1 College of Food Science and Engineering, Guangdong Ocean University, 529500 Yangjiang, Guangdong, China
2 Guangdong Strong Group Co., Ltd., 510000 Guangzhou, Guangdong, China
3 Yangjiang Strongfood Jelly Manufacturing Co., Ltd., 529500 Yangjiang, Guangdong, China
Abstract
The increasing global demand for sustainable hydrocolloids has positioned passion fruit peel as a strategic alternative to traditional citrus and apple sources, owing to the associated unique molecular composition, which is rich in rhamnogalacturonan-I (RG-I). However, important gaps remain in understanding how extraction-induced structural modifications influence specific nutritional outcomes. This review critically assesses the effects of novel green extraction methods, including ultrasonic, microwave, and enzyme-assisted techniques, on the extraction efficiency, molecular integrity, and degree of branching of passion fruit peel pectin (PFPP). In contrast to prior primarily descriptive compilations, this review analyzes available data to elucidate relationships between the fine structure of PFPP (e.g., degree of esterification) and the associated metabolic pathways in the intestine. Specifically, this review explores the effects of PFPP on the gut microbiota composition and short-chain fatty acid production, and proposes future research directions based on these findings. Additionally, we highlight challenges associated with industrial-scale PFPP production, including technical limitations and equipment costs, and offer recommendations for improvement. This review provides a new perspective for developing targeted prebiotic carbohydrates, transcending mere waste utilization toward precise nutritional interventions.
Keywords
- passion fruit peel
- pectin
- green extraction
- physicochemical properties
- application
Passion fruit (Passiflora edulis Sims), also known as egg fruit or
Brazilian fruit, is a perennial climbing vine belonging to the genus
Passiflora in the family Passifloraceae. According to the
latest report from the International Fruit Juice Association (IFU) in June 2025,
global passion fruit production in 2024 is estimated at 2 million metric tons.
The annual global volume of passion fruit waste is estimated to be 1.05 million
metric tons [1]. The peel of passion fruit accounts for approximately 55% of the
fresh fruit and is rich in various active components like pectin, polyphenols,
pigments, and other active ingredients [2]. The pectin in the peel is a natural
high-molecular-weight polysaccharide mainly found in plant cell walls and
intercellular layers. Unlike citrus and apple pomace, passion fruit peel pectin
(PFPP) is characterized by a high galacturonic acid content (
The current global commercial pectin is mainly derived from citrus and apple peel, yet the continuous growth in market demand for pectin has created an urgent need for alternative raw material sources such as passion fruit peel. Other pectin sources, including watermelon rind [3], acacia fruit rind [4], and dragon fruit rind [5], are currently attracting attention from the academic community and various industries. However, passion fruit peel is considered the most promising new source for industrialization, given its large production volume, superior composition, and easy accessibility. Recycling passion fruit peel is a promising new approach for developing natural biopolymers. However, the traditional extraction methods are unsatisfactory due to low yield, limited purity, compromised stability, and structural damage to the pectin [6, 7]. Furthermore, the use of strong acids and alkalis in pectin extraction often leads to environmental pollution and generates waste. In contrast, employing approaches such as green solvents, ultrasonic assistance, electrostatic field assistance, magnetic field assistance, and microwave assistance can not only mitigate these drawbacks but also improve pectin extraction rates. For instance, the extraction rate from deep eutectic solvents can reach 22.62%, and these solvents can be recycled by rotary evaporation [8].
A comprehensive comparison between traditional acid extraction and new green technologies for utilizing passion fruit peel is missing in the literature. In contrast to previous reviews, this study offers a detailed analysis of the impact of extraction methods on structural variability. The objectives of this review are to: (1) assess the trade-offs between yield and purity across various extraction techniques; (2) establish the relationship between extraction-induced structural modifications and functional characteristics; (3) evaluate the economic viability of upscaling these procedures; and (4) examine the metabolic effects on gut microbiota.
Pectin is a natural polysaccharide found in plant cell walls. Further, it is mainly distributed in the primary wall and the middle lamella [9]. Pectin is primarily composed of homogalacturonan (HG), rhamnogalacturonan I (RG-I), xylogalacturonan (XGA), and rhamnogalacturonan II (RG-II) [10]. Based on the esterification degree, pectin can be divided into high-methyl-esterified and low-methyl-esterified versions. The low-methyl-esterified pectin is mainly found in the intercellular layer, corners, and pores of cells, while the high-methyl-esterified pectin typically extends throughout the entire cell wall [11].
The main chain of HG is an unbranched polymer of D-galacturonic acid (GalA)
composed of
The XGA main chain consists of repeating GalA units linked by
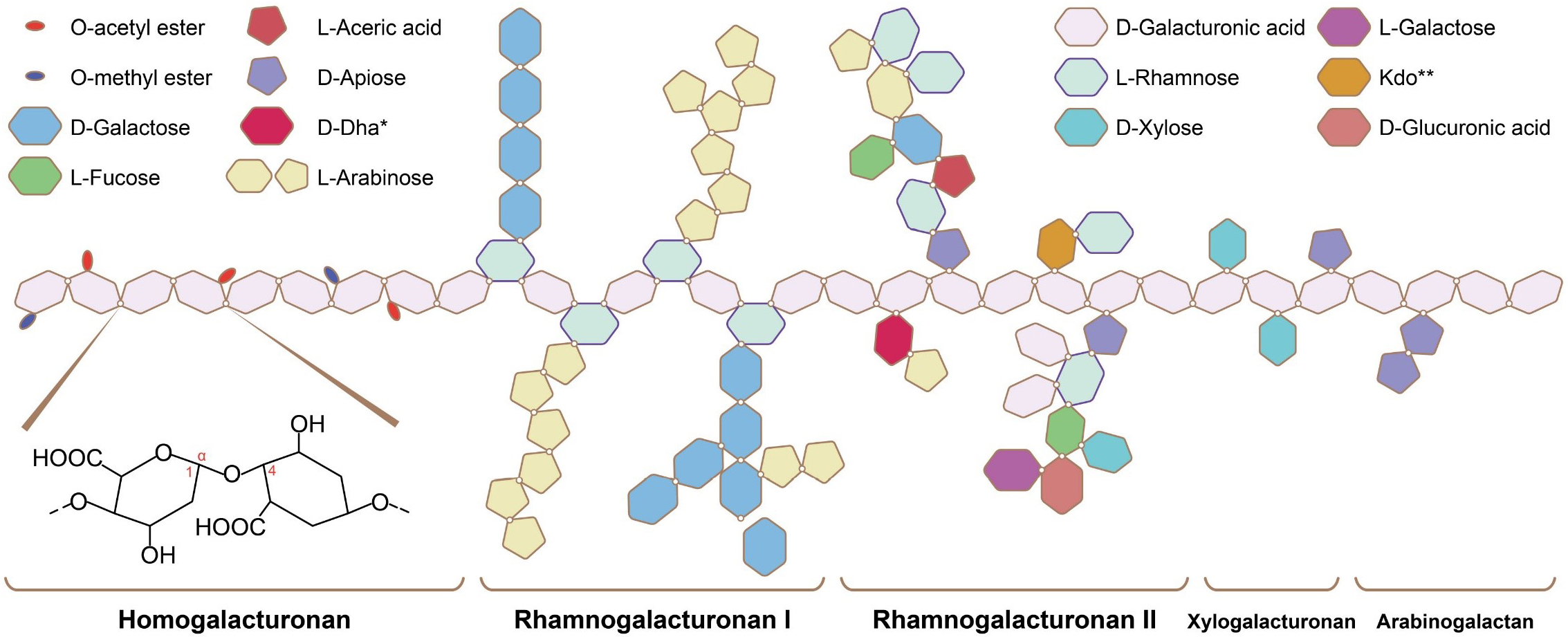 Fig. 1.
Fig. 1.
Pectin structure [20]. This figure shows the basic structure of pectin, which is primarily composed of homogalacturonan (HG), rhamnogalacturonan I (RG-I), xylogalacturonan (XGA), and rhamnogalacturonan II (RG-II), with homogalacturonan (HG) being the main component. The * and ** in the figure represent footnote markers, used to distinguish between two monosaccharides with longer names.
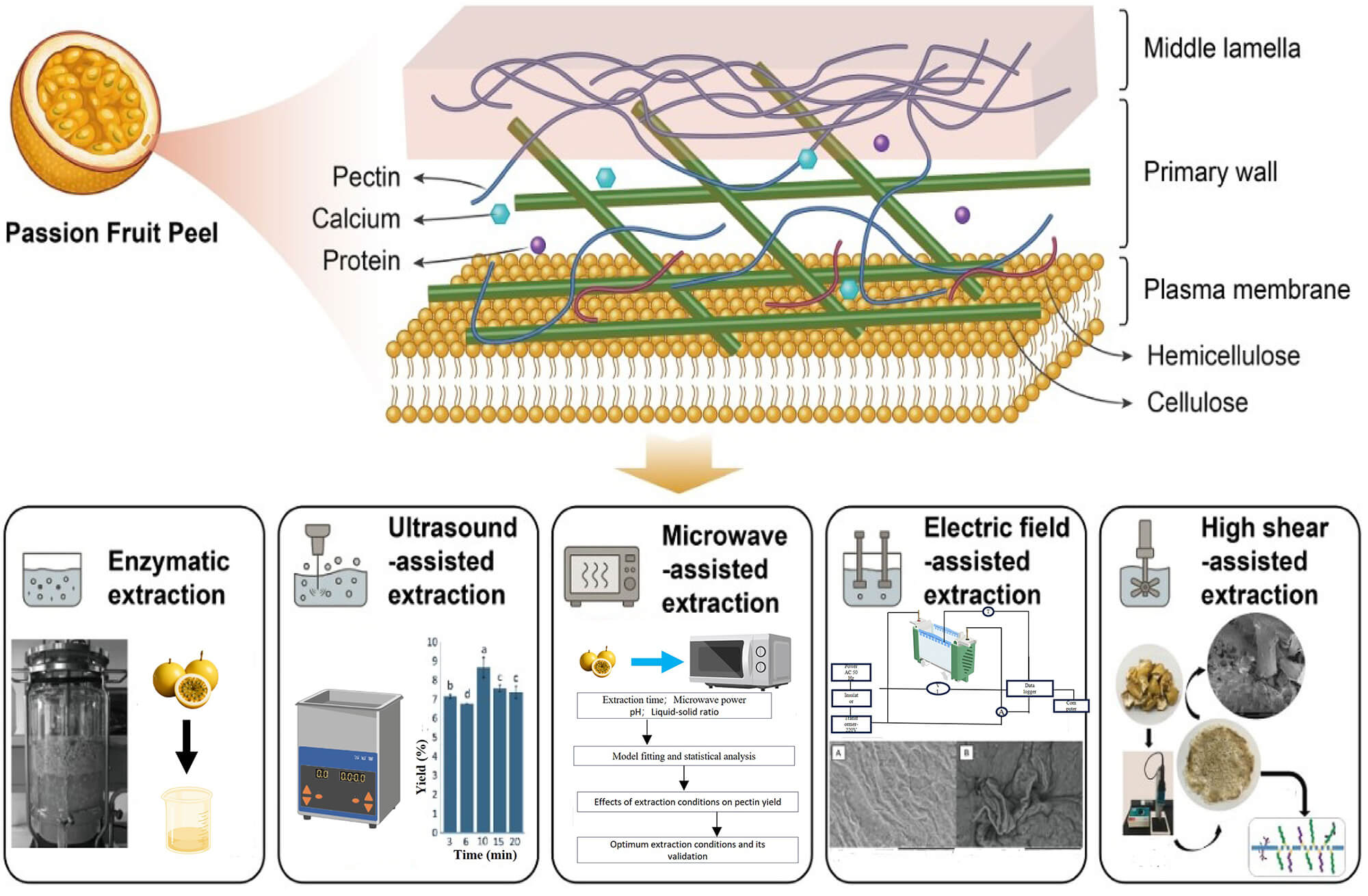 Fig. 2.
Fig. 2.
Structure of pectin in passion fruit (Anderson and Pelloux, 2025
[9]) and the effect of extraction. This diagram illustrates the distribution
and forms of pectin in plant cell walls and various extraction methods.
Extraction is a more efficient way to break these complex bonds between pectin
and cellulose, hemicellulose, calcium ions, and lignin. Different letters (a–d)
indicate significant differences (p
In scientific research, solvent extraction is one of the most commonly used methods for byproduct recovery and utilization. The process involves various solvents, including water, acid-base solutions, and novel solvents such as deep eutectic solvents. For example, Suleiman et al. [21] employed subcritical water as an extraction solvent to isolate pectin from the rambutan peel. Further, Sun et al. [22] utilized ternary deep eutectic solvents for extracting lignocellulose. Other commonly used extractants include hydrochloric acid, nitric acid, and citric acid. For example, hydrochloric acid is a solvent used to extract pectin from lemon peel [23]. Another study compared the extraction efficiency of four acids on pectin from orange peel and lime peel [24], while acid has been used to extract hawthorn pectin [25].
Hydrochloric and nitric acids are strong acids that can affect the structure of pectin [26], reducing its molecular weight and causing environmental pollution. With the current rise in the application of green solutions, the extraction rate of pectin has significantly improved [27]. These green solutions cause minimal environmental pollution, allow for recycling and reuse, and save costs.
Water extraction commonly employs hot and subcritical water to extract
by-products. This approach can accelerate substance dissolution and disrupt the
hydrogen bonds between substances, achieving the desired extraction effect. Since
pectin is highly soluble in water, hot water extraction is commonly used. Teng
et al. [28] demonstrated the effects of hot water extraction on the
extraction efficiency of purple passion fruit pectin, its structural
characteristics, anti-inflammatory, and antioxidant properties. The extraction
rate for hot water was 10.21
Critical water is the water that exists between the boiling point 100 °C and the critical temperature 374 °C, and is maintained in a liquid state by applying pressure [29]. Critical water has a higher ionic strength and a lower pH value, offering better acid hydrolysis effects and promoting bond breakage between molecules [30]. Thus, critical water is more environmentally friendly and advantageous for pectin extraction. Wang et al. [31] reported that subcritical water extraction of citrus and apple pectin achieved yields of 21.95% and 16.68%, respectively. Unlike acid-based methods, subcritical water causes minimal degradation of the pectin backbone, preserving the integrity of RG-I region. Consequently, the extracted pectin exhibits enhanced biological activity and emulsifying properties. Under combined acidic and high-temperature conditions, subcritical water further solubilizes pectin, with extraction efficiency strongly influenced by temperature and pH. Additionally, this method produces apple pectin with different degrees of methoxylation, where high-methoxyl pectin exhibits superior emulsification stability due to its robust molecular structure [32]. Klinchongkon et al. [33] showed that subcritical water increased the pectin extraction rate from passion fruit peel as the temperature increased, and ethanol addition retained RG-I. The optimal extraction conditions were as follows: subcritical water temperature of 140 °C, extraction time of 6.9 min, and ethanol concentration of 10% (v/v).
Traditional acid and alkali methods often employ strong acids and bases such as hydrochloric acid, nitric acid, and sodium hydroxide. These substances break down the structure of plant cell walls to facilitate pectin extraction. Kulkarni and Vijayanand [34] used concentrated hydrochloric acid to prepare varying concentrations of hydrochloric acid extractants for extracting pectin from passion fruit peel. By examining the effects of the extractant ratio, extraction time, and extraction temperature on the extraction rate and quality of pectin, they identified the optimal experimental conditions as: pH 2.0, a peel-to-extractant ratio of 1:30, a temperature of 98.7 °C, and a duration of 60 min. The pectin extraction rate was 14.8%, indicating that the galacturonic acid content was 88.2 g/100 g. This principle of pectin is similar to that of alkaline extraction. For instance, Le [35] used microwave-assisted alkaline method to extract passion fruit pectin by investigating NaOH concentration, solid-liquid ratio, extraction time, and microwave power. The alkali extraction conditions were: 180 mM NaOH, a solid-liquid ratio of 1:35, an extraction time of 7 min, and 376 W microwave power. These conditions yielded 14.2% pectin, 87.4% purity, and a 92.6% degree of esterification.
The extensive use of strong mineral acids and bases in industry poses significant environmental and sustainability challenges. These conventional methods produce large quantities of harsh byproducts that demand extensive neutralization, resulting in elevated salinity and increased chemical oxygen demand in wastewater [36]. Moreover, the corrosive properties of these substances mandate the use of costly corrosion-resistant alloys for equipment, leading to heightened environmental impact due to frequent maintenance and replacement. Additionally, the high energy consumption for prolonged heating further impedes sustainable valorization, conflicting with the principles of a circular bioeconomy [37]. These obstacles highlight the pressing necessity to shift towards bio-derived or ‘designer’ solvents like organic acids and deep eutectic solvents.
Citric acid is a natural organic acid, a currently popular extracting agent that can reduce environmental pollution and has minimal impact on the structure of pectin. Citric acid can effectively break down plant cell walls, subsequently releasing pectin. Compared to strong acids, citric acid effectively improves the extraction rate and pectin quality. For instance, Pinheiro et al. [38] used citric acid to extract passion fruit pectin, examining the effects of citric acid concentration, extraction time, and extraction temperature on the extraction rate. The optimal extraction conditions under the response surface methodology were: 0.086% (w/v) citric acid, 60 min extraction time, and 65 °C extraction temperature, realizing a 13.5% pectin extraction rate. Therefore, citric acid significantly impacts the degree of pectin esterification. Liew et al. [39] found that the yield of PFPP extracted from citric acid is 2.25–14.60%, with a 41.67–67.31% esterification degree. Besides, the pH value and extraction time influenced the structure of the pectin. Similarly, Guo et al. [40] used citric acid to extract low-methoxyl pectin from apple pomace, establishing that different pH levels extracted significantly different pectins. Additionally, when combined with Ca2+ under acidic conditions, the extracted pectin can enhance its gelling properties and stability.
The NAEDS are low-melting-point mixtures composed of two or more compounds and are currently popular because of the advantage of being recyclable. First proposed by Abbott et al. [41] the NAEDS concept involved mixing a specific ratio of choline chloride with organic acids to generate a solution with a low melting point, strong solubility, low volatility, and recyclability. Besides, Pereira et al. [42] employed three methods: conventional extraction, subcritical water extraction (SWE), and pressurized natural deep eutectic solvent (P-NaDES) to extract pectin from passion fruit peel. This method was used to compare pectin yield and structural characteristics in order to optimize the extraction conditions. At 120 °C, the pectin yield from P-NaDES (citric acid: glucose: water), SWE, and the conventional extraction method was 19.1, 15.7, and 10.9%, respectively. P-NaDES was the most effective extraction method, yielding pectin with a larger molecular weight. Additionally, the galacturonic acid content in the SWE extracted pectin reached 74–78%. Using DES to extract pectin and phenolic compounds from the passion fruit peel [8]. The optimal extraction condition is 120 min extraction time, a pH value of 2, a 90 °C extraction temperature, and a 20 v/w liquid-to-material ratio, resulting in a 22.63% final pectin extraction rate and a 737.04 KDa pectin molecular weight. Extraction using DES is more uniform and stable than using HCl. In citrus fruits, Rai et al. [43] extracted pectin from discarded sour orange peel by mixing different material-to-liquid ratios of DES, followed by ultrasonic-assisted extraction. The pectin extraction rate reached 37.21% and the DES recovery rate reached 85.49%.
Cellulase, hemicellulase, ligninase, and other enzymes are commonly used for
extracting by-products. For example, pectin forms a network structure with
cellulose, hemicellulose, and lignin. Enzymes can specifically decompose
substances by disrupting the structure of plant cell walls, making pectin easier
to release [44]. Cellulase decomposes cellulose, breaking the connection between
cellulose and pectin, thereby releasing pectin. This enzymatic hydrolysis
prevents the waste of large solvent amounts, offering environmental protection,
yet generating high-purity pectin [45]. Vasco-Correa and Zapata Zapata [46]
investigated the practical application of transitioning from laboratory to
large-scale pectin production from golden passion fruit by spontaneously
culturing the crude extract of the fungal enzyme PPase-SE versus pectin extracted
using traditional chemical methods. The pectin yield from passion fruit peel
using the laboratory enzyme method was 17.0–25.8 g/100 g of dry fruit peel,
whereas the yield from the chemical method was 15.9
UAE involves the propagation of ultrasonic waves in a liquid, generating shock
waves and high pressure, forming tiny bubbles that burst under high pressure to
release a large amount of energy and heat. This cavitation effect produced by
ultrasonic waves [48] can break down plant cell walls, allowing the components
within the cell walls to be extracted. For example, Freitas et al. [49]
utilized ultrasonic alkaline-assisted extraction of passion fruit peel to
evaluate the pectin yield alongside conventional extraction. Under ultrasonic
power (664 W) had an optimal yield of 12.67% at 85 °C, whereas the conventional
methods only yielded 7.95% under the same conditions. However, these conditions
yielded only 66.65 g/100 g GalA, with a DE value
Microwave heating can achieve higher extraction yields while maintaining low energy consumption. For example, increasing the microwave temperature from 80 °C to 100 °C increases the storage modulus of the pectin hydrogel accordingly, enhancing structural stability [51]. Seixas et al. [52] extracted pectin from passion fruit peel using a microwave-induced heating method by dissolving the peel powder in distilled water, adjusting the pH to 2, and placing the mixture in a microwave oven, with periodic replenishment of evaporated water. The supernatant was then collected and processed to obtain pectin before comparing tartaric, acetic, and nitric acid extraction effects. Tartaric acid achieved the highest pectin yield (30.3%), with relatively low purity and degree of esterification (DE 50.00%). Acetic and nitric acid extraction produced pectin with higher purity and degree of esterification (64.56 and 64.15%, respectively). Microwave extraction increased the porous structure of the passion fruit peel powder, rupturing cells and releasing pectin and other components readily. MAE yielded 18.73, 18.24, and 15.79% pectin from purple passion fruit, red dragon fruit, and white dragon fruit peel. In contrast, citric acid hot water extraction yielded 13.18, 15.12, and 14.11%, respectively (Dao et al. [53]).
Meanwhile, the optimal extraction conditions for the response surface methodology involve: 12 min extraction time, 218 W microwave power, pH of 2.9, and a 57:1 (v/w) liquid-to-solid ratio. However, the GalA content of microwave-extracted passion fruit pectin (71.39%) was lower than that of citric acid hot water (78.09%). The FTIR confirmed functional groups of the extracted pectin were similar to those of commercial pectin. Microwave-assisted extraction can achieve high extraction rates, maintain the structural integrity of pectin, reduce experimental time, minimize resource waste, and represent an environmentally friendly novel extraction method. These findings elucidate the synergistic mechanisms between microwave power and solvent polarity, providing a rigorous scientific basis for optimizing the extraction of high-methoxyl pectin with preserved backbone integrity. This optimization is critical for enhancing the gelling performance of PFPP in industrial applications.
There are a few studies on the electric field extraction of plant materials through electrodes. In this method, the cell membrane is pierced to form hydrophilic pores, increasing the permeability of the cell membrane [54] and accelerating pectin release [55]. De Oliveira et al. [56] obtained PFPP through moderate electric field-assisted extraction. The intensity of the electric field, extraction time, and pH significantly influence the extraction. Although pectin yield from electric field extraction is lower than conventional methods, the structure of the extracted pectin is superior. At pH 2, the pectin DE can reach 90.68% and the galacturonic acid content reaches 69.21 g/100 g. Petzold et al. [57] compared electric field-assisted extraction with conventional extraction methods. The electric field-assisted extraction produced pectin with a high degree of esterification (DE) content of 87–89% and exhibited excellent gelling properties. Additionally, this method required less time than conventional extraction, rendering it an effective approach for the food industry. The study examined the effects of various alternating current conditions on pectin extraction. Optimal results were obtained at 100 V, for 40 minutes, and at pH 5.0, resulting in a final yield of 7.68%; however, this extraction rate remains comparatively low when assessed against other methods.
This novel extraction technology, electric field-assisted extraction, still requires further exploration.
High-speed shearing generates enormous shear forces through the rapid rotation of the rotor, creating impact and end flow that ensures the material is dispersed, broken, and compressed. High-speed shearing (HSS-P) with 50 °C ultrapure water and flash extraction of pectin from passion fruit peel yielded similar pectin (Lin et al. [58]). The surface of HSS-P extracted pectin was loose, with a distinct honeycomb structure, whereas that from hot water extraction was smooth. Thus, HSS-P influenced the pectin structure, resulting in pectin with smaller molecular weights, shorter structural chains, and a higher degree of branching, which contributes to the stability of the pectin structure.
From an analytical perspective, while MAE provides the highest yield and shortest extraction time for passion fruit pectin, its energy consumption at an industrial scale presents a significant challenge due to the non-uniform distribution of electromagnetic fields in large reactors. In terms of sustainability, DES systems demonstrate considerable potential for preserving the bioactive side chains of pectin; however, the high energy costs associated with solvent recovery, such as through vacuum evaporation, must be mitigated to maintain economic competitiveness with traditional mineral acids. Table 1 (Ref, [8, 28, 34, 35, 38, 46, 50, 53, 56, 58]) summarizes and compares the various extraction methods.
| Extraction | Extraction conditions | Extraction rate | Pros and Cons | Industrial feasibility | References |
| Water | Temperature: 60 °C; Time: 2 hours; Liquid-to-material ratio: 1:20 (w/v) | 10.21 |
Pros: The solvent is non-toxic and harmless, and the method is simple. | Suitable | [28] |
| Cons: Low extraction yield, time-consuming, and high temperatures may damage the pectin structure. | |||||
| Acid and alkali | Acid: Temperature: 98.7 °C; Time: 1 h; Feed-to-solvent ratio: 1:30 (w/v) Alkali: Concentration: 180 mM NaOH; Time: 7 min; Liquid-to-material ratio: 1:35 (w/v); Microwave power: 376 W |
14.8% and 14.2% | Pros: It has a higher extraction rate than the water extraction method, effectively extracts pectin, and features mature technology and low costs. | Common industrial methods | [34, 35] |
| Cons: It pollutes the environment; acids and bases are corrosive and require specific handling methods; pectin is prone to esterification reactions. | |||||
| Citric acid | Temperature: 65 °C; Time: 1 h; Concentration: 0.086% (w/v) | 13.5% | Pros: Higher extraction efficiency than traditional methods; minimal impact on pectin structure; non-toxic and non-corrosive; stabilized by citric acid and resistant to decomposition. | Suitable | [38] |
| Cons: It is more expensive than hydrochloric acid and is suitable for production environments with stringent requirements, such as the pharmaceutical industry. | |||||
| Natural deep eutectic solvents (NADES) | Temperature: 90 °C; Time: 2 hours; Liquid-to-material ratio: 1:20 (w/v); pH: 2.0 |
22.63% | Pros: High extraction efficiency, solvent-recoverable, non-toxic and non-corrosive. | Suitable | [8] |
| Cons: Production and recycling equipment is not yet fully developed, and the process requires a large amount of solvents; it is suitable for products with high quality requirements. | |||||
| Enzymatic extraction | Enzyme concentration: 30 U/mL; Time: 2 hours; Temperature: 37 °C; pH: 3.0 | 17.0%–25.8% | Pros: High extraction efficiency, guaranteed safety, minimal damage to the pectin structure, and can be combined with other methods to further increase extraction efficiency. | It’s suitable, but the cost is relatively high. | [46] |
| Cons: Due to high costs, it is not currently suitable for large-scale production; however, it is possible to develop industrial enzymes that are low-cost and highly active. | |||||
| Ultrasound-assisted extraction | Temperature: 35 °C; Time: 30 min; Ultrasonic power: 240 W; Liquid-to-material ratio: 1:65 (w/v), Ammonium oxalate: 0.6% (w/v); pH: 2.0 |
10.05% | Pros: It takes little time and can be combined with various methods; low temperature. | Suitable for small-scale extraction; can be combined with other methods, such as enzymatic hydrolysis or citric acid | [50] |
| Cons: Not suitable for large-scale production; high frequencies may damage the pectin structure and cause noise pollution. | |||||
| Microwave-assisted extraction | Microwave power: 218 W; pH: 2.9; Time: 12 min; Liquid-to-solid ratio: 57:1 mL/g | 18.73% | Pros: High extraction efficiency, quick processing, and easy operation. | Suitable for small-scale extraction; can be combined with other methods, such as enzymatic hydrolysis or citric acid | [53] |
| Cons: The equipment is expensive and consumes a lot of energy; high temperatures may damage the pectin structure. | |||||
| Electric field-assisted extraction | Voltage: 100 V; Time: 40 min; pH: 2.0; Temperature: |
6.2% | Pros: It offers significant advantages in terms of extraction efficiency, processing time, energy consumption, and low-temperature protection. | Not suitable at this time | [56] |
| Cons: The equipment is expensive, the electrodes are prone to corrosion, and it cannot be used on a large scale. | |||||
| High shear-assisted extraction | Temperature: 50 °C; Time: 9 min | - | Pros: Low temperature, short processing time, no chemical reagents. | Not suitable at this time | [58] |
| Cons: Due to technical limitations, the pectin produced has a low molecular weight, and the extraction rate is not particularly high. |
PFPP exhibits high methoxylation, emulsifying stability, thermal stability, and gelling properties (de Moura et al. [59] and Liang et al. [60]). The extraction techniques outlined above not only have a substantial impact on yield but also influence the fundamental properties of pectin. Consequently, this section will present some of the basic properties of pectin, with the most direct correlation to the extraction techniques being the variation in the degree of esterification. These characteristics enable its widespread application in the food industry, such as thickeners and stabilizers.
Pectin with less than 50% methylation is classified as low methoxyl pectin and can form a stable gel under acidic conditions and high sucrose concentrations [60]. Conversely, high methoxyl pectin contains more than 50% methoxyl pectin. High methoxyl pectin can effectively stabilize the proteins in milk, preventing protein degradation and improving the stability and taste of milk [61]. PFPP is a high-ester pectin, with typical methylation of 59.3 to 83.40%.
PFPP demonstrates excellent emulsion stability by reducing the interfacial tension between water and oil. The mechanism involves pectin forming a stable biopolymer matrix around the oil droplets, preventing aggregation and separation. Pectin-created emulsions have enhanced shear resistance and reduced surface tension [62]. Emulsion stability influences the molecular weight and DE; pectin with a higher molecular weight and DE results in higher emulsion viscosity [63]. In milk chocolate, the emulsifying properties of pectin can enhance the sensory characteristics of the product [64].
Passion fruit pectin demonstrates excellent thermal stability due to its
relatively high molecular weight and compact molecular structure. Additionally,
the high content of galacturonic acid (
Gelling ability is one of the most important properties of pectin, closely related to its DE, molecular weight, and other factors. Gelation requires acidic conditions, as low pH promotes the formation of -COOH groups, reduces the repulsive forces between pectin molecules, and facilitates hydrogen bond formation [68]. The gelling properties of pectin can be combined with chitosan to form composite cryogels [69], and the unique textural structures formed by these properties can be effectively displayed in 3D food ink while also serving as a fat substitute [70].
The emulsifying, thickening, and gelling properties of pectin give it wide applications across various industries, including food and pharmaceuticals. Moreover, its gelling characteristics make it particularly suitable for pastries, jellies, and jams [52]. For instance, the gel hardness and viscosity of passion fruit pectin are comparable to those of commercial pectin, making it an excellent thickening agent [71]. In baking, pectin enhances the firmness and chewiness of bread [72], and adding pectin to dairy beverages prevents milk protein flocculation [73]. Given the growing global concern over plastic pollution, biofilms derived from PFPP have attracted considerable interest due to their biodegradability in natural environments, offering a promising avenue for reducing environmental contamination. Furthermore, pectin can serve as a sustainable alternative to conventional plasticizers such as glycerol [74], with pectin-based films demonstrating notable preservation capabilities. Beyond food and packaging applications, modified pectin from passion fruit peel has also shown potential in biomedical contexts. For example, Citrus aurantium pectin possesses strong antioxidant and antimicrobial properties [75]. Furthermore, melon rind pectin is a new source of prebiotics [76], and pectin is widely used in animal immunization [77]. Table 2 (Ref. [67, 78, 79]), Table 3 (Ref. [80, 81, 82]), Table 4 (Ref. [8, 83, 84, 85, 86]) and Fig. 3 summarize the relevant applications of passion fruit pectin. The schematic diagram (Fig. 4, Ref. [85]) illustrates the mechanism of the hypoglycemic effect of passion fruit pectin in the rat model.
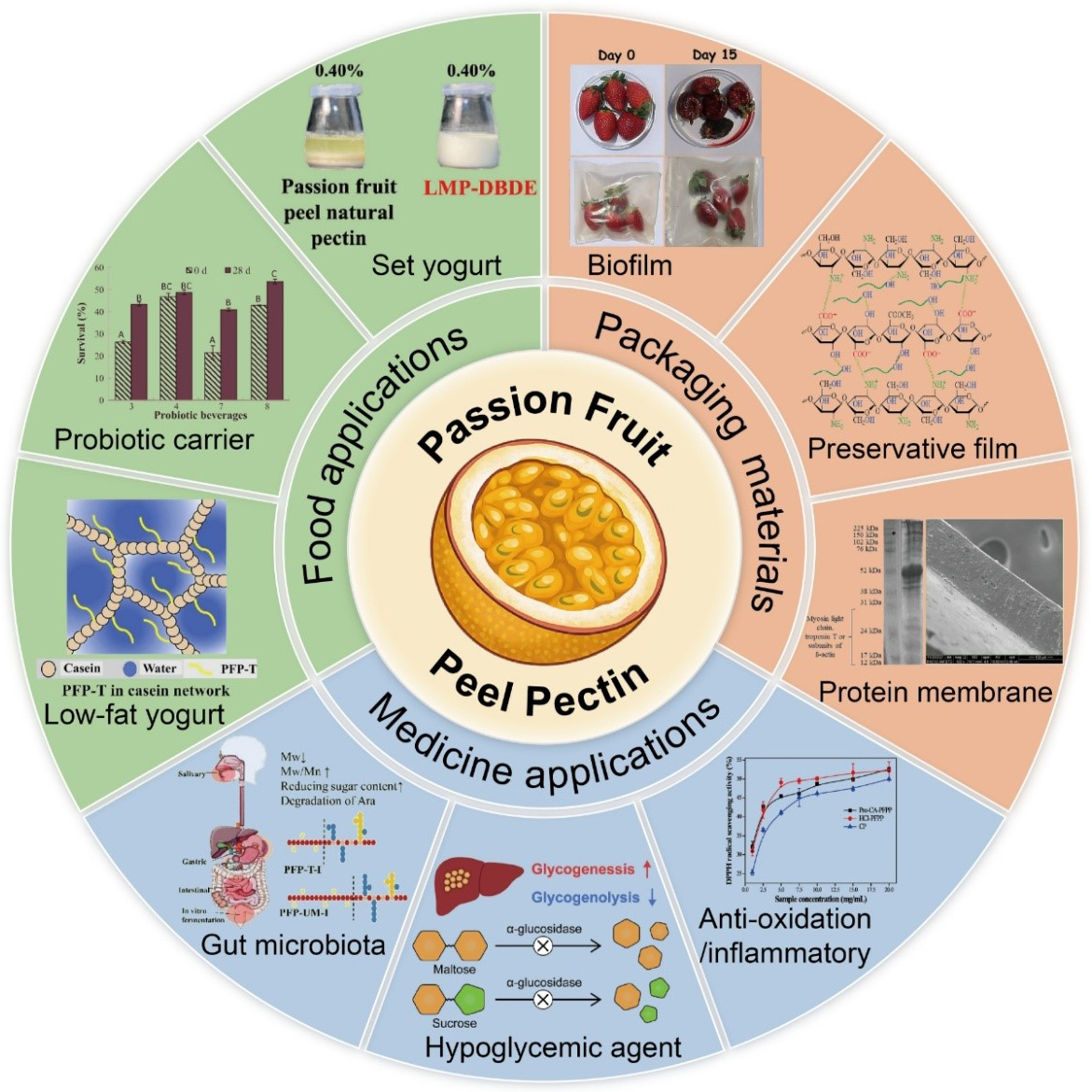 Fig. 3.
Fig. 3.
Applications of passion fruit pectin. This diagram systematically displays the multifunctional application prospects of PFPP, mainly divided into three directions: the food applications, packaging materials and medicine applications.
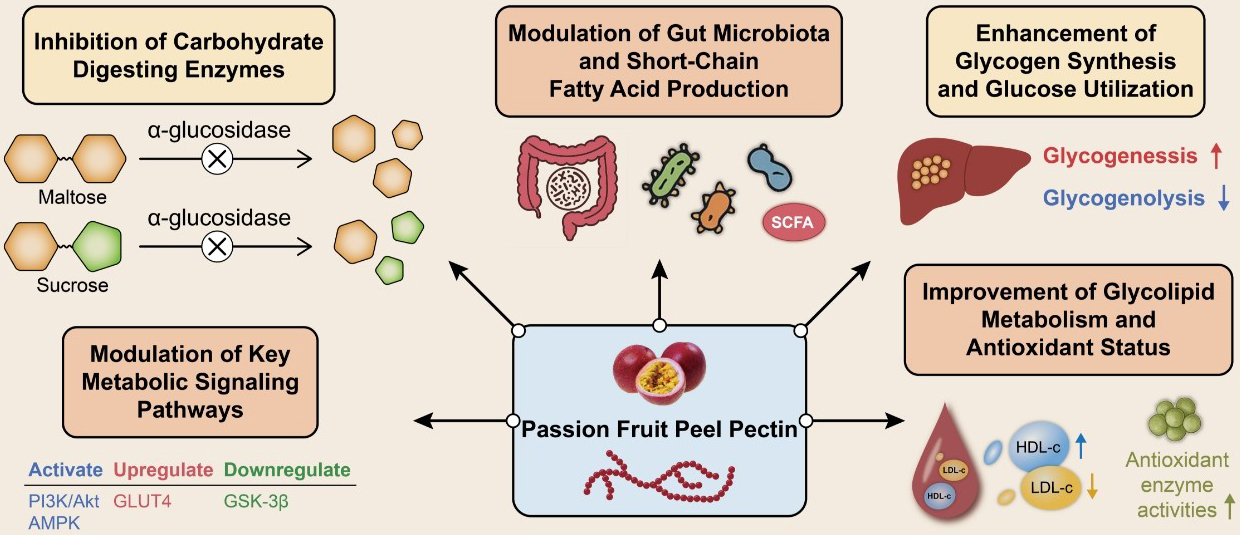 Fig. 4.
Fig. 4.
Schematic diagram of the mechanism of passion fruit pectin
hypoglycemic effect in rats [85]. This diagram shows the synergistic network of
gut interventions (enzyme inhibition, microbiota regulation)
| Type | Extraction | Galacturonic acid content | Degree of esterification | Application | Reference |
| Set yogurt | Enzymatic extraction, Solid-to-liquid ratio of 1:30, extraction time of 60 min | 47.81% | - | After de-esterification, pectin with a higher galacturonic acid content exhibits enhanced gelling properties, making it a promising ingredient for yogurt production. It can replace fat and sugar in yogurt, extending its shelf life. | [67] |
| Low-fat yogurt | - | - | - | Pectin can increase the fermentation rate of low-fat yogurt. Specifically, 0.025% passion fruit pectin enhances the number of lactic acid bacteria while interacting with casein to form complexes that strengthen the gel network structure, thereby extending the shelf life. | [78] |
| Probiotic carrier | Citric acid, Extraction temperature: 50 °C, extraction time: 2 h | - | 13.55% | Pectin extracted from passion fruit peel and pulp, and sucrose were added to fermented and non-fermented beverages, followed by inoculation with Lactobacillus rhamnosus. After 6 hours of incubation, the survival rate of the strains in beverages containing added passion fruit pectin reached 43% (non-fermented) and 47% (fermented). After 28 days of refrigeration, the beverages with added passion fruit pectin maintained higher strain survival rates. | [79] |
| Type | Extraction | Galacturonic acid content | Degree of esterification | Application | Reference |
| Biofilm | - | - | - | Polyvinyl alcohol (PVA), lemon pectin, PFPP, and rosemary essential oil (REO) were combined in various formulations. The film prepared with PVA, lime pectin, and passion fruit pectin (2:1:1 v/v) exhibited the best performance. | [80] |
| Preservative film | Citric acid, extraction time 90 min | 62.92 |
50.74 |
Edible preservation films were prepared using passion fruit pectin and chitosan, with the addition of Piper longum leaf extract to form composite biofilms via casting. The soluble solid content of eggplants was monitored; those wrapped with the film showed less leaching, while the eggplants without any preservation treatment exhibited the greatest changes. | [81] |
| Protein membrane | - | - | 88.95% | A novel packaging material was developed using pectin extracted from passion fruit peel and myofibrillar protein obtained from sawfish. The optimal formulation was 5% myofibrillar protein and 3% pectin. The biofilm prepared under these conditions exhibited uniformity, strength, flexibility, and biodegradability, while maintaining low solubility, low water vapor permeability, and excellent thermal stability. | [82] |
| Type | Extraction | Galacturonic acid content | Degree of esterification | Application | Reference |
| Antioxidation | DES, Extraction time 120 min, pH 2, extraction temperature 90 °C, liquid-to-solid ratio 20 v/w | - | 63.91% | The antioxidant activity of passion fruit pectin was determined through DPPH and ABTS free radical assays. The antioxidant capacity of pectin was concentration-dependent, with higher concentrations yielding stronger antioxidant effects. Pectin extracted using DES exhibited superior antioxidant activity compared to that obtained through dilute hydrochloric acid extraction. However, the antioxidant capacity of pectin proved to be comparable to ascorbic acid. | [8] |
| Intestine | - | - | - | Passion fruit pectin, a natural prebiotic, affects human intestinal microbiota. Fecal samples from four healthy donors were pooled and fermented independently in separate batches, with passion fruit and commercial pectin added to investigate the differences. The experimental group supplemented with passion fruit pectin exhibited a slower rate of pH decline than the commercial pectin group, accumulating more gas in the system. Therefore, passion fruit pectin is more readily utilized by intestinal microbiota, with enhanced effects observed upon probiotic supplementation. | [86] |
| Anticancer | - | - | - | Cell experiments revealed that modified pectin significantly inhibited colorectal cancer cell viability, while immunofluorescence studies showed that cancer cells could internalize pectin, demonstrating potential anticancer properties. | [83] |
| Anti-inflammatory/blood glucose reduction | - | - | - | Pectin can reduce blood glucose and triglyceride levels in diabetic rats, and decrease edema volume and myeloperoxidase release. Immunostaining revealed reductions in both tumor necrosis factor- |
[85] |
| Gut microbiota | ultrasound-microwave-assisted | - | 64.95% | PFP-T pectin promotes the proliferation of beneficial bacteria such as Bacillus and Megasphaera, while inhibiting the growth of Escherichia-Shigella, increasing the production of short-chain fatty acids. | [84] |
PFPP has been shown to enhance the production of short-chain fatty acids and influence specific bacterial strains. Research indicates that PFPP can improve glucose and lipid metabolism in obese individuals through the modulation of gut microbiota. Ju et al. [87] developed a high-sugar, high-fat mouse model to examine the hypoglycemic and hypolipidemic effects of PFPP. Their findings revealed that the inclusion of PFPP led to an increase in both short-chain fatty acid and probiotic concentrations, as well as a rise in the abundance of Bacteroides and Bifidobacterium.
This finding establishes a reference pathway for future research on PFPP. Additionally, Pimisa et al. [86] examined the effects of PFPP on gut microbiota through in vitro fermentation. The study compared four fermentation durations (0, 5, 24, and 48 hours) and revealed that PFPP produced elevated levels of acetic acid, contrasting with commercial pectin, which tends to generate more butyric acid. Furthermore, PFPP increased the abundance of the families Bifidobacteriaceae, Coriobacteriaceae, and Erysipelotrichaceae, while simultaneously reducing the prevalence of certain harmful bacteria. Given these findings, further research is necessary to explore whether the structure of PFPP correlates with alterations in the gut microbiome, potentially guiding the development of enhanced methods for modulating gut microbiota through various extraction techniques.
Although laboratory-scale extraction has demonstrated high yields, transitioning to industrial scales presents considerable engineering challenges. For example, in the UAE, the intensity of cavitation decreases with increasing distance from the transducer, necessitating the design of flow-through reactors to sustain efficiency. Likewise, microwave-assisted extraction (MAE) encounters issues with “hot spots” during large-batch processing, which may result in the thermal degradation of pectin chains.
The commercial success of PFPP relies on the integration of extraction facilities with fruit juice processing plants. This proximity reduces transportation costs and utilizes fresh rinds, preventing microbial spoilage. Substituting mineral acids with organic acids or Deep Eutectic Solvents (DES) not only aligns with green chemistry but also simplifies wastewater treatment processes, potentially lowering the overall operational expenditure (OPEX).
Commercialization efforts should emphasize the distinctive functional properties of PFPP, particularly its high degree of esterification. This compound demonstrates exceptional efficacy as a stabilizer in plant-based dairy alternatives, effectively preventing phase separation in soy-based and oat-based yogurts. Additionally, its potential as a targeted prebiotic carrier within the nutraceutical industry presents a significantly higher profit margin when compared to bulk food thickeners [88].
The extraction of pectin is diversifying, necessitating further research to
comprehend the impact of different methods on pectin’s structure and function.
This study highlights passion fruit peel as a superior option to citrus and apple
pomace, producing pectin with a notably higher proportion of rhamnogalacturonan-I
(RG-I) domains. Green extraction techniques like UAE and MAE quantitatively yield
18.5%–26.4% within 15–45 minutes, maintaining a degree of esterification (DE)
Passion fruit peel is a highly promising material for pectin extraction, exhibiting identical properties to commercial pectin. Since the production of passion fruit-related products currently discards the peel as a by-product, peel utilization can maximize its application and conserve resources, like other fruits, passion fruit faces challenges in ensuring extraction efficiency while minimizing the impact of extraction methods on the pectin structure. Additionally, research on passion fruit pectin extraction is limited, yet pectin applications are rapidly expanding. Therefore, further research on pectin is essential. Future novel processing methods combined with green solvents could be employed to extract pectin, improve its structure, and enhance bioavailability, while considering its practical applications. Pectin-related research can increase the utilization of waste materials and expand their application scope.
Although the prebiotic potential of PFPP is acknowledged, the ‘structure-activity’ relationship remains largely unresolved. Future investigations should employ high-resolution NMR and Atomic Force Microscopy (AFM) to elucidate how three-dimensional conformation influences metabolic cross-feeding among specific gut bacteria, such as Bifidobacterium and Faecalibacterium prausnitzii. Concurrently, digital technology can be integrated with artificial intelligence, utilizing advanced algorithms and analytics [89]. Regarding the effects of PFPP on gut microbiota, research should be refined to assess its impact on specific bacterial strains, broaden studies on its functional properties, and investigate its applications in food fermentation.
JM, YH, and PH designed this study. SW, JC, and ZL conducted the research. HZ, YD, and ZZ compiled the data. JM wrote this paper. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This work was supported by the Yangjiang Sci-tech Commissioner Program [grant numbers SDZX2022033 and SDZX2023026]; Additional support was provided by the Teaching Quality and Teaching Reform Project Construction Project of 2022 Guangdong Undergraduate Universities for the Guangdong Ocean University-Xizhilang Science and Technology Integration Practice Teaching Base [grant numbers 310117062201].
No conflict of interest exists in the submission of this manuscript, and the manuscript is approved by all authors for publication. All the authors listed have approved the manuscript that is enclosed. In addition, Shaoming Wu is an employee of Guangdong Strong Group Co., Ltd.; Jiezi Chen and Ziwen are employees of Yangjiang Strongfood Jelly Manufacturing Co., Ltd. I had full access to all of the data in this study and I take complete responsibility for the integrity of the data and the accuracy of the data analysis.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




