1 Huzhou Third Municipal Hospital, The Affiliated Hospital of Wenzhou Medical University, 313000 Huzhou, Zhejiang, China
2 Laboratory of Translational Medicine, Affiliated Cixi Hospital, Wenzhou Medical University, 315300 Ningbo, Zhejiang, China
3 School of Mental Health, Wenzhou Medical University, 325035 Wenzhou, Zhejiang, China
4 School of Pharmacy, Wenzhou Medical University, 325035 Wenzhou, Zhejiang, China
5 Beijing Hui-Long-Guan Hospital, Peking University, 100096 Beijing, China
6 Key Laboratory of Psychosomatic Medicine, Inner Mongolia Medical University, 010110 Hohhot, Inner Mongolia, China
7 The Affiliated Kangning Hospital, Wenzhou Medical University, 325035 Wenzhou, Zhejiang, China
†These authors contributed equally.
Academic Editor: Maria Pina Concas
Abstract
Background and Aims: Nicotine dependence (ND)-induced anxiety might be
modulated by genetic polymorphisms. The gene-by-environment interaction can be
fitted into the diathesis-stress and differential susceptibility models.
Nevertheless, knowledge of the interaction between adiponectin (ADPN)
polymorphisms and ND on the incident mental disorder is currently scarce. This
study aims to understand the role of ADPN rs266729 on anxiety in
patients with ND while elucidating the psychology model and the various reactions
across genotypes. Methods: We included 315 Chinese males with confirmed
ND, measured using the Fagerstrom test for nicotine dependence (FTND). Anxiety
was assessed using the Self-rating Anxiety Scale. Genomic DNA was extracted and
genotyped from peripheral blood. Hierarchical regression models were used to test
the interactions. Results: There was a significant interaction between
ADPN rs266729 and ND (
Keywords
- nicotine dependence (ND)
- anxiety
- polymorphism
- adiponectin
- differential susceptibility
Tobacco abuse is a global health problem associated with nicotine dependence
(ND) [1]. A 2012–2014 study found that the prevalence of cigarette smoking was
Adiponectin (ADPN) is a plasma protein secreted by adipocytes; it is connected
with ND or anxiety. Previous studies suggested that nicotine administration
decreased serum ADPN levels [11, 12, 13]. Regarding the relationship between
adiponectin and anxiety, decreased ADPN was associated with negative
emotion-related behaviors in chronic social defeat model mice. This reduction can
be reversed by rosiglitazone (a PPAR
The precise form of the interaction between the ND and ADPN rs266729
polymorphism has not been investigated. According to the literature on G
Our study aims to elucidate the effect of ADPN rs266729 on anxiety in
patients with ND, clarifying the psychological model and the varying reactions
associated with different genotypes. Specifically, the form of G
The 315 men in the study were drawn from our previous study [21], including 300 alcohol-dependent (AD) inpatients recruited from psychiatric hospitals in northern China and 15 healthy controls (HCs) from Inner Mongolia Autonomous Region were recruited from December 2009 to December 2012. All patients met the criteria for ND based on the Diagnostic and Statistical Manual of Mental Disorders, 4th edition (DSM-IV). The exclusion criteria were as follows: (1) other drug abuse or dependence, except alcohol; (2) severe cardiovascular disease, liver disease, or kidney disease; (3) participant, or a first-degree relative of the participant, with a severe mental illness.
The participants were asked to complete a series of questionnaires and provide a blood sample for DNA extraction. All staff involved in this study were trained before the study commenced. The Institutional Review Board of the Inner Mongolian Medical University approved the study (Ethic approval number: YKD2015003). All patients provided written informed consent and were told that the blood sample would be subjected to a gene assay.
The Fagerstrom test for nicotine dependence (FTND) was used to assess ND [22]. FTND is the most used scale for assessing ND. The FTND scores ranged from 0 to 10. The scale consists of six questions with scores at every answer related to the smoker’s level of ND. A score of 1–3 was categorized as low ND, 4–6 is medium ND, and 7–10 is high ND [23].
The Self-rating Anxiety Scale (SAS) was used to assess each participant’s level of anxiety. Zung introduced the SAS [24] for measuring what may be described as state anxiety, a term referring to “a transitory emotional state or condition of the human organism that is characterized by subjective, consciously perceived feelings of tension and apprehension and heightened autonomic nervous system activity” [25]. The questionnaire uses a four-point Likert score, ranging from 1 (none or a small amount of time) to 4 (most or all of the time). SAS has satisfactory psychometric properties, Cronbach’s alpha = 0.82 [26].
Alcohol dependence level was measured using the Michigan Alcoholism Screening Test (MAST) [27]. The MAST is a questionnaire containing a 25-item self-report in which respondents rated the severity of a range of dependence-related alcohol use behaviors using a 4-point scale ranging from 1 (not at all) to 4 (very much). The scale has high internal consistency with Cronbach’s Alpha value of 0.90 [28].
Genomic DNA was extracted from 5 ml of peripheral blood of each participant using the salting-out method. Primers were designed using Assay Design 3.1 software from Sequenom (San Diego, CA, USA) and synthesized by Gene-Cloud Biotechnology Co., Ltd. (Beijing, China). PCR-specific and single-base extension primer sequences are provided as follows: 5-ACGTTGGATGACACCTTGGACTTTCTTGGC-3, 5-ACGTTGGATGATGTGTGGCTTGCAAGAACC-3, and single-base extension primer: 5-GCTCATGTTTTGTTTTTGAAG-3. Reactions were carried out according to the manufacturer’s protocol [29, 30]. All laboratory procedures were carried out in a manner blind to case-control status. The conditions of PCR were as follows: 50 °C for 2 min, 95 °C for 10 min, followed by 50 cycles of 95 °C for 15 s and 60 °C for 1 min. Ten percent of the DNA samples were duplicated randomly and tested, and no-fault genotyping was found.
First, the Hardy-Weinberg equilibrium for genotype distributions of ADPN rs266729 was tested using the Chi-square test for goodness of fit. Then, Pearson correlations were examined between genetic polymorphisms, age, years of education, ND, and anxiety. Consistent with other studies [31, 32, 33], CG and GG genotypes were collapsed into a G allele group coded as one and the CC genotype coded as 0. Then, traditional linear regression was used to test the interaction between ND and the rs266729 polymorphism. When a significant interaction was found, region of significance (RoS) analysis was used to examine the form of the interaction [34].
Finally, a re-parameterized regression model was fitted to examine the specific
pattern of gene
Diathesis-Stress model is generated by the re-parameterized regression equations. In this regression equation, independent variable (X) is FTND scores, dependent variable (Y) is anxiety, age and years of education are demographic covariates (X2, X3). All variables are continuous variables and model were fitted using the least-squares method [35]. Crossover point C is a point where the slopes of the different groups cross. In differential-susceptibility, crossover point C is estimated by regression equations. What distinguishes the diathesis-stress and differential susceptibility models is the estimate and interval estimate of crossover point C. If the estimate and interval estimate of crossover point C fall within the range of ND, the model is consistent with the differential susceptibility model. Otherwise, if crossover point C is over the maximum of ND, the model is consistent with the diathesis-stress model. Results of the final best-fit models arrived at via model comparison, including F test, Akaike information criterion (AIC) and Bayesian information criterion (BIC).
The diathesis-stress and differential susceptibility models can be further subdivided into strong and weak versions. Strong versions assume that non-risk/non-plasticity allele carriers are not susceptible to the environment. Weak versions assume both allele carriers are susceptible to the environment; however, non-risk/non-plasticity carriers are less susceptible to the environment than risk/plasticity carriers. These models are nested within one another. Therefore, the F-test was used to compare the models and identify a difference in the parameter estimates. For non-nested models, the Akaike information criterion and Bayesian information criterion were compared to evaluate which model was a better fit.
Of the 315 male inpatients, 157 (49.8%) were CC homozygotes, 133 (42.2%) were
CG heterozygotes, and 25 (7.9%) were GG homozygotes. The genotype distribution
of ADPN rs266729 was consistent with Hardy-Weinberg equilibrium
(
| Genotype | Number of people | Percentage | |
| CC | 157 | 49.8% | |
| CG | 133 | 42.2% | |
| GG | 25 | 7.9% | |
| 0.19 | p | 0.67 | |
| Variables | M |
| Age | 45.22 |
| Educational Years | 10.25 |
| Nicotine Dependence (FTND scores) | 5.89 |
| Anxiety (SAS scores) | 35.03 |
| M, mean; SD, standard deviation. | |
| rs266729 polymorphism | Age | Educational Years | Nicotine Dependence (FTND) | Anxiety (SAS) |
| CC | 45.06 |
10.28 |
5.83 |
35.32 |
| G | 45.37 |
10.22 |
5.95 |
34.75 |
| t | –0.31 | 0.19 | –0.43 | 0.60 |
| p | 0.76 | 0.85 | 0.67 | 0.55 |
| CC, CC homozygote; G, G allele; t, independent t-test; p, p-value for t-test. | ||||
The descriptive statistics for each research variable are shown in Table 4. FTND
scores were positively correlated with SAS scores (r = 0.15, p
| rs266729 | Age | Educational Years | Nicotine Dependence (FTND) | Anxiety (SAS) | |
| rs266729 | 1 | ||||
| Age | 0.02 | 1 | |||
| Educational Years | –0.02 | –0.28 |
1 | ||
| Nicotine Dependence (FTND) | 0.03 | 0.03 | –0.10 | 1 | |
| Anxiety (SAS) | –0.04 | –0.3 | –0.09 | 0.15 |
1 |
| M | (–) | 45.22 | 10.25 | 5.89 | 35.03 |
| SD | (–) | 9.08 | 2.73 | 2.53 | 8.56 |
| M, mean; SD, standard deviation. Note: | |||||
Next, hierarchical regression models were used to predict anxiety from ND and
test the interaction between ND and rs266729 polymorphism on anxiety, with age
and years of education as covariates. Table 5 shows that ND had a significant
main effect on anxiety symptoms (
| Variables | Anxiety (SAS) | |||||
| ΔR |
B (SE) | t | p | 95% CI | ||
| Age | 0.01 | 0.01 (0.01) | 0.06 | 1.04 | 0.30 | [–0.01, 0.02] |
| Educational Years | 0.04 (0.02) | 0.10 | 1.74 | 0.08 | [–0.01, 0.08] | |
| Nicotine Dependence (FTND) | 0.02 | 0.14 (0.06) | 0.14 | 2.49 | 0.01 | [0.03, 0.25] |
| rs266729 | 0.07 (0.11) | 0.04 | 0.66 | 0.51 | [–0.15, 0.29] | |
| Nicotine Dependence×rs266729 | 0.02 | –0.26 (0.11) | –0.19 | 2.31 | 0.02 | [–0.48, –0.04] |
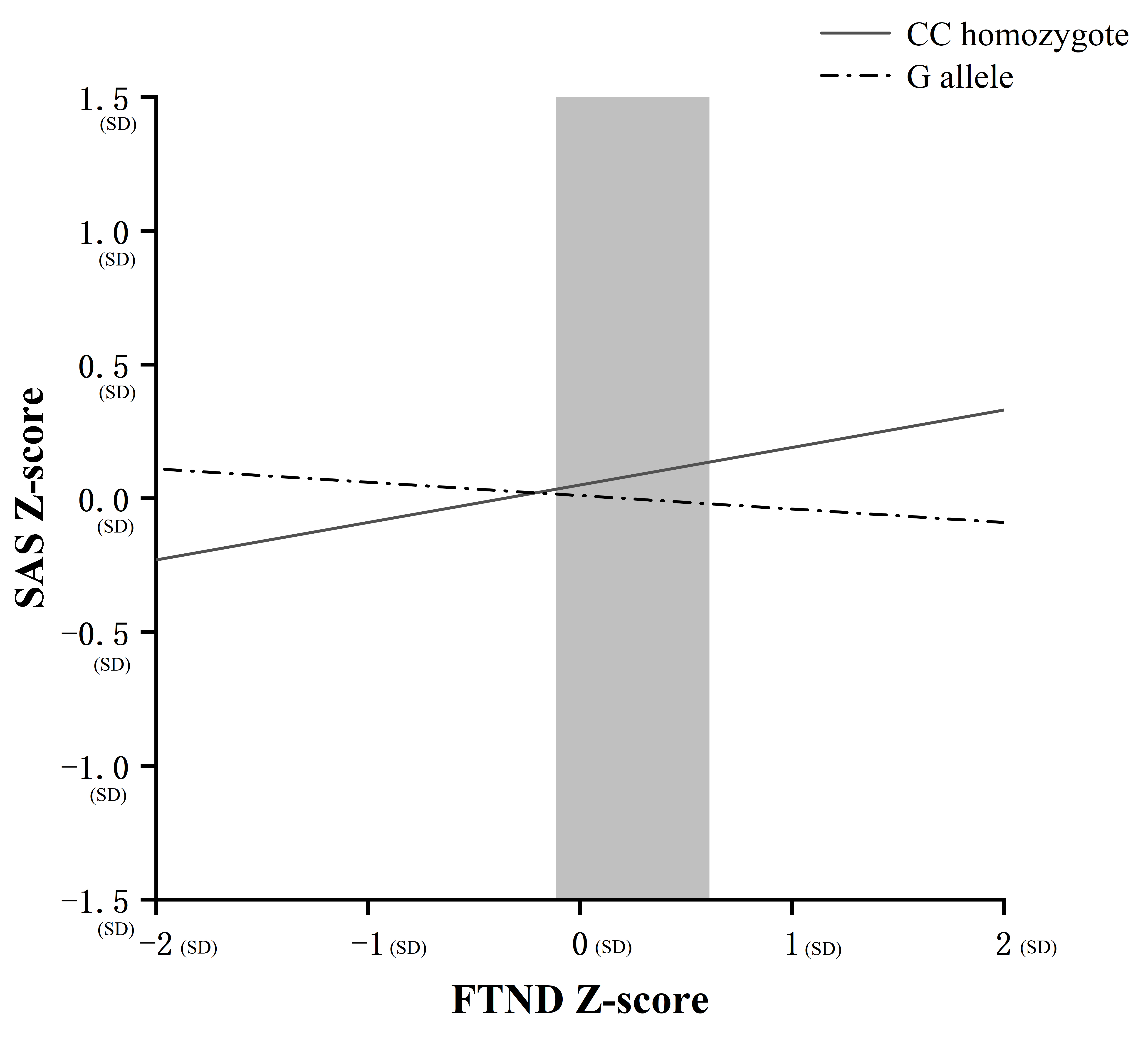 Fig. 1.
Fig. 1.Region of significance (RoS) test on anxiety from nicotine
dependence severity in rs266729 allelic groups. Note: Anxiety and nicotine
dependence: z scores (unit: standard deviations [SDs]); Simple slope at CC
homozygote: 0.14, t = 4.43, p
In addition, we re-analyzed the data to explain how alcohol use influences these
results. As shown in Supplementary Table 1, MAST scores were
positively correlated with SAS scores (r = 0.36, p
To improve the accuracy and robustness of the interaction results, a
re-parameterized regression model was fitted to examine the specific pattern of G
| Differential susceptibility | Diathesis-stress | |||
| Parameter | Strong: Model A | Weak: Model B | Strong: Model C | Weak: Model D |
| B0 | –0.61 (0.41) | –0.60 (0.42) | –0.55 (0.42) | –0.36 (0.43) |
| B1 | (–) | 0.02 (0.07) | (–) | 0.10 (0.06) |
| C | 0.27 (0.41) | 0.29 (0.45) | 1.93 (–) | 1.93 (–) |
| 95% CI of C | [–0.53, 1.07] | [–0.59, 1.17] | (–) | (–) |
| B2 | 0.28 |
0.28 |
0.12 |
0.19 |
| B3 | 0.01 (0.01) | 0.01 (0.01) | 0.01 (0.01) | 0.01 (0.01) |
| B4 | 0.03 (0.02) | 0.03 (0.02) | 0.04 (0.02) | 0.03 (0.02) |
| R² | 0.05 | 0.05 | 0.03 | 0.04 |
| F (df) | 3.85 |
3.86 |
3.15 |
3.07 |
| F vs.A (df) | (–) | 0.06 | 5.80 |
(–) |
| F vs.B (df) | 0.06 | (–) | 2.92 | 3.03 |
| AIC | 889.66 | 891.60 | 893.51 | 892.68 |
| BIC | 912.18 | 917.87 | 912.27 | 915.20 |
| Note: | ||||
Our study aimed to explore the interactive effect of ND and ADPN rs266729 on anxiety. First, our results show that ND positively correlates with anxiety, consistent with previous reports [9, 21, 37, 38]. Although previous studies remain mixed regarding the direction of the association between smoking and mental illness, more and more studies support the view that nicotine exposure is associated with the later onset of depression or anxiety [8, 39]. Second, our results revealed that ADPN rs266729 CC homozygous carriers were more likely to suffer from anxiety among ND patients than G allele carriers. In other words, the CC genotype is a high-risk allele or plasticity factor, while the G allele is a low-risk allele or non-plasticity factor. Further, the psychology model was confirmed to fit the robust differential susceptibility model.
ADPN is also associated with anxiety. Peripheral ADPN levels appear inversely associated with anxiety and other diseases and may be a promising biomarker for diagnosis and disease monitoring [8]. Serum ADPN levels throughout pregnancy were inversely associated with antenatal anxiety [40]. Evidence suggests that ADPN can cross the blood-brain barrier and act on specific neuronal populations through its receptors, AdipoR1, and AdipoR2. While AdipoR1 is widely expressed in the brain, AdipoR2 expression is restricted to a few brain regions, including the hippocampus and hypothalamus. ADPN decreases the activity of the Ventral tegmental area (VTA) dopamine neurons and induces anxiolytic responses through direct activation of AdipoR1 [15]. AdipoR1 and AdipoR2 are also closely associated with anxiety by modulating the hypothalamic-pituitary axis [41]. Nicotine is an agonist of the alpha-4 beta-2 nicotine receptor that induces dopamine secretion in the mesolimbic pathway and improves motivation; subjects with high anxiety tendencies are a potentially high-risk group susceptible to developing ND [42]. One review suggested that current smokers have reduced ADPN levels and that this reduction can be reversed by quitting smoking [12]. Another study suggested that tobacco use was significantly associated with a low ADPN level in community-dwelling young males [43]. The present study showed a significant interaction between ADPN rs266729 and ND. Compared to the G allele, ADPN rs266729 CC homozygous carriers were more likely to be affected by anxiety in ND patients. This result suggests that the GG genotype is a protective gene in anxiety induced by ND. These findings provide some guidance regarding preventing anxiety in people with ND. In the case of high-risk individuals (CC homozygous), we can intervene in advance to prevent exacerbations; for individuals with the G allele, we can enhance their ability to adjust themselves when facing adverse environments.
Re-parameterized regression models revealed that the interaction between ADPN rs266729 and ND fit the robust differential susceptibility model. In favorable environments, individuals with a putative high-risk allele show superior outcomes to individuals with the low-risk allele. In other words, individuals with “risk alleles” are sensitive to environmental conditions and may benefit from supportive environments; conversely, they may exhibit worse outcomes in unfavorable environments. The present study revealed that the robust differential susceptibility model was the best fit. This finding suggests that ADPN rs266729 CC homozygote carriers were more likely to be affected by ND-induced anxiety and might be a protective factor against anxiety in non-smoking people. Individual genetic variation and the interactions between genes and external factors may characterize neural circuits and neurochemical functions, representing adaptable individuals’ psychological strength. Stress-related events can increase an individual’s susceptibility to severe psychiatric problems such as anxiety. Our findings might be explained by the fact that individuals with genetic variation respond differently to different degrees of ND. Given that genetic factors contribute to recovery, it is critical to identify candidate genetic variations to explain genetic patterns.
Many reports of increased anxiety levels and smoking rates during the COVID-19 pandemic. Higher anxiety correlated with higher ND among university students during the pandemic [44]. These findings suggest that intervention with anxiety symptoms can reduce ND, reducing harm during the pandemic. Mental health prevention and intervention programs may reduce the risk for ND in teen smokers with and without symptoms of depression and anxiety [45]. One study showed that dependent smoking was positively associated with current anxiety/mood disorders; greater clinical attention could be directed toward the role of anxiety in smoking cessation [37]. Social interaction anxiety can also affect nicotine dependence via negative metacognitions about smoking [46]. In the long run, the probability of recurrence of ND can be reduced by improving anxiety symptoms.
This study has some limitations which should be considered. First, we investigated only one polymorphism; therefore, it is likely that differences across smoking groups could be related to internalizing outcomes in many ways other than their genotype at this ADPN variant. Other polymorphisms in the ADPN gene or other genes are likely to be involved. Second, the study is cross-sectional; therefore, causality cannot be inferred. Third, the baseline values of participants’ anxiety symptoms were not measured before admission and the diagnosis of nicotine dependence, which greatly limited the intensity of our study. Fourth, we did not consider dietary habits, physical activity, socioeconomic status, location of residence, or type of family. Finally, our findings may not be generalizable to other ethnic groups.
There was an interaction between ADPN rs266729 and ND, suggesting that rs266729 might correlate with anxiety symptoms among male adults with ND. These findings support the differential susceptibility model, in which the CC homozygote of rs266729 is a plasticity factor rather than a factor that only increases anxiety symptoms of individuals during ND. These empirical findings have important implications for understanding the genetic moderation of ND and its effect on individual differences in anxiety symptoms. Further work is required to explore the underlying mechanisms of anxiety modulation at the molecular level and functional studies of neural systems in a larger sample.
The datasets presented in this article are not readily available because data use sharing agreements would be necessary. Requests to access the datasets should be directed to benjaminlyl@wmu.edu.cn.
Conceptualization—LC, YL and FWang; methodology—XZ, FWu, GS, SY, YH and YW; software—KX; validation—LZ; formal analysis—WW; writing - original draft preparation—XZ; writing - review and editing—XS and LC; funding acquisition—XZ and FWang. All authors have read and agreed to the published version of the manuscript.
The study was approved by the Ethics Committee of Peking University Health Science Center. Written informed consent has been obtained from the patients to publish this paper (Ethic approval number: YKD2015003).
We thank all the participants in our study for their time and cooperation.
This research was funded by Natural Science Foundation of Xinjiang Uyghur Autonomous Region (2018D01C239), Project of Health Department in Zhejiang Province (2019KY681) and Huzhou Municipal Science and Tech Commission (2018GY20).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/j.fbl2711309.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

