1 Laboratory of Influenza Research, College of Veterinary Medicine, Chungnam National University, 34134 Daejeon, Republic of Korea
2 Institute of Influenza Virus, Chungnam National University, 34134 Daejeon, Republic of Korea
†These authors contributed equally.
Academic Editor: Vijay Kumar
Abstract
Background: The recently emerged variants of the severe acute
respiratory coronavirus 2 (SARS-CoV-2) pose a threat to public health.
Understanding the pathogenicity of these variants is a salient factor in the
development of effective SARS-CoV-2 therapeutics. This study aimed to compare the
expression patterns of genes involved in immune responses in K18-hACE2 mice
infected with the wild-type, Delta, and Omicron SARS-CoV-2 variants.
Methods: K18-hACE2 mice were intranasally infected with either wild-type
(B.1), Delta (B.1.617.2), or Omicron (B.1.1.529) variants. On day 6
post-infection, lung, brain, and kidney tissues were collected from each
variant-infected group. The mRNA expression levels of 39 immune response genes in
all three groups were compared by RT-qPCR. Viral titers were measured using the
median tissue culture infectious dose (TCID
Keywords
- apoptosis
- cytokines
- Delta
- Omicron
- SARS-CoV-2
- viral pathogenicity
The outbreak of the coronavirus-associated acute respiratory disease (COVID-19), caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), first emerged in China (Hubei Province) in December 2019 [1]. SARS-CoV-2 has a positive single-stranded, non-segmented RNA genome, and its virion comprises four major structural proteins: the nucleocapsid (N), transmembrane (M), envelope (E), and spike (S) proteins [2]. The receptor-binding domain (RBD) of the S proteins interacts with the host receptor, angiotensin-converting enzyme 2 (ACE2), invading epithelial cells in the respiratory and gastrointestinal tracts [3].
SARS-CoV-2 continuously evolves via genetic mutations, mainly in the spike genes, and circulates in the human population, causing viral adaptation to human cells [4]. In March 2021, the World Health Organization (WHO) declared nomenclature for global SARS-CoV-2 variants of concern (VOC), such as Delta and Omicron, and variants of interest (VOI), such as Alpha, Beta, and Gamma [5]. The SARS-CoV-2 Delta (B.1.617.2) variant is 40–60% more transmissible than the Alpha variant (B.1.1.7), increasing the risk of hospitalization [6]. Eight mutations have been identified in the S protein of the Delta variant (T19R, G142D, del157/158, L452R, T478K, D614G, P681R, and D960N), two of which (L452R and T478K) are in the RBD of the S protein [7]. Following the emergence of the Delta variant, a distinct variant emerged and was identified as Omicron (B.1.1.529) by the WHO on November 24, 2021 [8]. An epidemiological study has reported a 48% increased risk of transmission when infected with the Omicron variant than with the Delta variant, owing to the markedly high binding affinity of the Omicron variant with human ACE2 [1, 6]. However, the Omicron variant is associated with lower disease severity than the Delta variant, with a case fatality ratio of 1.9% versus 3.4% for Delta [9].
The immune system detects SARS-CoV-2 infection by recognizing
pathogen-associated molecular patterns (PAMPs), triggering the production of
several inflammatory cytokines and chemokines, including interleukin
IL-1
The pathogenicity and virulence of the SARS-CoV-2 ancestral strain and other VOCs, including Delta and Omicron, have been mainly investigated using clinical samples from hospitalized patients and in silico studies. However, in vivo studies on the pathogenicity of VOCs have been limited. Transgenic mice with human angiotensin I-converting enzyme 2 (hACE2) receptor and promoter cytokeratin 18 (K18; K18-hACE2) are well-established animal models that can reproduce human SARS-CoV-2 infection [12, 13, 14, 15]. Mouse models mimic the gene expression patterns of human inflammatory genes [16]; therefore, they have been used to evaluate the pathogenicity of human SARS-CoV-2 and the efficacy of prophylactics and therapeutics against infections [17, 18, 19].
SARS-CoV-2 associated complications have been reported to be triggered by the expression of different patterns of immune response genes. Therefore, a comprehensive understanding of these expression patterns is paramount for developing effective therapeutics for SARS-CoV-2 infections and terminating the pandemic. However, there is a lack of studies evaluating the pathogenicity of Delta and Omicron variants by comparing the gene expressions of immune-related genes. This study aimed to evaluate and compare the differences in the mRNA expression patterns of cytokines, chemokines, and apoptotic factors in K18-hACE2 transgenic mice infected with the ancestral strain, Delta, and Omicron variants.
Female K18-hACE2 transgenic mice [B6. Cg - Tg (K18-ACE2)2Prlmn/J; 6–7 weeks old] were acquired from the Jackson Laboratory (Bar Harbor, ME, USA). The mice were regularly fed a standard chow diet and water.
SARS-CoV-2 viruses were provided by the Korea Center for Disease Control (KCDC):
wild-type [SARS-CoV-2/Korea/KCDC03/2020(B.1)], Delta variant
[hCoV-19/Korea/KDCA119861/2021(B.1.617.2)], and Omicron variant
[hCoV-19/Korea/KCDC447321/2021(B.1.1.529)]. All viruses were propagated in
Vero-E6 cells obtained from American Type Culture Collection (VERO C1008,
Manassas, VA, USA). Vero E6 cell cultures were maintained in minimal essential
medium (MEM) supplemented with 10% fetal bovine serum (FBS) and 1
All experiments with SARS-CoV-2 were performed in a biosafety level -3 (BSL-3) facility certified by the Korean government.
K18-hACE2 transgenic mice (n = 10 per group) were intranasally infected with 50
The survival of the mice infected with the three SARS-CoV-2 variants was monitored for 14 d.p.i. The change in body weight of the surviving mice after infection was measured at a two-day interval for 14 d.p.i. or until death. Similarly, the body weight of PBS mock K18-hACE2 transgenic mice (n = 5) was measured as a control.
Lung, brain, and kidney tissues were isolated from wild-type-, Delta-, and
Omicron-infected mice on day 6 p.i. Each tissue (0.1 g) was homogenized with 1 mL
of PBS (pH 7.40). The homogenized solutions were filtered using 0.2
Vero cells were cultured in 96-well cell culture plates in MEM supplemented with
10% FBS and 1
After fixation, cells were permeabilized with 80% cold acetone (Samchun Pure Chemical Co., Seoul, Republic of Korea). SARS-CoV-2 nucleocapsid rabbit polyclonal antibody (Thermo Fisher Scientific, Waltham, MA, USA) was added to the cells, followed by incubation with fluorescently labeled goat anti-rabbit antibody (Thermo Fisher Scientific, Waltham, MA, USA). Endpoint titers were evaluated using the Muench and Reed method [20].
Total RNA was extracted from infected mice using the TRIzol RNA extraction kit.
Lung, brain, and kidney tissues were obtained from wild-type-, Delta-, and
Omicron-infected mice (n = 3 per group) at 6 d.p.i. Each tissue sample (0.1 g)
was treated with 1 mL TRIzol reagent (Invitrogen, Carlsbad, CA, USA) and
incubated at room temperature for 5 min. Chloroform (200
cDNA was synthesized using 2.5
RT-qPCR was performed to analyze the expression levels of genes encoding
cytokines, chemokines, TLRs, and apoptotic factors in the lung, brain, and kidney
of SARS-CoV-2 variant-infected mice. The RT-qPCR reaction was set up for a total
volume of 20
To determine tissue histopathology, the lung tissues of mice infected with
SARS-CoV-2 variants (n = 3 per group) were collected at 6 d.p.i., fixed in 10%
phosphate-buffered formalin (Triangle Biomedical Sciences, General Data
Healthcare, Cincinnati, OH, USA) for 6 h, washed with running tap water, and
embedded in paraffin. Tissue sections (5
Statistical significance was determined using the unpaired two-tailed t-test method, and one-way analysis of variance (ANOVA) alpha = 0.05. Data were analyzed using the GraphPad Prism software (version 8.0.1) (Dotmatics, San Diego, CA, USA).
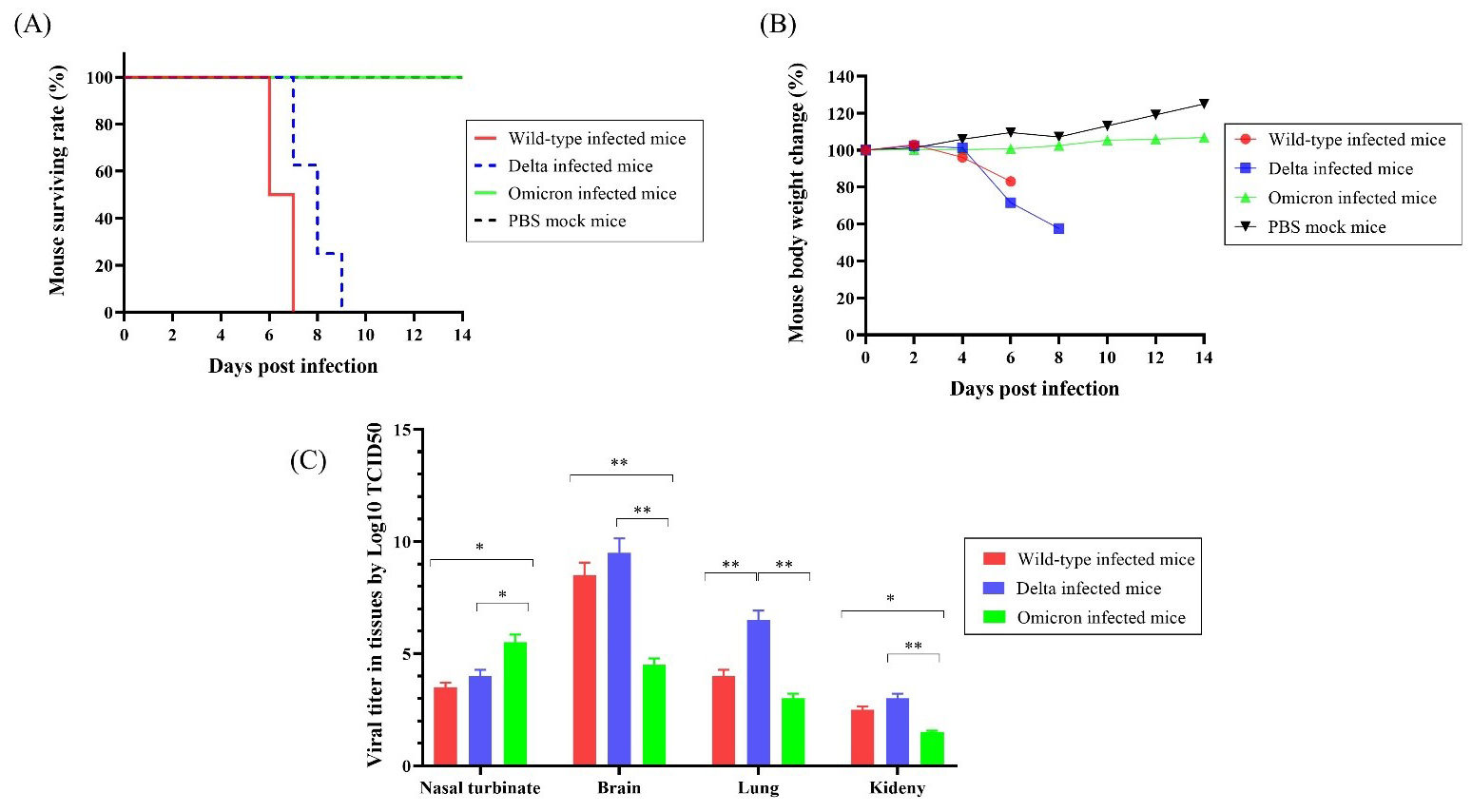
To compare the differences in survival rates after infections with the SARS-CoV-2 variants, we infected K18-hACE2 mice with wild-type, Delta, or Omicron variants (n = 10 per group) and observed mortality and body weight for 14 days. All mice that received the wild-type virus died within 7 days. Delta-infected mice showed 100% survival until 7 d.p.i, then died within two days. However, all mice infected with the Omicron variant and mock PBS exhibited a 100% survival rate until 14 d.p.i. (Fig. 1A).
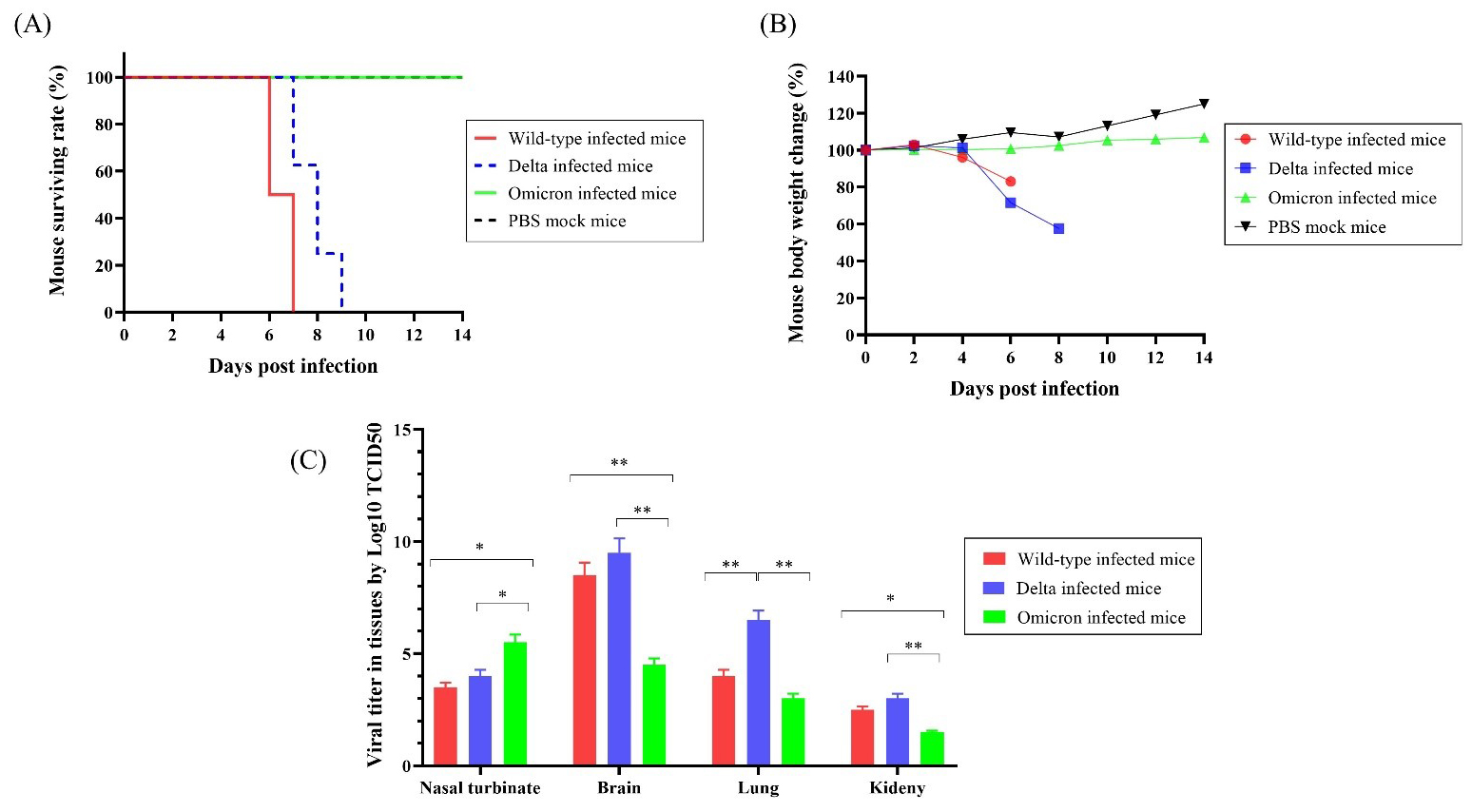 Fig. 1.
Fig. 1.The surviving rate, body weight changes, and viral titers of
K18-hACE2 transgenic mice infected with the SARS-CoV-2 variants (n = 10 per
group). (A) Survival rate of mice infected with wild-type, Delta, and Omicron
SARS-CoV-2 and uninfected mice. (B) Body weight changes after infection with
wild-type, Delta, Omicron, and PBS. (C) Viral titers in tissues of mice infected with
wild-type, Delta, or Omicron determine by Log10 TCID
Wild-type and Delta-infected mice started to lose body weight after 4 d.p.i. and showed 83% and 71% initial mean body weight at 6 d.p.i., respectively. Interestingly, the body weight of Omicron-infected mice increased at 6 d.p.i. and reached 110.5% of the initial mean body weight at 14 d.p.i., whereas mock mice reached 125% of the mean body weight (Fig. 1B). These results indicate that the Omicron group exhibited body weight gain, similar to the mock group, whereas the wild-type and Delta groups showed body weight reduction. However, these differences were not statistically significant.
The viral titers in each tissue of the infected mice were measured using the
Log10 median tissue culture infectious dose (TCID
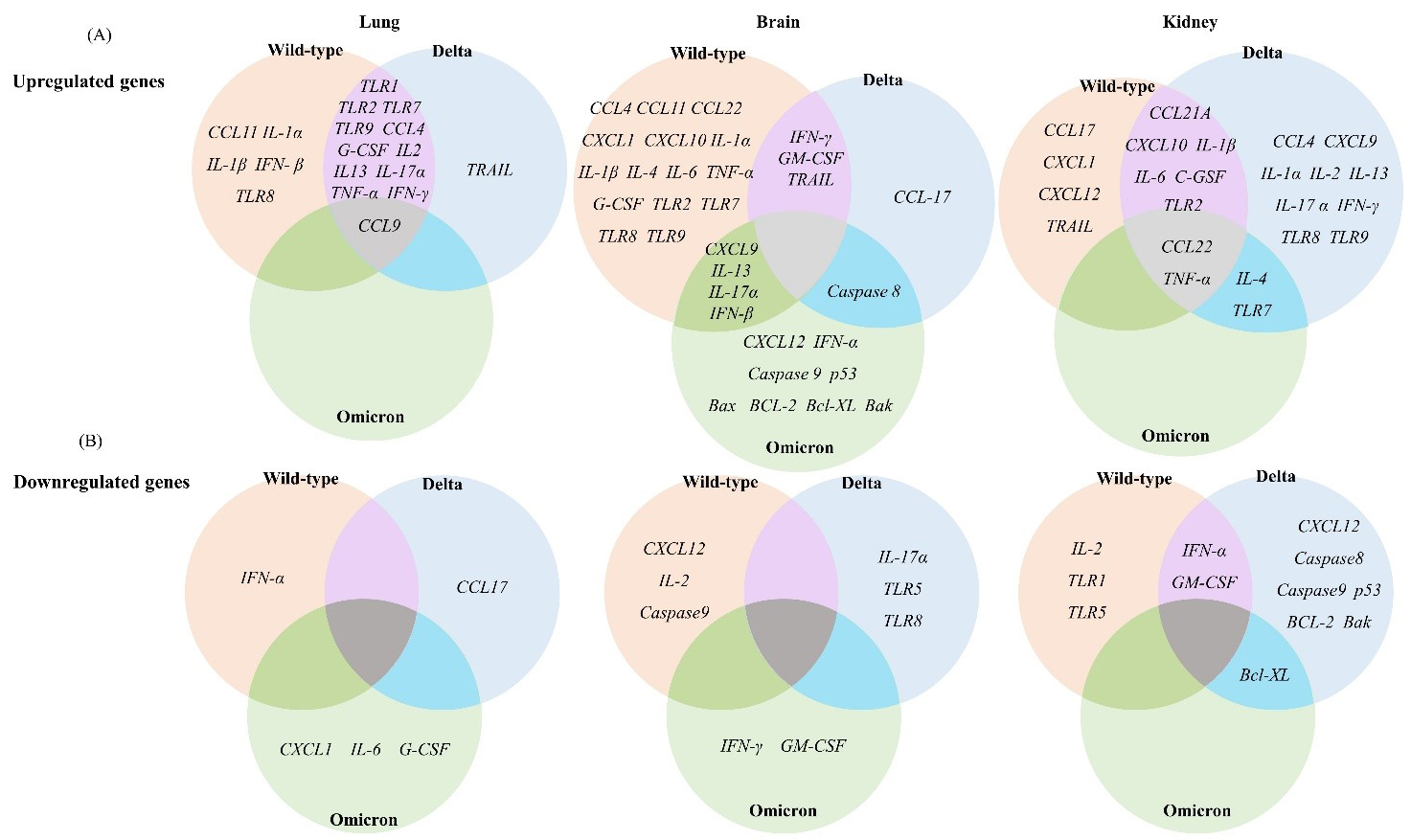
To evaluate the pathogenicity of the wild-type, Delta, and Omicron SARS-CoV-2 variants at the molecular level, we analyzed the expression levels of 39 immune response genes in the lung, brain, and kidney tissues of mice collected at 6 d.p.i. by RT-qPCR. The selected 39 immune response genes belong to various functional categories, including TLRs, cytokines, chemokines, and apoptotic factors. Relative gene expression levels were determined by relative fold change based on the gene expression levels of the PBS mock-treated group. The differential expression of each gene in different tissues of the wild-type, Delta, and Omicron groups is illustrated and summarized in a heatmap (Figs. 2,3,4,5,6 and Supplementary Figs. 1,2).
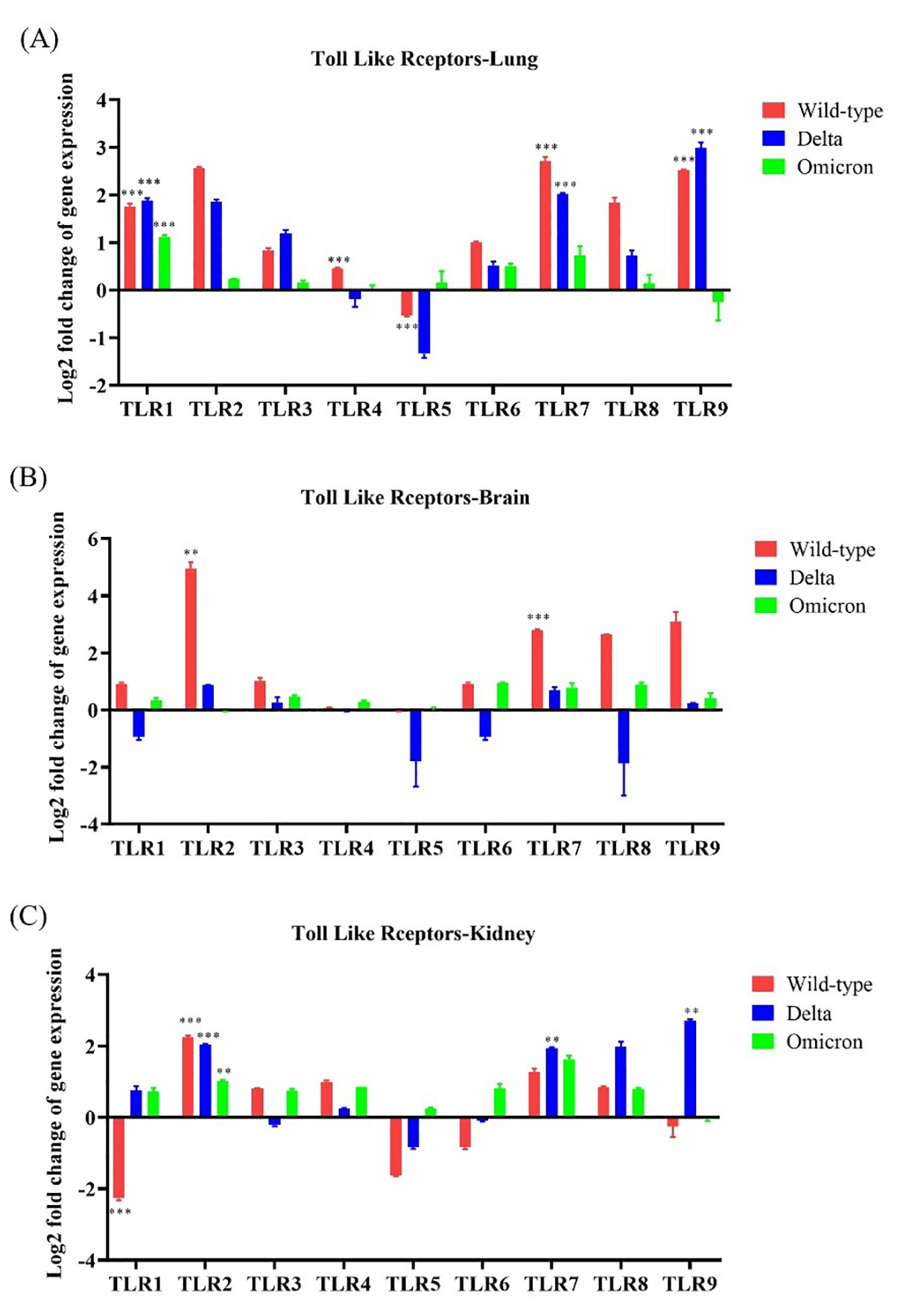
To identify innate immune activation following SARS-CoV-2 infection, we
evaluated TLR gene expression in the lung, brain, and kidney of wild-type-,
Delta-, and Omicron-infected mice. TLR1, TLR2, and
TLR7 were upregulated (
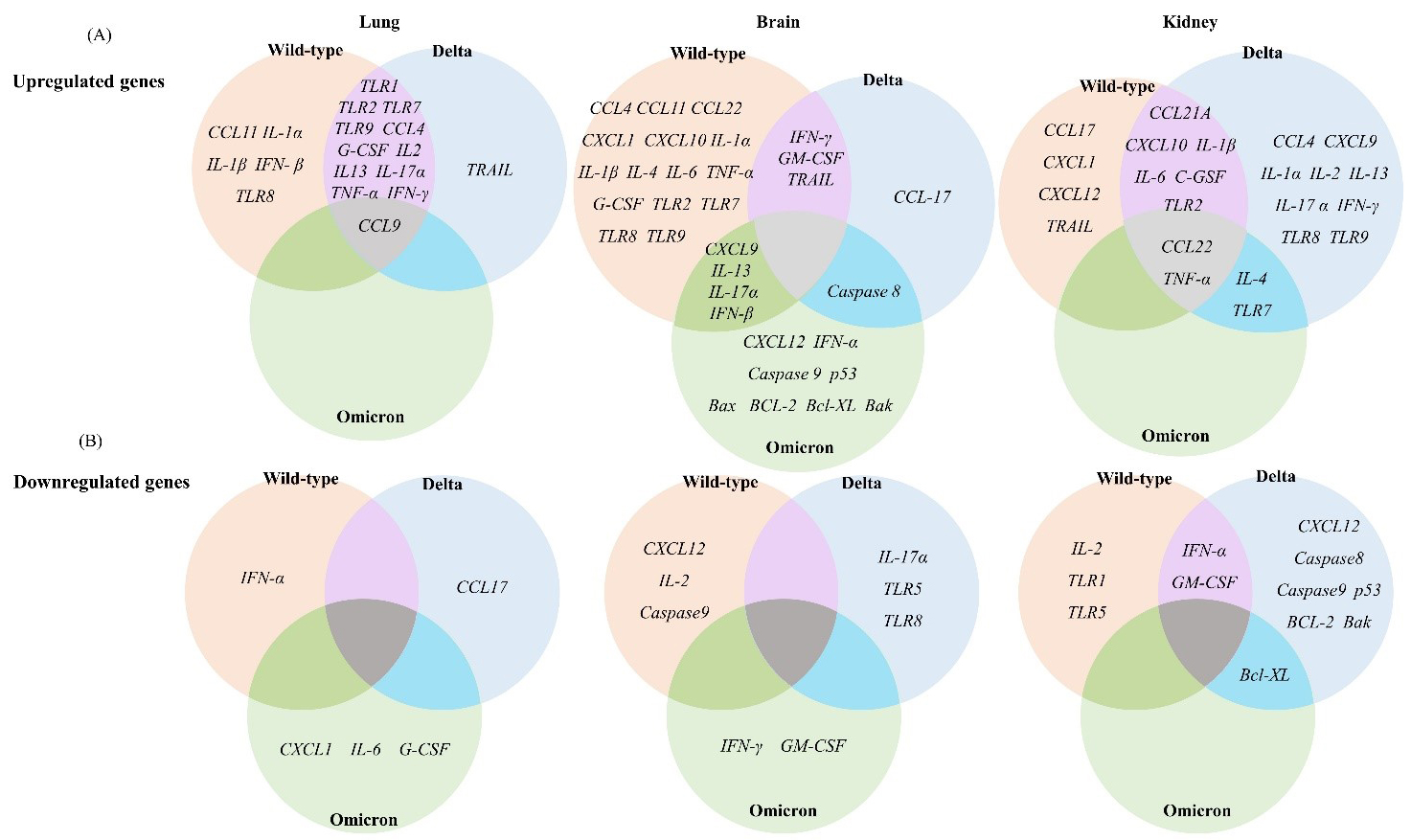 Fig. 2.
Fig. 2. Differential expression of immune response genes in K18-hACE2
transgenic mice after infections with the SARS-CoV-2 variants. (A) Upregulated
mRNA expression of genes related to the immune response in the lung, brain, and
kidney tissues of wild-type-, Delta-, or Omicron-infected mice. (B) Downregulated
mRNA expression of genes related to the immune response in the lung, brain, and
kidney after infection with wild-type, Delta, or Omicron (
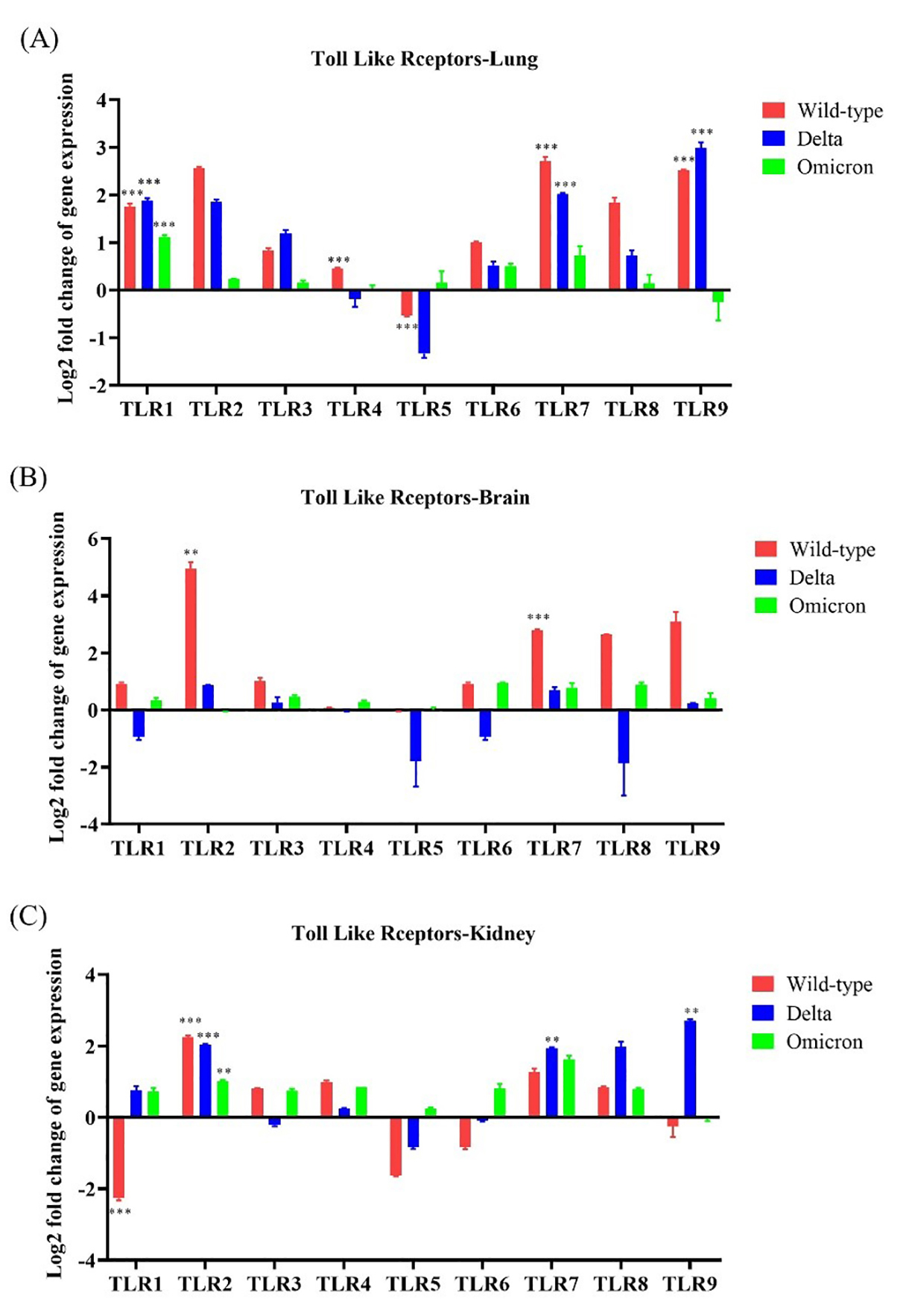 Fig. 3.
Fig. 3. Toll-like receptor (TLR) gene expression in the lung,
brain, and kidneys of K18-hACE2 transgenic mice infected with SARS-CoV-2
variants. The mRNA expression of each gene was normalized to that of mouse
Overall, these results indicate that infections with the wild-type and Delta SARS-CoV-2 resulted in increased TLR expression in the lung and brain tissues, whereas infection with the Omicron variant did not.
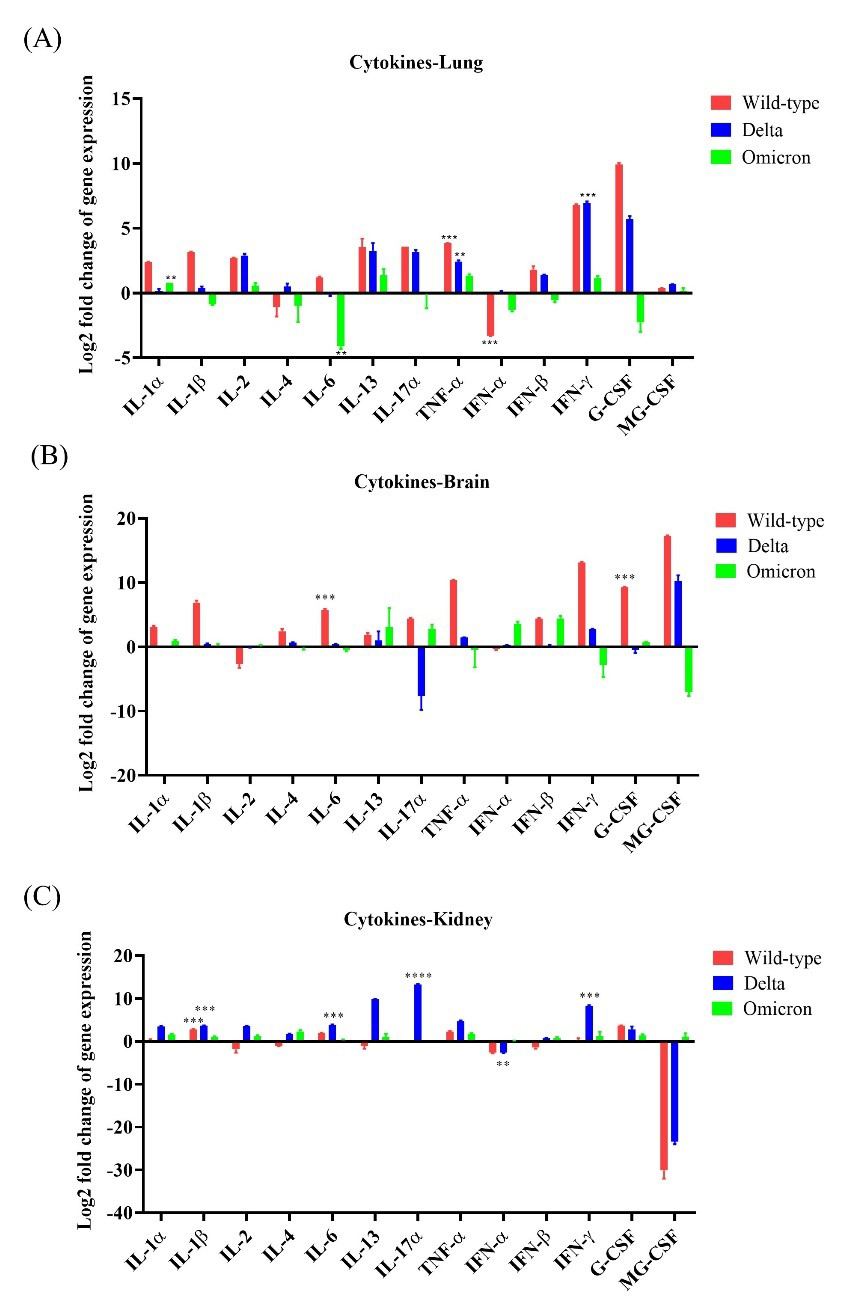
IL-1
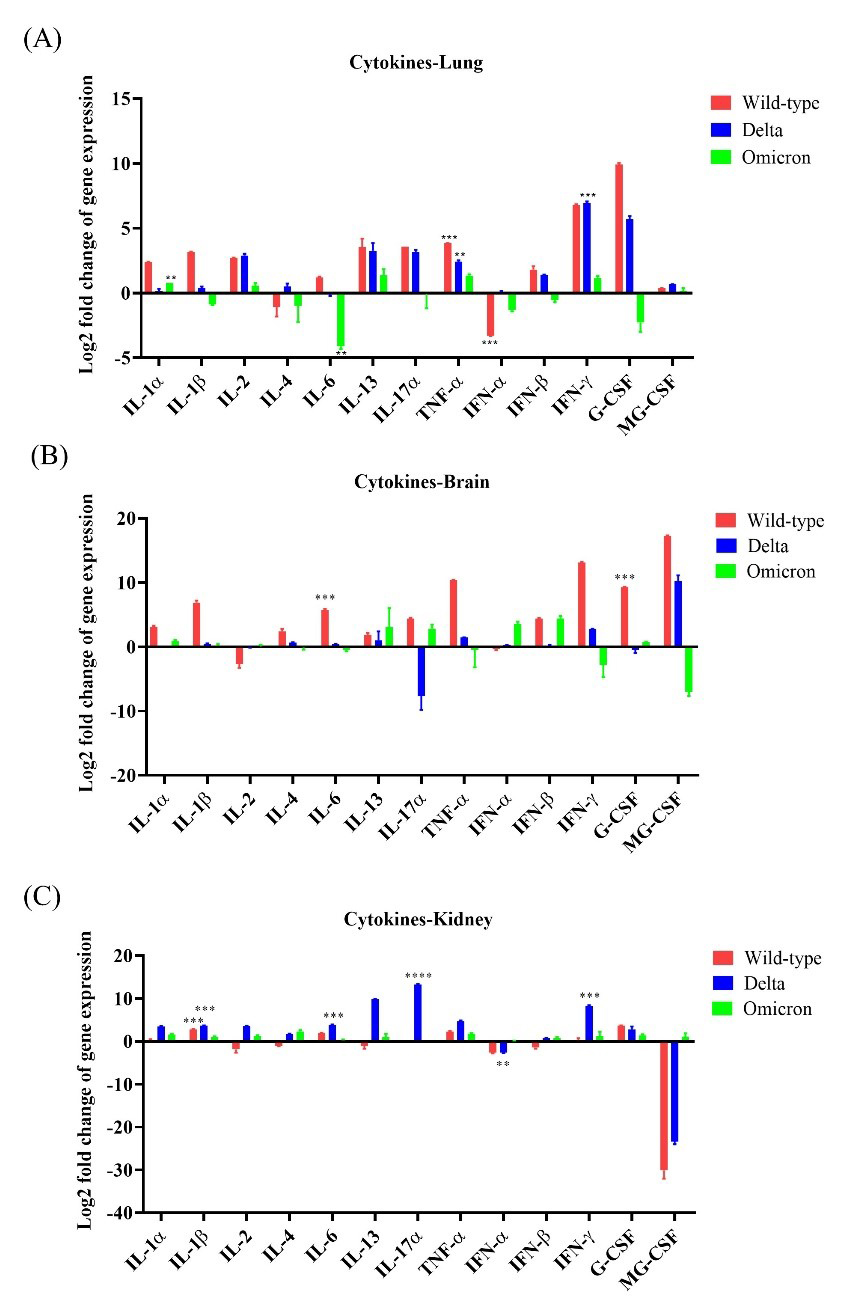 Fig. 4.
Fig. 4.Cytokine expression in K18-hACE2 mice after infections with
SARS-CoV-2 variants. The expression levels of 13 cytokine genes (seven
interleukins, TNF-
IL-4 and IL-6 expression levels were upregulated by 5.43 and
51.65, respectively, in the brain tissues following infection with the wild-type,
and the increase in IL-6 expression was statistically significant
(p
IL-17A was moderately upregulated in lung tissues after infection with the wild-type or the Delta variant by 12.0-fold and 8.91-fold, respectively. The brain tissues of wild-type- and Omicron-infected mice exhibited increased IL-17A expression levels by 21.27-fold and 7.40-fold, respectively, whereas those of Delta-infected mice showed drastic IL-17A downregulation compared to the control. Interestingly, IL-17A expression levels were markedly increased in the kidney after infections with wild-type, or Delta and Omicron variants compared to the control (Fig. 4A–C).
In addition, we evaluated the expression levels of IFN and TNF
in the lungs, brain, and kidneys of the mice infected with the three variants.
IFN-
Overall, these results indicate that cytokine production was the highest in the lungs of mice infected with wild-type compared to that in different tissues of the other groups.
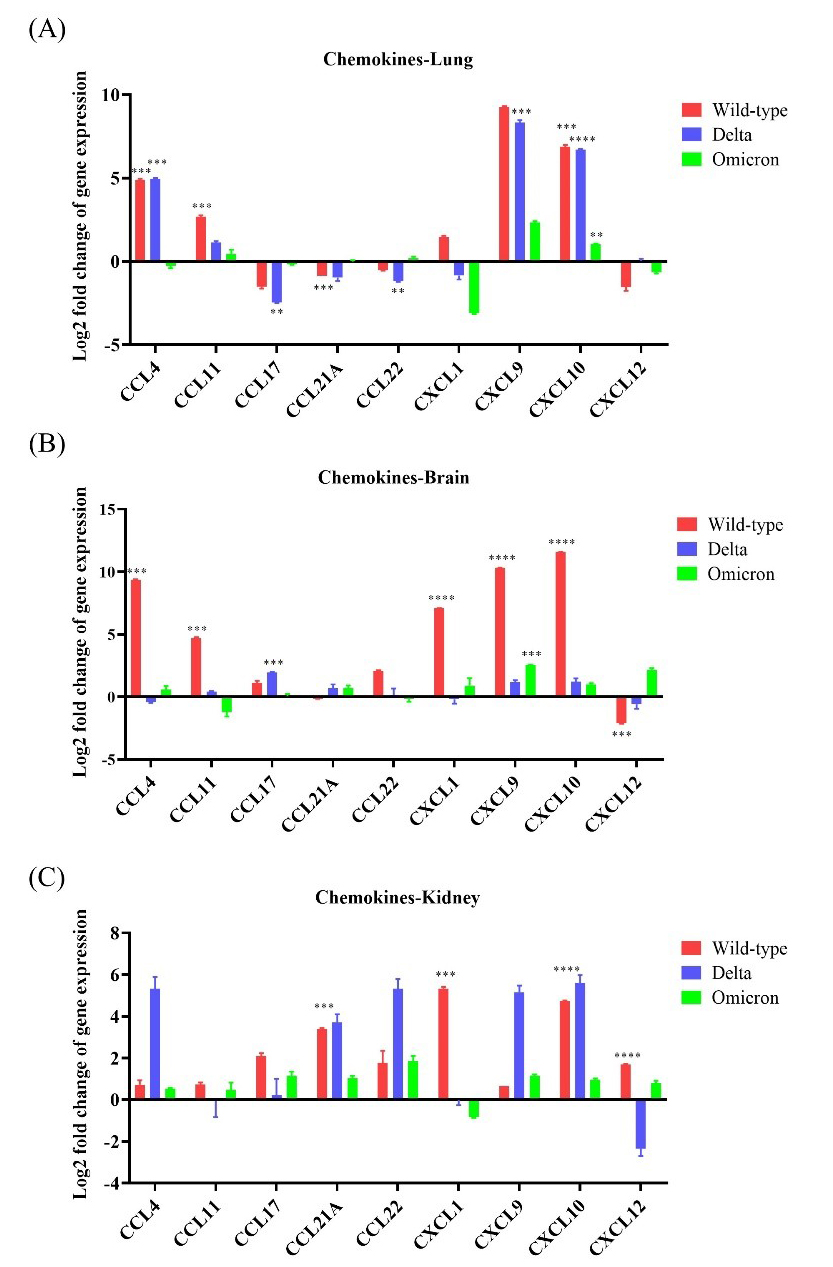
The lungs, brain, and kidneys of wild-type-infected mice showed the highest chemokine
expression compared to the other infected groups. Overall, CCL4,
CCL11, CXCL9, and CXCL10 were upregulated (
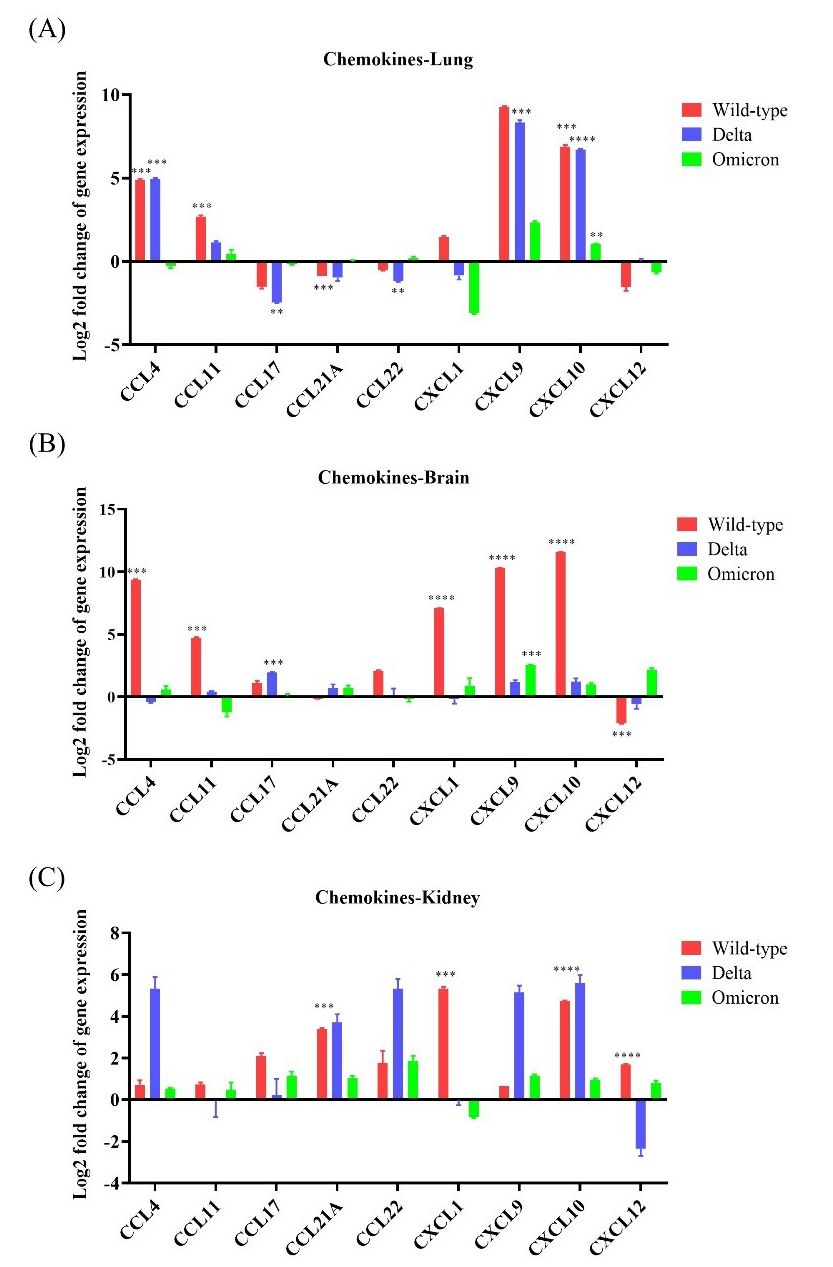 Fig. 5.
Fig. 5.Chemokine expression in K18-hACE2 mice infected with
SARS-CoV-2. The mRNA expression levels of nine chemokine genes (CCL4,
CCL11, CCL17, CCL21A, CCL22, CXCL1,
CXCL9, CXCL10, and CXCL12) were evaluated after
infection with wild-type, Delta variant, or Omicron variant. (A) mRNA
expression of the chemokine genes in the lung after inoculation with wild-type,
Delta, or Omicron. (B) mRNA expression of the chemokine genes in the brain after
inoculation with wild-type, Delta, or Omicron. (C) mRNA expression of the chemokine
genes in the kidney after inoculation with wild-type, Delta, or Omicron. * p
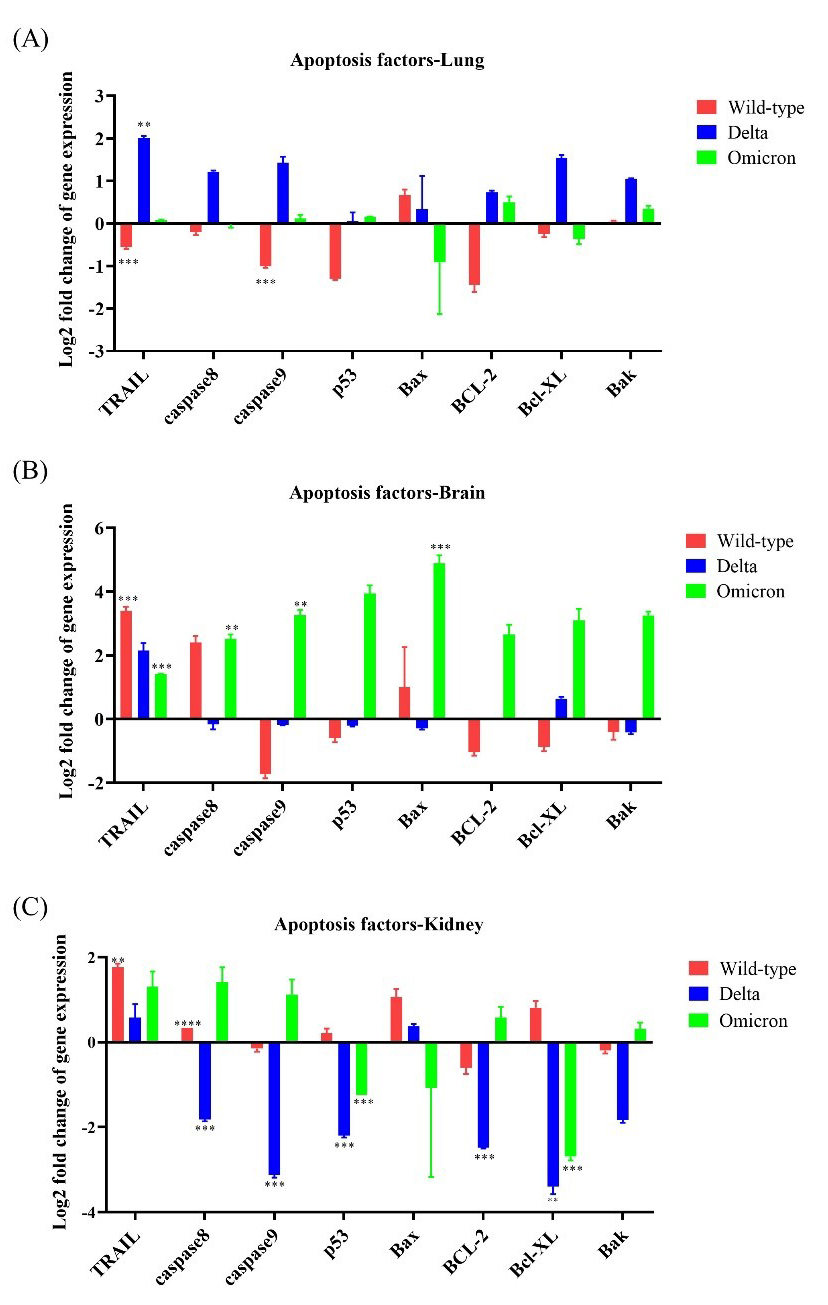
Apoptotic factors, including caspase 8, caspase 9,
p53, Bax, Bak, BCL-2, and Bcl-XL,
were mainly expressed in brain tissues of Omicron-infected mice; neither the lung
nor the kidney showed more than 3-fold upregulation of these apoptotic factors.
TRAIL was upregulated by
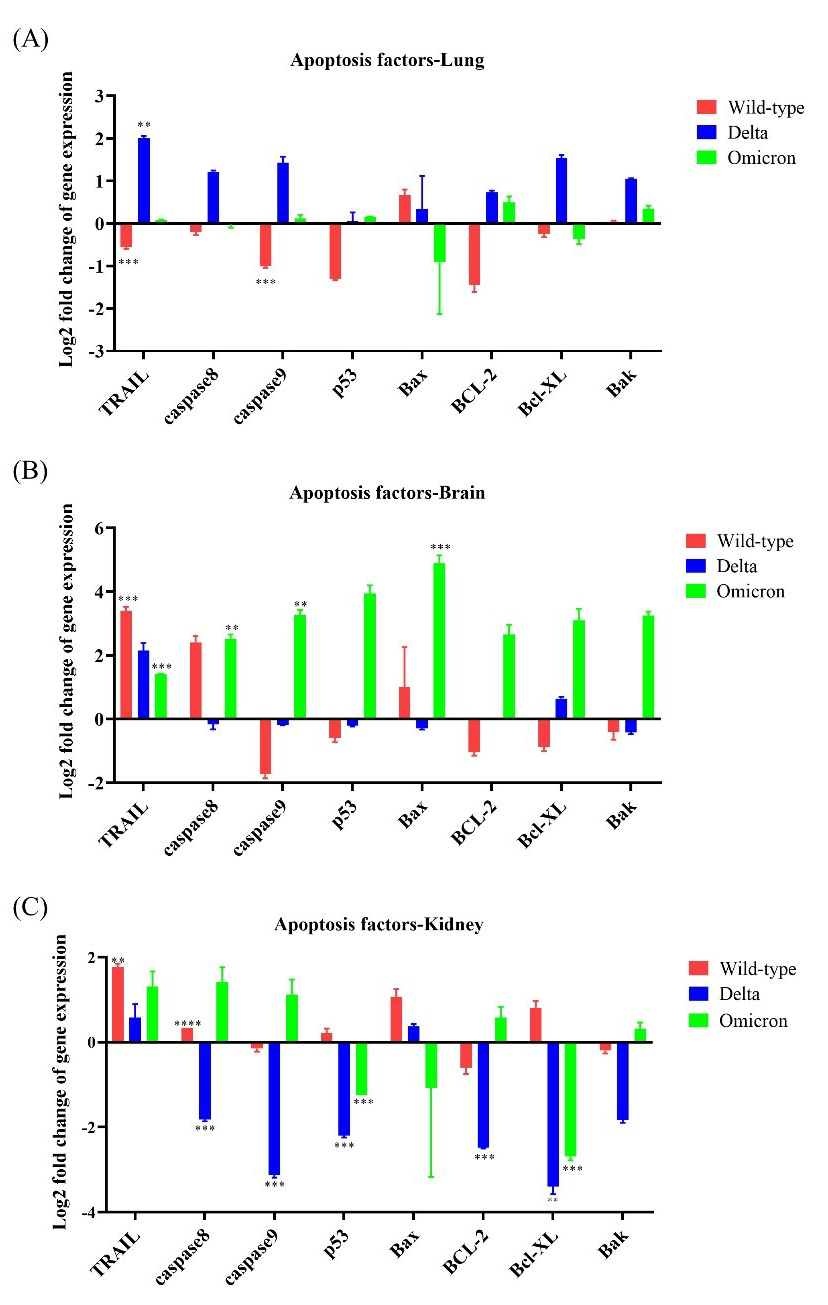 Fig. 6.
Fig. 6.Expression levels of apoptotic genes in K18-hACE2 mice infected
with SARS-CoV-2 variants. The expression levels of eight apoptotic
factor-related genes (TRAIL, caspase 8, caspase 9,
p53, Bax, BCL-2, Bcl-XL, and Bak)
were evaluated after infection with wild-type, Delta variant, or
Omicron variant. (A) mRNA expression of apoptotic genes in the lungs after
inoculation with wild-type, Delta, or Omicron. (B) mRNA expression of apoptotic genes
in the brain after inoculation with wild-type, Delta, or Omicron. (C) mRNA expression
of apoptotic genes in the kidney after inoculation with wild-type, Delta, or Omicron.
* p
To compare the disease severity caused by the SARS-CoV-2 variants, the lung pathology of K18-hACE2 transgenic mice infected with the wild-type, Delta, and Omicron was observed at 6 d.p.i. after H&E staining. No signs of pneumonia were observed in the lung tissues of PBS mock mice (Fig. 7A). In contrast, mice infected with the wild-type showed signs of severe interstitial pneumonia, which is characterized by inflammatory cell infiltration (Fig. 7B). The lung tissue of mice infected with the Delta variant exhibited mild pneumonia (Fig. 7C). Interestingly, the lung tissues of mice infected with the Omicron variant exhibited considerably milder interstitial pneumonia than those of mice infected with the Delta variant (Fig. 7D).
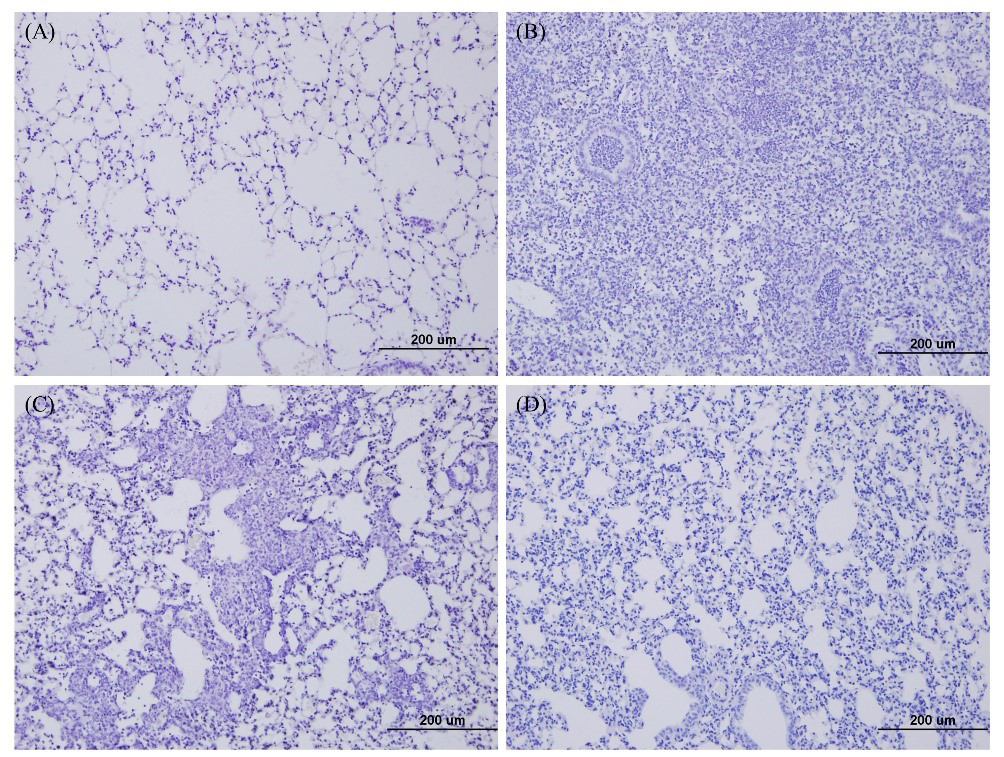 Fig. 7.
Fig. 7.Lung histopathology of K18-hACE2 transgenic mice infected with SARS-CoV-2 variants. At 6 d.p.i., the lung tissues of mice infected with the wild type, Delta, or Omicron variants were collected, and hematoxylin and eosin staining was performed. Lung histopathology was used to compare disease severity caused by each variant and wild-type. (A) Lung tissue of mock-infected mice. (B) Lung tissue of wild-type-infected mice. (C) Lung tissue of Delta-infected mice. (D) Lung tissues of Omicron-infected mice.
Among the known VOCs, the Delta and Omicron variants have been identified as dominant variants at different time points during the COVID-19 pandemic and are still persistent in the human population. Although the Delta variant has been reported to have overt pathogenicity, Omicron is the most recent variant reported to have remarkably attenuated pathogenicity and robust transmissibility [21, 22]. The pathogenicity of a viral pathogen depends on three factors: the accessibility of the virus to the host tissue, host cell susceptibility to virus multiplication, and virus susceptibility to host defense mechanisms [23].
Considering the outbreak indices, we focused on the pathogenicity profiles of the wild-type and the variants Delta and Omicron by analyzing the expression of immune response-related genes and the dynamics of infections in K18-hACE2 transgenic mice. Our results revealed that the wild-type and Delta variant resulted in a pronounced weight loss from 4 d.p.i. until death in vivo. In contrast, mice infected with the Omicron variant did not lose weight and survived until study termination. Moreover, our results indicated that the viral burden in the Omicron-infected K18-hACE2 mice was lower than that in the Delta- and wild-type-infected mice. Collectively, our results reveal that the Omicron variant exhibited lower pathogenicity in K18-hACE2 mice than the Delta variant and wild-type, which is consistent with the preliminary findings on the pathogenicity of SARS-CoV-2 in humans. Moreover, multiorgan tropism, including in the brain and kidney, was observed in the Delta- and Omicron-infected K18-hACE2 mice as well as in the infection of wild-type. The tissue tropism of the SARS-CoV-2 variants may be mainly attributed to the enhanced affinity and accessibility of the S protein to the ACE2 receptor. Although Omicron-infected mice showed lower viral loads in the lung, brain, and kidney tissues compared to those infected with the wild-type or Delta variant, the nasal turbinate of the Omicron-infected mice had significant viral titer even after 6 d.p.i. This may explain viral shedding and transmissibility associated with Omicron.
Other research works conducted using K18-hACE2 mice showed significantly attenuated replication of Omicron (B1.1.529) in lung and brain implying similar results to our findings [24, 25, 26]. As reported by Meng et al. [27] the impaired cleavage of spike protein and inefficient usage of TMPRSS2 cellular protein which is required in cell entry are the factors associated with the lower replication of Omicron compared to the other SARS-CoV-2 variants.
Most COVID-19 complications occur due to the dysregulated immune response caused by SARS-CoV-2 infection. Inflammation and cell death have been proposed as major mechanisms underlying severe COVID-19 [28, 29, 30, 31]. Our findings indicate that the wild-type and the Delta variant result in a more pronounced upregulation of TLR, cytokine, and chemokine genes compared to the Omicron variant. Moreover, genes that were differentially expressed in wild-type- and Delta-infected mice overlapped closely, showing similar disease progression patterns. Although TLR and cytokine expression was lower in Omicron-infected mice, high apoptotic factors-related gene expression levels were observed in these mice, especially in the brain, suggesting immune evasion. These results suggest that the Omicron variant is more evolved than the other SARS-CoV-2 variants.
In SARS-CoV-2 infections, TLRs act as double-edged swords that induce
immune-mediated pathology rather than preventing the progression of infection
[32, 33, 34]. Among TLRs, TLR2 recognizes SARS-CoV-2 and other
Cytokines are key drivers of inflammatory responses, which directly modulate
disease severity [37]. Many studies have described the increased expression of
cytokines, including IL-1/2/4/6/10/17, IFN-I/II/III,
TNF, G-CSF, GM-CSF, MCP-1,
MIP-1
In addition to cytokines, we also observed overexpression of chemokines in our study. Chemokine-mediated acute respiratory complications have been reported to be associated with 40% of deaths. Furthermore, we observed increased expression levels of TLRs, cytokines, and chemokines in the kidney, especially in Delta-infected mice, which may lead to acute kidney injury (AKI) as a complication of long-term COVID-19 infections. Our results are consistent with several studies reporting that patients with COVID-19 infected with the Delta variant experienced lingering AKI [49, 50].
Moreover, programmed cell death has been proposed as a pathway for SARS-CoV-2 pathogenesis [51]. In agreement with this hypothesis, in our study, Omicron-infected mice expressed higher levels of apoptotic factors compared to mice infected with the other two SARS-CoV-2 variants. Elevated expression levels of caspase 8 and caspase 9 in the brain tissues of the Omicron infected mice ratified the involvement of caspase mediated apoptotic pathways in SARS-CoV-2 infection. The experiments using Vero E6 cells have reported that the activation of caspase 8 and 9 and BID was induced by the SARS-CoV-2 ORF3a which ultimately results the cell death [52]. Upregulation of Bax, and BAK genes which cause the mitochondrial outer membrane permeabilization provide evidence for the activation of intrinsic pathway of apoptosis cell death by Omicron in the brain. However, the opening of the gateways of caspase 8 ultimately initiates apoptosis [53].
Our study stressed that even though the impaired Omicron viral replication in the brain, it can cause the upregulation of certain apoptosis factor related genes. This result complies with the findings of Frank et al. [54]; the S1 subunit of spike protein of SARS-CoV-2 is solely enough to act as a pathogen associated molecular pattern (PAMP) and drive the neuroinflammatory responses independent of viral infection. However, the low viral titers in brain shown with Omicron was not found to be associated with either the neuropathological changes or levels of immune response gene expression [55, 56]. Similarly, RT-qPCR and immunohistopathological analysis of brain tissues of de positive patients by Matschke et al. [57] has confirmed the presence of SARS-CoV-2 virus in brain even though it’s not correlated with the neuropathology of brain. Our results also corroborated with the above-mentioned research findings on differences of viral titer and the neuropathological changes.
As Omicron induce the programmed cell death associated immune responses in brain, the central nervous system associated manifestations might be occurred in the Long COVID sequelae. The experiment in mice provides evidence for persistent post-acute neurological complications after exposure to COVID-19 [58]. Another study which uses cohort of COVID-19 patients has reported the participants diagnosed with the neurological conditions including memory impairment, cerebrovascular disorders, cognition and sensory disorders in post-acute phase and furthermore COVID-19 patients have a 42% risk of developing neurological complications within a year post infection [59, 60]. Not only neurological complications but also pulmonary, cardiovascular, endocrine, hematologic, renal, gastrointestinal, dermatologic, immunological issues were encompassed by long COVID sequelae [60]. Therefore, therapeutic and diagnostic algorithms should be aided for the resolution of consequences of acute and post-acute COVID conditions.
In addition, our histopathology results suggest that severe pneumonia in the lungs of wild-type- and Delta-infected mice may have arisen due to a cytokine storm resulting from the increased levels of cytokines and chemokines.
Our study revealed that the pathogenicity of SARS-CoV-2 varies in the following
order: wild-type
The data are available from the corresponding author upon reasonable request.
SHS designed the research study. All authors performed the research. SHS provided help and advice on experiments. KAPPK and YJ analyzed the data and wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
All experiments involving animals were approved by the Internal Animal Use Committee at Chungnam National University (CNU) (Approval: 202206A-CNU-124). The research work was performed according to the guidelines and regulations of CNU, the Republic of Korea.
We would like to thank Editage (https://www.editage.com) for English language editing.
This work was partly supported by a grant by the National Research Foundation of Korea (NRF) funded by the Korean government (MSIT) (2019R1A2C2002166812).
The authors declare no conflict of interest. Sang Heui Seo was serving as Guest Editor of this journal. We declare that Sang Heui Seo had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Vijay Kumar.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/j.fbl2711316.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.