1 Haikou Xiuying Center for Disease Prevention and Control, 570311 Haikou, Hainan, China
2 Haikou Xiuying District Maternal and Child Health Institution, 570311 Haikou, Hainan, China
3 Business and Technology Department, Hainan Academy of Inspection and Testing, 570100 Haikou, Hainan, China
4 Institute of Food Testing, Hainan Academy of Inspection and Testing, 570100 Haikou, Hainan, China
5 Qionghai Branch of Hainan Academy of Inspection and Testing, 571442 Qionghai, Hainan, China
6 Danzhou Branch of Hainan Academy of Inspection and Testing, 571700 Danzhou, Hainan, China
Abstract
Excessive residues of organophosphorus pesticide pose a serious threat to consumer health and negatively impact the export trade of agricultural products in China. Therefore, establishing a rapid, accurate and reliable multiresidue detection method for organophosphorus pesticides to safeguard consumer health is highly necessary.
This study aimed to develop a combined detection method integrating surface-enhanced Raman spectroscopy (SERS) with molecularly imprinted polymer (SERS-MIP) technology. Quantitative analysis of two pesticides, trichlorfon and cypermethrin, as examples, enables rapid detection of pesticide residues in fruits and vegetables. The effects of four fruits and vegetables (oranges, tangerines, leeks, and spinach) on the detection of two pesticide residues were also investigated, with a focus on matrix effects on the SERS-MIP combined technique.
The limit of detection (LOD) for trichlorfon in oranges and tangerines was 1.27 μg/kg, with recovery rates ranging from 95.2% to 101.4%. The LOD for cypermethrin in leeks and spinach was 0.13 μg/kg, with recovery rates ranging from 95.2% to 100.3%. For all four matrices, the method enabled rapid quantitative detection with high specificity and sensitivity, meeting current market demands.
This approach holds promise for detecting other pesticide residues and provides a useful reference direction for developing rapid pesticide residue testing methods.
Keywords
- organophosphorus pesticides
- surface-enhanced Raman spectroscopy
- molecular imprinting
- solid-phase extraction
Trichlorfon is an organophosphorus insecticide with high efficiency and broad spectrum insecticidal characteristics. It is widely used in agriculture to control chewing mouthparts and biting mouthparts of agricultural, forestry, horticultural pests, underground pests, etc. [1, 2]. Cypermethrin is an organochlorine insecticide, mainly used to control lepidoptera pests that occur on crops. It is highly toxic and has a high insecticidal activity against many kinds of pests. It can be applied to various fruit trees, fruits and vegetables, etc., and has a good killing effect on many kinds of pests [1, 3]. In agricultural production, these two pesticides are usually used in combination, which has a synergistic effect [4].
At present, the major detection methods for the residues of these two pesticides mainly include chromatography, immunoassay, and sensor-based methods [2, 5]. Among these methods, chromatography demands relatively complex sample pretreatment procedures. Additionally, it incurs high costs, requires expensive instrumentation, and necessitates professional technical expertise [6, 7]. Immunoassay methods, on the other hand, have a long cycle. The biological activity of antibodies is susceptible to inactivation due to multiple influencing factors [8, 9]. Moreover, the preparation of pesticide antibodies involves the synthesis of complete antigens and immunization of animals, which poses challenges for industrial production [10, 11]. In most of the current biosensors, biomolecules (such as antibodies and enzymes) are utilized as recognition elements [12]. However, these biomolecules often exhibit poor stability and limited repeatability in detection. Chemical sensors typically rely on the polarity differences of analytes for detection, resulting in a lack of specificity in their detection capabilities [13, 14]. Consequently, the development of sensitive materials with specific recognition capabilities for the analytes of interest has emerged as a crucial aspect in the research and development of novel detection methods [15, 16].
In recent years, the number of articles published on the application of surface-enhanced Raman spectroscopy (SERS) for detecting pesticide residues has increased exponentially [17, 18, 19, 20]. Different pesticides possess distinct molecular structures and chemical bond configurations, resulting in varying intermolecular forces. These differences can be effectively distinguished through the characteristic peak positions, number of peaks, peak shapes, and peak intensities in SERS spectra. Combined with mathematical modelling, this approach establishes relationships between pesticide concentrations and characteristic peak intensities, thereby enabling both qualitative and quantitative analysis of pesticide residues in fruits and vegetables using SERS technology [4, 12]. Lin et al. (2018) [21] employed SERS to determine thiabendazole pesticide residues in rapeseed. They established a predictive model that links SERS spectral data to thiabendazole pesticide levels in rapeseed via partial least squares regression.
Molecular imprinting technology mimics the mechanism of antibody-antigen interaction. When the template molecule (imprinting molecule) comes into contact with the functional monomer, multiple interaction sites are formed [11, 22]. Through the polymerization process of the cross-linking agent, this interaction is memorized. After the template molecule is removed, stereospecific cavities complementary to the template molecule in terms of spatial structure, size and binding sites are left in the polymer network structure. Such cavities exhibit highly selective recognition characteristics for the template molecule and its analogues [23, 24]. In recent years, molecular imprinting has been used as a recognition unit to specifically recognize and bind target molecules, and combined with different types of transducers to produce molecularly imprinted sensors [25, 26]. It not only has the advantages of high sensitivity and simple, rapid operation of chemical sensors, but also has the advantages of mechanical stability, thermal stability, long service life and multiple reusability of molecularly imprinted polymers [16, 27]. Therefore, it has shown broad application prospects in the field of pesticide residue detection and has become an important research hotspot [11, 16].
With the rapid socio-economic development in China, there has been increasing public concern regarding quality of life, particularly heightened attention toward food quality and safety [7, 12, 15]. However, due to the extensive application of organophosphorus pesticides (OPs) in crop production and cultivation, the issue of OP pesticide residues exceeding maximum limits has emerged as a significant concern [23, 24, 25]. Although China has established stringent maximum residue limits (MRLs) for organophosphorus pesticides in fruits and vegetables, fruits, and other agricultural commodities, the misuse and overuse of these chemicals, along with violations involving restricted or banned OPs, remain prevalent. Moreover, there has been an increasing frequency of detecting multiple OP residues in produce [13, 22, 23]. The problem of excessive OP pesticide residues not only poses serious threats to consumer health but also adversely affects the export trade of Chinese agricultural products. Therefore, it is imperative to establish rapid, accurate, and reliable multi-residue detection methods for organophosphorus pesticides to ensure public health protection and enhance food safety monitoring [8, 23, 25].
Based on molecularly imprinted technology, this study establishes a rapid and effective method for the detection of multiple trace-level organophosphorus pesticide residues by combining molecularly imprinted solid-phase extraction with high-performance liquid chromatography (MISPE-HPLC). The proposed method not only significantly enhances detection sensitivity and lowers the limits of detection, but also represents a novel exploration into the application of molecularly imprinted solid-phase extraction technology for multi-residue analysis of organophosphorus pesticides.
This manuscript is the first to employ surface-enhanced Raman spectroscopy for the qualitative and quantitative analysis of two pesticides, trichlorfon and cypermethrin. Subsequently, by integrating surface-enhanced Raman spectroscopy with molecularly imprinted polymer technology, it conducts pesticide residue detection in actual vegetable samples. This approach overcomes the limitations of conventional SERS detection and offers broad research value and application prospects for achieving highly specific and sensitive SERS detection.
Trichlorfon (purity 90%), cypermethrin (purity 4.5%), methacrylic acid, ethylene glycol dimethacrylate, azobisisobutyronitrile, methanol, glacial acetic acid, chloroform, silver nitrate, trisodium citrate and acetone were purchased from Shanghai Aladdin Biochemical Technology Co., Ltd. High-purity nitrogen (99.999%) was purchased from Hainan Jiateng Chemical Gas Co., Ltd. Oranges, mandarins, spinach and leeks were purchased from Nanguo Supermarket, Haikou, Hainan Province.
The template molecules (0.2867 g of Trichlorfon and 9.2244 g of Cypermethrin) were dissolved in 12 mL chloroform in a three-neck flask, followed by the addition of 2.756 g of methacrylic acid (8 mmol) as the functional monomer. The mixture was stirred at room temperature for 30 minutes to ensure thorough mixing. Ethylene glycol dimethacrylate (3.964 g, 20 mmol) was added as the crosslinking agent, followed by 400 mg of azobisisobutyronitrile as the initiator. The reactions were configured with 0.5 mmol of template monomer, a template:functional monomer:crosslinker ratio of 1:4:20 (n:m:n), 0.44 mmol of initiator, and a pore-forming agent consisting of a mixed solution of acetonitrile/methanol (volume ratio = 3/1). After thorough mixing, the flask was sealed and sonicated for 15 mins. After nitrogen gas was introduced for 15 minutes to remove oxygen, the flask was sealed and placed in a 60 °C water bath for 24 h [11, 12].
Soxhlet Extraction for Removal of Pesticide Template Molecules from the Polymer: After the polymerization process was completed, an amber-colored solid was obtained. The solid was thoroughly ground in a mortar and sieved through a 200-mesh screen. The resulting powder was tightly wrapped in a filter paper thimble and placed into the extraction chamber of a Soxhlet apparatus. A mixture of methanol and glacial acetic acid (120 mL:40 mL, 3:1 v/v) was used as the extraction solvent. The extraction system was assembled and heated in a water bath at 93 °C. Siphoning occurred approximately every 20 min. After 12 h of continuous extraction, the thimble was removed. The extracted polymer was then washed with methanol until neutral pH was achieved and dried in an oven at 50 °C for 12 h to obtain the final molecularly imprinted polymer (MIP) [15, 16, 22].
The non-imprinted polymer (NIP) was prepared following the same procedure, except that no pesticide template molecule was added during the polymerization.
The surface morphologies of trichlorfon, the molecularly imprinted polymer (MIP), and the non-imprinted polymer (NIP) were characterized using scanning electron microscopy (SEM) to evaluate structural and topological features. A drop of nanometer compound was taken from the copper-covered network with membrane, subjected to natural air and 2% phosphotungstic acid dye for 30 min in the copper network carrying the nanometer compound. A filter paper was used to absorb the excess dye. Then, the sample was scanned with a transmission electron microscope using an acceleration volt-age of 80 kV [11, 12].
FTIR spectroscopy was employed to characterize trichlorfon, MIP, and NIP. By comparing the stretching vibration peaks of specific functional groups in the infrared spectra, the elution efficiency of the pesticide molecules was evaluated. The samples’ infrared spectrum analysis was performed using the KBr method, with a wave number in the range of 400 to 4000 cm-1. Scanning was conducted for 32 times at a resolution of 4 cm-1. This analysis also helped determine whether chemical bonding occurred between the trichlorfon molecules and the functional monomer, methacrylic acid [28, 29].
A standard aqueous solution of trichlorfon or cypermethrin at a concentration of 300 mg/L was accurately prepared. All experiments were performed in triplicate: 20 mg of molecularly imprinted polymer (MIP) was weighed into seven 50-mL volumetric flasks, followed by the addition of 10 mL of the 300 mg/L trichlorfon or cypermethrin standard solution. The flasks were shaken in an incubator at room temperature for periods ranging from 5 to 240 mins. After shaking, the mixtures were centrifuged at 4000 rpm for 30 mins. The supernatant was then carefully collected, and its absorbance was measured at 190 nm using a UV-Vis spectrophotometer. The same procedure was applied in parallel to evaluate the non-imprinted polymer (NIP) [10, 12].
To evaluate the adsorption performance of the two types of molecularly imprinted polymers, equilibrium binding studies were conducted with trichlorfon and cypermethrin. Exactly 20 mg of MIP was weighed into a 50-mL volumetric flask, and 10 mL of trichlorfon or cypermethrin solution at concentrations ranging from 0 to 300 mg/L was added. The mixtures were oscillated at room temperature for 4 hours and then centrifuged at 4000 rpm for 15 minutes. The supernatant was filtered through a 0.22 µm aqueous-phase membrane, and a 50 µL aliquot was injected for HPLC analysis at a detection wavelength of 200 nm [15, 23].
Measure 15 mL of 0.01 mol/L silver nitrate standard solution into a 150 mL volumetric flask. Dilute to volume with ultrapure water. Add a clean, dry magnetic stir bar and heat while stirring at 100 °C. Once boiling commences with uniform bubble formation, slowly add 3 mL of 1% trisodium citrate solution dropwise while maintaining stirring and heating for approximately 30 minutes. Cease heating and allow the mixture to cool naturally to room temperature. Take 40 g of the solution and centrifuge at 3500 rpm for 2 min to remove larger silver nanoparticle particles. Ultrasonically disperse the supernatant for 20 min to ensure thorough mixing, yielding the AgNPs. Store away from light [19, 30].
Five silver nitrate solutions were prepared at different concentrations: 0.2,
0.6, 1.0, 1.4, and 1.8
Raman and SERS spectra of the trichlorfon and cypermethrin standard solutions
were acquired using a HORIBA HR Evolution microconfocal Raman spectrometer. Prior
to the experiment, the instrument was calibrated using a silicon wafer. A 5
µL aliquot of AgNPs was mixed with 5 µL of the test solution on a
microscope slide, thoroughly mixed, and the slide was placed on the stage after 2
min. Exposure to light was avoided throughout. Measurements were performed using
the Raman spectrometer to collect the spectra. This method was applied to each
concentration of the pesticide solutions. Unless otherwise specified, the
spectral acquisition parameters were as follows: laser wavelength, 532 nm; laser
intensity, 50%; laser spot diameter, 200 µm; grating, 600 grooves/mm
(centre wavelength 500 nm); and a 50
Common leeks, which may retain residues of trichlorfon and cypermethrin, were selected as the actual samples for testing. The leek samples were spiked with known amounts of both pesticides. By combining the MIPs to adsorb the pesticide molecules with the prepared AgNPs as the SERS enhancement substrate, SERS spectra were acquired from the pesticide-contaminated leek samples. The leeks were rinsed with ultrapure water, air-dried in a fume hood for 30 min, immersed in the pesticide solutions for 5 min, removed and then left in a cool place for 24 h. This procedure ensured that the spiked pesticide concentrations closely approximated real-world contamination levels [17, 33].
Three grams of each of the two prepared pesticide-specific molecularly imprinted
polymers (MIPs) were weighed and dissolved in 3 mL of methanol. Chopped leeks
were placed on a glass slide, 5 µL of the molecularly imprinted polymer
solution was added, and the MIPs were allowed to adsorb fully onto the sample and
dry. Subsequently, 5 µL of AgNPs was added. After 2 min, surface-enhanced
Raman spectroscopy signals were collected from ten uniformly distributed points
on the sample surface. Measurements were taken multiple times at each
concentration, and the average value was selected once the signal intensity has
stabilised; this average was used as the final measurement result. The instrument
parameters were as follows: laser wavelength, 532 nm; laser intensity, 50%;
laser spot diameter, 200 µm; grating centred at 500 nm; and a 50
All experiments were performed in triplicate. Data were analyzed using SPSS 22.0
software (IBM Corp., Armonk, NY, USA). One-way ANOVA and paired sample
t-tests were employed to assess significant differences between groups.
A p-value
The molecularly imprinted polymers (MIPs) synthesized using trichlorfon and cypermethrin as template molecules, along with the non-imprinted polymer (NIP), were characterized by SEM. As shown in Fig. 1, molecularly imprinted polymer shown in the left figure has a microspherical surface with a large specific surface area. It is porous and has a rough, uneven texture. This morphological difference can be attributed to the interactions between the pesticide template molecules and the functional monomer during polymerization, leading to the formation of numerous microspheres complementary in size and shape to the templates [23, 25]. In contrast, the NIP, synthesized in the absence of template molecules, lacks such specific cavities, resulting in a comparatively smoother surface with fewer pores.
 Fig. 1.
Fig. 1.
Scanning electron micrographs of molecularly imprinted polymers. Scanning electron micrographs of molecularly imprinted polymers
(A) (
The FTIR spectra of the MIP, NIP, and trichlorfon are shown in Fig. 2. In the spectrum of MIP (Fig. 2A), the absorption peak at 1240.84 cm-1 corresponds to the stretching vibration of P=O [17]. Both the MIP (Fig. 2A) and NIP (Fig. 2B) show absorption near 1730 cm-1, attributed to the C=O stretching vibration. 2962.02 corresponds to C-H stretching vibrations of methyl (-CH3) or methylene (-CH2) groups. A broad absorption peak observed at 3503.93 cm-1 in the MIP spectrum (Fig. 2C) indicates the presence of O–H stretching vibrations due to intermolecular hydrogen bonding. This suggests that the P=O group of trichlorfon interacted with the –OH group of methacrylic acid, confirming successful binding between the mixed templates and the functional monomer during MIP synthesis [11, 15]. The similarity between the FTIR spectra of the MIP and NIP (Fig. 2A,B) indicates analogous molecular structures, confirming the effective removal of template molecules from the MIP.
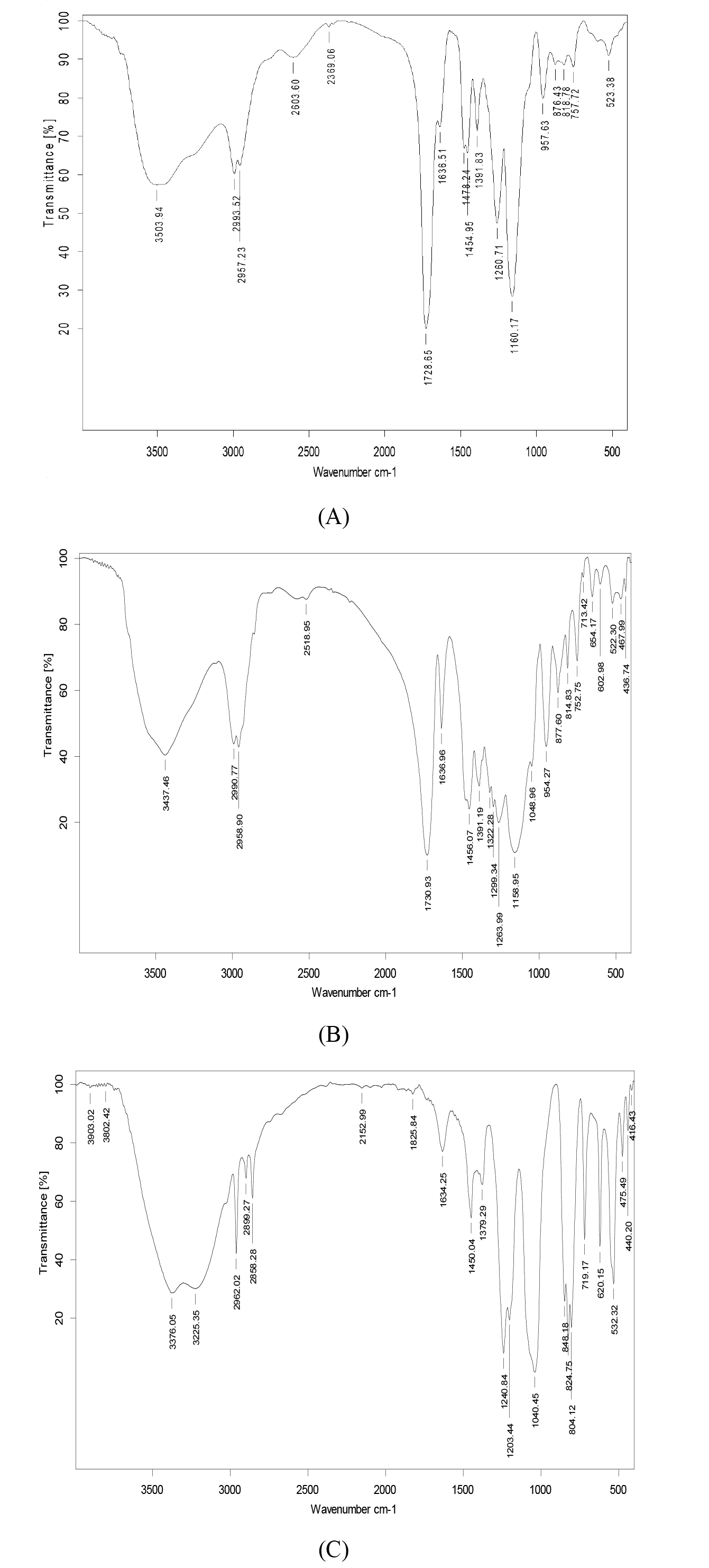 Fig. 2.
Fig. 2.
Infrared characterization of polymers and pesticides. Infrared characterization of polymers and pesticides. Note: (A) Molecularly imprinted polymer; (B) non-imprinted polymer; (C) trichlorfon.
The adsorption capacity was measured at different time intervals (Fig. 3). The results demonstrate that the adsorption capacity of the MIP increased over time, reaching a maximum value of 32.24 mg/g, indicating high adsorption affinity for trichlorfon [23, 25].
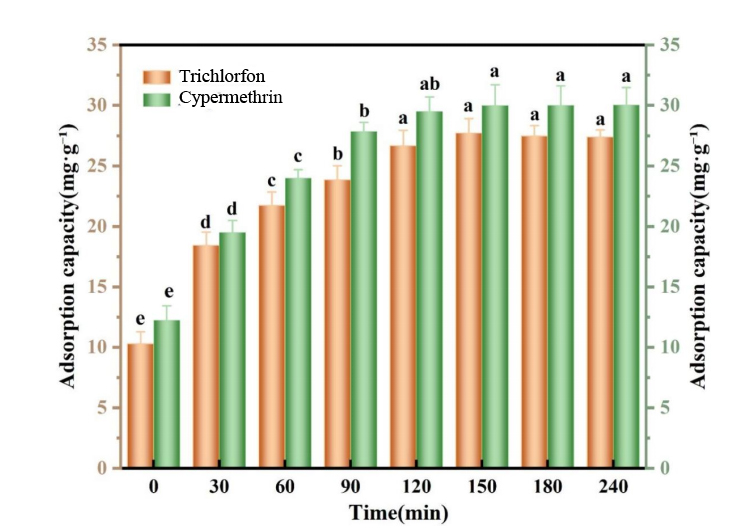 Fig. 3.
Fig. 3.
Kinetic uptake plots of the imprinted polymer towards trichlorfon and cypermethrin at 300 mg/L. Kinetic uptake plots of the imprinted polymer towards trichlorfon and cypermethrin at 300 mg/L. Note: Lowercase letters indicate a 0.05 significance level.
As shown in Fig. 3, after 30 minutes of shaking, the adsorption capacities of the common MIP toward trichlorfon and cypermethrin were 17.4 mg/g and 18.9 mg/g, representing 61.3% and 62.4% of the saturated adsorption capacity, respectively. Adsorption equilibrium was essentially reached within 180 mins. These results suggest that the common MIP exhibits rapid adsorption kinetics for organophosphorus pesticides and can be effectively used as a sorbent in matrix solid-phase dispersion extraction for rapid extraction of such pesticides from samples [24, 26].
The specific adsorption performance of Tri-MIPs and Cyp-MIPs was verified using
solutions of different pesticide molecules. Although trichlorfon and cypermethrin
are both benzene-containing compounds with similar molecular structures,
cross-adoption experiments were conducted followed by SERS detection. After
elution, Tri-MIPs, Cyp-MIPs, and the corresponding non-imprinted polymers (NIPs)
were separately exposed to 1
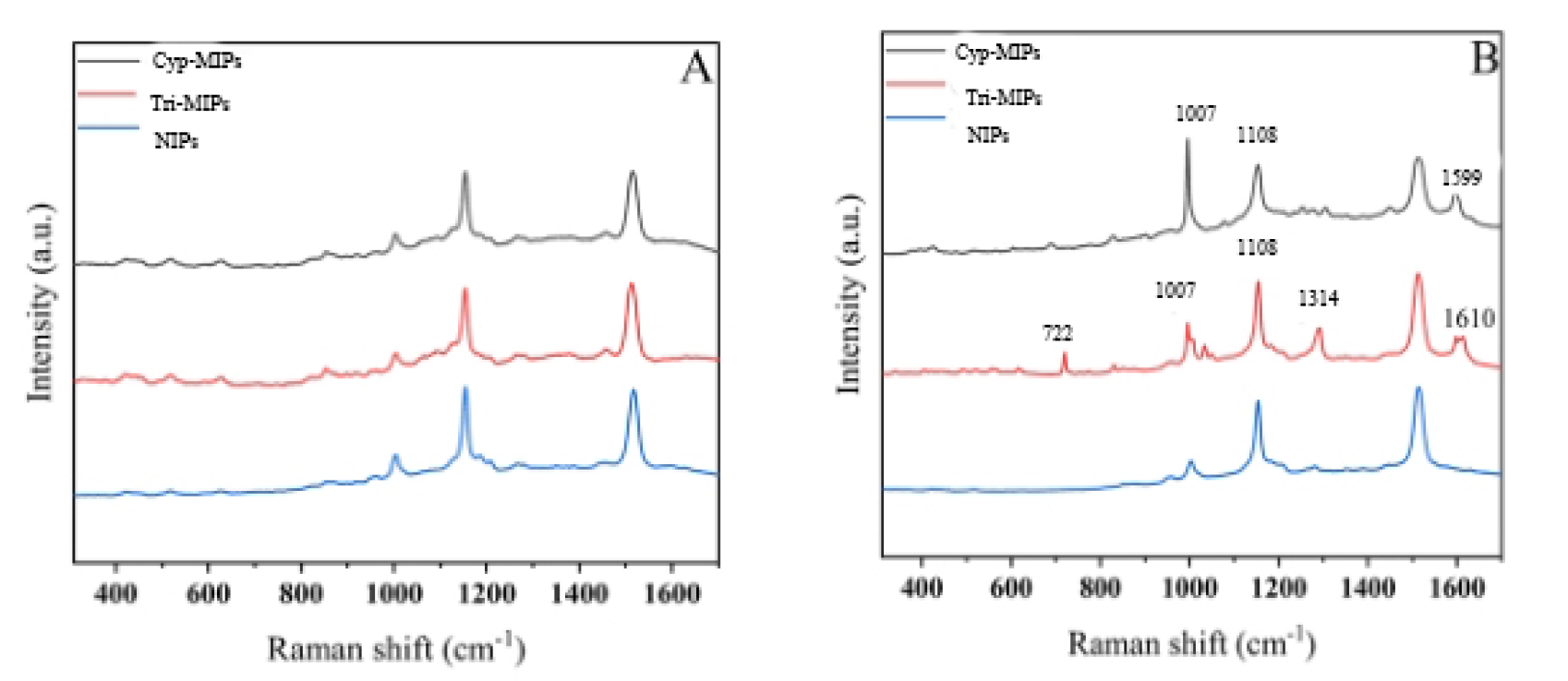 Fig. 4.
Fig. 4.
SEM images of MIP and non-MIP. (A) Raman spectra of Cyp-MIPs adsorb Cypermethrin, Tri-MIPs adsorb Trichlorfon, NIPs adsorb mixed solution. (B) Raman spectra of CIPC-MIPs adsorb CIPC, OPP-MIPs adsorb OPP, NIPs adsorb high concentration mixed solution. MIP, molecularly imprinted polymer; NIP, non-imprinted polymer; CIPC, Chlorpropham; OPP, O-Phenylphenol.
To evaluate the feasibility of the SERS-MIP method for rapid pesticide detection and to validate the practical application of the prepared MIPs and AgNP-enhanced substrates in real vegetable samples, spiked samples were analysed. Chinese chives were selected as the matrix for spiked recovery experiments. SERS spectra were acquired ten times for each spiked concentration.
Fig. 5A shows the SERS spectra of trichlorfon spiked into leeks at different
concentrations. Spectra a–e correspond to trichlorfon spiking concentrations of
1
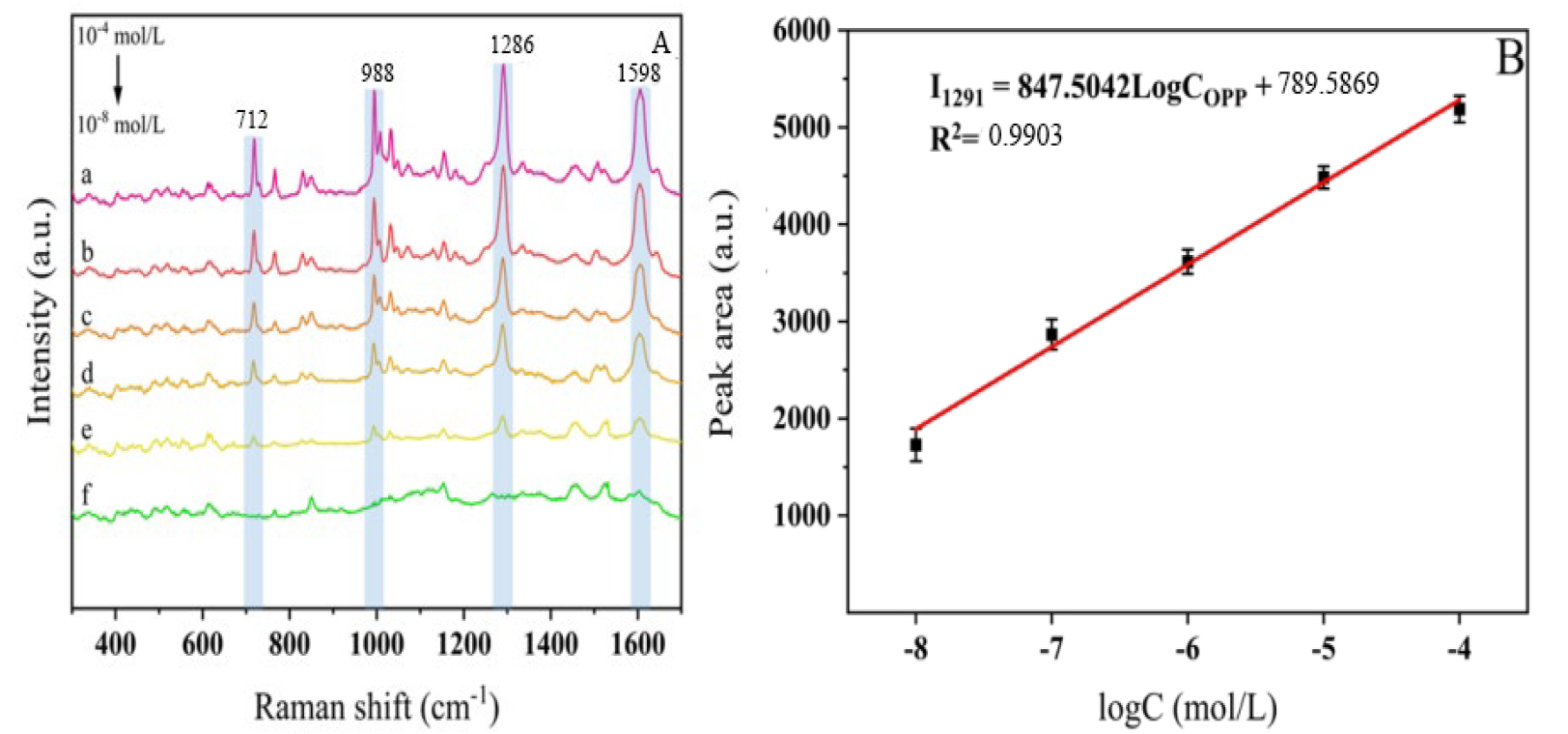 Fig. 5.
Fig. 5.
FTIR spectra of polymers and trichlorfon. (A) SERS spectra of trichlorfon solution with various
concentrations on leek. From a to e: 1
As shown in Fig. 6A, spectra a–g represents the SERS spectra of cypermethrin
spiked into leeks at concentrations ranging from 1
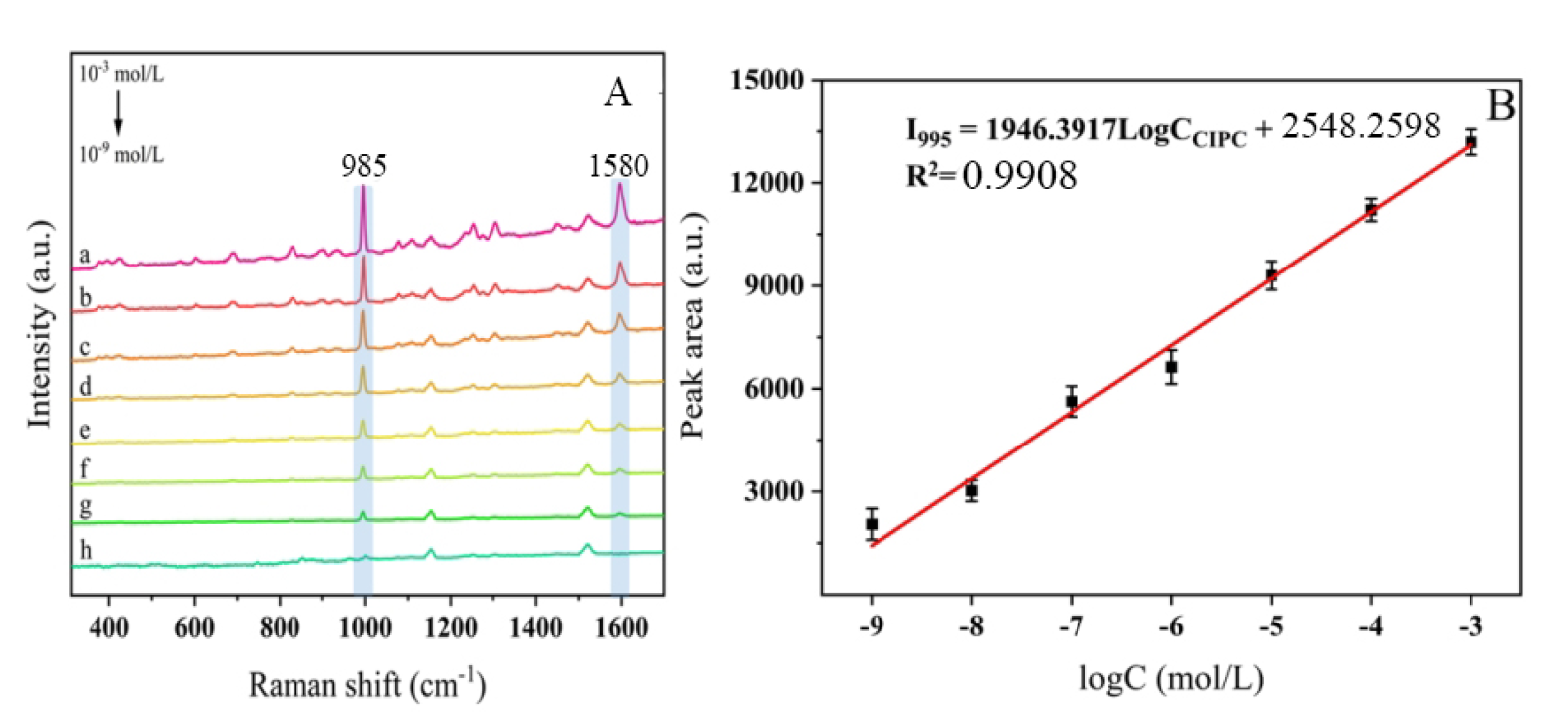 Fig. 6.
Fig. 6.
Kinetic uptake plots of MIP towards pesticides. (A) SERS spectra of cypermethrin solution with various
concentrations on Chinese chives. From a to g: 1
To determine the enhancement effect of AgNPs on the pesticides trichlorfon and
cypermethrin, enhancement factors were calculated based on the most prominent
characteristic peaks of the two pesticides at 1286 cm-1 and 995 cm-1,
respectively. Following the procedures described above, optimal enhancement
substrates for each pesticide were prepared, and their SERS spectral signals were
acquired. For trichlorfon, the surface-enhanced Raman spectroscopy detection
limit (CSERS) was 1.1
To validate the accuracy and precision of the calibration curves for various practical samples, three different concentrations of trichlorfon and cypermethrin standard solutions (2 µg/L, 5 µg/L, and 10 µg/L) were added to each of the four samples mentioned above. with eight parallel replicates for each concentration. After adsorption onto MIPs, the samples were enhanced using an AgNPs substrate. SERS spectra were collected, and corresponding concentrations were calculated by integrating the characteristic peak intensity into the standard curve. Recovery rates and RSD were calculated for each of the four fruit and vegetable matrices. Results are shown in Table 1. Recovery rates for oranges ranged from 95.2% to 101.4% with RSDs of 3.1% to 7.5%; for mandarins, 97.2% to 101.3% with RSDs of 1.4% to 4.3%; leeks showed recovery rates of 95.2%–98.1% with RSDs of 2.3%–3.1%; spinach exhibited recovery rates of 96.1%–100.3% with RSDs ranging from 3.2% to 5.0%. All four samples exhibited satisfactory recovery rates and low RSD values, yielding calibration curves with excellent accuracy. The experimental results comply with the requirements of national standards for physicochemical testing, indicating that the MIPs combined with SERS detection is suitable for the quantitative determination of trichlorfon and cypermethrin in real samples.
| Sample | 2 µg/L | 5 µg/L | 10 µg/L | |||
| Recovery/% | RSD/% | Recovery/% | RSD/% | Recovery/% | RSD/% | |
| Oranges | 95.2% | 7.5% | 101.4% | 3.5% | 98.1% | 3.1% |
| Mandarins | 97.2% | 2.0% | 97.5% | 1.4% | 101.3% | 4.3% |
| Leeks | 95.2% | 3.1% | 98.1% | 2.8% | 97.6% | 2.3% |
| Spinach | 96.1% | 5.0% | 100.3% | 3.9% | 98.7% | 3.2% |
RSD, Relative Standard Deviation.
The MIP coupled with SERS detection method established in this study was compared with other reported methods for detecting trichlorfon and cypermethrin, with results shown in Table 2 (Ref. [37, 38, 39, 40]). It can be seen that the SERS-MIP technology developed in this study exhibits high sensitivity for detecting both pesticides. It requires no cumbersome pretreatment, demonstrates strong specificity and high sensitivity in complex real-world systems, and necessitates only minimal sample quantities—covering the laser spot suffices for detection. This significantly reduces economic costs. Utilizing a portable Raman spectrometer enables real-time, rapid detection in outdoor settings.
| Pesticide | Method | LOD | LOQ | Reference |
| Tri | SERS-MIP | 1.27 µg/kg | 1.67 µg/kg | This work |
| LC-MS/MS | 0.2 mg/kg | 1 mg/kg | Yoshimitsu et al., 2020 [37]; | |
| GC-MS/MS | 2 µg/kg | 10 µg/kg | Liu et al., 2021 [38]; | |
| Cyp | SERS-MIP | 0.13 µg/kg | 0.22 µg/kg | This work |
| LC-MS/MS | 0.14 µg/kg | 0.43 µg/kg | Gomes et al., 2022 [39]; | |
| GC-MS/MS | 2.5 µg/kg | 3 µg/kg | Yang et al., 2022 [40]; |
LOD, Limit of Detection; LOQ, Limit of Quantitation; SERS-MIP, molecularly imprinted polymer technology with surface-enhanced Raman spectroscopy; LC-MS/MS, liquid chromatography tandem mass spectrometry; GC-MS/MS, Gas Chromatography-Mass Spectrometry.
This study utilized specific surface-enhanced Raman scattering combined with molecularly imprinted polymer (SERS-MIP) technology to detect carbaryl and cypermethrin pesticide residues in four types of fruits and vegetables. Although preliminary success has been achieved, extensive further experimentation is still required. Building on the existing research, the following areas of work can be pursued: (1) In SERS spectral analysis, relying solely on manual interpretation of peak positions and intensities is prone to errors; therefore, it is necessary to develop computational modeling to replace manual analysis and achieve greater precision. (2) This study focused on four common fruits and vegetables; however, given the scope of the pesticide residue issue, testing should be expanded to include a wider range of food products. Additionally, the SERS-MIP technique should be applied to the detection of other types of pesticide residues to establish a comprehensive detection system.
Molecularly imprinted polymers (MIPs) were synthesised via precipitation polymerisation using trichlorfon or cypermethrin as the template molecule, methacrylic acid as the functional monomer, acetonitrile and methanol as porogens, azobisisobutyronitrile as the initiator, and divinylbenzene or ethylene glycol dimethacrylate as the crosslinkers. SEM characterisation of the polymers revealed that both Cyp-MIPs and Tri-MIPs exhibited substantially higher specific surface area than the corresponding NIPs. A rapid SERS detection method incorporating pretreatment with Cyp-MIPs and Tri-MIPs was successfully established. Spiked tests were conducted on real samples of oranges, tangerines, leeks, and spinach using the prepared MIPs and AgNPs. Through spiked testing and quantitative analysis of four fruits and vegetables to establish standard curves, the detection results for trichlorfon and cypermethrin pesticides demonstrated good accuracy and reproducibility.
All data generated or analyzed during this study are included in this published article.
ST: Conceptualization. HC: Validation; Data Curation. GHT: Investigation. HRC: Methodology. QAM: Writing – review & editing; Formal Analysis. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This study was funded by Hainan Province Natural Science Fund Project (823MS172).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






