1 Unidad de Investigación UNAM-INC, División de Investigación, Facultad de Medicina, UNAM, Instituto Nacional de Cardiología Ignacio Chávez, 14080 Ciudad de México, Mexico
2 Unidad de Investigación Biomédica en Infectología e Inmunología, Hospital de Infectología, Centro Médico Nacional “La Raza”, IMSS, Col. La Raza, 02990 Ciudad de México, Mexico
3 Department of Experimental and Clinical Medicine, University of Florence, 50134 Florence, Italy
4 Interdisciplinary Internal Medicine Unit, Careggi University Hospital, 50134 Florence, Italy
Abstract
Depression is the leading cause of disability worldwide, contributing to the global disease burden. From above, it is a priority to investigate models that fully explain its physiopathology to develop new treatments. In the last decade, many studies have shown that gut microbiota (GM) dysbiosis influences brain functions and participate, in association with immunity, in the pathogenesis of depression. Thereby, GM modulation could be a novel therapeutic target for depression. This review aims to evidence how the GM and the immune system influence mental illness, particularly depression. Here, we focus on the communication mechanisms between the intestine and the brain and the impact on the development of neuroinflammation contributing to the development of Major Depressive Disorder (MDD). However, most of the current findings are in animal models, suggesting the need for studies in humans. In addition, more analysis of metabolites and cytokines are needed to identify new pathophysiological mechanisms improving anti-depression treatments.
Keywords
- gut-brain axis
- depression
- mental disorders
- dysbiosis
- microbiota
- microbiome
- immunity
- inflammation
Depression is a common mental disorder illness worldwide. The World Health Organization (WHO) estimates that globally 280 million people have depression. Major depressive disorder (MDD) represents a public health problem due is the most prevalent psychiatric disorder, leading to the third cause of morbidity in the world and one of the leading causes of disability time [1].
MDD is a chronic and relapsing neuropsychiatric disorder characterized by anxiety, decreased thinking, and delayed thinking. Several other symptoms are also present, including irritability, frustration, worthlessness or guilt, low energy, insomnia, hypersomnia, change in appetite or weight, difficulty concentrating, suicide attempts, or thoughts of death or suicide [2].
Regarding the factors that trigger depression, many authors have attributed them to the cumulative effects of both genetic and environmental factors. In recent years, understanding of MDD pathophysiology has progressed. Still, it is not fully understood because there are missing models which satisfactorily integrate all the mechanisms involved in developing this disease [3].
Recent experimental studies have widely described the relationship between mental and gastrointestinal illness. Murine models have revealed an important function of gut microbiota (GM) in the gut-brain axis (GBA) thanks to advanced techniques for studying metagenomic and metabolomic profiles. Due to this, it has been possible to investigate the interactions of GM and their metabolites with the host [4]. For that reason, it is known that GM composition is relevant for proper brain function, and GM alteration, known as gut dysbiosis (GD), can induce the development of mental illness, such as depression or anxiety [5]. There is evidence in mice that restoring GM eubiosis decreases the depression-like phenotypes by the modulation of the microbiota-gut-brain axis (MGBA); it represents a novel therapeutic goal in the treatment of this pathologic condition [6].
This review aims to integrate evidence of both influence of microbiota and immunity on depression, focusing on MDD pathogenesis, where the communication between the intestine and the brain contributes to depression development and modulation through neuroinflammation.
About 100 trillion symbiotic microorganisms inhabit the human body, including archaea, bacteria, parasites, and viruses. Commensal microorganisms inhabit the host after birth, and the composition and function of microbiota are influenced by age, sex, race, and diet [7]. GM is represented by over 1000 different species of bacteria [8]. GM plays an important role in metabolic and immune homeostasis, keeping the gut integrity, shaping the intestinal epithelium, and protecting against pathogens. When the GM composition is disrupted (dysbiosis), these functions can be disrupted and have been involved in developing different inflammatory diseases [8].
In the pathophysiology of mental disorders, the microbiota can modulate different mechanisms, including metabolic pathways like short-chains-fatty acids (SCFAs) production, bile acid (BA) metabolism, or the synthesis of neurotransmitter precursors, such as the tryptophan [7].
Additionally, GM dysbiosis, which is regulated by host and environmental factors, might impact the endocrine and neurological pathways. However, all the above factors are present when neuropsychiatric diseases develop [9].
The gut microbiota can alter the central nervous system by synthesizing SCFAs and BA, as well as the translocation of lipopolysaccharide (LPS) to the bloodstream. These metabolites can modulate hormonal secretion that alters brain functions, which can lead to the development of mental disorders [10]. These metabolites can directly affect the brain through the blood-brain barrier (BBB) or indirectly interact with the host’s immune, nervous, and endocrine systems [11].
The SCFAs are one of the most studied bacterial metabolites. GM, through gut fermentation, synthesizes SCFAs, mainly acetate, propionate, and butyrate [12]. The SCFAs have several functions in the intestinal lumen, like redox balance, maintaining intestinal barrier integrity, gut hormone production, and epigenetic regulation [13, 14, 15, 16]. However, in recent years, SCFAs have been related to psychiatric illnesses like depression.
The SCFAs are synthesized by intestinal microbiota, mainly by
Akkermansia, Bifidobacteria, Faecalibacterium, Lachnospiraceae,
Lactobacillus, and Ruminococcus species, among others [17]. Different
pathways are known by which SCFAs modulate the neural response, such as
stimulating the microglial cells maturation and homeostasis or inhibiting histone
deacetylase activity, which modifies gene expression [18, 19]. In addition, the
acetate can cross the BBB and decrease appetite while the butyrate acts as an
anti-inflammatory molecule by inducing cytokine secretion of interleukin-10
(IL-10) in regulatory T cells (Treg), which is important for immune homeostasis,
and its depletion has been related with increased depression-like symptoms [20, 21]. Also, the SCFAs induce the release of intestinal neuropeptides, for example,
YY peptide (YYP) and glucagon-like peptide 2 (GLP-2) peptides, as well as
hormones involved in the maintenance of intestinal barrier, cellular metabolism,
and satiety [22, 23]. On the other hand, valeric acid has been associated with
depression and is produced mainly by Oscillibacter. Valeric acid is
similar structure to
A substantial component of bile, BA, are produced from cholesterol in the liver, and they work in tandem with GM to regulate cholesterol metabolism to aid in lipid digestion and absorption. Cholesterol is metabolized into primary BA by hepatocytes; after that is transported to the gallbladder to finally be released in the duodenum. Upon reaching the intestine, the gut bacteria produce secondary BA–(deoxycholic and lithocholic acid) from primary BA. Only some bacteria, predominantly, Clostridium and Eubacterium, are responsible for secondary BA synthesis [28].
Although the main BA function is during cholesterol metabolism, these have also been associated with the regulation of neurotransmitters. In detail, the BA alter the function of neurotransmitters receptors, like M2 and M3 muscarinic acetylcholine, GABA, and N-methyl-D-aspartate (NMDA) receptors [29]. In vitro studies with cultured hypothalamic neurons, chenodeoxycholate acid inhibits GABA and NMDA receptors [30].
The relationship between BA and depression has been studied in humans. A study in China, patients with MDD showed higher levels of 2,3-Nordeoxycholic acid compared with healthy controls, as well, as lower levels of Taurolithocholic acid (TLCA), Glycolithocholic acid (GLCA) and Lithocholic acid 3-sulfate in patients with MDD, which, were correlated negatively with Hamilton Depression Rating Scale (HAM-D) score. Also in this study, the species Turicibacteraceae, Turicibacterales, and Turicibacter were correlated positively with TLCA and GLCA levels [31].
In other study in United States, patients with severe depression, chenodeoxycholic acid, a primary bile acid, showed reduced levels in comparison with less severe patients with depression [32]. Therefore, GM can induce central nervous system (CNS) illness, like MDD, via BA alterations [8]. A reduction of secondary BA synthesis can lead to dysbiosis and alters the permeability of the intestinal barrier, inducing a pro-inflammatory tone that leads to the pathogenesis of depression [33].
In the last decades, it has been shown that inflammation plays an important role
in MDD. Several cytokines such as IL-1
Several studies have postulated that LPS has been associated with several
diseases, including MDD [35]. In murine models, LPS can induce a systemic
proinflammatory response, decreasing thymus weight and increasing the production
of IFN-
In fact, LPS-treated mice are one of inflammation model depression and have been extensively used for the understanding of molecular pathogenesis and new treatments of MDD [35, 38, 39, 40]. Also, it has been described that LPS can stimulate microglia cells, immune cells of CNS, inducing inflammatory responses that cause death of dopaminergic neurons [41]. Thus, LPS translocation to the bloodstream via intestinal barrier can be associated with MDD patients and can be used as a therapeutic target to treat this mental illness. The gut microbiota plays an important role in the sickness symptoms of LPS-treated mice, since probiotics can reduce the LPS-stimulated markers [42].
The association between LPS and depression has also been described in patients.
In the NESDA (Netherlands Study of Depression and Anxiety) cohort, where
LPS-stimulated markers such as IL-2, IL-6, IL-10, MMP-2, IFN, TNF-
Cytokines can be produced by neurons or other components of CNS, such as microglia and astrocytes, but peripherally secreted cytokines can also gain access to the brain. Cytokines are large molecules, about 15–25 kDa, that do not pass through the BBB, so some hypotheses have been described to explain at least five mechanisms: (a) passage of cytokines through leaky regions of the BBB such as circumventricular organs or choroid plexus, it is considered a humoral pathway; (b) active transport via saturable by cytokine-specific transport molecules on brain endothelium; (c) second-messenger signals from the endothelial lining of the BBB (as well as other cerebral vasculature cells, cytokines stimulate microglia to produce monocyte chemoattractant protein 1(MCP-1)), both considered the cellular pathway; (d) cytokines binding to cytokine receptors in peripheral afferent nerve fibers (like vagus nerve during infections), which in turn transmits signals to brain nuclei (neural pathway) and (e) entry of activated monocytes from the periphery into the brain [35, 44, 45] (Fig. 1).
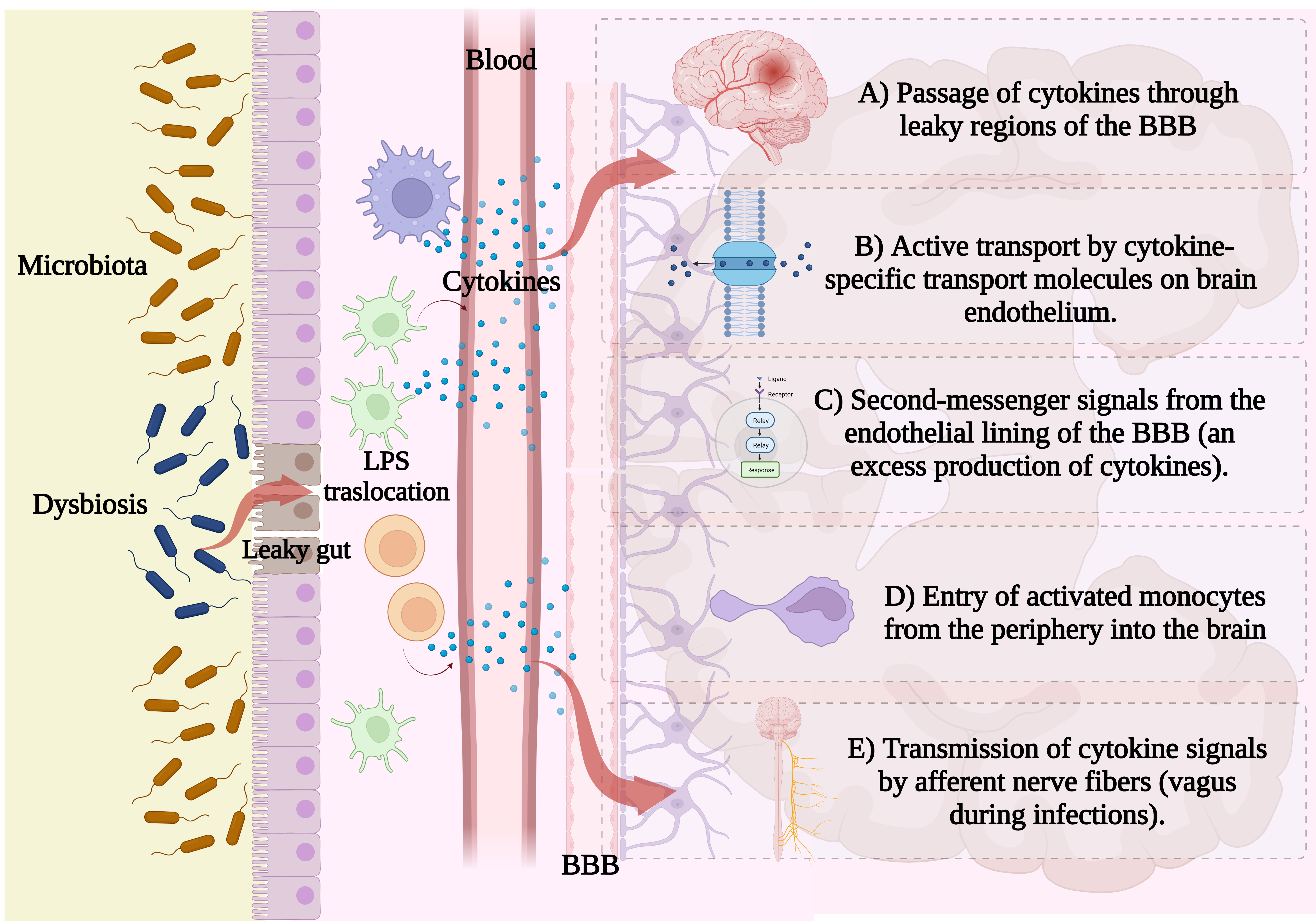 Fig. 1.
Fig. 1.Peripheral cytokine mechanisms in CNS signaling. The humoral pathway involves the (A) passage of cytokines through leaky regions of the blood-brain barrier; the cellular pathway describes in (B) active transport by cytokine-specific transport molecules on brain endothelium, (C) second-messenger signals from the endothelial lining of the BBB or (D) entry of activated monocytes from the periphery into the brain; and the neural pathway, including (E) the transmission of cytokine signals by afferent nerve fibers (Created with BioRender, https://www.biorender.com/).
The neural and neuroendocrine immune systems are associated with the
pathophysiology of depression. Activation of the hypothalamic–pituitary–adrenal
(HPA) axis results in increased cortisol secretion in the blood, which activates
immune cells and inflammatory signals are propagated through various cellular,
humoral and neural pathways and activate brain resident immune cells, which
disturb neuronal integrity by modifying neurotransmitters production [37]. Also,
the increased blood glucocorticoids (GC) induces sympathetic signaling inducing
immune cells mobilization from the bone marrow, lymph node and spleen in addition
to increasing the activation of monocytes and macrophages, which exert
pro-inflammatory effects by increasing pro-inflammatory mediators secretion
(IL-1
Several reports have shown the important role of inflammation in
neuropsychiatric illness. The evidence of inflammation in MDD has been argued in
different meta-analyses that evaluated the concentration of pro-inflammatory
cytokines or other inflammatory markers in patients with MDD. For example,
several studies report an increase in IL-6, IL-12, IL-1
Additionally, the MDD patients have been shown increased expression of
pro-inflammatory cytokines receptors in peripheral blood and cerebrospinal fluid
(CSF) [54], which can be reverted with antidepressant treatments [55]. Also, has
been found innate immune proteins genes such as IL-1
Immune cells also play an important role in the pathogenesis of MDD. Macrophages are immune cells that maintain homeostasis through modulation of inflammation, which are characterized as classically activated cells (M1) or as alternatively activated (M2), secreting pro-inflammatory cytokines or foment tissue repair, respectively. In any disease state of the CNS disorders involving inflammation, there is a shift of macrophage populations towards the M1 phenotype, being important contributors to inflammation neurodegenerative in patients with severe depression [57]. The peripheral blood gene expression profiles in subjects with depression present a pro-inflammatory “M1” macrophage phenotype and an over-expression of IL-6 [56].
The composition of the gastrointestinal microbiota is a mutual selection between the host and the microorganism. The gastrointestinal tract and host immune system maintain the homeostasis between commensals and pathogenic bacteria by a dynamic intestinal barrier with different components such as physical factors (mucus and epithelial layer), biochemical factors (enzymes and antimicrobial proteins) and immunological factors (epithelial associated immune cells).
Studies on the human microbiome suggest that the MGBA plays a role in mental disorders like depression [58], due to an inflammatory state associated with increased intestinal permeability. Some studies have suggested that the gut bacteria may affect neurological functions by altering behavior and the severity of nervous system disorders [59]. Different bacterial species, such as Enterococcus faecium promotes protection against enteric infections; or weaken this barrier, like the pathogenic Salmonella typhimurium and Clostridium difficile [60]. When the intestinal barrier is impaired, it allows products such as LPS to pass through leaky gut [61]. The resulting systemic inflammation is thought to influence brain functioning through cytokines that cross the BBB. Initially, it was assumed that the vagus nerve was the means of transmitting peripheral inflammation signals to the CNS. However, recent studies have identified distinct transport molecules present along the BBB that can actively transport the pro-inflammatory cytokines. In addition, systemic inflammation can alter BBB, making it more permeable to immune molecules [62].
To understand the role of GM in the MDD development is essential to establish the relationship between the gut and the brain. These organs communicate through a complex bidirectional system regulated and coordinated by the interplay of neural, hormonal, metabolic, and immune pathways. At the neural level, the autonomic nervous system (ANS) has the vagus nerve as the primary connection between the CNS and the intestine [10]. Across this connection, the brain receives gut information, and the hypothalamic–pituitary–adrenal (HPA) axis regulates the response to stress with the secretion of different hormones, mainly cortisol. In turn, the enteric nervous system (ENS) is found in the intestine, composed of a large neuronal network that shares neurotransmitters with the CNS and uses the vagus nerve as a central communication channel. Anatomically, the ENS is related to the gut-associated lymphoid tissue (GALT), which is part of the secondary lymphoid organs and is responsible for starting the immune responses against antigens in the intestinal tract. This relationship implies that immune modulators, such as cytokines, also regulate neuronal cell function, and can alter vagus nerve signaling and modify ENS neurotransmitter synthesis [63]. Also, subdiaphragmatic vagus nerve has an important role on Gut-Brain Axis in depression. The subdiaphragmatic vagotomy on LPS-stimulated mice reduces the IL-6 and TNF in comparison with control mice. In this same study, the alpha diversity indexes (Chao1 and Shannon) are reduced in LPS stimulated mice, however this reduction is not observed in LPS-stimulated mice with vagotomy. Firmicutes and Bacteroidetes are increasing in LPS-stimulated mice, but no in mice with vagotomy. These findings indicate that subdiaphragmatic vagus nerve plays an important role on inflammatory response to LPS injection, as well, modulating the gut microbiota composition [64].
In addition to the defensive barrier function, the intestinal epithelium also has an important enteroendocrine activity. Although they make up only 1% of intestinal cells, together they make up the largest endocrine organ in the body. In this context, 5-hydroxytryptamine (5-HT), which has been described as an important intestinal neuromodulator, regulates the whole intestinal physiology, and is involved in intestinal inflammatory processes (Fig. 2).
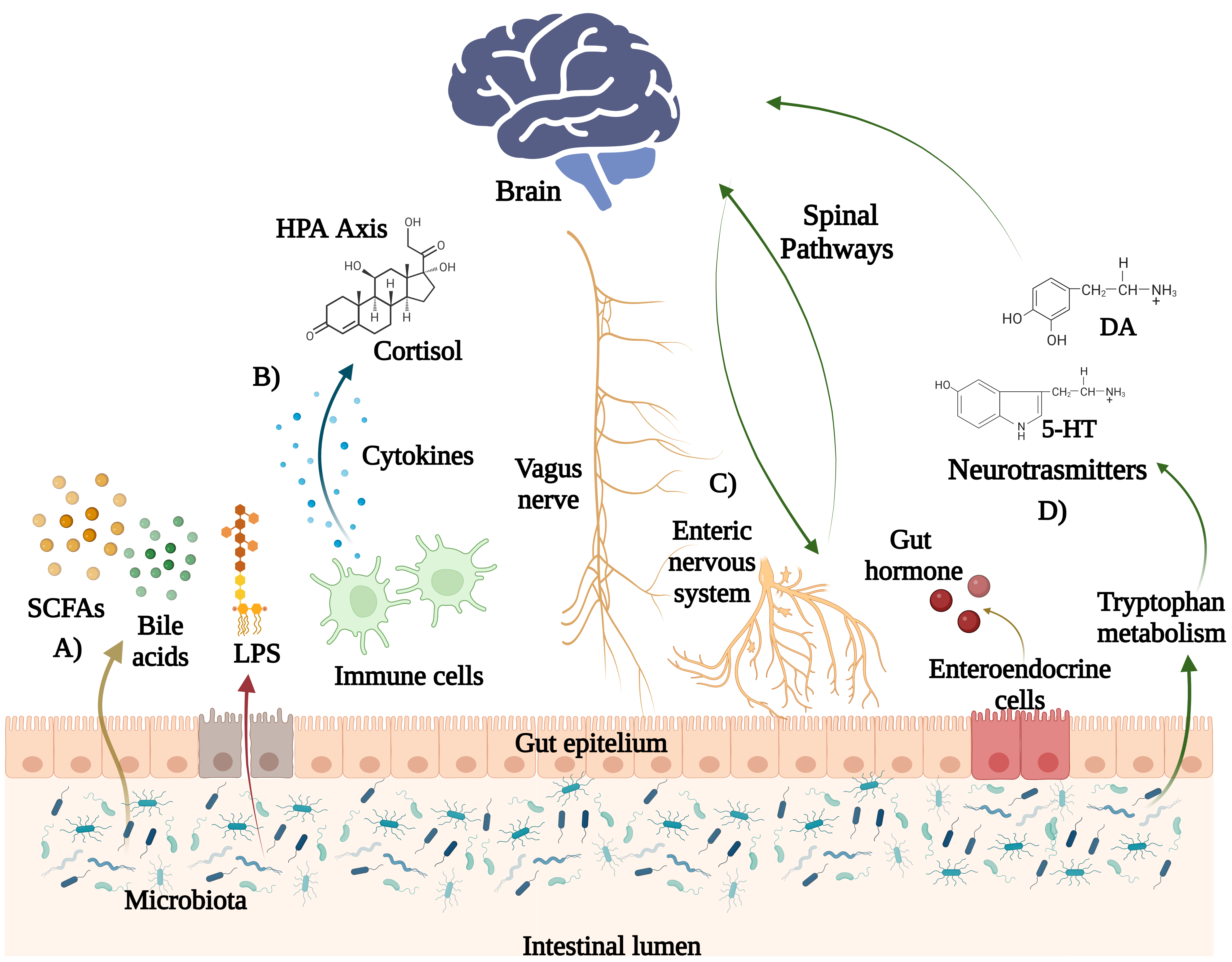 Fig. 2.
Fig. 2.Pathways in the brain-gut-microbiota axis. Some metabolites of gut microbiota include (A) short chain fatty acids (SCFAs), such as butyrate, acetate and propionate, which can modulate host immune cells functions. Bile acids (BA) are converted into secondary bile acids by the gut microbiota and are either transported from the systemic circulation to the brain. (B) Cytokines produced by resident immune cells in the large intestine can activate the hypothalamic-pituitary adrenal (HPA) axis and stimulate the brain through the cortisol. (C) The vagus nerve establishes bidirectional communication between the brain and the gut microbiota. Intestinal endocrine cells secrete intestinal hormones that act on the brain. (D) Gut microbes can also produce neurotransmitters (such as serotonin (5-HT), dopamine, and gamma-aminobutyric acid -GABA-) that can influence the activity of the CNS (Created with BioRender, https://www.biorender.com/).
Diverse studies have documented the importance of the gut microbiota during early life and its role in modulating neurodevelopment and behavior. The microbiome is initially developed via transmission of the placenta, amniotic fluid, and meconium. Early nutrition through breast milk also plays a role in the GM developing [65]. In detail, breastfeeding has been associated with the presence of bacteria in the Bifidobacterium genus present in the gut, as well as with high levels of IgA. Bifidobacterium together with species in the Lactobacillus genus, are capable of producing GABA, an inhibitory regulator of various neural pathways, so they are an important part of the infant microbiome [66]. Finally, a study showed that microbial disruption in early life selectively alters circulating immune cells and modifies neurophysiology in adolescence, including altered myelin-related gene expression in the prefrontal cortex and altered microglial morphology in the amygdala [67].
GM produces neuroactive metabolites acting as neuromodulators of immune homeostasis and modulating emotions. It is proposed that gut dysbiosis may contribute to decreasing monoamines and participate in the depression pathophysiology. The main monoamines are serotonin (5-HT), dopamine (DA), and GABA. Their depletion is considered a risk factor for developing depression. Therefore, most current antidepressants aim to increase their levels at the synapse [68].
The 5 Serotonin (5-hydroxytryptamine, 5-HT) is a monoamine synthesized by a two-step process of 5-HT biosynthesis; the first rate-limiting step is conversion of amino acid L-tryptophan into 5-hydroxytryptophan (5-HTP), a reaction catalyzed by the activity of the tryptophan hydroxylase (Tph) enzyme [69].
Two Tph genes have been described, TPH1 and TPH2. The former is primarily located in a variety of non-neuronal cells, such as the enterochromaffin cells of the gut and the pineal gland [70, 71]. At the same time, the latter is expressed in the intestinal myenteric plexus and within the serotonergic neurons in the raphe nuclei. Since Tryptophan hydroxylase 2 (Tph2) is the rate-limiting step for brain 5-HT biosynthesis, it possible to enhance Tph2 activity within the raphe nucleus after in vivo transfection in mice through the ocular instillation [72], which opens possibilities for new therapeutic alternatives. Besides that, the 5-HT plays a relevant role in regulating physiological function and has been implicated in several psychiatric and neurological disorders, like depression [73], HT depletion reduces melatonin production, which is responsible for sleep and regulating circadian rhythm, whose decrease is related to MDD [74].
About its metabolism, it is known that the amino acid tryptophan intake (ingested in the diet) can follow two pathways in the intestine: the production of 5-HT (3%) or the synthesis of kynurenine (90–95%). Production of quinolinic acid through the kynurenine pathway, is an endogenous agonist of NMDA receptors with neurotoxic and excitotoxic effects and inducing oxidative imbalance and neuronal apoptosis. Quinoline acid-mediated hippocampal volume loss is a distinctive finding in MDD [75]. In contrast, quinolinic acid, a product of the same pathway, can block the NMDA receptor with neuroprotective effects [76].
GM could degrade food-derived tryptophan with tryptophan decarboxylases enzyme and convert it to tryptophan amine, thus limiting the host tryptophan availability thus, decreasing serotonin levels, and leading to mood changes that are MDD symptoms [77]. This was characterized in a metabolomic study in mice that the microbiota has a role in the availability of tryptophan in blood, finding that plasma concentrations of tryptophan in plasma from conventional mice were 40 and 60% lower than in germ-free mice plasma. The authors also found 2.8-lower plasma serotonin levels in germ-free mice in comparison with conventional mice [78].
On the other way, the roles of the kynurenine pathway (KP) in major depressive disorder (MDD) are associated to serotonin deficiency and an inflammatory state. Antidepressants with anti-inflammatory properties may inhibit IDO induction by lowering levels of proinflammatory cytokines in immune activated patients. Using molecular docking in silico, Dawood S et al. [79], demonstrated salicylate and celecoxib strongly dock to the crystal structure of tryptophan 2,3-dioxygenase (TDO). IDO results in immunoactive patients with MDD can be described by IDO induction through changes in KP enzymes affecting glutamatergic function.
In addition, the inflammation induced by dysbiosis contributes to alterations in
tryptophan metabolism. IFN-
Dopamine (DA) is the most abundant catecholamine neurotransmitter in the brain. It is synthesized in dopaminergic neurons from tyrosine in the diet and transported to the brain via the blood-brain barrier. In the human gut, Staphylococcus can take up the precursor L-3,4-dihydroxy-phenylalanine (L-DOPA) and convert it into dopamine by staphylococcal aromatic amino acid decarboxylase (SadA) [83]. More than 50% of dopamine in the human body is synthesized in the gut [83], influencing mucosal blood flow, motility, and gastric secretion [84, 85].
DA is important in regulating anhedonia, a characteristic symptom in patients
with MDD. The mechanism by which peripheral dopamine acts to decrease the
cytokine levels is through activating receptors on Natural Killer lymphocytes
(NKT) that regulate liver immunity. The addition of IFN-
In mice, E. faecium was shown to modulate the immune system and influence the host through dopaminergic pathways. Mice treated with Bifidobacterium in the long term exhibited increased DA and 5-HT, decreasing depression-like behaviors [87]. On the other hand, Bacillus spp. also produced dopamine [81, 82].
Also, as mentioned above, the GM can transform BA, essential to maintain brain homeostasis, through NMDA and GABA signaling pathways [88].
GABA is the main inhibitory neurotransmitter and participates in multiple physiological and psychological processes. Thus, GABA system dysfunction is associated with various neuropsychiatric disorders, including depression.
The ability of the GM to affect behavior has become gradually investigated and recognized. “Psychobiome” has been associated with many neurologic and psychological diagnoses, including autism, Parkinson’s disease, and MDD. The strains of Lactobacillus brevis and Bifidobacterium dentium are efficient GABA producers. The number of Lactobacillus and Bifidobacterium species is decreased in depressed mouse models. In addition, using Lactobacillus rhamnosus reduces the mRNA expression of GABA receptors, which are associated with depressive disorders [89]. Despite the ability of multiple human gut bacterial species to synthesize neurotransmitters, authors have reported one species has ever been sufficient to induce depression [90].
Nonetheless, Gomez-Nguyen A et al. [90], demonstrated that administration of Parabacteroides distasonis induces depressive-like behavior in SAMP1 mice models. Additionally, P. distasonis was not associated with increased intestinal inflammation or other behavior alterations. They also observed that a Crohn’s disease (CD) gut environment in mice could conduct the colonization of P. distasonis and subsequent induction of depressive-like behavior.
The study of MGBA has elucidated the mechanisms by which the GM participates in MDD pathogenesis. This has been directed towards the search for targeted treatments that involve GM. Recent studies of the human microbiota in cognitive functioning have led to the hypothesis that GM-based treatments prevent or relieve depression.
One approach is the use of prebiotics. In a randomized, double-blind, placebo-controlled study, the patients with irritable bowel syndrome were treated with a Short-chain fructo-oligosaccharides (SCFOs) supplemented diet; and the anxiety score of these patients was reduced, alongside increasing Bifidobacteria spp. in the feces, suggesting that SCFOs in diet change the GM composition and reducing anxiety symptoms [91]. In other words, diet could be an essential feature that can help to prevent or treat depression. In another USA study, the patients who received fructo-oligosaccharides as prebiotics versus those treated with Bimuno®GOS (65% galacto-oligosaccharide content) showed reduced cortisol awakening response, which modulates the HPA axis and helps to increase attention to emotional stimuli [92].
Numerous probiotics have shown psych-modulatory capabilities; these so-called psychobiotics, probiotic strains that, when ingested sufficiently, have favorable psychiatric effects in psychopathological diseases, are now known for this property [93].
In Iran, a randomized, double-blind, placebo-controlled clinical trial was conducted. Patients with MDD received probiotics (Lactobacillus acidophilus, Lactobacillus casein, and Bifidobacterium bifidum). The probiotic administration had beneficial effects on Beck Depression Inventory compared with the placebo, decreasing insulin resistance and serum C reactive protein, indicating a lower proinflammatory state [94]. In another clinical trial in Iran, patients with moderate depression were treated with fluoxetine plus probiotic capsule (L. casei, L. acidofilus, L. bulgarius, L. rhamnosus, B. breve, B. longum, and Streptococcus thermophiles). The treatment decreased the Hamilton Depression Rating Scale (HAM-D) score compared with only fluoxetine, suggesting that using probiotics as an adjuvant effectively treats mental disorders like moderate depression [95].
There is evidence that gut dysbiosis is involved in the development of MDD. In summary, gut microbiota and its metabolites regulate the production and availability of neurotransmitters that contribute to different processes of neurogenesis and neuromodulation, commonly affected in MDD.
In addition, gut dysbiosis triggers systemic inflammation where the deterioration of the epithelium and bacterial filtration stimulates the release of proinflammatory cytokines that will travel through the bloodstream and reach the central nervous system, where they will cross the blood-brain barrier, causing depression symptoms. Although enough studies demonstrate an association between gut microbiota and MDD, most of these findings have been made in animal models. Therefore, more clinical studies in humans are needed especially focused on bacterial metabolites and proinflammatory cytokines.
Regarding the included clinical trials, it is essential to determine whether a longer intervention time could decrease other symptoms of MDD or even an improvement in the diagnostic disease stage. The two clinical trials, including metabolomic and proinflammatory cytokine studies, would also allow us to know if the findings in animal models on changes in pro-inflammatory cytokines, secondary bile acids, and SCFA are found in humans.
An integral vision of the individual is required to advance in anti-depression treatments, including new pathophysiological mechanisms, such as gut dysbiosis and systemic inflammation, to complement therapeutic interventions. All this is to improve the people´s quality of life disabled by this condition, whose prevalence is increasing.
Finally, the diet is an essential factor contributing to the GM composition, suggesting the potential for therapeutic dietary strategies to manipulate microbial diversity, stability, and composition. On the other hand, prebiotic and probiotics could restore gut eubiosis in patients with different psychological disorders, like depression.
SRM, LSR, APGG, LACJ and ETC did the literature search and wrote the first draft of the manuscript. SRM provided and created images. MMAG and AA conceptualized the idea and developed the outline for the review. MMAG, AA and ETC critically revised and edited the manuscript for submission in its final form. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
Not applicable.
Not applicable.
This paper is part of the requirements for obtaining a Doctoral Degree at the Posgrado en Ciencias Biológicas, UNAM, was funded by CONACyT fellowship (CVU 742855), the postdoctoral program of 2022–2023 of the DGAPA-UNAM, Facultad de Medicina, Universidad Nacional Autónoma de México, the Programa de Apoyo y Fomento a la Investigación Estudiantil (AFINES) of the research division of the UNAM Medical School, and DGAPA-PAPIIT (IN212422).
The authors declare research was conducted without any commercial or financial relationships construed as a potential conflict of interest. Amedeo Amedei is serving as one of the Editorial Board members of this journal. We declare that Amedeo Amedei had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Gernot Riedel.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


