1. Introduction
Diabetic
nephropathy (DN) is considered to be the main cause of end-stage renal disease,
which is prone to high morbidity and mortality. The typical manifestation of DN
is proteinuria, and other features include thickened glomerular basement membrane
(GBM) and renal tubulointerstitial fibrosis [1, 2]. Podocyte injury is considered
to be the core link to cause proteinuria in the early stage of DN [3]. Podocytes
are highly differentiated cells attached to the lateral side of the GBM and play
an important role in maintaining renal functions [4]. Studies have shown that
ferroptosis is involved in DN progression both in vivo and in
vitro [5, 6]. Ferroptosis is a programmed cell death mode that depends on
reactive oxygen species (ROS) and iron [7].
Previous studies have suggested that the accumulation of iron and lipid peroxides
may increase renal injury in DN by increasing oxidative stress [8]. In addition,
epithelial-mesenchymal transition (EMT) in podocytes is also closely related to
DN progression. EMT of Podocytes has been reported to cause renal
tubulointerstitial fibrosis in DN [9, 10]. Besides, EMT of podocytes has also
been associated with the excretion of urinary albumin in DN [11]. At present,
antihypertensive and hypoglycemic drugs are mainly applied to treat DN
clinically, while drugs targeting the pathogenesis of DN are various but suffer
from poor efficacy and safety [12]. For this situation, Chinese herbs and their
extracts provide more options for the clinical treatment of DN [13].
Rhein, an anthraquinone compound isolated from Chinese herbal
medicine such as Rheum palmatum L, cassia seed, polygonum multiflorum, aloe, and
polygonum cuspidatum mainly by the ultrasonic nebulization extraction [14] and
the microwave-assisted extraction [15]. It has been widely used for the treatment
of various diseases with pharmacological properties including antibacterial,
anti-inflammatory, kidney preserving, and antifibrosis [16]. Studies have found
that rhein is effective in the treatment of experimental DN, and its mode of
action is the comprehensive result of multiple targets and multiple pathways
[17, 18, 19]. Rhein can improve renal lesions and dyslipidemia in db/db DN mice [20].
Clinical studies have shown that the levels of total cholesterol, triglycerides,
and transforming growth factor beta1 (TGF-1) in glomerular mesangial
cells are markedly reduced in DN patients after 4 and 6 months of oral rhein
supplements [21]. In the further study of the mechanism of rhein on DN, it was
found that rhein could inhibit the apoptosis and EMT of renal tubular epithelial
cells and produce the antifibrotic effect, thus preventing the progressive
development of DN [22]. However, the specific mechanism of rhein on DN remains to
be clarified, which is one of the main causes limiting rhein to clinical
application.
Ras-related C3 botulinum toxin substrate 1 (Rac1) is a member of the Rho family
of small G proteins. Rac1 hyperactivation in glomerular podocytes is associated
with the pathogenesis of familial proteinuria nephropathy [23]. Studies have
shown that Rac1 deficiency can ameliorate podocyte injury and urinary albumin
induced by streptozocin (STZ) in DN mice [24]. In addition, the Rac1/PAK1
signaling pathway promoted high glucose-induced EMT of podocytes in
vitro by triggering -catenin transcriptional activity [25]. NADPH
oxidase 1 (NOX1) is a key factor in mediating ferroptosis [26]. In DN studies,
inhibition of the NOX1/ROS/NF-B pathway improved inflammation and
extracellular matrix accumulation [27]. In addition, Rac1 regulated the
production of NOX1-dependent ROS [28]. The wnt/-catenin signaling
pathway participates in rhein-ameliorating renal injury in DN mice [29]. These
pieces of evidence suggest that the Rac1/Nox1/-catenin axis may be
involved in the development of DN. Studies have found that rhein could stably
bind Rac1 by molecular docking [30, 31]. However, it has not been investigated
whether rhein inhibits the ferroptosis and EMT of podocytes to alleviate DN by
regulating the Rac1/Nox1/-catenin axis.
Therefore, this study explored the role and specific mechanism of rhein in the
treatment of DN by constructing the podocyte high glucose (HG) model and mouse DN
model and using rhein for intervention treatment, to provide new insights for the
treatment of DN.
2. Materials and Methods
2.1 Cell Culture
The mouse glomerular podocyte (MPC5) (AW-CNM109, Abiowell, Changsha, China) was
cultured in Dulbecco’s modified Eagle’s medium (DMEM) (D5796,
Sigma, St. Louis, MO, USA) containing 10% fetal bovine serum
(FBS) (10099141, Gibco, Waltham, MA, USA) and 1% Penicillin/Streptomycin
(SV30010, Beyotime, Shanghai, China) and placed in a humidified incubator
containing 5% CO at 37 °C. When the cell confluence reached 70–80%,
trypsin digestion, passage, and transfection experiments were carried out.
2.2 Construction of HG Injury Model and Intervention
In experiment 1, MPC5 was divided into 4 groups: normal glucose (NG), NG+Rhein, HG, and HG+Rhein. MPC5 was cultured in a serum-free medium for 12 h.
MPC5 in the NG group was treated with 5.5 mM glucose and 24.5 mM mannitol for 48
h as control. MPC5 in the HG group was treated with 30 mM HG for 48 h to
construct the HG injury model [32, 33]. MPC5 in the NG+Rhein group was treated
with 5.5 mM glucose, 24.5 mM mannitol, and 25 g/mL rhein (extracted from
the root of Rheum palmatum L of Polygonaceae) (98%, CAS#478-43-3,
R7269, Sigma, USA) for 48 h [29]. MPC5 in the HG+Rhein group
was treated with 30 mM HG and 25 g/mL rhein for 48 h.
In experiment 2, MPC5 was divided into 3 groups: HG, si-NC, and si-Rac1. Before
HG induction, Rac1 silencing (si-Rac1) plasmid and its negative control (si-NC)
were transfected into MPC5 of the si-NC group and si-Rac1 group using
Lipofectamine 2000 (11668019, Invitrogen, Carlsbad, CA, USA).
Then, MPC5 in HG, si-NC, and si-Rac1 groups were treated with 30 mM HG for 48 h,
respectively.
In experiment 3, MPC5 was divided into 4 groups: HG, HG+oe-NC, Rhein+oe-NC,
and Rhein+oe-Rac1. Before HG induction, Rac1 overexpression (oe-Rac1) plasmid
and its negative control (oe-NC) were transfected into MPC5 of the HG+oe-NC
group, Rhein+oe-NC group, and Rhein+oe-Rac1 group using Lipofectamine 2000.
MPC5 in the HG+oe-NC group was treated with 30 mM HG for 48 h. MPC5 in
Rhein+oe-NC and Rhein+oe-Rac1 groups were treated with 30 mM HG and 25
g/mL rhein for 48 h.
2.3 Construction of DN Model and Intervention
C57BL/6J male mice (6–8 weeks, 18–20 g) were bought from Hunan SJA Laboratory
Animal Co., Ltd (Changsha, China). Mice were first accepted one week of adaptive feeding and then
divided into 5 groups: Sham, DN, Rhein, Rhein+LV-NC, and Rhein+LV-Rac1, with
6 mice in each group. Mice in the DN group were intraperitoneally injected with
STZ (50 mg/kg) (AWH0492, Abiowell, Changsha, China) and
deprived of food for 4–6 h every day for 5 consecutive days. On day 3 after
injection, the glucose level of tail blood was detected, and the DN model was
constructed when it was greater than 16.7 mmol/L [34]. Mice in the Sham group
were given the same dose of citrate buffer. Mice in the Rhein, Rhein+LV-NC, and
Rhein+LV-Rac1 groups were gavaged with rhein at a dose of 150 mg/(kgd)
while the other groups were given the same dose of normal saline [20]. Besides, 2
weeks before STZ injection, mice in Rhein+LV-NC and Rhein+LV-Rac1 groups were
injected with Rac1 Lentivirus (LV-Rac1) and its negative control (LV-NC) (titer 2
10 TU/mL) through the tail vein, respectively. The body weight
and blood glucose levels of mice in different groups were measured before STZ
intervention, on days 3, 7, 14, 21, and 28, respectively. Four weeks after the
construction of the DN model, the mice were sacrificed, and their blood, urine,
and kidneys were collected for detection (fasting for 12 hours before sampling).
The kidneys were fixed with 4% paraformaldehyde, embedded in paraffin, and
sectioned for staining experiments. All procedures were approved by the Animal
Experimental Ethical Committee of the First Hospital of Human University of
Chinese Medicine (No. ZYFY20220223).
2.4 Cell Counting Kit-8 (CCK-8) Assay
CCK-8 kit was utilized to detect the viability of MPC5 in different groups. MPC5
in the logarithmic growth phase was digested by trypsin (AWC0232, Abiowell,
Changsha, China) and counted. The cells were seeded in 96-well plates at a
density of 5 10 cells/well, 100 L per well. After cell
adhesion, the cells were treated respectively. Then, 100 L of medium
containing 10% CCK-8 (NU679, DOJINDO, Kyushu, Japan) was added to each well, and
cultured for another 4 h in a humidified incubator containing 5% CO at 37
°C. Optical density (OD) at 450 nm was analyzed with a multifunctional microplate
reader.
2.5 Flow Cytometry
Flow cytometry was applied to detect the levels of lipid ROS in different groups
of MPC5. MPC5 was digested with trypsin to prepare the cell suspension. The cells
were centrifuged at 2000 rpm for 5 min, then the supernatant was discarded
followed by washing with PBS twice. C11-BODIPY (50 M) (MX5211-1MG, MKBIO,
Shanghai, China) was added and incubated at 37 °C for 1 h. The cells
were washed twice with PBS and the supernatant was discarded. The precipitates
were suspended in 300 L PBS and detected by flow cytometry.
2.6 Immunofluorescence (IF) Staining
IF staining was used to detect the expression of GPX4, -smooth muscle
actin (-SMA), and Nephrin in podocytes and Podocin in mouse kidneys.
For renal tissue detection, the slices were first placed in xylene for 20 min for
dewaxing. Dehydration was carried out with gradient ethanol (75–100%), 5 min
for each level. The slices were then immersed in EDTA buffer (pH 9.0) and boiled
for antigen repair. They were placed in sodium borohydride solution for 30 min
and rinsed with tap water for 5 min. The slices were immersed in 75% ethanol
solution for 1 min and then incubated in Sudan black dye solution for 15 min.
After rinsing with tap water for 5 min, the slices were blocked with 5% BSA for
1 h. For podocyte detection, slides of cells were fixed with 4% paraformaldehyde
for 30 min. After transparency with 0.3% triton at 37 °C for 30 min, the slides
of cells were washed with PBS for 3 min. Then, they were blocked with 5% BSA at
37 °C for 1 h and rinsed with PBS for 3 min. The
tissue slices were incubated with the primary antibody of Podocin (20384-1-AP,
PTG, Chicago, IL, USA) overnight at 4 °C. The slides
of cells were incubated with primary antibodies of GPX4 (67763-1-IG, Proteintech,
Chicago, IL, USA), -SMA (55135-1-AP, Proteintech, Chicago, IL, USA),
and Nephrin (Ab216341, Abcam, Cambridge, UK) overnight at 4 °C. Then 100
L CoraLite488-conjugated AffiniPure Goat Anti-Rabbit IgG (SA00013-2,
Proteintech, Chicago, IL, USA) or CoraLite594-conjugated Affinipure Goat
Anti-Mouse IgG (SA00013-4, Proteintech, Chicago, IL, USA) was added separately
and incubated for 1.5 h at 37 °C. The nucleus was stained with DAPI
(AWI0331a, Abiowell, Changsha, China) at 37 °C for 20 min. The tissue
slices and slides of cells were sealed with glycerin and observed under a
fluorescence microscope.
2.7 Hematoxylin-Eosin (HE) Staining
HE staining was used to detect renal morphological lesions in different groups
of mice. First, the slices were dewaxed by placing them in xylene for 20 min.
Then dehydration was carried out with gradient ethanol (75–100%), 5 min for
each level. The slices were stained with hematoxylin (AWI0001a, Abiowell,
Changsha, China) for 1 min, rinsed with distilled water, and then returned to
blue in PBS. Next, the slices were stained with eosin (AWI0029a, Abiowell,
Changsha, China) for 1 min and rinsed with distilled water. Dehydration was
carried out with gradient alcohol (95–100%), 5 min for each level. The slices
were cleared in xylene for 10 min and then sealed with neutral gum (AWI0238a,
Abiowell, Changsha, China) for observation by microscope.
2.8 Immunohistochemistry (IHC) Staining
IHC staining was applied to detect the expression of -SMA and Nephrin
in the kidneys of different groups of mice. First, the slices were dewaxed by
placing them in xylene for 20 min. Then dehydration was carried out with gradient
ethanol (75–100%), 5 min for each level. The slices were immersed in citrate
buffer (0.01 M, pH 6.0) (AWI0206a, Abiowell, Changsha, China), and boiled for
antigen repair. Subsequently, 1% periodic acid was added to inactivate the
endogenous enzyme. The slices were incubated with antibodies of -SMA
(55135-1-AP, Proteintech, Chicago, IL, USA) and Nephrin
(Ab216341, Abcam, Cambridge, UK) overnight at 4 °C separately. Then, the
slices were incubated with 100 L HRP-anti-Rabbit-IgG for 30 min at 37
°C. Next, 100 L DAB was added to slices and incubated for 5 min
for color development. The slices were counterstained with hematoxylin for 5 min,
rinsed with distilled water, and returned to blue in PBS. Dehydration was carried
out with gradient alcohol (60–100%), 5 min for each level. The slices were
cleared in xylene for 10 min and then sealed with neutral gum for observation by
microscope.
2.9 Masson Staining
Renal tubulointerstitial fibrosis in different groups of mice was detected by
Masson staining. First, the renal slices were dewaxed to water. Hematoxylin stain
solution was added to cover the slices and then stained for 1 min. The slices
were washed with tap water and distilled water in turn. Then, the slices were
soaked in PBS (pH 7.2–7.6) or ammonia for 10 min to make the nucleus return
blue. Acid fuchsin stain solution was added and stained for 5 min. After that,
the slices were reacted with a phosphomolybdic acid differentiation solution for
about 30 s. The tissue was covered by a drop of aniline blue counterstain,
stained for 3 min, and rinsed with absolute ethanol. The slices were blow-dried,
cleared in xylene, and then sealed with neutral gum for observation by
microscope.
2.10 Biochemical Detection
According to the instructions of the superoxide dismutase (SOD) assay kit
(A001-3, NJJCBIO, Nanjing, China), malondialdehyde (MDA) assay
kit (A003-1, NJJCBIO, Nanjing, China), reduced glutathione
(GSH) assay kit (A006-2-1, NJJCBIO, Nanjing, China), iron assay kit (ab83366,
Abcam, Cambridge, UK), creatinine assay kit (C011-2-1, NJJCBIO, Nanjing, China),
blood urea nitrogen (BUN) assay kit (C013-2-1, NJJCBIO, Nanjing, China), urinary
albumin quantitative assay kit (C035-2-1, NJJCBIO, Nanjing, China), glucose assay
kit (F006, NJJCBIO, Nanjing, China), respectively, the contents of SOD, MDA, GSH,
Fe, creatinine, BUN, urinary albumin, and blood glucose in experimental
samples were measured.
2.11 Quantitative Real-Time Polymerase Chain Reaction (RT-qPCR)
RT-qPCR was employed to measure the expression of Rac1, NOX1, -catenin,
E-cadherin, Vimentin, Snail, and Desmin in different groups of MPC5. Firstly,
total RNA was extracted by using the Trizol total RNA extraction kit (15596026,
Thermo, Waltham, MA, USA). mRNA was reverse transcribed into
cDNA according to the guidance of the mRNA reverse transcription kit (CW2569,
CWBIO, Beijing, China), followed by RT-qPCR. The primers used were as follows:
-actin: F: ACCCTGAAGTACCCCATCGAG, R: AGCACAGCCTGGATAGCAAC; Rac1: F:
TGACCCTCTTTACCTCGCCCAC, R: AACATCGTCAGCACTAGCACAG; NOX1: F: TAAAGGCTCACAGACCCTGC,
R: GAGCCCTTCTAGGCAACAGG; -catenin: F: ATTCTTGGCTATTACGACAGACT, R:
AGCAGACAGATAGCACCTT; E-cadherin: F: AGCCATTGCCAAGTACATCCTC, R:
CGCCTTCTGCAACGAATCCC; Vimentin: F: GTCCACACGCACCTACAGTCT, R:
AAGTCCACCGAGTCTTGAAGC; Snail: F: TGCTTTTGCTGACCGCTCCAAC, R:
GCACTGGTATCTCTTCACATCCGAGT; Desmin: F: CCATTGCCCTGGGATGAACT, R:
GACTGTCCCCATCCCTACCT. The expression levels of target genes were calculated by
the 2 method with -actin as the internal
reference.
2.12 Western Blotting
Western blotting was utilized to detect the expression of
Recombinant Solute Carrier Family 7, Member 11 (SLC7A11), Transferrin Receptor 1
(TFR1), acyl-CoA synthetase long-chain family member 4 (ACSL4), E-cadherin,
Vimentin, Snail, Desmin, Rac1, NOX1, -catenin, and GPX4. Different
groups of samples were treated with RIPA lysate (AWB0136, Abiowell, Changsha,
China) to extract total proteins. The protein was separated by SDS-PAGE and
transferred to the nitrocellulose (NC) membrane. The NC membrane was blocked with
5% skimmed milk (AWB0004, Abiowell, Changsha, China) for 1.5 h and then
incubated with primary antibodies at 4 °C overnight separately. Primary
antibodies were as followed: SLC7A11 (1:1,000, 26864-1-AP, Proteintech, Chicago,
IL, USA), TFR1 (1:5,000, ab269513, Abcam, Cambridge, UK), ACSL4 (1:10,000,
ab155282, Abcam, Cambridge, UK), E-cadherin (1:5,000, 20874-1-AP, Proteintech,
Chicago, IL, USA), Vimentin (1:5,000, 10366-1-AP, Proteintech, Chicago, IL, USA),
Snail (1:500, 13099-1-AP, Proteintech, Chicago, IL, USA), Desmin (1:10,000,
16520-1-AP, Proteintech, Chicago, IL, USA), Rac1 (1:500, 24072-1-AP, Proteintech,
Chicago, IL, USA), NOX1 (1:1,000, 17772-1-AP, Proteintech, Chicago, IL, USA),
-catenin (1:10,000, 51067-2-AP, Proteintech, Chicago, IL, USA), GPX4
(1:1,000, 67763-1-Ig, Proteintech, Chicago, IL, USA), and -actin
(1:5,000, 66009-1-Ig, Proteintech, Chicago, IL, USA). The NC membrane was
incubated with HRP-goat anti-mouse IgG (1:5,000, SA00001-1, Proteintech, Chicago,
IL, USA) or HRP-goat anti-rabbit IgG (1:6,000, SA00001-2, Proteintech, Chicago,
IL, USA) for 1.5 h. Finally, the NC membrane was incubated with ECL reagent
(AWB0005, Abiowell, Changsha, China) and followed by imaging. The expression
levels of each protein were analyzed by Quantity One 4.6.6
(Bio-Rad Inc., Hercules, CA, USA) with -actin as the reference protein.
2.13 Data Analysis
GraphPad Prism 8.0 (GraphPad Software Inc.,
San Diego, CA, USA) was used for data analysis. Experimental data were expressed
as mean standard deviation (SD). One-way analysis of variance (one-way
ANOVA) and two-way analysis of variance (two-way ANOVA) were used for comparison
between groups, and p 0.05 was considered statistically significant.
3. Results
3.1 Rhein Inhibited HG-Induced Ferroptosis in Podocytes
To screen the suitable concentration of rhein, podocytes were treated with a
range of concentrations of rhein (0, 5, 15, 25, 50, and 100 g/mL) for 24 h
and 48 h. The half maximal inhibitory concentration (IC) of rhein at 24
and 48 h was 115.92 g/mL and 89.14 g/mL, respectively (Fig. 1A).
When the concentration of rhein exceeded 25 g/mL, its inhibitory effect on
podocyte proliferation was very significant, so it was selected to carry out
follow-up experiments. To investigate the effect of rhein on HG-induced
ferroptosis in podocytes, we first constructed the HG injury model and
co-cultured it with rhein for 48 h. CCK-8 detection results in Fig. 1B displayed
that podocytes viability in the HG group was obviously reduced, indicating the HG
injury model was successfully constructed. However, the viability of HG-treated
podocytes was recovered by co-culture with rhein. Moreover, flow cytometry
further revealed that rhein reduced HG-induced lipid ROS accumulation (Fig. 1C).
Next, biochemical detection results displayed that rhein reduced the content of
MDA and Fe in HG-treated podocytes, but promoted the synthesis and
accumulation of SOD and GSH (Fig. 1D). It was found by IF staining that rhein
co-culture reversed the inhibitory effect of HG on GPX4 synthesis in podocytes
(Fig. 1E). The expression of SLC7A11 in HG-treated podocytes was increased after
co-culture with rhein, while TFR1 and ACSL4 were the opposite (Fig. 1F). These
results indicated that rhein could inhibit HG-induced ferroptosis in podocytes.
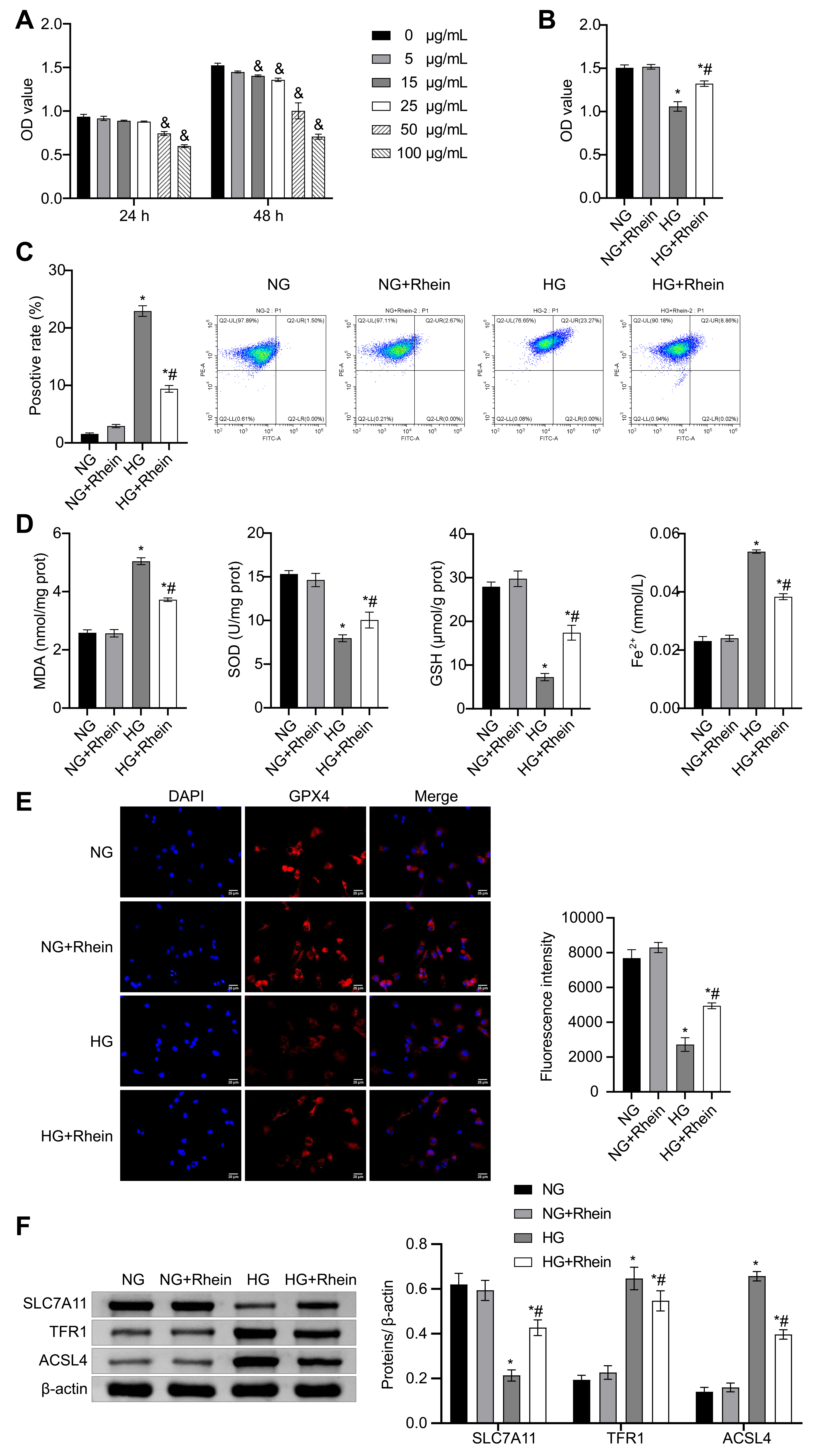 Fig. 1.
Fig. 1.
Rhein inhibited HG-induced ferroptosis in podocytes. (A,B)
CCK-8 assay of podocytes viability. (C) Flow cytometry detection of lipid ROS
levels. (D) Biochemical detection of MDA, SOD, GSH, and Fe contents. (E)
IF staining detection of GPX4 content. (F) Western blotting analysis of SLC7A11,
TFR1, ACSL4 expressions. & p 0.05 vs. 0 g/mL, * p
0.05 vs. NG, # p 0.05 vs. HG, one-way ANOVA, two-way ANOVA.
Notes: IC, half maximal inhibitory concentration; NG, normal glucose; HG,
high glucose; CCK-8, Cell Counting Kit-8; ROS, reactive oxygen species; MDA,
malondialdehyde; SOD, superoxide dismutase; GSH, glutathione; IF,
immunofluorescence; SLC7A11, Recombinant Solute Carrier Family 7, Member 11;
TFR1, Transferrin Receptor 1; ACSL4, acyl-CoA synthetase long-chain family member
4.
3.2 Rhein Inhibited HG-Induced EMT in Podocytes
We then investigated the effect of rhein on HG-induced EMT in podocytes.
Podocytes were subjected to HG and co-cultured with rhein for 48 h, and the
expression of the marker proteins of EMT was analyzed. As shown in Fig. 2A,
-SMA content decreased while Nephrin content increased in HG-treated
podocytes after co-culture with rhein. It was further found that rhein
significantly inhibited the expression of Vimentin, Snail, and Desmin at the mRNA
and protein levels in HG-treated podocytes. Moreover, rhein intervention promoted
the high expression of E-cadherin in HG-treated podocytes (Fig. 2B). These
results indicated that rhein could inhibit HG-induced EMT in podocytes.
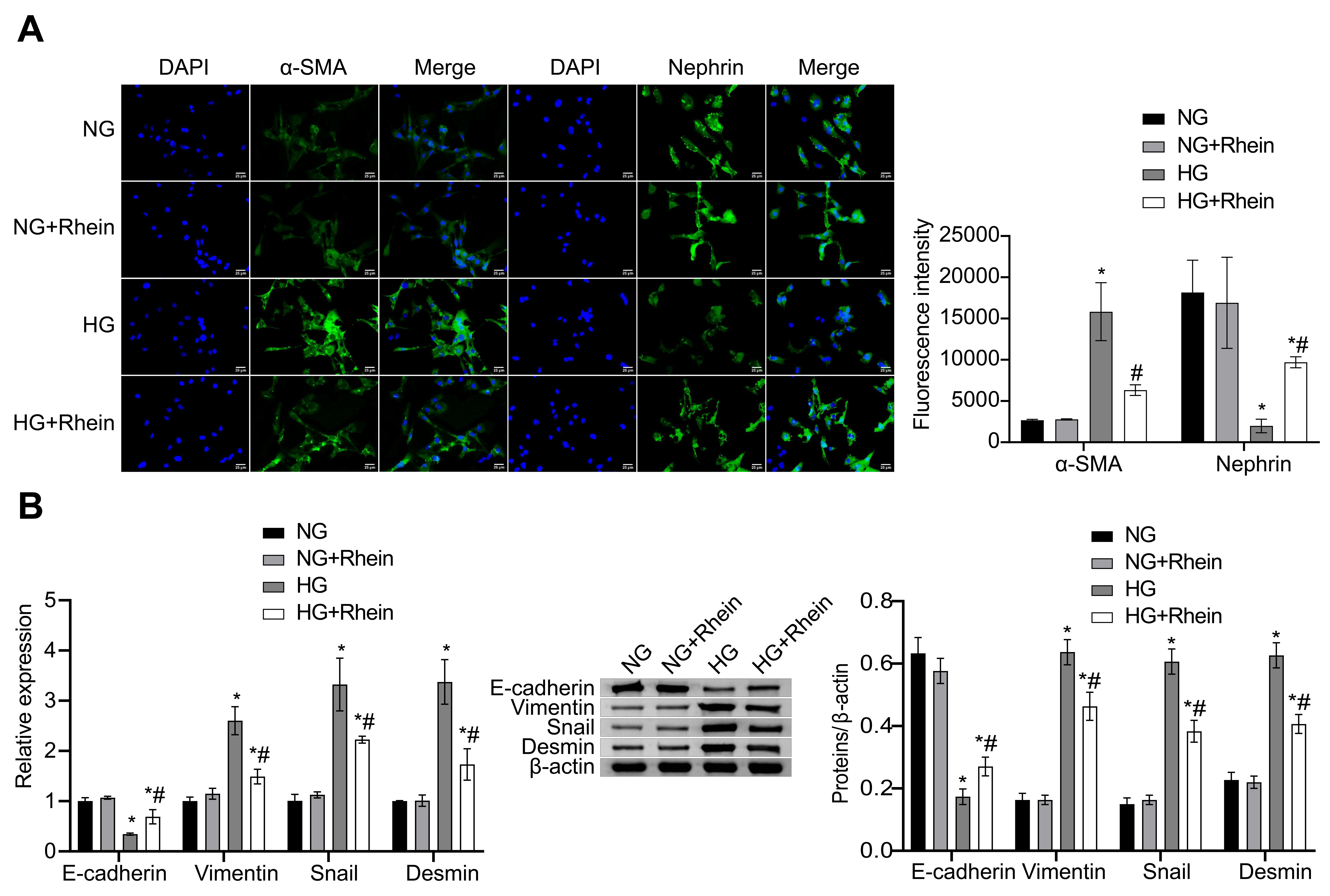 Fig. 2.
Fig. 2.
Rhein inhibited HG-induced EMT in podocytes. (A) IF staining
detection of -SMA and Nephrin contents. (B) RT-qPCR and Western
blotting analysis of E-cadherin, Vimentin, Snail, and Desmin
expressions. * p 0.05 vs. NG, # p 0.05 vs. HG, one-way
ANOVA. Notes: NG, normal glucose; HG, high glucose; EMT, epithelial-mesenchymal
transition; IF, immunofluorescence; -SMA, -smooth muscle
actin; RT-qPCR, quantitative real-time polymerase chain reaction.
3.3 Rhein Inhibited the Rac1 Pathway
To further investigate the specific mechanism of rhein in alleviating HG-induced
podocyte injury, we analyzed the expression of Rac1 and its downstream targets
NOX1 and -catenin respectively. As displayed in Fig. 3A, the mRNA
expression levels of Rac1, NOX1, and -catenin in HG-treated podocytes
were significantly decreased after rhein intervention. Consistently, the protein
expression levels of Rac1, NOX1, and -catenin were also downregulated in
response to rhein (Fig. 3B). Together, these results indicated that rhein could
inhibit the Rac1 pathway.
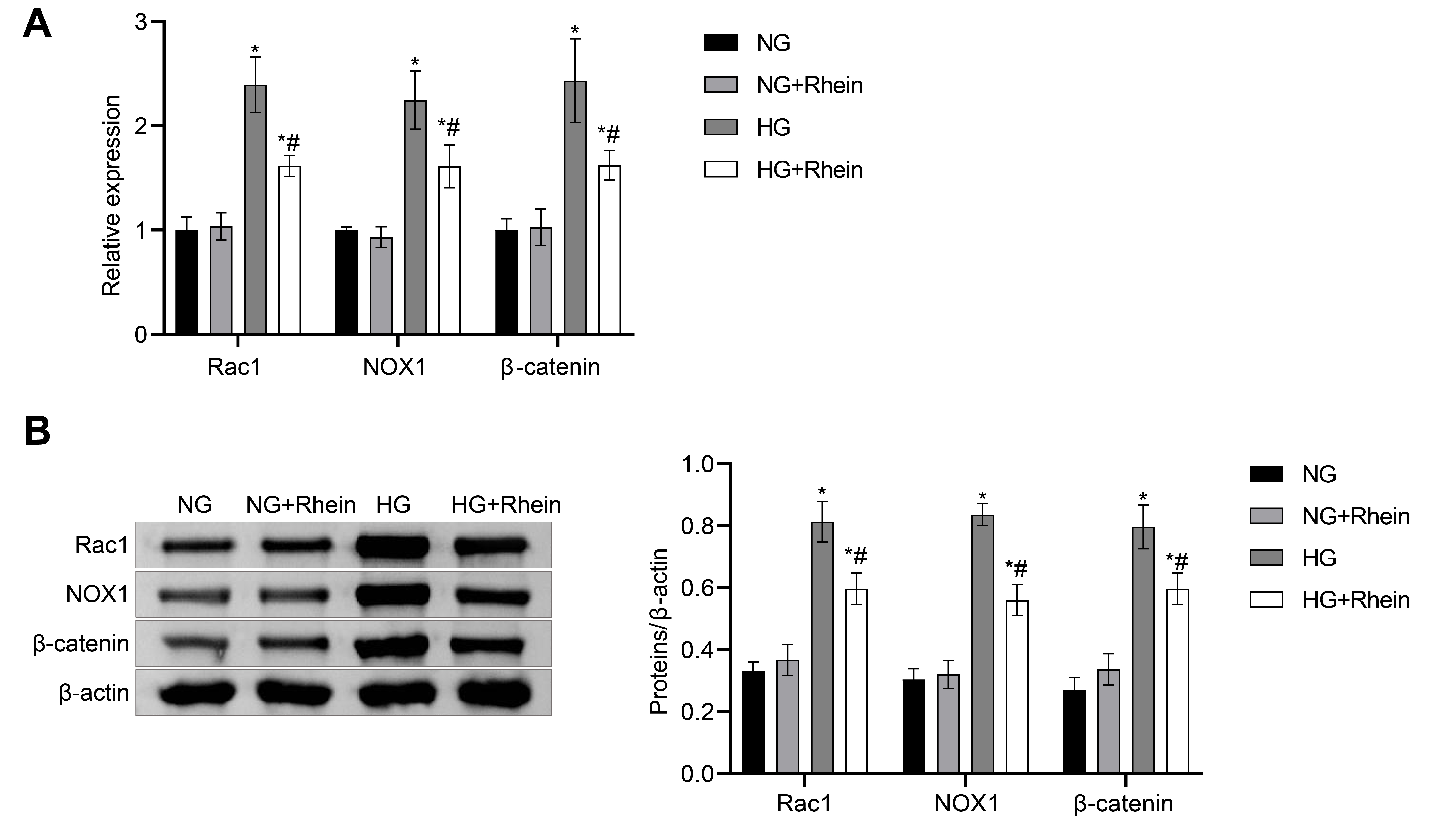 Fig. 3.
Fig. 3.
Rhein inhibited the Rac1 pathway. RT-qPCR (A) and Western
blotting (B) analysis of Rac1, NOX1, and -catenin expressions. *
p 0.05 vs. NG, # p 0.05 vs. HG, one-way ANOVA. Notes: NG,
normal glucose; HG, high glucose; Rac1, Ras-related C3 botulinum toxin substrate
1; NOX1, NADPH Oxidase 1; RT-qPCR, quantitative real-time polymerase chain
reaction.
3.4 Rac1 Pathway Activation Mediated HG-Induced Ferroptosis and EMT
in Podocytes
To explore the role of the Rac1 pathway in HG-induced ferroptosis and EMT,
podocytes were transfected with si-Rac1 first and then treated with HG. Western
blotting results displayed that si-Rac1 significantly decreased the expression of
Rac1 and its downstream targets NOX1 and -catenin (Fig. 4A). In
addition, biochemical detection results displayed that si-Rac1 reduced MDA and
Fe contents in HG-treated podocytes, but significantly promoted the
synthesis and accumulation of SOD and GSH (Fig. 4B). By Western blotting analysis
of ferroptosis-related proteins, we further found that the expression of SLC7A11
and GPX4 in HG-treated podocytes was significantly increased after si-Rac1
transfection, while TFR1 and ACSL4 were opposite (Fig. 4C). In addition, the IF
staining of the marker proteins of EMT revealed that the EMT in HG-treated
podocytes was inhibited under the action of si-Rac1, which was manifested as the
decrease of -SMA and the increase of Nephrin (Fig. 4D). These results
indicated that Rac1 pathway activation could mediate HG-induced ferroptosis and
EMT in podocytes.
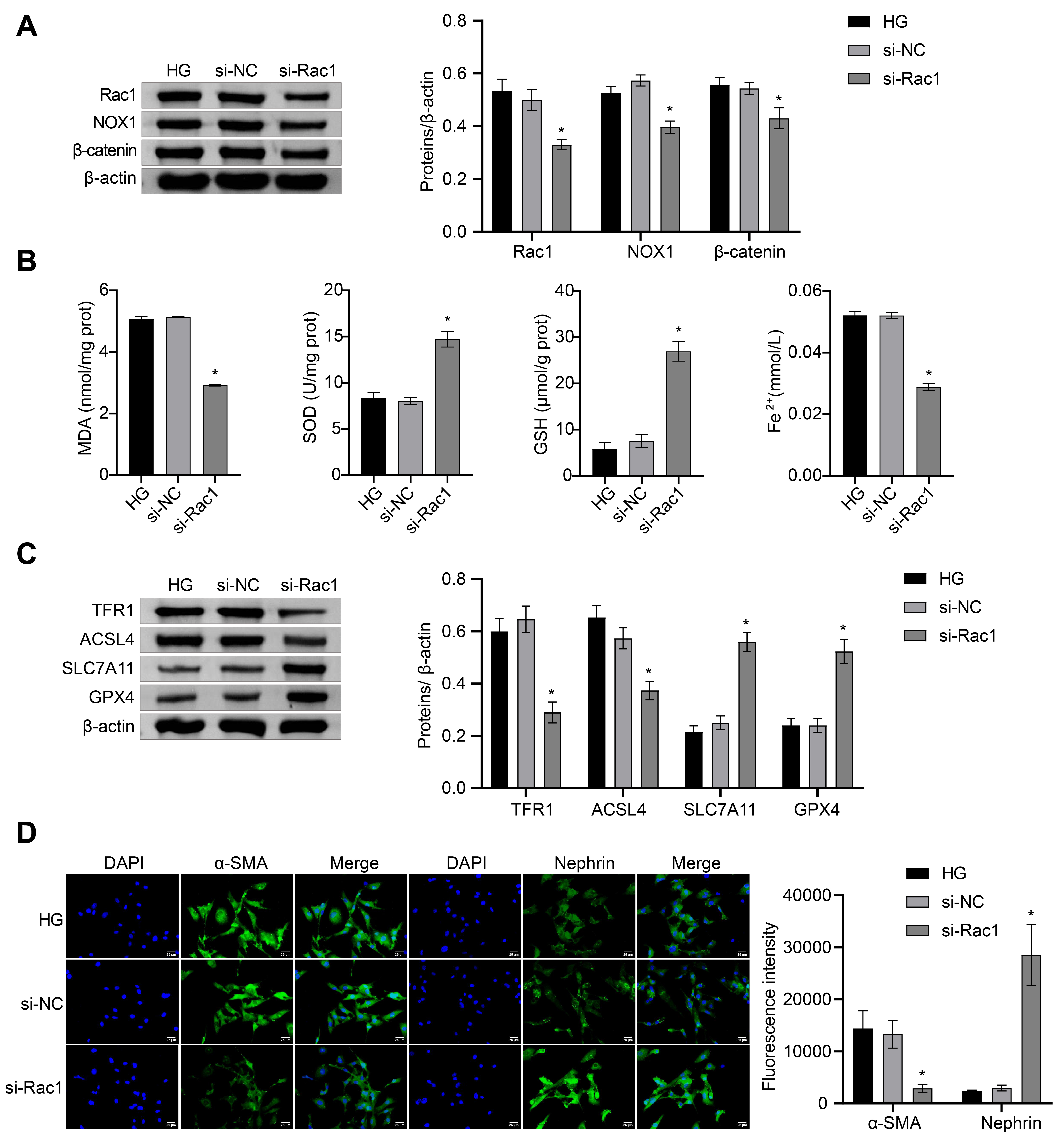 Fig. 4.
Fig. 4.
Rac1 pathway activation mediated HG-induced ferroptosis and EMT
in podocytes. (A) Western blotting analysis of Rac1, NOX1, and -catenin
expressions. (B) Biochemistry detection of MDA, SOD, GSH, and Fe contents.
(C) Western blotting analysis of TFR1, ACSL4, SLC7A11, and GPX4 expressions. (D)
IF staining detection of -SMA and Nephrin contents. * p
0.05 vs. HG, one-way ANOVA. Notes: HG, high glucose; si, silencing; NC, negative
control; Rac1, Ras-related C3 botulinum toxin substrate 1; EMT,
epithelial-mesenchymal transition; NOX1, NADPH Oxidase 1; MDA, malondialdehyde;
SOD, superoxide dismutase; GSH, glutathione; TFR1, Transferrin Receptor 1; ACSL4,
acyl-CoA synthetase long-chain family member 4; SLC7A11, Recombinant Solute
Carrier Family 7, Member 11; GPX4, glutathione peroxidase 4; IF,
immunofluorescence; -SMA, -smooth muscle actin.
3.5 Rhein Inhibited HG-Induced Ferroptosis and EMT in Podocytes by
Regulating the Rac1 Pathway
To further determine the role of rhein in inhibiting HG-induced ferroptosis and
EMT in podocytes by regulating the Rac1 pathway in vitro, podocytes were
transfected with oe-Rac1 before HG induction and rhein intervention. As shown in
the biochemical detection results in Fig. 5A, the contents of MDA and Fein podocytes co-cultured with rhein decreased remarkably while the contents of
SOD and GSH increased. However, oe-Rac1 transfection reversed the inhibitory
effect of rhein on HG-induced ferroptosis. In addition, Western blotting results
displayed that rhein inhibited the HG-induced overexpression of TFR1 and ACSL4.
However, oe-Rac1 transfection displayed the opposite result, with upregulated
expression of TFR1 and ACSL4 and downregulated expression of SLC7A11 and GPX4
(Fig. 5B). IF staining detection further revealed that rhein inhibited HG-induced
EMT in podocytes, but oe-Rac1 transfection reversed this situation, resulting in
increased -SMA content and decreased Nephrin content (Fig. 5C). These
results indicated that rhein could inhibit HG-induced ferroptosis and EMT in
podocytes by regulating the Rac1 pathway.
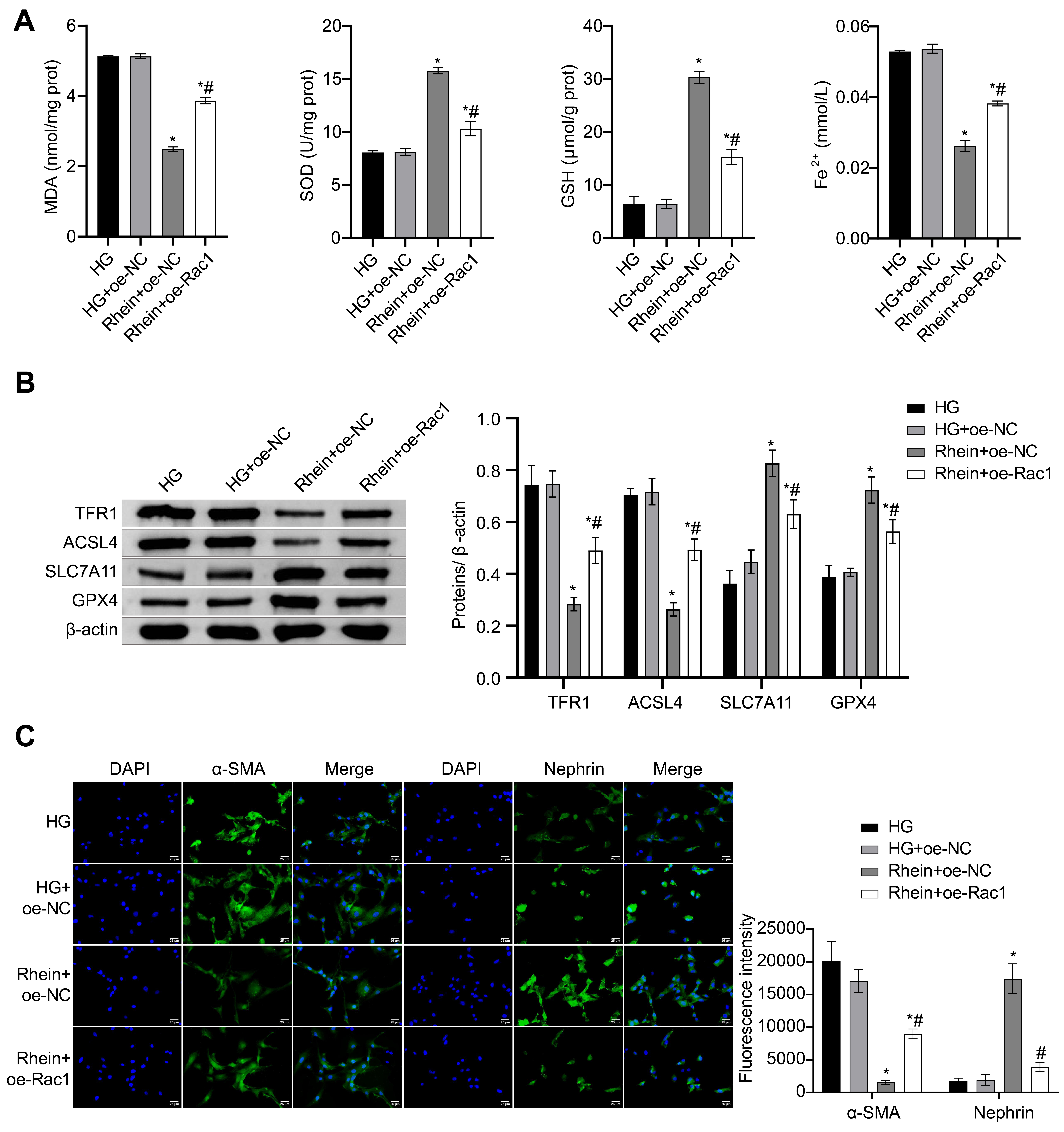 Fig. 5.
Fig. 5.
Rhein inhibited HG-induced ferroptosis and EMT in podocytes by
regulating the Rac1 pathway. (A) Biochemical detection of MDA, SOD, GSH, and
Fe contents. (B) Western blotting analysis of TFR1, ACSL4, SLC7A11, and
GPX4 expressions. (C) IF staining detection of -SMA and Nephrin
contents. * p 0.05 vs. HG, # p 0.05 vs. Rhein+oe-NC,
one-way ANOVA. Notes: HG, high glucose; oe, overexpression; NC, negative control;
EMT, epithelial-mesenchymal transition; Rac1, Ras-related C3 botulinum toxin
substrate 1; MDA, malondialdehyde; SOD, superoxide dismutase; GSH, glutathione;
TFR1, Transferrin Receptor 1; ACSL4, acyl-CoA synthetase long-chain family member
4; SLC7A11, Recombinant Solute Carrier Family 7, Member 11; GPX4, glutathione
peroxidase 4; IF, immunofluorescence; -SMA, -smooth muscle
actin.
3.6 Rhein Inhibited Ferroptosis and EMT in DN Mice
by Regulating the Rac1 Pathway
To further explore the role of rhein in inhibiting DN-induced ferroptosis and
EMT by regulating the Rac1 pathway in vivo, LV-Rac1 was injected through
the tail vein of mice 2 weeks before the construction of the DN model, and then
rhein was administered by gavage. Compared with the Sham group, the body weight
of mice in the DN group was sharply decreased, accompanied by persistent
hyperglycemia, indicating that the DN model was successfully constructed. Mice in
the Rhein group had higher body weight and lower blood glucose than those in the
DN group, but LV-Rac1 injection changed these conditions (Fig. 6A). The renal
index and urinary albumin levels of mice in the DN group were significantly
higher than those in the Sham group, which verified the successful induction of
DN. In addition, LV-Rac1 injection significantly inhibited the relieving effect
of rhein on DN (Fig. 6B). Biochemical detection results displayed that rhein
intervention significantly reduced DN-induced high BUN and serum creatinine,
whereas LV-Rac1 injection presented the opposite result (Fig. 6C). Furthermore,
HE staining displayed thickened GBM and hypertrophic glomeruli in DN mice. Masson
staining displayed marked renal tubulointerstitial fibrosis in DN mice. These
conditions were alleviated by rhein intervention. However, the injection of
LV-Rac1 reversed the therapeutic effect of rhein on DN (Fig. 6D). Using IF
staining, Podocin content was further found to be significantly increased after
rhein intervention but significantly decreased after LV-Rac1 injection (Fig. 6E).
Western blotting results revealed that GPX4 and SLC7A11 were highly expressed and
RAC1, NOX1, -catenin, TFR1, and ACSL4 were lowly expressed in the Rhein
group. However, the expression of these proteins was opposed in the Rhein+LV-Rac1
group (Fig. 6F,H). In addition, Rhein intervention inhibited the accumulation of
MDA and Fe but significantly promoted the synthesis of SOD and GSH.
However, LV-Rac1 injection reversed this trend (Fig. 6G). IHC staining further
found that the content of -SMA was low and the content of Nephrin was
high in the Rhein group, while the situation was opposite in the Rhein+LV-Rac1
group (Fig. 6I). These results indicated that rhein inhibited ferroptosis and EMT
in DN mice by regulating the Rac1 pathway.
 Fig. 6.
Fig. 6.
Rhein inhibited ferroptosis and EMT in DN mice
by regulating the Rac1 pathway. (A) Body weight and blood glucose. (B) Renal
index and urinary albumin level. (C) Biochemical detection of BUN and serum
creatinine contents. (D) HE staining and Masson staining. (E) IF staining
detection of Podocin content. (F) Western blotting analysis of Rac1, NOX1, and
-catenin expressions. (G) Biochemical detection of MDA, SOD, GSH, and
Fe contents. (H) Western blotting analysis of TFR1, ACSL4, SLC7A11, and
GPX4 expressions. (I) IHC staining detection of -SMA and Nephrin
contents. * p 0.05 vs. Sham; # p 0.05 vs. DN,
& p 0.05 vs. Rhein+LV-NC, one-way ANOVA, two-way ANOVA. Notes:
EMT, epithelial-mesenchymal transition; DN, diabetic nephropathy; LV, Lentivirus;
NC, negative control; Rac1, Ras-related C3 botulinum toxin substrate 1; BUN,
blood urea nitrogen; HE, Hematoxylin-eosin; IF, immunofluorescence; NOX1, NADPH
Oxidase 1; MDA, malondialdehyde; SOD, superoxide dismutase; GSH, glutathione;
TFR1, Transferrin Receptor 1; ACSL4, acyl-CoA synthetase long-chain family member
4; SLC7A11, Recombinant Solute Carrier Family 7, Member 11; GPX4, glutathione
peroxidase 4; IHC, immunohistochemistry; -SMA, -smooth muscle
actin.
4. Discussion
In the study of DN, both ferroptosis and EMT have been involved in the
progression of DN. With in-depth exploration, it has been found that rhein, as
the main extract of rhubarb, has a potential
therapeutic effect on DN [35]. However, the specific mechanism of rhein in
improving DN remains to be explored. Here we studied the effect of rhein on DN by
constructing a podocyte HG model and mouse DN model and further analyzed the
specific mechanism of Rac1 and its downstream NOX1/-catenin pathway in
the inhibition of DN development by rhein.
Ferroptosis is involved in the pathological process of serious diseases,
including tumors, cardiovascular and cerebrovascular diseases, neurodegenerative
diseases, and renal diseases [36]. ROS accumulation and iron overload in
ferroptosis are important determinants in DN development [6]. Previous studies
proved that iron accumulation increased DN-induced renal injury by increasing
oxidative stress or decreasing antioxidant capacity [8]. In DN, massive ROS is
produced due to the disorder of glucose metabolism, which induces renal
structural and functional damage. Meanwhile, abnormal lipid metabolism leads to
the accumulation of MDA [5]. Due to the sensitivity of podocytes to ROS,
excessive ROS can cause irreversible changes in podocyte structure and function
[37]. In this study, HG-induced podocytes produced massive ROS, and MDA, and
inhibited the synthesis and accumulation of SOD and GSH. However, rhein
intervention noticeably repaired HG-induced damage in podocytes. In the study of
rhein, it was found that rhein could protect cells from HO-induced
damage, reduce the content of MDA, and increase the activities of SOD and GSH-PX
[38]. Here we found HG-induced ferroptosis in podocytes, as shown by the increase
of Fe, TFR1, and ACSL4 and the decrease of GPX4 and SLC7A11, which were
consistent with previous studies [26]. However, HG-induced podocyte ferroptosis
was inhibited in the presence of rhein. The catalytic activity of GPX4
contributes to reducing the level of lipid peroxidation to inhibit the occurrence
of ferroptosis [39]. SLC7A11 is involved in the exchange of extracellular cystine
and glutamate and plays an inhibitory role in ferroptosis. Related studies have
shown that activation of the SLC7A11/GPX4 axis can inhibit ferroptosis [40]. TFR1
is considered a marker protein for the occurrence of ferroptosis, which can be
prevented by a TFR1 inhibitor or knockdown of TFR1 [41]. ACSL4 is a known
promoter of ferroptosis, and knockdown of it has a protective effect against
ferroptosis-mediated acute renal injury [42].
Studies have shown that EMT is one of the important mechanisms of renal
tubulointerstitial fibrosis in the progression of renal lesions [43, 44].
Podocytes may undergo phenotypic transformation through EMT under certain
injuries. Therefore, the occurrence of EMT in podocytes may be one of the
important mechanisms of podocyte loss in DN [45]. When EMT occurs,
epithelial-like phenotypic markers such as E-cadherin are lost, and mesenchymal
phenotypic markers such as Vimentin and -SMA are expressed [46]. Snail
is an important promoter of the EMT process and can directly inhibit E-cadherin
expression at the transcriptional level [47]. In the present study, the
expressions of -SMA, Vimentin, Snail, and Desmin were up-regulated and
those of E-cadherin and Nephrin were down-regulated in HG-treated podocytes.
Rhein could antagonize EMT and reverse the above trends, which were consistent
with the results of related studies [48]. These results suggest rhein could be
effective in the treatment of DN by inhibiting EMT in podocytes.
Aberrant activation of Rac1 plays an important role in renal diseases [49].
During DN pathogenesis, Rac1 overactivation induces changes in podocyte structure
and function [50]. Here, HG activated the Rac1 pathway in podocytes and promoted
the high expression of downstream NOX1 and -catenin, but it was
inhibited in the presence of rhein intervention. Besides, si-Rac1 transfection
verified that the activation of the Rac1 pathway mediated ferroptosis and EMT in
HG-induced podocytes. In addition, in vitro results displayed that DN
mice had decreased body weight, increased blood glucose, and high renal index.
However, the symptoms were relieved under rhein gavage treatment. A high glucose
environment is an important cause of diabetic complications. Therefore,
controlling blood glucose can reduce the incidence of DN [51]. Moreover, the
biochemical detection results displayed that the levels of urinary albumin, BUN,
and serum creatinine in DN mice were high, which was consistent with the
performance of DN in the literature [52, 53]. However, these symptoms were
relieved by rhein treatment. In related studies, it was found that rhein could
inhibit the wnt/-catenin signaling pathway and increase Nephrin
expression in podocytes of db/db mice, to reduce the excretion of urinary albumin
and repair HG-induced podocyte injury [29]. HE staining and Masson staining
further revealed that GBM thickening, glomerular hypertrophy, and renal
tubulointerstitial fibrosis were observed in DN mice, which were relieved after
rhein intervention. In DN, GBM thickening is one of the main features [54]. The
appearance of renal tubulointerstitial fibrosis results in renal dysfunction
[12]. In previous studies, rhein improved the autophagy of mouse renal tubular
cells and delayed the progression of renal tubulointerstitial fibrosis by
regulating the AMPK/mTOR signaling pathway [55]. Podocin is an important protein
in the hiatal membrane of podocytes, which can maintain the structure and
function of podocytes. Here, Podocin expression was reduced in kidney tissue of
DN mice but was rescued by rhein intervention. In addition, overexpression of
Rac1 reversed the protective effect of rhein against renal injury. These results
suggested that rhein inhibited ferroptosis and EMT in DN mice through the Rac1
pathway.
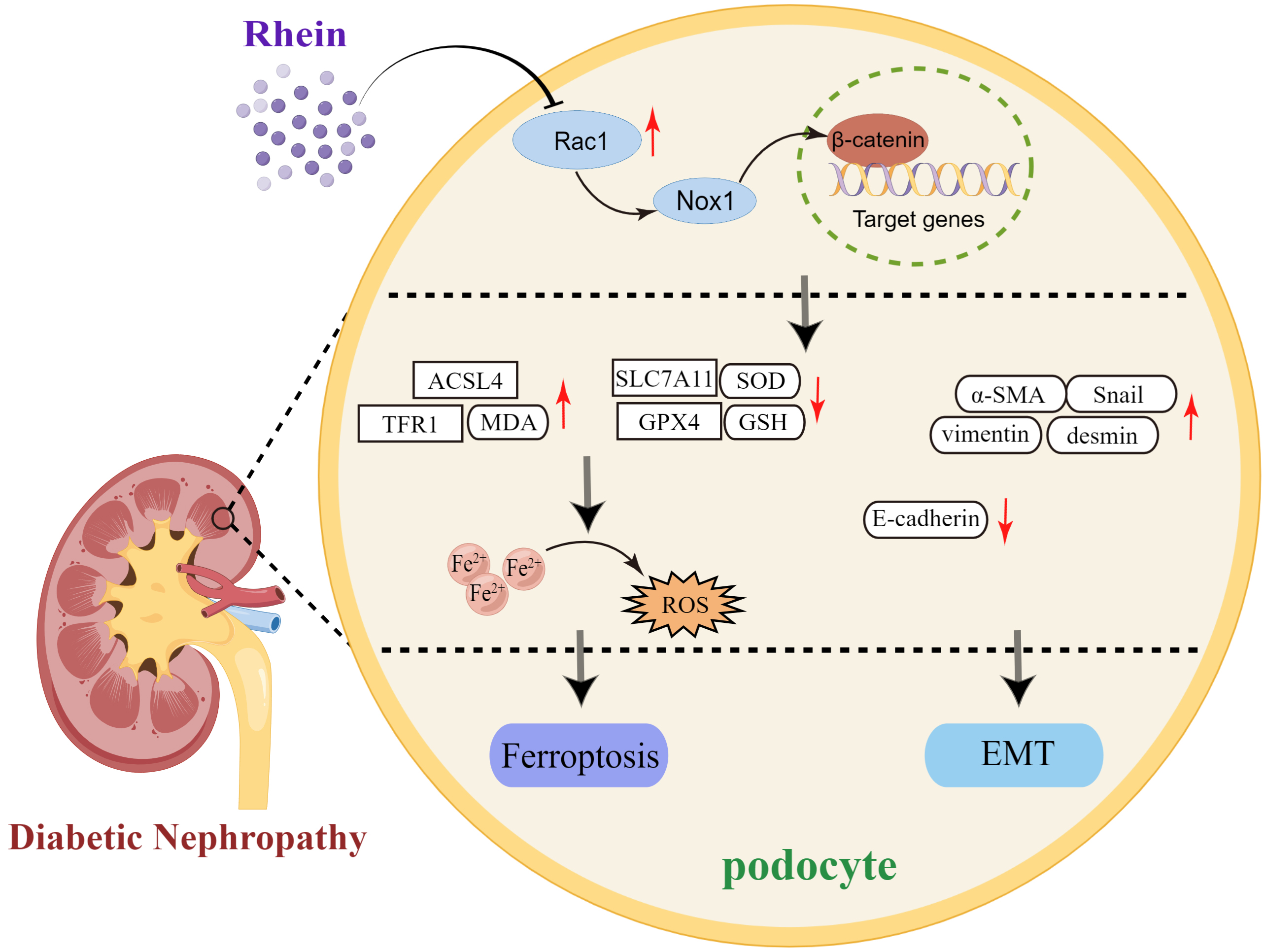
 Fig. 1.
Fig. 1. Fig. 2.
Fig. 2. Fig. 3.
Fig. 3. Fig. 4.
Fig. 4. Fig. 5.
Fig. 5. Fig. 6.
Fig. 6.





