Academic Editor: Federico Ronco
There are limited data regarding the use of angiotensin converting enzyme inhibitors/angiotensin receptor blockers (ACEi/ARBs) in acute heart failure (AHF). The purpose is to determine the patterns of ACEi/ARB use at the time of admission and discharge in relation to invasive hemodynamic data, mortality, and heart failure (HF) readmissions. This is a retrospective single-center study in patients with AHF who underwent right heart catheterization between January 2010 and December 2016. Patients on dialysis, evidence of shock, or incomplete follow up were excluded. Multivariate logistic regression analysis was used to analyze the factors associated with continuation of ACEi/ARB use on discharge and its relation to mortality and HF readmissions. The final sample was 626 patients. Patients on ACEi/ARB on admission were most likely continued on discharge. The most common reasons for stopping ACEi/ARB were worsening renal function (WRF), hypotension, and hyperkalemia. Patients with ACEi/ARB use on admission had a significantly higher systemic vascular resistance (SVR) and mean arterial pressure (MAP), but lower cardiac index (CI). Patients with RA pressures above the median received less ACEi/ARB (P = 0.025) and had significantly higher inpatient mortality (P = 0.048). After multivariate logistic regression, ACEi/ARB use at admission was associated with less inpatient mortality; OR 0.32 95% CI (0.11 to 0.93), and this effect extended to the subgroup of patients with HFpEF. Patients discharged on ACEi/ARB had significantly less 6-month HF readmissions OR 0.69 95% CI (0.48 to 0.98). ACEi/ARB use on admission for AHF was associated with less inpatient mortality including in those with HFpEF.
The inhibition of the Renin Angiotensin Aldosterone System (RAAS) plays a key role in reducing morbidity and mortality in patients with heart failure, particularly in those patients with reduced ejection fraction (HFrEF). The benefits of the RAAS inhibition (RAASi) have been proven in landmark trials resulting in their incorporation as key elements of guideline directed medical therapies (GDMT) for HFrEF [1, 2, 3]. While there is less certainty on the benefits with RAASi in patients with heart failure with preserved ejection fraction (HFpEF), there may be a potential role in reducing hospitalizations and mortality with careful patient selection [4].
There are limited high-quality data to guide use in terms of initiation, continuation, or withdrawal of ACEi/ARB in the setting of acute heart failure, which is typically characterized by escalated and maladaptive activation of the RAAS [5] regardless of underlying ejection fraction (EF). While continuation of RAASi therapy after hospital discharge in patients with HFrEF has been associated with lower mortality and readmission rates in prior observational studies [6], there is still a high rate of discontinuation of ACEi/ARB therapy during AHF due to complications such as worsening kidney function, hypotension, hyperkalemia, and angioedema [5]. Therefore, we aimed to investigate further the patterns of ACEi/ARB use among patients with AHF, reasons for stopping these agents, hemodynamic changes associated with use, and impact on clinical outcomes of mortality and HF readmissions.
This study is a retrospective single center analysis of patients with clinical diagnosis of acute heart failure (AHF)-this included patients with characteristic symptoms of shortness of breath/orthopnea/paroxysmal nocturnal dyspnea and with clinical evidence of volume overload on physical examination as well as on imaging based on records review who were admitted and underwent right heart catheterization (RHC) at any point during the index admission at Einstein Medical Center, Philadelphia between January 2010 to December 2016. We included patients that had complete hemodynamic parameters from the concomitant RHC, and information on medication use including initiation of ACEi/ARBs during admission and discharge. We excluded patients with end stage renal disease (ESRD) and those who presented with hemodynamic or clinical criteria of shock (including cardiogenic shock) as these conditions reflect where the use of ACEi/ARB or other guideline directed medical therapy for heart failure therapy are limited see (Fig. S1). Patients who died were excluded from the HF readmissions rate analysis. The study protocol was approved by the local Institutional Review Board.
De-identified demographic, clinical, and cardiac catheterization hemodynamic
data were collected by review of medical records. Serum creatinine values on the
day of admission and discharge were obtained; and eGFR was calculated using the
CKD-EPI equation. Heart failure with preserved ejection fraction was defined as
EF
Descriptive statistics were used to analyze the demographic and
outcome variables. Chi square tests were used for categorical variables.
Demographic data were compiled and compared between patients discharged on or off
ACEi/ARBs. Differences in hemodynamic measurements were compared using
independent t-tests among patients on ACEi/ARBs started on admission compared to
those not on these medications. For significantly different means, analysis of
covariance was used to adjust for age, gender, race and ejection fraction. Rates
of HF related readmissions at 1 month and 6 months were compared between patients
discharged on or without ACEi/ARBs using Chi square. Multivariate logistic
regression analysis was used to analyze factors associated with ACEi/ARB
continuation on discharge. The multivariate logistic regression between mortality
or HF readmissions and ACEi/ARB use on discharge was performed after adjusting
for age, race, gender, diabetes, hypertension, other comorbidities,
EF
Of the final sample of 626 patients see (Fig. S1), mean age
(+/-SD) was 66.92
| Discharge with ACE/ARB (n = 314) | OFF ACE/ARB (n = 312) | P value | |
| Mean Age (years old |
65.54 |
68.31 |
0.01 |
| Gender | 0.472 | ||
| Male, n (%) | 168 (53.5) | 157 (50.3) | |
| Female, n (%) | 146 (46.5) | 155 (49.7) | |
| Race | |||
| African American, n (%) | 64.6 | 51.6 | |
| Caucasian, n (%) | 26.4 | 35.9 | |
| Hispanic, n (%) | 6.7 | 4.8 | |
| Others, n (%) | 2.2 | 7.7 | |
| Diabetes, n (%) | 149 (47.6) | 155 (50) | 0.575 |
| Hypertension, n (%) | 286 (91.4) | 266 (85.8) | 0.032 |
| Coronary Artery disease, n (%) | 143 (45.5) | 143 (46.1) | 0.936 |
| Atrial fibrillation, n (%) | 67 (21.3) | 78 (25.2) | 0.297 |
| Smoking, n (%) | 116 (37.5) | 95 (31) | 0.106 |
| Ejection fraction EF |
143 (45.5) | 238 (76.3) | |
| BMI | 32.89 |
32.41 |
0.875 |
| eGFR on admission | 73.60 |
58.33 |
0.024 |
| eGFR on discharge | 74.72 |
59.85 |
0.001 |
| Medications used (%) | |||
| ACE/ARB | 229 (72.9) | 69 (22.1) | |
| Beta blocker use | 257 (82) | 228 (73) | 0.01 |
| Diuretics | 232 (52.3) | 212 (47.7) | 0.141 |
| Nitrates | 78 (25) | 66 (21.3) | 0.296 |
| Hydralazine | 63 (20) | 71 (23) | 0.436 |
| Digoxin | 33 (10.5) | 19 (6.1) | 0.059 |
| MRA | 53 (17) | 23 (7.4) |
There were 298 (47.6%) patients that were on ACEi/ARBs at home prior to admission. Eighty percent of patients on ACEi/ARB were continued on these agents on admission and 77% were discharged on it (Fig. 1). In the other hand, 328 patients were not on ACEi/ARB at home. 42 (13%) patients were started newly on ACEi/ARB on admission and 85 (26%) of these patients were discharged on ACEi/ARBs (Fig. 2). Among those on ACEi/ARB at home and those newly started during the hospitalization, majority of the reasons for discontinuation were worsening renal function (WRF) (60%), hypotension (31%), and hyperkalemia (2%) (Fig. 3).
 Fig. 1.
Fig. 1.Rates of Inpatient Use of ACEi/ARB and Continuation on Discharge among Patients with ACEi/ARB use at home (far left bar graph). The rates of ACEi/ARB continued use on admission and on discharge parallel the rates of usage at home and are on the succeeding bar graphs respectively.
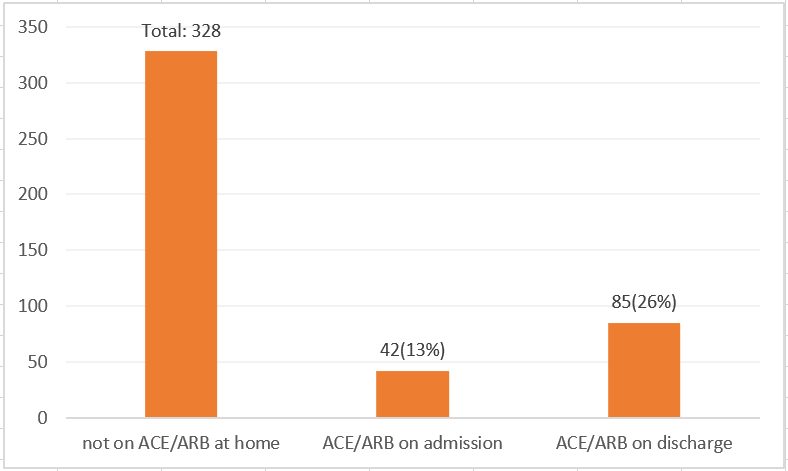 Fig. 2.
Fig. 2.Rates of Inpatient Use of ACEi/ARB and Continuation on Discharge among Patients not on ACEi/ARB use at home (far left bar graph). This graph shows lower rates of ACEi/ARB use on admission and on discharge among those not originally on ACEi/ARB at home.
 Fig. 3.
Fig. 3.Reasons for Stopping ACEi/ARB on Admission with AKI and hypotension as the leading causes.
Independent factors associated with continuation of ACEi/ARBs on discharge were analyzed using multivariate logistic regression analysis. Existing ACEi/ARB use at home was highly associated with ACE/ARB on discharge OR 9.68 95% CI (6.36-14.76). Higher eGFR on discharge was also correlated with ACEi/ARB use on discharge OR 1.014 95% CI (1.008 to 1.021). Ejection fraction (EF) was inversely associated with continuation of ACEI/ARB wherein higher EF was associated with less odds of ACEi/ARB use on discharge OR 0.96 95% CI (0.946 to 0.972). Racial minority (non-African American, non-Caucasian or non-Hispanic ethnicity) also predicted less ACEi/ARB on discharge OR 0.3 95% CI (0.101- 0.897) (Table 2).
| OR | 95% CI | P value | |
| Age | 0.999 | 0.982 to 1.015 | 0.863 |
| Male | Referrant | ||
| Female | 1.309 | 0.854 to 2.005 | 0.216 |
| African American | Referrant | ||
| Caucasian | 0.677 | 0.424 to 1.082 | 0.103 |
| Hispanic | 0.697 | 0.290 to 1.674 | 0.419 |
| Others | 0.301 | 0.101 to 0.897 | 0.031 |
| eGFR on discharge | 1.014 | 1.008 to 1.021 | |
| EF | 0.958 | 0.946 to 0.972 | |
| Diabetes | 0.721 | 0.475 to 1.095 | 0.125 |
| Hypertension | 1.175 | 0.607 to 2.273 | 0.633 |
| On ACEi/ARB at home | 9.684 | 6.355 to 14.759 |
The right atrial pressures (RAP) were elevated in both groups (on ACEi/ARB vs no
ACEi/ARB) with mean of 12.05
| Hemodynamics (mean |
ACE/ARB on admission (n = 346) | OFF ACE/ARB (n = 280) | P value |
| RA pressure | 11.71 |
12.33 |
0.264 |
| mPAP | 31.7 |
31.92 |
0.816 |
| PCWP | 19.88 |
19.98 |
0.892 |
| Transpulmonary gradient | 12.18 |
12.29 |
0.882 |
| PVR (dynes-sec-cm-5) | 224.99 |
223.49 |
0.921 |
| SVR (dynes-sec-cm-5) | 1598.31 |
1431.94 |
0.002 |
| SVR adjusted (Standard error) | 1573 (38.06) | 1459 (34.24) | 0.027 |
| MAP | 96.32 |
91.07 |
|
| MAP adjusted (Standard error) | 96.34 (0.99) | 91.34 (0.90) | |
| CI | 2.22 |
2.48 |
0.001 |
| CI adjusted (Standard error) | 2.24 (0.056) | 2.47 (0.050) | 0.003 |
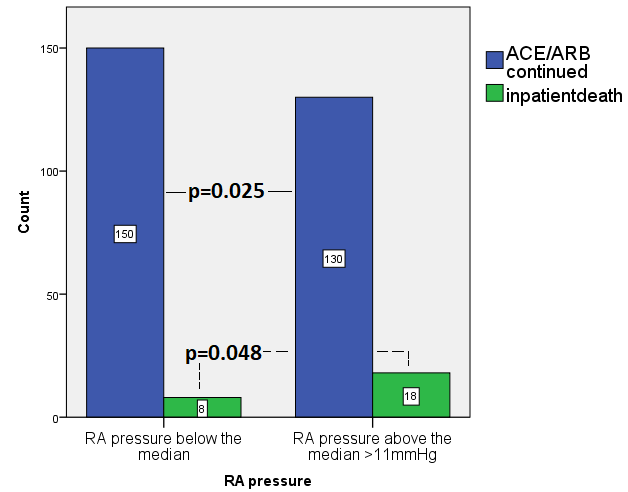 Fig. 4.
Fig. 4.ACEi/ARB use and Inpatient Mortality Stratified by Median RA pressure showing that among patients with higher RA pressure above the median, there was a significantly lower rates of continued ACEi/ARB use but higher rates of inpatient death.
There were 26 recorded-inpatient deaths in this cohort. 39% were from acute coronary syndromes, 23% were from respiratory arrest related to heart failure, 19% were from heart failure with multiorgan failure, 15% were from arrhythmias and 4% were non-cardiac causes. After adjusting for demographics and baseline comorbidities, patients who were on ACEi/ARB at home had a significantly lower odds of inpatient mortality OR 0.32, 95% CI (0.11 to 0.93) (see Table S1). In a subgroup analysis looking at patients with HF with preserved EF (HFpEF), patients on ACEi/ARB had lower inpatient deaths compared to those without (1.3% vs 5.4% P = 0.037). On multivariate logistic regression, ACEi/ARB use among patients with HFpEF was still significantly associated with lower inpatient mortality OR 0.10, 95% CI (0.01 to 0.908) P = 0.041 (see Table S2).
Among those who survived and were discharged without ACEi/ARB in the full
cohort, 136 (22.7%) were readmitted within 1 month and 255 (42.5%) were
readmitted within 6 months. There was a significantly lower rate of 1-month HF
readmission among patients discharged on ACEi/ARB (23.6% vs 36.2%) P = 0.032
while 6-month HF readmission rates were borderline significant (38.9% vs 46.5%)
P = 0.069. In analysis of a composite of 6-month readmissions and inpatient
mortality, those with RA pressure above the median and without ACEI/ARB had
significantly higher number of poor outcomes 55% vs 38% P
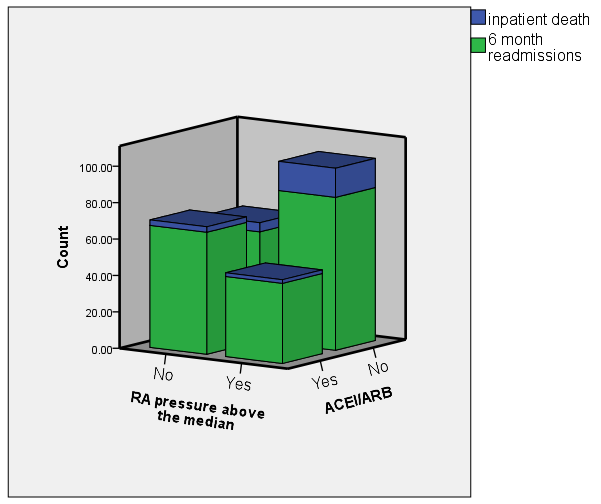 Fig. 5.
Fig. 5.Outcomes of inpatient death and 6 month readmissions stratified by ACEi/ARB use and RA pressure above or below the median. This figure shows more rates of inpatient death and 6 month readmissions in the subset with both RA pressure above the median and at the same time not on ACEi/ARB.
After adjusting for age, gender, comorbidities, eGFR at discharge and HF medication use, ACEi/ARB on discharge was associated with lower rates of 6-month HF readmission OR 0.69 95% CI (0.48 to 0.98) and borderline significant for 1-month HF readmission OR 0.66 (0.44 to 1.02) (Tables S3,4).
Our study found that ACEi/ARB use was associated with significantly lower inpatient mortality as well as lower 6 month HF readmission rates among patients admitted with AHF. This mortality benefit also extended to the subgroup of patients with HFpEF with acute HF exacerbation. While the clinical benefits in the use of ACEi/ARB for patients with chronic HFrEF are well established [2, 3], there are less high-quality data guiding the management of RAASi in patients admitted with acute HF. This results in marked heterogeneity in clinical practice with respect to continuation, escalation or cessation of RAASi in acute HF in patients with HFrEF [5]. There is also less evidence supporting routine use of RAASi in patients with chronic HFpEF, which remains difficult to treat due to its different phenotype and unique multifactorial pathophysiology [7, 8, 9, 10, 11]. Various studies have shown that ACEi/ARB mainly showed benefits in reduced HF hospitalizations, but no significant effect on all-cause mortality and cardiovascular mortality [9, 10, 11]. However, in a meta-analysis, the use of ACEI in HFpEF showed an all-cause mortality benefit (RR = 0.91, 95% CI = 0.87-0.95) [4]. Several factors may contribute to the lack of demonstrable benefits with RAASi in HFpEF including the lack of a clear consensus on the definition of HFpEF which can lead to significant heterogeneity in clinical profiles, exercise capacity, and relevant clinical end points [12]. It should also be noted that the major trials looking at ACEi/ARB in patients with HFpEF were not done in the setting of AHF [9, 10, 11]. In an analysis of the DOSE-AHF (Diuretic Optimization Strategies in Acute Heart Failure) and CARRESS-HF (Cardiorenal Rescue Study in Acute Decompensated Heart Failure) trials [13, 14] by Mentz et al., although patients with HFpEF tended to have lower baseline aldosterone levels compared to HFrEF, biomarkers for RAAS activation (plasma renin activity and serum aldosterone levels) were nevertheless elevated [15], suggesting that at least in AHF, there is a similar mal-adaptive activation of the RAAS. This provides plausibility to the hypothesis that RAASi may be beneficial in AHF with potential to modify the vulnerable post hospitalization course regardless of underlying EF. In addition to the effects of ACEi/ARBs directly on the RAAS system, ACEi were also shown to decrease inflammatory chemokines such as interleukins that may be useful in targeting the inflammatory pathway of heart failure and cardiorenal syndrome [16]. ACEi inhibit bradykinin breakdown which may have actions including vasodilation, fibrinolysis, and inhibition of cell growth that may contribute to its HF benefits but at the same time side effects such as cough and angioedema [17]. Meanwhile, angiotensin type I receptor blockade with ARBs can lead to more angiotensin II receptor stimulation which also has vasodilatory and antiproliferative effects that may be beneficial with less associated adverse effects from the bradykinin system [17, 18]. However, whether clinical outcomes among patients who received ACEi vs ARBs were different in this analysis due to plausible neurohormonal effects and/or any component of the effects of ACEi/ARB on the inflammatory pathway in HF needs further study.
Serial measurements by Packer et al. showed that ACEi acutely lowered right and left ventricular filling pressures, mean arterial pressure, heart rate, systemic and pulmonary vascular resistance in severe HF. RAASi decreases preload and afterload through blocking vasoconstrictive effect of angiotensin II [19]. However, hemodynamic parameters in our study revealed patients in AHF (in the absence of cardiogenic shock) and on ACEi/ARB therapy had significantly higher mean SVR and MAP, and lower cardiac index compared to those not on these medications. Although temporal relationships between these hemodynamic parameters and ACEi/ARB use could not be established in this study, patients in AHF usually have precipitating factors that lead to decompensation. Especially in patients with HFpEF, higher blood pressure reflecting a higher SVR and afterload may trigger AHF wherein ACEi/ARB use is appropriate and potentially therapeutic [20]. We also found that patients with RAP above the median received less ACEi/ARB but had significantly higher rates of inpatient mortality and readmissions. This is consistent with previous studies that an elevated central venous pressure or right atrial pressure was directly associated with renal outcomes and all-cause mortality in patients with cardiovascular disease [21, 22]. These findings also suggest that there is really no standardized clinical guidance on the use of ACEi/ARB in patients with AHF and oftentimes, these medications are suspended. These patients with high right atrial pressure likely have concomitant cardiorenal syndrome [23] and that these findings reflect a reflexive tendency of clinicians to indiscriminately stop these agents due to concerns for WRF, despite the potential for optimizing the neurohormonal changes seen with acute HF with the use of these drug classes.
The main reasons for ACEi/ARB discontinuation in our study were azotemia (60%), hypotension (31%), and hyperkalemia (2%). Our findings are consistent with multiple prior studies have described same factors for withdrawal or as barrier for initiation of RAASi [5, 6, 24, 25]. Perceived risk for or real decline of renal function is one of the common triggers for RAASi withdrawal in clinical practice. However, elevation of serum creatinine and BUN to a greater extent in AHF likely only reflect hemodynamic changes in the setting of cardiorenal syndrome, and not necessarily intrinsic renal damage [26]. Elevated right sided filling pressures (as what was seen in our sample population), translating to elevated renal venous pressures and subsequent decreased renal blood flow, coupled with elevated levels of RAAS and neurohormonal activation leading to preglomerular vasoconstriction may lead to the reduced GFR characteristic of cardiorenal syndrome [23]. This pathophysiologic condition actually benefits from careful ACEi/ARB administration rather cessation. In the CONSENSUS trial, there was a marked reduction in HF-associated mortality and symptom burden [3] and that trends of rise in serum creatine were mostly during early phase and returned within 30% of baseline thereafter [27]. A meta-analysis by Flather et al. on ACEi in AHF showed that discontinuation was rarely necessary despite higher rates of azotemia in the treatment versus placebo arms [28]. The critical need to ensure optimal delivery of ACEi/ARB, especially during and after vulnerable period of AHF despite elevation of serum creatinine, are missed opportunities to improve clinical outcomes with these patients [5].
In the setting of WRF and AHF, biomarkers of intrinsic kidney injury are valuable in clinical decision-making on RAASi use [23]. Validated biomarkers of tubular injury, urine microscopy and urinary sediment analysis are simple and readily available tests to distinguish intrinsic AKI (typically with superimposed sepsis or shock) from functional changes in serum creatinine [5, 23]. The use of commercially available biomarkers of tubular injury such as serum and urine NGAL (neutrophil gelatinase-associated lipocalin), and the combination of TIMP-2 and IGFBP7 (tissue inhibitor of metalloproteinase-2 and insulin-like growth factor-binding protein 7 respectively) can detect early intrinsic tubular injury and may play a role when they are negative in ensuring appropriate continuation of RAASi in the setting of AHF despite benign fluctuations in markers of glomerular injury such as serum cystatin and serum creatinine [29, 30].
Hyperkalemia is another legitimate yet infrequent reason for discontinuation of ACEi/ARB in this study. Collins et al. demonstrated that the presence of HF considerably increased fatal risks of hyperkalemia in patients on RAASi therapy [31]. In the Hyperkalaemia Randomized Intervention Multidose SZC Maintenance (HARMONIZE) trial, a subgroup analysis in patients with HF and hyperkalemia showed that use of sodium zirconium cyclosilicate achieved normokalemia in 93% of patients within 48-h without the need to adjust the RAASi doses [32]. Hence, the use of novel oral anti-hyperkalemic agents such as sodium zirconium cyclosilicate or patiromer can prove as valuable tools in optimizing ACEi/ARBs use in AHF [33, 34].
Finally, contextual prescription patterns seem to be a factor in the continuation of ACEi/ARB use in AHF. Our study found that independent of baseline co-morbidities, prior usage was a factor that is strongly associated with discharge therapy of ACEi/ARB. The same pattern of use was reported in a large observational data using Get With the Guidelines-Heart Failure (GWTG-HF) registry, to which 88.5% usage of ACEi/ARB prior to hospitalization persisted from admission through discharge of AHF in those with reduced EF [35]. Physician reluctance to alter a patient’s existing medical regimen may have contributed to these findings [35]. Continued physician and provider education about the benefits of continuation of ACEi/ARB is warranted to achieve improved outcomes in AHF.
Our study has all the limitations of a retrospective and single-center analyses. Data were collected from electronic medical records and some laboratory values such as the actual potassium levels were not provided and included as part of the analyses. The temporal relationships between the invasive hemodynamic parameters and use of ACEi/ARBs cannot fully be established. Data on whether ACEi/ARBs were stopped midway during the course of admission was not available and may potentially influence some clinical outcomes. ACEi/ARBs were also analyzed together and differences in the mechanism and relation of ACEi compared to ARB in heart failure and cardiorenal syndrome pathophysiology may also influence outcomes. Overall patient compliance to GDMT was not assessed. The specific doses used for each individual ACEi/ARBs were not considered in this analysis. Compliance with the other components of the guideline-directed medical therapy for heart failure was not a part of the scope of this study but were partly accounted for their effect in the multivariate analyses of the outcomes. Use of other anti-hypertensive medications such as calcium channel blockers and centrally acting agents were not accounted for and may influence blood pressure and heart failure outcomes. Our institution predominantly serves patients from the low socio-economic stratum, compliance to GDMT and diuretics is a limitation. Time to diuretic use on admission was also not documented. Nevertheless, this study has a reasonably large sample size with females (48%) and African Americans (58%) adequately represented. These are the population groups who are usually under-represented in cardiovascular studies [36]. This study is also the first to our knowledge to explore hemodynamic parameters in relation to ACEi/ARB use and quantify evidence that use of ACEi/ARB in the setting of AHF can have clinical benefits across strata of ejection fraction.
Patients on ACEi/ARB at home are most likely to continue these medications on admission and upon discharge after AHF. Most common causes cited for withdrawal of RAASi while in-hospital were WRF, hypotension, and hyperkalemia. ACEi/ARB use on admission was associated with significantly less inpatient mortality while ACEi/ARB on discharge was associated with less 6-month HF readmissions, including in patients with HFpEF.
All authors were involved with study conceptualization and planning. KBL, HMT, GS, JJ, RB, EQ collected data, KBL summarized and analyzed the data. KBL GS and EQ drafted the first version. All authors contributed edits, read and approved the final manuscript version.
The study was approved by the Institutional Review Board at Einstein Medical Center Philadelphia IRB-2020-431. Waiver of consent was done for this retrospective study.
There are no acknowledgments to disclose.
None declared, no funding, this study was approved by our local Institutional Review Board. All authors included in this manuscript contributed significantly in various aspects of the study from study conceptualization, analysis, drafting and review or editing of the manuscript.
