1 Department of Nursing Management, The First Affiliated Hospital of Soochow University, 215006 Suzhou, Jiangsu, China
2 Department of Gastroenterology, The First Affiliated Hospital of Soochow University, 215006 Suzhou, Jiangsu, China
Academic Editor: Azhar Ali
Abstract
Background: It has been reported that ubiquitin specific peptidase 4
(USP4) was functional in several tumors, but its function and mechanism in
gastric cancer were still unknown. Methods: Bioinformatic tools were
used to predict the prognosis of gastric cancer patients and the expression
levels of USP4 in gastric cancer. Quantitative real-time polymerase chain reaction (qRT-PCR) and immunoblotting were carried out to
detect the messenger RNA (mRNA) and protein levels. Cell viability of gastric cancer was
evaluated by Cell Counting Kit-8 (CCK-8) assay. Cell line-derived xenograft models were established to
evaluate the tumor growth of gastric cancer. Luciferase assay and immunoblotting
were used to determine the activation of nuclear factor kappa B (NF-
Keywords
- USP4
- gastric cancer
- PRL-3
- NF-κB
- PR-619
Gastric cancer is a common malignant tumor of digestive tract, which was reported to be the second leading cause of tumor-related mortality worldwide, especially in China [1, 2]. In 2018, there were 1,033,701 new cases and 782,685 deaths of gastric cancer worldwide [3, 4]. Gastric cancer has been proven to be a complex disease, which was reported to be related to many factors, including environmental exposure and genetic factors [1]. Despite the continuous development of medical strategies, the treatment of gastric cancer is still mainly limited to surgery, radiotherapy and chemotherapy [5]. Therefore, it is particularly important to find novel targets for the diagnosis and treatment of gastric cancer.
The ubiquitin-proteasome system (UPS) is an important regulator in cells, and dysregulation of UPS is closely related to the occurrence and development of diseases, including tumors [6]. The UPS is composed of at least 6 components, including ubiquitin, ubiquitin-activating enzyme, ubiquitin-conjugating enzyme, ubiquitin ligase, deubiquitinating enzyme (DUB) and 26S proteasome [7]. Among these, DUBs are widely expressed in cells, which are closely related to the modulation of tumorigenesis by removing ubiquitin chains from macromolecules involved in cancer regulation [8]. Thus, targeting DUBs has been considered as a potential strategy for anti-tumor drug discovery.
Nuclear factor kappa B (NF-
It has been also reported that phosphatase of regenerating liver-3 (PRL-3) was an upstream molecule of NF-
To predict the overall survival (OS) and post-progression survival (PPS) for gastric cancer patients with low or high USP4 (Affymetrix ID: 202681_at, 211800_s_at), the online database Kaplan-Meier Plotter was used (http://kmplot.com/) [18]. The expressions of USP4 in STAD based on individual cancer stages, tumor grade, H.pylori infection status or nodal metastasis status were analyzed by UALCAN online tool (http://ualcan.path.uab.edu/) [19, 20]. The correlation analyses between USP4 and cyclin D2 (CCND2), CCND3, B cell leukemia/lymphoma 2 (BCL2) or XIAP in stomach cancer were analyzed by GEPIA 2 database online (http://gepia2.cancer-pku.cn/).
Gastric cancer cell lines MKN-45 and SNU-1 were obtained from Cell Resource Center, Institute of Basic Medicine, Chinese Academy of Medical Sciences. HEK293T cell line was purchased from American Type Culture Collection, Manassas, VA, USA. All cell lines were cultured in RPMI 1640 medium (Meilunbio, Dalian, China) with 10% FBS (Biochannel, Nanjing, China) and 1% penicillin/streptomycin (Beyotime, Beijing, China). The chemical PR-619 (Cat. No. HY-13814) was purchased from MedChemExpress, New Jersey, USA. The primary gastric cancer tissues were received from the First Affiliated Hospital of Soochow University, and the case information was listed in the Supplementary Table 1. The collection and use of human gastric cancer tissues for this study were approved by the Review and Ethics committee of the First Affiliated Hospital of Soochow University.
The qRT-PCR was carried out to evaluate the messenger RNA (mRNA) levels of USP4, CCND2, BCL2 and GAPDH according to the previous study [21]. The primers used were as follows: USP4, forward 5′-AAGGAAGCCTGGGAGAAT-3′ and reverse 5′-GCAGTGGCAGCGTTAGAT-3′; CCND2, forward 5′-CTGTCTCTGATCCGCAAGCAT-3′ and reverse 5′-CCCACACTTCCAGTTGCGAT-3′; BCL2, forward 5′-GAACTGGGGGAGGATTGTGG-3′ and reverse 5′-CATCCCAGCCTCCGTTATCC-3′; GAPDH, forward 5′-GCACCGTCAAGGCTGAGAAC-3′ and reverse 5′-TGGTGAAGACGCCAGTGGA-3′.
Immunoblotting was carried out to evaluate the protein levels as described
previously [22]. The anti-USP4 (Cat. No. sc-376000) and anti-PRL-3 (Cat. No.
sc-130355) antibodies were purchased from Santa Cruz Biotechnology, Santa Cruz, CA, USA.
Anti-phospho-NF-
Gastric cancer cells were also lysed for immunoprecipitation (IP) analysis. In brief, cells were lysed by using IP lysis (Beyotime, Shanghai, China, Cat. No. P0013J). Then, PRL-3 protein was purified by using an anti-PRL-3 antibody (Santa Cruz, Cat. No. sc-130355) and Protein A/G PLUS-Agarose (Santa Cruz, Cat. No. sc-2003). At last, the interacting protein USP4 was detected by using immunoblotting with an anti-USP4 antibody (Santa Cruz, Cat. No. sc-376000).
The cell viability of gastric cancer cells was detected by Cell Counting Kit-8 (CCK-8) assay referenced to the manufacturer’s instructions (Cat. No. B34302, Bimake, Houston, TX, USA).
Lentivirus-delivered shRNAs against USP4 (shUSP4) and a negative control shNC were synthesized from GeneChem Co., Ltd. (Shanghai, China). The target sequences of shUSP4#1 and shUSP4#2 were 5′-GGTCGCAGATGTGTATAAT-3′ and 5′-GCAGCCACTATTGCTTTCT-3′. Lentiviral particles were generated in HEK293T cells, and gastric cancer cells were infected by the lentivirus according to the previous study [23].
The luciferase reporter pNF
SNU-1 and MKN-45 cells infected with shNC or shUSP4#1-derived lentivirus (3
SNU-1 and MKN-45 cells were transfected with luciferase reporter
pNF
In the experiments, to compare the differences between two groups, student’s t test was used. One-way ANOVA with Tukey’s Multiple Comparison Test was used to compare the differences among multiple groups. In this study, a p value less than 0.05 was considered to be statistically significant.
In order to examine the relationship between USP4 and clinical prognosis of gastric cancer patients, we tried Kaplan-Meier Plotter database for analysis. As shown in Fig. 1A,B, the Kaplan-Meier Plotter with Affymetrix microarray (ID: 202681_at) showed that the gastric cancer patients with high expression level of USP4 had the shorter overall survival (OS) and post-progression survival (PPS) than the patients with low expression. In addition, the Kaplan-Meier Plotter with Affymetrix microarray (ID: 211800_s_at) also showed that the gastric cancer patients with high expression level of USP4 had the shorter OS and PPS (Fig. 1C,D).
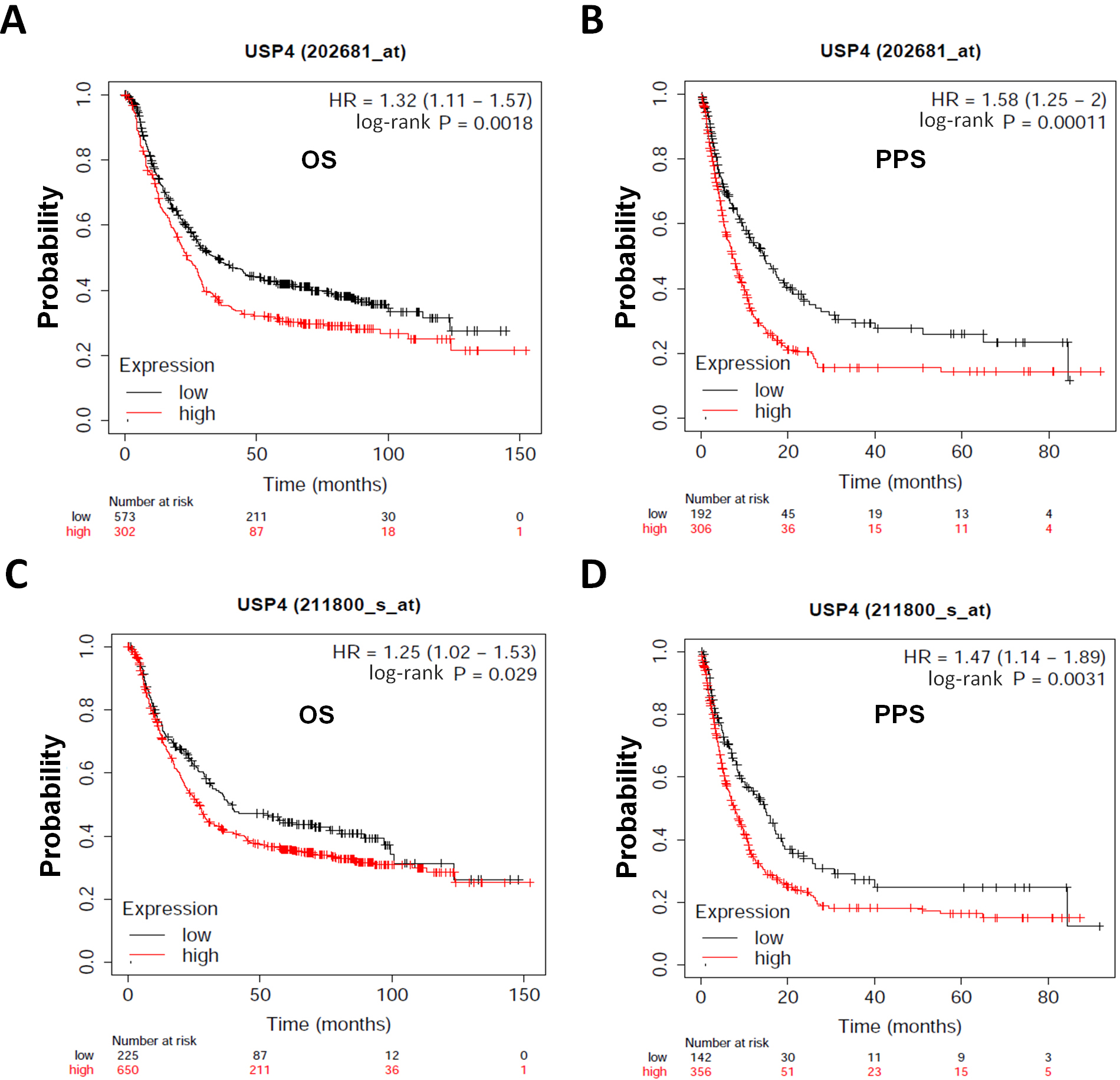 Fig. 1.
Fig. 1.USP4 predicts a negative index for gastric cancer patients. (A,B) The overall survival (OS) and post-progression survival (PPS) for gastric cancer patients with low or high USP4 (Affymetrix ID: 202681_at) were analyzed by Kaplan-Meier Plotter database online. (C,D) The OS and PPS for gastric cancer patients with low or high USP4 (Affymetrix ID: 211800_s_at) were analyzed by Kaplan-Meier Plotter.
Above results showed that USP4 predicted a poor prognosis for gastric cancer patients, which suggested that USP4 may have therapeutic significance for gastric cancer in clinic. Then, to further investigate the expression levels of USP4 in gastric cancer based on individual cancer stages, tumor grade, H.pylori infection status or nodal metastasis status, the public database UALCAN was used online. As shown in Fig. 2A, USP4 was significantly upregulated in patients with Stage 4 gastric cancer. USP4 was also significantly increased in patients with Grade 3 gastric cancer (Fig. 2B). It was also shown that gastric cancer patients without H.pylori infection had a higher expression of USP4 (Fig. 2C). In addition, the expression level of USP4 in gastric cancer was also related with the nodal metastasis status (Fig. 2D).
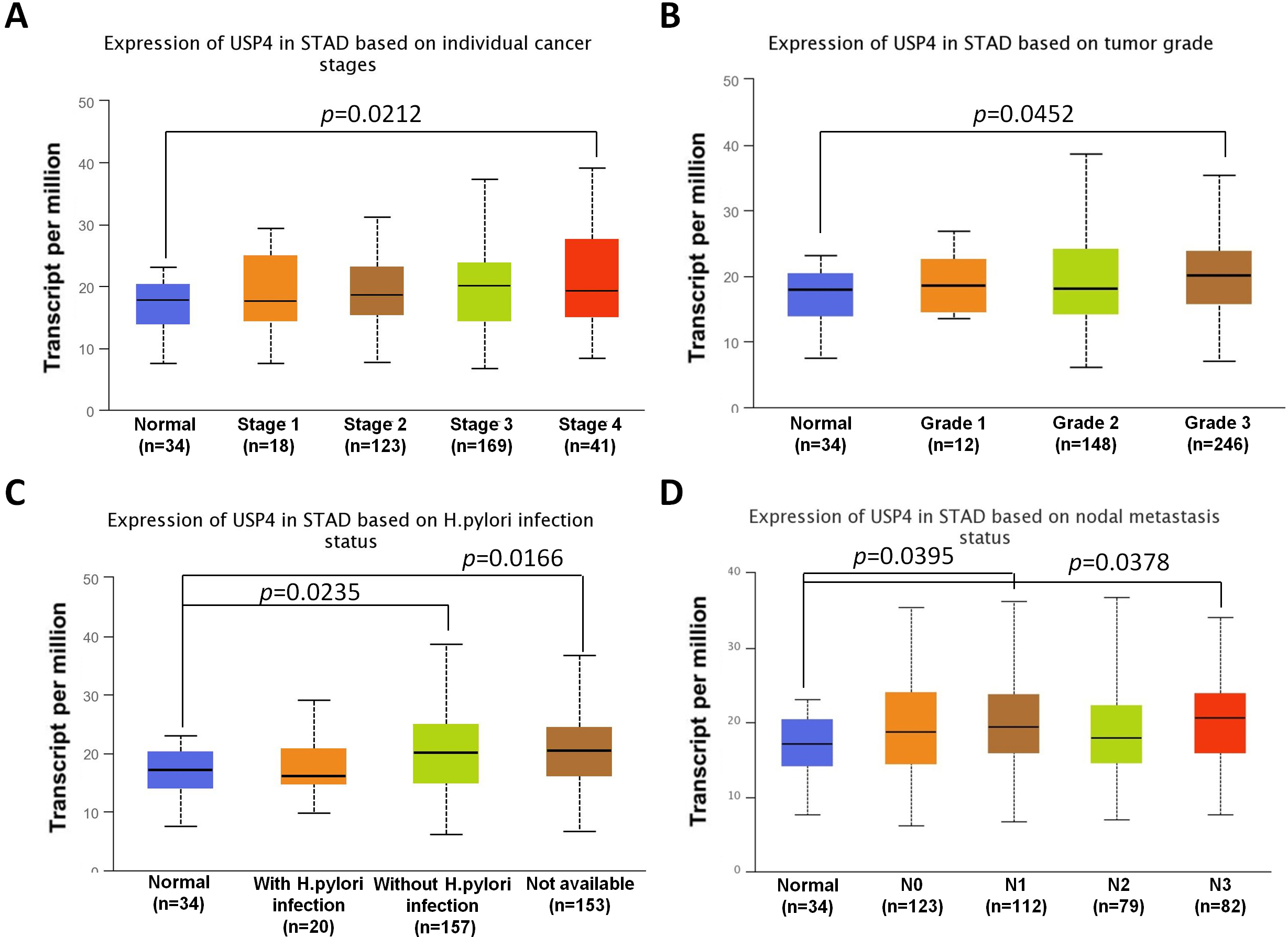 Fig. 2.
Fig. 2.The expression of USP4 in gastric cancer patients. (A–D) The expressions of USP4 in STAD based on individual cancer stages (A), tumor grade (B), H.pylori infection status (C) or nodal metastasis status (D) were analyzed by UALCAN database online.
Then, several gastric cancer tumor tissues were collected for qRT-PCR analysis, and it showed that USP4 was increased in tumor tissues compared with the adjacent normal tissues (Fig. 3A). To further investigate whether USP4 was functional in gastric cancer, USP4 was knocked down by shRNAs. As shown in Fig. 3B,C, the qRT-PCR analysis showed that USP4 was successfully knocked down in both of SNU-1 and MKN-45 cells. Then, infected cells were prepared for CCK-8 assay to investigate whether USP4 knockdown affected cell growth of gastric cancer. As shown in Fig. 3D,E, knockdown of USP4 significantly suppressed cell growth in both of gastric cancer cell line SNU-1 and MKN-45 cells. These results further confirmed that USP4 was functional in gastric cancer.
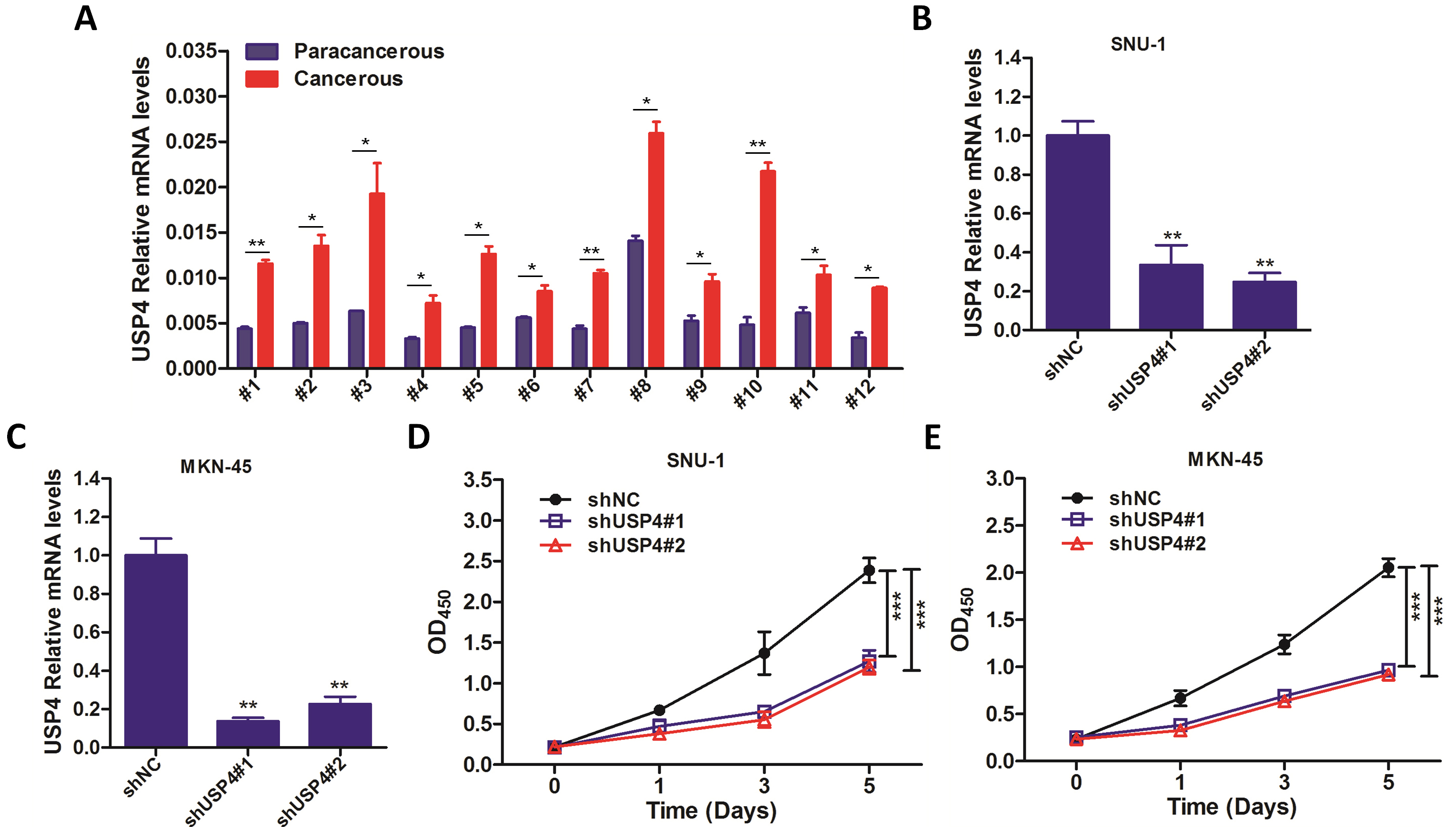 Fig. 3.
Fig. 3.Downregulation of USP4 suppresses cell growth of gastric cancer.
(A) Twelve pairs of gastric cancer tumor tissues (cancerous)
and adjacent normal tissues (paracancerous) were prepared for qRT-PCR against
USP4. GAPDH was used as an internal control. (B,C) SNU-1 (B)
and MKN-45 (C) cells were infected with shNC, shUSP4#1 or shUSP4#2 lentivirus
for 3 days, followed by qRT-PCR against USP4. (D,E) SNU-1 (D)
and MKN-45 (E) cells were infected with shNC, shUSP4#1 or shUSP4#2 lentivirus
for indicated time, followed by CCK-8 assay at day 0, 1, 3 or 5.
Moreover, we also establised the xenograft models to evaluate the function of USP4 in vivo. The animal studies showed that knockdown of USP4 markedly suppressed the tumor growth of gastric cancer (Fig. 4). These results further confirmed that USP4 was functional in gastric cancer.
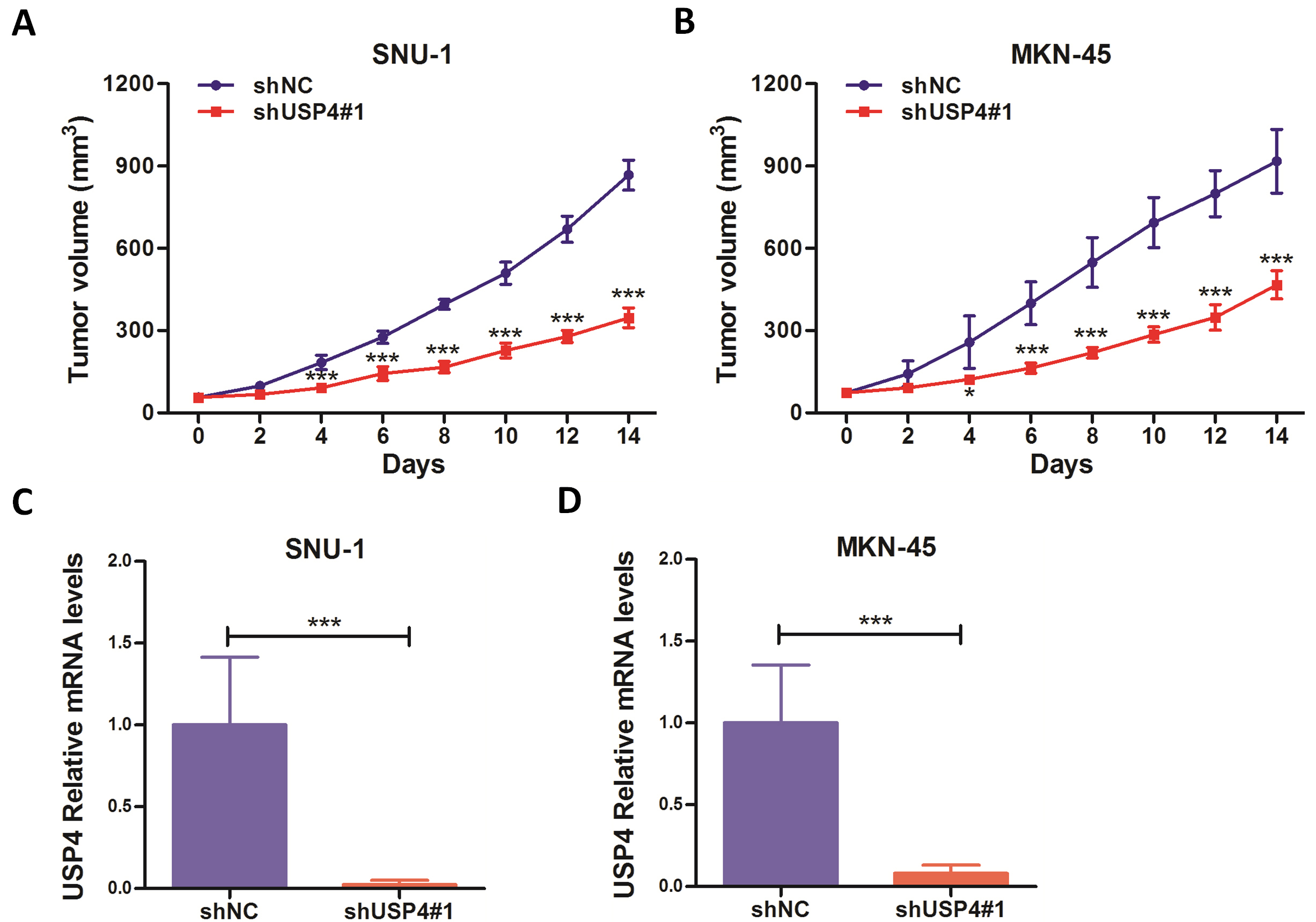 Fig. 4.
Fig. 4.Downregulation of USP4 suppresses tumor growth in gastric cancer
cell line-derived xenografts. (A,B) The tumor volume curves.
(C,D) The excised tumors were prepared for qRT-PCR analysis
against USP4. GAPDH was used as an internal control.
Next, our further study showed that knockdown of USP4 decreased the expression
of PRL-3 (Fig. 5A), which was previously reported to be a substrate of USP4 [13].
It has been also reported that PRL-3 was an upstream molecule of NF-
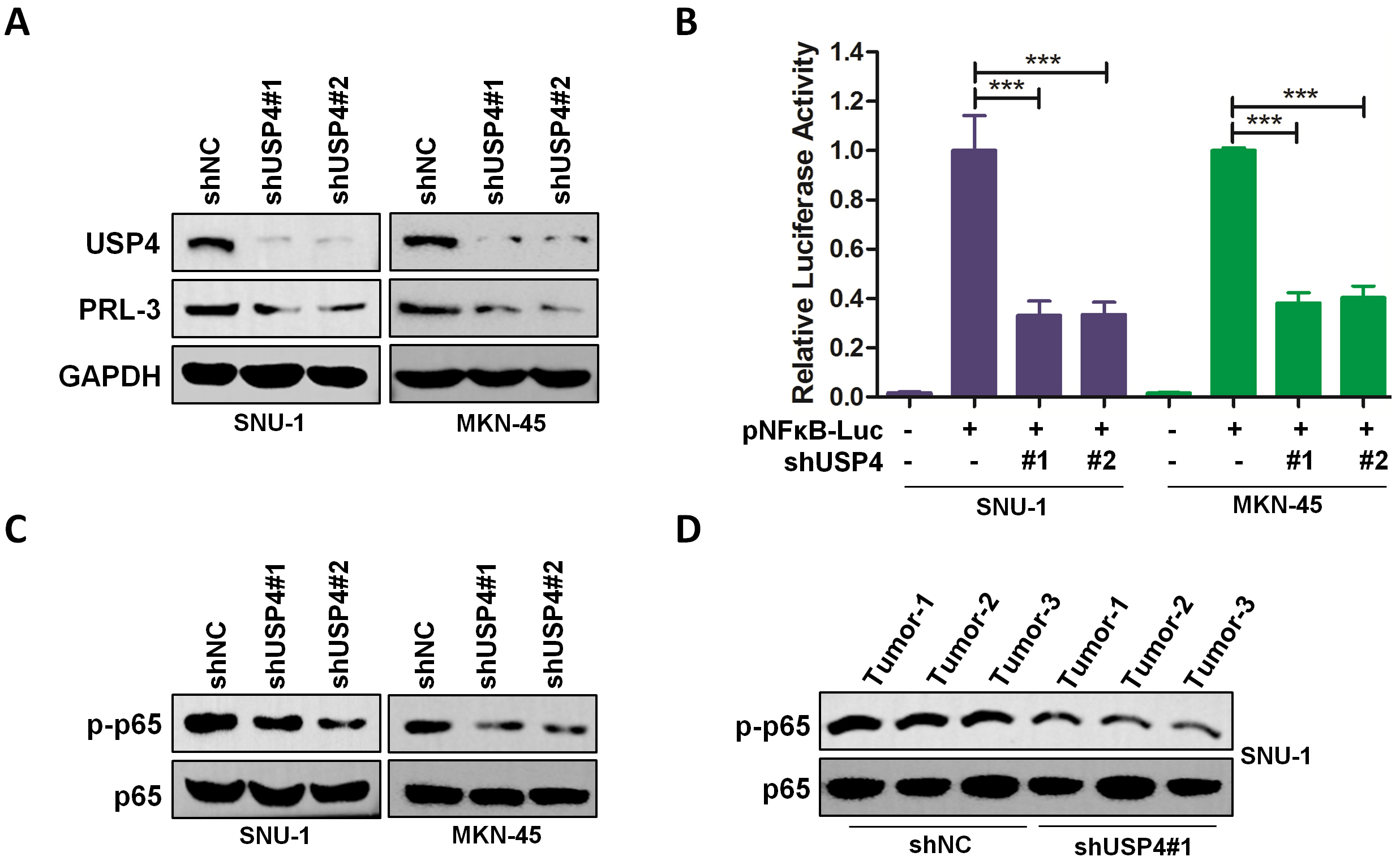 Fig. 5.
Fig. 5.Downregulation of USP4 inhibits PRL-3 expression and
NF-
In addition, our findings revealed that USP4 expression was positively
correlated with the target genes of NF-
In order to further confirm whether PRL-3 was a target for USP4 in gastric
cancer, co-immunoprecipitation assay was firstly performed (Fig. 6A). In
addition, PRL-3 was overexpressed by plasmid transfection (Fig. 6B). As shown in
Fig. 6C,D, the luciferase assay showed that overexpression of PRL-3 could
significantly attenuate the inhibitory effects of USP4 knockdown on
NF-
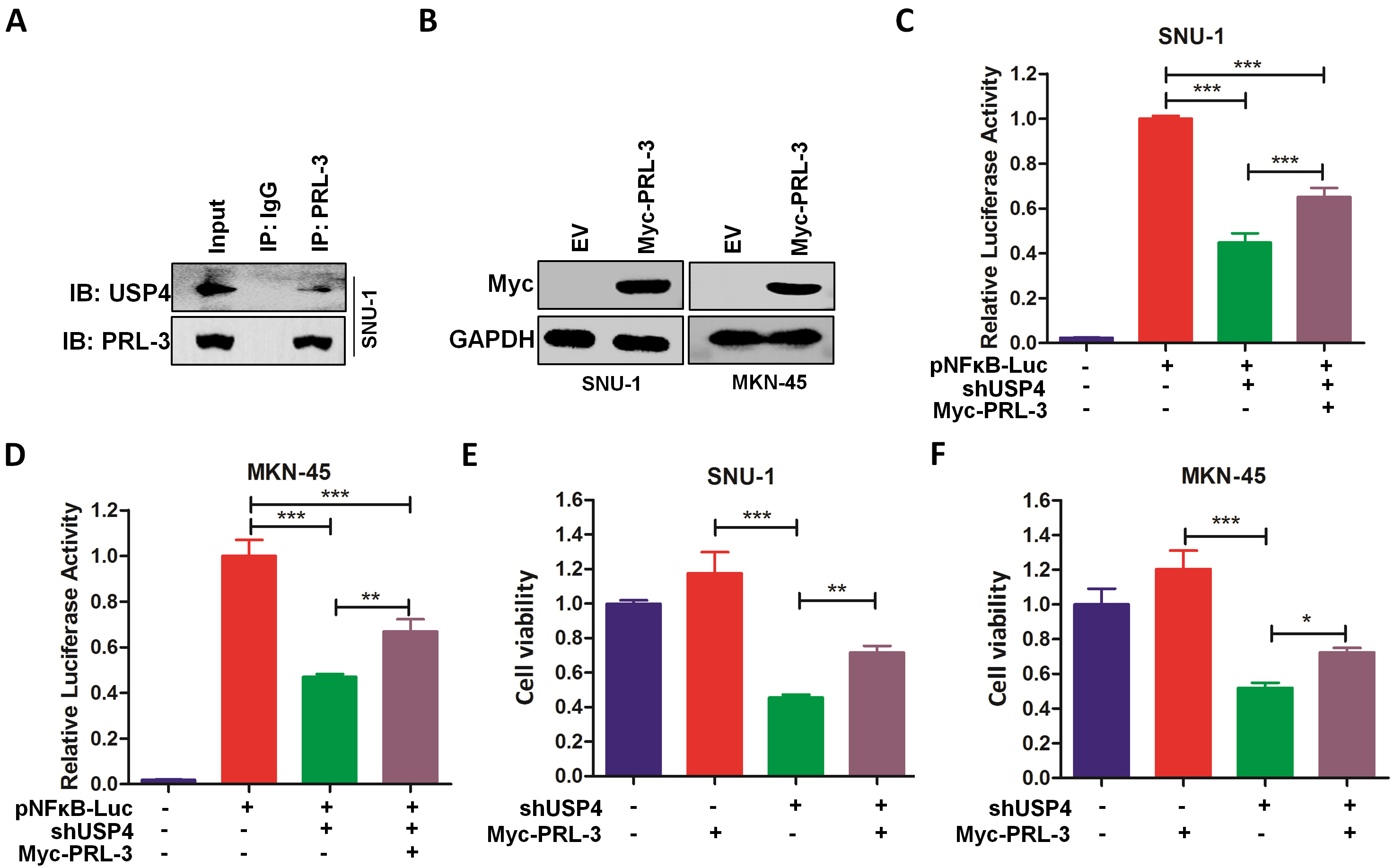 Fig. 6.
Fig. 6.Overexpression of PRL-3 attenuates the inhibitory effects of
USP4 knockdown in gastric cancer cells. (A) SNU-1 cells were
lysed for co-immunoprecipitation. IP, immunoprecipitation; IB, immunoblotting.
(B) The overexpressing efficacy of Myc-PRL-3 plasmids was
detected by immunoblotting in SNU-1 and MKN-45 cells. (C,D)
SNU-1 (C) and MKN-45 (D) cells transfected with pNF
It has been proven that PR-619, as a broad-range deubiquitinase inhibitor,
inhibited the activities of USP4 and other deubiquitinases, and exerted its
anti-tumor activity in several tumors [24, 25]. Based on these, we finally
evaluated whether the cell viability of gastric cancer could be inhibited by
PR-619. As shown in Fig. 7A,B, the cell viability of gastric cancer cells was
significantly inhibited by PR-619 treatment. In addition, the luciferase activity
driven by NF-
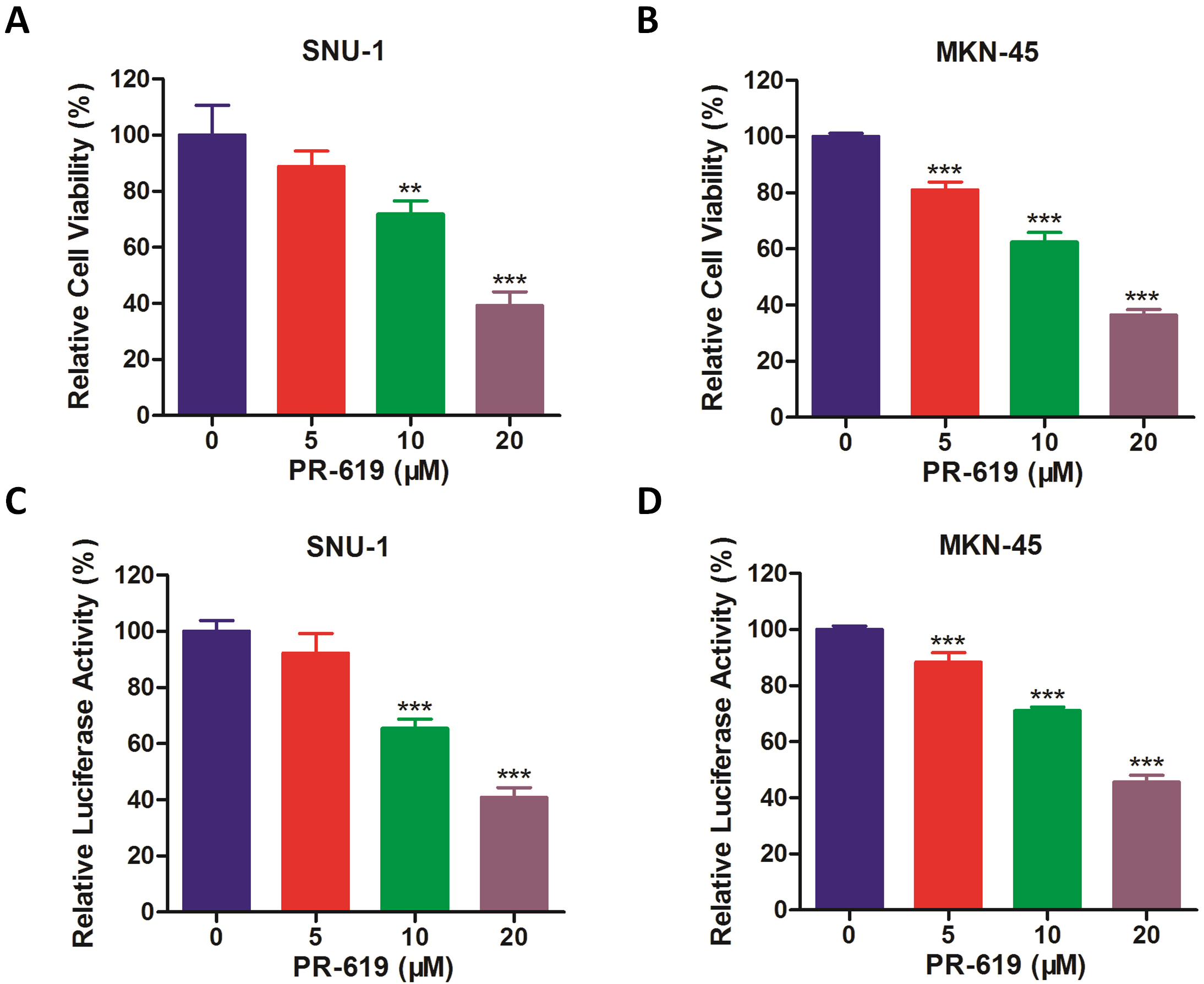 Fig. 7.
Fig. 7.Deubiquitinase inhibition by PR-619 inhibits cell viability of
gastric cancer and NF-
It has been reported that USP4 was functional in several tumors mostly as an oncogene. For example, in glioblastoma multiforme (GBM), USP4 was found markedly upregulated in GBM tissues and patients with high USP4 had a poor prognosis [26]. Moreover, knockdown of USP4 significantly suppressed cell growth of GBM by inhibiting ERK signaling both in vitro and in vivo [26]. Additionally, in glioblastoma, USP4 could induce temozolomide (TMZ) chemoresistance by suppressing cell apoptosis in a p53-dependent manner [27]. USP4 was also reported elevated in both of melanoma tumor tissues and cell lines, and knockdown of USP4 could increase the sensitivity to cisplatin as well as attenuating the migratory and invasive abilities of melanoma cells by suppressing epithelial-mesenchymal transition (EMT), which suggested that USP4 displayed an oncogenic role in melanoma [17]. However, the function of USP4 has not been investigated in gastric cancer. In our present study, USP4 was also found upregulated in gastric cancer tumor tissues, and knockdown of USP4 significantly inhibited cell growth and tumor growth of gastric cancer, which indicated that USP4 was a novel oncogene in gastric cancer.
At present, targeting DUBs, especially ubiquitin-specific proteases, for the development of anticancer drugs has become an effective strategy. Take USP4 as an example, neutral red (NR) was identified as an uncompetitive inhibitor of USP4 through the in vitro deubiquitinating activity assays, and it showed that NR markedly reduced cell migration and colony formation of colorectal cancer cells as well as inhibiting tumor growth of colorectal cancer in a mouse xenograft model [28]. Thus, this study further supported the possibility of developing USP4 inhibitors as therapeutic agents in gastric cancer, and our future work will focus on this aspect.
So far, many studies have proven that NF-
Collectively, our present study indicated that targeting
USP4/PRL-3/NF-
All data generated or analyzed during this study are included in this article.
WDY participated in the conception and design of the study. YYT and WDY performed the experiments. WDY interpreted the data and produced the main document. All authors read and approved the final manuscript.
This study was approved by the Review and Ethics committee of the First Affiliated Hospital of Soochow University (Ethic Approval Number: 2019, 127).
Not applicable.
This study was supported by the National Natural Science Foundation of China (No.81902399).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.







