1 Department of Neurosurgery, General Hospital of Northern Theater Command, 110016 Shenyang, Liaoning, China
2 Department of Reproductive Endocrinology, Xi'an International Medical Center Hospital, Northwest University, 710127 Xi’an, Shaanxi, China
3 Department of Pathology, General Hospital of Northern Theater Command, 110016 Shenyang, Liaoning, China
4 State Key Laboratory of Cancer Biology, Department of Pathology, Xijing Hospital and School of Basic Medicine, Fourth Military Medical University, 710068 Xi’an, Shaanxi, China
†These authors contributed equally.
Abstract
Background: Glioma has a high incidence in young and middle-aged adults
and a poor prognosis. Because of late diagnosis and uncontrollable recurrence of
the primary tumor after failure of existing treatments, glioma patients tend to
have a poor prognosis. Recent advances in research have revealed that gliomas
exhibit unique genetic features. Mitogen-activated protein kinase 9 (MAPK9) is
significantly upregulated in mesenchymal glioma spheres and may be a new target
for glioma diagnosis. This study aimed to investigate the potential diagnostic
significance and predictive value of MAPK9 in glioma. Methods:
Paraffin-embedded tumor tissues and paracancerous tissues were collected from 150
glioma patients seen at the General Hospital of Northern Theater Command.
Immunohistochemistry and western blot assays were used to detect the expression
levels of MAPK9. Prognosis and survival analyses were performed using SPSS 26
software for univariate/multivariate analysis and
log-rank analysis. Cellular models were used to assess the effect of MAPK9
overexpression and knockdown in vitro. Results: MAPK9
expression was higher in glioma tissues than in paraneoplastic tissues.
Prognostic and survival analyses revealed that the MAPK9 expression level is an
independent prognostic factor in glioma patients. In addition, overexpression of
MAPK9 significantly promoted the proliferation and migration of primary glioma
cells, possibly via the Wnt/
Glioma has been studied extensively in recent years. However, despite many significant research advances [1, 2], insufficient progress has been made in addressing the disease, and patient outcomes remain poor. The global prevalence of glioma is approximately 6 in 100,000. The number of glioma cases among men is 1.6 times that among women [3]. In accordance with the most recent guidelines in the 2021 WHO Classification of Tumors of the Central Nervous System, the glioma classification system has been significantly updated. Common subtypes include astrocytoma withthe isocitrate dehydro-genase (IDH) mutation (which is usually WHO grade 2, 3, or 4), oligodendroglioma with IDH mutation and 1p/19q codeletion (usually WHO grade 2 or 3), and glioblastoma with wild-type IDH (typically in WHO grade 4) [4, 5]. Molecular information on glioma, such as IDH mutation status and 1p/19q codeletion status, has become increasingly important in diagnosis and treatment, as these features provide information on the molecular mechanism of glioma development [6, 7]. In histological classification, gliomas are commonly classified into high-grade gliomas (HGGs), whose most common type is glioblastoma, and low-grade gliomas (LGGs). Although studies in recent years have improved the treatment of glioma, the overall survival rate of glioma remains low. Glioblastoma (GBM) has the lowest overall survival among brain tumors, with a 5-year survival rate of only 5.8% after diagnosis [8]. Gliomas with an oligodendroglial component have a higher survival rate than gliomas with an astrocytic component. LGG has 5-year survival rates ranging from 55% to 86%, and the median survival time of LGG patients ranges from 7.5 to 10 years. The prognosis of gliomas commonly depends on multiple factors, such as cytological type, central nervous system (CNS) WHO grade, age, and Karnofsky performance score [9, 10, 11]. Current clinical diagnostic methods include imaging technology, intraoperative and postoperative histopathology, genetic testing [12], and molecular testing [13]. The current gold standard for diagnosing brain tumors remains brain MRI, including T2-weighted sequences, T2-weighted fluid-attenuated inversion recovery sequences, and 3D T1-weighted sequences before and after gadolinium contrast [3, 14]. However, imaging modalities for glioma classification, prognosis and judgment do not currently meet all needs, so more sensitive molecular-assisted diagnostic methods are still needed.
Mitogen-activated protein kinase 9 (MAPK9) belongs to the MAPK family and is also known as c-Jun N-terminal kinase (JNK2); the JNK signaling pathway at the core related pathway and can be activated in a variety of ways, such as by growth factors and multiple stressors [15]. MAPK9 participates in a variety of cellular regulatory mechanisms, such as stress responses, apoptosis, proliferation and differentiation, and has been found to promote tumor formation in mammalian studies [15]. Using tumor specimens from 18 proneural and 12 mesenchymal high-grade glioma patients, Kim et al. [16] compared the expression of 349 kinase-encoding genes in primary cultures of glioma stem cells in a genome-wide expression study and found that the MAPK9 gene was significantly upregulated in mesenchymal tumor specimens. Recent studies have found that the MAPK9 inhibitor RGB-286638 causes loss of activity in a variety of basement membrane cell culture models and that RGB-286638 may retard tumor growth [17].
However, there is still a lack of more in-depth studies on the relationship between MAPK9 expression and the clinical features and prognosis of glioma and the possible mechanisms involved. Therefore, in our study, we examined glioma and paraneoplastic specimens from 150 clinical patients. Immunohistochemical staining revealed that MAPK9 protein expression was significantly enhanced in glioma tissue, could be used for diagnosis, and was closely related to a poor prognosis. We also established a primary glioma cell model of MAPK9 overexpression or knockdown and investigated the role of MAPK9 in glioma cell proliferation and migration as well as its possible mechanisms. The results of clinical analysis combined with cell studies suggest that MAPK9 may be a good prognostic predictor for glioma and is involved in tumor progression.
We collected paraffin-embedded tumor tissues from 150 glioma cases originally diagnosed and treated in the General Hospital of Northern Theater Command. Among them, 99 cases were LGG (WHO Grade 2–3), and 51 cases were GBM (WHO Grade 4). Tumors and paired paracancerous tissues from six glioma patients (4 LGG cases, 2 GBM cases) who underwent surgical treatment at the General Hospital of Northern Theater Command from March to June 2022 were also collected. These tissues were frozen in liquid nitrogen and analyzed by western blotting. We reviewed medical records to collect case information and to define overall survival from the date of diagnosis to the event endpoint or study completion date. In these cases, we used immunohistochemistry to detect brain tumor molecular expression and WHO grade, next-generation sequencing to test IDH mutation status, fluorescence in situ hybridization to determine 1p/19q codeletion status, and methylation-specific PCR to detect MGMT methylation status. Ethical approval was obtained from the Ethics Committee of the General Hospital of Northern Theater Command, according to the principles of the Declaration of Helsinki. In addition, we included 147 cases from the CGGA database (http://www.cgga.org.cn/index.jsp) in the validation set.
Six pairs of glioma tissues frozen in liquid nitrogen were first removed with paraneoplastic tissues, and MAPK9 expression was detected. Brain tissues were homogenized in RIPA lysis buffer (Cat. No.: P0013B, Beyotime Biotechnology, Shanghai, China) to lyse for 30 minutes. Next, the samples were centrifuged at 12,000 rpm for 10 minutes at 4 °C, and the supernatant was collected for measurement. The protein samples were initially separated using electrophoresis after using a BCA kit ( Cat. No.: P0012, Beyotime Biotechnology) and transferred to Polyvinylidene Fluoride (PVDF) membranes. After Bovine Serum Albumin(BSA) blocking, the PVDF membranes (Cat. No.: ISE00010, Merck Millipore Ltd., Darmstadt, Germany) were incubated with MAPK9 antibody (Cat. No.: sc-7345, 1:200, Santa Cruz Biotechnology, Dallas, TX, USA) and secondary antibody of the corresponding species (Cat. No.: A0216, 1:1000, Beyotime Biotechnology) and exposed using BeyoECL luminescent buffer. Grayscale values were analyzed using ImageJ V1.8.0 software (National Institutes of Health, Bethesda, MD, USA).
Immunohistochemistry was performed to assess MAPK9 expression in surgically
resected specimens and normal brain tissues from 150 glioma patients. After the
specimens were isolated, they were fixed in 10% formalin for 48 h. Then, we
embedded the samples using paraffin and sectioned them at 4
The expression score for MAPK9 was obtained by multiplying the percentage of
tumor-positive cells by the staining intensity ranging from 0 to 12. We scored
the samples as 0 when there was no staining under the microscope, as 1 when there
was yellowish staining, as 2 when there was yellowish-brown staining, and as 3
when there was brown staining. The percentage of positive staining was scored as
1 for
We extracted total RNA with Beyozol reagent (Beyotime Biotechnology, Shanghai, China) and detected reverse transcription polymerase chain reaction (RT–PCR) products using SYBR Green fluorescent probes. Finally, the expression level of the GAPDH gene was used as the standard to compare the expression level of MAPK9 mRNA.
Recently we have isolated and reported two primary cell lines from GBM patients
named G62 and G91 [18]. We used Dulbecco’s Modified Eagle Medium (DMEM) plus 10% fetal bovine serum (FBS) to culture G62 and G91 cells
routinely at 37 °C and 5% CO
At 37 °C and 5% CO
Stably transfected glioma cells were inoculated at 1
We used SPSS 26 software (SPSS, Inc., Chicago, IL, USA) and GraphPad Prism 9 software (GraphPad Software, Inc.,
La Jolla, CA, USA) for statistical analysis. The relationship between MAPK9
expression and clinicopathological variables was assessed by the
We divided 150 cases into two groups (high MAPK9 expression and low MAPK9
expression). Table 1 shows the statistical analysis and summary of the
correlation between MAPK9 expression and glioma clinicopathological parameters.
Statistical analysis showed that there were significant associations between the
expression of MAPK9 and WHO grade (p
| Clinicopathologic variables | No. of case | MAPK9 expression | p-value | ||
|---|---|---|---|---|---|
| Low | High | ||||
| Age | 0.055 | ||||
| 129 | 89 | 40 | |||
| 21 | 10 | 11 | |||
| Sex | 0.625 | ||||
| Male | 105 | 68 | 37 | ||
| Female | 45 | 31 | 14 | ||
| WHO grades | |||||
| LGG | 96 | 88 | 8 | ||
| GBM | 54 | 11 | 43 | ||
| Site | 0.343 | ||||
| Parietal and occipial lobe | 27 | 13 | 14 | ||
| Frontal and temporal lobe | 96 | 59 | 27 | ||
| Ventricle | 12 | 7 | 5 | ||
| Cerebellar lobe | 10 | 6 | 4 | ||
| Basis cranii | 5 | 4 | 1 | ||
| IDH mutation status | |||||
| Mutant | 103 | 93 | 10 | ||
| Wildtype | 47 | 6 | 41 | ||
| IDH mutation status and 1p/19q codeletion status | |||||
| IDH-wildtype | 47 | 6 | 41 | ||
| IDH-mutant and noncodel | 9 | 4 | 5 | ||
| IDH-mutant and codel | 94 | 89 | 5 | ||
| 1p/19q codeletion status | |||||
| Codel | 94 | 89 | 5 | ||
| Non-codel | 56 | 10 | 46 | ||
| MGMT methylation status | |||||
| Methylated | 93 | 71 | 22 | ||
| Un-methylated | 57 | 28 | 29 | ||
| 3-year survival rate | |||||
| 31 | 1 | 30 | |||
| 119 | 98 | 21 | |||
| 5-year survival rate | |||||
| 43 | 4 | 39 | |||
| 107 | 95 | 12 | |||
To verify whether MAPK9 expression was increased in glioma tissues, we used
western blotting to detect MAPK9 expression in tumors and paired paraneoplastic
tissues of six glioma patients. As shown in Fig. 1A,B, MAPK9 protein expression
was significantly higher in glioma tissues than in paraneoplastic tissues. We
further verified the results of MAPK9 in different grades of gliomas in 150
samples using immunohistochemical methods and revealed that MAPK9 expression was
significantly higher in glioma tissues than in paraneoplastic tissues, and MAPK9
protein expression in GBM tumor tissue was significantly higher than that in LGG
(Fig. 1C). Immunohistochemical evaluation more directly demonstrated the
expression of MAPK9 in different grades of gliomas in 150 samples (Fig. 1D). In
addition, the PCR results showed that the mRNA expression of MAPK9 in the IDH
wild-type group was higher than that in the IDH mutant group (Fig. 1E, p
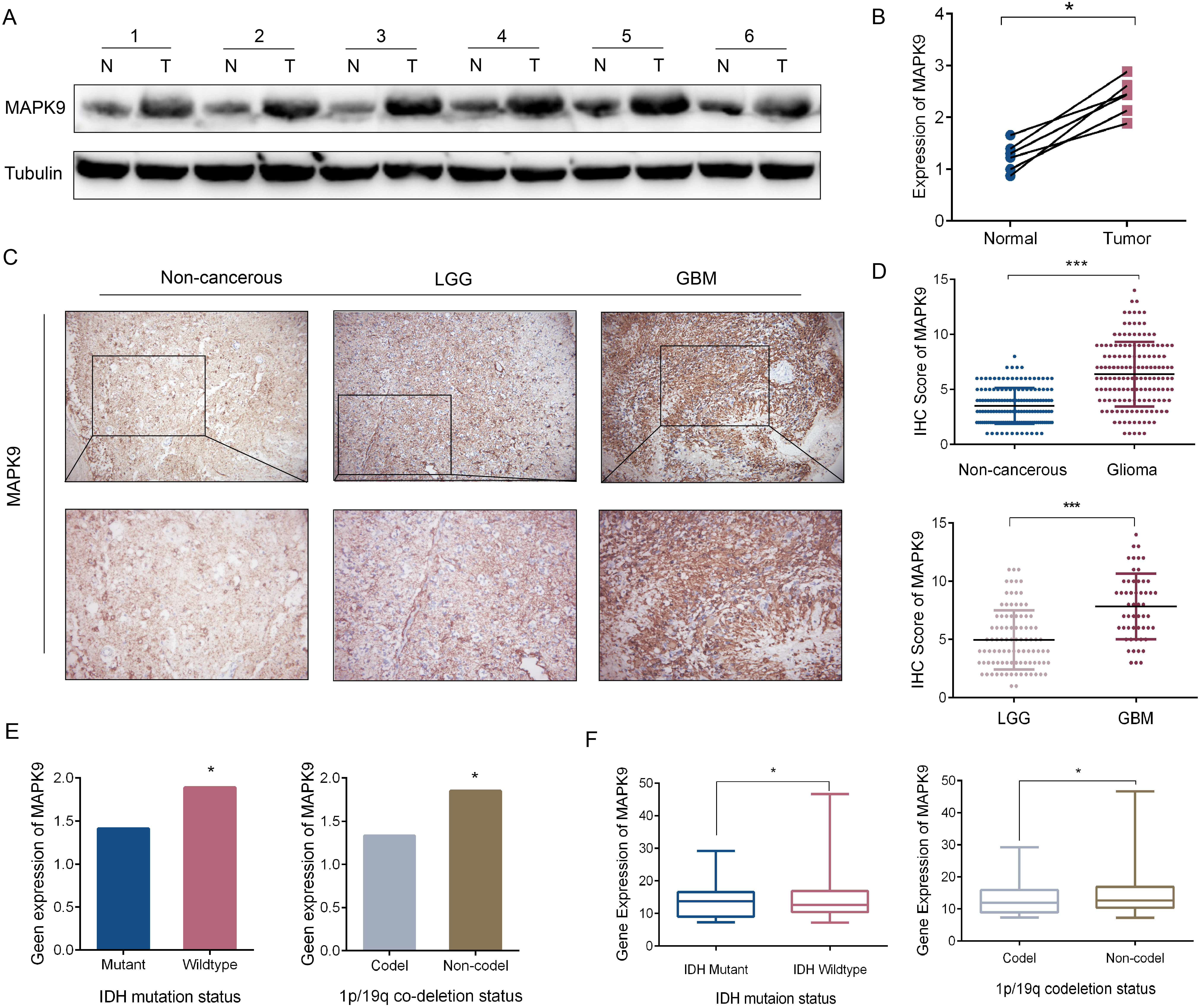 Fig. 1.
Fig. 1.Expression of MAPK9 in different grades of glioma. (A) Western
blotting images of MAPK9 expression in 6 glioma cases and paired paracancerous
tissues. (B) MAPK9 expression in glioma and paired paracancerous tissues,
*p
To study the effect of MAPK9 on the prognosis of glioma patients, we used Kaplan‒Meier analysis to analyze the association between MAPK9 expression level and patient survival. The results suggested that the patients with high MAPK9 expression had a worse prognosis than the patients with low MAPK9 expression (Fig. 2A). In addition, MAPK9 remained a meaningful prognostic predictor in LGG and GBM patients (Fig. 2B,C). MAPK9 also predicted a worse prognosis in the IDH-wildtype and IDH-mutant/codel groups (Fig. 2D,F). Although not statistically significant, high MAPK9 expression showed a trend toward worse survival in IDH-mutant/non-codel group because of the small sample size (n = 9) (Fig. 2E).
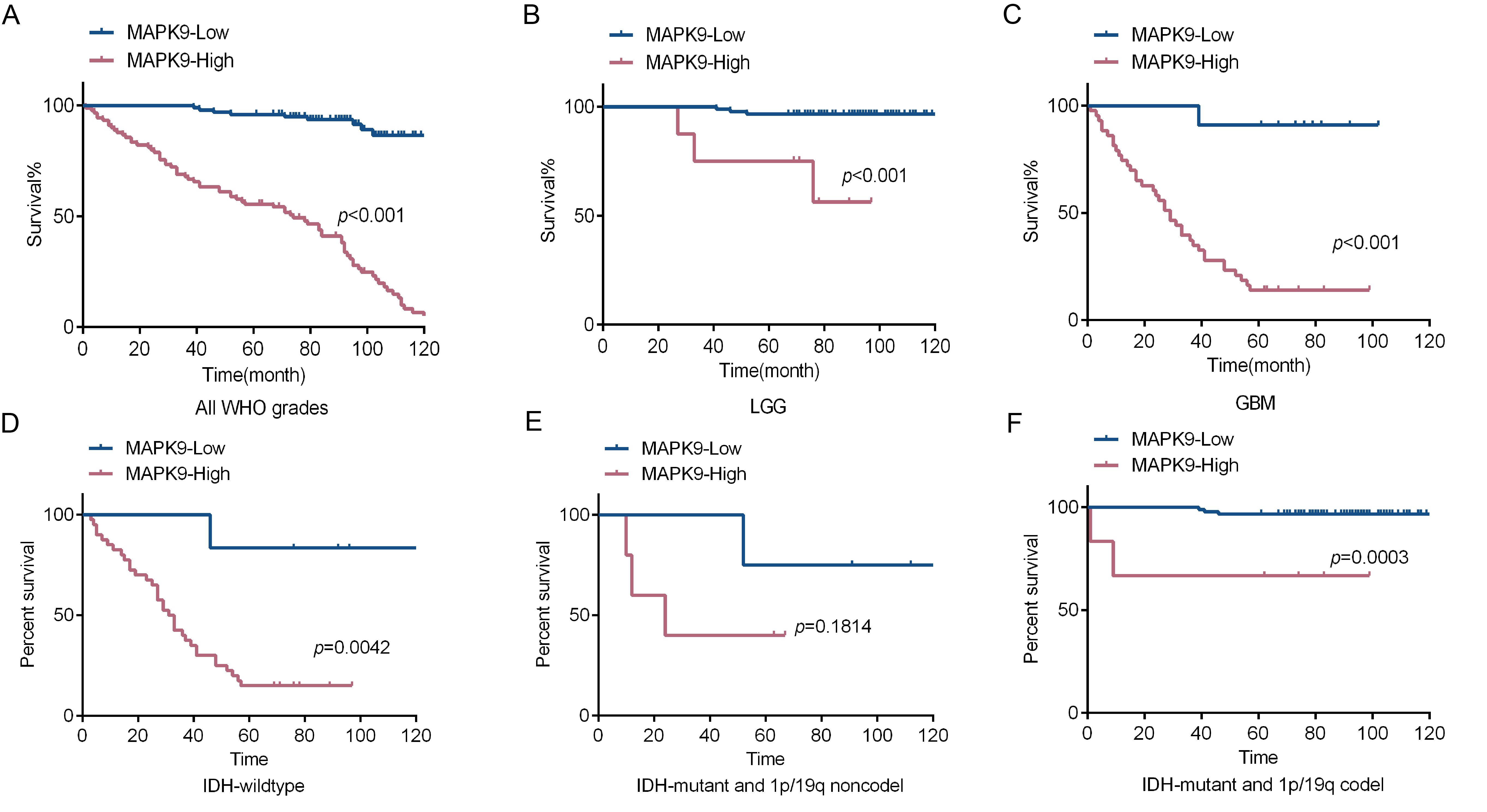 Fig. 2.
Fig. 2.The expression of MAPK9 affects the prognosis of glioma patients. (A) Kaplan‒Meier assay to assess MAPK9 expression in 150 glioma specimens. (B) Kaplan‒Meier assay to detect MAPK9 expression in 96 LGG specimens. (C) Kaplan‒Meier assay to detect MAPK9 expression in 54 GBM specimens. (D) Kaplan‒Meier assay to assess MAPK9 expression in 47 IDH-wildtype specimens. (E) Kaplan‒Meier assay to detect MAPK9 expression in 9 IDH-mutant/non-codel specimens. (F) Kaplan‒Meier assay to detect MAPK9 expression in 94 IDH-mutant/codel specimens.
To explore the possible mechanism of MAPK9 in glioma progression, we used lentivirus to knock down and overexpress MAPK9 in primary glioma cell lines G62 and G91. First, the effectiveness of cell transfection was verified by western blotting. Compared with that in the control group, the expression level of MAPK9 in the shMAPK9 group was obviously decreased (Fig. 3A). Compared to those in control cells, MAPK9 expression levels were significantly increased in the MAPK9 overexpressing cells (Fig. 3A). We then used the CCK-8 assay to assess the proliferative capacity of glioma cells. The results suggested that the cell growth of the shMAPK9 group was significantly lower than that of the control group, while that of the MAPK9 overexpressing group was higher than that of the control group (Fig. 3B). In addition, the migration ability of glioma cells was assessed using a transwell assay. As shown in Fig. 3C, MAPK9 knockdown significantly reduced migration compared with that in the control group, and MAPK9 overexpression increased glioma cell migration. These results suggest that MAPK9 may be a potential promoter of glioma cell progression.
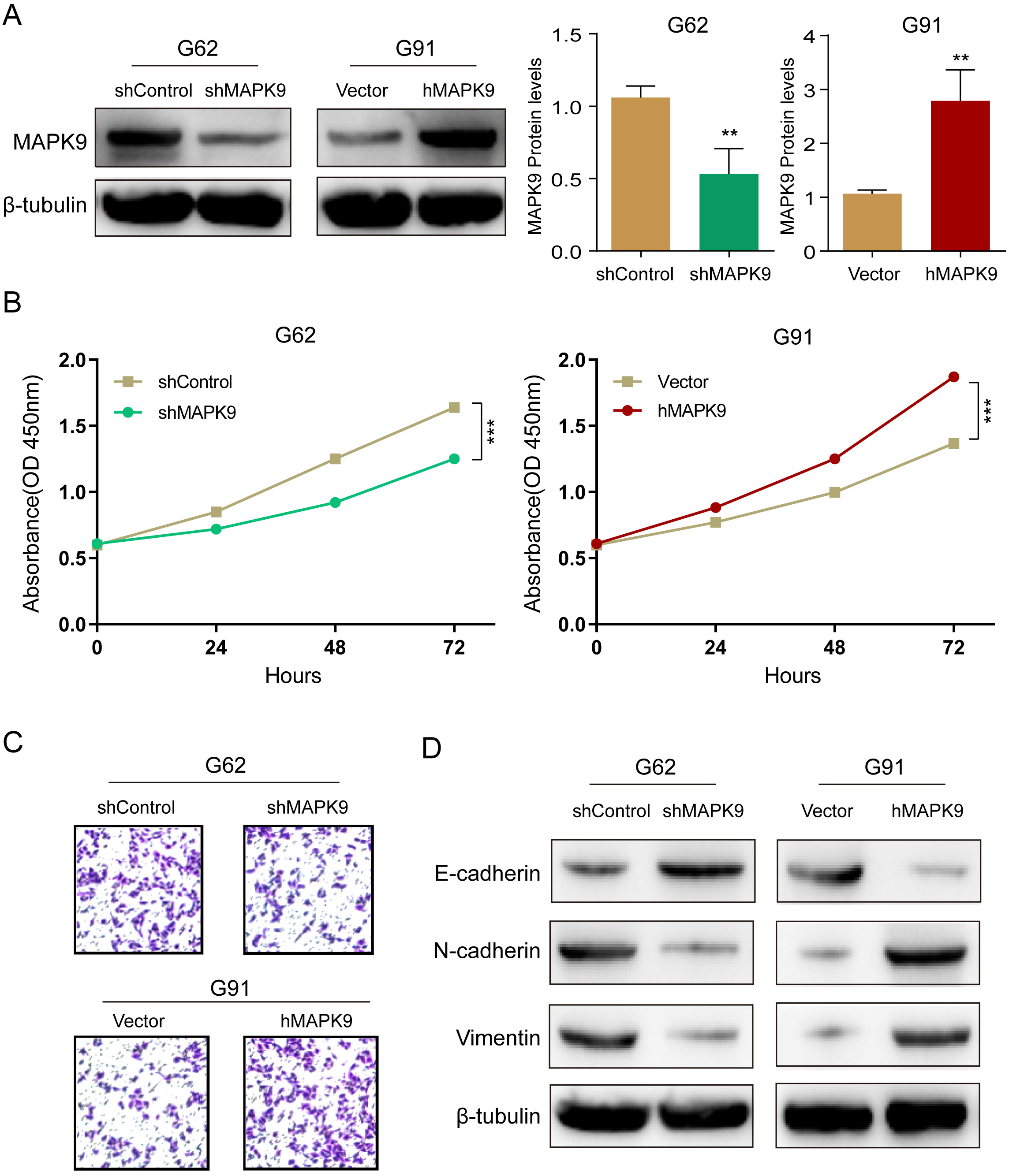 Fig. 3.
Fig. 3.Overexpression of MAPK9 increases glioma cell
proliferation and migration. (A) Western blot validation of MAPK9
knockdown and overexpression in G62 and G91 cell lines, **p
Furthermore, Western blot assays demonstrated that G62 cells with MAPK9 knockdown exhibited decreased N-cadherin and vimentin expression and increased E-cadherin expression, and MAPK9 overexpressing G91 cells exhibited the opposite trends (Fig. 3D). In general, these findings suggest that MAPK9 promotes glioma cell migration by altering EMT-related protein expression.
To further investigate the mechanism of the cancer-promoting effect of MAPK9 on
glioma cells, we assessed
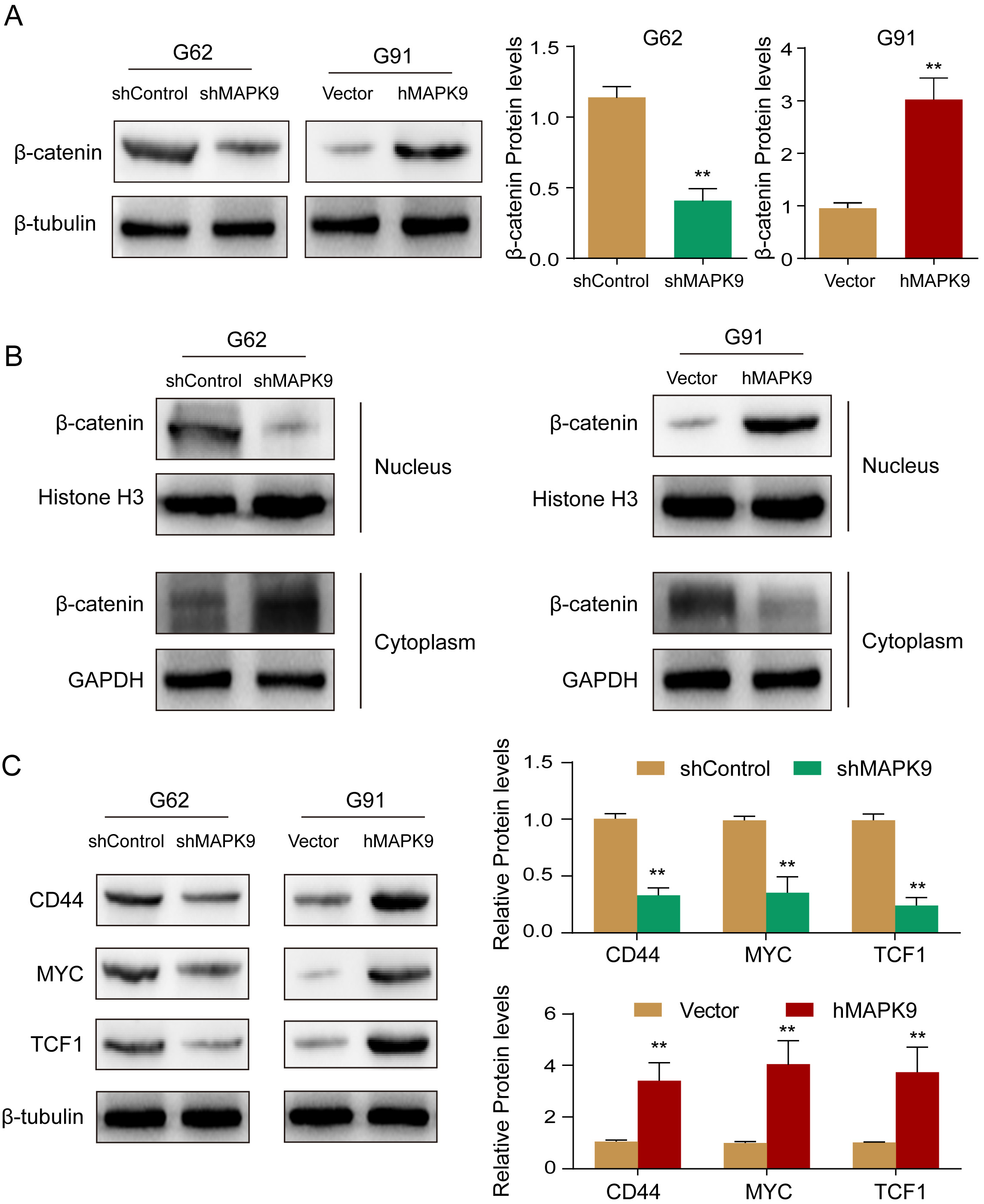 Fig. 4.
Fig. 4.MAPK9 increases glioma cell progression via
There was a significant decrease in the expression of the
Our study revealed that (1) the expression of MAPK9 is high in glioma tissues and low in paracancerous tissues; (2) MAPK9 overexpression is closely associated with WHO grade, IDH mutation and 1p/19q codel status, 3-year survival rate, and 5-year survival rate in glioma, providing new insights into the prediction of glioma prognosis; and (3) MAPK9 overexpression increases the cell viability as well as the migration and invasion ability of glioma cells. Brain MRI, including T2-weighted sequences, T2-weighted FLAIR sequences, and 3D T1-weighted sequences, is the gold standard for diagnosing brain tumors. Electroencephalography can help monitor tumor-related epilepsy and determine the cause of altered consciousness [3]. Genetic testing and molecular testing are helpful for the diagnosis and prognosis evaluation of glioma and, to a certain extent, can determine the follow-up treatment. However, genetic testing and molecular testing are not as popular as imaging diagnosis. Most importantly, the number of targets that can be used for clinical testing is too small, and they are not sensitive enough. Therefore, we need to identify target genes and molecules that can be applied in the clinic. Recently, Kim et al. [16] collected 18 proneural and 12 mesenchymal HGG tumor specimens. They found six significantly upregulated genes (MAPK9, MLK4, LYN, MST4, VRK2 and PRKCH) from 349 protein kinase-encoding genes based on a genome-wide expression dataset for mesenchymal glioma spheres [16], and there is growing evidence that stem cells in these cancers significantly promote cancer initiation and treatment resistance [19]. Therefore, the expression of MAPK9 in glioma warrants further investigation.
C-Jun amino-terminal kinases (JNKs) belong to the typical mitogen-activated protein kinase (MAPK) family. JNKs regulate multiple functions of cells. Stress, growth factors, and cytokines are common activators of JNK2 (MAPK9) expression in humans [20]. MAPK9 has been shown to be overexpressed in various tumors and to promote tumor progression. Wang Zet al. [21] showed that CircRNF20 stimulates the proliferation of non-small cell lung carcinoma (NSCLC) through activation of MAPK9. MAPK9 expression is upregulated in NSCLC patients and correlates with T staging, and MAPK9 overexpression significantly promotes the proliferative capacity of NSCLC cells [21]. In human squamous cell carcinoma (SCC), JNK2 is activated in over 70% of cases, and genetic or pharmacological inhibition of JNK2 prevents tumorigenesis in human SCC cells. It is important to note that JNK2 binds to oncogenic Ras and transforms primary human epidermal cells into SCC malignant tumors [22]. A study by Lili Du et al. [23] demonstrated that JNK2 activity promotes the proliferation and invasiveness of melanoma cells.
In our study, MAPK9 expression was first analyzed in relation to the clinical
characteristics of 150 glioma patients, and we found that MAPK9 expression was
correlated with IDH mutation and the 1p/19q codel, MGMTp methylation and glioma
survival. IDH mutant and 1p/19q codel are molecular phenotypes that have been
included in the WHO classification of gliomas in recent years. IDH mutation,
1p/19q codel and MGMTp methylation are associated with better survival rate in
glioma patients [4]. Our data suggest that MAPK9 is mostly expressed at low
levels in IDH mutant and 1p/19q codel cases, whereas it is highly expressed
(p
Although there has been extensive research, the mechanism by which MAPK9
promotes tumor progression has not been elucidated and may have different
pathways of action in different cancers. In our in vitro experiments, we
demonstrated that MAPK9 knockdown suppressed the tumor phenotype of
primary glioma cells. Epithelial-mesenchymal transition (EMT) and
Wnt/
In summary, MAPK9 may be a meaningful prognostic predictor in glioma and a molecular diagnostic target for glioma.
Our results showed that MAPK9 expression was significantly increased in glioma tissues compared to paraneoplastic tissues. By knocking down and overexpressing the MAPK9 gene in G62 and G91 cell lines, it was demonstrated that MAPK9 may have a facilitative effect on glioma development by activating the Wnt/β-catenin-regulated EMT signaling pathway. The detection of MAPK9 could be used as a strategy to diagnose glioma and predict glioma prognosis.
All data generated or analyzed during this study are included in this published article. Some of the datasets generated and/or analyzed during the current study are available in the CGGA repository, [http://www.cgga.org.cn/index.jsp].
LC and DF conceived and designed the experiments; XY, XL, QJ, WW and ZZ performed the experiments; JS, XY, QJ and YH analyzed the data; XY and ML wrote the manuscript. LC gave the final approval for the manuscript to be published. The manuscript has been read and approved by all the authors, that the requirements for authorship have been met, and that each author believes that the manuscript represents honest work.
The glioma tissue collection was approved by the Ethics Committee of the General Hospital of Northern Command (NO. (2022)028). Each patient has written an informed consent to use part of the tissue for scientific research. All experimental schemes are in accordance with the National Institutes of Health guide for the care and use of Laboratory animals (NIH Publications No. 8023, revised 1978).
We wish to thank Dr. Liang for helpful and valuable suggestions.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




