Frontiers in Bioscience-Landmark (FBL) is published by IMR Press from Volume 26 Issue 5 (2021). Previous articles were published by another publisher on a subscription basis, and they are hosted by IMR Press on imrpress.com as a courtesy and upon agreement with Frontiers in Bioscience.
1 Department of Gastroenterology, Xiangya Hospital, Central South University, Xiangya Rd, Changsha, Hunan 410008, P. R. China
Abstract
The ubiquitously expressed multifunctional protein, CUEDC2 (CUE domain-containing 2), is involved in many physiological and pathological processes, including the cell cycle regulation and inflammation. Although it is known that CUEDC2 is expressed disparately in breast cancer, ovarian carcinoma, hepatocellular carcinoma, cholangiocarcinoma, glioma, lung adenocarcinoma, colon cancers, and is involved in the Warburg’s effect, its role in oncogenesis remains to be further explored. In this review, we examine the expression of CUEDC2 in various tumors, and discuss several fundamental signaling pathways that are impacted by CUEDC2.
Keywords
- CUEDC2
- Tumorigeneses
- Cell cycle
- Inflammation
- Tumor progression
- Prognosis
- Review
CUEDC2, a novel CUE domain-containing protein, has been reported to contain a small and somewhat conserved ubiquitin-binding motif comprised of approximately 40 amino acids (1). The CUE domain has been observed in multiple eukaryotic proteins and is involved in the recognition of mono-ubiquitin and poly-ubiquitin as well promoting intramolecular mono-ubiquitination. CUEDC2 was originally implicated in the regulation of progesterone receptor (PR) degradation in breast cancer via the ubiquitin-proteasome pathway (2). The current evidence suggests that CUEDC2 plays a pivotal role in many cellular events (as shown in Figure 1), such as regulation of the cell cycle, inflammation, and carcinogenesis. Man et al. have reported that CUEDC2 is widely expressed in tissues and organs, including the brain, heart and testis (3). Intriguingly, in contrast to the high levels of expression of CUEDC2 in different cancer cells, such as ovarian serous carcinoma (1) and cholangiocarcinoma cells (4), which are similar to those of H19, one of the first reported long non-coding RNAs (5) (6), glioma (7), lung adenocarcinoma (8) and infiltrating macrophages in colon cancer (9) have noticeably decreased levels expression, suggesting that it plays a significant role in tumorigenesis.
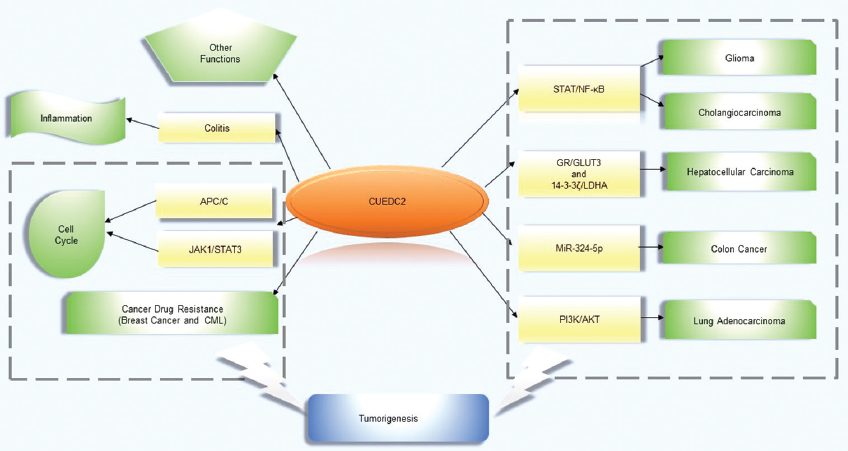 Figure 1
Figure 1Furthermore, despite our understanding of the role of CUEDC2 still being considered to be a ‘work in progress’, there have been several varying reports concerning CUEDC2-associated tumorigenesis pathways, including the NF-κB pathway (10), the Janus kinase (JAK)/signal transducer and activator of transcription(STAT) pathway (11) and the miR-324-5p axis (9). Last, but not least, some other novel functions of CUEDC2 are also described in this article. In the following sections, we would like to describe the expression pattern of CUEDC2 in different cancers, the role of CUEDC2, the regulation of CUEDC2, the molecular mechanisms of CUEDC2 and recent advances in CUEDC2.
The expression of CUEDC2 in tumorigenesis has been well studied. An increasing number of studeies have suggested that CUEDC2 exerts a crucial role in different cancer types with distinct expression patterns. A summary of these studies is provided below.
Pan et al. and Zeng et al. have shown that CUEDC2 expression levels are higher in breast cancer tissues than in normal breast tissues (12) (13). In addition, Zhang et al. discovered that CUEDC2 binds to PR in breast cancer, subsequently inhibiting its transcription and blocking progesterone signaling. This binding also showed correlation with cell proliferation and breast cancer progression. Zhang et al. also showed that the CUE domain of CUEDC2 (180–226 aa) may be important for the efficient repression of PR transcriptional activity, whereas the 133–180 aa region may be sufficient for association with PR. It has also been found that CUEDC2 blocks PR transactivation and prevents progesterone-induced cell proliferation by decreasing MAPK activity and, consequently impeding the effect of progesterone on breast cancer cell growth (2).
An important finding is that endocrine resistance (ER) critically interferes with the success of hormonal therapy for breast carcinoma (14). Pan et al. have shown that CUEDC2 decreases the stability of the ER-α (estrogen receptor-α) protein via the ubiquitin-proteasome pathway (12) (15), which strongly suggest that CUEDC2 plays a crucial role in the resistance of breast cancer to endocrine therapies. In addition, it has been reported that the overexpression of CUEDC2 in breast tumors may result in a poor response to tamoxifen treatment and may be responsible for the relatively high rate of relapse (12).
Ovarian carcinoma has long been recognized as the most fatal gynecological malignancy, causing approximately 140,000 deaths annually worldwide (16) (17). The factors in the early stages of ovarian serous carcinoma that contribute to this high mortality rate are not fully understood, and there is a lack of dependable biomarkers to assess its prognosis. Wang et al. preformed an immunohistochemical study on 101 cases of ovarian serous carcinoma tissues and found that CUEDC2 overexpression was observed in 60 cases (about 59.4%). Furthermore, the upregulation of CUEDC2 in ovarian serous carcinoma tissue samples is significantly associated with histopathological grade, patient age at diagnosis, FIGO stage, and recurrence. A survival follow-up study from the same research group evaluating its clinical relevance showed that the upregulated CUEDC2 expression levels are associated with shorter disease-free survival; the median disease-free survival time of CUEDC2-positive patients was 36.0 months compared with 53.9 months for CUEDC2-negative patients (1). Taken together, this study concluded that CUEDC2 may be a novel biomarker that can be used to evaluate the progression of serous ovarian carcinoma and to predict potential relapse.
Hepatocellular carcinoma (HCC) is one of the most common cancers of the digestive system, ranking fifth among malignant tumors and third in cancer-related deaths worldwide (18). Factors impacting the high mortality rate of HCC are that there is an incomplete understanding of the molecular biology of the disease and the issue of early diagnosis. Zhong et al. found that there are upregulated aerobic glycolysis in HCC cancer cells, which may be assciated with tumor cells progression. And this upregulation results from activating two glycolytic proteins glucose transporter 3(GLUT3) and lactate dehydrogenase A(LDHA), which might be mediated by glucocorticoid receptor(GR) or 14-3-3ζ respectively. In 14 pairs of HCC and adjacent normal tissues, CUEDC2, together with the GR/GLUT3, and 14-3-3ζ/LDHA, which are all critical for CUEDC2-regulated tumor progression, were more highly expressed in HCC tissues than in normal tissues. Moreover, through a statistical analysis of 213 HCC samples, this reaseach group discovered that CUEDC2 expression is positively associated with that of GLUT3 (64%) and LDHA (68%). These data suggest the likelihood that CUEDC2 expression might be highly correlated with the expression of GR/GLUT3 and 14-3-3ζ/LDHA in HCC cells (19). However, the exact role of CUEDC2 in HCC formation and progression requires further study.
Wang et al. discovered that HCC tumors are detected greater than 80% CUEDC2-positive. In addition, CUEDC2 expression is related to serum alpha-fetoprotein (AFP) levels, tumor size, tumor number, tumor differentiation, and TNM stage. HCC patients with high levels of CUEDC2 expression have a shorter overall survival and recurrence-free survival than those with low CUEDC2 expression levels. Collectively, these data suggest that CUEDC2 could be a new valuable molecular marker for predicting prognosis in HCC patients (20).
Zhou showed that CUEDC2 is also highly expressed in cholangiocarcinoma. Its expression appears to be associated with tumor differentiation, invasion, and TNM stage. It was also shown that in different stages during the progression of cholangiocarcinoma, the expression levels of CUEDC2 increase, paralleling the elevation in the levels of inflammatory factors tumor necrosis factor (TNF)-α and STAT/NF-κB. More interestingly, it has been shown that CUEDC2 inhibits the conversion of inflammatory hyperplasia to atypical hyperplasia in cholangiocarcinoma (4).
Tumors in the central nervous system are considered to be some of the most complicated and high-risk malignancies. Recently, Li et al. found that CUEDC2 is expressed at relatively lower levels in the glioma tissue and cell lines than in normal tissue and astrocyte cells. This decrease in CUEDC2 levels in glioma cell lines has been shown to promote tumor cell migration, invasion, and proliferation. Further study by this research group has revealed that the decreased levels of CUEDC2 expression in glioma results in the activation of the STAT3 and NF-κB signaling pathways, which may be related to gliomagenesis and tumorigenic behavior (7).
Evidence provided by Sun et al. has shown that CUEDC2 expression is reduced at the mRNA and protein levels in both lung adenocarcinoma cell lines and tissues, and this reduction is associated with tumor T classification and tumor size. Intriguingly, Sun et al., using multivariate analysis, reported that the median survival time of lung adenocarcinoma patients with lower levels of CUEDC2 expression was 31 months compared with 61 months for patients with the same cancer, but with higher levels of CUEDC2 expression; this implies that CUEDC2 expression could be an independent indicator of the prognosis of patients with lung adenocarcinoma. CUEDC2 knockdown experiments using an short hairpin RNA (shRNA) revealed that it is involved in facilitating tumor growth and is associated with inactivation of the PI3K/Akt pathway, the induction of p21, and the reduction in cyclin D1 expression levels. Taken together, the decreased expression of CUEDC2 results in lung adenocarcinoma growth, contributing to poorer overall clinical survival (8).
Macrophages, which are differentiated from monocytes, play an indispensable role in host defense and inflammation (21). CUEDC2 is upregulated during the course of monocyte to macrophage differentiation (9). To explore whether CUEDC2 is associated with colon carcinogenesis, Chen et al. performed an immunohistochemistry analysis with 36 paired colon cancer samples. To their surprise, there turned out to be no difference in expression between colon cancer cells and adjacent nomal epithelial cells. However, the expression level of CUEDC2 in the macrophage infiltrating the tumors were notably lower than in normal adjacent tissues. Hence, these data suggest the innovative idea that the downregulation of CUEDC2 expression in macrophages might play an important role in the formation of colon cancer. However, this requires further investigation (9).
As the distinct expression patterns of CUEDC2 in cancer have recently been extensively explored, a vital role for CUEDC2 in the cell cycle and inflammation has emerged suggesting the CUEDC2 might be involved in tumorigenesis.
CUEDC2 has been identified as a novel cell cycle regulator, the deregulation of which could lead to various disorders, including tumor development. The anaphase-promoting complex or cyclosome (APC/C), a multisubunit E3 ubiquitin ligase, is considered to be a vital regulator of protein degradation in the cell cycle and is activated via binding to Cdc20 (a cell division cycle protein) or a Cdc20 homolog 1 (Cdh1). The spindle assembly checkpoint (SAC), which is activated by improperly attached kinetochores, plays a supervisory role to ensure accurate chromosome segregation and inhibit the activation of APC/CCdh1. It has been reported that excessive exposure to UV light, which disrupts genomic integrity, increases the probability of skin cancer progression (22). Specifically, when cells are overexposed to UV light, CUEDC2 undergoes ERK1/2-mediated phosphorylation and degradation. As a consequence, the decrease in CUEDC2 levels facilitates the activation of APC/CCdh1, which is essential for the DNA damage response (23, 24) and stimulates APC/CCdh1-induced cyclin A ubiquitination, subsequently leading to CDK2 inactivation and a block at G1. These data suggest that the degradation of CUEDC2 triggered by UV light exposure might contribute to genomic instability in the cell cycle, which is considered to be responsible for tumor initiation and promotion (22).
It is well known that persistent activation of the JAK1-STAT3 signaling pathway is important in tumourigenisis, incuding for cell cycle progression, apoptosis, and tumor cell invasion. Li et al. have reported that in the development of gliomas CUEDC2 downregulation facilitates the G1-S and S-G2 phase transitions in the cell cycle partially via JAK1-STAT3 signaling pathway (7).
IBDs (inflammatory bowel diseases), including ulcerative colitis and Crohn’s disease, are chronic inflammatory gastrointestinal disorders. Although the etiology of IBDs remains unknown, studies have shown that they might be associated with infections, immune disorders, and so on. The continuous relapse of IBDs could directly increase the possibility of developing colorectal cancer. CUEDC2 is known to modulate inflammation, which supports the hypothesis that CUEDC2 might regulate inflammatory diseases such as colitis. Wang et al. have found that the degree of colonic inflammation is more severe in CUEDC2 knockout (KO) mice than in wild type colitis mouse models with respect to body weight loss, increase in bowel movements, and bloody stools, which suggests CUEDC2 could alleviate colonic inflammation. The level of pro-inflammatory cytokines, such as interleukin (IL)-6 and TNF-α, continue to rise in the colon tissues of CUEDC2 KO mice at different stages of colonic inflammation, leading to more severe inflammation and, potentially acting via the NF-κB and STAT3 signaling pathways (25). Together, these data indicate that CUEDC2 potentially plays a role in protecting the colon from excessive inflammation, and could be a useful clinical marker for colitis, or even colitis-associated cancer.
A number of studies have further explored the mechanisms regulated by CUEDC2 with respect to its physiological and pathological functions. CUEDC2 has been suggested to have an important role in tumorigenesis by modulating several processes, such as mitosis, G1 arrest and the Warburg effect,
A dysregulation of mitosis may lead to apoptosis or rampant cell proliferation, eventually initiating cancer. It has been suggested that CUEDC2 may modulate the progression through mitosis. Gao et al. have demonstrated that during mitosis, CUEDC2 is phosphorylated by Cdk1, which is essential for the metaphase-anaphase transition, and then interacts with Cdc20 to stimulate the release of Mad2 from APC/C–Cdc20, subsequently leading to APC/C activation. Conversely, an earlier activation of APC/C has been attributed to the overexpression of CUEDC2, leading to chromosome missegregation and aneuploidy (26). As mentioned earlier, CUEDC2 is relatively highly expressed in many somatic tumors. Hence, we assume that the incorrect mitotic modulation of CUEDC2 might cause tumor progression through an induction of chromosomal instability.
To maintain genomic stability, cell cycle arrest, especially G1 arrest, is initiated in response to DNA damage and, provides adequate time for DNA repair and inhibition of the anomalous replication of damaged DNA. If the cell fails to induce cell cycle arrest, genomic instability and carcinogenesis can result. In the normal cell cycle, CUEDC2 promotes G1–S transition, for which CDK2 is a pivotal kinase. In addition, CUEDC2 may contribute to augmented CDK2 activity. During the G1–S transition, several factors have been shown to be of great significance in the activation of CDK2, such as cyclin A and Skp2, the expression levels of which increase in response to CUEDC2 overexpression. These data indicate that CUEDC2 could modulate CDK2 activity by regulating cyclin A and Skp2 levels during G1–S transition. Conversely, when cells are exposed to UV light, there is an impressive reduction in the cyclin A and Skp2 protein levels and a noticeable G1–S block, for which CUEDC2 downregulation is potentially required. This indicates that CUEDC2 regulates UV-induced G1 arrest by decreasing cyclin A levels and inactivating CDK2 (21) (22).
The Warburg effect, or aerobic glycolysis, h as been shown to be enhanced in a variety of cancer cells, which results in alterations in metabolism, and is considered to be an emerging hallmark of oncogenic lesions. An analysis performed by Zhong et al. has shown that CUEDC2 is involved in the switch from glucose metabolism to glytic metabolism. The Warburg effect is upregulated in cancer cells through the activation of two glycolytic proteins GLUT3 and LDHA. The elevated Warburg effect in turn facilitates cancer progression. And CUEDC2 could regulate aerobic glycolysis in cancer cell survival and proliferation(19). Apart from that, regarding the upward-trend researches of metabolism, CUEDC2 might interact with phosphoprotein phosphatase 1-interacting proteins to act as an potential therapeutic target in prostate cancer. In addition, as the androgen receptor (AR) plays an important role in the hormonal progression of prostate cancer, CUEDC2 may promote AR degradation by regulating its transcriptional activity (27). Collectively, these data suggest it is likely that CUEDC2 act as a bridge between cellular metabolism and tumorigenesis.
Although a full understanding of its molecular mechanism remains incomplete, evidence to date suggests that CUEDC2 contributes to a range of signaling pathways, such as the NF-κB pathway, the JAK/STAT pathway, the miR-324-5p axis, as well as others.
The NF-κB transcription factor is a vital regulator of various cytokine-mediated cellular responses, such as cell proliferation and inflammation (28). In most cases, the NF-κB protein remains inactive via binding to the inhibitory IκB protein. Different stimuli, including TNF-α and IL-1, contribute to the activation of IKK by the IκB kinase (IKK) complex, which consists of IKKα, IKKβ, and IKKγ (also called NEMO), resulting in IκB phosphorylation and its proteosomal degradation. Following this, NF-κB translocates to the nucleus and activates the expression of NF-κB-regulated genes, such as the proinflammatory cytokine IL-6. However, CUEDC2, together with GADD34, a regulatory subunit of protein phosphatase (PP)-1, was found to constitutively interact with IKKα and IKKβ and inhibit NF-κB activation by decreasing IKK phosphorylation and activation (10). In addition, NF-κB can be activated through IKK-independent pathways, such as ultraviolet irradiation treatment, however, surprisingly, CUEDC2 expression does not inhibit the IκBα degradation in HeLa cells induced by ultraviolet irradiation, suggesting that CUEDC2 does not prevent NF-κB activation upon exposure to ultraviolet irradiation (10) (21). Thus, CUEDC2 might block only IKK-dependent NF-κB activation. Zhang et al. have also reported that CUEDC2 could sensitize CML (chronic myeloid leukemia) cells to imatinib therapy in part through the NF-κB signaling pathway (29). We hypothesize that CUEDC2 might affect inflammatory diseases and be a potential therapeutic target for such disorders or inflammation-associated tumors.
The JAK/STAT pathway, which is one of the most well recognized oncogenic signaling pathways, was initially regarded to be activated by IFNs (interferons), capable of impacting the formation of blood, cell proliferation, differentiation, and the immune response (30) (31). When any dysregulation in JAK/STAT activation occurs, a variety of diseases, including tumor development, could be initiated. The STAT protein family consists of six members, namely, STAT1–6. STAT3 is considered to be a DNA binding protein that is activated in response to several cytokines and growth factors, such as IFN and leptin. In normal cells, STAT3 activation is transient, which is similar to that of the other STAT family members, whereas the persistent activation of STAT3 might promote cellular transformation, tumor angiogenesis and block antitumor immune responses, further facilitating tumor development. Zhang et al. have suggested that CUEDC2 impedes IFN-induced STAT3 transcription activity in a dose-dependent manner, whereas CUEDC2 has no such effect on STAT1-mediated transcription, showing that CUEDC2 specifically inhibits STAT3 at the transcriptional level. In addition, CUEDC2 attenuates JAK1 and STAT3 phosphorylation by binding to suppressor of cytokine signaling-3 (SOCS-3), thereby preventing SOCS-3 proteosomal degradation and leading to the suppression of JAK1/STAT3 signaling (11).
Apart from this, the activation of STAT proteins, especially STAT5 and STAT3, is critical in the pathogenesis of lymphoid and myeloid malignancies (32-35). Recently, Wu et al. have reported that in acute myeloid leukemia (AML), downregulation of suppressor of cytokine signaling-1 (SOCS-1) counteracts the JAK1-STAT3 pathway activation. Through a mass spectrometry (MS) analysis of SOCS1, overexpressed CUEDC2 was identified to be a SOCS-1 interacting partner that might regulate the ubiquitin-mediated degradation of SOCS1 through its SH2 domain, thus inhibiting the JAK1-STAT3 signaling pathway and leukemogenesis in AML. However, further studies revealed that low CUEDC2 expression levels in AML results in SOCS1 degradation, an increase in JAK1-STAT3 pathway activation, and AML progression, hence accounting for lower overall survival and event-free survival rates (36).
Studies have suggested that microRNAs, which control cancer initiation, progression and biological processes, regulate gene expression during monocyte-macrophage differentiation (37). CUEDC2 was found to be upregulated during the differentiation from monocytes to macrophages. To determine whether the expression of CUEDC2 in these two types of cells is modulated by microRNAs, Chen et al. evaluated three microRNAs, termed miR-324-5p, miR-326, and miR-330-5p, with target sequences in the 3’ UTR of CUEDC2 mRNA. Although the three selected microRNAs showed relatively higher expression levels in monocytes than in macrophages, only miR-324-5p had a distinct inhibitory effect on CUEDC2 expression. MiR-324-5p is likely to regulate CUEDC2 expression at the translational level, and CUEDC2 deficient mouse models are more prone to colitis-associated cancer. Importantly, a further analysis of infiltrating macrophages in colon cancer tissue indicated that IL-4 upregulates miR-324-5p and downregulates CUEDC2 expression, suggesting that IL-4 in colon cancer cannot be ruled out as a contributor to the decreased CUEDC2 expression in macrophages via its ability to induce the miR-324-5p mimic (9). Ashish et al. reported their novel finding that miR-324-5p expression is down-regulated after cells are infected with the RNA virus H5N1. And miR-324-5p could inhibit H5N1 replication by specifically targeting a viral gene (PB1) and a host gene (CUEDC2) in the process of host defense(38).
Additionally, Song et al. have shown that sinomenine could decrease the levels of IL-4 and miR-324-5p (also called the IL-4/miR-324-5p/CUEDC2 axis) to elevate CUEDC2 expression, which prevents breast cancer cell invasion and migration by inhibiting NF-κB activation (39).
CUEDC2 is a versatile protein that can significantly regulate multiple precesses. However, recent advances have identified a role in modulating the activity of cardiomyocytes that have provided a more comprehensive understanding of the function of CUEDC2.
Jian et al. identified a potential role for CUEDC2 in the modulation of cardiomyocyte death. When ischemia occurs, CUEDC2 levels decrease in the heart. Knockout of CUEDC2 reduces reactive oxygen species (ROS) levels and upregulates glutathione peroxidase 1 (GPX1) protein levels, thereby enhancing the antioxidant capacity of cardiomyocytes. CUEDC2 knockdown not only decreases the generation of ischemia/reperfusion (I/R)-induced ROS to promote cardiomyocyte survival, but also enhances GPX1 protein levels through a ubiquitin-proteasome-dependent pathway to enhance the antioxidant ability of cardiomyocytes. In addition, high ROS levels can accelerate cardiomyocyte death and thus serve a central role in myocardial remodeling, heart failure, and aging-induced cardiomyopathy. Therefore, this provids an impetus to create a therapeutic method for reducing that decreases CUEDC2 levels in myocardial injury and aging-associated diseases (40).
Doxorubicin (DOX), originally used to treat solid tumors and some types of leukemia, is a broad-spectrum anthracycline antibiotic that can impede DNA replication (41) (42). However, there are many side effects of DOX, including vomiting, hair loss and heart injury, as well as others, that place restrictions on its clinical use (43). Among these side effects, cardiomyopathy, resulting from increased ROS generation, is the worst (44). As noted previously, CUEDC2 is extensively expressed in the heart. Zhang et al. have shown that the decreased expression of CUEDC2 prevents DOX-induced cardiotoxicity in H9c2 cells (myocardial phenotype). Thus, CUEDC2 may be a potential target for the prevention of DOX-induced cardiotoxicity (45).
In summary, the multifunctional protein CUEDC2 plays critical roles not only in the modulation of cell cycle and inflammation, but most interestingly, in the processes of tumor promotion. Similar to the function of Apollon (46) and eIF4E (eukaryotic translation initiation factor 4E) (47), CUEDC2 expression has different effects in a wide range of cancers. Specifically, CUEDC2 is overexpressed in HCC, ovarian serous carcinoma and cholangiocarcinoma but shows decreased expression in glioma, lung adenocarcinoma and macrophages infiltrated in colon cancer. The question remains as to what role CUEDC2 will play in other malignancies or systems, as a diagnostic agent or a therapeutic tool? Hence, we have provided a review of the reported research on CUEDC2 and a summary of the signaling mechanism for CUEDC2 in tumor progression, which will highlight it as a great potential target for cancer therapy.
This work was supported by Xiangya Hospital of Central South University. Jun-bo Xiao wrote the article; Guan-nan Ye, Yan-qiu Zhang, Zhen Wen, and Jian He helped with the preparation of the manuscript, firgures and references and the critical revision of the literature, respectively.
Abbreviations: CUEDC2 (CUE domain-containing 2); PR (progesterone receptor); JAK/STAT (the Janus kinase/signal transducer and activator of transcription); ER (endocrine resistance); ER-α (estrogen receptor-α); Hepatocellular carcinoma (HCC); AFP (alpha-fetoprotein); GR (glucocorticoid receptor); GLUT3 (glucose transporter 3); LDHA (lactate dehydrogenase A); shRNA (short hairpin RNA); TNF (tumor necrosis factor); (AR) androgen receptor; IBDs (inflammatory bowel diseases); APC/C (the anaphase-promoting complex or cyclosome); Cdc20 (cell division cycle protein); Cdh1 (Cdc20 homolog 1); SAC (the spindle assembly checkpoint); PP-1 (protein phosphatase-1); CML (chronic myeloid leukemia); IFNs (interferons); SOCS3 (suppressor of cytokine signaling-3); AML (acute myeloid leukemia); MS (mass spectrometry); TNF (tumor necrosis factor); ROS (reactive oxygen species); GPX1 (glutathione peroxidase 1); I/R (ischemia/reperfusion); DOX (Doxorubicin); eIF4E (eukaryotic translation initiation factor 4E)

