1 Tianjin Key Laboratory of Ionic-Molecular Function of Cardiovascular Disease, Department of Cardiology, Tianjin Institute of Cardiology, Second Hospital of Tianjin Medical University, 300211 Tianjin, China
2 National Institute of Health Data Science at Peking University, Peking University, 100871 Beijing, China
3 Institute of Medical Technology, Peking University Health Science Center, 100871 Beijing, China
4 Cardio-Oncology Research Unit, Cardiovascular Analytics Group, PowerHealth Limited, 999077 Hong Kong, China
5 Department of Cardiology, First Affiliated Hospital of Dalian Medical University, 116011 Dalian, Liaoning, China
6 Heart Failure Center, State Key Laboratory of Cardiovascular Disease, Fuwai Hospital, National Center for Cardiovascular Diseases, Chinese Academy of Medical Sciences and Peking Union Medical College, 100037 Beijing, China
7 Liverpool Centre for Cardiovascular Science, University of Liverpool, Liverpool John Moores University and Liverpool Heart & Chest Hospital, L69 3BX Liverpool, UK
8 Danish Center for Health Services Research, Department of Clinical Medicine, Aalborg University, 999017 Aalborg, Denmark
9 Section of Cardio-Oncology & Immunology, Division of Cardiology and the Cardiovascular Research Institute, University of California San Francisco, San Francisco, CA 94143, USA
10 School of Nursing and Health Studies, Hong Kong Metropolitan University, 999077 Hong Kong, China
Abstract
A growing body of evidence on a wide spectrum of adverse cardiac events following oncologic therapies has led to the emergence of cardio-oncology as an increasingly relevant interdisciplinary specialty. This also calls for better risk-stratification for patients undergoing cancer treatment. Machine learning (ML), a popular branch discipline of artificial intelligence that tackles complex big data problems by identifying interaction patterns among variables, has seen increasing usage in cardio-oncology studies for risk stratification. The objective of this comprehensive review is to outline the application of ML approaches in cardio-oncology, including deep learning, artificial neural networks, random forest and summarize the cardiotoxicity identified by ML. The current literature shows that ML has been applied for the prediction, diagnosis and treatment of cardiotoxicity in cancer patients. In addition, role of ML in gender and racial disparities for cardiac outcomes and potential future directions of cardio-oncology are discussed. It is essential to establish dedicated multidisciplinary teams in the hospital and educate medical professionals to become familiar and proficient in ML in the future.
Keywords
- cardio-oncology
- machine learning
- cardiotoxicity
- inequity
- multidisciplinary team
In recent years, advances in cancer diagnosis and treatment have significantly improved the survival and quality of cancer patients. However, this has been accompanied by a significant increase in the incidence of cardiotoxicity associated with cancer therapies [1, 2, 3]. A population-based study conducted on the causes of cardiovascular disease (CVD) death in the US has found that among 3.2 million cancer survivors, 38.0% eventually died from cancer and 11.3% died from CVDs, with 76.3% of CVD deaths were caused by heart disease [4]. CVD has become a significant cause of mortality and morbidity among cancer survivors [4]. CVD and malignancy share common risk factors, including age, obesity, and diabetes mellitus, and biological mechanisms such as increased oxidative stress and a pro-inflammatory milieu [5, 6]. Their clinical convergence led to the emerging discipline of cardio-oncology, which mainly focuses on the detection, monitoring, and treatment of cardiovascular disease occurring in the context of cancer treatment, encompassing both chemotherapy and radiotherapy.
Over the past decade, artificial intelligence (AI), particularly machine learning (ML), has changed medical practice and research to some degree [7]. By leveraging massive amounts of data, AI provides personalized opportunities for disease diagnosis, classification, risk stratification, and management [8]. Unlike human-coded time-to-event analysis, which relies on the expertise of the researcher to develop accurate and reliable coding criteria, ML algorithms use complex mathematical models to automatically identify patterns in the data. Statistical methods used in ML include regression analysis, clustering, and classification. Regression analysis enables the modeling of the relationship between a dependent variable and one or more independent variables. Clustering, on the other hand, is utilized to group similar data points into clusters, whereas classification is a technique for categorizing data points into distinct classes based on their features or characteristics [9]. ML algorithms often require large datasets to train and test the models. This is because the accuracy and effectiveness of ML models often increase with the amount of data used by them. Using data-trained learning algorithms, ML can make judgments about new situations, including but not limited to evaluating radiographic images, electronic medical records and pathology slides [10].
There are many branches of ML, including random forest (RF), artificial neural networks (ANN), convolutional neural networks and deep learning (DL) [11, 12], each having unique properties useful in cardio-oncology. ANN can simulate human neurons and process electrocardiogram (ECG) and echocardiographic data. In the cardiovascular field, the application of ANN mainly focuses on the multi-layer ANN used to simulate the human brain to operate in DL. It is also widely used in the analysis of imaging data, drug dosing and patient survival [12, 13, 14, 15]. Moreover, RF is commonly used in coronary computed tomography (CT) image processing, readmission of patients with heart failure, and the development of prediction models [16, 17, 18].
In this article, we systematically review the application of ML approaches in cardio-oncology, comprehensively describe the cardiotoxicities identified by ML, and outline the role of ML in the prediction, diagnosis and treatment of cardiotoxicities in cancer patients. In addition, we discuss the role of ML in better understanding the gender and racial disparities for cardiac outcomes among cancer patients. Given the current application of ML in clinical practice, we provide a perspective on the future development direction and challenges in cardio-oncology (Fig. 1).
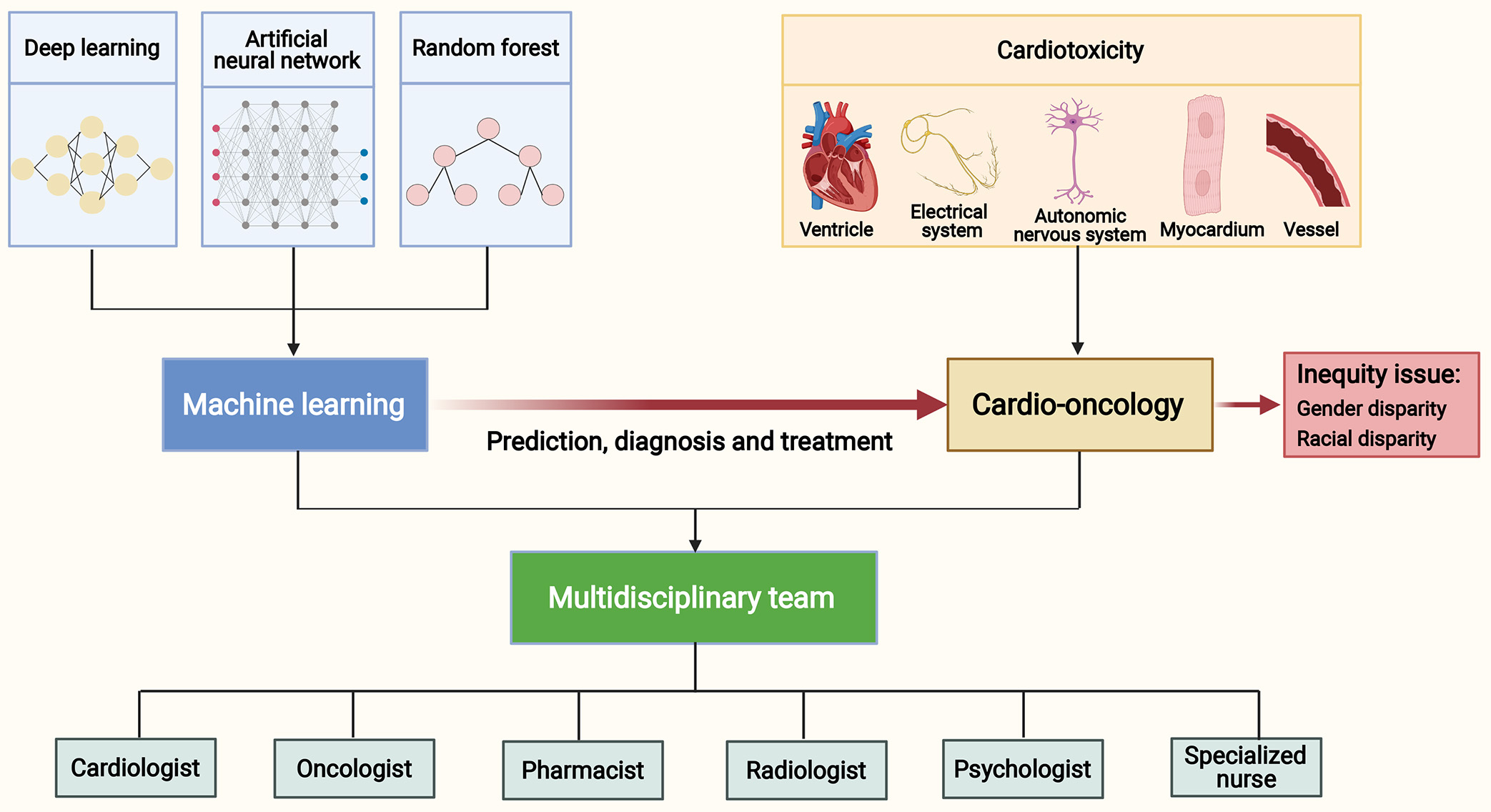 Fig. 1.
Fig. 1.Machine learning in cardio-oncology and a multidisciplinary team. Anticancer therapies may cause cardiotoxicities to the ventricle, electrical system, autonomic nervous system, myocardium and vessel. Machine learning approaches, including deep learning, artificial neural network and random forest, have an exciting application in the prediction, diagnosis and treatment of cardiotoxicities. Comprehensive management of cancer patients with cardiovascular diseases requires an equitable approach and the development of multidisciplinary teams. These teams should include experts such as cardiologists, oncologists, pharmacists, radiologists, psychologists, and specialized nurses.
Iatrogenic treatment harm refers to the harm caused by medical treatments or procedures [19]. In the context of cancer treatment, iatrogenic harm refers specifically to the harm caused by cancer treatments, such as chemotherapy, radiation therapy, and surgery. These treatments can result in both direct and indirect damage to the heart and cardiovascular system, and leading to a range of complications [20]. ML has the potential to significantly reduce the iatrogenic treatment harm. By analyzing large and complex datasets of patient information, ML algorithms can detect patterns and correlations that may not be easily discernible by humans [21]. This analytical capability can help predict which patients are at a higher risk of experiencing cardiotoxicity from cancer treatments, thus enabling healthcare providers to implement targeted interventions and personalized care. Different ML approaches in cardio-oncology are discussed as follows.
DL is a type of ML that combines statistics, computer science, and decision theory [22]. It is useful for analyzing hemodynamic and electrophysiological metrics which are increasingly obtained through wearable devices and cardiac imaging segmentation [22, 23, 24, 25, 26]. DL has shown exciting potential in automating complex image analysis [27], with broad applications in ultrasound, computed tomography, and magnetic resonance imaging [28]. The application of ML approaches in cardio-oncology is summarized in Table 1 (Ref. [29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40]).
| ML types | Sample | Cancer types | Conclusions | Reference |
| DL | 273 | Lung cancer | [29] | |
| DL | 2085 | Lung cancer | [30] | |
| DL | 180 | Lung cancer | [31] | |
| DL | 55 | NSCLC | [32] | |
| DL | 217 | NSCLC | [33] | |
| DL | 129 | Breast cancer | [34] | |
| DL | 127 | NSCLC | [35] | |
| DL | 49 | Breast cancer | [36] | |
| ANN | 489 | NSCLC | [37] | |
| RF | 49,864 | Prostate cancer | [38] | |
| RF | 45,000 | Breast cancer | [39] | |
| RF | 42,257 | Lung cancer | [40] |
ANN, artificial neural network; COD, causes of death; CVDs, cardiovascular diseases; DL, deep learning; ML, machine learning; LDCT, low-dose computed tomography; NSCLC, non-small cell lung cancer; RF, random forest; CT, computed tomography.
To date, the application of DL in the field of cardio-oncology has focused mainly on LDCT [29, 30]. LDCT is effective in lung cancer screening in clinical trials [41, 42], and screening for CVD comorbidities in high-risk populations undergoing LDCT is vital for reducing overall mortality. In high-risk patients, LDCT based on DL can screen for lung cancer and estimate CVD risk simultaneously [29]. In addition, DL can be applied to conventional chest CT imaging to quantify left atrial volume and predict adverse outcomes: a study [30] found that DL-measured left atrial volume index was significantly related to the higher risks of new-onset atrial fibrillation (AF), heart failure, and major adverse cardiovascular events within five years. The predicted values showed high agreeability with manual quantification, reinforcing its potential clinical applicability. Furthermore, inputting the risk of plaque and coronary artery calcification (CAC) in chest LDCT images as scores into a hybrid neural network algorithm, which is a subtype of DL, can predict all-cause mortality in lung cancer patients. Compared with other neural networks that input images alone and conventional semi-automatic scoring methods, the hybrid neural network achieved better performance [31].
Radiation therapy for patients with thoracic malignant tumors can significantly reduce the local recurrence rate of patients. However, for patients whose tumors are in close proximity to the heart, the heart will be irradiated inevitably, potentially resulting in cardiac damage. Emerging evidence has shown that cardiotoxicity from radiotherapy may be associated with specific cardiac substructures, but manual delineation of these substructures can be challenging. Harms et al. [32] proposed a DL-based algorithm for automatic delineation of cardiac substructures, including ventricles, great vessels, coronary arteries, heart valves, and the whole heart, all of which could be segmented within five seconds. This provided a tool for investigating associations between the radiation dose on cardiac substructures and resultant toxicity. Additionally, Haq et al. [33] established and validated a similar model for twelve cardio-pulmonary substructures, reducing the segmentation time from one hour to ten seconds per patient; importantly, evaluation by radiation oncologists determined its clinical acceptability for use in treatment planning and clinical outcomes analysis. Recently, several studies have confirmed the reliability of DL models for automatic contouring of cardiovascular substructures on CT images of radiotherapy planning [34, 35, 36]. DL models can accurately and rapidly outline the heart and vessels in large datasets of chest CT images, and are expected to be time-saving in future clinical practice.
Analysis of confounding behavior of dosimetric variables predicts overall survival in cancer patients. Cardiac dosimetry of cardiac subvolumes is associated with decreased overall survival in patients with early-stage non-small cell carcinoma undergoing stereotactic ablative radiotherapy [43]. Using ML to optimize radiotherapy treatment plans for non-small cell cancer patients can reduce mean cardiac dose without increasing pulmonary dose [44]. It is thus feasible and desirable to apply these tools during radiotherapy to optimize dosimetric tradeoffs and minimize irradiation of the heart.
ANNs are highly distributed and interconnected networks of computer elements modeled after biological nervous systems. ANNs simulate human neurons and combine neurons to form neural networks, use complex neural networks to make predictions, and perform regression analysis [45].
In patients with cancer, surgery can minimize tumor burden and is the most crucial treatment for solid tumors. However, postoperative inflammation may contribute to the development of adverse postoperative cardiovascular events, which are not rare and should be monitored closely. Studies have shown that tumor resection may be associated with cardiovascular complications such as arrhythmia, myocardial ischemia, and heart failure [46, 47, 48]. A limited number of neural networks trained for the same target can be assembled into a neural network ensemble, which can be used to predict morbidity. A recently proposed ANN ensemble predicted the occurrence of postoperative cardiovascular complications in non-small cell carcinoma patients undergoing pneumonectomy with satisfactory performance [37]. Before tumor surgery, attention should be paid to cardiovascular preparation and perioperative management. Such tools may aid pre-operative stratification of cardiovascular risk and optimization, which will likely reduce the risk of cardiovascular complications.
RF is a classifier that uses multiple trees to train and predict samples. The current application of RF in cardio-oncology mainly focuses on predicting the cause of cardiovascular death in cancer patients, and it has shown advantages in classification capabilities and accuracy compared with other ML and regression models [38]. Causes of death in cancer patients include cancer, non-cancer, infection, CVD, endocrine and blood diseases, and specific deaths because of other factors [38, 39, 40, 49]. A population-based study showed that CVD was the most common cause of death within the first year of cancer diagnosis [4]. It is thus essential to understand and prevent CVD in cancer patients. Three studies [38, 39, 40] used RF to predict causes of death, including CVD in breast cancer, lung cancer, and prostate cancer patients respectively, all of which found that RF was superior to multinomial logistic regression in predicting causes of death. RF allows systematic prediction of the cause of death in cancer patients and comprehensively analyzes the inter-relationships between risk factors, facilitating prevention of CVD amongst cancer patients.
As the most common adverse drug events, drug-induced cardiovascular complications are the leading cause of discontinuation for many post-marketing drugs or of restrictions of their use [50]. A recent study [50] developed a combined classifier framework using several ML algorithms, including RF, support vector machine, k-nearest neighbors, and neural network. This classifier was validated for 63 anticancer agents using human pluripotent stem cell-derived cardiomyocytes, providing a powerful tool for systemic risk evaluation of drug-induced cardiovascular complications that can be applied to anticancer drug clinical trials and post-marketing surveillance [50].
CTRCD is defined as asymptomatic cardiac insufficiency or symptomatic heart failure manifested by adverse effects of cancer treatment on cardiac structure and function [51]. Cancer therapies, including conventional chemotherapeutics such as anthracyclines, as well as targeted therapies comprising small-molecule kinase inhibitors, agents targeting human epidermal growth factor receptor 2 (HER-2), and specific proteasome inhibitors, have been demonstrated to be associated with CTRCD [52, 53, 54, 55]. The prevention, detection and treatment of cardiac dysfunction patients before, during and after cancer therapy play significant roles in precision cardio-oncology.
Clinically, the diagnosis of CTRCD usually relies on standard echocardiographic monitoring, most often left ventricular ejection fraction (LVEF) and global longitudinal strain (GLS) [56]. However, inter-reader variability in reporting of LVEF with its implications in defining cancer therapy-related cardiotoxicity, and variability for GLS measurement among various ultrasound system vendors limit their application. Comfortingly, AI plays an important role in improving LVEF and GLS assessment and inter-vendor agreement. Studies have shown that automated magnetic resonance imaging (MRI)-based left ventricular contractility analysis tools can provide accurate estimates of cardiotoxic impairment associated with chemotherapy for cancer [57]. Troponin, natriuretic peptides, systolic global longitudinal and circumferential strain have been used to assess CTRCD risk. A recent prospective cohort study [58] identified an integrated approach combining three-dimensional echocardiographic LVEF, GLS, and global circumferential strain to diagnose CTRCD using a conditional inference tree model. Deep convolutional neural networks can provide important information on ventricular function, detection, and pathologies related to myocardial dysfunction, which can be used for routine monitoring to identify cardiotoxicity in cardiac image analysis [59, 60, 61, 62, 63, 64]. The DeepLabV3+ deep convolutional neural network and ResNet-50 backbone network can measure parameters such as left ventricular end-diastolic diameter and LVEF in cancer patients, suitable for automatic left ventricular quantification of cardiotoxicity [65]. A supervised ML model using RF regression can identify left ventricular segmental strain from acquired echocardiograms of patients undergoing cancer treatment and estimate the nadir of LVEF after treatment completion, thereby predicting CTRCD [66]. The cardiotoxicities identified by ML are summarized in Table 2 (Ref. [48, 58, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83]).
| CVDs | Sample | Cancer types | ML types | Conclusions | Reference |
| CTRCD | 136 | Breast cancer | Conditional inference tree models | [58] | |
| CTRCD | 42 | Breast cancer | DeepLabV3 + deep CNN | [65] | |
| CTRCD | 237 | Breast cancer | RF | [66] | |
| Arrhythmias | 472 | Colorectal cancer | Support vector machine | [48] | |
| Arrhythmias | 1149 | Chronic lymphocytic leukemia | AI-ECG algorithm using CNN | [67] | |
| Arrhythmias | 210,414 | Prostate cancer | AI-ECG algorithm using CNN | [68] | |
| Heart rate variability | 77 | Breast cancer, prostate cancer, colorectal cancer, lung cancer, and pancreatic cancer | RF, linear discriminant analysis, and naive bayes | [69] | |
| ICIs related immunotoxicities | 4960 | Non-small cell lung cancer, melanoma, and renal cell carcinoma | Decision tree | [71] | |
| ICIs related immunotoxicities | 1152 | Various cancers | Natural language processing | [70] | |
| Cardiomyopathy | 1217 | NA | AI-ECG | [72] | |
| Cardiomyopathy | 471,9591 | Lung cancer and breast cancer | ML algorithm | [73] | |
| Coronary atherosclerosis | 480 | Lung cancer, prostate cancer, breast cancer, and hematological malignancies | Neural network ML | [74] | |
| Coronary atherosclerosis | 480 | Various cancers | Neural network ML | [75] | |
| CAC | 15,915 | Breast cancer | DL algorithm | [76] | |
| CAC | 12,332 | Lung cancer | ML algorithm | [77] | |
| CAC | 1825 | NA | ML-based ECG | [78] | |
| CAC | 16,000 | Breast cancer | DL algorithm | [79] | |
| CAC | 428 | Lung cancer | DL algorithm | [80] | |
| CAC | 2289 | Breast cancer | DL algorithm | [81] | |
| CAC | 1700 | Breast cancer | DU-Net model | [82] | |
| CAC | 840 | Breast cancer | CNN | [83] |
AI, artificial intelligence; CAC, coronary artery calcification; CNN, convolutional neural network; CTRCD, cancer treatment-related cardiac dysfunction; CVDs, cardiovascular diseases; DL, deep learning; ECG, electrocardiogram; ICIs, immune checkpoint inhibitors; ML, machine learning; RF, random forest; 2D, two dimensional; 3D, three dimensional; LDCT, low-dose computed tomography; CT, computed tomography. NA means that the article didn’t specify the cancer type of included patients.
Palpitation is a common complaint in cancer patients. Conventional cytotoxic chemotherapy, targeted drugs, and immunotherapies alike can lead to cancer therapy-related arrhythmias. The most common arrhythmia is AF, followed by ventricular arrhythmias, bradycardia and atrioventricular block [3, 54, 84, 85, 86, 87, 88].
The development of a rapid and inexpensive point-of-care method for AF screening using AI-enabled ECG has the potential to improve patient outcomes by enabling earlier detection and treatment of AF. Attia et al. [89] described the development of an AI-enabled ECG to identify patients with AF using standard 10-second, 12-lead ECGs. The AI algorithm was developed using a convolutional neural network and trained on a dataset of over 649,931 normal sinus rhythm ECGs from over 180,000 patients. The AI-enabled ECG was able to identify AF with an accuracy of 79.4% using a single ECG, and 83.3% when all ECGs acquired during the first month of each patient’s window of interest were included. Therefore, with high accuracy, AI-ECG could be used for AF screening in clinical practice. To evaluate the effectiveness of ML algorithms trained on ECG signals to predict patient outcomes after AF ablation, Tang et al. [90] used a convolutional neural network and a multimodal fusion framework to analyze data from 156 patients who underwent catheter ablation. The study highlights the importance of multimodal data analysis, which combines electrogram, ECG, and clinical features to improve the accuracy of prediction.
For cancer patients, AI-ECG is also a cost-effective and readily available tool for predicting cardiac arrhythmias. Christopoulos et al. [67] evaluated the role of AI-ECG in predicting AF in patients with ibrutinib-induced chronic lymphocytic leukemia and ibrutinib-independent AF. The AI-ECG algorithm, developed from a convolutional neural network, was used to monitor the rhythm of the two groups during the follow-up period, and the ECG characteristics of AF were used to predict the future risk of AF in the two groups of patients. The study demonstrated the feasibility and potential value of using AI-ECG in the management of patients with cancer. The role of AI algorithms in identifying androgen deprivation therapy (ADT)-induced changes in ECGs in prostate cancer patients has also been reported. A study [68] used a convolutional neural network to develop predictive signatures for cardiac pathologies, including the ability to predict the “estimated sex” of the patient. The results showed that patients who received ADT had a lower estimated male sex value in ECG compared to those who did not receive ADT, which was associated with decreased serum testosterone. This study highlights the potential of AI algorithms to detect changes in ECG parameters associated with ADT treatment for prostate cancer. The ability to monitor treatment effects and physiological changes non-invasively using ECGs has significant clinical implications, particularly for patients with cardiac morbidity and mortality associated with ADT. The findings also suggest the importance of considering the potential impact of ADT on serum testosterone levels and its association with ECG changes.
Drug-induced QT interval prolongation is a prevalent issue in various therapeutic interventions and can result in severe clinical consequences [91]. A relevant study [92] discussed the use of ML algorithms to predict the risk of drug-induced QT prolongation in inpatients. The researchers used harmonized data from the UCHealth electronic health record to compare multiple ML methods and found that a deep neural network demonstrated superior classification accuracy, which provided a reasonable predictive performance for identifying individuals with a high susceptibility to drug-induced QT prolongation. The further study compared the deep learning model, which had high accuracy but low interpretability, and the interpretable model based on cluster analysis, which was less accurate but more clinically applicable [93]. Both types of models have their own advantages and limitations, and that the choice of model should depend on the specific clinical context and the intended use of the prediction tool.
It is common for cancer patients to exhibit autonomic dysfunction with reduced heart rate variability, which has prompted investigations into the potential value of heart rate variability (HRV) in cancer detection. Based on five-minute ECG recordings, Vigier et al. [69] showed that ML-analyzed algorithm heart rate variability parameters could be used to distinguish cancer patients from healthy individuals with an accuracy of 79%–85%. However, circadian rhythms may increase heart rate variability measurement variance due to differences in timing and equipment of the subjects’ ECG recordings, and sleep alterations may also cause changes in heart rate variability signatures. Uniform standardization of ECG recording time and equipment as well as questionnaires on sleep patterns are required. Future studies should be conducted in more diverse and more extensive samples of cancer patients to explore the robustness of heart rate variability-based cancer detection.
ICIs have been one of the most promising types of anticancer drugs in recent years. ICIs therapy such as programmed death receptor-1(PD-1) or programmed death ligand-1(PD-L1) inhibitors can significantly improve the prognosis of cancer patients [94]. However, ICIs can induce a series of immune-related adverse events (irAEs) [95], including myocarditis, pericarditis, pericardial effusion, and acute vascular events [88, 96, 97, 98, 99], with myocarditis being the most frequently reported cardiac irAE [88, 100, 101, 102, 103]. Though uncommon, cardiac irAE may be fatal in up to 30% of cases [95], and the incidence of cardiac irAE associated with ICIs may be under-reported [96, 98, 104, 105, 106]. Therefore, it is important to identify risk factors for adverse cardiac events in patients receiving ICI therapy.
AI in the forms of ML and natural language processing (NLP) provides great analytical aptitude. The foundation of high-quality AI lies in data. As a considerable proportion of clinically relevant information is present in unstructured data, NLP assumes a critical role in extracting and analyzing information to inform decision-making, facilitate administrative reporting, and advance research efforts. This may be valuable when assessing complications or rare treatment-related events. Lu et al. [70] used NLP software to identify patients with various cancers treated with ICIs, and showed NLP could identify ICIs treatment-related cardiotoxicity events. NLP software can also be used to identify ICI-associated myocarditis in cancer patients and describe their clinical course and outcomes. Given the increasing popularity of ICIs treatment among various cancers, there is an unmet clinical need for identifying prognostic factors for myocarditis to facilitate clinical risk stratification, early diagnosis, and management.
Using a sizeable cross-sectional database of cancer patients, Heilbroner et al. [71] created a ML model for predicting cardiac events in PD-1/PD-L1-treated patients. The model comprehensively analyzed 356 potential risk factors and identified immunological, oncological, and cardiac risk factors associated with cardiac events. Although PD-1 treatment significantly increased the risk of cardiac events, PD-1 was not among the 40 most important predictors.
Cardiomyopathy is a heterogeneous entity that includes numerous subtypes, such as dilated, hypertrophic, and restrictive cardiomyopathy [107]. Early identification of cancer patients at high risk of treatment-related cardiomyopathy may improve outcomes by intervening before heart failure development.
Güntürkün et al. [72] conducted a prospective study of 1217 childhood cancer survivors reaching adult age in the St Jude Lifetime Cohort, in which seven AI methods and an extreme gradient boosting approach were applied to 12-lead ECG to predict cardiomyopathy during a mean follow-up duration of 5.2 years. A resultant model based on ECG and clinical characteristics achieved a cross-validation area under curve (AUC) of 0.89, with a specificity of 81% and a sensitivity of 78%. AI-ECG may therefore help identify cancer survivors at increased risk of developing cardiomyopathy.
Stress cardiomyopathy is characterized by transient, reversible, local, or global myocardial dysfunction without ischemic perfusion defects [108, 109]. Catecholamine surge, epicardial coronary artery spasm and/or diffuse coronary vasoconstriction, and microvascular dysfunction are considered to be crucial mediating processes in the pathophysiology of stress cardiomyopathy [110]. Stress cardiomyopathy can be activated by various stressors, including infection, surgery, emotional or psychological stress, worsening chronic diseases, and medication [111]. Studies have shown that cancer patients with stress cardiomyopathy have a higher in-hospital mortality rate than those with only stress cardiomyopathy [112, 113]. However, a ML analysis by primary tumor types showed that stress cardiomyopathy was not associated with the in-hospital mortality of active cancer patients, with lung cancer and breast cancer both being associated with an increased likelihood of stress cardiomyopathy, and breast cancer patients with stress cardiomyopathy having a significantly reduced mortality [73]. Further prospective researches are needed to confirm these findings and reveal possible protective factors in breast cancer patients with stress cardiomyopathy.
Cancer causes a hypercoagulable and pro-inflammatory state, sometimes accompanied by systemic infection which can lead to changes in inflammatory cytokines and massive release of chromatin. Meanwhile, cancer can increase the levels of peripheral blood neutrophils, predisposing to the formation of extracellular traps which are procoagulant and prothrombotic. The released chromatin and extracellular traps promote endothelial injury, followed by platelet aggregation, vasospasm, and possibly accelerated atherosclerosis [114, 115, 116, 117, 118]. ML can be used to assess whether specific malignancies can alter the natural progression and location of coronary artery diseases. A study [74] of both cancer patients and non-cancer patients undergoing coronary angiography showed that ML analysis was useful for identifying the locations of significant coronary stenoses. The study showed that lung cancer patients had higher odds of significant left anterior descending and right coronary artery stenoses than patients without lung cancer. However, the results remain controversial. Another ML-based analysis [75] showed that when compared to patients without cancer, patients with cancer had significantly fewer left anterior descending and left circumflex lesions, but were less likely to have multiple coronary arteries and acute left circumflex artery diseases. However, these findings were limited by potential bias by indication. More prospective studies should be conducted to reveal the longitudinal relationship between cancer and the development of coronary lesions.
Measured from computed tomography scans, CAC is an independent risk factor for CVD [119, 120, 121]. Traditional CAC scoring is often manual, which is tedious and time-consuming [76]. Recently, several studies have shown that ML-based automated algorithms may be used for evaluating CAC scores, which, in turn, can be used to predict CVD morbidity and mortality in cancer patients [76, 77, 78, 79, 80, 81]. A DL-based CAC scoring algorithm was developed for automatic CAC scoring [122, 123], providing a rapid and low-cost tool for cancer patients at increased risks of CVD. Gernaat et al. [81] used a DL algorithm to score CAC automatically, and found that the prevalence of CAC in cancer patients was relatively high and increased with age. Comparisons with manual scoring confirmed the reliability of DL in measuring CAC. Similarly, Gal et al. [76] used a DL algorithm to automatically extract CAC scores, demonstrating a direct correlation between CAC scores and CVD risks among cancer patients.
Breast arterial calcification as detected by mammography may be evidence of general atherosclerosis, and may be a valuable marker of CVD [124]. Studies have shown that DL can be used to detect breast arterial calcification effectively and assess patients at high cardiovascular risks [82, 83], with performance similar or better than manual detection. Further large-scale studies are needed to test and improve these models across different experimental settings [125].
During chemotherapy and targeted therapy, cancer patients will develop adverse reactions of various tissues and organs in the body, among which cardiotoxicity is one of the most severe complications. Related studies have shown that ML can be used to predict the risk of cardiotoxicity in cancer patients after anticancer therapy. Li et al. [126] enrolled colorectal cancer patients treated with fluoropyrimidine and developed ML models including extreme gradient boosting, RF, and logistic regression to predict the subjects’ risk of cardiotoxicity. The study showed that among 36,030 colorectal cancer patients, 18.74% developed cardiotoxicity within 30 days after the first dose of fluoropyrimidine. All three ML models demonstrated high prediction accuracy with extreme gradient boosting having the best prediction performance. Thus, the ML model can accurately predict the occurrence of cardiotoxicity within a certain period after cancer patients start chemotherapy. Application of ML to positron emission tomography (PET) scans in cardio-oncology patients is also an emerging avenue. It has been demonstrated that risk prediction using ML applied to PET is more effective at predicting patients at high risk of major adverse cardiovascular events than logistic regression [127]. Additionally, PET scans in another study showed that coronary flow reserve was inversely related to radiation dose to specific coronary regions, which suggested that PET could identify coronary arteries damage after radiation therapy [128].
ML-based algorithms may be emerging diagnostic tools in cardio-oncology. For
example, systolic dysfunction remains one of the significant side effects in
patients with HER-2-positive breast cancer treated with trastuzumab. Therefore,
current guidelines recommend that patients undergo echocardiography every three
months during treatment. However, AI-augmented ECG using ML algorithms has good
diagnostic performance in predicting abnormal ejection fraction while on
trastuzumab therapy: a study [129] showed that the AI 12-lead ECG algorithm could
reduce echocardiography by 15% when screening HER-2-positive breast cancer
patients without missing a single patient with an LVEF of
Sometimes the lack of baseline LVEF can be a challenge when assessing the likelihood of CTRCD. ML algorithms can predict CTRCD in cancer patients based on clinically relevant variables, effectively circumventing the lack of LVEF. A classification model was trained to evaluate six cardiovascular outcomes, showing that clinical variables such as age, hypertension, blood glucose levels, creatinine and aspartate aminotransferase levels were all significantly associated with CTRCD [130]. Compared with traditional cardiac function monitoring methods, ML provides powerful tools for cardiac risk stratification in cancer patients by leveraging longitudinal, large-scale patient data from healthcare systems.
Some cardio-protective agents can prevent or reduce cardiotoxicity during
anticancer therapy. A recent meta-analysis [131] showed that
angiotensin-converting enzyme inhibitors, angiotensin receptor
blockers, and beta-blockers could preserve LVEF and protect against
cardiotoxicity during trastuzumab and anthracycline treatment, with statistically
significant outcomes with beta-blockers. Various anticancer drugs can produce
off-targeted effects that negatively affect cardiac function and reduce LVEF.
Heart failure with reduced ejection fraction should be concomitant with specific
anticancer therapy to prevent cardiotoxicity. The American and European
Cardio-Oncology Guidelines state that in patients with LVEF
An RF-based study [134] that performed exome sequencing of 289 childhood cancer survivors exposed to anthracyclines for at least three years, showed that almost 90% of patients without cardiotoxicity harbored rare/low-frequency variants in cardiac injury pathways that likely protected them from the damaging effects of anthracycline. In contrast, less than 50% of patients with cardiotoxicity harbored these variants. Compared with models using only clinical variables, cardiotoxicity risk prediction models incorporating clinical and genetic risk factors were more precise and had lower misclassification rates. The study using RF [134] found that in vitro gene inhibition of related pathways such as phosphoinositide-3-kinase regulatory subunit 2 (PI3KR2) and zinc finger protein 827 (ZNF827) protected cardiomyocytes from cardiotoxicity. The discovery of variant genes that protect against cardiotoxicity in cardiac injury pathways provides information for establishing predictive models for late-onset cardiotoxicity of anthracyclines, and autophagy gene targets to exploit cardio-protective drugs.
Sex is an independent risk factor that increases the risk of adverse cardiovascular events in cancer patients. A recent systematic review and meta-analysis [135] found that among 13,975 patients with Hodgkin’s Lymphoma who received radiation therapy, cardiovascular events mortality was significantly higher in women than in men with an odds ratio of 3.74. Propensity scores use existing data to make statistically significant inferences and are of great value for real-world data analysis. However, the level of evidence in evidence-based medicine is still insufficient. As the accuracy and operational advantages of ML in large-scale medical data analysis are increasingly recognized, ML can be used to improve propensity scores, so that the two can be deeply combined to improve the selection accuracy of covariates in propensity scores [136].
One multicenter case-control study [137] used ML-augmented propensity score to analyze the outcomes of 30,195,722 hospitalized patients, revealing that percutaneous coronary intervention significantly reduced overall mortality, especially for cancer patients. Furthermore, percutaneous coronary intervention considerably reduced total hospitalization costs of cancer patients. However, a nationally representative case-control analysis using ML-generated propensity scores showed that women were less likely to undergo percutaneous coronary intervention and survive than men in cancer patients [138]. The above findings underscore the need for enhanced surveillance, and ML is expected to be better applied to monitor the incidence of adverse cardiovascular events in female cancer patients.
The inadequate representation of women in clinical trials, the higher dose of radiation required by female patients for specific types of cancer, and women’s higher degree of microvascular coronary artery diseases may account for the gender disparities [135, 139]. Further studies should focus on the age of the patients, treatment dose, duration, and methods of diagnosis for CVD to arrive at definitive conclusions about gender risks. The related guidelines also suggest optimizing gender equality in therapy access and outcomes. Future investigations on gender disparities should be expanded to other gender-discriminated groups, including gays, lesbians, bisexuals, asexuals, transgender, and intersex patients.
Though only early reports have been published, racial disparities may have adverse effects on the cardiovascular outcome of patients who receive cancer therapy. Limited data have shown that compared with non-Hispanic White, the incidence of cardiovascular adverse outcomes increases in black cancer patients. African American men patients are 2.5 times more likely to die from prostate cancer than white men [140], and black women patients with breast cancer had a 25% higher risk of cardiovascular death than non-Hispanic White [141]. A large case-control study [142] conducted a ML-optimized and propensity score-adjusted study about the mortality of cancer patients who received percutaneous coronary intervention treatment in the United States in 2016, revealing that although mortality was comparable, racial disparities in outcomes remained in cancer patients with percutaneous coronary intervention treatment. A case-control study [143] using propensity score analysis and neural network ML-augmented multivariable regression in cancer patients with CVDs showed a significant increase in mortality among Hispanics and Asians as compared with Caucasians. The current coronavirus disease-2019 pandemic has further enlarged the gap on the influence of the marginal population in history, strengthening the importance of determining and resolving this inequality [144].
The root causes of racial disparities are multifactorial, complex and interleaved. The previously described factors include racism, prejudice, limited health care, distrust of the medical profession due to historical experience, related genetic and molecular basis, and underrepresentation in clinical trials [145, 146, 147, 148]. One clinical trial study found that although African Americans constitute 22% of all US cancer patients, they only account for 3.1% of the trial participants [149].
Using ML approaches to identify racial disparities in patients with cardio-oncology diseases and improve the representation of different ethnic groups in clinical trials may therefore be critical for improving outcomes and reducing adverse CVD events. A multidisciplinary approach is needed to eliminate the racial disparities in cardio-oncology patients, including joint efforts of critical stakeholders, healthcare policymakers, clinicians, patients, and scientists [144].
At present, ML has a wide range of applications in the cardiovascular field, especially in many aspects of cardiac imaging, including but not limited to detection, characterization, and segmentation [150]. ML can take the place of many regular detection, characterization, and quantification work performed by clinicians using the cognitive ability and complete the integration of electronic medical records data mining [151, 152, 153]. It has been shown to outperform human experts in specific situations [31, 82]. This may raise a question amongst clinicians: is ML a threat or opportunity? Currently, most experts are of the opinion that although ML has dramatically improved its capabilities and applications, it will not replace clinicians at least in the near future [154, 155, 156]. Intelligent medical technology exists to improve patient management and support physician decision-making. ML has limitations, such as the need for large datasets for training and validation, and difficulty in identifying initial clustering patterns. As with any technology, ML is not infallible and there is a risk that it could fail. Some of the potential causes of ML failure include poor data quality, lack of domain knowledge, inappropriate modeling techniques, and overfitting of models to training data [157]. If we rely solely on advanced statistics, there is a risk of over-reliance on the models developed by ML algorithms, which can lead to poor decision-making. Human domain knowledge and expertise are also critical in interpreting the results of ML models and making informed decisions. In addition, the dehumanization of medicine is one of the most significant barriers to the application of intelligent medical technology [158]. Cancer not only brings physical pain to patients, but also many psychological and social challenges. Clinicians need to use appropriate information communication methods and content to provide psychological support and humanistic care. To this end, ML should assist clinicians as an adjunct, rather than being a replacement. Using AI is an iterative learning process, and the important thing is to harness and use this technique correctly and avoid misuse.
Cardio-oncology is still in its infancy in many countries and its development has been lagging. The increasingly active research field of cardio-oncology warrants wider dissemination and integration into routine clinical practice. To this end, the formation of cardio-oncology teams with multidisciplinary input from cardiologists, oncologists, pharmacists, radiologists, psychologists, and nurse specialists is imperative. Besides, the opening of multidisciplinary joint diagnosis and treatment clinics in cardio-oncology is expected. Recently, the view of educating augmented doctors has been raised, and some universities have begun to innovate educational models to meet the need to train future doctors for the challenges of AI in medicine [158]. Although the risk of clinicians being replaced by ML in the near future is remote, clinicians are recommended to learn more about ML to rely on digital expertise and clinical experience to solve medical practice problems better. Researchers and clinicians can employ statistical software packages such as R or Python to construct and train ML models. The quantity of data necessary to build an ML model is contingent upon several factors, including the intricacy of the problem, the data quality, and the selected algorithm. Typically, larger datasets are preferred since they yield more informative content for the model to learn from and enhance the accuracy of predictions [159].
In conclusion, cardiotoxicities of cancer therapies cannot be ignored. ML plays a certain role in predicting, diagnosing and treating cardiotoxicities in cancer patients. As essential parts of ML, DL, RF, and ANN have tremendous applications in cardio-oncology. Inequalities exist in cardio-oncology, and ML is expected to optimize equality in treatment opportunities and outcomes. Medical education should cultivate interdisciplinary talents proficient in oncology, cardiovascular medicine and ML to meet future challenges.
ADT, androgen deprivation therapy; AF, atrial fibrillation; AI, artificial intelligence; ANN, artificial neural networks; CAC, coronary artery calcification; CTRCD, cancer treatment-related cardiac dysfunction; CVD, cardiovascular disease; DL, deep learning; ECG, electrocardiogram; GLS, global longitudinal strain; HER-2, human epidermal growth factor receptor 2; ICIs, immune checkpoint inhibitors; LDCT, low-dose computed tomography; LVEF, left ventricular ejection fraction; ML, machine learning; NLP, natural language processing; PD-1, programmed death receptor-1; PD-L1, programmed death ligand-1; PET, positron emission tomography; RF, random forest.
YZ and TL designed the paper; YZ wrote the paper; ZLC, SH, NZ and YYW provided help and advice on literature search and acquisition of data; SDH, JSKC, KYC, YLX, YHZ, GYHL, JQ, GT and TL revised the manuscript critically for important intellectual content and made contributions in conceptualization. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We would like to express our sincere gratitude to those who helped us during the writing of the manuscript and thanks to all the peer reviewers for their opinions and suggestions.
This work was supported by the National Natural Science Foundation of China (81970270, 82170327 to TL), Tianjin Natural Science Foundation (20JCZDJC00340, 20JCZXJC00130 to TL) and Tianjin Key Medical Discipline (Specialty) Construction Project (TJYXZDXK-029A).
The authors declare no conflict of interest. Gary Tse and Tong Liu are serving as Guest Editor of this journal. We declare that Gary Tse and Tong Liu had no involvement in the peer review of this article and have no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Poul F. Høilund-Carlsen.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.