1 Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, No.1095 Jiefang Avenue, Qiaokou District, 430030 Wuhan, Hubei, China
2 School of Nursing, Tongji Medical College, Huazhong University of Science and Technology, 430030 Wuhan, Hubei, China
Academic Editor: Takatoshi Kasai
Abstract
Our objective was to provide evidence for exercise-based cardiac rehabilitation (ECR) for patients with aortic dissection (AD), so as to better improve the prognosis of patients and improve the quality of life (QoL) after discharge. The database PubMed, Embase, MEDLINE, Web of Science, Cochrane Library, WanFang Chinese database, ZhiWang Chinese database, Chinese Clinical Trials Registry from establishment of each database until February 2021 were included. A total of 1684 records were found by searching the database and clinical trial registry, 178 duplicate records were deleted, and 11 records met the inclusion criteria according to the screening process. We can conclude that ECR for patients with AD can effectively reduce complications and shorten the course of the disease. In addition, it is very safe because there are no serious adverse events occurring. Further research should be developed from three aspects, including the development of systematic evaluation indicators and standardized clinical exercise rehabilitation pathway, more randomized controlled trials, and the development of individualized exercise program so as to help patients with AD better improve the prognosis and QoL.
Keywords
- Aortic dissection
- Exercise-based cardiac rehabilitation
- Scoping review
Aortic dissection (AD) is a pathological change that the blood in the aortic cavity enters the aortic media from the aortic intima tear and expands along the long axis of the aorta, resulting in the separation of the true and false aortic cavities [1]. It is a serious cardiovascular emergency with the characteristics of sudden onset, rapid progress and high mortality.
In recent years, with the improvement of diagnostic methods and surgical techniques, the survival rate of AD has improved, but the medium-term prognosis is still poor [1]. A post-dissection aorta is almost invariably dilated and may thus experience greater associated wall stress, so the risk of AD recurrence, rupture and cardiovascular disease is higher in this group [2]. Generally, the quality of life (QoL) of AD patients is not ideal, their physiological function and role function are decreased [3]. In addition, patients with AD are also prone to psychological problems, such as anxiety, depression and so on [4]. Also, the growing number of survivors is usually younger, they have doubts about lifestyle, return to normal physical activity and even exercise. Therefore, too many physical restrictions will have a huge impact on their daily life [5, 6].
In 2007, American Association of Cardiovascular and Pulmonary Rehabilitation/American Heart Association (AACVPR/AHA) defined cardiac rehabilitation (CR) as a comprehensive and coordinated long-term plan, including medical evaluation, exercise prescriptions, interventions for cardiac risk factors, health education, counseling, behavioral interventions, and so on [7]. Exercise-based cardiac rehabilitation (ECR) as the core part of cardiovascular rehabilitation program [8], it can low blood pressure, delay the development of atherosclerosis, reduce the occurrence of cardiac events, control risk factors, promote mental health, and improve the QoL and the prognosis of cardiovascular disease [9, 10, 11]. Moderate physical exercise can reduce blood pressure and prevent the progression of aortic disease, but intense exercise will aggravate the expansion of aorta [12]. At present, the recommendation for type and intensity of ECR of patients with AD still represents a dilemma [13].
Therefore, our group conducted a scoping review of the literature on AD exercise rehabilitation. The key aims of this scoping review were to provide evidence for ECR for patients with AD, so as to better improve the prognosis of patients and improve the QoL after discharge.
We used the Arksey and O’Malley five step framework [14] as revised by Levac et al. [15] to outline the methods for our scoping review. The completed review is reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis extension for Scoping Reviews [16]. It was justified to summarize systematically primary research as there is no relevant summary describing the effect of ECR for AD. Findings will provide reference suggestions for ECR of patients with AD.
The research questions posed for this review were:
(1) What are the measurement indexes of ECR for AD?
(2) What are the sport types of ECR for AD?
(3) What is the safety and effectiveness of ECR for AD?
To ensure that sufficient information was captured, a search was implemented in eight databases including PubMed, Embase, MEDLINE, Web of Science, Cochrane Library, WanFang Chinese database, ZhiWang Chinese database, Chinese Clinical Trials Registry (Appendix). The search strategy included a combination of the National Library of Medicine Subject Headings (MESH), in addition to exploring keywords representing the concepts of “aortic dissection”, “exercise rehabilitation”. There were no restrictions on language, date and type of study.
The retrieved articles were imported into Endnote citation management system, and the repeated ones were eliminated. Microsoft Excel was used to screen title, abstract, and full-text.
Initially, titles and abstracts identified in the search were screened by two blinded independent authors (DNF, JK) and excluded those that did not irrelevant studies. Next, two authors read the full text independently to determine whether this study should be included. A third reviewer (SFH) adjudicated discrepancies when opinions at odds. Two authors (DNF, JK) extracted data from the included articles. The process of identification, screening, eligibility, inclusion of studies is induced in Fig. 1.
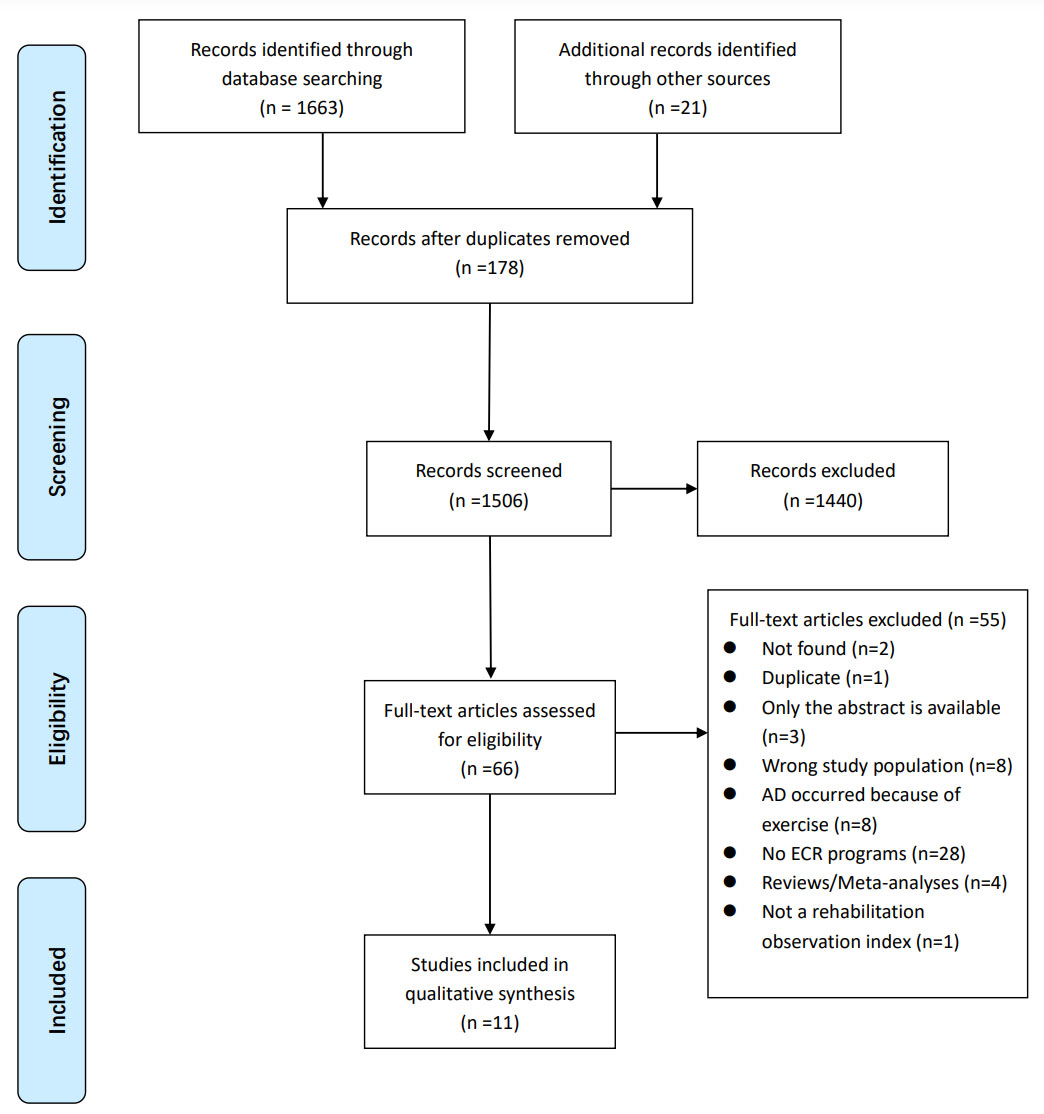 Fig. 1.
Fig. 1.PRISMA flow diagram.
Articles meeting the following conditions were included in this review: (1) research articles that included patients with AD, this review also included patients with AD and other cardiovascular diseases due to the few articles retrieved; (2) studies that carried on the ECR; (3) studies that included corresponding observation indexes; and (4) published in English and/or Chinese and included trials registering in Clinical Trials Registry. Publication date and research type were no restriction.
Articles meeting the following conditions were excluded in this review: (1) intervention contents such as therapeutic measures; (2) unable to get full text article; (3) studies were reviews or meta-analyses; (4) studies were repetitive.
All relevant papers published up to February 2021 were retrieved.
According to the guidelines for the systematic scoping review [17], the objective of the scoping review is to determine the scope and type of literature, so no quality assessment has been conducted.
The following data were extracted and classified: each individual study (first author, year, and country of publication), study type, time and location for taking ECR, major evaluation indexes, main types, study purpose, the programs of ECR, summary of results and main conclusions.
A total of 1684 records were found by searching the database and clinical trial registry, 178 duplicate records were deleted, and 11 records met the inclusion criteria according to the screening process. The process of screening and the reasons for excluding studies are presented in Fig. 1. The research contents are summarized in 1~5.
Table 1 summarizes the characteristics of the articles, including case study (n = 3), cohort (n = 6), randomized control trial (n = 2).
| Characteristic | Articles (N = 11) | |
| Year of publication | ||
| 2009 | 1 | |
| 2016 | 2 | |
| 2020 | 7 | |
| 2021 | 1 | |
| Country of publication | ||
| USA | 3 | |
| Mexico | 1 | |
| France | 2 | |
| Denmark | 1 | |
| Japan | 1 | |
| China | 3 | |
| Type of study | ||
| Case study | 3 | |
| Cohort | 6 | |
| Randomized control trial | 2 | |
| Patients with types of aortic dissection | ||
| Type A aortic dissection | 5 | |
| Type B aortic dissection | 2 | |
| Both | 1 | |
| DeBakey type I aortic disecction | 1 | |
| DeBakey type III aortic disecction | 1 | |
| Not described | 1 | |
| Time to start ECR | ||
| In hospital rehabilitation: 10–14 days after the event | 5 | |
| Out of hospital early rehabilitation: 3–6 months after the event, continued to 1 year after the event | 2 | |
| Long term rehabilitation out of hospital: 1 year after the event | 2 | |
| Location of program implementation | ||
| The local YMCA | 1 | |
| Cardiac rehabilitation centers | 3 | |
| Testing laboratory | 1 | |
| Hospital | 6 | |
Table 2 showed the evaluation indexes of patients with AD after participating in ECR. They could be divided into four parts, including indicators reflecting cardiopulmonary function (n = 8), reflecting adverse events (n = 5), reflecting in-hospital clinical course (n = 4), reflecting psychosocial function (n = 3).
| Indicators reflecting cardiopulmonary function (n = 8) | Indicators reflecting adverse events (n = 5) | Indicators reflecting in-hospital clinical course (n = 4) | Indexes reflecting psychosocial function (n = 3) |
| Aerobic threshold (AT) | Major cardio-vascular events (MCE) | Usage of oxygen (day) | Degree of satisfaction |
| Rate of perceived exertion (RPE) | Aortic event recurrences | Usage of i.v. antihypertensive agents (day) | Degree of comfort |
| Metabolic equivalents (METs) | Serious adverse events | Febrile duration (day) | Self-Rating Depression Scale (SDS) |
| Chronotropic response (CR) | Abnormal exercise tests | First defecation (day) | Self-Rating Anxious Scale (SAS) |
| Heart rate recovery (HRR) | Postoperative complications | The length of ICU stay | Quality of life (QOL) |
| Rate-pressure product (RPP) | Adverse aortic events | Postoperative hospital stay | Activities of daily living (ADL) |
| Oxygen consumption | Occurrence of pneumonia and delirium | Initiation of walking (day) | |
| Oxygen pulse (O |
ADL deterioration | Initiation of standing (day) | |
| Maximum Workload | Late adverse aortic events within 3 years after discharge | In-hospital cost (million yen) | |
| Respiratory exchange ratio (RER) | |||
| VO |
|||
| VE/VCO |
|||
| Breathing Reserve | |||
| VO |
|||
| Left ventricular ejection fraction (LVEF), % | |||
| 6 min walking ability | |||
| Exercise time | |||
| Exercise test duration, sec | |||
| Heart rate | |||
| Blood pressure | |||
| Peak breathing frequency | |||
| The Borg scale |
Table 3 showed the main types of ECR for patients with AD, including early activities (n = 3), aerobic exercise (n = 8), resistance exercise (n = 3), flexibility exercise (n = 1) and coordinated exercises (n = 1).
| Stage I rehabilitation (n = 3) | Stage II and III rehabilitation | |||
| Aerobic exercise (n = 8) | Resistance exercise (n = 3) | Flexibility exercise (n = 1) | Coordinated exercises (n = 1) | |
| Trunk control training: sitting, standing, body position transfer, balance training | Running on an elliptical or treadmill | Standing resistance exercises | Stretching exercises: bow back stretch, hand side leg press, bend up stretch, squat up stretch, bend down stretch, suspension stretch | Swiss ball, single leg standing exercises |
| Limb motor function training: range of motion, muscle strength training | Swimming | Bodyweight exercises: bodyweight squats, single leg quarter squats, bridging, quadruped, front plank, side plank, and ball hyperextension | ||
| Physical training: simple aerobic exercise based on walking | Calisthenics | Muscle strength training: bicep curls, triceps extensions, resistance bands, deadlift, single arm exercises | ||
| Respiratory function training: inspiratory muscle training | Walking | Closed kinetic chain training sessions | ||
| Stage I rehabilitation: in hospital rehabilitation; Stage II rehabilitation: out of hospital early rehabilitation; stage III rehabilitation: long term rehabilitation out of hospital. | ||||
Table 4 showed nine researches of ECR for patients with AD [18, 19, 20, 21, 22, 23, 24, 25, 26], which were described from four aspects, including the purpose of study, the programs of ECR, the effectiveness of exercise and the safety of exercise. In view of the main types of ECR were listed, so only the purpose of study, the effectiveness of exercise and the safety of exercise were discussed below.
| Author | Purpose of the study | The programs of ECR | Results | Main conclusions |
| DeFabio et al. [18] | To recover cardiac fitness and improve core stability and appendicular strength, ultimately aiming toward a potential to return to recreational sport involving short duration, high intensity activity. | Duration: 12 months | The aortic dilation demonstrated slight change, as observed on CT scan, with a maximal diameter of 5.0 mm, while the distal thoracic and abdominal aorta did not demonstrate any increase in dilation. Mood, quality of life and cardiovascular function improved. In addition, fatigability with ADL and while exercising decreased. | Monitoring HR, BP, and RPE can be safely applied in the TBAD patient. Optimal BP management with medication is essential as well as an individualized exercise prescription to maintain a beneficial cardio-vascular risk profile. |
| ① Elliptical and treadmill running: 4–7 RPE or 5–7 METs for 30 minutes at least 3X/week with brisk walking for 20–30 minutes on the alternate days. | ||||
| ② HIT (3 min) at an RPE of 7.5. | ||||
| ③ Resistance exercises: 3X/week and limited to three sets of 12–15 repetitions. | ||||
| ④ Bodyweight exercises: 1 and 3 sets of 15 reps. | ||||
| ⑤ Core stability exercises: 3–5 second with a rest interval of 60–90 seconds. | ||||
| ⑥ Single leg weight bearing exercises: 8–10 with a 90 second rest. | ||||
| Castañeda-López et al. [19] | To achieve this goal with a 96 bpm heart rate target, BORG 12, double product (Rate-pressure product) target of 12,840, and a 4 metabolic equivalents of tasks load. | With 24 sessions of aerobic and anaerobic training | ① Aerobic exercise: the maximum heart rate average of 95 bpm (99% of prescribed) and double maximum product average of 10,393 (83% of prescribed). There were no cardiovascular adverse events during training sessions. | The patient achieved an adequate tolerance to exercise and good progression on load work through cardiac rehabilitation. Physical training programs such as methods for exercise prescription (cardiopulmonary stress test) are safe. |
| ① Aerobic exercise: training sessions on a treadmill were performed with a workload progression of 3.3 METs to 5.3 during sessions. | ② Anaerobic exercise: There was also improvement in orthotic symmetry, tolerance to resistance changes, and a decrease of muscular contracture. All sessions were performed within pre-established risk parameters with no adverse events on training sessions. | |||
| ② Anaerobic exercise: including strength, equilibrium, coordination and elasticity with resistance bands, Swiss ball, and closed kinetic chain training sessions. | ||||
| Bartee et al. [20] | To recover the high-intensity | Approximately 6 months of testing | The initial setting was 24 steps per minute, and the patient ended the test at 71 steps per minute. He achieved 9 metabolic equivalents. The peak RPE was 7. During the 3 days of exercise testing, the patient had no adverse signs or symptoms that required him to discontinue any session. | Approximately 18 months after undergoing AD repair, the patient reached his goals without cardiovascular symptoms. |
| outdoor activities the patient had long enjoyed: lifting and manipulating a 50-pound suitcase, hiking, scuba diving. | ① perform a deadlift, military press maneuver, the upright row, the bicep curl. | |||
| ② complete a peak treadmill stress test and an additional metabolic stress test. | ||||
| ③ walk 15 feet toward a swimming pool, step off to enter the water, swim 164 feet, and climb a ladder to exit the pool. | ||||
| Delsart et al. [21] | To test the prognostic significance of CPET regarding aortic and cardiovascular events. | Incremental exercise tests were performed on a calibrated electromagnetically braked cycle ergometer, using a 1-min step protocol at 10 W min-1 until exhaustion with the subjects maintaining a pedaling frequency of 60 |
Among the 165 patients who underwent CPET, no adverse event was observed during exercise testing. Peak oxygen pulse was 1.46 (1.22–1.84) mLO |
CPET is safe in post AD patients should be used to not only to personalize exercise rehabilitation, but also to identify those patients with the highest risk for new aortic events and MCE not directly related to aorta. |
| Hornsby et al. [22] | To evaluate serious adverse events, abnormal CPET event rate, CRF (peak oxygen pulse, VO |
CPET was performed using an electronic/motorized treadmill until patient request to stop, general/leg fatigue, clinical decision to terminate, or a VO |
No serious adverse events were reported, although 1 abnormal exercise test (3% event rate) were observed. Median measured VO |
There were no serious adverse events with an |
| abnormal CPET event rate of only 3% 3 mo following repair for AD. | ||||
| Fuglsang et al. [23] | To assess changes in peak oxygen uptake, maximal workload, and quality-of-life after completion of an exercise-based cardiac rehabilitation program. | The ECR program started between six and twelve weeks postoperatively, had a duration of 12 weeks. The training sessions included fitness, muscle strength training and stretching. All tests were performed in using an ergometer cycle, with increases varying from 2 watts per 6 seconds to 4 watts per 8 seconds. | significant increases in peak oxygen uptake, maximal workload and quality-of-life after ended ECR. | TAAD patients have hemodynamic responses to exercise that are comparable to other cardiovascular patients. |
| Corone et al. [24] | To evaluate the feasibility of subsequent physical exercise and the incidence of events over a 1-year follow-up period in patients undergoing rehabilitation. | Warm up 5 min, aerobic activity 30 min, cooling down 5 min. Exercise on a bicycle ergometer (3 |
Systolic BP during exercise remained |
Moderate intensity, noncontact aerobic activity is likely safe and effective for selected post‐AD patients. |
| Zhang et al. [25] | To explore the effect of perioperative rehabilitation exercise guidance on the rehabilitation of patients with AD and adult congenital heart disease undergoing interventional surge | ① Slowly and forcefully lift the toes up to the maximum extent, and then let the toes down, each movement for 10s, 5 min per time, 3 times per day. | The quality of life, comfort and satisfaction of the observation group were significantly increased, while the SDS score and SAS score were significantly decreased (P |
Perioperative rehabilitation exercise training guidance can effectively reduce the complications of AD and adult patients with congenital heart disease, it is worth recommending. |
| ② Lift the waist hard, 5 seconds each time, 5 minutes each time, 3 times a day. | ||||
| ③ Raise your hips 2 cm up for 5 minutes, then slowly lower your hips 50 times a day. | ||||
| Kato et al. [26] | To confirm the usefulness and safety of our first-track rehabilitation program for UTBAAD in the acute phase. | Oral intake and assuming a sitting position from day 1 after the onset, standing by the bed from day 2, walking in their room from day 4, and discharge from day 16 if all goes smoothly. | The first defecation time of the observation group was significantly earlier than that of the control group, and the incidence of pneumonia complications was lower than that of the control group. | The fast-track rehabilitation program for patients with UTBAAD resulted in a better in-hospital clinical course and lower expense than conventional medical treatment without any adverse aortic events. |
| Abbreviations: METs, Metabolic Equivalent; HIT, High intensity training; RPE, Rate of perceived exertion; CT, Computed tomography; ADL, Activities of daily living; HR, Heart rate; BP, Blood pressure; TBAD, Type B aortic dissection; AD, Aortic dissection; CPET, Cardiopulmonary exercise testing; MCE, Major cardio-vascular events; CRF, Cardiorespiratory fitness; ECR, Exercise-based cardiac rehabilitation; TAAD, Type A aortic dissection; SDS: Self-Rating Depression Scale; SAS, Self-Rating Anxious Scale; UTBAAD, Uncomplicated type B acute aortic dissection. | ||||
In these three studies [18, 19, 20], the purposes of ECR were to recover cardiac fitness and return to the goal of high-intensity outdoor activities. Cardiopulmonary exercise testing (CPET) is the gold standard for the evaluation of exercise capacity, which can comprehensively evaluate the overall function and reserve capacity of the organ system such as the heart and lung [27]. Two studies were conducted to evaluate the safety of CPET and to test the prognostic significance of CPET regarding aortic and cardiovascular events [21, 22]. The two studies included a control group that did not perform standard ECR programs [23, 24]. The purposes of them were to evaluate the feasibility of subsequent physical exercise and to observe the effect of ECR on physical condition compared with the control group. In addition, two studies implemented early ECR, beginning after surgery [25, 26]. The objectives of the two studies were to explore the effect of perioperative rehabilitation exercise guidance on the rehabilitation of patients [25] and to confirm the usefulness and safety of our first-track rehabilitation program [26].
DeFabio et al. [18] pointed mood, QoL and cardiovascular function improved through ECR. In addition, the fatigue of activities of daily living and exercise also decreased. ECR can achieve an adequate tolerance to exercise and good progression on load work [19]. A study in France found patients with type A dissection have hemodynamic responses to exercise that are comparable to other cardiovascular patients, also patients who participated in CPET and ECR program at the same time with better significant increases in peak oxygen uptake, maximal workload and quality-of-life [23]. ECR can also increase the mean baseline maximum physical work capacity [24]. A study in China showed perioperative rehabilitation exercise training guidance can effectively reduce the complications of AD, improve the comfort of patients, the QoL of patients, get higher satisfaction [25]. A study in Japan also showed the early rehabilitation can get better in-hospital clinical course such as first defecation time, duration of fever, length of hospital stay and reduced the cost of hospitalization [26].
In the three case studies [18, 19, 20], the patient achieved the exercise goal without adverse events. Castañeda-López et al. [19] pointed physical training programs such as methods for exercise prescription (cardiopulmonary stress test) are safe. CPET is safe in post AD patients should be used to not only to personalize exercise rehabilitation, but also to identify those patients with the highest risk for new aortic events and major cardio-vascular events not directly related to aorta [21].
Corone et al. [24] also pointed moderate intensity, noncontact aerobic activity is likely safe and effective for selected post-AD patients. What’s more, Kato et al. [22] found that there were no any adverse aortic events during the fast-track rehabilitation program for patients with uncomplicated type B acute aortic dissection (UTBAAD) [26]. However, a study in USA reported no serious adverse events but an abnormal exercise test (about 3% event rate) were observed during the process of test.
Table 5 showed two studies from the clinical trial registry. Although the research has not yet been completed, the purpose of is also to evaluate the effectiveness and safety of early bicycle training as well as early rehabilitation. The intervention contents of rehabilitation included early mobilization, cycling, cardiopulmonary rehabilitation by physiotherapist.
| Name | Reference ID | Source | Purpose of the study | Physical rehabilitation programs |
| Early mobilization of postoperative patients with Stanford type A acute aortic dissection: a real-world study | ChiCTR2000039559 | Chinese Clinical Trials Registry | To evaluate the effect and safety of early cycling training after TAAAD surgery. | Early mobilization and cycling |
| Early rehabilitation to acute standford type A aortic dissection after surgery | ChiCTR2000034123 | Chinese Clinical Trials Registry | To explore the effectiveness and safety of early rehabilitation to TAAD after surgery. | Cardiopulmonary rehabilitation by physiotherapist or self-administrated pulmonary rehabilitation |
| Abbreviations: TAAAD, Type A acute aortic dissection; TAAD, Type A aortic dissection. | ||||
The key aims of this scoping review were to provide evidence for ECR for patients with AD, so as to better improve the prognosis of patients and improve the QoL after discharge. Findings make an essential contribution to provide guidances and references for further development of ECR for patients with AD. We read 66 full texts and found that most of the articles were excluded because no ECR was implemented (n = 28). In addition, we found that 8 articles reported that the occurrence of AD was due to exercise. Only 11 eligible studies were included in this scoping review, the publication time of these articles were relatively new, randomized controlled trials were few, and these researches were mostly concentrated in Europe, Asia and North America, showing the paucity of literature on this topic. However, we also found that the recent literatures on this topic are gradually increasing, and the clinical trial registration center has also carried out correlative researches on this aspect, which shows that the ECR of patients with AD is more and more concerned by the medical community, and it is a topic worthy of study. Finally, we introduced the scoping review from the five aspects of description of the articles, evaluation indexes of ECR, main types of ECR, nine researches on ECR of AD, two studies from the clinical trial registry.
The most important point of postoperative treatment of AD is to control blood pressure, which is very important to avoid acute adverse events and chronic aortic dilatation [28]. Studies have reported that regular exercise can significantly reduce blood pressure [28]. Therefore, we should encourage patients with AD to take regular exercise [29]. In the process of exercise, the measurement indexes are very important for evaluating the curative effect. It is well known that the rehabilitation of cardiovascular diseases is a comprehensive medical management model. Multiple outcome criterion (MOC) score standard [30] is a way to measure the effect of CR, it considers that the evaluation standard of CR efficacy should be taken as a whole, which includes cardiovascular disease risk factors, exercise ability and subjective health. Subjective health assessment included anxiety, depression, pain, physical health and mental health [31]. It is pointed out in the guidelines that patients with AD often have some cardiovascular risk factors. Even if the patients are screened before and during the whole training, unexpected complications may still occur in the process of exercise training [7]. Therefore, we should consider the incidence of adverse events in the evaluation process. What’s more, Kato et al. [26] reported the fast-track rehabilitation program for patients with UTBAAD and put in-hospital clinical course into the evaluation index. Based on the above aspects, this scoping review divided the evaluation indexes of ECR into the following aspects, including indicators reflecting cardiopulmonary function, adverse events, in-hospital clinical course, psychosocial function. However, there is still a lack of a systematic measurement index for the rehabilitation of AD. This aspect needs to be further studied by researchers in the future.
According to the guideline of CR and secondary prevention revised in 2007, CR
can be divided into three stages: stage I rehabilitation (in hospital
rehabilitation), stage II rehabilitation (out of hospital early rehabilitation),
stage III rehabilitation (long term rehabilitation out of hospital) [7]. The goal
of each stage is different, stage II is the key period for patients to recover,
the formulation of individualized exercise prescription in real sense starts from
stage II rehabilitation [7]. The composition of the prescription includes
exercise form, intensity, time and frequency [7]. The classic exercise
rehabilitation procedure includes three steps: preparation, training and
relaxation. The types of exercise include aerobic exercise, impedance exercise,
flexibility exercise and coordination exercise [32]. Dynamic exercise can
increase cardiac output, heart rate, only a small increase in systolic blood, and
diastolic blood pressure remained stable [33]. Conversely, static exercise can
lead to a significant increase in systolic and diastolic blood pressure, but a
small increase in cardiac output and heart rate [34, 35]. Therefore, this type of
exercise would seem to be contraindicated for these patients. However, Spanos in
the European Journal of Vascular and Endovascular Surgery pointed that patients
in AD can maintain normal physical and sexual activities, including lifting
weights to 50% of body weight, as well as all daily activities [36]. Thijssen
et al. [37] recommend patients perform mild-moderate activities, and
avoid heavy static activity. Chaddha encouraged patients with AD to take
moderate-intensity aerobic exercise (3–5 METs) for
CPET is a method of objectively and quantitatively assessing cardiorespiratory
reserve function [39]. It is widely used in a variety of cardiovascular diseases,
such as in small abdominal aortic aneurysm [40], chronic heart failure [41], and
so on. CPET is a combination of exercise and gas metabolism testing technology,
based on the principle of coupling internal respiration and external respiration
[42]. The core measurement indicators of CPET include VO
According to the scoping review, ECR for patients with AD is safe and the probability of abnormal events is very low. However, due to the particularity of AD group, having the characteristics of sudden onset, rapid progress and high mortality. For those older patients with underlying diseases, even if they are screened comprehensively before training, it is still possible that unexpected complications occur [7]. Therefore, we should carefully observe the patient’s condition during the whole process of ECR in order to identify the emergency situation in time. AACVPR requires medical staff to update their knowledge and skills every 3 years [45]. What’s more, CR needs multidisciplinary cooperation, a complete team should include cardiologists, specialist nurses, pharmacists, physiotherapists, nutritionists and clinical psychologists [7]. Therefore, it is very necessary to train all the medical staff involved in ECR on the core competence of training rehabilitation, teach them to master the skills of high-risk electrocardiogram identification as well as sports risk identification and accept the training of life support treatment technology. It is also essential to formulate the corresponding warning symptoms, emergency plan processing flows and place first-aid equipment in corresponding CR places, so as to ensure the safety and effectiveness of ECR [7].
The prognosis of patients with AD after discharge still has some problems and can be improved if appropriate measures are taken. This review has proved the safety and effectiveness of ECR for patients with AD. Our results should promote future research in three aspects. First of all, systematic evaluation indicators and standardized clinical exercise rehabilitation pathway are warranted to measure the effect of ECR and improve the prognosis of patients. Secondly, more randomized controlled trials are needed to fill the gaps in this field. Finally, due to the cultural differences in different regions, we need to take more assessment methods (such as CPET) to assess the exercise tolerance and maximum exercise load of patients, so as to help patients develop more individualized exercise programs and ensure the safety of exercise.
AACVPR, American Association of Cardiovascular and Pulmonary Rehabilitation; AD, Aortic dissection; AHA, American Heart Association; CPET, Cardiopulmonary exercise testing; CR, Cardiac rehabilitation; ECR, Exercise-based cardiac rehabilitation; MESH, Medicine Subject Headings; MOC, Multiple outcome criterion; QoL, Quality of life; TAAD, Type A aortic dissection; UTBAAD, Uncomplicated type B acute aortic dissection.
DNF: conception of the study, major drafting of the work, final approval and agreeing to the accuracy of the work. SFH: conception of the study, major drafting of the work, final approval and agreeing to the accuracy of the work. JK: help in design of the study, drafting of the work, final approval and agreeing to the accuracy of the work. XRL: supervision, critical revision of the manuscript, final approval and agreeing to the accuracy of the work. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
Not applicable.
The authors acknowledge the role of all support staff in the study.
This research was funded by the National Natural Science Foundation of China, grant number 71874063.
The authors declare no conflict of interest.
The process of searching on PubMed:
#1 “aneurysm, dissecting” [MeSH Terms] OR Dissecting Aneurysm[Title/Abstract] OR Aneurysms, Dissecting[Title/Abstract] OR Dissecting Aneurysms [Title/Abstract] OR Dissection, Blood Vessel[Title/Abstract] OR Blood Vessel Dissection[Title/Abstract] OR Aortic Dissection[Title/Abstract] OR Aortic Dissections[Title/Abstract] OR Dissection, Aortic[Title/Abstract] OR Dissections, Aortic[Title/Abstract]
#2 “Exercise Therapy” [MeSH Terms] OR Remedial Exercise[Title/Abstract] OR Exercise, Remedial[Title/Abstract] OR Exercises, Remedial[Title/Abstract] OR Remedial Exercises[Title/Abstract] OR Therapy, Exercise[Title/Abstract] OR Exercise Therapies[Title/Abstract] OR Therapies, Exercise[Title/Abstract] OR Rehabilitation Exercise[Title/Abstract] OR Exercise, Rehabilitation[Title/Abstract] OR Exercises, Rehabilitation[Title/Abstract] OR Rehabilitation Exercises [Title/Abstract] OR Sport[Title/Abstract] OR Exercise[Title/Abstract] OR Training[Title/Abstract] OR Physical fitness[Title/Abstract] OR Physical activity[Title/Abstract]
#3 #1 AND #2

