1 Beijing Huilongguan Hospital, Peking University Huilongguan Clinical Medical School, 100191 Beijing, China
2 Early Childhood Integrated Development Center, Beijing Fengtai Maternal and Child Health Care Hospital, 100069 Beijing, China
3 Department of Psychiatry, Beijing Jishuitan Hospital, 102200 Beijing, China
4 Department of Psychiatry, Beijing Children’s Hospital, Capital Medical University, National Center for Children Healthy, 100101 Beijing, China
Abstract
Objectives: Noninvasive brain stimulation (NIBS) has been shown to
effectively alleviate negative and positive symptoms in patients with
schizophrenia. However, its impact on depressive symptoms and general
psychopathology symptoms (GPSs), which are crucial for functional outcomes,
remains uncertain. We aimed to compare the efficacy of various NIBS interventions
in treating depressive symptoms and GPSs. Methods: We conducted a
comprehensive search of multiple databases and performed a meta-analysis to
evaluate the efficacy of NIBS in treating depressive symptoms and GPSs in
schizophrenia. The effect sizes of NIBS for depression symptoms and GPSs were
estimated using standard mean differences (SMDs) with 95% confidence intervals
(CIs). Subgroup analyses were employed to examine potential influencing factors
on the pooled SMD of NIBS for GPSs. Results: Our search yielded 35
randomized controlled trials involving 1715 individuals diagnosed with
schizophrenia. The protocol of this systematic review was registered with INPLASY
(protocol ID: INPLASY202320082). Neither repetitive transcranial magnetic
stimulation (rTMS) nor transcranial direct current stimulation (tDCS)
demonstrated significant improvements in depressive symptoms
compared to sham controls. NIBS exhibited a small-to-moderate effect size for
GPSs, with a pooled SMD of –0.2956 (95% CI: –0.459 to –0.132) and a
heterogeneity (I
Keywords
- noninvasive brain stimulation
- SMD
- depressive symptoms
- general psychopathology symptoms
- schizophrenia
- meta-analysis
Schizophrenia is a chronic, recurrent, and highly disabling mental illness [1]. Currently, the first-line treatment for schizophrenia is antipsychotic medication [2]. While these medications effectively address positive symptoms, their efficacy in treating negative and other symptoms of schizophrenia remains limited [3]. Additionally, the adverse effects of antipsychotics may lead to reduced treatment compliance among some patients with schizophrenia [4]. As a result, nonpharmacological interventions, such as noninvasive brain stimulation (NIBS), have emerged as innovative and crucial approaches in the treatment of schizophrenia [5]. NIBS technologies, particularly repetitive transcranial magnetic stimulation (rTMS) and transcranial direct current stimulation (tDCS), have been extensively researched [6, 7].
rTMS induces an electric field in a discrete area of the brain by applying a repetitively pulsed magnetic field over the scalp. This electric field modulates neuronal activity in the area where rTMS is applied. tDCS involves the application of a weak electrical current through two or more electrodes placed on the scalp to stimulate underlying brain tissue. The biological mechanisms underlying the effects of rTMS and tDCS on neuropsychiatric disorders are very complicated and remain unclear. However, the concept of neuroplasticity has been emphasized most often [8]. There is evidence that rTMS produces long-lasting neuroplastic changes and beneficial clinical effects across a variety of neuropsychiatric disorders [9], while tDCS can stimulate neuroplasticity by modulating changes in neuronal membrane potential and increasing cortex excitability [10]. Neuroplasticity refers to the capacity of the brain to change and reorganize itself in response to internal and/or external influences [11]. In summary, the rationale behind rTMS or tDCS therapy is to modulate cortical excitability, increase neural plasticity, and ultimately improve functional outcomes.
Several meta-analyses have evaluated the benefits of NIBS on the negative symptoms of schizophrenia [12, 13]. Numerous studies have also investigated the effects of NIBS on positive symptoms [14, 15, 16]. However, current research data indicate that limited attention has been given to the treatment of general psychopathology symptoms (GPSs) in schizophrenia. General psychopathology symptoms, as measured separately from the Positive and Negative Syndrome Scale (PANSS), provide a separate but parallel measure of the severity of schizophrenic illness [17]. These symptoms encompass a wide range of conditions, including somatic concerns, anxiety, feelings of guilt, depression, motor retardation, poor attention, disturbance of volition, poor impulse control, and active social avoidance; all of which contribute to functional outcomes.
Existing evidence suggests that depression in schizophrenia is linked to a reduced quality of life and an increased risk of suicide [18, 19]. Anxiety in schizophrenia has also been correlated with adverse outcomes, including heightened suicide risk, sleep disturbances, reduced quality of life, increased depression, and neuropsychological impairments [20, 21]. Recent reviews have demonstrated that psychiatric symptoms (psychotic symptoms and GPSs) negatively impact the quality of life in patients with schizophrenia [22]. A previous study found no correlation between suicide attempts and PANSS positive and negative scores, while PANSS general psychopathology scores were associated with suicide attempts [23]. The presence of GPSs is likely to affect patients’ functional outcomes and quality of life [24, 25]. Therefore, GPSs intervention is also critical for the clinical treatment of schizophrenia. However, current NIBS technology primarily targets the main positive and negative symptoms [26, 27], with few intervention studies focusing on GPSs. Treatment of GPSs in schizophrenia is an essential yet often overlooked aspect of schizophrenia management.
Some studies of NIBS interventions for negative symptoms also report changes in GPSs. For example, Zheng et al. [28] found that 10 Hz rTMS could improve both negative symptoms and general psychopathology symptoms. Gomes’s research emphasized the therapeutic effects of tDCS for treating negative symptoms in schizophrenia, noting a significant reduction in general PANSS scores from baseline to post-tDCS compared to the sham control group [29]. Another study on rTMS for treatment of auditory hallucinations did not observe significant improvements in general psychopathological symptoms [30]. However, these are individual studies, and no meta-analysis has specifically focused on the effects of NIBS on GPSs.
Furthermore, we discovered that different targets and intervention techniques can yield varying results. Ray et al. (2015) [31] utilized 1 Hz rTMS to stimulate the left temporal-parietal cortex (TPC) and observed no significant improvement in total PANSS scores or general psychopathological scores. Similarly, Bais et al. (2014) [30] applied 1 Hz rTMS to the left or bilateral temporoparietal junction area and found no notable improvement in general psychopathological symptoms. In contrast, Li et al. (2020) [32] employed 10 Hz rTMS to stimulate the left dorsolateral prefrontal cortex (DLPFC) and reported significant improvements in both total PANSS scores and general psychopathological scores compared to the control group. Moreover, Lisoni et al. (2022) [33] observed significant improvements in the PANSS general psychopathology subscales following active tDCS in comparison to sham tDCS. These findings suggest that the effectiveness of NIBS interventions on GPSs may be influenced by several factors. Identifying factors that impact NIBS technology in GPSs intervention could prove valuable in designing specialized intervention techniques for GPSs in the future.
This meta-analysis aimed to examine the effectiveness of NIBS in treating General Psychopathology Scale symptoms in schizophrenia and to identify potential moderators influencing the effectiveness of NIBS treatment on GPSs in schizophrenia. We hypothesize that NIBS exerts a mild-to-moderate effect size on GPSs in schizophrenia, and factors such as varying intervention techniques, targets, and other variables may influence the intervention’s efficacy.
We conducted a search of five databases, including PubMed, Web of Science, PsycINFO, Google Scholar, and the China National Knowledge Infrastructure (CNKI). Only studies published between January 1, 1999, and December 1, 2022, were included in our search. The following search terms were used: “transcranial magnetic stimulation”, “TMS”, “transcranial direct current stimulation”, “tDCS”, “brain stimulation”, “schizophrenia”, “psychotic disorder”, “psychosis”, “general symptom”, “general psychopathology”, “positive and negative syndrome scale”, “PANSS”, “randomized controlled trial”, and “RCT”. Additionally, we reviewed the references of the retrieved articles to identify any other relevant studies and searched for corresponding terms in Chinese in CNKI.
In this study, the following inclusion and exclusion criteria were employed:
Inclusion Criteria:
(1) Utilization of a randomized sham-controlled study design.
(2) Diagnosis of schizophrenia in patients according to standardized criteria, such as the Diagnostic and Statistical Manual of Mental Disorders (DSM), International Statistical Classification of Diseases and Related Health Problems (ICD), or Chinese Classification of Mental Disorders (CCMD).
(3) Implementation of rTMS or tDCS interventions.
(4) Employment of the PANSS to evaluate general psychopathology symptoms as outcome measures.
(5) Maintenance of consistent psychotropic medication dosages before and throughout the intervention.
(6) Articles written in English or Chinese.
Exclusion Criteria:
(1) Participants exhibited significant positive or negative symptoms.
(2) Patients demonstrated additional psychotic symptoms.
(3) General psychopathology symptom scores were not reported.
(4) Articles consisted of duplicate records or contained overlapping samples.
(5) Articles were case reports, editorials, commentaries, or review papers.
(6) The study lacked a control group, or essential information for the control group was missing (e.g., symptom presence or age data).
(7) Participants were under the age of 18.
The quality of each study was evaluated using the modified Jadad scale [34]. The assessment criteria included randomization, blinding strategy, withdrawals/dropouts, inclusion/exclusion criteria, adverse effects, and statistical analysis. Two authors independently assessed each trial, and any discrepancies were resolved through discussion to reach a consensus. All the studies incorporated in this analysis had Jadad scores of 5 or higher.
We extracted the following information from the included studies: first author’s name, year of publication, demographic, and clinical characteristics (sample size, male and female distribution, mean age), study location, diagnostic criteria, outcome measurements, participant groups, and the number of rTMS or tDCS sessions. These data were extracted independently by two authors, and any discrepancies were discussed with a third author to reach a consensus.
The Global Psychopathology Scale scores were measured independently from the positive and negative symptoms assessed by the PANSS. These scores offer a distinct yet complementary evaluation of the severity of schizophrenia, which is useful for interpreting syndrome scores [17]. The GPSs covers a range of symptoms, including somatic concerns, anxiety, feelings of guilt, depression, motor retardation, poor attention, disturbance of volition, poor impulse control, and active social avoidance, all of which are critical to functional outcomes.
The standardized mean difference (SMD) for each study was calculated, along with
the pooled SMD. A SMD between 0.2 and 0.5 indicated mild-to-moderate efficacy of
NIBS, while SMD values between 0.5 and 0.8 suggested moderate-to-large efficacy
[35]. The I
The choice of a computational model for meta-analysis depends on whether studies are expected to share a common effect size, as well as the objectives of the analysis [36]. A fixed-effect meta-analysis estimates a single effect, assumed to be common across all studies, while a random-effects meta-analysis estimates the mean of a distribution of effects. In this review, various types of NIBS studies collected from the published literature were incorporated into the meta-analysis, potentially leading to differences in effect size among the studies. Consequently, the random-effects model was a more suitable choice for this meta-analysis.
Pre- and post-PANSS-G (General Psychopathology Scale of PANSS) differences (mean
and standard deviation values) were extracted from the studies.
All analyses were conducted in R (version 3.5.3, The website:
https://www.r-project.org) using the “meta” and “metafor” packages, with a
p value
First, the Jadad scale was applied to evaluate the quality of the included studies. Studies with Jadad scale scores below 4 were excluded. Second, publication bias for the included studies was assessed using Egger’s test and illustrated with a funnel plot. Third, a sensitivity analysis identified studies contributing to high heterogeneity. Studies were excluded when the change in heterogeneity associated with a particular study exceeded 5%. Fourth, the pooled effect size was calculated based on the SMD. Fifth, subgroup analysis explored the heterogeneity in the effect sizes of NIBS for depressive symptoms and GPSs. These two methods (including the subgroup analysis and sensitivity analysis) also helped identify potential influencing factors of the efficacy of NIBS for treating GPSs.
The flow diagram in Fig. 1 illustrates the search and selection process results. This study was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [37] and the protocol of this systematic review was registered with INPLASY (protocol ID: INPLASY202320082). Ultimately, 35 studies were identified and incorporated into the meta-analysis. For a detailed view of the study identification process, please refer to Fig. 1. PRISMA checklist is shown in Supplementary Material-PRISMA checklist.
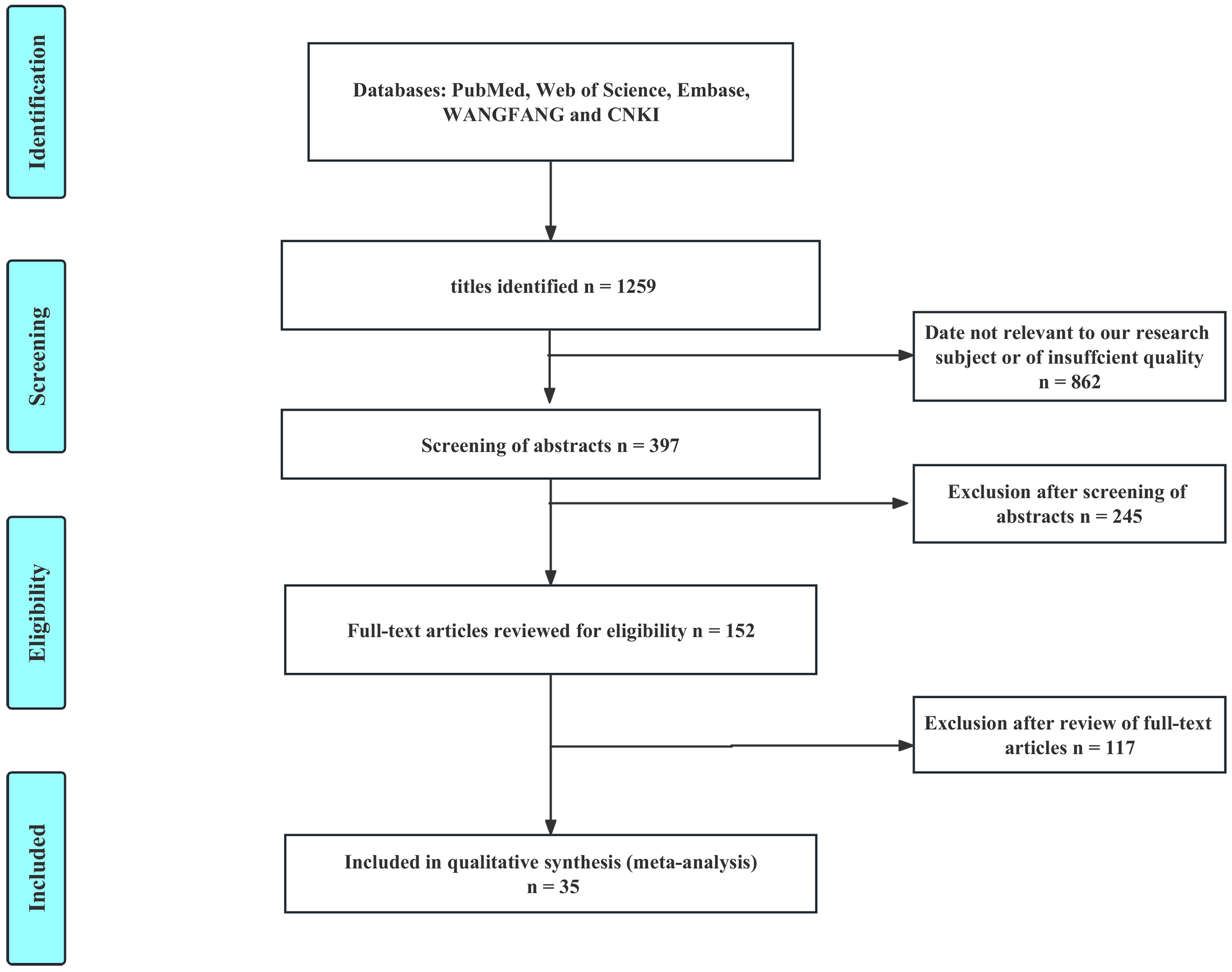 Fig. 1.
Fig. 1.Flowchart of the identification of included studies.
We have compiled all the extracted data in Table 1 (Ref. [28, 29, 30, 31, 32, 33, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66]). Out of the 35 studies, 12 were conducted in East Asia, 10 in Europe, 5 in South Asia, 4 in North America, 3 in South America, and 1 in Western Asia. The intervention methods featured in these studies consisted of 25 rTMS studies and 10 tDCS studies. For more information, please refer to Table 1.
| No. | First author | Year | Age (Years) | Area | Male/Female | Diagnosis criteria | Sample size | Comparison group | Outcome measurements | Sessions | Stimulation site |
| 1 | Lisoni [33] | 2022 | tDCS: 40.96 |
Italy | 39/11 | DSM-V | 50 | Sham | PANSS, CGI, SUMD, BACS | 15 sessions | anode: left DLPFC; cathode: right orbitofrontal region |
| Sham: 44.44 |
|||||||||||
| 2 | Du [40] | 2022 | rTMS: 45.9 |
Mainland China | 20/21 | ICD-10 | 41 | Sham | SANS, PANSS, PRM | 20 sessions | left DLPFC |
| Sham: 45.1 |
|||||||||||
| 3 | Gupta [41] | 2021 | rTMS: 29.70 |
India | 39/0 | N/A | 39 | Sham | PANSS, PGI-MS | 10 sessions | left temporo-parietal cortex |
| Sham: 31.26 |
|||||||||||
| 4 | Wen [42] | 2021 | rTMS: 41.4 |
Mainland China | 25/20 | DSM-IV | 45 | Sham | PANSS, RBANS, SCWT, UKU | 20 sessions | left DLPFC |
| Sham: 38.8 |
|||||||||||
| 5 | Dharani [43] | 2021 | tDCS: 39.14 |
India | 12/2 | ICD-10 | 14 | Sham | SANS, PANSS, CGI-S | 10 sessions | anode: left DLPFC |
| Sham: 33.85 |
|||||||||||
| 6 | Valiengo [44] | 2020 | tDCS: 34.6 |
Brazil | 80/20 | DSM-IV | 100 | Sham | PANSS, CDSS, AHRS, GAF, SANS | 10 sessions | anode: left prefrontal cortex; cathode: left temporoparietal junction |
| Sham: 35.9 |
|||||||||||
| 7 | Guan [45] | 2020 | rTMS: 51.9 |
Mainland China | 41/0 | DSM-IV | 41 | Sham | PANSS, RBANS | 40 sessions | left DLPFC |
| Sham: 56.0 |
|||||||||||
| 8 | Kumar [46] | 2020 | rTMS: 32.4 |
India | 57/43 | ICD-10 | 100 | Sham | PANSS, SANS, CGI-S, CDSS | 20 sessions | left DLPFC |
| Sham: 30.8 |
|||||||||||
| 9 | Li [32] | 2020 | rTMS: 23.9 |
Mainland China | 47/50 | DSM-IV | 97 | Sham | MCCB, PANSS | 10 sessions | left DLPFC |
| Risperidone: 24.0 |
|||||||||||
| 10 | Xiu [47] | 2020 | 10 Hz rTMS: 50.7 |
Mainland China | 97/0 | DSM-IV | 97 | Sham | RBANS, PANSS | 40 sessions | left DLPFC |
| 20 Hz rTMS: 52.0 |
|||||||||||
| Sham: 54.7 |
|||||||||||
| 11 | Zhuo [48] | 2019 | rTMS: 28.97 |
Mainland China | 41/19 | DSM-IV | 60 | Sham | SANS, PANSS, MCCB, CGI | 20 sessions | left DLPFC |
| Sham: 30.63 |
|||||||||||
| 12 | Gomes [29] | 2018 | tDCS: 39.17 |
Brazil | 17/7 | DSM-IV | 24 | Sham | PANSS, CDSS, GAF, MATRICS | 10 sessions | anode: left prefrontal cortex; cathode: contralateral area |
| Sham: 33.75 |
|||||||||||
| 13 | Jeon [49] | 2018 | tDCS: 40.00 |
Korea | 25/27 | DSM-V | 52 | Sham | PANSS, CGI, CDSS, MCCB, WCST | 10 sessions | anode: left DLPFC; cathode: right DLPFC |
| Sham: 39.86 |
|||||||||||
| 14 | Mellin [50] | 2018 | tDCS: 29.57 |
United States | N/A | DSM-IV | 14 | Sham | AHRS, PANSS, BACS | 10 sessions | anode: left DLPFC |
| Sham: 38.86 |
|||||||||||
| tACS: 47 |
|||||||||||
| 15 | Lindenmayer [39] | 2019 | tDCS: N/A | New York | 24/4 | DSM-V | 28 | Sham | PANSS, MCCB, AHRS, CGI-S | 40 sessions | anode: frontal cortex on the left side; cathode: left auditory cortex |
| Sham: N/A | |||||||||||
| 16 | Hasan [51] | 2017 | rTMS: 33.88 |
Germany | 60/13 | N/A | 73 | Sham | PANSS, CGI, GAF, MADRS, MRI | 15 sessions | left DLPFC |
| Sham: 36.00 |
|||||||||||
| 17 | Garg [52] | 2016 | rTMS: 32.40 |
India | 33/7 | ICD-10 | 40 | Sham | PANSS, CDSS | 10 sessions | the vermal part of cerebellum |
| Sham: 30.75 |
|||||||||||
| 18 | Fröhlich [53] | 2016 | tDCS: 43.38 |
USA | 22/4 | DSM-IV | 26 | Sham | AHRS, PANSS | 5 sessions | anode: left DLPFC; cathode: left temporo-parietal junction |
| Sham: 40.00 |
|||||||||||
| 19 | Huang [54] | 2016 | rTMS: 40.58 |
Mainland China | 37/0 | DSM-IV | 37 | Sham | PANSS, WCST, MADRS | 21 sessions | left DLPFC |
| Sham: 39.39 |
|||||||||||
| 20 | Dlabac-de Lange [38] | 2015 | rTMS: 41.8 |
The Netherlands | 26/6 | DSM-IV | 32 | Sham | SANS, PANSS, MADRS, WHOQOL-BREF, BIS | 30 sessions | the bilatera-l DLPFC |
| Sham: 32.3 |
|||||||||||
| 21 | Mondino [55] | 2016 | tDCS: 36.7 |
France | 15/8 | DSM-IV | 23 | Sham | PANSS, AHRS, fMRI | 10 sessions | anode: left DLPFC; cathode: left temporo-parietal junction |
| Sham: 37.3 |
|||||||||||
| 22 | Gan [56] | 2015 | rTMS: 28 |
Mainland China | 44/23 | DSM-IV | 67 | Sham | PANSS, TESS, VAS | 20 sessions | left DLPFC |
| Sham: 29 |
|||||||||||
| 23 | Quan [57] | 2015 | rTMS: 46.87 |
Mainland China | 72/45 | DSM-IV | 117 | Sham | PANSS, SANS, CGI, UKU | N/A | left DLPFC |
| Sham: 46.87 |
|||||||||||
| 24 | Ray [31] | 2015 | rTMS: 31.35 |
India | N/A | ICD-10 | 40 | Sham | AHRS, PANSS, CGI | 10 sessions | left temporo-parietal region |
| Sham: 29.30 |
|||||||||||
| 25 | Smith [58] | 2015 | tDCS: 46.76 |
United States | 22/8 | DSM-IV | 30 | Sham | MCCB, PANSS | 5 sessions | anode: left DLPFC; cathode: the contralateral supraorbital ridge |
| Sham: 44.88 |
|||||||||||
| 26 | Bais [30] | 2014 | Left rTMS: 37.2 |
The Netherlands | 27/20 | DSM-IV | 47 | Sham | PANSS, AHRS | 12 sessions | left temporo-parietal junction area |
| Bilateral TMS: 33.9 |
|||||||||||
| Sham: 37.3 |
|||||||||||
| 27 | Prikryl [59] | 2014 | rTMS: 30.40 |
Czech Republic | 35/0 | ICD-10 | 35 | Sham | PANSS, MADRS, CDSS | 21 sessions | left DLPFC |
| Sham: 34.58 |
|||||||||||
| 28 | Zhao [60] | 2014 | 10 Hz rTMS: 48.0 |
Mainland China | 33/36 | DSM-IV | 69 | Sham | PANSS, SANS, TESS | 10 sessions | left DLPFC |
| 20 Hz rTMS: 49.1 |
|||||||||||
| Sham: 46.7 |
|||||||||||
| 29 | Prikryl [61] | 2012 | rTMS: 30.47 |
Czech Republic | 30/0 | ICD-10 | 30 | Sham | PANSS, VFT, fMRI | 15 sessions | left DLPFC |
| Sham: 34.55 |
|||||||||||
| 30 | Zheng [28] | 2012 | 10 Hz rTMS: 56.5 |
Mainland China | 45/0 | CCMD-3 | 45 | Sham | PANSS, VSWM, VFT | 5 sessions | left DLPFC |
| 20 Hz rTMS: 56.8 |
|||||||||||
| Sham: 55.6 |
|||||||||||
| 31 | Prikryl [62] | 2007 | rTMS: 31.36 |
Czech Republic | 22/0 | ICD-10 | 22 | Sham | PANSS, SANS, SAPS, MADRS, CDSS | 15 sessions | left DLPFC |
| Sham: 36.46 |
|||||||||||
| 32 | Rosa [63] | 2007 | rTMS: 29.83 |
Brazil | 6/5 | DSM-IV | 11 | Sham | PANSS, CGI, AHRS, VAS | 10 sessions | the left temporo-parietal cortex |
| Sham: 33.00 |
|||||||||||
| 33 | Saba [64] | 2006 | rTMS: 30.7 |
France | 13/3 | DSM-IV | 16 | Sham | PANSS, CGI | 10 sessions | the left temporo-parietal cortex |
| Sham: 30.6 |
|||||||||||
| 34 | Holi [65] | 2004 | rTMS: 38.5 |
Finland | 19/3 | DSM-IV | 22 | Sham | PANSS, MMSE, SCL-90 | 10 sessions | left DLPFC |
| Sham: 34.8 |
|||||||||||
| 35 | Klein [66] | 1999 | rTMS: 30.2 |
Israel | 11/20 | DSM-IV | 31 | Sham | CGI, PANSS, BPRS, HDRS | 10 sessions | the right prefrontal area |
| Sham: 29.5 |
Abbreviations: AHRS, Auditory Hallucinations Rating Scale; CDSS, Calgary Depression Scale for Schizophrenia; GAF, Global Assessment of Functioning; NA, not applicable; PANSS, Positive and Negative Syndrome Scale; SAPS, Scale for the Assessment of Positive Symptoms; SANS, Scale for the Assessment of Negative Symptoms; tDCS, transcranial direct current stimulation; RBANS, Repeatable Battery for the Assessment of Neuropsychological Status; WHOQOL-BREF, World Health Organization Quality of Life-BREF; BIS, Birchwood Insight Scale; PRM, pattern recognition memory; VAS, visual analog scale; BACS, Brief Assessment of Cognition in Schizophrenia; UKU, Udvalg for Kliniske Under sogelser; VFT, verbal fluency task; VSWM, visual spatial working memory; PGI-MS, Postgraduate Institute Memory Scale; DSM-IV, Diagnostic and Statistical Manual of Mental Disorders, 4th. Edition; DSM-V, Diagnostic and Statistical Manual of Mental Disorders, 5th edition; WCST, Wisconsin Card Sorting Test; rTMS, repetitive transcranial magnetic stimulation; CGI, Clinical global impression; CGI-S, Clinical global impression-Severity scale; SUMD, Scale to Assess Unawareness of Mental Disorder; DLPFC, dorsolateral prefrontal cortex; MATRICS, Measurement and Treatment Research to Improve Cognition in Schizophrenia; SCWT, Stroop Color and Word Test; BPRS, Brief Psychiatric Rating Scale; SCL-90, Symptom Checklist-90; MMSE, Mini-mental State Examination; TESS, Treatment Emergent Symptom Scale; MCCB, MATRICS Consensus Cognitive Battery; MRI, Magnetic resonance imaging; fMRI, functional magnetic resonance imaging; HDRS, Hamilton Depression Rating Scale; MADRS, Montgomery-Asberg Depression Rating Scale; ICD-10, The International Statistical Classification of Diseases and Related Health Problems 10th Revision; CCMD, Chinese Classification and Diagnostic Criteria of Mental Disorders.
The quality assessment scores for the included studies based on the Jadad scale all exceeded 5. Details regarding the individual Jadad scale items for each study can be found in Supplementary Table 1.
The included studies were assessed for publication bias. A funnel plot was employed to visually represent potential publication bias. Additionally, Egger’s test was conducted to determine the presence of any publication bias. The resulting p value of 0.20 suggests that no publication bias was detected (refer to Supplementary Fig. 1).
We determined the effect size of NIBS for depressive symptoms using the SMD. The
pooled SMD and confidence interval (CI) for NIBS in relation to depressive
symptoms was –0.0249 (95% CI: –0.2447 to –0.1950). We observed a
heterogeneity (I
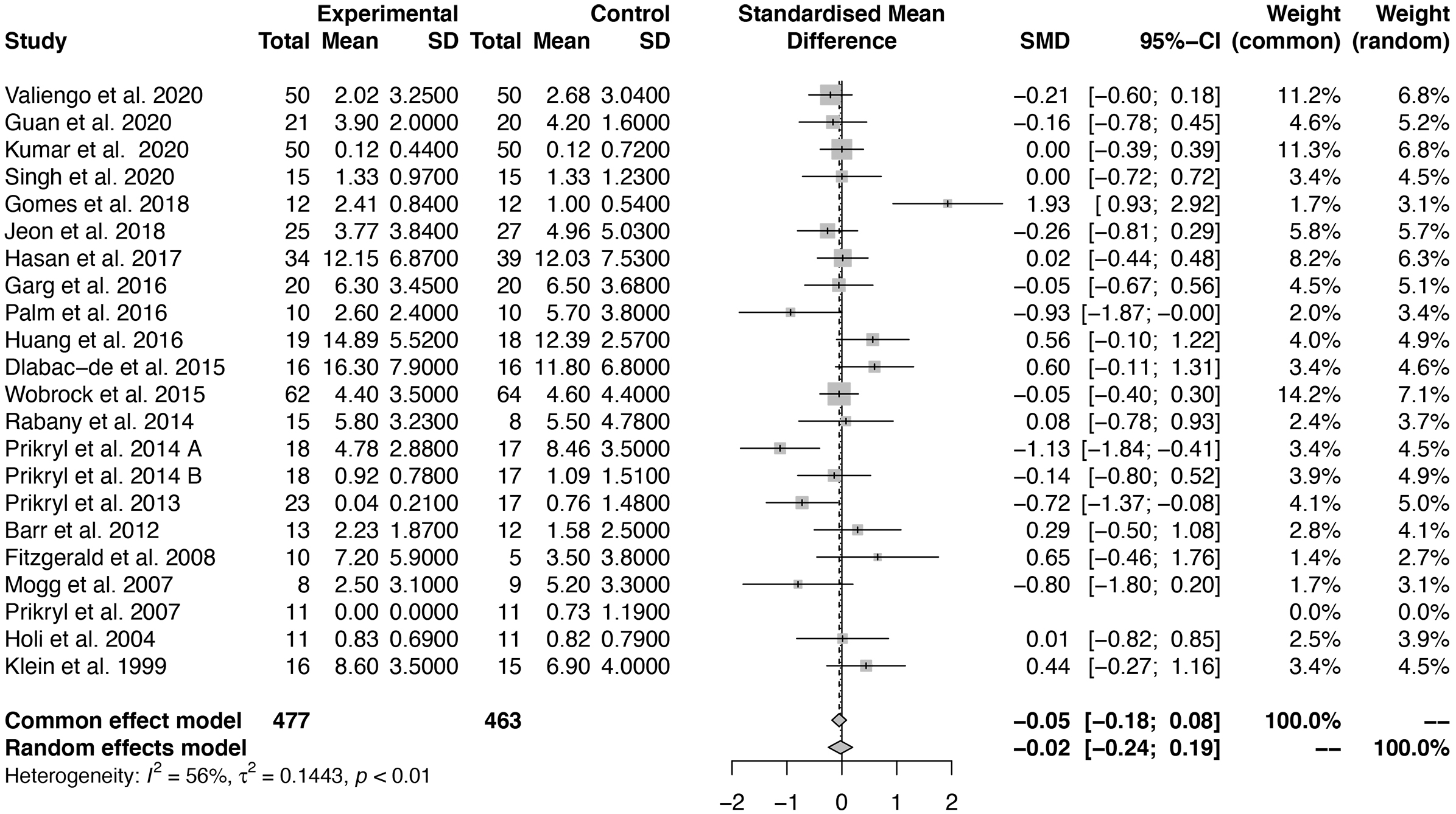 Fig. 2.
Fig. 2.Forest plot of the effect size of noninvasive brain stimulation (NIBS) for depressive symptoms. SMD, Standard mean difference; SD, Standard deviation; CI, Confidence interval.
We also evaluated the effect size of NIBS on GPSs by calculating the SMD. The
pooled SMD and CI for NIBS in addressing GPSs was –0.296 (95% CI: –0.459 to
–0.132), with a heterogeneity (I
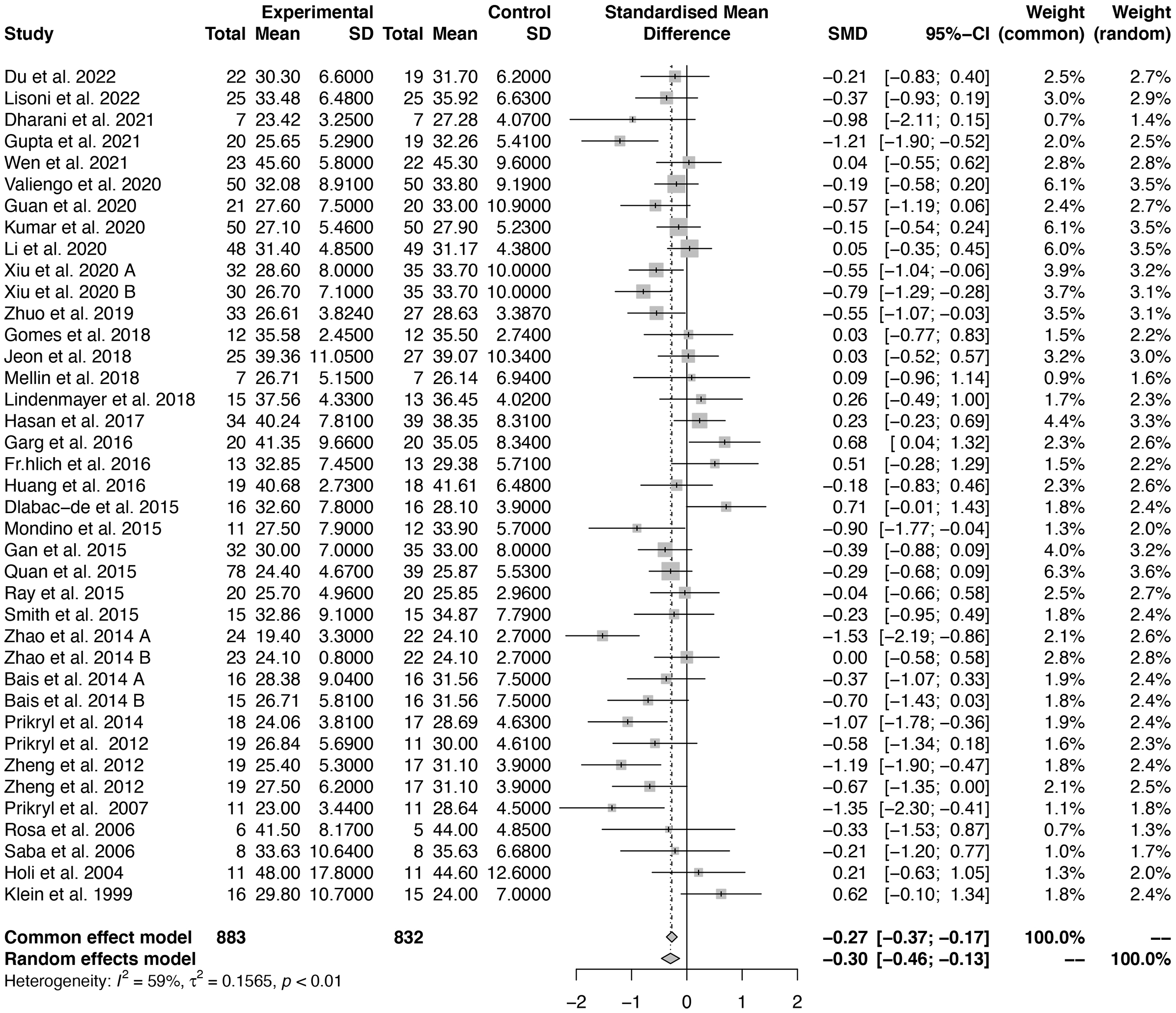 Fig. 3.
Fig. 3.Forest plot of the effect size of NIBS for general psychopathology symptoms (GPSs).
The subgroup analysis showed no significant difference (p = 0.824) in improvement of depressive symptoms between the rTMS and tDCS groups (rTMS: SMD = –0.032, 95% CI: –0.224 to 0.161; tDCS: SMD = 0.099, 95% CI: –1.040 to 1.239). These findings suggest that neither rTMS nor tDCS contributed to the improvement of depressive symptoms. For further information, please refer to Supplementary Fig. 2.
We conducted a subgroup analysis of the pooled SMD of NIBS for GPSs to compare
the effects of rTMS and tDCS. The heterogeneity test revealed significant
differences between the studies (I
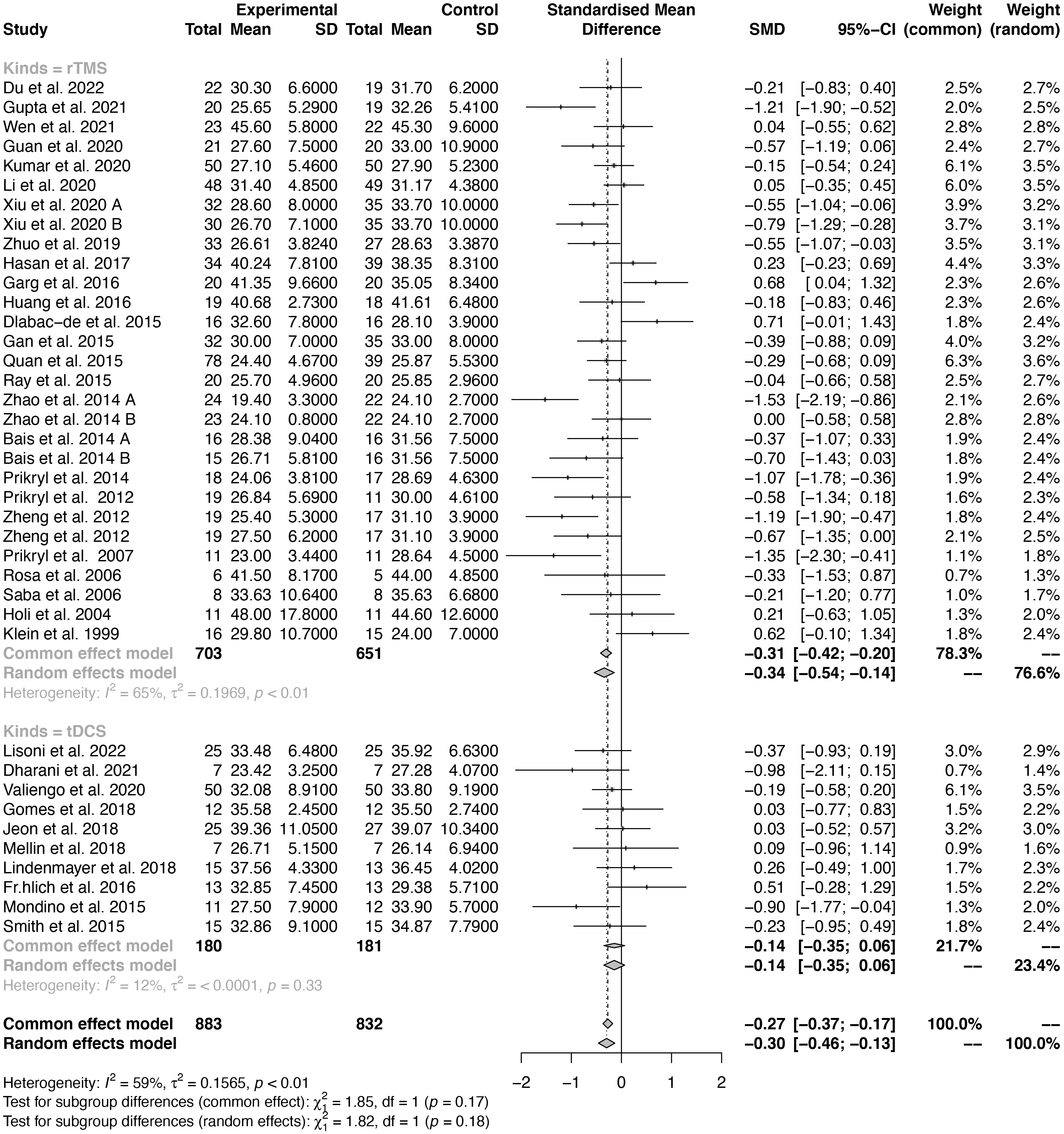 Fig. 4.
Fig. 4.Forest plot of different types of effect sizes of NIBS for GPSs.
Moderate heterogeneity was observed among the 25 included rTMS RCTs
(I
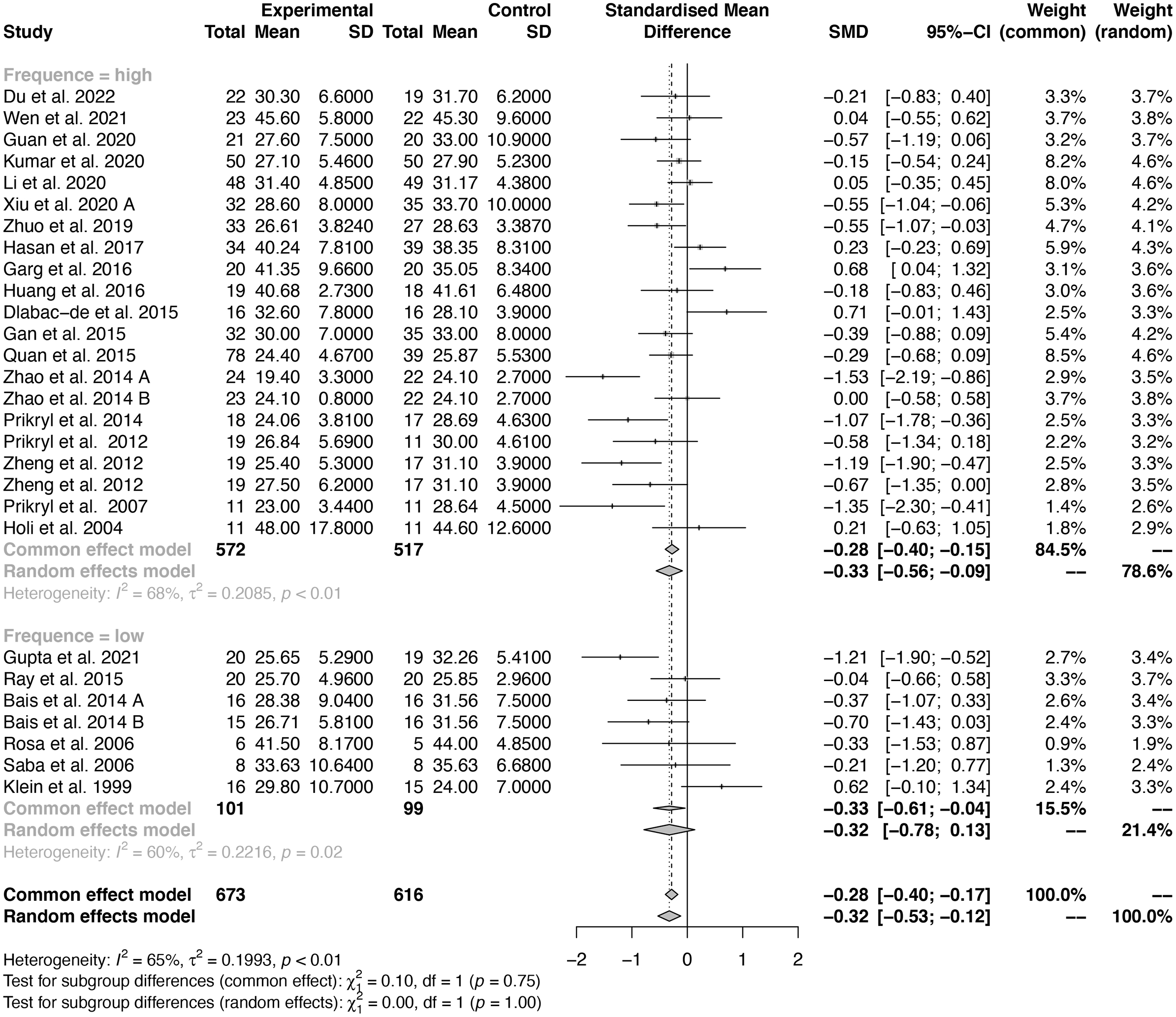 Fig. 5.
Fig. 5.Forest plot of different frequencies of rTMS for General Psychopathology Scale.
A subgroup analysis was performed to examine the pooled SMD of various rTMS
treatment stimulation sites for GPSs. The heterogeneity test revealed significant
disparities between studies (I
For other associated continuous variables (including mean age, sex, and the number of sessions) that might have potential influences on the effect size of NIBS for GPSs, a meta-regression analysis was used to identify whether these associated continuous variables could significantly predict the effect size of NIBS for GPSs.
Only sex had a significant influence on the effect size of NIBS for general
psychopathology symptoms (p
| Moderators | tau |
I |
H |
R |
Test of moderators (p) |
| Mean age | 0.134 | 58.02% | 2.38 | 14.47% | 0.059 |
| Sex | 0.144 | 60.14% | 2.51 | 13.63% | 0.048* |
| Number of sessions | 0.176 | 63.43% | 2.73 | 0.00% | 0.840 |
tau
To our knowledge, this meta-analysis is the most recent and largest study to directly investigate the potential efficacy of NIBS for both depressive and GPSs in schizophrenia. Our study produced several important findings. First, neither rTMS nor tDCS showed a significant improvement in the depressive symptoms associated with schizophrenia compared to the sham controls. However, the main finding of this meta-analysis is that NIBS was effective for GPSs in schizophrenia. The pooled SMD of NIBS for GPSs was small to moderate across 35 studies.
In our study, we found a small-to-moderate effect size of NIBS with rTMS or tDCS on GPSs in the treatment groups when compared to the controls. This finding is consistent with the results of a related meta-analysis conducted by Lee et al. (2022) [67], indicating that NIBS has potential therapeutic effects on GPSs in schizophrenia. Human magnetic resonance spectroscopy (MRS) studies showed that tDCS could modulate the concentration of gamma-aminobutyric acid (GABA), which is a neurotransmitter acting at inhibitory synapses in the brain [68]. rTMS and tDCS have been shown to increase GABA levels in the DLPFC [69, 70]. Accordingly, we speculate that rTMS and tDCS may induce changes in neuroplasticity by modulating the concentration of GABA in stimulated brain regions, which ultimately leads to changes in pathological symptoms. The site of GPSs is related to the pathophysiology of the target symptom. The efficacy of NIBS on GPSs was an additional result of most clinical trials. This may be the reason why the effect of NIBS on GPSs is not strong, with only a small-to-moderate effect.
Since rTMS and tDCS are distinct types of stimulation, further subgroup analysis was conducted in this study. The results indicated that while rTMS had a mild-to-moderate impact on improving GPSs, tDCS had no effect. These results suggest that rTMS may be more effective than tDCS in addressing GPSs in individuals with schizophrenia. Although evidence is currently stronger for rTMS than tDCS, this may be due to the limited number of studies conducted on tDCS. The divergent effects of rTMS and tDCS on symptom dimensions underscore the importance of investigating these treatments separately. Combining their analyses may obscure subtle differences between the two modalities that may have implications for disease characteristics and treatment mechanisms, as well as for guiding the selection of different neuroregulatory interventions for different symptom groups of schizophrenia. Details regarding the specific effects of rTMS and tDCS on symptom dimensions can be found in the study’s report.
In a study utilizing functional magnetic resonance imaging (fMRI) to assess activation during a planning task, increased frontal activation was observed in patients with schizophrenia following stimulation of the DLPFC with rTMS [38]. Therefore, it is speculated that the improvement in GPSs may be related to activation of frontal lobe function in patients with schizophrenia. It is possible that rTMS can regulate neuronal activity and produce a potential therapeutic effect on GPSs.
tDCS has shown promise in alleviating both positive and negative symptoms in schizophrenia. However, its effect on functional outcomes is less clear than that of rTMS. No effect of tDCS on GPSs was observed in this study, which could be attributed to the short treatment duration and limited number of stimulus sessions used. Previous research has predominantly focused on 1–2 weeks of stimulation with 5–10 sessions, which is likely influenced by practical considerations surrounding subject compliance. However, some tDCS studies have reported significant positive effects with twice-daily stimulation [39, 71], indicating that tDCS may only be effective with frequent applications. Despite the current lack of robust evidence to support its effectiveness, we are unable to advise against the use of tDCS for schizophrenia patients, as no reports have suggested that it worsens GPSs poststimulation. In 2022, a meta-analysis was carried out to specifically investigate the impact of tDCS treatment on GPSs. This review included only 8 relevant studies [67]. Notably, Lee et al. [67] reported a pooled SMD of 0.31 (0.05 to 0.57) for GPSs across the 8 studies, while our meta-analysis of 10 studies showed a pooled SMD of –0.1437 (–0.35 to 0.07). Lee et al. [67] also reported a significant reduction in General Psychopathology Scale scores from PANSS after active tDCS treatment compared to sham treatment and examined 5 trials that reported having followed up with their patients. The conclusion drawn by Lee et al. [67] was that tDCS improved GPSs in the short term, but there was no evidence to suggest that the treatment worked in the long term. The inconsistency between our findings and Lee et al. [67] may be attributed to several factors. First, as Lee et al. [67] observed, GPSs encompass a broad range of symptoms, and individual differences in symptom profiles may influence the efficacy of tDCS. Second, our meta-analysis included two additional tDCS studies from 2021 and 2022 that were not included in the Lee et al. [67] analysis. To clarify the findings on the efficacy of tDCS in treating GPSs, further studies with larger sample sizes are needed.
In our meta-analysis, we did not impose restrictions on rTMS parameters during
study selection, which resulted in the inclusion of studies utilizing different
stimulus frequencies (ranging from 1 Hz to 20 Hz) and stimulus locations
(including left DLPFC, bilateral DLPFC, and left TPC). Our subgroup analysis
focusing on the different frequencies of rTMS treatment showed that
high-frequency rTMS was effective in improving general psychopathology symptoms
in schizophrenia, while low-frequency rTMS was not found to be effective. It
should be noted that rTMS can be divided into high-frequency stimulation (5–20
Hz) and low-frequency stimulation (
Although the left DLPFC has been the most studied target region for NIBS in the treatment of negative symptoms due to its significant role in the pathophysiology of schizophrenia [73, 74], our meta-analysis results indicate that NIBS has no significant effect on improving depressive symptoms in schizophrenia. There are several possible explanations for this finding. First, depressive symptoms in schizophrenia are different from other depressive disorders, and in schizophrenia patients, reductions in prefrontal cortex grey matter (GM) volume are associated with depressive symptoms and auditory verbal hallucinations (AVHs) [75]. GM damage is more severe in patients with first-episode schizophrenia who have depressive symptoms than in those who do not [76], indicating that depressive symptoms in schizophrenia may be a nonnegligible factor in treatment resistance. Schizophrenia patients with depressive symptoms do not respond as well to current medications and have a worse long-term prognosis than those without depressive symptoms [77]. Therefore, we speculate that schizophrenia patients with depressive symptoms are less sensitive to NIBS.
Second, depression is not a negative symptom but a common confound for negative symptoms of schizophrenia due to their overlapping conditions. Negative symptoms may mask depressive symptoms, making it difficult to distinguish them clinically. Third, the efficacy of NIBS for depression may be affected by the frequency of stimulation, duration of treatment, and other factors. For example, a previous RCT study applied bimodal tDCS with bi-anodal stimulation over the DLPFC on both sides and demonstrated that this mode could reduce negative and depressive symptoms in patients with schizophrenia [78]. This finding may indicate that improving depressive symptoms in schizophrenia requires a stronger electrical dosage as well as deeper brain stimulation. Fourth, the assessment tools used were not uniform. The Calgary Depression Scale for Schizophrenia (CDSS) is an ideal tool for the assessment of depressive symptoms in people with schizophrenia [79]. However, the CDSS has not been widely used, and some of the included studies adopted other scales for the assessment of depressive symptoms in patients with schizophrenia, for example, the PANSS-Depression score and Montgomery-Asberg Depression Rating Scale. Finally, it is worth noting that most of the research designs and target populations for NIBS do not involve GPSs. The studies we included mostly concerned NIBS treating positive or negative symptoms of schizophrenia, and there were few studies directly investigating GPSs or depressive symptoms of schizophrenia. Therefore, it cannot be ruled out that there is a floor effect with negative outcomes; that is, NIBS is ineffective for depressive symptoms in schizophrenia. Given the high prevalence of depressive symptoms in schizophrenia, there is an urgent need for an understanding of the underlying neural mechanisms to identify therapeutic targets for its effective treatment.
Our study focused on the effect of NIBS on the GPSs and depressive symptoms of schizophrenia. General psychopathology symptoms encompass a broad range of symptoms, and some of these symptoms overlap with negative symptoms of schizophrenia, such as anhedonia. This also suggests that anhedonia may be a more common symptom in people with schizophrenia. Our findings may indicate a direction for future large-scale randomized controlled trials. Future studies should also investigate the neural basis of GPSs in more detail, such as MRI or combined transcranial magnetic stimulation and electroencephalography (TMS-EEG) techniques, which may provide insights into its underlying mechanisms and clues for more targeted interventions.
For individuals with obvious GPSs rTMS therapy may be the preferred therapeutic technique. However, effective treatment involves considering numerous parameters, such as stimulus intensity, frequency of stimulus train, site of stimulation, and course of treatment. Further research is necessary to test and optimize these settings and explore the maintenance effect of rTMS after treatment. Given challenges in the treatment of schizophrenia patients with GPSs and depressive symptoms, it is essential to conduct in-depth neural mechanistic studies to identify targets for the development of effective therapies.
While our study provides evidence supporting the efficiency of rTMS as an adjunctive treatment for GPSs in schizophrenia, it is important to acknowledge several limitations. First, the credibility of our results may be reduced due to the limited number of trials included and their small sample sizes. Second, the subgroup analysis was limited to only three related factors due to the lack of available data. Third, the potential therapeutic effect of concomitant antipsychotic medication cannot be entirely excluded, as no RCTs excluded them from their study design. Finally, the limited reporting of follow-up data a month or more after treatment prevents conclusions from being drawn about the duration of effects, which is a significant limitation. Despite these limitations, our study provides valuable insights into the use of rTMS as an adjunctive treatment for schizophrenia.
In conclusion, our meta-analysis demonstrates that rTMS is effective in treating GPSs in schizophrenia, while the efficacy of tDCS in addressing these symptoms requires further exploration. Psychiatrists should prioritize the management of GPSs during physical interventions, and rTMS may provide advantages in this regard. However, more conclusive evidence is needed to support this claim.
The datasets generated and/or analysed in this study are publicly available. They can be obtained from the corresponding authors upon reasonable request.
WQH, HW, NH, JBC, and XZZ were involved in the interpretation of results and manuscript preparation. FQL and YL were involved in the conceptualization and design of the study. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We would like to thank the participants in this study, who generously gave their time.
This work was supported by the National Natural Science Foundation of China (NSFC) under Grant No. 82171538, 82001445 and the Natural Science Foundation of Beijing Municipality under Grant No. 7212035, 7232057, Beijing Hospitals Authority Youth Programme Grant No. QML20211203, Long Yue Program of Hospital Science Fund for Young Scholar, grant number LY202103.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





