1 Department of Cardiology, Zhongshan Hospital, Fudan University, Shanghai Institute of Cardiovascular Diseases, Key Laboratory of Viral Heart Diseases, National Health Commission, 200032 Shanghai, China
2 Department of Echocardiography, Zhongshan Hospital, Fudan University, Shanghai Institute of Cardiovascular Diseases, Shanghai Institute of Medical Imaging, 200032 Shanghai, China
3 Department of Urology, Shanghai General Hospital, Shanghai Jiao Tong University School of Medicine, 200080 Shanghai, China
Abstract
Background: The cardiotoxicity of doxorubicin (DOX) limits its use in
cancer treatment. To address this limitation, we developed a novel animal model
that uses beagle dogs to investigate DOX-induced cardiac disorders.
Unfortunately, the lack of effective cardioprotection strategies against
DOX-induced cardiotoxicity poses a significant challenge. To
establish a canine model for low-mortality DOX-induced cardiac dysfunction and
explore the relationship between inflammatory reprogramming and DOX-related
cardiotoxicity. Methods: Twenty male beagle dogs aged two years were
randomly assigned into the DOX (N = 10) and control (CON) (N = 10) groups. DOX
was infused (1.5 mg/kg) every two weeks until doses cumulatively reached 12
mg/kg. Serum biomarkers and myocardial pathology were evaluated, while real-time
fluorescence-based quantitative polymerase chain reaction (RTFQ-PCR), two- and
three-dimensional echocardiography (2DE and RT3DE), functional enrichment, and
matrix correlation were also performed. Results: In the DOX group,
high-sensitive cardiac troponin T (hs cTnT) and N-terminal pro-brain natriuretic
peptide (NT-proBNP) were significantly increased. Myocardial pathology indicated
early to medium myocardial degeneration via a decreased cardiomyocyte
cross-sectional area (CSA). Increased levels of inflammatory gene transcripts
(interleukin 6 (IL6), tumor necrosis factor (TNF), transforming growth factor
Keywords
- doxorubicin (DOX)
- inflammatory reprogramming
- cardiotoxicity
- cardio-oncology
- RT3DE
- beagle dog
Doxorubicin (DOX), a prototype agent of anthracycline, has been proven to be efficacious against a wide range of malignant neoplasms [1]. However, its dose-dependent chronic and irreversible cardiotoxicity [2] has limited its clinical application, thus, close surveillance should be applied to its use [3].
It is pivotal to establish an animal model that can be used to explore mechanisms and clinical strategies. Previous anthracycline-related toxicity models on small animals [4, 5, 6] were all inferior in reflecting echocardiographic and hemodynamic alterations since their hearts were too small to be accurately assessed. Beagle dogs can be monitored using the aforementioned indicators, and therefore, represent an improvement on previous methods [5, 7]. Thus, we established a simple and safe new model for DOX-induced cardiac disorder in beagle dogs.
Unfortunately, effective protections for DOX-induced cardiotoxicity are still lacking. Bisdioxopiperazine dexrazoxane (ICRF-187), an iron-once chelator, is the only approved cardio-protectant that targets anthracyclines-related congestive heart failure [8]; however, its use has been limited owing to its induction of secondary malignancies (SMNs) [9].
Inflammatory reprogramming is reportedly associated with myocardial remodeling, which in turn affects the cardiac function of a variety of cardiomyopathies [10, 11]. This previous finding prompted our study to explore the relationship between inflammatory reprogramming and DOX-induced cardiomyopathy in this new beagle dog model, to identify further mechanisms and treatments.
Twenty male beagle dogs aged two years, weighing 23–28 kg, were obtained from Jambo Biological Technology Co., Ltd (Shanghai, China). They were randomized using an online system (https://www.lifeguideonline.org) and placed into the DOX or control (CON) groups in a 1:1 ratio. Prior to commencing the experiment, pre-existing cardiac conditions were excluded in both groups, including pulmonary stenosis. All animals were individually housed in clean cages and were provided with free water and food. Indwelling needles (24G) were used to establish intravenous access through the medial cephalic vein of the forelimb for Telazol anesthesia (10 mg/kg, intravenously) and through the saphenous vein in the hind limb for DOX or 0.9% sodium chloride injection.
Owing to the fatal DOX-induced gastrointestinal bleeding (unpublished data: Supplementary Fig . 1), adequate attention was required to add gastrointestinal protective agents during DOX administration. In this protocol, a digestive tract protective mixed pharmaceutic preparation was applied for DOX hydrochloride (EnergyChemical, Shanghai, China), omeprazole sodium for injection (Luoxin Pharmaceutical, Shandong, China), and Bacillus licheniformis (Jingxin Pharmaceutical, Zhejiang, China). Subjects allocated to the DOX group received a 30 min intravenous infusion mixture of 1.5 mg/kg DOX and both 0.5 mg/kg omeprazole sodium and Bacillus licheniformis dissolved in 250 mL 0.9% sodium chloride. Synchronously, the control group received an injection of only 250 mL of 0.9% sodium chloride. Repeated infusion was performed every two weeks for a total of eight infusions (Fig. 1).
 Fig. 1.
Fig. 1.Study protocol of the new DOX-related cardiomyopathy beagle dog model. DOX, doxorubicin; iv., intravenous administration; q2w, every 2 weeks; 2DE, two-dimensional echocardiography; RT3DE, real-time three-dimensional echocardiography; hs cTnT, high-sensitive serum cardiac troponin T; NT-proBNP, N-terminal pro-brain natriuretic peptide; N = 10 dogs per group.
All animals participated in the study with signed owner consent and the protocols were approved by the Animal Ethics Committee of Zhongshan Hospital, Fudan University.
Whole blood samples (15 to 25 mL) were drawn from the saphenous vein of the hind limb before anesthesia at weeks 0, 12, and 28 in both groups, left to stand at room temperature for 2 hours, and then centrifuged at 1000 g for 20 minutes to collect the serum. The serum levels of high-sensitive cardiac troponin T (hs cTnT) and N-terminal pro-brain natriuretic peptide (NT-proBNP) were determined to assess cardiomyocyte damage and myocardial stretch, respectively, using available enzyme-linked immunosorbent assay (ELISA) kits (Ze Ye Biological Technology Co., Ltd., Shanghai, China). Optical density (OD) values were measured for each sample at a wavelength of 490 nm (for hs cTnT) or 450 nm (for NT-proBNP) using an enzyme immunoassay analyzer (BioTek, Winooski, VT, USA).
The dogs were sacrificed at week 28. The hearts were perfused with
phosphate-buffered saline (PBS, Hyclone, Logan, UT, USA) and then isolated to
measure their weight. To collect the pathological specimen, myocardial tissue
blocks of 5 mm
Functionally annotated enrichment analysis was conducted using the Metascape (https://metascape.org/gp/index.html) database [12]. Sangerbox (http://sangerbox.com/about.html) platform was used to analyze and create transcript chord plots and matrix correlation heat maps [13]. Spatial transcriptome and single nucleus RNA sequencing (snRNA SEQ) data were obtained via the STOmicsDB (https://db.cngb.org/stomics/) assets [14].
To extract mRNA from cells or heart tissue, we used RNAiso Plus#9109 (Takara Bio, Kusatsu, Japan). Next, RNA was reverse transcribed into cDNA using the PrimeScript™ reverse transcription kit# RR036A (Takara Bio, Kusatsu, Japan). The RTFQ-PCR kit (Applied Biosystems, Foster City, CA, USA) was used for the destination analysis. RTFQ-PCR was performed by an ABI 7300 (Thermo Fisher Scientific, Waltham, MA, USA) detection system using the SYBR Green PCR detection method. We prepared 20 µL RTFQ-PCR reaction mixture containing 10 µL qPCR SYBR Green Master Mix (Takara Bio, Kusatsu, Japan), 0.4 µL F primer, 0.4 µL R primer, 1 µL cDNA template, and 1 µL sterile deionized water. The reaction cycle conditions were 95 °C for 30 seconds followed by 40 cycles of 95 °C for 10 seconds and 60 °C for 30 seconds.
Two-dimensional echocardiography was performed using an iE33 echocardiographic system (Philips Medical Systems, Andover, MA, USA) with an S5-1 transducer (1–5 MHz) at baseline (week 0) in both groups, then again after the second infusion (week 4), the fourth infusion (week 8), the sixth infusion (week 12), the eighth infusion (week 16), and three months after the eighth infusion (week 28) (Fig. 1). Two-dimensional echocardiograms were acquired from the parasternal and apical views to monitor regional myocardial function. The transverse diameter of the right ventricular basal segment (RVD1), middle segment (RVD2), and apical segment (RVD3) were collected for analysis. Tricuspid annular plane systolic excursion (TAPSE) was measured by M-mode tracings from the apical four-chamber view. Moreover, mitral inflow was recorded by pulsed-wave Doppler to measure peak velocities of early (E) and late (A) filling; mitral annular early (E’) and late (A’) peak velocities were determined from tissue Doppler imaging.
After examining the 2DE, RT3DE was performed in both groups using the iE33 echocardiographic system equipped with an X3-1 transducer (1–3 MHz). Standard apical four-chamber views were recorded and 3D full-volume dynamic images of three consecutive cardiac cycles were stored for offline analysis by TomTec 4D LV (4.6.0.411, TomTec Imaging Systems GMBH, Unterschleißheim, Germany). The software semiautomatically traced the endocardial contours of the LV and RV at end-diastole and end-systole and allowed for manual modification when needed. The right ventricular end-diastolic volume (RVEDV), right ventricular end-systolic volume (RVESV), right ventricular ejection fraction (RVEF), right ventricular longitudinal free-wall strain (RVLFS), right ventricular longitudinal septal strain (RVLSS), left ventricular end-diastolic volume (LVEDV), left ventricular end-systolic volume (LVESV), left ventricular ejection fraction (LVEF), left ventricular global longitudinal strain (LVGLS) and left ventricular global circumferential strain (LVGCS) were calculated.
Data are expressed as mean
The percentage variation was calculated using the following formula:
Statistical significance was determined using SPSS (version 25.0, IBM Corp.,
Chicago, IL, USA) by paired Student’s t-test or one-way ANOVA with
Tukey’s post hoc test after applying Benjamini–Hochberg correction for numerical
variables. One-way repeated measures ANOVA with Tukey’s post hoc test was used in
analyzing related groups data. p values and correlation coefficient were
calculated using Spearman correlation analysis. GraphPad Prism (version 7.02 for
Windows, GraphPad Software, Inc., La Jolla, CA, USA) was utilized to generate the
statistical graph. A p value
The CON group experienced no adverse effects during the whole process.
Conversely, three out of ten beagle dogs in the DOX group experienced three to
four days of appetite loss and hematochezia after the fourth injection, although
they recovered after receiving a soft and low-residue diet and this situation did
not affect the fifth injection. To more effectively avoid gastrointestinal
hemorrhage, all animals were fasted for 1 day before the following injections and
all of them were provided soft and low-residue diets following the injections.
All the beagle dogs survived to the end of the experiment without any significant
change in weight (vs. week 0 or vs. the CON group at each follow-up, all
p
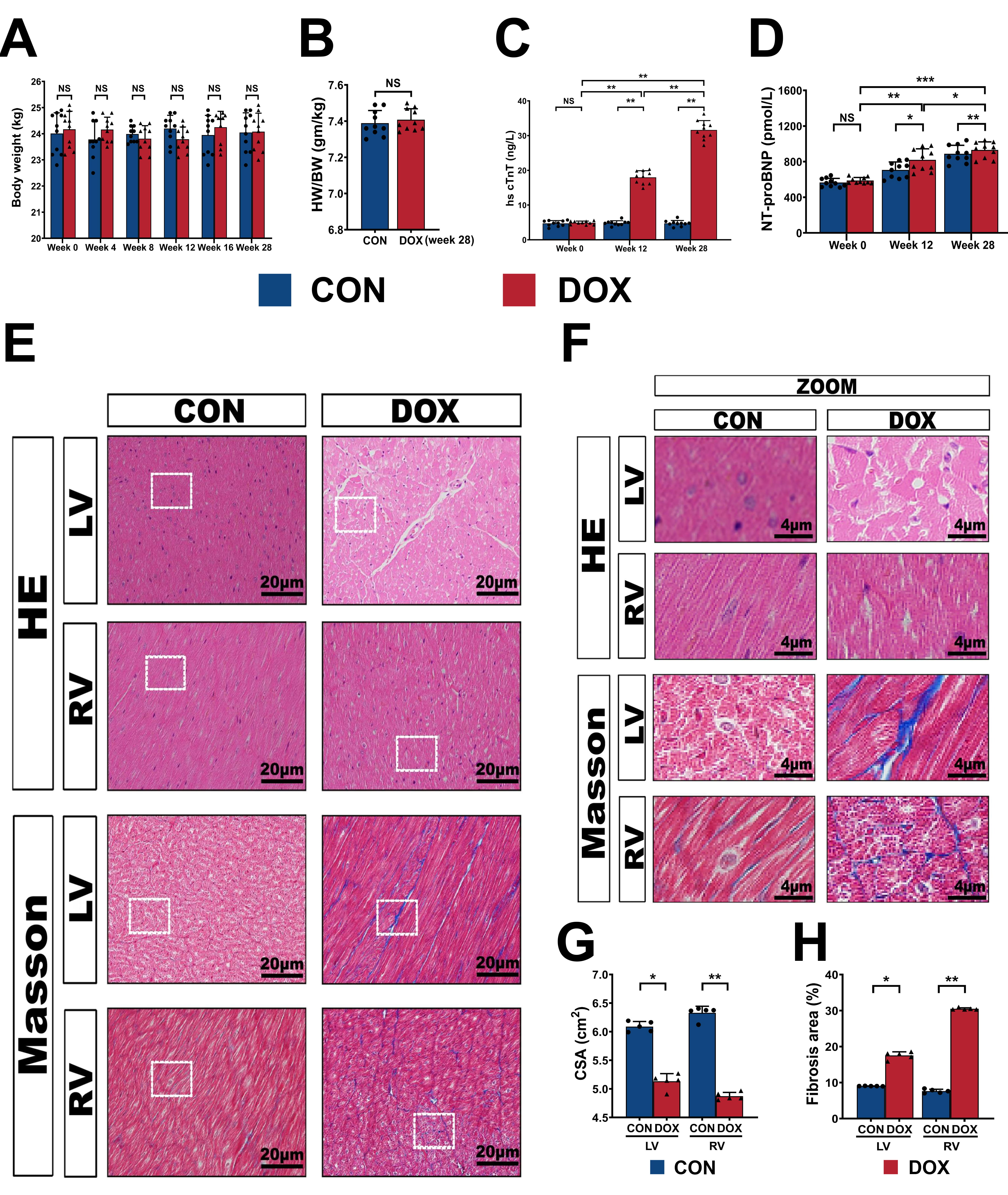 Fig. 2.
Fig. 2.General characteristics of the novel beagle dog model. (A,B) Body weight and heart weight/body weight ratio before and after DOX infusion.
(C,D) Serum hs cTnT and NT-proBNP levels before and after DOX infusion. (E)
Representative HE and Masson’s trichrome staining light micrographs (400
amplification) from CON and DOX groups. (F) Magnified regions from (E). (G,H) Quantification of the cross-sectional area and the area percentages of
fibrosis. CON, control group; DOX, doxorubicin group; HW/BW, heart weight/body
weight; hs cTnT, high-sensitive serum cardiac troponin T; NT-proBNP, N-terminal
pro-brain natriuretic peptide; LV, left ventricle; RV, right ventricle; HE,
Hematoxylin–eosin staining; Masson, Masson’s trichrome staining; CSA,
cross-sectional area; ZOOM, magnified regions. ⚫ represents data from
CON group; ▲ represents data from DOX group. Values are expressed as the
mean
To assess the effect of DOX-induced myocardial injury and heart failure, we
examined the expression of hs cTnT and NT-proBNP in the serum at weeks 0, 12, and
28. Compared with week 0 (4.92
Necropsy examinations (Fig. 2E) and regional magnification images (Fig. 2F) in the DOX group showed moderate myocardial disease. The degenerative changes observed in the HE staining were characterized by the early to medium multifocal invasion of the myocardium, generation with evaporation of myocardial fibers, areas of fiber disruptors, and scattered inflammatory cell infiltration (Fig. 2F). Similarly, CSA of RV and LV were strongly decreased in the DOX group compared to the CON group (Fig. 2G). Furthermore, DOX caused a marked increase in collagen deposits under Masson’s trichrome staining, which were observed predominantly via the fibrosis of the interstitium in the cardiomyocytes (Fig. 2E,F). In the DOX group, the percentage areas of fibrosis in both the RV and LV increased significantly compared to the control group (Fig. 2H), thereby implying that the myocardium was in the medium stage of the DOX-induced pathological changes.
Due to the absence of spatio-temporalomic data concerning DOX-induced injuries in beagles, we obtained spatial transcriptome data linked to the protective and restorative effects of Mus musculus hearts in response to injury via the STOmicsDB (https://db.cngb.org/stomics/) assets, from which we extracted significant single nucleus RNA sequencing (snRNA SEQ) information for further analysis (https://db.cngb.org/stomics/datasets/STDS0000110).
The transcript chord plot and the enrichment of the related gene pathways (Fig. 3A,B) suggested that changes are concentrated primarily in multiple enzyme-linked
receptor protein signaling pathways and collagen metabolism during the process of
coping with injury. This observation led us to hypothesize that the interaction
between doxorubicin toxicity and myocardial stress could regulate myocardial
inflammatory programming and induce various signal pathway imbalances, leading to
subsequent collagen metabolism disorders. To assess this hypothesis, we used
RTFQ-PCR to measure the transcript levels of hallmark inflammatory genes in the
DOX group, including interleukin 6 (IL6) and tumor necrosis factor-alpha
(TNF
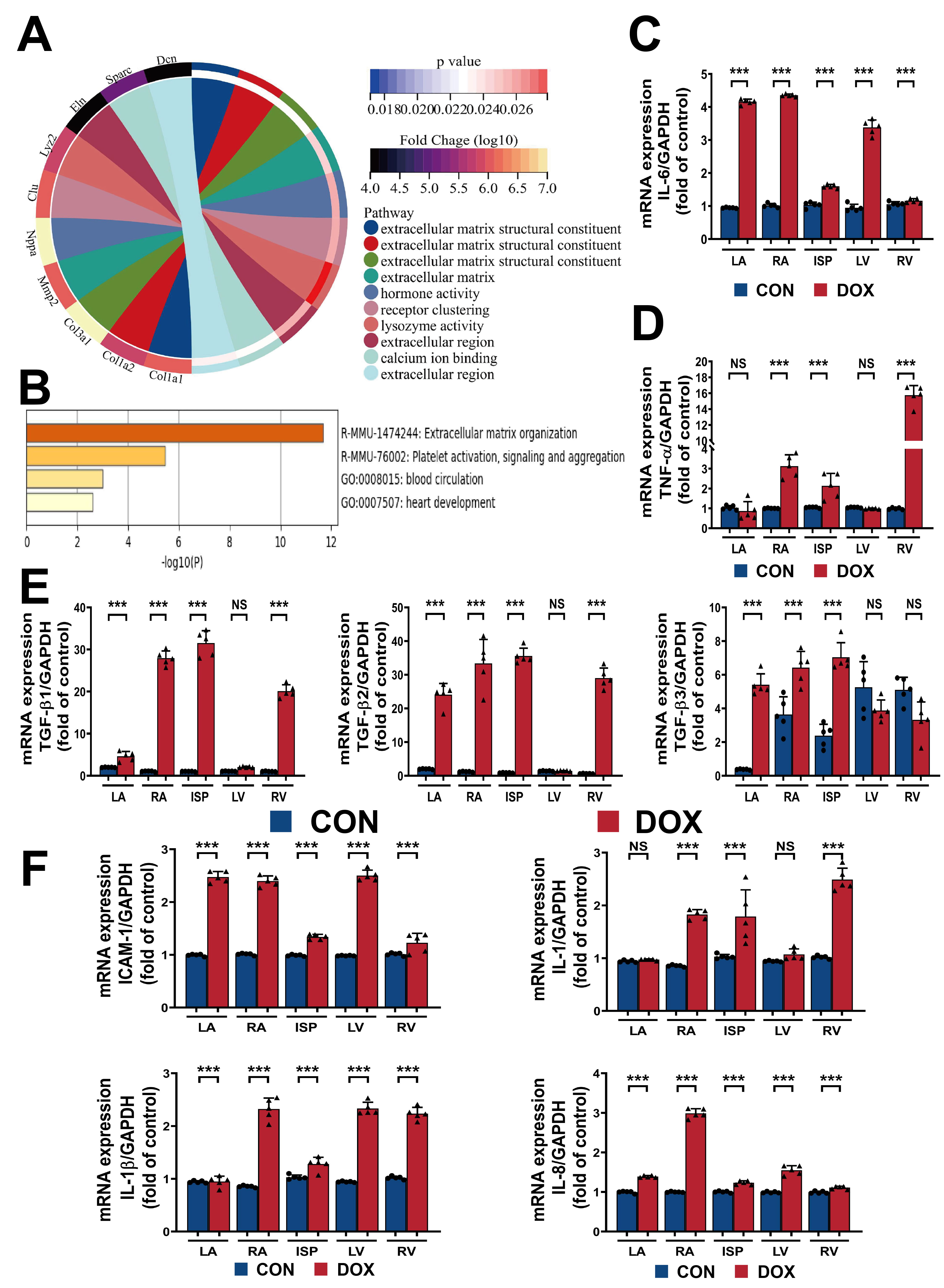 Fig. 3.
Fig. 3.Doxorubicin toxicity triggers inflammatory reprogramming in the
myocardium. (A,B) The transcript chord plot and the pathway enrichment
adapted from STOmicsDB assets and Metascape database. (C,D) Quantification
of IL-6 and TNF-
In the DOX group, HE and Masson’s trichrome staining revealed cardiomyocyte
structural disorder, patchy interstitial inflammatory infiltration, and obvious
interstitial collagen deposition in various parts of the myocardium (Fig. 4A).
The statistical results confirmed the occurrence of myocardial structural
remodeling under the effect of informatory reprogramming. Specifically, the CSA
in the DOX group cardiomyocytes decreased significantly at all points in the
heart, and the percentage area of fibrosis increased significantly (all
p
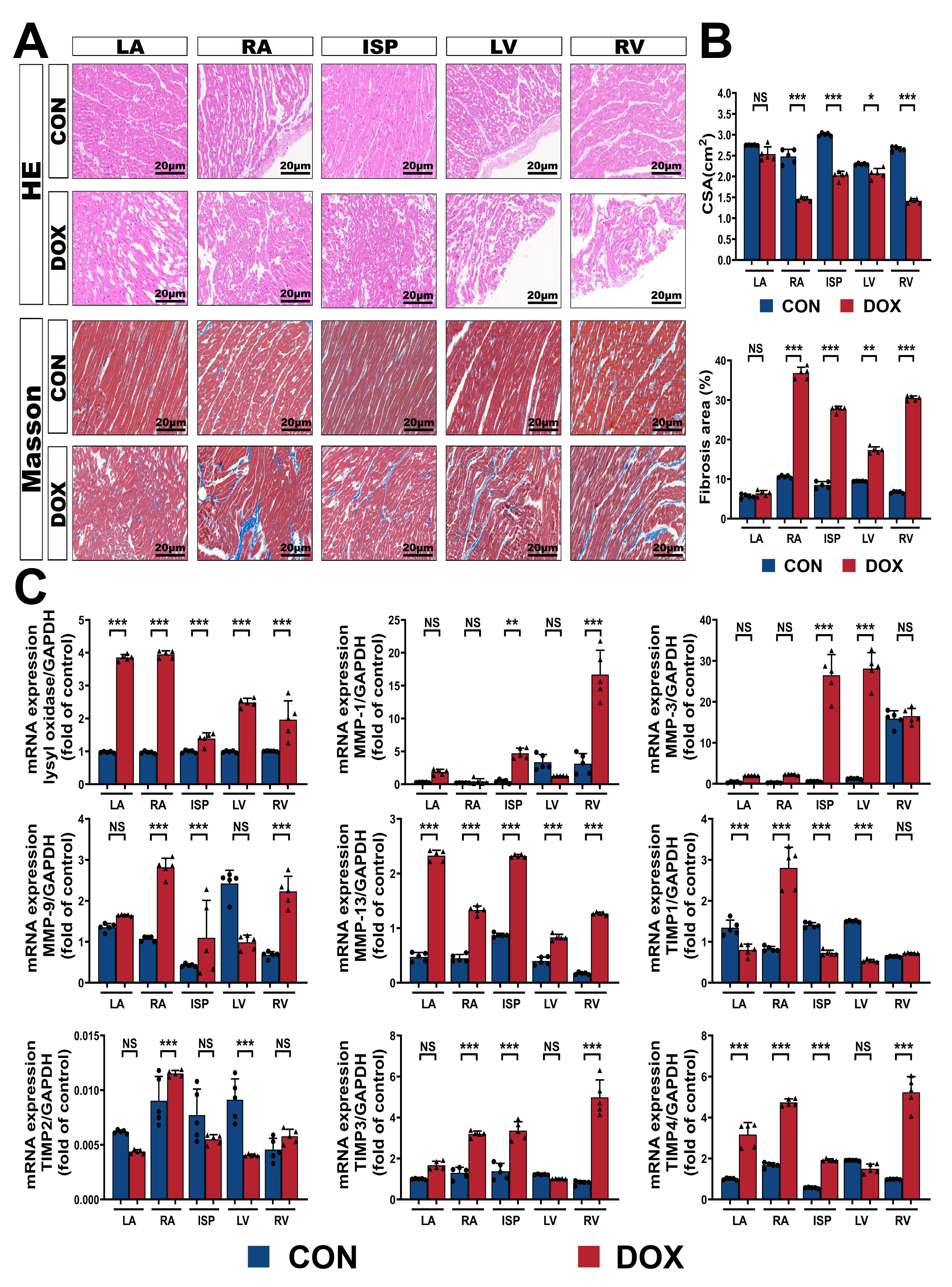 Fig. 4.
Fig. 4.Inflammatory reprogramming activation contributes to myocardial
remodeling in DOX-related cardiomyopathy. (A) Representative HE and Masson’s
trichrome staining light micrographs (400 amplification) from CON and DOX groups.
(B) Quantification of the CSA and the percentage areas of fibrosis from (A). (C)
Quantification of lysyl oxidase, MMP family, and TIMP family transcript levels
normalized to GAPDH in dog heart sections. HE, Hematoxylin–eosin staining;
Masson, Masson’s trichrome staining; CON, control group; DOX, doxorubicin group;
LV, left ventricle; RV, right ventricle; ISP, interventricular septum; LA, left
atrium; RA, right atrium; CSA, cross-sectional area; GAPDH,
Glyceraldehyde-3-phosphate dehydrogenase; MMP-1, matrix metalloproteinase 1; MMP-3,
matrix metalloproteinase 3; MMP-9, matrix metalloproteinase 9; MMP-13, matrix
metalloproteinase 13; TIMP-1, tissue inhibitor of matrix metalloproteinase 1;
TIMP-2, tissue inhibitor of matrix metalloproteinase 2; TIMP-3, tissue inhibitor of
matrix metalloproteinase 3; TIMP-4, tissue inhibitor of matrix metalloproteinase
4. Values are expressed as the mean
Inflammatory cascades, fibrosis, and myocardial remodeling frequently cause changes in ventricular and circulatory capacity, leading to a close association with the natriuretic peptides family (NPS). This family plays a vital role in natriuretic, diuretic, vasodilator, anti-sympathetic, and renin–aldosterone inhibiting activities. Under this premise, we detected the natriuretic peptide A (NPPA) transcription levels in this family, as well as natriuretic peptide B (NPPB) and natriuretic peptide (NPPC), which play similar roles as key markers of ventricular volume load change in response to inflammation and fibrosis. Unlike inflammatory reprogramming, which was widely upregulated in almost all parts of the heart, the NPPA, NPPB, and NPPC transcript levels tended to change significantly for the right side and interventricular septum (Fig. 5A), suggesting that further effects of myocardial remodeling varied in different parts of the heart. To judge the impact of DOX on typical parts in terms of inflammation, collagen metabolism, and volume load more intuitively and comprehensively, we analyzed their multidimensional relationships by thermogram. It could be seen that DOX-related myocardial toxicity and changes appeared to play a more important role in the right ventricle, followed by the interventricular septum, whereas there were only a few significant changes in the left heart (Fig. 5B).
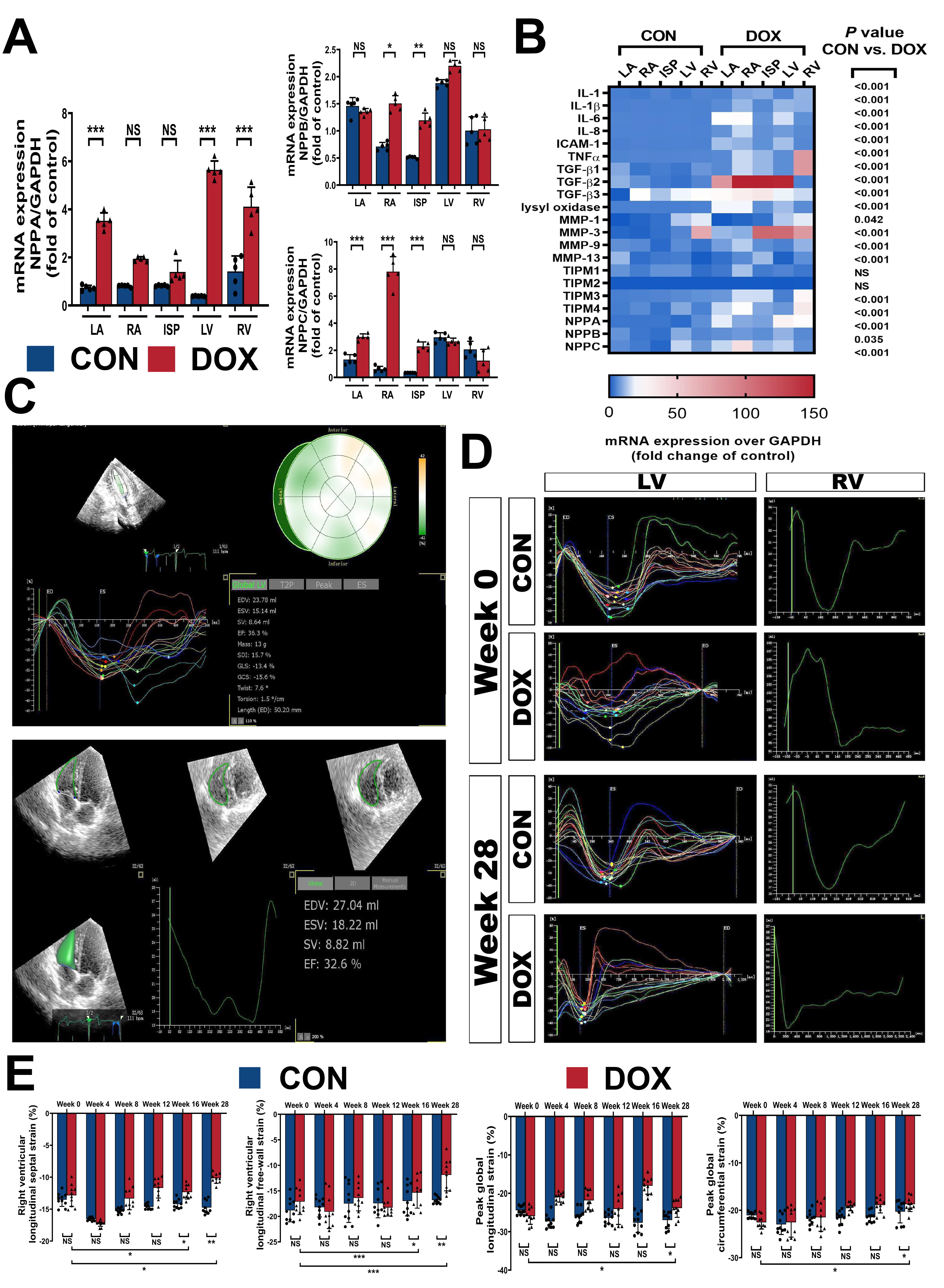 Fig. 5.
Fig. 5.Myocardial remodeling and volume changes influence the 3D strain
capacity. (A) Quantification of NPPA, NPPB, and NPPC transcript levels
normalized to GAPDH in dog heart sections. (B) Heat map of inflammation, collagen
metabolism, and volume load in the right- or left side of the heart in the CON
and DOX groups. (C) Examples of 3D echocardiographic studies for the LV (above)
and RV (below) strain parameters using TomTec offline analysis software. (D)
Three-dimensional echocardiographic studies of LV and RV strain curves at
baseline (week 0) and endpoint (week 28) using TomTec offline analysis software.
(E) Quantification of 3D echocardiographic parameters for RVLSS, RVLFS, LVGLS,
and LVGCS. LV, left ventricle; RV, right ventricle; ISP, interventricular septum;
LA, left atrium; RA, right atrium; NPPA, natriuretic peptide A; NPPB, natriuretic
peptide B; NPPC, natriuretic peptide; GAPDH, Glyceraldehyde-3-phosphate
dehydrogenase; CON, control group; DOX, doxorubicin group; IL1, interleukin 1;
IL1
The three-dimensional speckle tracking method could more accurately and dynamically reconstruct the cardiac chamber structure and has irreplaceable advantages in evaluating the strain capacity and cardiac function of large animals. Therefore, we used three-dimensional echocardiography to record the typical endocardial map of the left and right ventriculares during the cardiac cycle and combined it with the corresponding interpolation function to calculate the fitting curve and key parameters (Fig. 5C). Fig. 5D illustrates the standard left and right ventricular strain curves in the control and DOX groups at baseline (week 0) and endpoint (week 28).
Significant strain abnormalities were observed in the RVLSS, RVLFS, LVGLS, and
LVGCS at week 28 (vs. week 0 or CON group, p
Hence, a correlation between myocardial remodeling, volume load adjustments, and alterations in ventricular strain capacity could be detected. Over the observation period, the cardiac damage caused by DOX led to modifications in the three-dimensional cardiac strain capacity of beagles, notably in the right ventricle.
Previous literature has reported a comprehensive regulatory relationship between myocardial remodeling and strain disorder that can mutually reinforce and exacerbate circulatory challenges [15], both of which significantly impact cardiac function. Given the evidence of remodeling and strain abnormalities, as well as their potential connections in a canine model, we aimed to investigate their effect on cardiac function. Specifically, the investigation aimed to determine whether remodeling and strain abnormalities correlated to changes in cardiac function, which is critical in understanding the clinical features of doxorubicin cardiomyopathy. The important parameters of left and right ventricular systolic function were measured and analyzed from week 0 to week 28 using Tomtec software (Fig. 6A).
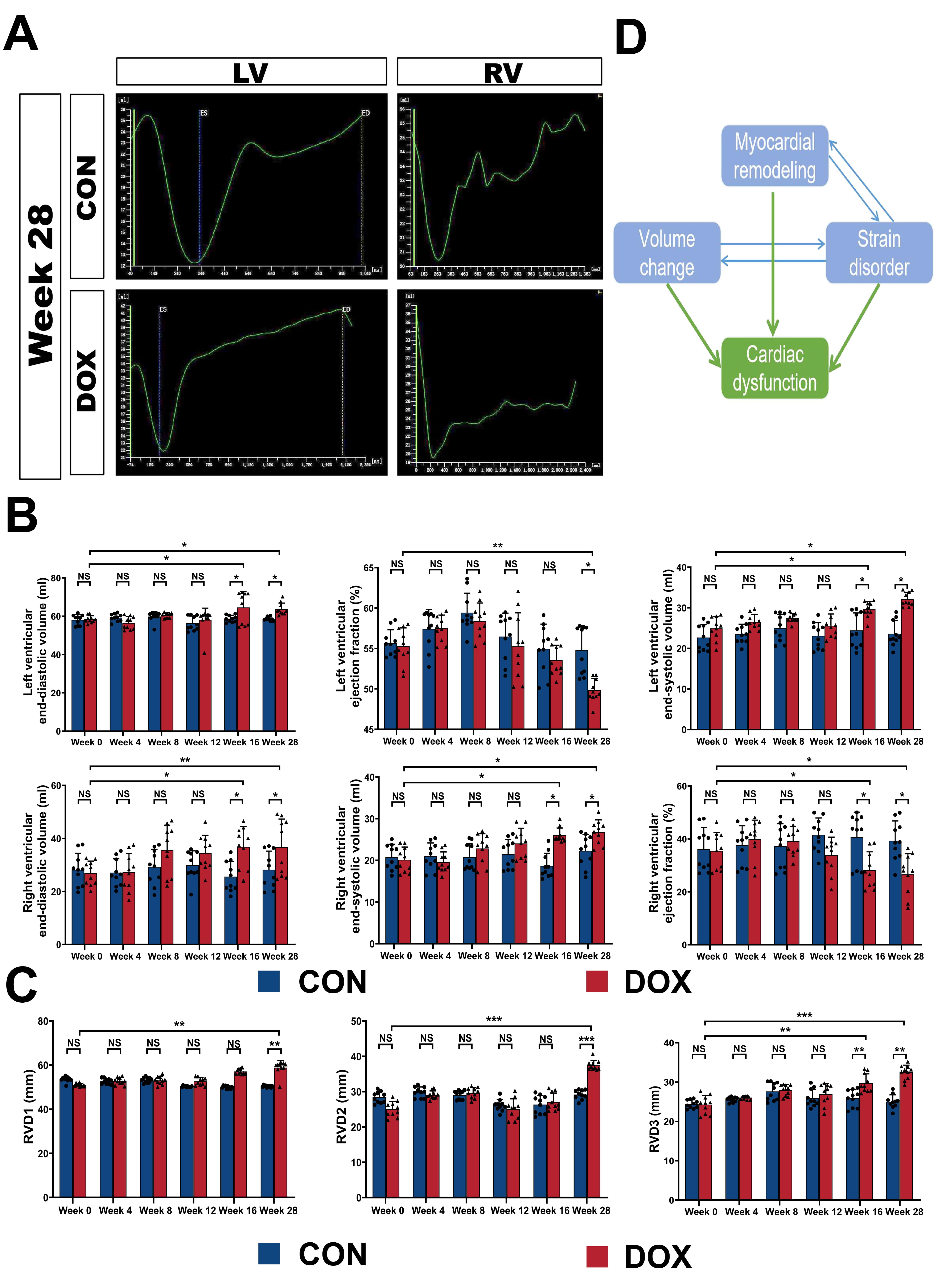 Fig. 6.
Fig. 6.Myocardial remodeling and strain disorders influence the 3D
cardiac function. (A) Examples of 3D echocardiographic studies of LV and RV
function curves using TomTec offline analysis software. (B) Quantification of 3D
echocardiographic parameters for LVEDV, LVESV, LVEF, RVEDV, RVESV, and RVEF in
the CON and DOX groups. (C) Quantification of 3D echocardiographic parameters for
transition diameters in the right ventricular basal (RVD1), middle (RVD2), and
apical (RVD3) segments in the CON and DOX groups. (D) The relationship among
myocardial remodeling, volume load, strain disorder, and cardiac dysfunction. LV,
left ventricle; RV, right ventricle; CON, control group; DOX, doxorubicin group;
LVEDV, left ventricular end-diastolic volume; LVESV, left ventricular
end-systolic volume; LVEF, left ventricular ejection fraction; RVEDV, right
ventricular end-diastolic volume; RVESV, right ventricular end-systolic volume;
RVEF, right ventricular ejection fraction; RVD1, transition diameters of the
right ventricular basal segment; RVD2, transition diameters of the right
ventricular middle segment; RVD3, transition diameters of the right ventricular
apical segment. N = 10 dogs per group. Values are expressed as the mean
Compared to week 0 or for each in the CON group, both of RVEDV and RVESV were
considerably increased at week 16 and persisted until week 28 (all p
The transition diameters of the right ventricular basal (RVD1), middle (RVD2),
and apical (RVD3) segments were also reflective of the lateral systolic function
by the right heart, thereby exhibiting a similar trend. The results demonstrated
that RVD3 increased significantly from week 16 (vs. control group, p
In the present study, the LV diastolic function and tricuspid annular plane
systolic excursion (TAPSE) were explored via 2D Doppler echocardiography. In
Table 1, no significant change was found in E, A, E’, and A’ (p
| Variables | Week 0 | Week 4 | Week 8 | Week 12 | Week 16 | Week 28 | ||||||
| CON | DOX | CON | DOX | CON | DOX | CON | DOX | CON | DOX | CON | DOX | |
| E (cm/s) | 51.87 |
52.69 |
52.79 |
53.46 |
49.05 |
48.96 |
53.90 |
55.14 |
58.92 |
58.70 |
56.42 |
55.30 |
| A (cm/s) | 54.20 |
53.08 |
51.01 |
50.35 |
46.83 |
47.06 |
49.91 |
50.75 |
52.19 |
50.64 |
55.92 |
55.50 |
| E’ (cm/s) | 11.70 |
12.57 |
11.27 |
10.74 |
11.15 |
10.63 |
10.32 |
9.95 |
11.08 |
10.76 |
11.86 |
11.11 |
| A’ (cm/s) | 16.43 |
16.04 |
14.83 |
15.40 |
15.46 |
14.48 |
13.63 |
14.71 |
13.12 |
13.71 |
16.93 |
17.36 |
| E/A ratio | 1.47 |
1.05 |
1.29 |
1.13 |
1.15 |
1.13 |
1.21 |
1.14 |
1.19 |
1.24 |
1.10 |
1.04 |
| E/E’ ratio | 5.18 |
4.72 |
4.96 |
5.09 |
5.03 |
4.87 |
5.37 |
5.73 |
5.91 |
6.06 |
5.86 |
5.16 |
| TAPSE (mm) | 19.80 |
20.90 |
22.10 |
21.40 |
22.30 |
21.70 |
20.70 |
20.50 |
19.60 |
19.80 |
21.70 |
21.10 |
Data are expressed as the mean
Collectively, these findings suggest that during the observation period, there is an alteration in cardiac function indicators in response to doxorubicin. The transversal systolic function had been affected initially, primarily on the right ventricle. Effects on diastolic and longitudinal systolic functions were relatively small or might occur later.
Considering the comprehensive relationship between myocardial remodeling, strain, and cardiac function, we proposed the following hypothesis: In adriamycin cardiomyopathy, the remodeling of the myocardium itself and the change in the volume load of the chamber jointly led to the change in myocardial strain capacity, while the strain disorder further aggravated the process of myocardial remodeling and the increase in the volume load. The interaction of the two finally contributed to the imbalance in cardiac function (Fig. 6D).
Although the above results provided compelling evidence that the effects on
cardiac strain and function by DOX relied on subsequent cascade reactions in
inflammatory reprogramming, the correlation between them lacked statistical
support, meaning it needs to be further explored in a multidimensional context.
Thus, we investigated the relationship between reprogramming-related transcript
changes, 3D strain capacity, and cardiac function, which were visualized by
correlation matrix thermograms (Figs. 7A,6B). The gram between
inflammatory reprogramming and strain capacity revealed that, firstly, the right
ventricular strain damages strongly correlated with inflammatory reprogramming
(most Spearman’s coefficients
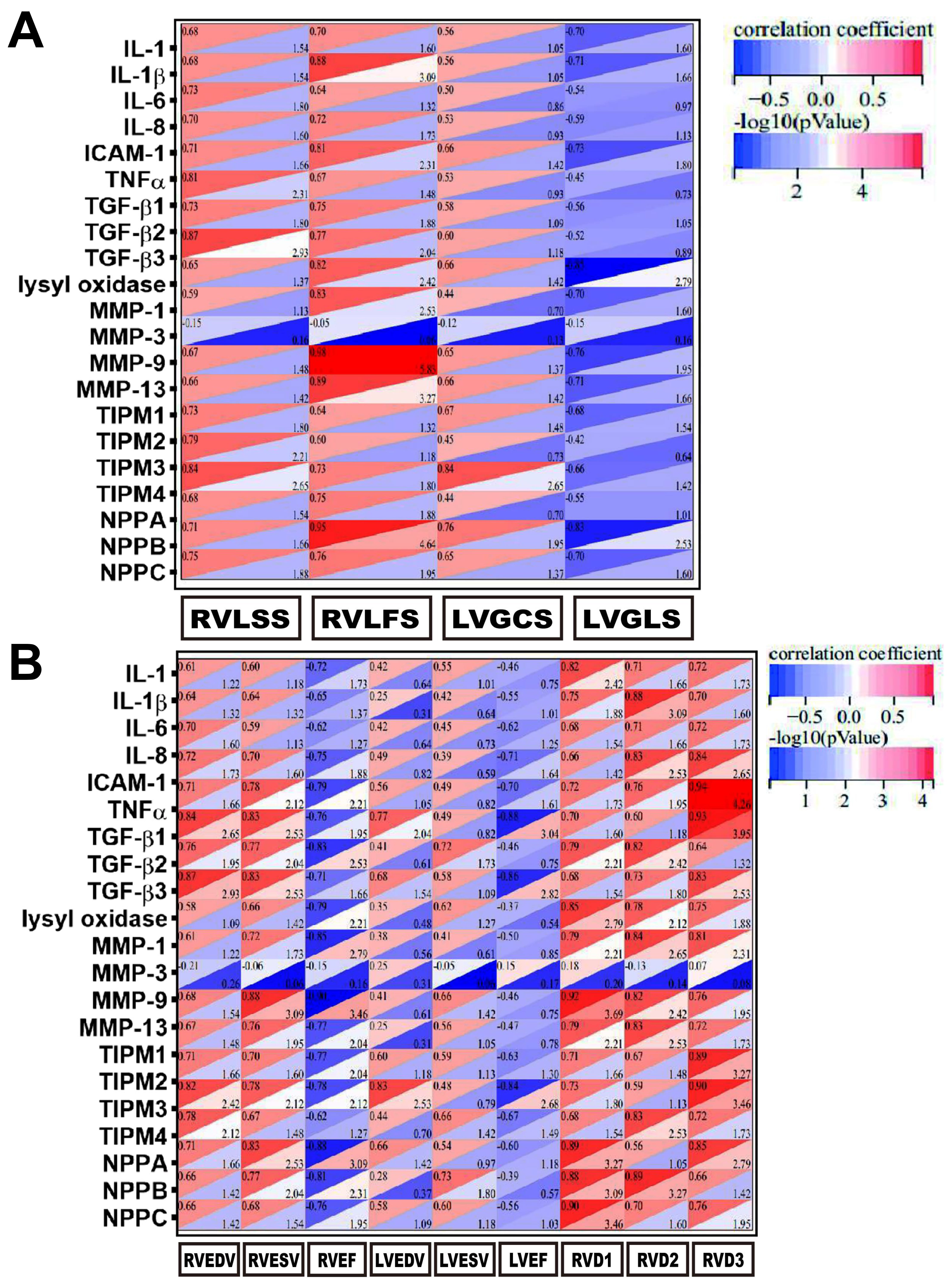 Fig. 7.
Fig. 7.Statistical correlation between inflammatory reprogramming,
strain capacity, and cardiac function. (A) Correlation matrix thermograms
between inflammatory reprogramming and strain disorders. (B) Correlation matrix
thermograms between inflammatory reprogramming and cardiac dysfunction. IL1,
interleukin 1; IL1
The disease process in the novel DOX-induced cardiomyopathy beagle dog model has been summarized in Fig. 8. Doxorubicin myocardial toxicity extensively activated and upregulated the inflammatory reprogramming process, causing myocardial remodeling and changes in cardiac volume load. Under dual pressure, myocardial strain capacity became disorderly, resulting in a disrupted state of cardiac function, which finally cascaded into functional imbalance.
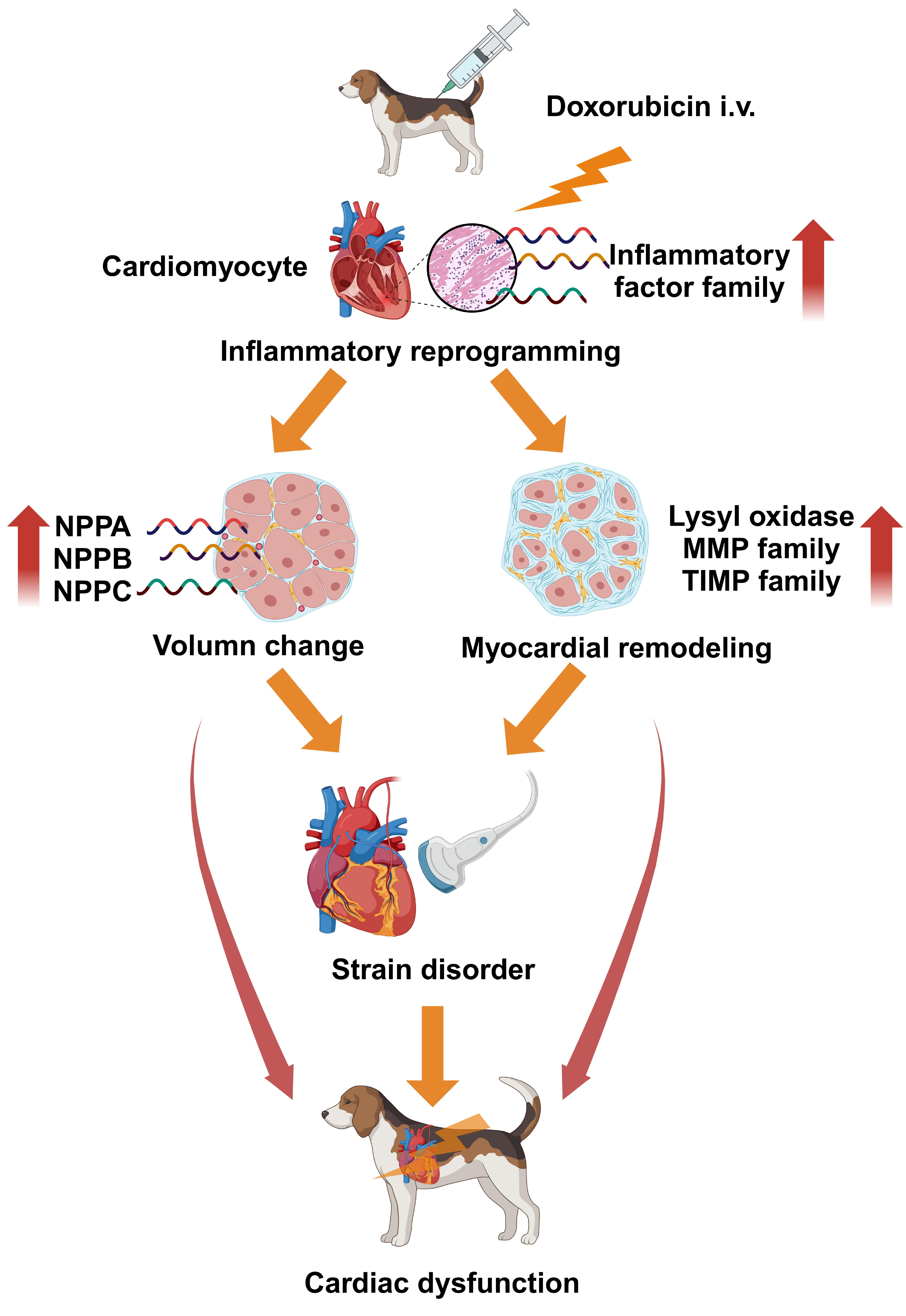 Fig. 8.
Fig. 8.Inflammatory reprogramming mediated disorders in strain capacity and cardiac function in the DOX-related cardiomyopathy beagle dog model. (created with https://www.biorender.com/). i.v., intravenous administration; NPPA, natriuretic peptide A; NPPB, natriuretic peptide B; NPPC, natriuretic peptide; MMP, matrix metalloproteinase; TIMP, tissue inhibitor of matrix metalloproteinase; DOX, doxorubicin.
DOX still plays a crucial role in tumor treatment, despite it causing long-term and serious side effects, via myocardial injuries [1]. Currently, there is no consensus as to the mechanism related to the onset of myocardial injury owing to the lack of any accurate experimental models [7, 16]. Therefore, cardio-oncology calls for an efficient and stable new model to assist clinical exploration.
Due to the obscurity and refractoriness of congestive heart failure caused by DOX, precise and systematic early monitoring is required, with techniques such as echocardiography, magnetic resonance imaging (MRI), and single-photon emission-computed tomography (SPECT) [5, 6]. However, using small animal models, such as mice, rats, and rabbits, to measure these indicators is limited due to the significant structural deviations between their hearts and humans [2, 17, 18]. Beagle dogs, on the other hand, are considered a more suitable model because their heart characteristics are similar to humans.
However, the currently utilized beagle model, which involves DOX being infused into the coronary artery, requires surgical methods, such as bilateral thoracotomy [19], or the insertion of a femoral artery catheter using a fluoroscope [7]. Both operation techniques require a high level of skill and have mortality rates that exceed 40%. Intravenous injection seems to be a more convenient and practical method. Inspired by the study by Cheng et al. [20], our final plan administered only 1.5 mg/kg of DOX every two weeks, at a concentration of 0.4 mg/mL, with an injection duration of 30 to 45 minutes. Additionally, omeprazole and Bacillus were added at a 1:1 ratio as gastrointestinal protective agents to prevent the fatal gastrointestinal bleeding caused by DOX, which was observed in our previous experiments (unpublished data: Supplementary Fig. 1).
Our new protocol has also led to a low-mortality DOX-induced cardiotoxicity beagle dog model, characterized by cardiac dysfunction, right and left ventricular strain impairment, and myocardial disorders.
Multiple indicators were used during the model evaluation. An example is hs cTnT, which can predict myocardial injury in clinical practice [21, 22]. Similar to the findings of Cove-Smith et al. [23], our study observed an increase in hs cTnT levels that correlated positively with DOX after a cumulative dose of 9 mg/kg and preceded the detection of echocardiographic abnormalities, which were present only in doses higher than 12 mg/kg. Similar trends were observed in NT-proBNP levels, although the magnitudes of changes were comparatively smaller. This finding might be partially due to a delayed response of NT-proBNP to changes in cardiac volume and pressure resulting from the self-compensatory effects seen in the early-to-mid stages of cardiac dysfunction [24].
Our study suggested that DOX triggers inflammatory reprogramming, resulting in a proinflammatory state and myocardial remodeling. By referring to the spatial transcriptome from mice coping with injury-induced stress, the main upregulated genes were involved in collagen metabolism and enzyme-linked receptor protein signaling pathways, we assumed that broad signaling disorders can lead to unbalanced collagen metabolism.
The present experiment demonstrated an extensive upregulation in proinflammatory
factors, including IL6 and TNF
Myocardial remodeling and changes in volume load can result in alterations to the three-dimensional strain capacity. By heat mapping all inflammation, fibrosis, and brain natriuretic peptide transcripts, we found that transcriptional upregulation tended to be more pronounced in the right heart than in the left. Since structural disorders are often accompanied by functional alterations, we posited that misalignments in echocardiographic indicators may arise in respective spatial locations.
RT3DE has been proven to yield plentiful information on cardiac strain parameters as well as provide more accurate evaluations of LVEF and RVEF than 2DE [25]. However, in terms of ventricular strain, it is noteworthy that RVLFS and RVLSS began to decline as early as week 16; earlier than LVGLS and LVGCS, which declined at week 28. The asynchronous changes in strain may be due in part to the thickness of the ventricular wall, which affects wall stress. According to Laplace’s law, wall stress increases with pressure and radius but decreases with wall thickness [26, 27]. Considering that the right ventricular wall is thinner than the left, it experiences greater wall stress and appears more susceptible to spatial strain degradation.
Myocardial remodeling is a factor that affects cardiac function by altering the myocardial fiber properties during systole and diastole [10], whereas strain disorder disrupts the balance of the regulatory effects, which are multi-segmental and multi-temporal [28]. In terms of systolic function, LVEF remained relatively stable over the initial 16-week period but experienced a significant and sustained decline from week 16 to week 28. In contrast, RVEF demonstrated more significant DOX-induced impairment at an earlier time point (week 16), with a higher percentage variation (13.27% for RVEF vs. 5.26% for LVEF at week 28). To further support this finding, the right ventricular systolic function was assessed using the transverse diameters of the right ventricular basal, middle, and apical segments (RVD1, RVD2, and RVD3), all of which were notably higher than the baseline values and indicative of myocardial toxicity at an early stage. Specifically, this interesting finding was novel in the canine model, as prior studies had not commonly observed early anomalies in the right ventricular systolic function. Prior research by Nagata et al. [29] proposed that contractile impairment could be compensated for until DOX exposure resulted in decompensated cardiotoxicity and symptomatic heart failure. It is hypothesized that the thinner right ventricular wall may provide fewer compensation reserves and yield earlier manifestations of reduced RVEF. In contrast, the indices for diastolic function did not exhibit significant changes during the follow-up period, implying that DOX-induced myocardial toxicity may have limited effects on these indicators, or they may experience a late-stage injury.
To investigate the correlation among strain capacity, cardiac function, inflammatory reprogramming, and myocardial remodeling, we utilized multiple correlation matrix thermograms to depict the relationship intuitively. Here, extensive inflammation and remodeling had a greater correlation to the right ventricular strain and function parameters. Moreover, we discovered that DOX toxicity had a minimal impact on the left and right ventricular longitudinal contractions or strain capacities, or that they might experience a time-lag effect. Hence, we need to prolong the follow-up to observe any long-term changes.
In summary, our study reviews the critical role of inflammatory reprogramming in promoting changes to the three-dimensional strain capacity and cardiac function in beagle dogs during the development of DOX-related cardiomyopathy.
One limitation of this study was that the potential late-occurring diastolic and TAPSE abnormalities by DOX were not evaluated. A previous study showed that impaired diastolic function occurred in 60% of breast cancer participants treated with DOX by 1 year, 70% by 2 years, and 80% by 3 years [30], thereby demonstrating that diastolic dysfunction develops gradually over time and TAPSE abnormality might also be delayed. For that reason, follow-ups of over 1 year are also needed for the beagle dog model in the future to ensure that myocardial stiffness reaches the degree where it can be detected by echocardiography.
Another limitation to consider is that all myocardial pathological changes in the study are still in the early to medium stages. Considering this, follow-up experiments can use increased dosages and extend the course of DOX administration to discover more evident variations in the phenotype and their relationship with inflammatory reprogramming.
We have successfully established a stable DOX-induced myocardial toxicity canine model with low mortality, which was characterized by increased hs cTnT and NT-proBNP levels alongside predominantly right ventricular systolic and strain dysfunction. Inflammatory reprogramming is believed to be one of the initial causes during the myocytic injury process. Therefore, our findings provide a valuable foundation for further research on the mechanisms involved. Ultimately, such initiatives are essential for ensuring humane animal treatment in research settings.
All data points generated or analyzed during this study are included in this article and there are no further underlying data necessary to reproduce the results.
YC and XW designed animal experiments, conducted animal modeling and evaluating, collected data and write the first draft and revised the article and contributed equally in the works above, they took public responsibility for the content. RZ, YS, HZ, XW, YX and JZ assisted in conducting animal experiments, animal parameter evaluation and statistics, reviewing the drafts for major intellectual content. WZ helped in data processing, statistics, secondary inspection of intellectual content and article polishing. ZL, LC and JG designed the technological roadmap of the article together, guided the experiment and participated in the revision of the manuscript. All the authors above have given final approval of the version to be published and agreed to be fully accountable for all aspects of the work, ensuring thorough investigation and resolution of any inquiries regarding its accuracy or integrity.
All institutional and national guidelines for the care and use of laboratory animals were followed and approved by Animal Ethics Committee of Zhongshan Hospital, Fudan University (Ethics approval No. 2016-0817).
Not applicable.
This study was supported by the National Natural Science Foundation of China (No. 81201095), Clinical Research Project of Zhongshan Hospital (No. 2020ZSLC21) and Smart medical treatment project of Zhongshan Hospital (No. 2020ZHZS16), Shanghai Science and Technology Commission (No. 18411950200).
The authors declare no conflict of interest. Leilei Cheng is serving as the Guest editors of this journal. We declare that Leilei Cheng had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Yan Topilsky and John Lynn Jefferies.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.








