1 Internal Medicine Unit, Department of Advanced Medical and Surgical Sciences, University of Campania “Luigi Vanvitelli", Piazza L. Miraglia, 80138 Naples, Italy
2 Department of Internal Medicine, Betania Evangelical Hospital, Via Argine, 80147 Naples, Italy
3 Facility of Epidemiology and Biostatistics, Gemelli Generator, Fondazione Policlinico Universitario A. Gemelli IRCCS, 00168 Rome, Italy
4 Division of Cardiology, A.O.R.N. “Sant'Anna e San Sebastiano", Caserta, Italy
5 Department of Translational Medical Sciences, University of Campania “Luigi Vanvitelli", 80138 Naples, Italy
Academic Editors: Massimo Volpe, Filippos Triposkiadis, Grigorios Korosoglou and Matteo Cameli
Abstract
Type 2 Diabetes Mellitus (T2DM) is associated with an elevated incidence of cardiovascular and renal diseases, responsible for mortality rates significantly higher than in the general population. The management of both cardiovascular risk and progression of kidney disease thus seem crucial in the treatment of the diabetic patient. The availability of new classes of drugs which positively affect both cardiovascular and renal risk, regardless of the glycemic control, represents a revolution in the treatment of T2DM and shifts the attention from the intensive glycemic control to a holistic management of the diabetic patient. Among these, sodium-glucose cotransporter-2 inhibitors (SGLT2i) have been associated with a remarkable reduction of cardiovascular and renal mortality, lower hospitalization rates for heart failure and lower progression of renal damage and albuminuria. Thus, their use in selected subpopulations seems mandatory. Aim of this review was the assessment of the current evidence on SGLT2i and their related impact on the cardiovascular and renal profiles.
Keywords
- SGLT2i
- gliflozin
- heart failure
- chronic kidney disease
- diabetes
Type 2 Diabetes Mellitus (T2DM) is among the major risk factors for both cardiovascular (CVDs) and renal diseases [1]. T2DM accounts for a 2-fold increase of CVDs, the main cause of death among T2DM patients, and, as well, it represents the main cause of renal damage and end-stage kidney disease (ESKD) [1, 2, 3]. In fact, almost 35% of T2DM patients display a chronic kidney disease (CKD), and the concomitant presence of T2DM and renal disease results in an overall mortality risk 23% higher than in non-diabetic patients with preserved renal function [4]. Despite the current pharmacological (e.g., renin-angiotensin-aldosterone system blockers - RAAS, optimization of both glycemic and blood pressure control) and other (e.g., lifestyle modifications) interventions, the risk of both CVDs and CKD among individuals with diabetes still remains elevated [5].
The recent introduction of new anti-hyperglycemic drugs has marked a conceptual turning point in T2DM treatment. In fact, sodium-glucose cotransporter inhibitors (SGLT2i) and glucagon-like peptide 1 receptor agonists (GLP1-RA) have been proven to significantly affect both cardiovascular (CV) and renal outcomes in individuals with diabetes, regardless of glycemic control, and are now indicated by international guidelines among the first choice drugs for patients with T2DM as secondary prevention in patients with established atherosclerotic CVDs or for primary prevention of CV events in high-risk patients [6, 7]. In particular, SGLT2i have been associated with a remarkable reduction of CV and renal mortality, lower rates of hospitalization for heart failure (HF) and lower progression of renal damage [8].
This review aims to assess SGLT2i effects on both cardio- and renal protection and the pharmacodynamic mechanisms potentially involved.
CV risk is almost doubled in T2DM patients than in the general population [1]. In fact, T2DM enhances the risk of coronary artery disease (CAD), HF (also in absence of significant CAD), ischemic stroke and peripheral arteriopathy, by mechanisms often mediated by both the induction and progression of atherosclerosis phenomena [2, 3, 9]. Thus, the impact of CV events on overall mortality rates renders CV risk reduction mandatory in the management of T2DM patients [10]. In this view, besides the already known treatments, the clinician must apply all measures suitable to reduce the CV risk of diabetic patients [11].
In this regard, the two new classes of antihyperglycemic drugs, SGLT2i and GLP1-RA, have represented a radical change in the therapeutic algorithm, due to their demonstrated ability to reduce both CV and renal events in patients with T2DM and CKD. In particular SGLT2i, beyond their glucose-lowering effect due to the reduced reabsorption and the increased renal excretion, have demonstrated a remarkable favorable impact on CV mortality [12, 13, 14, 15, 16, 17, 18, 19]. The reduction of atherosclerotic CVDs occurs by a significant decrease of CV death rates. On the contrary, data on both ischemic stroke and myocardial infarction (MI) reduction seems controversial, though the overall trend favorable to a reduction of the risk of cardio and cerebrovascular events. SGLT2i exert the maximum beneficial effect on the reduction of hospitalization rates due to HF so significantly that particularly dapagliflozin, empagliflozin and sotagliflozin are recommended by the European guidelines in all patients with reduced left ventricular ejection fraction (LVEF) (class of recommendation: Ia), even in non-diabetic patients [7]. Although the heterogeneous enrollment criteria among the different trials cited so far and the focus of some trials on primary prophylaxis and others on secondary, CV risk reduction seems almost comparable among all SGLT2i (empagliflozin, canagliflozin, dapagliflozin, ertugliflozin, sotagliflozin). However, clinical trials aimed at direct comparison between drugs seem mandatory to confirm this hypothesis.
Empagliflozin Cardiovascular Outcome Event Trial in Type 2 Diabetes Mellitus Patients (EMPA-REG OUTCOME) assessed the CV risk reduction of Empagliflozin (Table 1, Ref. [12, 13, 14, 15, 16, 17, 18]) on T2DM patients affected by either coronary, peripheral or cerebrovascular disease versus placebo, after a median follow-up of 3.1 years [12]. Empagliflozin treatment resulted in a 38% reduction of CV death and 32% of death from any cause, and a 35% reduction of hospitalization for HF, without any efficacy on MI and stroke events.
| Empagliflozin | Canagliflozin | Dapagliflozin | Ertugliflozin | Sotagliflozin | |||
| RCT | EMPA-REG | CANVAS | CREDENCE | DECLARE-TIMI 58 | DAPA-HF | VERTIS-CV | SCORED |
| Reference | [12] | [13] | [14] | [15] | [16] | [17] | [18] |
| Publication date | 2017 | 2017 | 2019 | 2019 | 2019 | 2020 | 2021 |
| No. of patients | 7020 | 10142 | 4401 | 17160 | 4744 | 8246 | 10584 |
| Median follow up (years) | 3.1 | 2.4 | 2.62 | 4.2 | 1.52 | 3.0 | 1.33 |
| eGFR, mL/min/m |
30 |
25 | |||||
| Reduced Composite Outcome (CV death, fatal/non-fatal MI and/or ischemic stroke) | –14% | –14% | –20% | –7% | n.a. | –3% | n.a. |
| Reduced Composite Outcome (CV death, hospitalization for HF and/or urgent visits for HF) | n.a. | n.a. | n.a. | n.a. | n.a. | n.s. | –26% |
| Reduced CV Death | –38% | –13% | –22% | –2% | –18% | –8% | n.s. |
| All-cause death | –32% | –13% | –17% | –7% | –17% | –7% | n.s. |
| Reduced Fatal/non-fatal MI | n.s. | -15% | n.a. | –11% | n.a. | +4% | n.a. |
| Reduced ischemic stroke | n.s. | -10% | n.a. | +1% | n.a. | +6% | n.a. |
| Reduced hospitalization for HF | –35% | –33% | –39% | –27% | –30% | –30% | –33% |
| Reduced progression of renal damage | n.a. | –40% | –34% | –47% | –29% | –19% | n.a. |
| Reduced Proteinuria | n.a. | –27% | –40% | n.a. | n.a. | n.a. | n.a. |
| Any cause hospitalization | n.a. | –6% | n.a. | n.a. | n.a. | n.a. | n.a. |
| Abbreviations: RCT, Randomized Controlled Trial; eGFR, estimated glomerular filtration rate; CV, cardiovascular; MI, Myocardial Infarction; HF, Heart Failure, n.a., not applicable; n.s., not significant. | |||||||
CANVAS (Canagliflozin Cardiovascular Assessment Study) Program trial, includes two separate sub-studies: the CANVAS, investigating CV outcomes, and the CANVAS-R, to examine benefit on albuminuria (Table 1), instead focused on Canagliflozin [13, 19]. T2DM patients with either CVD or CV risk factors, after a median follow-up of 3.6 years, showed a 14% reduction of the composite outcome (CV death, non-fatal MI/stroke), as well as a reduced incidence of HF hospitalization. No significant effect was instead observed as for all-cause and CV mortality.
Dapagliflozin potential CV benefits were instead analysed in the Dapagliflozin
Effect on Cardiovascular Events (DECLARE-TIMI 58) (Table 1) [15]. After a median
follow-up of 4.2 years, a population of T2DM patients with established
atherosclerotic CVDs or multiple CV risk factors showed, as compared to placebo,
a 17% reduction of the composite outcome (CV death/hospitalization for HF).
Despite a 7% reduction rate, major adverse cardiac events (MACEs: death from CV
causes, non-fatal MI or ischemic stroke) did not significantly varied with
respect to placebo. Individual analysis of the composite outcome clarified that
this effect was mainly driven by a strong reduction in hospitalization for HF,
without any evidence of change in CV death. A further focus on HF population is
provided by the DAPA-HF (Dapagliflozin and Prevention of Adverse Outcomes in
Heart Failure) randomized clinical trial, designed to prospectively assess both
efficacy and safety of Dapagliflozin on 4744 patients (both diabetic and not)
with HF, ejection fraction
The VERTIS CV clinical trial (Evaluation of Ertugliflozin Efficacy and Safety Cardiovascular Outcomes Trial) focused on CV outcome in T2DM patients at high CV risk, after treatment with Ertugliflozin, as compared to placebo (Table 1) [17]. After a median follow-up of 3.5 years, Ertugliflozin demonstrated non-inferior to placebo as for the composite primary outcome (CV death, non-fatal MI, or stroke). Moreover, consistent with the other SGLT2i, it revealed superior to placebo in reducing hospitalization for HF. Other secondary endpoints (e.g., CV death and all-cause mortality) were instead not reached.
Similar impact on CV outcomes was also demonstrated by Sotagliflozin, underlining how this effect is comparable among all SGLT2i. The SCORED (Sotagliflozin on Cardiovascular and Renal Events in Patients with Type 2 Diabetes and Moderate Renal Impairment Who Are at Cardiovascular Risk) trial showed that Sotagliflozin is able to significantly reduce the composite outcome of deaths from CV causes, hospitalizations for HF, and urgent visits for HF in patients with T2DM, CKD and risk for CV disease, independently by the presence of albuminuria [18]. On the other hand, the rate of deaths from CV events did not show statistically significant differences compared to placebo, although the trial was stopped early due to lack of funds. However, in the SOLOIST-WHF trial, Sotagliflozin has been shown to significantly reduce the rate of CV mortality and re-hospitalization in T2DM patients with recent HF worsening [20].
A recent metanalysis including findings from EMPA-REG OUTCOME, CANVAS Program
and DECLARE-TIMI 58 furtherly investigated the cardioprotective role of SGLT2i
[8]. SGLT2i confirmed beneficial in reducing MACEs (HR 0.89; 95% CI:
0.83–0.96), with an overall risk reduction of about the 11% as compared to
placebo. Further meta-analyses, in addition to confirming the reduced incidence
of HF and HF hospitalization, has shown how treatment with SGLT2i also positively
impacts on the incidence of MACEs (MI and stroke) and on CVD-related and
all-cause mortality [21, 22]. Moreover, SGLT2i treatment appears to reduce the
risk of arrhythmias, in particular atrial fibrillation and ventricular
tachycardia [23]. Toyama et al. [24] focused on the role exerted by
SGLT2i in CKD patients (eGFR
Although the effects of SGLT2i both on glycemic control and on CV risk and the progression of renal damage appear homogeneous, there are significant differences in the mechanisms of action of the individual molecules which could result in a different clinical impact. In this regard, Sotagliflozin, compared to other SGLT2i, is also an inhibitor of SGLT1, which is present not only in the proximal renal tubule, but also in the heart and small intestine [35, 36]. This peculiarity determines a reduced intestinal absorption of glucose and consequently a reduction in postprandial glycaemia, as well as an enhancement of the glycosuric effect (about three times greater) [35, 37]. A recent meta-analysis showed that non-selective SGLT2i such as Sotagliflozin may be superior to highly selective SGLT2i in terms of HF outcomes [38]. However, specific comparative clinical trials appear necessary to evaluate the possible superiority of Sotagliflozin in terms of reduction of CV risk and progression of renal damage compared to other SGLT2i.
The gliflozin pleiotropic effect in modifying CV outcomes has been widely investigated, though always new hypothesises have been arising. Mechanisms potentially involved in the impact of gliflozins on CV outcomes can be differentiated into three main groups: haemodynamic effects, metabolic effects, and impact on known CV risk factors (Fig. 1).
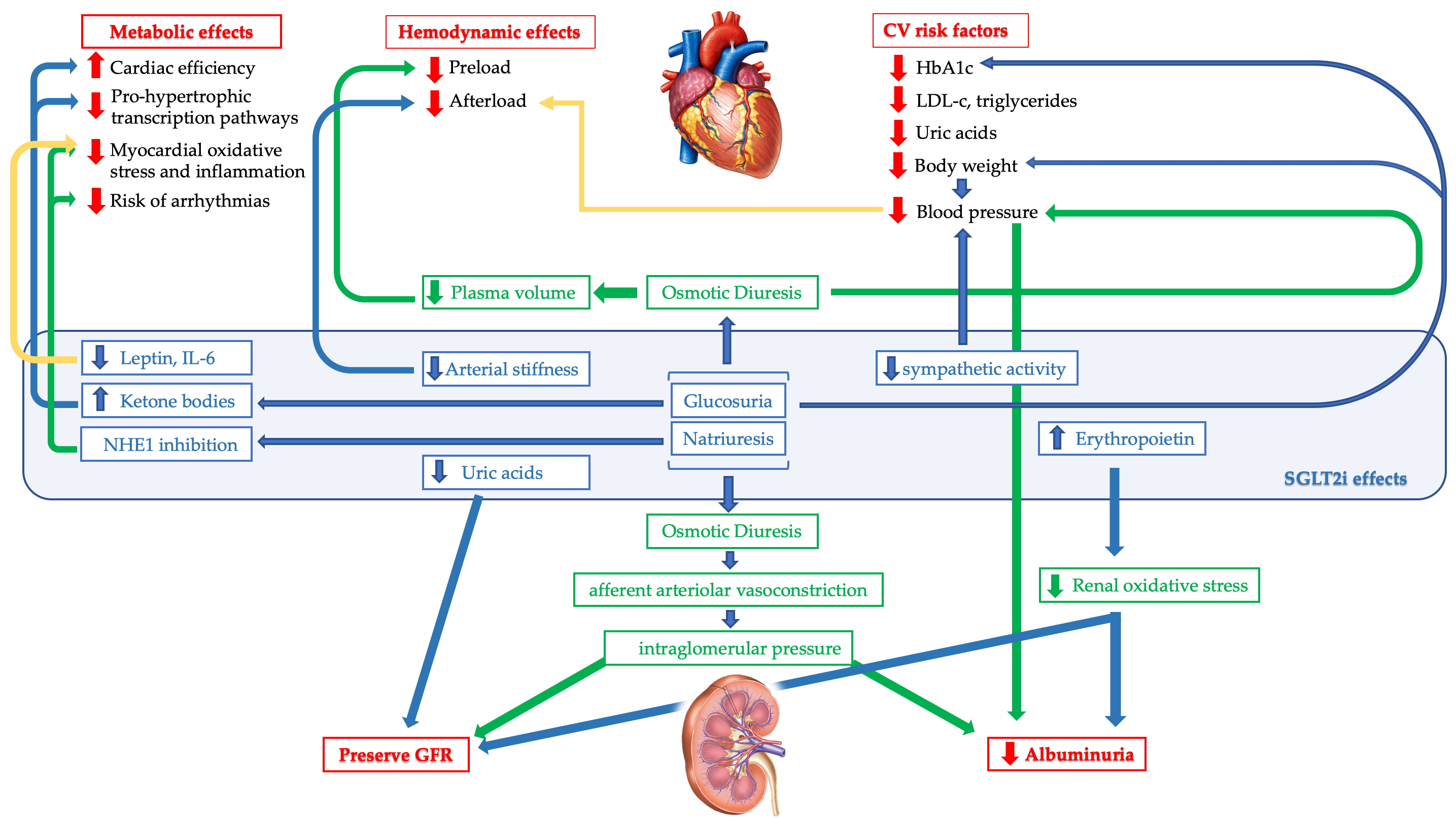 Fig. 1.
Fig. 1.Map of the main mechanisms potentially involved in the cardiorenal impact of SGLT2i. The haemodynamic and metabolic effects of glyflozines and their effect on independent cardiovascular risk factors are intertwined with a positive impact on cardiovascular outcomes. Furthermore, their glycosuric and natriuretic effects favor a reduction in the degree of albuminuria and preserve the GFR.
Impact on cardiac preload. T2DM patients have an increased tissue sodium content [39]. Muscle and skin sodium reservoir are the most relevant in contributing to fluid retention and preload [40]. In addition, elevated tissue sodium levels have shown as independently associated with left ventricular (LV) hypertrophy in patients with CKD [41]. According to a double-blind randomized study, sodium tissue levels of diabetic patients decrease after 6 weeks treatment with Dapagliflozin [39] and Empagliflozin [42], reducing estimated extracellular and plasma volume. A mediation analysis of the EMPA-REG OUTCOME trial suggested an association between a reduction in plasma volume and a CV benefit [43]. Gliflozins, by inhibiting SGLT2 in the renal proximal tube, would induce natriuresis and glucosuria [44, 45]. The presence of not reabsorbed glucose and sodium in the tubule stimulate osmotic diuresis. As a result, both sodium and chloride concentration are lower into the tubular fluid, thus inhibiting the NA-K-2Cl cotransporter and preventing sodium reabsorption in the loop of Henle [46]. Consequently, there is a reduction in plasma volume and total body sodium content [41], modifying cardiac preload condition, with a positive impact on Franklin-Starling curve of left ventricle [47]. Indeed, SGLT2i have been shown to determine a significant improvement in LV volumes, LV mass and LV systolic function [48] and in pulmonary congestion through the reduction of lung fluid volumes [49]. However, other diuretics (e.g., thiazides and loop diuretics) have not shown any benefit in CV outcome, as confirmed in two studies comparing dapagliflozin vs hydrochlorothiazide and bumetanide, respectively [45, 50]. SGLT2i have been suggested to induce an electrolyte-free water clearance from the interstitial fluid higher than blood volume, and a decreased activation of the RAAS through the neurohormonal pathway [45]. More recent evidence contrasts with the data of the first trials [51, 52]. When evaluated in patients with T2DM with preserved kidney function on a controlled standardized sodium diet (150 mmol/day), SGLT2i seem to be able to reduce blood pressure without a clear increase in urinary sodium excretion [51]. Therefore, the osmotic diuresis induced by them could be mainly due to an increased glycosuria rather than to an effect on natriuresis [52]. Further studies appear necessary to clarify the preload reduction mechanisms exercised by SGLT2i.
Impact on cardiac afterload. SGLT2i may affect afterload by acting on both blood pressure (BP) and arterial stiffness. In fact, SGLT2i have been shown to be one of the most effective classes of glucose-lowering drugs in reducing systemic BP [53]. The beneficial effect on BP is achieved most likely by natriuresis and osmotic diuresis, weight loss and reduction of sympathetic nervous activity [54]. EMPAREG-OUTCOME firstly assessed the role of SGLT2 inhibitors as BP lowering drugs [12], as later confirmed by 2 meta-analyses, showing a reduction of systolic BP by 2.46 mmHg and diastolic BP by 1.46 mmHg, and 24-h outpatient systolic and diastolic BP by 3.76 mmHg and 1.83 mmHg, respectively. A more significant reduction in blood pressure was also highlighted during treatment with Dapagliflozin [55], while indirect data from a metanalysis showed that Canagliflozin 300 mg better performed in reducing systolic BP than other SGLT2i, whilst no difference was observed for the diastolic pressure [56]. SGLT2i effect was more prominent during night than daytime [57], and a particular benefit using Empagliflozin was found in Asian high-risk patients with uncontrolled nocturnal hypertension [58], and black patients [59]. While the glomerular filtration rate (eGFR) affects the glycosuric function of SGLT2i, the hypotensive effect is preserved even in patients at a more advanced stage of CKD [60]. This occurs due to the persistence of both natriuretic and diuretic effects of these drugs inside the residual nephrons. Likewise, also the kaliuretic effect increases. This, together with the reduction of reducing potassium retention in the diabetic patient with CKD, contemporarily improves the sensitivity to RAAS inhibitors, with consequent potential synergic effects of these two classes of drugs in BP control [43].
Arterial stiffness can be estimated by pulse wave velocity and is associated with future CV outcomes [61]. In a small cohort of T2DM patient, after 48-h of Dapagliflozin administration a reduction of pulse wave velocity, and subsequently of the arterial stiffness, was observed. Alongside, beneficial effect of Dapagliflozin has been shown also on the endothelial function [61], as further assessed in other studies with Dapagliflozin and in in vitro models [62, 63]. Other markers of arterial stiffness (e.g., central systolic BP, forward/backward pulse wave amplitude) have been investigated with empagliflozin and dapagliflozin, with encouraging results [55, 64, 65].
Metabolic substrates modifications. Heart adenosine triphosphate (ATP)
consumption is physiologically very high due to its continuous contraction to
sustain life. About 90% of ATP production derives from fatty acids and
carbohydrates oxidation, with the further use of only a few other metabolic
substrates (e.g., ketone bodies and amino acids) to sustain ATP demand in normal
circumstances [66]. Glucose metabolism is efficient, also improving myocardial
oxygen demand. In T2DM, high free fatty acids (FFAs) levels and impaired glucose
uptake due to insulin resistance lead to a reduction in glucose oxidation.
Therefore, there is a shift in fatty acid metabolism, which becomes less
efficient. This occurs due to a higher myocardial oxygen demand, lipotoxicity
from increased free oxygen radical production and sarcoplasmic reticulum calcium
uptake impairment, thus boosting development of diastolic dysfunction [66].
Ketone bodies represent a good alternative substrate, as assessed by a study
comparing ketone infusion and placebo in patient with HF, showing an increased
stroke volume, cardiac output and LVEF [67]. SGLT2i are known to increase ketone
bodies production, mostly
Inhibition of Sodium-Hydrogen Exchange. Myocardial intracellular sodium
regulates calcium cycling which, in turn, modulates contractility, oxidative
state, and may potentially lead to arrhythmias. The balance between both
electrolytes is impaired in HF, with increased levels of myocyte intracellular
sodium. As a result, there is an increased activity of the Na
Impact on adipokines levels. Epicardial fat cells in the heart play an
important role in paracrine regulation. Adipokines production may contribute to
CAD development [77]. In depth, leptin production promotes myocardial
inflammation, while adiponectin displays anti-inflammatory and cardioprotective
effects. Adipokines are altered in obesity, insulin resistance, and T2DM, in
favour to a proinflammatory state. SGLT2i have been suggested to restore the
unpaired balance between both states, by reducing leptin levels [78]. A reduction
of serum leptin levels by Canagliflozin was demonstrates, as compared to
glimepiride [79]. In addition, also a reduction in the pro-inflammatory cytokine
interleukin-6 (IL-6) was observed, while Tumor Necrosis Factor-
Cardiac remodelling. Cardiac fibrosis is among the most important pathways in HF development. Aberrantly activated fibroblasts induce an increased extracellular matrix proteins secretion in myocardium. This alteration leads to a reduced ventricular performance and contractility [81]. In addition, also LV hypertrophy, inflammation, and cardiomyocyte cell death play an important role in cardiac remodelling. SGLT2i role in cardiac remodelling has been largely suggested. Empagliflozin have shown an attenuation in extracellular matrix remodelling and suppression of several profibrotic markers in in vitro study on human cardiac fibroblast [82]. Improvement in cardiac function and fibrosis was also assessed in in vivo studies in several animal models [83, 84, 85]. SGLT2i showed also an improvement of LV remodelling and mass, interstitial myocardial fibrosis and epicardial adipose tissue in both patients with [86, 87] or without T2DM [80]. Further studies by cardiac magnetic resonance imaging on T2DM patients with CAD showed a positive impact of Empagliflozin in reducing LV mass [88] and extracellular volume [89] after a 6-months treatment as compared to placebo.
Ischemia/Reperfusion (I/R) injury improvement. Data from animal studies
have hypothesized an improvement in I/R injury also in humans. Canagliflozin has
been shown to reduce infarct sizes, apparently in a glucose independent way [90].
Moreover, I/R injury have been suggested as mediated by the
Ca
Circulating Pro-Vascular Progenitor Cells. Pro-vascular progenitor cell depletion is believed as the underlining condition for impaired vascular repair in T2DM patients. EMPA-HEART CardioLink-6 trial showed an improvement in sub-populations of circulating pro-angiogenic progenitor cells (CD133+) and atherogenic monocytes, with high aldehyde dehydrogenase activity levels with Empagliflozin [94]. This mechanism may play a role in improving T2DM-associated regenerative cell depletion, with a benefit in CV outcomes.
Body weight. Obesity is among major independent risk factors for CV events [95]. SGLT2i are among the most effective hypoglycaemic drugs in reducing body weight [53], being able to reduce approximately 300 kcal/day, through glycosuria, which causes a 2–3 kg weight loss, regardless of background therapy. This benefit is reached during the first weeks of therapy, with the maximum effect after 6 months and a successive stabilization [96]. However, the expected weight loss would have been higher than the current reports. This imbalance between observed and expected weight-loss might be due to a compensatory increase in energy intake, as also suggested by several animal study [96, 97, 98]. In a network metanalysis, the higher weight reduction was achieved with Canagliflozin 300 mg as compared to Dapagliflozin 5 mg, whereas no difference was found for Canagliflozin 100 mg as compared to other SGLT2i [99].
Blood pressure. The role played by SGLT2i as blood lowering drugs has been already discussed in “hemodynamic effects” section. The concomitant use of SGLT2i with other antihypertensive drugs may exert a synergistic effect on both blood pressure control and cardio-renal outcomes. In fact, the kaliuretic effect of SGLT2i improves sensitivity to RAAS inhibitors, thus enhancing their ability to control blood pressure [43]. Moreover, the ability of SGLT2i to trigger a reduction in the intraglomerular pressure due to a vasoconstriction of the afferent arteriole (see below) seems complementary to RAAS inhibitors ability to induce vasodilation of the efferent arteriole, which results in a reduction of renal hyperfiltration and, consequently, a limitation of GFR decline and improvement of albuminuria degree.
Dyslipidaemia. Lipid profile disorders are common both in the general population and in T2DM individuals, with an increased CV risk. SGLT2i are associated with a slight increase in both low-density lipoprotein cholesterol (LDL-C) and high-density lipoprotein cholesterol (HDL-C), with decreased triglycerides and small dense LDL levels [100]. This issue has been largely discussed in several trials (CANVAS and EMPAREG OUTCOME) and metanalysis [12, 13, 101, 102]. It has recently been hypothesized that SGLT2i may stimulate an increase in cholesterol absorption, resulting in an increased HDL-C levels [103]. Beyond the changes in lipid serum levels, also benefit from changes in lipoprotein sub-fraction have been observed. As an example, in a Japanese study, Dapagliflozin have been associated with suppression of atherogenic small dense LDL-C and increased HDL-C and of the less atherogenic large buoyant LDL-C [104].
Hyperuricemia improvement. Uric acid plays a different role according to where it acts. In fact, in plasma it has antioxidant properties whilst, on the contrary, in the cytoplasm or in an atherogenic plaque it plays a pro-oxidant role, promoting oxidative stress and thus contributing to CVDs development. A recent metanalysis assessed that Empagliflozin manage to reduce uric acid levels when compared with controls [105]. Moreover, a sub-analysis of the EMPA-REG OUTCOME study on the association between uric acids levels and cardiorenal outcomes disclosed a reduction in hospitalization for HF and approximately a 25% decrease of CV death [43]. However, further studies are needed to better explain cause-effect relationships.
Chronic kidney disease (CKD). CKD is burdened by an increased CV risk both in the general population and in T2DM patients and will be discussed in depth below.
CKD, defined by persistent albuminuria or decline in eGFR, occurs in
approximately 35% of diabetic patients, with a subsequent significant increase
in mortality [4]. As compared to T2DM alone, presence of both T2DM and CKD is
responsible for a higher CV risk and, subsequently, for higher CV mortality rates
[106]. Moreover, an increased mortality rate was observed also in patients with
early signs of CKD progression (e.g., reduced eGFR), as well as in those with
asymptomatic microalbuminuria [107]. In fact, the presence of albuminuria
represents a significant risk factor for both CV morbidity and mortality, most of
all during the early phases of CKD, and it is also associated with the other
microvascular complications of T2DM [108, 109]. Moreover, individuals with T2DM
and CKD with proteinuria are exposed to a much higher CV risk than those without
[4]. Also poor glycaemic control seems strictly associated with diabetic
nephropathy (DN) and represents the major cause of both CKD and ESKD [110]. In
fact, lower blood glucose levels (HbA1c
The choice of the most appropriate antihyperglycemic therapeutic regimen in
individuals with diabetes is always guided by various factors (e.g., HbA1c
levels, risk of hypoglycaemia and CV risk profile). However, a pivotal role is
played by the renal function, as many antihyperglycemic drugs require a dose
reduction according to eGFR, and some are even not recommended with an eGFR
SGLT2i have been largely proven to reduce the progression of renal damage in T2DM patients (Table 1) [8, 13, 14, 15, 114, 115]. In fact, a secondary analysis of the EMPA-REG OUTCOME trial showed a significant association between Empagliflozin treatment and a lower progression of renal damage, as well as of a reduced rate of renal adverse events (–39% as for incident nephropathy or worsening of a pre-existing nephropathy and –38% as for progression of albuminuria) [114]. Empagliflozin was also associated with a 55% reduction risk of renal replacement therapy [114]. A similar finding was reported also for canagliflozin (CANVAS program), with a significant reduction of composite outcome (sustained doubling of serum creatinine, ESKD and death from renal causes) as compared to placebo [115], as further confirmed in the CREDENCE trial [14]. The DECLARE-TIMI 58 confirmed the reduction of renal outcomes also in patients treated with Dapagliflozin [15], as well as DAPA-HF TRIAL in patients with chronic HF [16]. A recent metanalysis largely confirmed the ability of SGLT2i to induce a 45% risk reduction of renal damage progression [HR 0.55; 95% CI: 0.48–0.64] and an overall reduction of composite outcome (worsening of renal function, ESKD, and mortality by renal causes) [8].
The known close relationship between the SGLT2i glycosuric effect and the renal
function at baseline has suggested the hypothesis that their impact on renal
outcomes might be affected by the degree of renal failure. The aforementioned
metanalysis also shows a maximum reduction of renal damage progression in the
individuals either without any kidney disease at baseline or with a mild kidney
disease (estimated reduction of renal outcomes of 56% with a eGFR
SGLT2i show a bi-phasic effect on eGFR, determining an initial and reversible
reduction in the short-term, followed by a substantial preservation on the
long-term rather than placebo, as shown both in EMPAREG OUTCOMES and CANVAS
trials [114, 115]. EMPA-REG OUTCOMES assessed effect of Empagliflozin on eGFR at
the beginning, during, and on the long-term after the discontinuation of the drug
[114]. After an initial reduction of eGFR during the first 4 weeks, during a
follow-up of 192 weeks we observe a stabilization of eGFR, likewise to what
reported for other classes of drugs (e.g., ACE-inhibitors). Moreover, after the
discontinuation of treatment, we can assist to a restoration of initial values of
eGFR, despite what occurs among patients treated with placebo. In fact, unlike
the initial decline (0.19
Albuminuria is both a marker of glomerular damage and a CV risk factor. Two recent trials reported a reduction in albuminuria in patients receiving either Dapagliflozin vs. placebo [119], or Empagliflozin in add-on to standard of care [120], respectively over a 104- and 52-weeks follow-up. We have already reported the 38% relative risk reduction of macroalbuminuria progression observed in the EMPA-REG OUTCOME trial, as compared to placebo, though the onset de novo was not demonstrated. Also Canagliflozin, as reported by the CANVAS Study, disclosed a 18% risk reduction of albuminuria, with modifications directly proportional to the degree of albuminuria (–9% in patients with normoalbuminuria at baseline, –34% in those with microalbuminuria and –36% in macroalbuminuric patients) [115].
However, unlike Empagliflozin, Canagliflozin was also able to reduce the
de novo onset of albuminuria. In fact, SGLT2i seem able both to prevent
the onset of proteinuria in diabetic patients and to face its worsening in
patients albuminuric at baseline. Likewise, also Dapagliflozin has been proven to
significantly reduce the degree of albuminuria (39.6% at a dosage of 5 mg and
33.9% for 10 mg, vs 15.8% of placebo) [15]. As well, the 18.9% of patients
treated with Dapagliflozin 10 mg and the 17.8% of those treated with
Dapagliflozin 5 mg displayed a reduction of albuminuria till normality ranges.
Similar results are also demonstrable in patients with CKD. In the DAPA-CKD trial
[121], Dapagliflozin has been shown to reduce the urinary albumin-to-creatinine
ratio (UACR) by 35.1% in patients with eGFR
SGLT2i improves albuminuria and slow down the progression of renal damage. This effect more likely occurs through a multifactorial mechanism, including reduced arterial and intraglomerular pressure, renal hyperfiltration, increased natriuresis, uric acid reduction, and anti-inflammatory effects (Fig. 1). The nephroprotective effect of a better glycemic compensation by SGLT2i more likely occurs in the long-term. However, it is independent of the drugs class used.
Reduced glomerular hyperfiltration via tubule-glomerular feedback. Renal hyperfiltration represents both a marker of intraglomerular hypertension and a risk factor for renal disease progression in T2DM [123]. Thus, it is a central mechanism involved in the pathogenesis of DN. SGLT2i induce a significant reduction of renal hyperfiltration [124]. The reduced renal absorption of sodium and glucose induced by SGLT2i in the proximal tubules causes an increased sodium delivery to macula densa, perceived by the juxtaglomerular apparatus as an increase of circulating plasma. The activation of this tubule-glomerular feedback is triggered by a paracrine mechanism mediated by adenosine, an afferent arteriolar vasoconstriction and a reduction in intraglomerular hypertension and renal hyperfiltration [114, 124, 125, 126]. Thus, SGLT2i mechanism of reduction of the intraglomerular pressure (afferent arteriolar vasoconstriction) seems essentially different, more likely complementary, to the one induced by RAAS (efferent arteriolar vasodilatation) [116]. The reduction of renal hyperfiltration determines a suppression of albuminuria. Moreover, the active tubular transport work caused by hyperfiltration induces oxygen consumption. The subsequent hypoxia is involved in the development of renal interstitial fibrosis. By the reduction of glomerular hyperfiltration, SGLT2i favor a reduced oxygen consumption, thus contributing to preserve renal function. Moreover, the reduction of intraglomerular pressure by SGLT2i could also trigger the suppression of both inflammation and glomerular fibrosis mechanisms, typical of DN [127].
Blood pressure reduction. A remarkable role in nephroprotection by SGLT2i is closely related to their ability to significantly reduce systemic blood pressure. Unlike the effects on the reduction of the intraglomerular pressure, which seems immediate, those on systemic blood pressure are more likely deployed on the long-term, as already largely discussed (see previous sections).
Increased erythropoietin (EPO). SGLT2i have also stimulate the renal production of EPO which, in turn, triggers an increase in haematocrit [50]. EPO has a direct nephroprotective effect by the suppression of inflammatory response and oxidative damage, which determines a reduction of tubular injury, interstitial fibrosis and inflammation, as well as attenuated albuminuria [128].
Lowering uric acid level. Uric acid plays a key role in the development of tubular damage during DN. This more likely occurs due to mechanisms mediated by either inflammation or endothelial dysfunction [129]. Therapies aimed to reduce uric acid levels during DN seem related to a lower decline of eGFR [129]. SGLT2i have been proven able to exert this role, with an approximatively 10–15% reduction rate of plasma uric acid levels thanks both to the exchange of glucose and uric acid by glucose transporter 9 (GLUT 9) and to the subsequent secretion of uric acid [105, 130]. This mechanism might thus represent a further pathway of the nephroprotection exerted by SGLT2i [105].
Glomerulosclerosis reduction. Inflammation and oxidative stress during DN stimulate the synthesis of extracellular matrix, thus triggering a pro-sclerotic state [131]. The aforementioned antioxidant and anti-inflammatory effects of SGLT2i on kidneys might contribute to a reduction of the renal glomerulosclerosis.
SGLT2i availability represents a “conceptual revolution” in the management of T2DM patients. Beyond their anti-hyperglycemic effect, SGLT2i are able to reduce CV risk and progression of kidney disease in individuals with or without diabetes. The remarkable effects observed in selected subpopulations (e.g., individuals with HF) render their use mandatory in these subjects, even at the cost of a lower HbA1c reduction.
ATP, adenosine triphosphate; BCAA, branched-chain aminoacid;
RN contributed to the study conception and design. Material preparation and data collection were performed by MA and AC. The first draft of the manuscript was written by RN, MA, AC, CB and PCP. LEA, LR, FCS, FG, PC supervised the project, editing the draft and revised the manuscript for important intellectual content. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Not applicable.
We would like to express our gratitude to all the peer reviewers for their opinions and suggestions.
This research received no external funding.
The authors declare no conflict of interest. Ferdinando Carlo Sasso is serving as one of the Editorial Board members of this journal. We declare that Ferdinando Carlo Sasso had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Massimo Volpe, Filippos Triposkiadis, Grigorios Korosoglou and Matteo Cameli.

