1 Laboratory of Angiopathology, Institute of General Pathology and Pathophysiology, 125315 Moscow, Russia
2 Diabetes Research Center, School of Traditional Chinese Medicine, Beijing University of Chinese Medicine, 100029 Beijing, China
3 Petrovsky National Research Centre of Surgery, 119991 Moscow, Russia
Abstract
Psoriasis cannot be completely cured and is often difficult to diagnose, which is why the search for new effective therapies and diagnostics is a highly relevant area of research. To identify new therapeutic compounds, the first step is to study the role of various factors underlying the development of psoriasis. One such factor is oxidative stress. In this review, we will consider the role of oxidative stress at different stages of psoriasis development, as well as biomarkers of oxidative stress that can potentially be used in the diagnosis of psoriasis and antioxidants, which are likely to be applied in the treatment of this disease.
Keywords
- psoriasis
- oxidative stress
- skin
Psoriasis is a chronic non-infectious skin disease with unclear etiology and lack of sufficiently effective therapy [1]. Psoriasis is a global health problem. There are more than 100 million people in the world affected by this disease [1]. Psoriasis is characterized by an unpredictable course of the disease, as well as a high likelihood of developing a number of serious comorbidities including arthritis, Crohn’s disease, ulcerative colitis, cardiovascular disease, metabolic syndrome, and depression [1]. Psoriasis has an autoimmune nature, its key feature is the occurrence of stable inflammatory response as a result of increased infiltration of immune cells, which, in turn, leads to increased and uncontrolled proliferation of keratinocytes, which make up to 90% of epidermal cells, present from the basal to the stratum corneum, and are the primary protection of the skin from external factors [2]. The pathogenesis of psoriasis is very complex and is based on the intercellular interaction between keratinocytes, leukocytes, and other types of resident skin cells [2]. At the same time, keratinocytes play an important role both in the initial phase, accelerating inflammation, and during the maintenance phase, contributing to the development of a chronic inflammatory response [3]. The etiology of psoriasis is not completely clear, but includes both hereditary and non-hereditary factors. To date, numerous loci associated with predisposition to psoriasis have been identified, among which PSORS1 locus located on chromosome 6p21 plays the greatest role in predicting the development of this disease [4]. Non-hereditary factors include sunburn, trauma, various infectious diseases, alcohol and tobacco consumption, certain medications, and obesity [4]. Currently, there are no therapeutic agents that can completely cure psoriasis [5]. However, there are drugs that alleviate the condition of patients with psoriasis. The first line of defense for the treatment of mild to moderate psoriasis is the use of topical therapies, among which ointments and creams based on corticosteroids are most commonly used [5]. Phototherapy can also be used to treat moderate and severe psoriasis, which consists in exposing the skin to a certain dose of natural or artificial radiation [6]. For the treatment of moderate and severe psoriasis in the absence of a positive reaction to other drugs, injectable systemic drugs based on steroid hormones, retinoids, cytostatics, as well as biologics are used [6]. Each drug has side effects and should be used with caution, especially systemic drugs, for example, a known side effect of the cytostatic methotrexate is hepatotoxicity [7], for cyclosporine severe side effects include nephrotoxicity and myelosuppression [8] and for monoclonal antibodies immunosuppression may occur, which increases the risk of infection [6]. The existing imperfections of current therapeutic strategies make us think about new options for the treatment of psoriasis, which could level out the key targets of pathological foci of the disease and, thus, stop or even completely cure psoriasis. In this review, we propose to consider oxidative stress (OS), one of the significant pathological conditions of the cell, as a basis for the search for new therapeutic targets for psoriasis.
Reactive oxygen species (ROS) are important cell signaling molecules, which,
however, at high concentrations can cause severe damage to biological
macromolecules and lead to the development of a pathological condition known as
OS [9]. OS plays an important role in the pathogenesis of various chronic
diseases [10], including psoriasis, which will be discussed in more detail below.
At acceptable concentrations, ROS perform an antimicrobial function and is
involved in the activation of various important signaling pathways, including
cell proliferation and differentiation, induction of apoptosis, and regulation of
autophagy [9]. The main types of ROS include superoxide anion radical (O
OS occurs as a result of an imbalance between the production of ROS and the
functioning of the antioxidant system. A decrease in the antioxidant capacity of
the cell may result from a decrease in the activity of enzymes of the antioxidant
system, including (SOD), catalase (CAT), and(GSH-Px), as well as a decrease in
the concentration of compounds that have an antioxidant effect, both endogenous
(glutathione (GSH)) and exogenous (carotenoids, tocopherols) [16]. In a mouse
model, it was shown that genetic knockout of extracellular SOD led to
IL-23-mediated skin inflammation, which was determined by increased accumulation
of immune cells: CD4
The main symptom of autoimmune diseases, including psoriasis, is the destruction
of the organism’s own intact tissues by the immune system as a result of impaired
antigen recognition, during which autoantigens are recognized as foreign. Both
innate and adaptive immune cells are involved in the development of an autoimmune
reaction in psoriasis [16]. Under conditions of increased OS, keratinocytes begin
to secrete proteins into the extracellular space, which are recognized by immune
cells as autoantigens. The characterized autoantigens in psoriasis are proteins:
LL-37, ADAMTSL5, PLA2G4D, and keratin 17 [25]. Dendritic cells are a key
population of immune cells responsible for the development of the autoimmune
response in psoriasis. The autoantigens taken up by dendritic cells are
fragmented and exposed to the surface of the cell membrane as part of the MHC,
which leads to antigen presentation to T-cells. OS also contributes at this stage
of psoriasis pathogenesis, since ROS are stimulators of antigen presentation by
dendritic cells [26]. Skin dendritic cells are represented by epidermal dendritic
cells (called Langerhans cells) and dermal dendritic cells (myeloid and
plasmacytoid) [27]. The main pathological population of dendritic cells in
psoriasis is myeloid CD11c
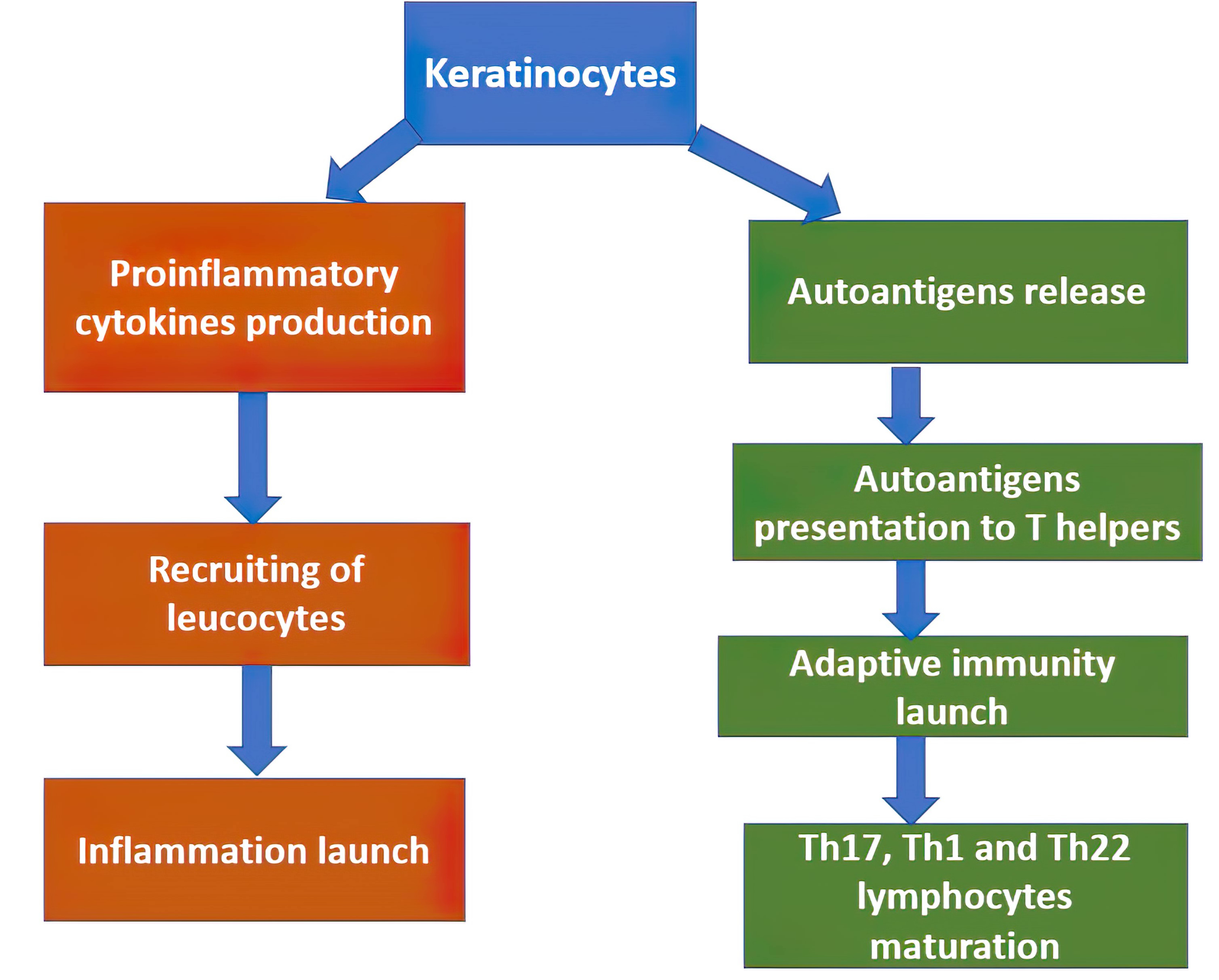 Fig. 1.
Fig. 1.Two ways of immune cells involvement in the development of psoriasis.
Keratinocytes are cells inducing an autoimmune reaction for psoriasis. In this case, the inducing effect of keratinocytes can be divided into 2 branches. The first is connected with the products of pro -inflammatory cytokines, which lead to the involvement of various leukocytes, such as: macrophages, neutrophils, dendritic cells, lymphocytes and further development with their participation of self -sustaining inflammation. The second branch is associated with the release of autoantigens, which are captured and processed by dendritic cells and are presented on their surface with T-lymphocytes, which cause an adaptive response, in which in psoriasis development Th1, Th17 and Th22 lymphocytes play the major role. At the same time, ROS directly affect the initiation and strengthening of autoimmune processes at each stage of the development of the disease.
Keratinocytes play a direct role in the intensification and subsequent development of the chronic phase of inflammation. As a result of exposure to pro-inflammatory cytokines, keratinocytes, in addition to high proliferative activity, acquire the ability to produce high levels of cytokines, such as: CXCL1/2/3, CXCL8, CCL2 and CCL20, which lead to increased recruitment of leukocytes involved in autoimmune inflammation, including: dendritic cells, macrophages, neutrophils, and Th17 lymphocytes [3]. In addition, keratinocytes secrete antimicrobial peptides, such as S100A7/8/9/12, hBD2 and LL37, which contribute to additional stimulation of the innate immune system and increased inflammation [3]. Keratinocytes produce new portions of ROS, which act as strong chemoattractants for neutrophils, promoting their invasion and accumulation in skin psoriatic lesions [32]. Neutrophils, like other immune cells in the inflammatory focus, in turn release high levels of ROS, which lead to an even greater increase in OS and the development of the chain reaction of increased inflammation, which eventually develops into a state of chronic inflammation. The general scheme of the pathogenesis of psoriasis based on the effects of ROS is shown in Fig. 2.
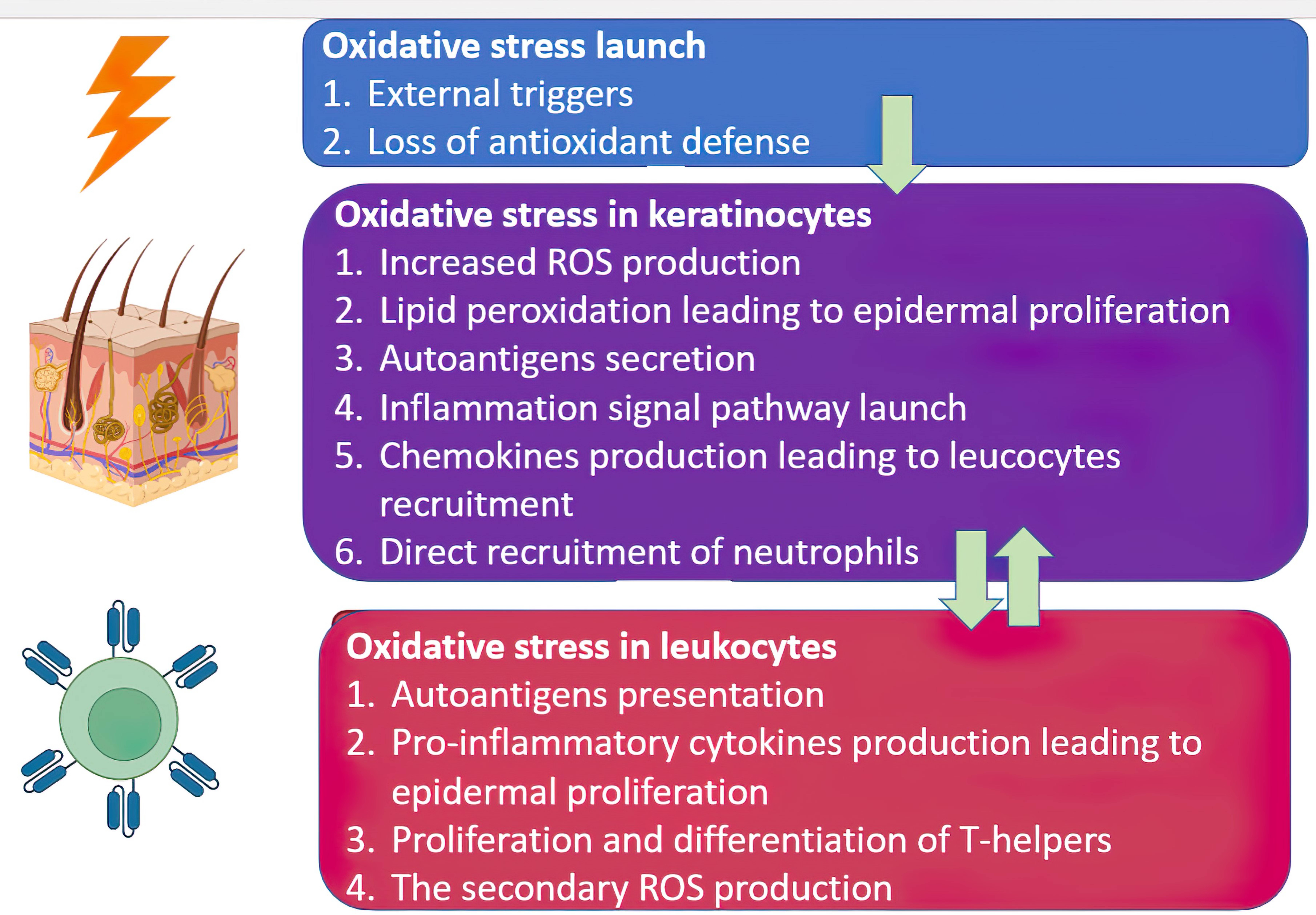 Fig. 2.
Fig. 2.General scheme of psoriasis pathogenesis based on ROS effects.
External factors (for example, UV) cause an increase in ROS production in keratinocytes and a weakening of antioxidant protection, which initiates OS, epidermal proliferation,the development of inflammation, which subsequently involves leukocytes and autoantigens released by keratinocytes resulting in an autoimmune reaction onset that becomes chronic, enhancing total inflammatory immune response.
OS is a concomitant factor in many inflammatory diseases, but the level of OS biomarkers can vary for different diseases, so finding specific biomarkers can contribute to a more accurate diagnosis of psoriasis. Thus, in a study [33], it was shown that patients with atopic dermatitis had a high level of nitrates in the urine, and patients with psoriasis were characterized by a high level of both nitrates and 8-hydroxydeoxyguanosine (8-OHdG). Thereby the potential using of the 8-OHdG detection method as a convenient test for the diagnosis of psoriasis was demonstrated. 8-OHdG is a signal molecule that detects DNA damage [34]. A high level of 8-OHdG in psoriasis can be detected not only in urine, but also in serum, which was shown in a study [35].
Another promising biomarker of OS in psoriasis is the enzyme paraoxonase-1
(PON1), which has anti-inflammatory and antioxidant activity [23]. The function
of PON1 is associated with the protection of high-density lipoproteins (HDL) from
ROS peroxidation [36]. In a study [37], it was shown that the level of PON1 in
tissues and serum was significantly lower in patients with psoriasis compared
with the control group. It was also found that a low level of PON1 was inversely
correlated with the main inflammatory markers: TNF
Another biomarker of OS in psoriasis is the sirtuin 1 protein (SIRT1) [23].
SIRT1 is an NAD-dependent deacetylase. It inhibits the inflammatory response
through interference with MAP or NF-
Thiols are compounds containing a sulfidhydryl group; the most common compound of thiols in the cell is GSH [23]. Thiols are indicators of the redox balance inside the cell, which makes them effective markers of OS [23]. The mechanism underlying the indication of OS is based on the reversible reaction of the conversion of thiols into disulfides as a result of exposure to ROS [40]. The thiol/disulfide concentration ratio just makes it possible to determine the level of OS. It has been noted that thiol/disulfide balance is disturbed in psoriasis [41]. Based on this mechanism, a quantitative colorimetric test was developed to quickly determine the level of thiol/disulfide homeostasis in plasma [42]. When testing , serum samples were analyzed from healthy volunteers, patients with degenerative and proliferative diseases. The highest concentration of disulfides was found in patients with proliferative diseases, and the lowest in healthy volunteers.
In a study [43], compounds of the biopyrrin group were identified as biomarkers of OS in psoriasis. This clinical study was conducted by measuring urinary levels of biopyrrins in patients with chronic plaque psoriasis and healthy controls. Bilirubin, which is a cellular antioxidant, is able to bind to ROS, which leads to the formation of a number of hydrophilic metabolites, which are biopyrrins and are excreted in the urine due to their hydrophilic properties. There was a significant difference between the level of biopyrrins in the control group and patients with psoriasis. Also, a certain level of biopyrrins correlated with the severity of the disease. It should be considered that the described markers of OS are not precisely markers of the development of psoriasis, however, as can be seen from the described studies, the definition of their levels can be used as one of the tools in the diagnosis of psoriasis. Data on biomarkers of OS in psoriasis are summarized in Table 1.
| Biomarkers | Detectable medium | Determination algorithm |
|---|---|---|
| 8-hydroxydeoxyguanosine | urine and serum | Increased level |
| Paraoxonase-1 | tissues and serum | Decreased level |
| Sirtuin 1 | skin fibroblasts | Decreased level |
| Thiols | serum | Transformation to disulfides |
| Biopyrrins | urine | Increased level |
Curcumin is a natural polyphenol found in the roots of the Curcuma longa plant. It is a strong antioxidant and is currently widely used in dietary supplements [44]. At the same time, there are a number of clinical studies that have shown the potential therapeutic efficacy of using curcumin as a therapeutic agent in the treatment of psoriasis. In studies with topical application of curcumin, an improvement in skin lesions was observed in patients with psoriasis [45, 46]. Clinical efficacy has also been observed in studies with oral curcumin, including when curcumin is combined with conventional treatments for psoriasis. Thus, in a study [47], therapy of methylprednisolone in combination with oral curcumin was more effective than single therapy of methylprednisolone with the addition of placebo. In addition to a decrease in the Psoriasis Area and Severity Index (PASI), patients treated with curcumin, compared with the control group without curcumin, had a decrease in serum IL-22, however, when comparing IL-17, no differences were observed between groups. At the same time, the anti-inflammatory effect of curcumin, based, among other things, on a decrease in IL-17, was shown in a mouse model [48]. However, there is also evidence from a study in which curcumin had no significant effect in the treatment of patients with moderate to severe psoriasis [49]. The advantage of the potential use of curcumin in the treatment of psoriasis is the minimal side effects noted in several clinical studies [50, 51]. It should be noted that the results of the use of curcumin monotherapy for the treatment of psoriasis are available only for phase I and II clinical trials, the results of phase III and IV clinical trials are available for combination therapy with curcumin and conventional treatments. Based on this, curcumin may find use in combination therapy as an adjuvant for the treatment of psoriasis in the coming years, but the introduction into clinical practice as a standalone drug will not happen soon in our opinion.
Coenzyme Q10 (CoQ10), also known as ubiquinone, is a component of the mitochondrial electron transport chain, as well as a strong antioxidant molecule, and is currently included in many dietary supplements [52]. As with curcumin, the most promising strategy to enter clinical practice is the combined use of CoQ10 in combination with drugs already proven effective in the treatment of psoriasis. For example, in a clinical study [53], it was shown that combined therapy with the biological drug adalimumab used in conjunction with CoQ10 led to an improvement in the correlation between PASI and Dermatology Life Quality Index (DLQI) indicators. At the same time, the combined use of several antioxidant agents may also have a high potential for further use. Thus, in a study [54], after the introduction of the nutritional supplements complex containing CoQ10, vitamin E and selenium to patients with psoriasis, the improvement in the clinical parameters of patients was observed, and the normalization of the value of OS markers was also shown.
Selenium is a trace element widely used in food supplements. It is known that
the level of selenium in patients with psoriasis is lower than in healthy people
[55]. It was found that selenium is able to modulate the immune response by
changing the expression of cytokines and their receptors, and also helps to
increase the resistance of immune cells to OS [55]. In the clinical study [56],
it was noted that the combined intake of selenium and vitamin E increased the
activity of (GSH-Px), an enzyme with antioxidant activity, in serum and whole
blood.It should be noted, that taking these supplements did not reduce the
severity of psoriasis. But the prospective study [57] showed that a decrease in
serum selenium levels was associated with an increase in the severity of
psoriasis. In another prospective study [58], it was found that daily intake of
400 mkg of selenium for 6 weeks led to an increase in the number of CD4
Omega-3 polyunsaturated fatty acids (PUFAs) are the main components of fish oil. The role of these compounds in reducing OS has been demonstrated in various tissues [60, 61]. At the same time, the main omega-3 polyunsaturated fatty acids considered for the treatment of psoriasis are docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA) [62]. Two ongoing prospective studies have shown that supplementation with omega-3 fatty acids improved PASI scores in patients with psoriasis [63, 64]. In a study [65] in patients receiving daily capsules containing a mixture of EPA and DHA, it was shown to reduce itching, erythema and the area of the affected skin. In another clinical study, which also used a mixture of EPA and DHA, in addition to clinical improvement, the mechanisms underlying it were demonstrated: inhibition of the synthesis of leukotriene B4 in peripheral leukocytes in vitro, as well as a decrease in the production of malondialdehyde by platelets and a decrease in abnormalities in the structure of the lipid membrane in erythrocytes [60]. In a randomized trial [66] for patients treated with PUFAs as an additional treatment for psoriasis, it was possible to reduce the use of non-steroidal anti-inflammatory drugs (NSAIDs). Also, for these patients, compared with the control group, the levels of leukotriene B4 were reduced. In a clinical study [67], patients with psoriasis used a moisturizer containing linoleic acid and ceramide, which improved the therapeutic effect of topical glucocorticoids and delayed the occurrence of relapses. It has been shown that the use of this agent improves the PASI score. It is known that the imbalance between the formation and destruction of the extracellular matrix (ECM) is an important step in the pathogenesis of psoriasis, so a change in protein metabolism in the ECM can be a sign of both recovery and disease intensification. However, a clinical study on the level of protein biomarkers in the ECM in psoriasis did not reveal any differences between the control group and the group receiving PUFAs as therapy [68]. In addition, a number of studies have shown the effectiveness of combination therapy when taking omega-3 polyunsaturated fatty acids in conjunction with other treatments for psoriasis [62]. Although several studies have shown no clinical improvement in psoriasis patients with DHA and EPA, and some specific side effects such as fishy taste, nausea, and diarrhea, these compounds are promising treatment options for psoriasis [62]. However, phase III and IV clinical trials with a large number of participants and medical centers are required to fully demonstrate their clinical effectiveness.
Quercetin is a plant flavonoid used in many dietary supplements. Due to its
antioxidant and anti-inflammatory properties, quercetin is often considered by
researchers as a potential therapeutic agent for a number of inflammatory and
autoimmune diseases, including psoriasis. In a study [69], a mouse model of
imiquimod-induced psoriasis was created. It was found that administration of
quercetin to mice reduced tissue damage and temperature, as well as reduced the
inflammatory response, which was measured by reducing the concentration of
TNF-
Baicalein, another plant flavonoid, is also being considered as a therapeutic
agent for psoriasis. Baicalein was investigated in a study similar in design to
that previously described for quercetin in an imiquimod-induced mouse psoriasis
model [73]. In addition to improving the clinical picture, expressed in a
decrease in erythema and scaling, baicalein significantly reduced the production
of cytokines responsible for the development of psoriasis: IL-17A, IL-23, IL-22a
and TNF-
The effects of the described antioxidants on the course of psoriasis are summarized in Table 2.
| Antioxidant | Influence on the psoriasis pathogenesis | Clinical manifestation | Potential for introduction into clinical practice |
|---|---|---|---|
| Curcumin | IL22 and IL17 (in mouse model) decrease | Improvement in skin lesions, PASI decrease | Use in combination therapy |
| CoQ10 | OS decrease | PASI and DLQI values normalization | Use in combination therapy |
| Selenium | Increase activity of GSH-Px, CD4 |
Not detected | Unknown |
| PUFAs | Malondialdehyde and leukotriene B4 decrease | PASI decrease, Reducing of itching and erythema, Late relapses, Reducing of NSAIDs taking | Possible use in monotherapy |
| Quercetin | Increase activity of GSH, CAT and SOD, Inhibition of NF-κB, IKK |
PASI decrease, Skin protection from UV and chemical compounds | Possible use as a prophylactic agent |
| Baicalein | Increase production of cytokines: IL-17A, IL-23, IL-22 b and TNF- |
Decrease in erythema and scaling | Unknown |
PUFAs, Polyunsaturated fatty acids; GSH-Px, Glutathione Peroxidase; PASI, Psoriasis Area and Severity Index; DLQI, Dermatology Life Quality Index; NSAIDs, Non-steroidal anti-inflammatory drugs; UV, Ultraviolet light; CAT, Catalase; SOD, Superoxide dismutase; MMP-1, Metalloproteinase-1; COX-2, Cyclooxygenase-2; MDA, Malondialdehyde.
OS is an important factor in the pathogenesis of psoriasis, on the basis of
which tools for the diagnosis and therapy of this disease can be developed, as
shown in the previous sections. Nevertheless, several important questions remain,
the answers to which will help shed light on the further fight against psoriasis,
as well as other chronic autoimmune diseases. First of all, an important step is
the search for new biomarkers of OS, which can be effectively used as useful
tools for differential diagnosis of psoriasis, OS as was shown in the example of
8-OHdG. It is also important to differentiate the quantitative values of the
biomarkers content depending on the severity of psoriasis. The creation of these
test systems will contribute to more accurate prognosis of the disease, as well
as a more informed prescription of therapy, which will help to avoid
“unnecessary” side drug effects. It should be noted that the biomarkers presented
in this review were selected based on the criterion of a high probability of
introduction into clinical practice based on the available studies in our
opinion, however, this choice is subjective and does not mean that other
compounds are less promising (see review [76]). It is also worth pointing out
that despite the fact that the considered biomarkers of OS can be used to
diagnose psoriasis, none of them is the specific marker of psoriasis, so they
cannot be used as the only tool for diagnosing psoriasis. With regard to
antioxidant therapy, it makes sense to understand which combinations of
antioxidants and traditionally used drugs provide the best therapeutic effect in
the treatment of psoriasis. The use of antioxidant monotherapy currently does not
seem appropriate, as stated in the previous section. An important direction is
the search for compounds capable of restoring the natural antioxidant function of
cells. In this regard, the question arises: what factors are predominant in the
occurrence of OS in psoriasis (external, including exposure to ultraviolet
radiation, or internal, associated with a decrease in the activity of antioxidant
enzymes)? In the context of the described studies, a promising strategy may be to
search for compounds with theragnostic properties, which will allow combining
efforts to develop new drugs and diagnostic tests. Also an important direction in
the treatment of psoriasis is the search for new therapeutic targets that play a
significant effect in the pathogenesis of this disease. An example of these
targets is the transcription factor NRF2, the exact mechanism of which in the
context of psoriasis requires a thorough study. On the one hand, the NRF2
activates the genes of an antioxidant response (ARE) [77], on the other hand, it
triggers the proliferation of keratinocytes in psoriasis [78]. Another issue
requiring attention is the degree of development of OS in different types of
immune cells, which may allow the identification of new subtypes of leukocytes
that play a key role in the pathogenesis of psoriasis. It is also worth paying
attention to additional factors that initiate excessive proliferation of
keratinocytes, for example, a shift in the prevailing metabolic pathways. In this
regard, it is important to consider the relationship between OS and the work of
mitochondria under these conditions, as well as to identify the possibility of
developing mitochondrial dysfunction and its effect on metabolic changes. This
strategy could potentially lead to the discovery of new therapeutic targets in
the treatment of psoriasis. So, in the study [79], a change in the content of
some metabolites was revealed, as well as the modulation of the main metabolic
pathways in response to OS in the skin. So, an increase in the pentose phosphate
pathway and
OS is a detectable phenomenon in cells for various chronic diseases; its development is based on an increase in the concentration of ROS, which shifts the redox balance in cells and leads to a stress state. In psoriasis, ROS are important participants in all stages of pathogenesis, from the initial triggering of OS in keratinocytes to the development of a chronic inflammatory response. Various compounds can be used as markers of OS in psoriasis, while the detection signal indicating the onset of pathology can manifest itself in various ways: an increase in concentration, a decrease in concentration, and also the transformation of the marker into another compound. Several antioxidants are potentially being considered as potential therapies for psoriasis in combination with drugs that have proven efficacy. The use of antioxidants as monotherapeutic agents is unlikely to provide the desired therapeutic effect.
ROS, reactive oxygen species; OS, oxidative stress; ETC, electron transport chain; SOD, superoxide dismutase; RNS, reactive nitrogen species; CAT, catalase; GSH, glutathione; LDL, low-density lipoproteins; HDL, high-density lipoproteins; MHC, major histocompatibility complex; NETs, Neutrophil extracellular traps; PASI, Psoriasis Area and Severity Index; DLQI, Dermatology Life Quality Index; PUFAs, Omega-3 polyunsaturated fatty acids; DHA, docosahexaenoic acid; EPA, eicosapentaenoic acid; MDA, malondialdehyde; MMP-1, metalloproteinase-1; COX-2, cyclooxygenase-2; JAK-2, Janus kinase 2; AP-1, protein activator-1.
AB and AO designed the review plan. SG and DZ provided help and advice as experts in this phield. IE ana-lyzed the data. VS interpreted data for the clinical and pre-clinical studies cited in the manuscript. AB, IE, VS and AO wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
Not applicable.
Not applicable.
This work was supported by the Russian Science Foundation (Grant # 23-45-00031).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


