, Shanghai, China) and troponin (Tn)-I (ml003202, Mlbio, Shanghai, China) ELISA kits were used to determine the cytokine levels in rat cardiomyocytes and fibroblasts. Briefly, the cells were centrifuged at 1000 g for 10 minutes to obtain a supernatant. The standards and cell supernatants were diluted and added to the ELISA plates for incubation for 30 minutes. The biotin-labeled antibody was used to unbind the antigen in the cell supernatant, followed by binding to biotin by affinity streptavidin–horseradish Peroxidase (HRP). After the color was developed by the substrate developing solution, the reaction was stopped. The absorbance at 450 nm was determined with a microplate reader and the cytokines levels were calculated according to the instructions.
The Annexin V-FITC/PI Apoptosis Detection Kit (556547, BD, Franklin Lakes, NJ,
USA) was used to determine the apoptosis of rat cardiomyocytes and fibroblasts.
The cells were collected and the cell concentration was adjusted to 1
Total protein was extracted from rat cardiomyocytes and fibroblasts by the
Protein Extraction Kit (G2002, Servicebio, Wuhan, Hubei, China). Then the protein
was quantified by the BCA kit (T14514, Topscience, Shanghai, China) was separated
by SDS-PAGE gel and electroblotted to the PVDF membrane (WJ001, Epizyme,
Cambridge, MA, USA). After blocking, the membrane was probed with the primary
antibody and the secondary antibody which are shown in Table 1. Finally, the
protein signals were detected by the ECL luminescence reagent (SQ202, Epizyme,
Cambridge, MA, USA) using the Imaging System (610020-9Q, Qinxiang, Shanghai,
China).
Total RNA was extracted from rat cardiomyocytes and fibroblasts by the Total RNA
Purification Kit (EZB-RN4, HiFunBio, Shanghai, China). Then the reverse
transcription and qPCR was conducted by the cDNA Synthesis Kit (EZB-RT2,
HiFunBio, Shanghai, China) and SYBR Green qPCR Mix (A0001, HiFunBio, Shanghai,
China) on a Real-Time PCR system (CFX Connect, Bio-rad, Hercules, CA, USA). The
mRNA levels were normalized to
| List of oligonucleotide sequences | 5 |
| Primers for qRT-PCR | |
| PDE-5 Forward | GCTATGTTGCCCTTTGGAGAC |
| PDE-5 Reverse | TAGCGATCCGCAGAGATGAGT |
| cGMP Forward | TGATGAGGAGTGATGGGGCAG |
| cGMP Reverse | TGCAGAGAAGTGGCATCCAAG |
| PKG Forward | CTCCACAAATGCCAGTCGGT |
| PKG Reverse | AAGGTGTCTCCTCTTGCACC |
| AAGGTCGGTGTGAACGGATTT | |
| CTTTGTCACAAGAGAAGGCAGC |
Data are presented as mean
As shown in Fig. 1A, cardiomyocytes and fibroblasts began to adhere to the wall
after 4 hours of culture, and their shapes gradually changed from round to
spindle. After 24 hours of culture, cardiomyocytes began to extend out from
pseudopodia, showing a triangular or polygonal shape. After 72 hours of culture,
cardiomyocytes were interconnected and interwoven into a network, forming
single-layer cardiomyocytes or cell clusters. After 24 hours and 48 hours of
culture, fibroblasts also showed fusiform or polygonal shape, and scattered
growth. After 72 hours of culture, cells proliferated faster, grew and gathered,
and showed spiral and radial shapes. Cardiomyocytes specifically express Cardiac
troponin I, Nkx2.5 and fibroblasts specifically express Vimentin,
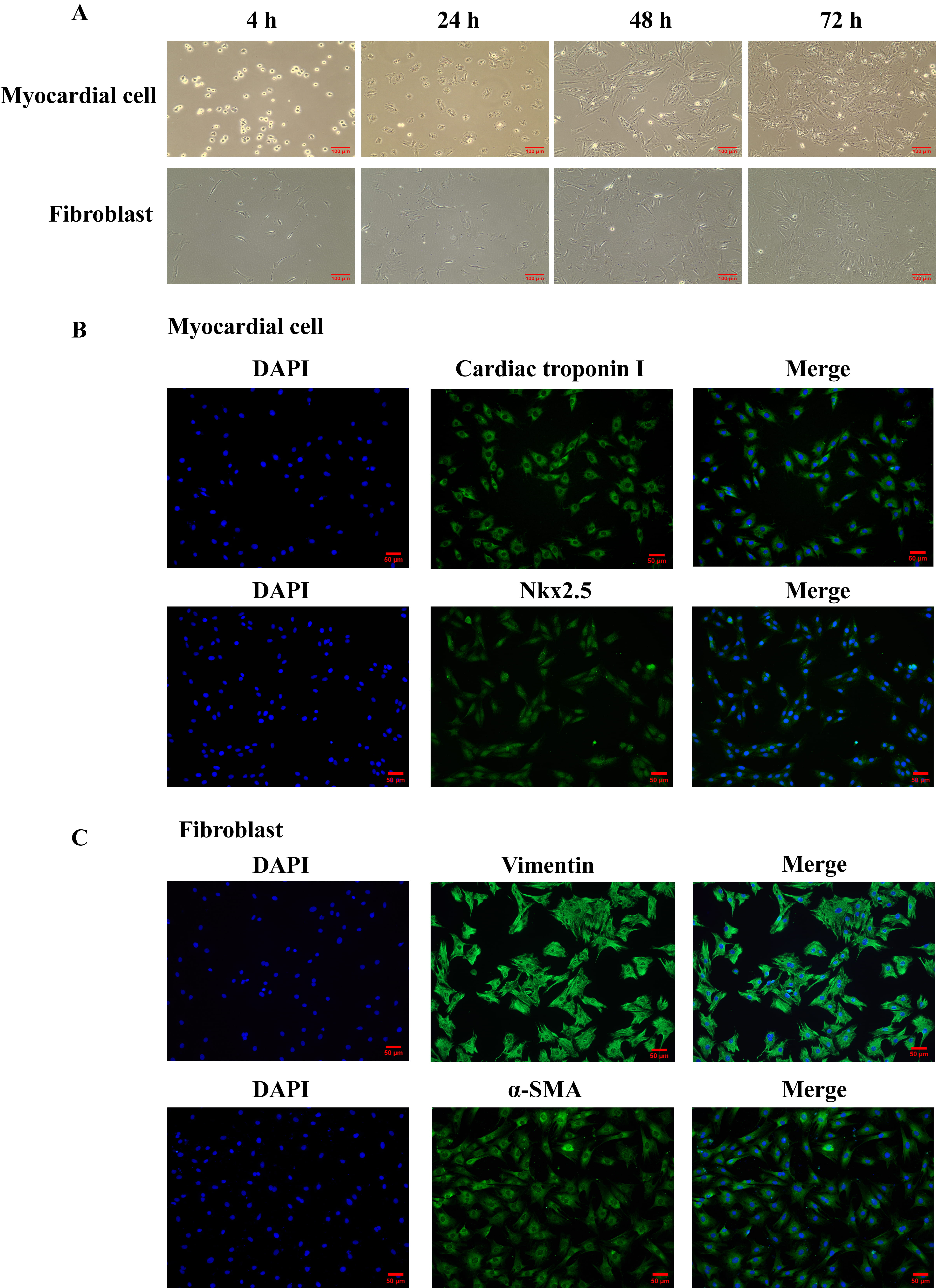 Fig. 1.
Fig. 1.Isolation and identification of neonatal rat primary
cardiomyocytes and fibroblasts. (A) The morphology of rat primary cardiomyocytes
and fibroblasts cultured for 4 hours, 24 hours, 48 hours and 72 hours was
observed under inverted microscope (magnification, 100
| Markers | Total number of cells | Number of positive cells | Positive percent (%) |
| Cardiac troponin I | 86.67 |
84.33 |
97.33 |
| Nkx2.5 | 133.00 |
131.00 |
98.49 |
| Vimentin | 90.67 |
88.00 |
97.21 |
| 103.33 |
102.00 |
98.72 |
Data were presented as mean
We determined the overexpression efficiency of PDE-5 by qRT-PCR and showed that
the expression of PDE-5 significantly increased after BMSCs were transfected with
PDE-5 overexpression plasmid (Fig. 2A, p
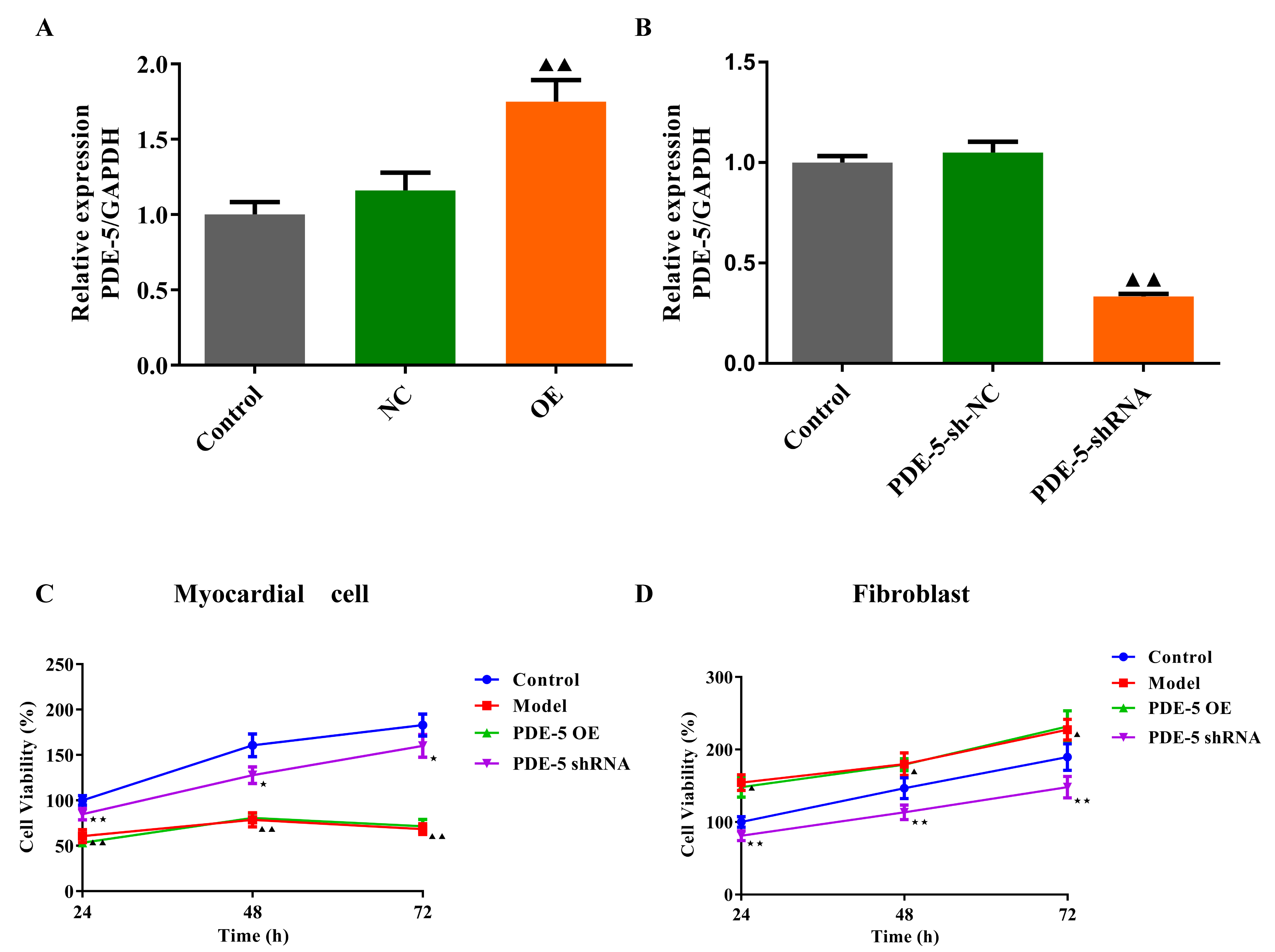 Fig. 2.
Fig. 2.The effects of PDE-5 on the viability of rat fibroblasts and
cardiomyocytes. The transfection efficiency of PDE-5 overexpression plasmid (A)
and the PDE-5 sh-RNA (B) in rat BMSCs (mean
After HG culture, the contents of Col-I, Col-III, TIMP-1 and Desmin secreted by
fibroblasts gradually increased, while MMP-1 secreted by fibroblasts and Tn-I
secreted by cardiomyocytes decreased in a time-dependent manner (Fig. 3A–F,
p
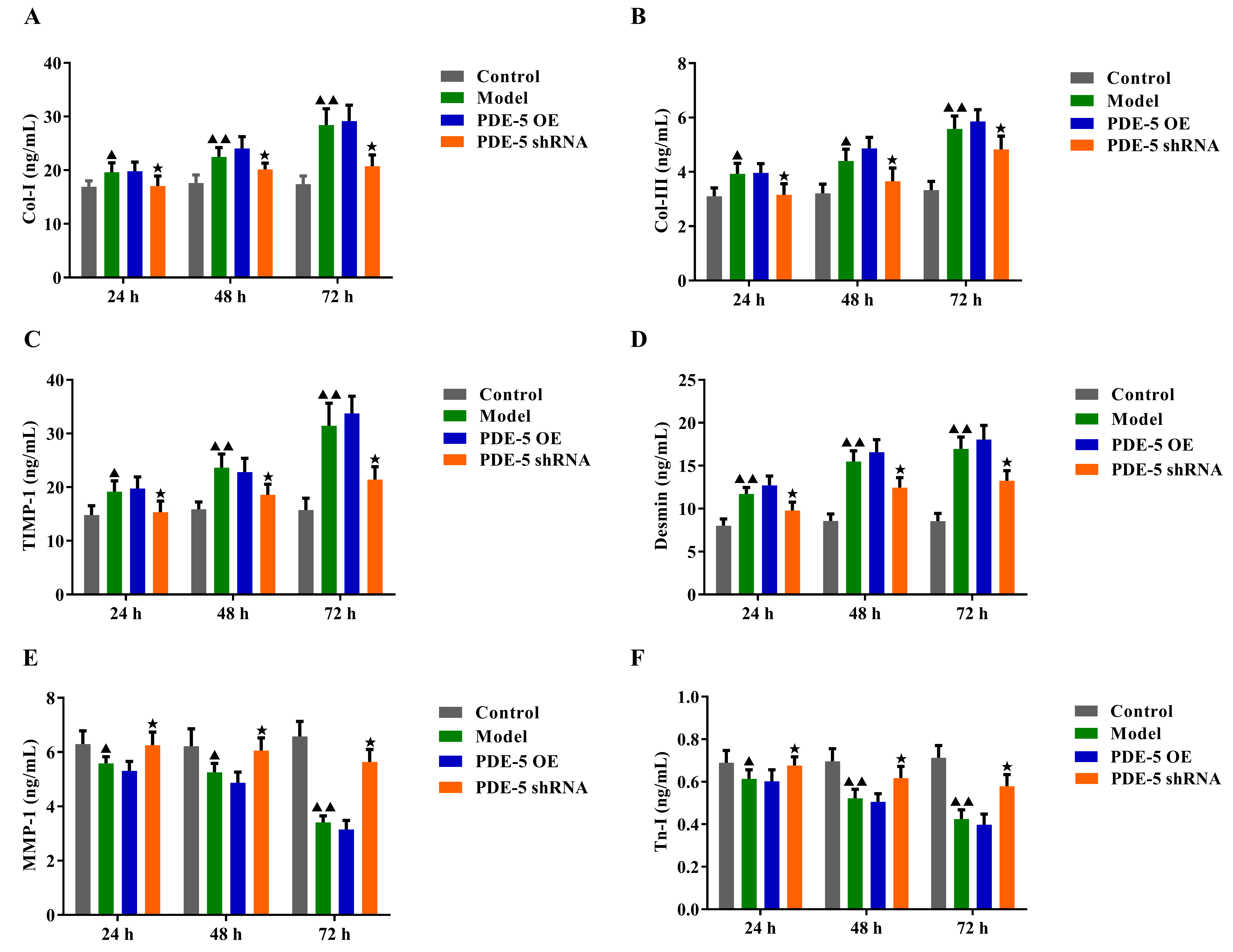 Fig. 3.
Fig. 3.The effects of PDE-5-modified MSCs on the contents of
Col-Ⅰ, Col-III, Desmin, MMP-1, TIMP-1 and Tn-I. (A–E) ELISA
was used to detect the contents of Col-Ⅰ, Col-III, Desmin, MMP-1, and TIMP-1 in
the supernatant of rat fibroblasts. (F) ELISA was used to detect the contents of
Tn-I in the supernatant of rat cardiomyocytes (mean
HG culture increased the apoptosis of rat fibroblasts and cardiomyocytes
compared to the normal group cells (Fig. 4A,B, p
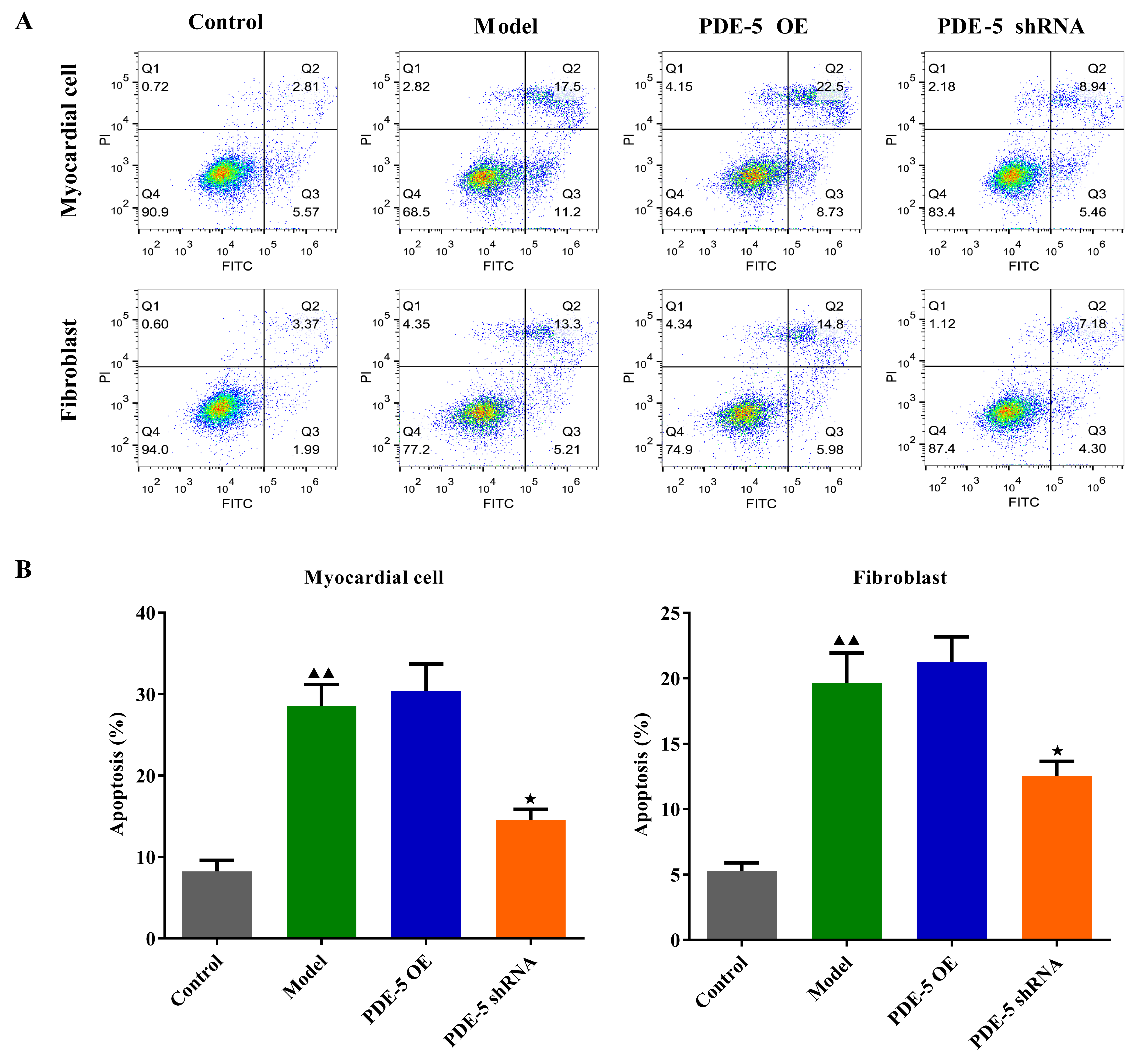 Fig. 4.
Fig. 4. The effects of PDE-5-modified MSCs on apoptosis of fibroblasts
and cardiomyocytes. (A,B) The apoptosis of fibroblasts and cardiomyocytes in
each group was determined by flow cytometry (mean
As shown in Figs. 5A–D,6A,B, we found that the protein and mRNA levels
of PDE-5 were increased (p
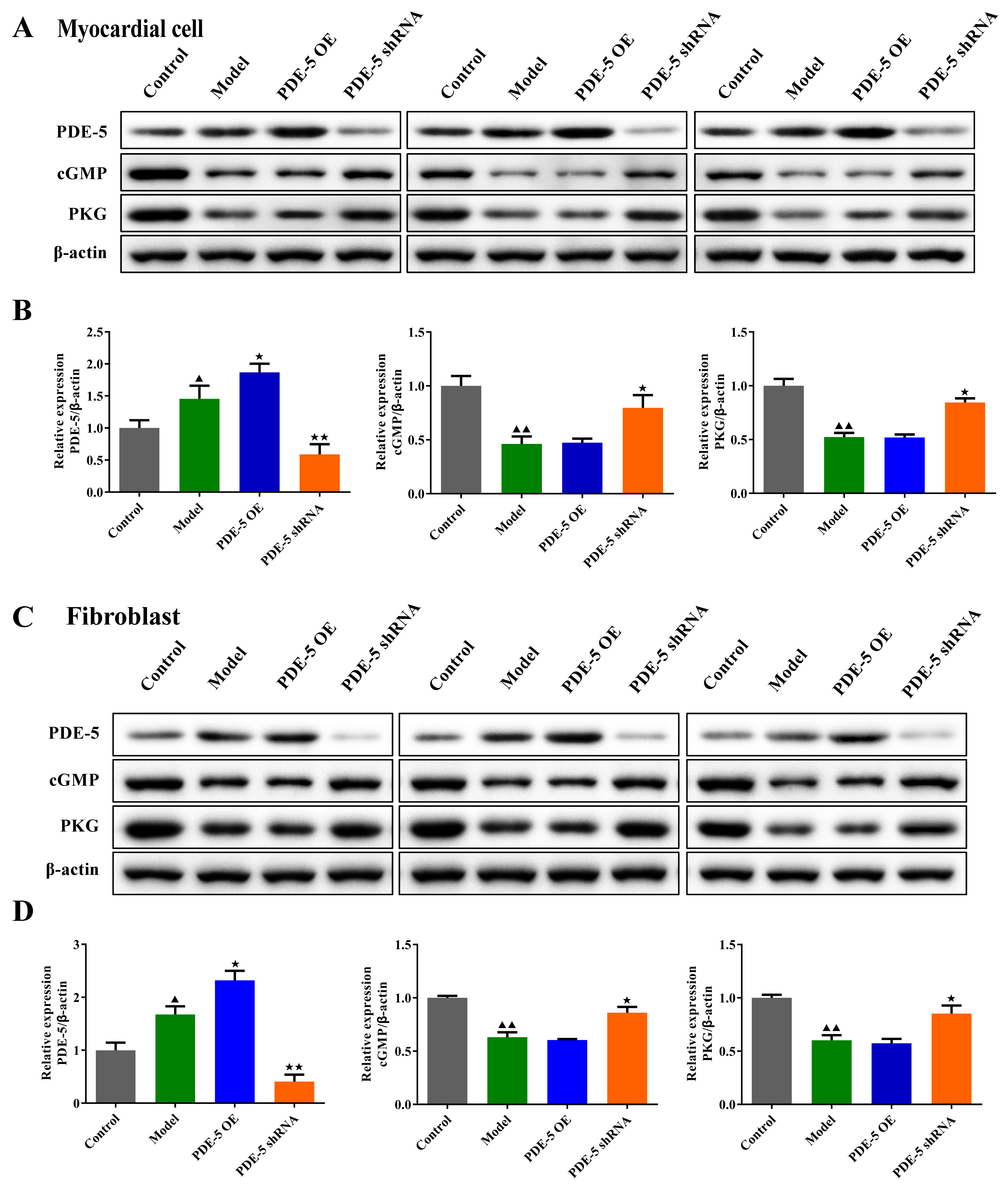 Fig. 5.
Fig. 5.The effects of PDE-5-modified MSCs on the protein levels of
PDE-5, cGMP and PKG in fibroblasts and cardiomyocytes. Western blot was used to
detect the protein levels of PDE-5, cGMP and PKG in rat cardiomyocytes (A,B) and
fibroblasts (C,D).
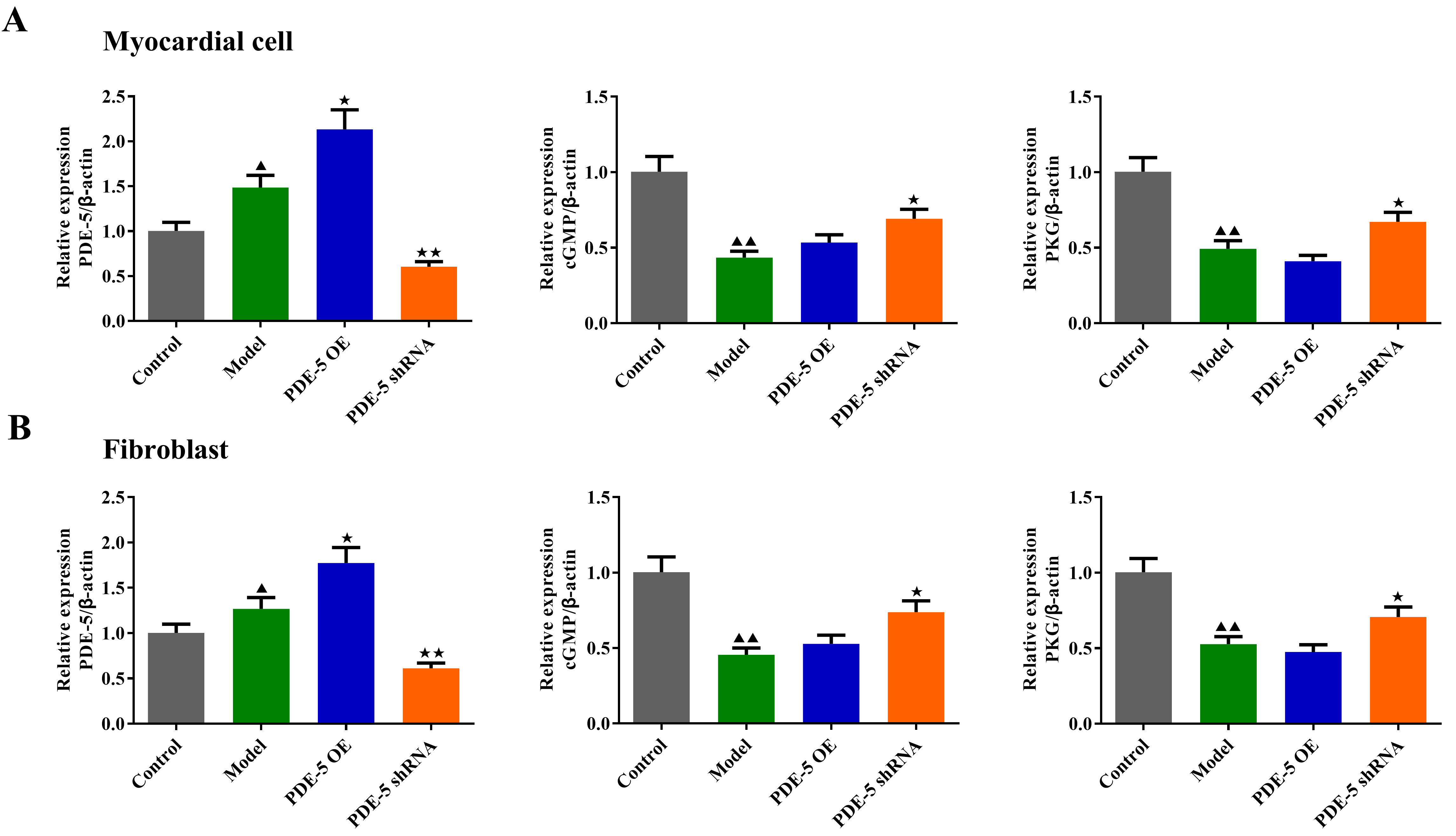 Fig. 6.
Fig. 6.The effects of PDE-5-modified MSCs on the mRNA levels of PDE-5,
cGMP and PKG in fibroblasts and cardiomyocytes. qRT-PCR was used to detect the
mRNA levels of PDE-5, cGMP and PKG in rat cardiomyocytes (A) and fibroblasts (B).
Cardiac fibrosis, the hallmark of DC, contributes to the structural remodeling of the heart, culminating in decreased cardiac function, heart failure, and arrhythmias. It is reported that the process of cardiac fibrosis is mainly mediated by the activation of cardiac fibroblasts. Several studies have shown that there is crosstalk between cardiac fibroblasts and cardiomyocytes, which promotes the development of DC [15, 16]. Therefore, in this study, fibroblasts and cardiomyocytes were cultured separately, in order to further explore the mechanism of myocardial fibrosis in DC.
It is well known that fibroblasts and cardiomyocytes can be regulated by HG. HG can improve the activity of fibroblasts and activate fibroblasts, and at the same time, it can induce cardiomyocyte apoptosis [17, 18]. Activated fibroblasts can affect cardiac function through direct and indirect effects on cardiomyocytes. Under the induction of HG, fibroblasts will secrete more fibrous collagen, such as Col-I and III, which are responsible for the changes of cardiac function [19]. MMP-1 can mediate the degradation of Col-I, while TIMP-1 is the inhibitor of MMP-1. Therefore, HG-induced fibroblasts will secrete more TIMP-1 and reduce the release of MMP-1, thus promoting the production of Col-I [20]. Desmin is the protein of mesenchymal cells, promoting extracellular matrix remodeling and mediating collagen degradation [21]. Tn-I is a specific biomarker of myocardial cells, and the down-regulation of Tn-I expression induced by HG indicates myocardial cell necrosis [22]. Consistent with a previous study [21], in our study, HG-induced fibroblasts secreted more Desmin, which indicates that fibroblasts are more likely to change to the myofibroblast phenotype. HG induction led to fibroblast activation, collagen deposition and phenotype, as well as cardiomyocyte apoptosis, which indicates that protecting cardiomyocytes by limiting fibrosis might be an important strategy for the treatment of DC.
Previous studies have shown that BMSCs can treat DM and heart disease [23]. However, the effect of BMSCs on reducing cardiac fibrosis needs to be further investigated. Ke Meng et al. [13] suggested that transplantation of adiponectin-modified BMSCs into DM rats could alleviate HG-induced cardiac fibrosis. In this study, we proposed a strategy of PDE-5-silenced BMSCs to treat cardiac fibrosis. The cardioprotective effect of PDE-5 inhibitor has been widely confirmed [24]. Hoke et al. [25] reported that implantation of PDE-5-silenced adipose-derived stem cells into a myocardial infarction model in mice could significantly improve cardiac function and reduce myocardial fibrosis and cardiomyocyte apoptosis. Similarly, we demonstrated the therapeutic effect of PDE-5-silenced BMSCs on DC by co-culture with HG-induced fibroblasts and cardiomyocytes, that PDE-5-silenced BMSCs significantly inhibited fibroblast activation, collagen deposition, and cardiomyocyte apoptosis.
The cardio-protective effect (improvements in the pulmonary circulation, cardiac remodeling and diastolic function) of PDE-5 inhibitors is mainly mediated by activating the cGMP/PKG pathway [26]. Saihakoka et al. [27] found that PDE-5 inhibitors can increase cGMP and PKG activities in the heart, thus alleviating adriamycin-induced cardiomyopathy. However, it has not been reported whether PDE-5 inhibitors can alleviate myocardial fibrosis in DC patients by activating the cGMP/PKG pathway. Wei et al. [28] reported that myocardial injury in DM rats could be alleviated by regulating the cGMP/PKG pathway. Qin et al. [29] emphasized that activating the cGMP/PKG pathway in cardiac fibroblasts can alleviate HG-induced cardiac fibrosis [28]. Consistent with previous studies, in this study, we found that HG-induced inactivation of the cGMP/PKG pathway in cardiac fibroblasts and cardiomyocytes was reversed by PDE-5-silenced BMSCs, suggesting that PDE-5 silenced-BMSCs may alleviate HG-induced cardiac fibrosis and myocardial injury by regulating the cGMP/PKG pathway. However, there are limitations in this study, such as the effect of PDE-5 on BMSCs in vivo. In the future, we will study the effect of PDE-5 on myocardial fibrosis in DC rats under physiological conditions by constructing a diabetic rat model.
This study shows that PDE-5-silenced BMSCs can alleviate the HG-induced collagen deposition, myocardial fibrosis and cardiomyocyte apoptosis by activating the cGMP/PKG pathway, suggesting that PDE-5 inhibitor-transfected BMSCs have the potential to prevent and treat diabetic cardiomyopathy. These findings may contribute to the development of therapeutic stem cell transplantation for DC.
The relevant data to support the methods and results are available on Figshare (DOI: 10.6084/m9.figshare.21501054).
XF, QH and JM designed the research study. QH, JZ, and LJ performed the research. QH, HW and LJ analyzed the data. XF, QH, HW and JM QH interpreted the data, QH, JZ, LJ and HW wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work to take public responsibility for appropriate portions of the content and agreed to be accountable for all aspects of the work in ensuring that questions related to its accuracy or integrity.
The study was approved by Animal Experimentation Ethics Committee of Zhejiang Eyong Pharmaceutical Research and Development Center (Certificate No. SYXK (Zhe) 2021-0003, Hangzhou, China).
The authors thank all those who supported them in animal experiments and helped them to manage the data.
This research was funded by the Public Welfare Technology Application Research Project of Zhejiang Province, China, grant number LGD20H070002 and LGD21H020004 granted to HQ and FX. Respectively, the Research Project of Zhejaing Provincial People’s Hospital, grant number ZRY2020B014 granted to HQ. The Medicine and Health Science and Technology Project of Zhejiang Province, grant number 2022KY523, 2020KY022, and 2021KY060 granted to HQ and FX, respectively. All the funders were not involved in the manuscript writing, editing, approval, or decision to publish.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






