1 Department of Food Science and Nutrition, College of Sciences, Taif University, 21944 Taif, Saudi Arabia
Abstract
The growing demand for clean-label meat products has intensified interest in plant-derived antioxidants as alternatives to synthetic preservatives.
This study aimed to evaluate the potential of chia seed (Salvia hispanica) extract (CSE) as a natural antioxidant to enhance quality attributes and extend the refrigerated shelf-life of beef burgers stored at 4 °C for 15 days.
Five beef burger formulations were prepared: a negative control without antioxidants (T0), a positive control containing a synthetic antioxidant (SET), and three treatments incorporating CSE at 0.5% (T1), 1.0% (T2), and 1.5% (T3). Chia seed extract was characterized using high-performance liquid chromatography (HPLC), which identified gallic acid, caffeic acid, and quercetin as major phenolic constituents. Burgers were assessed for lipid oxidation (TBARS), water-holding capacity (WHC), cooking yield, shrinkage, texture profile attributes, and color stability throughout storage.
CSE supplementation markedly reduced lipid oxidation, with the T3 formulation showing the lowest TBARS value (0.28 mg MDA/kg) at day 15 compared with the control (1.27 mg MDA/kg) (p < 0.05). CSE-treated burgers exhibited improved WHC, reflected in higher cooking yield (91.27%) and reduced shrinkage. Texture profile analysis revealed better retention of hardness, springiness, cohesiveness, and resilience across storage. Additionally, CSE preserved color stability by mitigating reductions in L* and a* values. These enhancements are attributed to the antioxidant activity of chia phenolics, which inhibit lipid peroxidation and protein degradation.
This study provides the first comprehensive assessment of chia seed extract as a natural antioxidant in beef burgers. CSE—particularly at 1.5%—effectively improved oxidative stability, texture, and overall storage quality, demonstrating its potential as a clean-label alternative to synthetic antioxidants in meat preservation.
Keywords
- chia seed extract
- Salvia hispanica
- beef burger
- TBARS
- lipid oxidation
- natural antioxidant
- texture profile
- shelf-life
- refrigerated storage
- clean-label meat product
The growing consumer awareness of food safety, nutrition, and health has driven
the global meat industry to reformulate traditional products with functional and
natural additives. Synthetic antioxidants such as butylated hydroxytoluene (BHT)
and butylated hydroxyanisole (BHA), although effective in delaying oxidative
spoilage, have raised concerns regarding their potential toxicological effects
and long-term health risks [1]. As a result, the focus has shifted toward natural
antioxidant sources derived from plants, herbs, and seeds, which are rich in
polyphenols and bioactive compounds that align with clean-label demands [2, 3].
Lipid oxidation is among the most critical challenges affecting the quality,
safety, and shelf-life of meat and meat products. The oxidative degradation of
unsaturated fatty acids leads to rancid flavors, discoloration, texture
deterioration, and the formation of potentially harmful compounds [4]. Such
reactions begin during meat processing and continue throughout storage,
especially under aerobic and refrigerated conditions. Consequently, controlling
lipid peroxidation is essential to preserve key quality attributes such as color,
texture, flavor, and nutritional value [1, 5]. Among natural antioxidants
investigated, chia seeds (Salvia hispanica L.), a member of the
Lamiaceae family, have gained significant attention due to their health-promoting
properties and multifunctional roles in food applications. Native to Central and
South America, chia seeds are a rich source of dietary fiber, polyunsaturated
fatty acids—particularly
Several studies have confirmed the potential of chia (Salvia hispanica) to inhibit lipid and protein oxidation in food systems. For example, chia extract significantly reduced thiobarbituric acid reactive substances (TBARS) values in chicken patties during refrigerated storage [10], while chia-derived hydrocolloids enhanced water-holding capacity, minimized cooking losses, and improved texture stability in processed meats [11]. These findings underscore the dual role of chia derivatives as natural antioxidants and functional binders. Beef burgers, widely consumed processed meat products, are particularly susceptible to oxidative spoilage due to their high fat content and oxygen exposure during grinding and shaping. Rising consumer demand for healthier formulations with reduced synthetic additives has prompted exploration of natural alternatives that preserve quality and sensory attributes [12, 13]. In this context, chia seed extract offers a promising strategy for reformulating burgers, providing both antioxidant protection and functional benefits. Despite extensive research on the nutritional and health effects of chia seeds, few studies have systematically investigated the impact of concentrated chia seed extracts on the physicochemical, textural, and oxidative stability of beef burgers during refrigerated storage. Prior work has primarily focused on whole or ground seeds, leaving a research gap in evaluating chia extracts as functional additives for meat preservation [14, 15].
Furthermore, chia seed extract exhibits multifunctional properties that can simultaneously influence various quality attributes of meat products. The hydrophilic nature of chia mucilage improves moisture retention, cooking yield, and structural resilience during thermal processing, while its abundance of phenolic antioxidants suppresses lipid oxidation, thereby prolonging shelf-life and preserving desirable color and flavor. These combined effects align with the increasing trend of incorporating plant-based ingredients to enhance both the technological performance and functional quality of processed foods [16, 17]. Accordingly, the present study was designed to evaluate the effect of chia seed powder extract on the physicochemical, oxidative, and textural properties of beef burgers stored under refrigerated conditions for 15 days. Key parameters assessed included cooking yield, shrinkage, color stability (L*, a*, b*), texture profile (hardness, springiness, cohesiveness, chewiness, resilience), and lipid oxidation as indicated by TBARS values. The outcomes of this investigation are expected to contribute to the development of functional meat products with enhanced storage stability and consumer acceptability, while supporting the application of chia as a natural preservative and quality enhancer in meat processing.
Fresh ground beef (80% lean, 20% fat) was obtained from a local meat processing facility in Taif, Saudi Arabia. Chia seeds (Salvia hispanica L.) were purchased from Al-Dawaa Pharmacy (Taif, Saudi Arabia). Seeds were manually cleaned to remove foreign materials and stored in airtight containers at ambient temperature until further use. All analytical-grade reagents were obtained from Sigma-Aldrich (St. Louis, MO, USA), including thiobarbituric acid (TBA; cat. no. T5500), trichloroacetic acid (TCA; cat. no. T6399), sodium chloride (NaCl; cat. no. S9888), butylated hydroxytoluene (BHT; cat. no. W218405), and malondialdehyde (MDA) standard for TBARS analysis (cat. no. MDA325). Microbiological media were obtained from Merck (Darmstadt, Germany), including plate count agar (PCA; cat. no. 105463), MacConkey agar (cat. no. 105465), and potato dextrose agar (PDA; cat. no. 110130). Distilled water was used for the preparation of all solutions and for equipment cleaning.
Chia seeds (Salvia hispanica) were dried in a hot-air oven at 40
The proximate and bioactive composition of chia seed (Salvia
hispanica L.) powder was analyzed prior to extract preparation using Association
of Official Analytical Collaboration (AOAC) methods [18] and
complementary instrumental techniques. All analyses were performed in triplicate,
and results were expressed as mean
The chemical composition of chia seed (Salvia hispanica) powder was analyzed before extract preparation to determine its nutritional profile. The analyses were carried out according to AOAC (2005) official methods [18].
100 – (% moisture + % protein + % fat + % ash)
Additionally, the dietary fiber content was analyzed using the
enzymatic-gravimetric method [19], and results were expressed as g/100 g dry
weight. All analyses were performed in triplicate, and results were reported as
mean
Fatty acid composition of chia seed oil was determined using gas chromatography
(GC-FID). Lipids were extracted by cold maceration with hexane (cat. no. 34859,
Sigma-Aldrich, St. Louis, MO, USA), and the extracted oil was converted into
fatty acid methyl esters (FAMEs) by transesterification using methanolic
potassium hydroxide (cat. no. 60325, Sigma-Aldrich, St. Louis, MO, USA). FAMEs
were separated and quantified using a gas chromatograph (Model GC-7890A, cat. no.
N/A, Agilent Technologies, Santa Clara, CA, USA) equipped with a flame ionization
detector and a capillary column (HP-88, 100 m
Mineral elements such as calcium (Ca), magnesium (Mg), potassium (K), sodium (Na), iron (Fe), and zinc (Zn) were determined using atomic absorption spectrophotometry (AAS; PerkinElmer AAnalyst 400, Waltham, MA, USA) after dry ashing of samples at 550 °C followed by dissolution in nitric acid.
Individual phenolic compounds were identified and quantified using
high-performance liquid chromatography with a diode-array detector (HPLC-DAD).
Methanolic extracts of chia seed powder were filtered through a 0.45
µm syringe filter (cat. no. SLHV033RS, Millipore, Burlington, MA,
USA) and injected into an HPLC system (Shimadzu LC-20AT, cat. no. N/A, Shimadzu
Corporation, Kyoto, Japan) equipped with a reverse-phase C18 column (250 mm
2.3.5.1 HPLC Analysis of Phenolic Compounds in Chia Seed Extract.
High-performance liquid chromatography (HPLC) was employed to identify and quantify individual phenolic compounds present in chia seed (Salvia hispanica) extract. The analysis was carried out using a reverse-phase HPLC system (Shimadzu LC-20AT, Japan) equipped with a diode array detector (DAD) and an autosampler. Chia seed powder extract was dissolved in methanol (HPLC grade), filtered through a 0.45 µm PTFE syringe filter, and stored at 4 °C until injection.
Chromatographic Conditions:
Phenolic compounds were identified by comparing retention times and UV spectra
with those of authentic standards, including gallic acid, caffeic acid,
chlorogenic acid, ferulic acid, rutin, quercetin, and others. Quantification was
based on external calibration curves for each standard. Results were expressed as
mg of compound per gram of dry extract (mg/g). All measurements were performed in
triplicate and presented as mean
2.3.5.2 Quantification of Total Phenolic Content and Antioxidant Activity.
(a) Total Phenolic Content (TPC)
The total phenolic content (TPC) of chia seed extract was determined using the Folin–Ciocalteu method. Briefly, 0.5 mL of extract solution (1 mg/mL) was mixed with 2.5 mL of Folin–Ciocalteu reagent (diluted 1:10 with distilled water; cat. no. F9252, Sigma-Aldrich, St. Louis, MO, USA) and incubated for 5 min at room temperature. Subsequently, 2 mL of 7.5% sodium carbonate solution (cat. no. S7795, Sigma-Aldrich, St. Louis, MO, USA) was added. After incubation in the dark for 30 min, absorbance was measured at 765 nm using a UV–visible spectrum spectrophotometer (UV-1800, cat. no. N/A, Shimadzu Corporation, Kyoto, Japan). Gallic acid (cat. no. G7384, Sigma-Aldrich, St. Louis, MO, USA) was used as the calibration standard, and results were expressed as mg gallic acid equivalents (GAE) per g of extract.
(b) Antioxidant Activity (DPPH Assay)
The radical scavenging activity of chia seed extract was evaluated using the DPPH (2,2-diphenyl-1-picrylhydrazyl) assay. A 0.1 mM DPPH solution was prepared in methanol, and 1 mL of extract (1 mg/mL) was added to 2 mL of DPPH solution. The mixture was incubated in the dark at room temperature for 30 min, and absorbance was measured at 517 nm. The percentage inhibition of DPPH radicals was calculated relative to a blank. Ascorbic acid served as a positive control.
The vitamin content of chia seed (Salvia hispanica) powder was quantified using standardized methods to assess its nutritional contribution as a functional ingredient.
Water-soluble vitamins, including thiamine (vitamin B1), riboflavin (vitamin
B2), niacin (vitamin B3), pyridoxine (vitamin B6), and ascorbic acid (vitamin C),
were extracted using acid and enzymatic hydrolysis methods. Approximately 2 g of
chia seed powder was homogenized with 25 mL of 0.1 M hydrochloric acid (HCl; cat.
no. 258148, Sigma-Aldrich, St. Louis, MO, USA) and incubated in a water bath
(Model WB-22, cat. no. N/A, Memmert, Schwabach, Germany) at 100 °C for
30 min, followed by enzymatic digestion with takadiastase (cat. no. D0256,
Sigma-Aldrich, St. Louis, MO, USA) at 37 °C for 3 h. The extract was
filtered through Whatman No. 1 filter paper (cat. no. 1001-110, Cytiva/Whatman,
Marlborough, MA, USA) and analyzed by high-performance liquid chromatography with
a UV–visible spectrum (HPLC-UV; Shimadzu LC-20AT, cat. no. N/A, Shimadzu Corporation,
Kyoto, Japan) using a reverse-phase C18 column (250 mm
Fat-soluble vitamins, including
The antioxidant capacity of chia seed (Salvia hispanica) powder extract was evaluated using three complementary in vitro assays: ABTS+ radical scavenging activity, DPPH radical scavenging activity, and ferric reducing antioxidant power (FRAP). All measurements were performed in triplicate, and results were expressed as µmol Trolox equivalents (TE) per gram of extract.
The ABTS+ assay was conducted with slight
modifications using the method described previously [19] . The ABTS+ radical cation was produced by mixing 7 mM ABTS solution
with 2.45 mM potassium persulfate and allowing the mixture to stand in the dark
at room temperature for 12–16 hours. Before use, the ABTS+ solution was diluted
with methanol to an absorbance of 0.70
The DPPH (2,2-diphenyl-1-picrylhydrazyl) assay was performed as described by [20]. A 0.1 mM DPPH solution was prepared in methanol. Then, 1 mL of chia extract was mixed with 2 mL of the DPPH solution and incubated in the dark for 30 minutes at room temperature. Absorbance was recorded at 517 nm. The percentage inhibition of DPPH radicals was calculated using the formula:
DPPH inhibitory (%) = (Abs. C – Abs. S)/(Abs. C)
Where Abs. C is the absorbance of the control and Abs. S is the absorbance of the sample.
The FRAP assay was conducted according to [21] Benzie and Strain (1996). The
FRAP reagent was freshly prepared by mixing 300 mM acetate buffer (pH 3.6), 10 mM
TPTZ (2,4,6-tripyridyl-s-triazine) solution in 40 mM HCl, and 20 mM
FeCl3
The tannic acid content of chia seed (Salvia hispanica) powder extract was determined spectrophotometrically using the Folin–Denis method, as described by [18, 19] with minor modifications. An aliquot of 1 mL of chia seed extract (1 mg/mL) was mixed with 1 mL of Folin–Denis reagent (cat. no. F9252, Sigma-Aldrich, St. Louis, MO, USA), followed by 2 mL of saturated sodium carbonate solution (cat. no. S7795, Sigma-Aldrich, St. Louis, MO, USA). The mixture was diluted to 10 mL with distilled water and incubated at room temperature for 30 min in the dark. The absorbance of the resulting blue color was measured at 760 nm using a UV–Vis spectrophotometer (UV-1800, cat. no. N/A, Shimadzu Corporation, Kyoto, Japan). Tannic acid (cat. no. T0377, Sigma-Aldrich, St. Louis, MO, USA) was used as the standard, and a calibration curve was prepared using concentrations ranging from 0 to 100 µg/mL. The tannic acid content was calculated from the standard curve and expressed as milligrams of tannic acid equivalents per gram of extract (mg TAE/g extract).
Fresh ground beef (80% lean, 20% fat) was used for burger preparation. The meat was sourced from a local processing facility in Taif, Saudi Arabia, and transported under chilled conditions (4 °C) to the laboratory. Visible fat and connective tissue were manually trimmed upon arrival. The burger formulation consisted of 90% ground beef, 5% cold water, 2% corn starch (cat. no. 33292, Sigma-Aldrich, St. Louis, MO, USA), 1.5% salt (cat. no. S9888, Sigma-Aldrich, St. Louis, MO, USA), 0.5% mixed spices (black pepper, coriander, garlic powder; sourced from a local supplier, Taif, Saudi Arabia), and 0.1% sodium tripolyphosphate (STPP; cat. no. 22103, Sigma-Aldrich, St. Louis, MO, USA). All ingredients were thoroughly mixed in a food mixer (Kenwood Chef XL, Model KVL4100S, cat. no. N/A, Kenwood Appliances, Havant, UK) for 5–7 minutes to form a uniform emulsion. The mixture was then shaped into patties of approximately 80 g and 1.5 cm thickness using a stainless steel mold (cat. no. N/A, local supplier, Taif, Saudi Arabia). Each patty was individually wrapped in polyethylene film (cat. no. 1098-100, Sigma-Aldrich, St. Louis, MO, USA) and stored at 4 °C prior to chia extract incorporation (Table 1).
| Ingredient | SET (Positive Control) | T0 (Negative Control) | T1 (0.5% CSE) | T2 (1.0% CSE) | T3 (1.5% CSE) |
| Ground beef | 90.00 | 90.00 | 89.50 | 89.00 | 88.50 |
| Cold water | 5.00 | 5.00 | 5.00 | 5.00 | 5.00 |
| Corn starch | 2.00 | 2.00 | 2.00 | 2.00 | 2.00 |
| Salt | 1.50 | 1.50 | 1.50 | 1.50 | 1.50 |
| Mixed spices* | 0.50 | 0.50 | 0.50 | 0.50 | 0.50 |
| Sodium tripolyphosphate (optional) | 0.10 | – | 0.10 | 0.10 | 0.10 |
| Chia seed extract powder | – | – | 0.50 | 1.00 | 1.50 |
| Total | 99.10 | 99.00 | 99.10 | 99.10 | 99.10 |
* Mixed spices include black pepper, coriander, and garlic powder in equal proportions.
• SET = Positive control with synthetic antioxidant (sodium tripolyphosphate).
• T0 = Negative control with no antioxidant.
• T1–T3 = Experimental treatments with increasing levels of chia seed extract (CSE).
The prepared burger patties were divided into four groups as follows:
The chia seed extract was homogenized into the meat mixture during mixing,
before shaping into patties. All treated and control patties were packed in
low-density polyethylene (LDPE) bags, labeled, and stored at 4
The physicochemical, microbiological, and sensory properties of burger samples
treated with chia seed extract were evaluated at regular intervals (0, 3, 6, 9,
and 12 days) during refrigerated storage at 4
Approximately 10 g of each burger sample was homogenized with 90 mL of distilled water using a laboratory homogenizer (Ultra-Turrax T25, cat. no. 00700354, IKA, Staufen, Germany). The pH of the homogenate was measured using a digital pH meter (Model MP220, cat. no. 30200222, Mettler-Toledo, Greifensee, Switzerland) calibrated with standard buffer solutions at pH 4.0 and 7.0.
Lipid oxidation was evaluated by the TBARS assay as described by [21], with modifications. A 10 g sample was blended with 50 mL of distilled water and 2.5 mL of 4% HCl. The mixture was distilled, and 5 mL of the distillate was reacted with 5 mL of 0.02 M thiobarbituric acid in a boiling water bath for 35 minutes. Absorbance was measured at 532 nm using a UV-Vis spectrophotometer. TBARS values were expressed as mg malondialdehyde (MDA)/kg of sample.
Sensory evaluation was conducted to determine the effects of chia seed extract (CSE) on the organoleptic quality of beef burger patties during refrigerated storage.
A semi-trained panel consisting of 15 members (aged 22–45 years), including postgraduate students and staff from the Department of Food Science, Taif University, participated in the study. All panelists were regular consumers of meat products and received orientation on the evaluation criteria and use of the hedonic scale prior to testing sessions.
Burger patties were pan-fried to an internal temperature of 72 °C using a calibrated digital thermometer, cooled to approximately 40 °C, and cut into uniform pieces (~25 g). Samples were served warm on white plates coded with random three-digit numbers to ensure unbiased evaluation. The serving order was randomized to minimize positional effects. Panelists were provided with water and unsalted crackers to cleanse the palate between samples.
Panelists rated each attribute on a 9-point hedonic scale (1 = dislike extremely, 9 = like extremely). Mean scores and standard deviations were calculated.
Cooking yield was determined by weighing the beef burgers before and after cooking and expressed as a percentage:
All experimental data were analyzed using SPSS software (version 25.0; IBM
Corp., Armonk, NY, USA). Results are expressed as mean
The proximate composition of chia seed powder demonstrates its exceptional
nutritional value, supporting its use in functional food systems (Table 2).
Moisture content was low (5.84
| Parameter | Value (Mean | |
| Proximate composition (g/100 g) | ||
| Moisture | 5.84 | |
| Crude protein | 20.31 | |
| Crude fat | 31.66 | |
| Ash | 4.56 | |
| Total carbohydrates* | 37.63 | |
| Crude fiber | 23.40 | |
| Soluble dietary fiber | 7.12 | |
| Insoluble dietary fiber | 16.28 | |
| Fatty acid composition (% of total fat) | ||
| 62.3 | ||
| Linoleic acid (C18:2, ω-6) | 19.5 | |
| Oleic acid (C18:1) | 10.7 | |
| Palmitic acid (C16:0) | 4.2 | |
| Stearic acid (C18:0) | 2.1 | |
| Bioactive compounds | ||
| Total phenolic content (mg GAE/g) | 38.72 | |
| Total flavonoid content (mg QE/g) | 22.18 | |
| Tannic acid content (mg TAE/g) | 12.94 | |
| Antioxidant activity | ||
| DPPH inhibition (%) | 75.61 | |
| ABTS+ inhibition (%) | 81.47 | |
| FRAP (µmol Trolox equivalents/g) | 62.53 | |
| Phenolic compounds by HPLC (mg/g) | ||
| Gallic acid | 8.37 | |
| Caffeic acid | 4.15 | |
| Ferulic acid | 3.89 | |
| Chlorogenic acid | 2.91 | |
| Rutin | 2.46 | |
| Quercetin | 1.74 | |
| Mineral content (mg/100 g) | ||
| Calcium (Ca) | 590.4 | |
| Magnesium (Mg) | 335.7 | |
| Potassium (K) | 722.1 | |
| Phosphorus (P) | 744.5 | |
| Sodium (Na) | 8.3 | |
| Iron (Fe) | 7.62 | |
| Zinc (Zn) | 4.27 | |
| Copper (Cu) | 0.89 | |
| Vitamin content | ||
| Vitamin C (mg/100 g) | 7.28 | |
| Thiamine – B1 (mg/100 g) | 0.82 | |
| Riboflavin – B2 (mg/100 g) | 0.27 | |
| Niacin – B3 (mg/100 g) | 5.63 | |
| Pyridoxine – B6 (mg/100 g) | 0.48 | |
| Vitamin A (µg/100 g) | 142.5 | |
| Vitamin E – |
12.84 | |
| Vitamin K (µg/100 g) | 3.51 | |
*Carbohydrates calculated by difference: 100 – (protein + fat + ash + moisture).
All values are expressed as mean
Chia seeds were also rich in bioactive compounds: total phenolic content (TPC)
was 38.72
The high bioactivity of chia seed extract, demonstrated through multiple assays and a diverse phenolic profile, highlights its dual functionality: it can retard lipid oxidation and enhance oxidative stability in lipid-rich foods, including ground beef, while contributing nutritional and health-promoting benefits. Incorporation of chia extract into meat products thus represents a promising natural alternative to synthetic antioxidants, aligning with consumer demand for clean-label, functional ingredients [29].
Regarding vitamin composition, chia seed powder exhibited a diverse profile,
providing both water- and fat-soluble vitamins. Vitamin C content was 7.28
High-performance liquid chromatography (HPLC) was employed to identify and
quantify the major phenolic compounds present in chia seed (Salvia
hispanica L.) extract, providing insights into its antioxidant potential and
suitability as a functional food ingredient (Fig. 1; Tables 3,4). The analysis
was performed using a Shimadzu LC-20AT HPLC system equipped with a diode array detector (DAD; Shimadzu Corp.,
Kyoto, Japan), allowing precise profiling of phenolic constituents based on
retention time and UV-visible spectra. The chia extract was dissolved in
HPLC-grade methanol, filtered through a 0.45 µm PTFE syringe filter,
and stored at 4 °C until analysis. Separation was achieved on a C18
reverse-phase column (250 mm
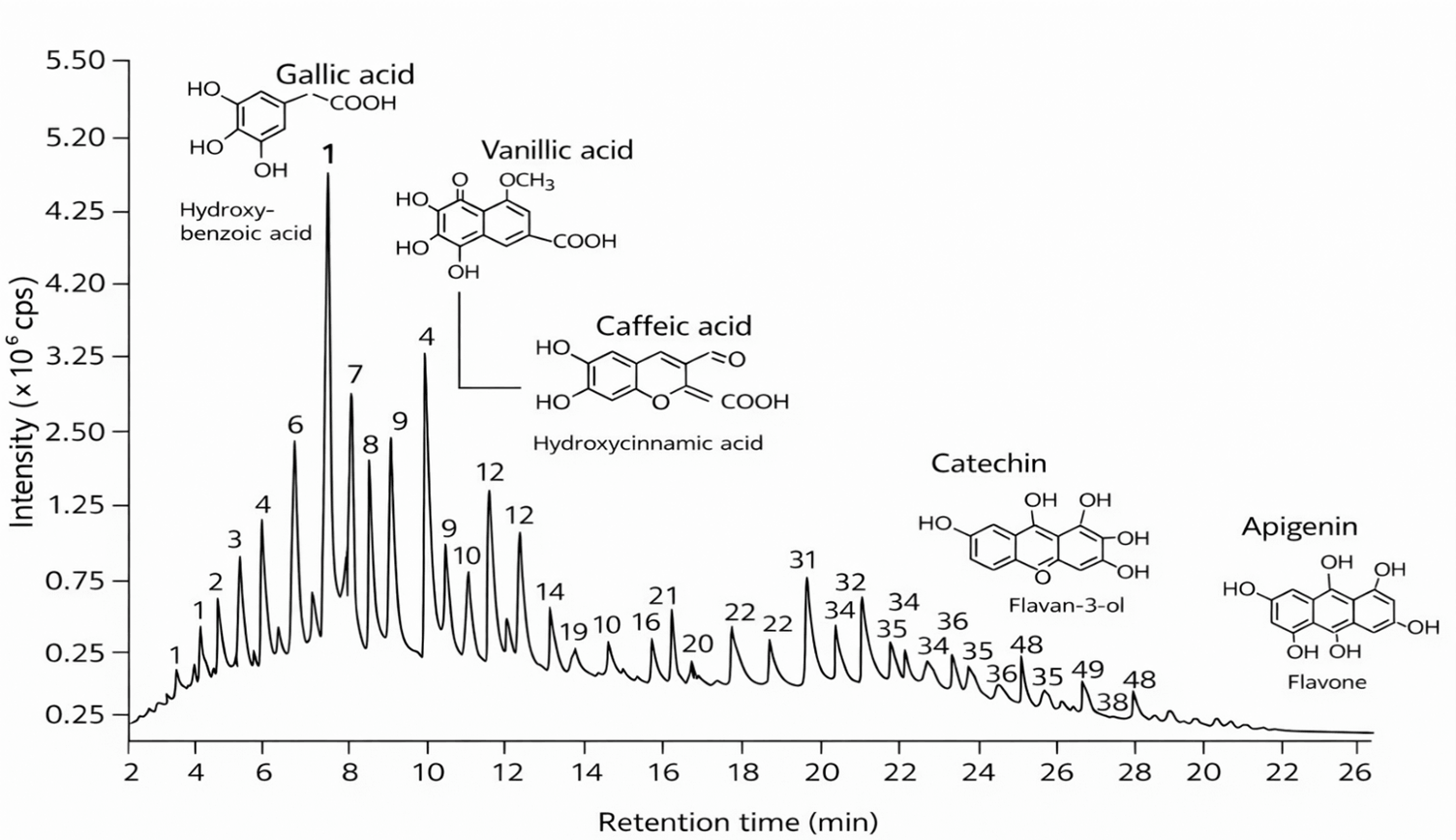 Fig. 1.
Fig. 1.
Base peak chromatogram of chia seed (Salvia hispanica L.) extract obtained by high-performance liquid chromatography with diode array detection (HPLC-DAD). The chromatogram exhibits a complex phenolic profile comprising 48 distinct peaks eluting between 2.1 and 26.0 minutes. Early-eluting peaks (2–10 min), including gallic acid, protocatechuic acid, and p-hydroxybenzoic acid, correspond to low-molecular-weight, highly polar hydroxybenzoic acids, while later peaks represent less polar phenolics, including ferulic acid, rutin, and quercetin. The most intense peaks were observed between 22 and 24 minutes, corresponding to high-intensity flavonoid compounds (see Table 4). Peaks 13 and 15 represent to minor early-eluting traces of rutin and quercetin; however, their major quantitative signals were recorded at 32.48 and 39.72 min, respectively. UV absorbance was monitored at 280 nm and 320 nm for optimal detection of phenolic acids and flavonoids. The chromatographic pattern, consistent with Table 3, reflects the diverse composition of hydroxybenzoic, hydroxycinnamic, and flavonol derivatives, which collectively contribute to the potent antioxidant potential of chia seed extract.
| Peak No. | Retention time (min) | Tentative compound | Compound class/Remark |
| 1 | 2.1 | Gallic acid | Hydroxybenzoic acid |
| 2 | 2.5 | Protocatechuic acid | Hydroxybenzoic acid |
| 3 | 3.1 | p-Hydroxybenzoic acid | Hydroxybenzoic acid |
| 4 | 3.8 | Vanillic acid | Hydroxybenzoic acid |
| 5 | 4.6 | Caffeic acid | Hydroxycinnamic acid |
| 6 | 5.1 | Chlorogenic acid | Hydroxycinnamic acid |
| 7 | 6.3 | Syringic acid | Hydroxybenzoic acid |
| 8 | 7.0 | Ferulic acid | Hydroxycinnamic acid |
| 9 | 8.1 | Sinapic acid | Hydroxycinnamic acid |
| 10 | 9.2 | p-Coumaric acid | Hydroxycinnamic acid |
| 11 | 10.4 | Catechin | Flavan-3-ol |
| 12 | 11.1 | Epicatechin | Flavan-3-ol |
| 13 | 12.2 | Rutin (trace) | Flavonol glycoside (early eluting trace) |
| 14 | 13.5 | Myricetin | Flavonol |
| 15 | 14.6 | Quercetin (trace) | Flavonol (early eluting trace) |
| 16 | 15.3 | Kaempferol | Flavonol |
| 17 | 16.0 | Naringenin | Flavanone |
| 18 | 17.1 | Apigenin | Flavone |
| 19 | 18.3 | Rosmarinic acid | Polyphenolic ester |
| 20 | 19.0 | Luteolin | Flavone |
| 21 | 19.5 | Caffeic acid derivative | Hydroxycinnamic acid conjugate |
| 22 | 19.9 | Ferulic acid derivative | Hydroxycinnamic acid conjugate |
| 23 | 20.5 | Rutin derivative | Flavonol glycoside conjugate |
| 24 | 21.0 | Quercetin-3-O-glucoside | Flavonol glycoside |
| 25 | 21.3 | Kaempferol derivative | Flavonol glycoside |
| 26 | 21.6 | Luteolin-7-O-glucoside | Flavone glycoside |
| 27 | 21.9 | Apigenin-7-O-glucoside | Flavone glycoside |
| 28 | 22.3 | Unidentified phenolic compound | Possible flavonoid derivative |
| 29 | 22.6 | Unidentified compound | Co-eluting phenolic glycoside |
| 30 | 22.8 | Chlorogenic acid derivative | Hydroxycinnamic conjugate |
| 31 | 23.0 | Rosmarinic acid derivative | Polyphenolic conjugate |
| 32 | 23.4 | Rutin (major) | Flavonol glycoside (confirmed in Table 4, 32.48 min) |
| 33 | 23.6 | Quercetin (major) | Flavonol (confirmed in Table 4, 39.72 min) |
| 34 | 23.8 | Myricetin derivative | Flavonol conjugate |
| 35 | 24.0 | Kaempferol derivative | Flavonol conjugate |
| 36 | 24.2 | Rosmarinic acid isomer | Phenolic ester derivative |
| 37 | 24.4 | Luteolin derivative | Flavone conjugate |
| 38 | 24.6 | Apigenin derivative | Flavone conjugate |
| 39 | 24.8 | Chlorogenic acid isomer | Hydroxycinnamic acid isomer |
| 40 | 25.0 | Rutin-like compound | Flavonol glycoside derivative |
| 41 | 25.1 | Quercetin-like compound | Flavonol derivative |
| 42 | 25.3 | Rosmarinic acid conjugate | Polyphenolic compound |
| 43 | 25.5 | Unidentified compound | Tentative phenolic conjugate |
| 44 | 25.6 | Unidentified compound | Tentative flavonoid derivative |
| 45 | 25.7 | Unidentified compound | Tentative phenolic polymer |
| 46 | 25.8 | Unidentified compound | Tentative phenolic polymer |
| 47 | 25.9 | Unidentified compound | Tentative phenolic polymer |
| 48 | 26.0 | Unidentified compound | Tentative phenolic polymer |
Notes:
| Phenolic compound | Class | Retention time (min) | Concentration (mg/g DW) |
| Gallic acid | Hydroxybenzoic acid | 6.27 | 8.37 |
| Caffeic acid | Hydroxycinnamic acid | 11.92 | 4.15 |
| Ferulic acid | Hydroxycinnamic acid | 21.34 | 3.89 |
| Chlorogenic acid | Hydroxycinnamic acid | 18.76 | 2.91 |
| Rutin | Flavonol glycoside | 32.48 | 2.46 |
| Quercetin | Flavonol | 39.72 | 1.74 |
Notes:
Quantitative analysis (Table 4) identified gallic acid (8.37 mg/g), caffeic acid (4.15 mg/g), and ferulic acid (3.89 mg/g) as the predominant phenolic compounds, accompanied by appreciable levels of chlorogenic acid (2.91 mg/g), rutin (2.46 mg/g), and quercetin (1.74 mg/g). Overall, hydroxybenzoic acids (e.g., gallic acid) and flavonols (e.g., quercetin, rutin) were the most abundant phenolic classes. These compounds are recognized for their potent antioxidant, anti-inflammatory, and antimicrobial properties. From a functional perspective, gallic and caffeic acids are known inhibitors of lipid peroxidation, while flavonols such as quercetin and rutin contribute cardiovascular, neuroprotective, and anti-carcinogenic effects. The phenolic composition observed in chia seed extract, therefore, underscores its potential as a natural antioxidant source capable of enhancing lipid stability, delaying rancidity, and improving the nutritional and functional quality of processed meat products. These findings highlight chia seed extract’s promise as a clean-label alternative to synthetic antioxidants, supporting current trends toward natural preservation strategies in the food industry. High-performance liquid chromatography with diode array detection (HPLC-DAD) was employed to characterize and quantify the major phenolic constituents of chia seed (Salvia hispanica L.) extract, providing insight into its antioxidant potential and suitability as a natural functional ingredient. The chromatographic analysis revealed a rich and complex phenolic profile, as illustrated in Fig. 1, comprising a total of 48 distinct peaks across the retention range of 2.1–26.0 minutes. The identified compounds were tentatively assigned based on retention times, UV spectral characteristics, and comparison with authentic standards and previously reported chromatographic data (Table 3).
Early-eluting peaks, particularly those appearing between 2 and 10 minutes, corresponded predominantly to low-molecular-weight, highly polar phenolic acids. These included gallic acid, protocatechuic acid, p-hydroxybenzoic acid, vanillic acid, caffeic acid, chlorogenic acid, and ferulic acid, among others. Such compounds belong primarily to the hydroxybenzoic and hydroxycinnamic acid classes, which are well known for their strong antioxidant and radical-scavenging activity. Mid-to-late eluting peaks, detected between 15 and 26 minutes, represented larger, less polar compounds such as flavonols and flavones, including myricetin, quercetin, kaempferol, luteolin, and apigenin. The presence of both phenolic acids and flavonoids underscores the chemical diversity of chia seed extract, reflecting its multifunctional bioactivity potential.
Notably, peaks 13 and 15 corresponded to early-eluting traces of rutin and quercetin; however, their major quantitative signals were observed at 32.48 min and 39.72 min, respectively. These later peaks were used for quantification and are presented in Table 4. The chromatogram thus exhibits a bimodal pattern typical of plant extracts, where early peaks indicate polar phenolic acids and later peaks represent high-molecular-weight flavonoids. UV absorbance was monitored at 280 nm and 320 nm, which are optimal wavelengths for detecting phenolic acids and flavonoids, respectively.
Quantitative analysis (Table 4) revealed that gallic acid (8.37
Chlorogenic acid (2.91
The coexistence of hydroxycinnamic acids and flavonols in chia seed extract highlights its multifunctional role in food systems. These compounds not only enhance the extract’s antioxidant activity but also contribute to technological benefits, including improved lipid stability, color retention, and moisture preservation. In meat formulations, for instance, chia seed extract can significantly suppress oxidative deterioration, extend product freshness, and reduce reliance on synthetic antioxidants such as BHT and BHA. Functionally, the extract offers dual benefits—enhancing nutritional value through bioactive phenolics while simultaneously improving product stability by mitigating oxidative processes. Additionally, the phenolic matrix contributes to flavor and color stabilization, both crucial for maintaining consumer acceptability during storage.
The chromatographic and quantitative findings (Tables 3,4) demonstrate that chia seed extract is a potent source of both hydroxybenzoic and hydroxycinnamic acids, complemented by biologically active flavonols. The predominance of gallic acid, together with significant levels of caffeic, ferulic, and chlorogenic acids, ensures robust radical-scavenging activity, while rutin and quercetin further enhance biological efficacy and processing stability. This chemical composition confirms chia seed extract as a natural, clean-label antioxidant with high potential for application in oxidation-sensitive foods, particularly processed meats. Such findings substantiate the extract’s dual role as a nutraceutical and natural preservative, supporting the development of healthier and more sustainable food preservation strategies in response to increasing consumer demand for synthetic additive-free products.
This analytical approach enabled a comprehensive characterization of the
bioactive composition of chia seed (Salvia hispanica L.) extract,
forming the foundation for understanding its antioxidant efficacy and potential
applications in food systems. The identified phenolic compounds (Table 4)
emphasize chia’s dual functionality as both a nutraceutical and a natural
preservative, supporting its incorporation into functional and shelf-stable food
formulations. Among the detected compounds, gallic acid emerged as the most
abundant constituent (8.37
The co-occurrence of hydroxycinnamic acids and flavonols in chia seed extract underscores its multifunctionality in food systems. These compounds not only provide potent antioxidant protection but also influence technological attributes such as lipid stability, moisture retention, and color preservation. Their presence contributes to the improvement of organoleptic properties and storage stability in formulated products. In meat-based systems, for instance, the addition of chia extract has the potential to suppress oxidative degradation, prolong product freshness, and reduce the dependence on synthetic antioxidants such as butylated hydroxytoluene (BHT) and butylated hydroxyanisole (BHA). From a formulation perspective, chia extract fulfills a dual role—enhancing nutritional quality through its diverse bioactive compounds, while simultaneously improving oxidative stability through its phenolic matrix. This duality extends to functional outcomes such as flavor protection, color stabilization, and maintenance of sensory quality throughout storage. The synergistic contribution of gallic, caffeic, ferulic, and chlorogenic acids ensures robust radical-scavenging performance, while rutin and quercetin further amplify biological benefits and thermal resilience.
Overall, the diversity and abundance of phenolic acids and flavonoids identified in chia seed extract confirm its potential as a multifunctional, clean-label additive for oxidation-sensitive foods. The compositional profile (Tables 3,4) illustrates a balance between polar phenolic acids and less polar flavonoids, reflecting a phytochemical spectrum capable of broad antioxidant coverage. These findings substantiate chia seed extract’s application as a natural preservative and nutraceutical, capable of enhancing both the nutritional and technological quality of food products. In particular, its efficacy in stabilizing lipid systems and suppressing oxidative rancidity positions chia extract as a promising alternative to synthetic antioxidants, aligning with current industry trends favoring healthier and more sustainable preservation strategies.
The proximate composition of beef burger samples enriched with chia seed
(Salvia hispanica L.) extract was evaluated on day one of refrigerated
storage to determine its influence on product nutritional quality. Moisture
content ranged from 69.60 to 71.16 g/100 g across all formulations, with no
significant differences observed among treatments (p
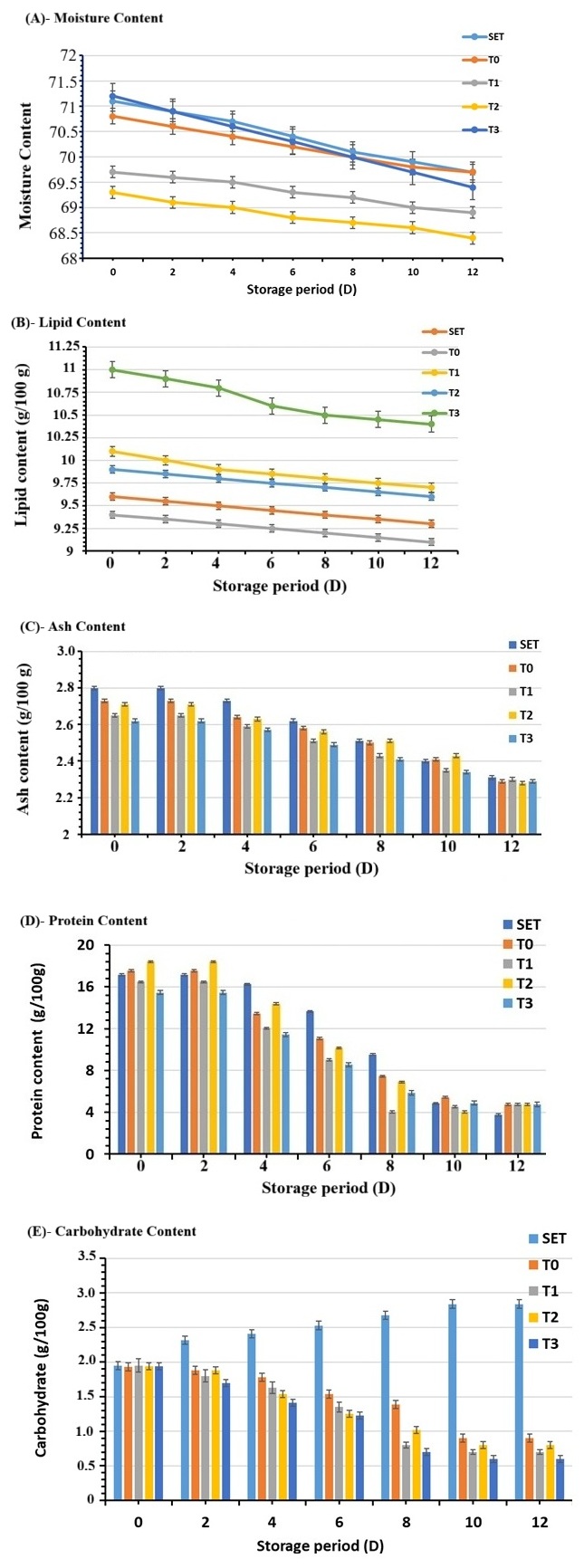 Fig. 2.
Fig. 2.
Proximate composition of beef burgers prepared with chia seed extracts during cold moisture content decreased gradually in all beef burger treatments during refrigerated storage, a typical trend attributed to water migration and evaporation. (A) Moisture content: Shows changes in moisture percentage of control and chia-supplemented beef burgers (T1–T3) during cold storage, reflecting water migration and retention behavior. (B) Protein content: Illustrates the effect of chia seed extract incorporation and storage time on protein levels of beef burgers. (C) Fat content: Demonstrates variations in fat percentage among treatments during refrigerated storage. (D) Ash content: Represents changes in total mineral (ash) content of beef burgers as affected by chia seed extract concentration and storage duration. (E) Carbohydrate content: Displays the influence of chia seed extract addition on calculated carbohydrate content during storage. Chia-supplemented samples (T1–T3) initially exhibited slightly lower moisture values, likely due to the water-binding and gel-forming properties of chia polysaccharides. By day 12, however, T3 (1.5% chia extract) demonstrated superior moisture retention compared with the other chia treatments, suggesting that higher chia incorporation may stabilize water distribution within the meat matrix.
| Parameter | SET | T0 | T1 (0.5%) | T2 (1.0%) | T3 (1.5%) |
| Moisture (g/100 g) | 71.13 |
71.16 |
69.81 |
69.60 |
70.66 |
| Protein (g/100 g) | 15.64 |
15.92 |
16.00 |
16.02 |
16.02 |
| Lipid (g/100 g) | 9.59 |
9.34 |
9.53 |
10.10 |
10.98 |
| Ash (g/100 g) | 2.69 |
2.71 |
2.74 |
2.80 |
2.78 |
| Carbohydrates (g/100 g) | 0.94 |
0.87 |
1.35 |
1.34 |
1.01 |
| pH | 6.10 |
6.07 |
6.00 |
6.04 |
6.11 |
Note: Values are expressed as mean
A statistically significant increase in lipid content was observed in treatment
T3 (10.98
Thus, higher levels of chia extract incorporation not only increase total lipid
content but may also enhance the fatty acid profile of the product, contributing
to its nutritional value. Ash content remained consistent across all
formulations, indicating that chia addition did not markedly alter total mineral
content. Minor increases in carbohydrate levels were noted in treatments T1 and
T2, though these changes were not statistically significant (p
Lipid content also declined slowly across all groups throughout storage, most
likely as a result of oxidative degradation. T3, which began with the highest
lipid content, retained more fat than other treatments during storage. This
observation suggests a lipid stabilization of chia seed extract, attributable to
its richness in polyphenols, flavonoids, and tocopherols that may inhibit lipid
oxidation. Table 6 (Ref. [39]) presents the changes in cooking yield and
shrinkage percentage of beef burgers fortified with chia seed extracts (CSEs)
over 15 days of refrigerated storage. Cooking yield, an essential technological
parameter, reflects the ability of a product to retain water and fat during heat
treatment, while shrinkage corresponds to the dimensional and weight losses that
occur during cooking. Both parameters are critical indicators of product quality,
consumer acceptability, and industrial profitability [40, 41]. At day 0, the
incorporation of chia seed extracts significantly improved (p
| Parameter | Period (days) | Control | SET | T1 | T2 | T3 |
| Cooking yield (%) | 0 | 83.61 |
88.92 |
86.81 |
89.45 |
87.47 |
| 5 | 83.66 |
84.39 |
86.63 |
88.26 |
86.02 | |
| 10 | 73.80 |
74.92 |
78.11 |
79.81 |
84.34 | |
| 15 | 83.89 |
82.87 |
82.13 |
87.11 |
91.27 | |
| Shrinkage (%) | 0 | 12.92 |
13.35 |
13.53 |
13.06 |
11.35 |
| 5 | 13.93 |
13.06 |
13.46 |
12.84 |
13.20 | |
| 10 | 15.97 |
15.74 |
15.99 |
15.16 |
13.10 | |
| 15 | 13.31 |
13.30 |
14.87 |
14.64 |
11.35 |
Note:
By day 5, a gradual reduction in cooking yield was observed across all samples,
likely associated with early protein denaturation and oxidative interactions that
reduce the water-holding capacity of myofibrillar proteins. Nonetheless,
CSE-treated burgers maintained significantly higher yields compared to the
control, confirming the stabilizing role of chia bioactives during short-term
storage. The yield values for T2 (88.26%) and T1 (86.63%) remained particularly
high, indicating that chia extract effectively mitigates the moisture and fat
loss commonly seen during initial storage stages. As storage progressed to day
10, all samples showed a marked decline in cooking yield, with the control and
SET dropping to 73.80% and 74.92%, respectively. This reduction can be
attributed to protein oxidation, structural weakening, and increased water
mobility within the meat matrix. However, chia-supplemented burgers demonstrated
a significantly improved performance. Notably, T3 (84.34%) retained
substantially higher yield compared with all other treatments (p
Interestingly, by day 15, a notable increase in cooking yield was recorded for all chia-supplemented samples, with T3 reaching the highest yield (91.27%), followed by T2 (87.11%), both markedly outperforming the control (83.89%) and SET (82.87%). This apparent recovery may be explained by the water-binding and film-forming capacity of chia mucilage, which may continue to reinforce the meat matrix structure during cold storage. The chia extracts’ polyphenolic components, particularly gallic, caffeic, and ferulic acids, may also inhibit oxidative degradation of muscle proteins and lipids, thereby preserving the physicochemical conditions necessary for optimal water retention [43, 44]. These interactions likely prevent excessive protein cross-linking and preserve a more hydrated network, contributing to higher yields at the final storage stage.
Overall, the data indicate that chia seed extract supplementation significantly enhances the technological functionality of meat systems by improving cooking yield. The improvement is both immediate (day 0)—due to hydrocolloid formation—and sustained (up to day 15)—through antioxidant protection and protein–polysaccharide interactions. Treatments containing higher concentrations of chia extract (particularly T3) consistently achieved superior performance throughout storage, underscoring a dose-dependent relationship between chia content and yield stability.
In contrast, the control and SET samples exhibited greater fluctuations and overall lower yields, suggesting that chia’s combination of hydrophilic fibers and bioactive compounds provides a more comprehensive stabilizing effect than conventional synthetic antioxidants. The ability of chia components to form viscous networks and retain moisture also contributes to better product juiciness, mouthfeel, and consumer acceptability. From a mechanistic perspective, chia’s hydrocolloids act as natural water binders, reducing drip loss during cooking, while the antioxidants and phenolic acids (e.g., gallic, ferulic, and chlorogenic acids) counteract oxidative stress, maintaining protein functionality. This synergistic behavior enhances the product’s structural integrity, yielding higher cooking recovery rates and lower post-cooking deformation. Collectively, the results demonstrate that chia seed extract supplementation serves a dual functional role in meat systems:
(1) Technological: by improving cooking yield and preserving structural integrity through hydrocolloidal gel formation and moisture entrapment; and
(2) Nutritional: by introducing natural antioxidants and omega-3 fatty acids that mitigate oxidation and improve overall product stability.
These outcomes validate chia seed extract as a promising clean-label alternative to synthetic antioxidants (e.g., BHT, BHA) in processed meats. Beyond yield enhancement, the presence of chia bioactives contributes to extended shelf-life, improved sensory attributes, and a more sustainable, health-conscious product formulation [44, 45]. Future studies exploring the molecular interactions between chia polysaccharides and meat proteins, as well as the sensory perception of CSE-enriched products, will be essential to further optimize formulation levels and validate consumer acceptance. Nonetheless, the present findings clearly establish that chia seed extracts, particularly at higher inclusion levels (T3), offer substantial benefits in terms of cooking performance, product yield, and storage stability—confirming their suitability as multifunctional ingredients in modern meat processing.
Color is a crucial quality parameter for meat products, as it strongly influences consumer perception of freshness and overall acceptability [46]. The lightness (L*) values of beef burgers enriched with chia seed extracts (CSEs) were monitored throughout 15 days of refrigerated storage (Table 7). At day 0, burgers supplemented with CSE, particularly T3 (54.51), showed slightly lower L* values than the control (57.30). This modest reduction may be attributed to the presence of chia pigments and phenolic compounds, which impart a natural brownish tint and reduce surface reflectance [35, 42].
| Parameter | Day | Control | SET | T1 | T2 | T3 |
| L* (Lightness) | 0 | 57.30 |
58.25 |
57.07 |
56.11 |
54.51 |
| 5 | 56.12 |
56.75 |
55.23 |
53.60 |
54.48 | |
| 10 | 55.56 |
56.31 |
54.49 |
53.09 |
54.58 | |
| 15 | 57.44 |
54.38 |
54.21 |
54.31 |
54.64 | |
| a* (Redness) | 0 | 6.14 |
7.72 |
5.33 |
4.18 |
3.75 |
| 5 | 6.12 |
7.41 |
4.37 |
3.12 |
4.06 | |
| 10 | 5.48 |
7.40 |
4.83 |
4.76 |
4.29 | |
| 15 | 6.62 |
6.71 |
3.94 |
3.96 |
3.92 | |
| b* (Yellowness) | 0 | 11.84 |
13.65 |
20.24 |
22.18 |
22.94 |
| 5 | 11.30 |
12.73 |
17.16 |
20.25 |
22.42 | |
| 10 | 10.24 |
12.84 |
17.02 |
19.46 |
21.95 | |
| 15 | 11.65 |
10.16 |
15.47 |
18.80 |
21.00 |
Note:
Despite these initial differences, no significant decline in lightness was
observed during storage across all treatments (p
The a* parameter, reflecting the intensity of red coloration, decreased progressively in all samples during refrigerated storage, consistent with the oxidative conversion of oxymyoglobin to metmyoglobin. At day 0, the control (6.14) and SET (7.72) samples exhibited significantly higher a* values compared to chia-enriched formulations, with T3 showing the lowest initial redness (3.75). This reduction may result from partial dilution of meat pigments by chia extract or interactions between chia-derived antioxidants and myoglobin chemistry. Despite their lower initial redness, chia-containing treatments displayed improved color stability during storage. Particularly after day 10, T2 and T3 exhibited less pronounced declines in a* compared to the control, suggesting that bioactive constituents in chia, including polyphenols, flavonoids, and rosmarinic acid, helped slow pigment oxidation. This stabilization effect implies that chia seed extract contributes to preserving meat redness over time, even if the initial intensity appears reduced.
The b* values were strongly influenced by chia incorporation. At day 0, chia-enriched burgers showed significantly higher yellowness, with T3 reaching 22.94 compared to 11.84 in the control. This increase is attributable to the natural yellowish-brown pigments in chia extracts, likely derived from phenolic and carotenoid-like compounds [47]. Although a gradual decline in b* values occurred during storage, chia-containing treatments consistently maintained higher yellowness than the control. Overall, T3 demonstrated the most balanced color performance, combining minimal changes in L*, attenuated loss of redness, and sustained higher yellowness throughout storage. This indicates that chia seed extracts not only influence the initial appearance of meat products but also contribute to stabilizing color quality during shelf-life.
Although chia seed extract caused minor initial darkening and reduced redness, it markedly enhanced color stability and oxidative resistance throughout storage. This effect is attributed to the synergistic action of chia-derived antioxidants, including rosmarinic acid, caffeic acid, and tocopherols, which can scavenge reactive oxygen species and stabilize myoglobin pigments. Consequently, chia extract not only modifies the initial appearance of beef burgers but also plays a protective role in maintaining desirable color attributes during refrigerated storage.
Chia seed extract acted as an effective natural color stabilizer in beef burgers, maintaining visual quality during refrigerated storage. Among the instrumental color parameters, b* (yellowness) was most affected by chia addition. At day 0, the control sample showed a b* value of 11.84, while T3 exhibited a significantly higher value of 22.94, reflecting the presence of yellow–brown pigments associated with chia polyphenols and fiber-bound compounds [48]. Unlike the L* and a* values, which declined gradually with storage, the b* values in T2 and T3 remained stable, indicating a protective effect against oxidative discoloration. This sustained color intensity enhances product shelf-life and supports the use of chia extract as a clean-label antioxidant in meat formulations (Table 7).
The incorporation of chia seed extracts (CSEs) noticeably influenced the color stability of beef burgers during refrigerated storage. While initial redness (a*) values were slightly lower in chia-treated samples compared to controls, these formulations exhibited enhanced oxidative stability, maintaining more consistent redness over time. This outcome can be attributed to the antioxidant activity of chia-derived polyphenols, flavonoids, and tocopherols, which delay myoglobin oxidation. Yellowness (b*) values were significantly higher in CSE-enriched samples, particularly T3, reflecting the natural yellow–brown pigments and fiber-bound phenolics inherent in chia. Importantly, this attribute remained stable throughout storage, suggesting a protective effect against oxidative discoloration. Lightness (L*) values showed minimal change, indicating that chia incorporation did not negatively affect product brightness.
Overall, these findings suggest that chia seed extracts serve as effective natural color stabilizers, mitigating oxidative changes and supporting visual quality retention. Such color enhancement may improve the visual distinctiveness of functional meat products, offering a potential marketing advantage in health-conscious product lines. From a technological perspective, the ability of chia extracts to reduce lightness loss and limit color fluctuations positions them as multifunctional, clean-label ingredients in meat product development [47, 48]. Further consumer-focused studies are warranted to determine the concentration levels that balance appealing color characteristics with optimal nutritional and oxidative stability.
Texture is one of the most critical determinants of meat product quality, influencing both consumer acceptability and industrial performance. In this study, the influence of chia seed extract (CSE) on the texture profile of beef burgers was evaluated through five key parameters—hardness, springiness, cohesiveness, chewiness, and resilience—over 15 days of refrigerated storage at 4 °C. A three-dimensional (3D) response surface methodology (RSM) approach was employed to visualize the interactive effects of CSE concentration and storage duration on texture characteristics (Fig. 3A–E). This analytical approach provided an integrated understanding of how chia extracts, through their hydrocolloid-forming capacity and bioactive compounds, modulate the mechanical behavior of meat matrices during storage (Table 8). Table 8 presents the evolution of texture parameters in beef burgers supplemented with different concentrations of CSE (T1–T3), compared to the control (without additives) and SET (synthetic antioxidant-treated) samples. Texture attributes are vital indicators of the structural integrity, protein–water interactions, and fat-binding capacity of the meat system. These properties can deteriorate during cold storage due to protein denaturation, lipid oxidation, and moisture migration. However, the addition of CSE, rich in phenolics, polysaccharides, and mucilage, may mitigate these changes and improve texture stability through its antioxidant and water-retention capabilities.
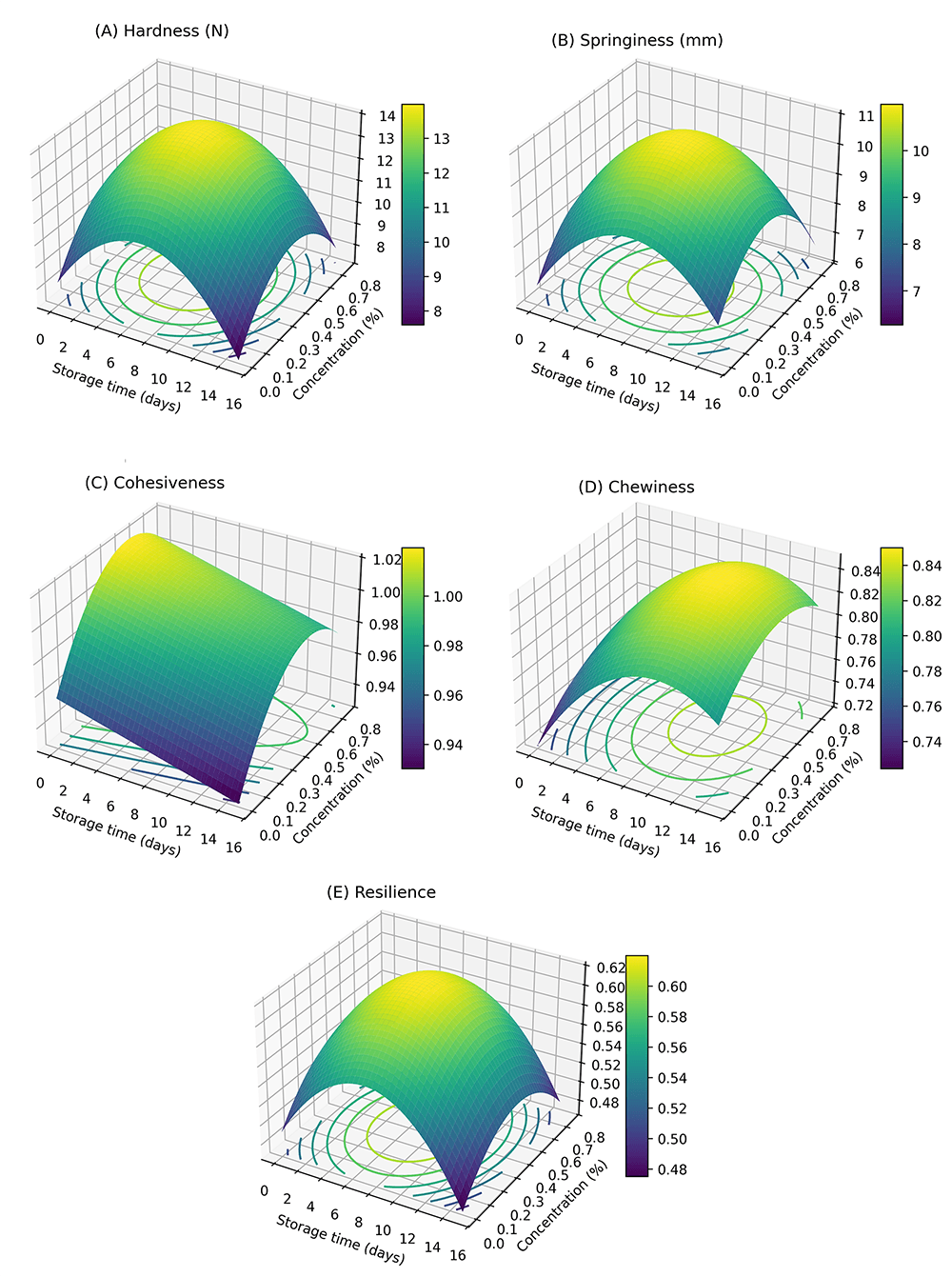 Fig. 3.
Fig. 3.
Three-dimensional (3D) response surface plots from texture profile analysis of beef burgers prepared with chia seed extracts during 15 days of cold storage at 4 °C. (A) Hardness: Illustrates the interactive effects of CSE concentration and storage duration on burger firmness. (B) Springiness: Shows how CSE concentration and storage time influence the elastic recovery of burgers. (C) Cohesiveness: Depicts the combined effects of CSE and storage on internal structural integrity. (D) Chewiness: Represents the influence of CSE and storage duration on the energy required to chew the burgers. (E) Resilience: Displays the effect of CSE and storage on the ability of burgers to recover their shape after deformation.
| Parameter | Day | Control | SET | T1 | T2 | T3 |
| Hardness (N) | 0 | 12.41 |
11.73 |
9.35 |
12.09 |
12.43 |
| 5 | 15.62 |
12.91 |
9.26 |
14.93 |
12.86 | |
| 10 | 14.90 |
13.21 |
10.01 |
14.43 |
13.86 | |
| 15 | 13.43 |
12.99 |
10.35 |
13.42 |
11.49 | |
| Springiness (mm) | 0 | 6.30 |
6.42 |
6.40 |
6.48 |
6.35 |
| 5 | 6.36 |
6.40 |
6.32 |
6.33 |
6.38 | |
| 10 | 6.34 |
6.43 |
6.36 |
6.10 |
6.37 | |
| 15 | 6.29 |
6.28 |
6.41 |
6.40 |
6.39 | |
| Cohesiveness | 0 | 0.87 |
1.00 |
0.99 |
0.93 |
0.96 |
| 5 | 0.91 |
0.99 |
0.94 |
0.95 |
0.95 | |
| 10 | 0.94 |
0.96 |
0.95 |
0.96 |
0.94 | |
| 15 | 0.96 |
0.99 |
0.94 |
0.97 |
0.94 | |
| Chewiness (N·mm) | 0 | 68.24 |
75.12 |
61.18 |
72.46 |
73.94 |
| 5 | 70.12 |
73.33 |
62.43 |
75.58 |
72.11 | |
| 10 | 72.40 |
71.36 |
65.29 |
74.11 |
70.34 | |
| 15 | 71.31 |
72.05 |
66.52 |
71.87 |
68.74 | |
| Resilience | 0 | 0.44 |
0.46 |
0.43 |
0.48 |
0.42 |
| 5 | 0.45 |
0.47 |
0.44 |
0.45 |
0.41 | |
| 10 | 0.43 |
0.45 |
0.42 |
0.44 |
0.38 | |
| 15 | 0.44 |
0.46 |
0.43 |
0.45 |
0.41 |
Notes:
Hardness values varied significantly (p
Fig. 3A, the three-dimensional response surface plot for hardness illustrates
the interactive effects of chia seed extract (CSE) concentration and storage
duration on the firmness of beef burgers during 15 days of refrigerated storage
at 4 °C. A gradual decline in hardness was observed in all formulations
over time, reflecting typical softening associated with protein denaturation,
moisture migration, and weakening of the myofibrillar network during cold storage
[35, 42]. Notably, burgers fortified with moderate CSE levels (T2: 1.0%)
maintained hardness values comparable to the control, indicating good structural
preservation. In contrast, higher inclusion (T3: 1.5%) produced a slightly
softer texture (p
This firmness retention effect is mainly linked to the hydrocolloid-forming capacity of chia mucilage and its high dietary fiber content, which together generate a gel-like network within the meat matrix. This structure improves water-holding capacity (WHC) and minimizes structural disintegration during storage [35, 42, 48]. Furthermore, chia-derived phenolics—particularly gallic, ferulic, and caffeic acids—are likely to form non-covalent and covalent interactions with muscle proteins and connective tissue, contributing to cross-linking that reinforces matrix integrity and resists oxidative or enzymatic degradation [35, 42].
Conversely, hardness in the control and SET samples decreased more rapidly, reflecting the absence of hydrocolloid reinforcement and limited oxidative protection. These findings agree with previous reports indicating that plant extracts rich in polyphenols and soluble fibers synergistically preserve textural stability and extend the sensory shelf-life of meat products during refrigerated storage [49]. Collectively, the enhanced hardness retention in CSE-enriched burgers underscores the multifunctional role of chia extract as both an antioxidant and a natural hydrocolloid, thereby improving product quality and consumer acceptability throughout cold storage.
Springiness values were relatively consistent across treatments and days
(6.10–6.48 mm), showing no significant differences (p
The response surface plot for springiness (Fig. 3B) shows that no significant
differences (p
This effect can be attributed to the hydrophilic polysaccharides in chia mucilage, which form a gel-like network within the meat matrix, entrapping water and reinforcing protein interactions, thereby minimizing structural collapse [44, 45, 51]. Additionally, the presence of polyphenolic and flavonoid compounds such as quercetin, rutin, and chlorogenic acid may inhibit protein oxidation, preserving the integrity of myofibrillar proteins responsible for elasticity [35, 42]. The stable springiness observed during storage highlights chia seed extract’s multifunctional ability to sustain mechanical strength and enhance the perceived juiciness and freshness of beef burgers during cold storage.
Cohesiveness describes the internal bonding strength of the product’s matrix. At
day 0, the control exhibited the lowest cohesiveness (0.87
The response surface plot for cohesiveness (Fig. 3C) demonstrates that the
changes in all treatments during 15 days of refrigerated storage were not
statistically significant (p
Cohesiveness in meat systems primarily depends on protein–protein and protein–water interactions, which can deteriorate under oxidative or enzymatic stress [45, 52]. The enhanced cohesiveness observed in CSE-fortified burgers can be attributed to the dual functionality of chia seed components: (i) phenolic acids such as gallic, ferulic, and caffeic acids protect against protein oxidation by minimizing carbonyl formation and preserving sulfhydryl groups [39, 46], and (ii) chia mucilage, rich in soluble polysaccharides, forms hydrocolloid-like gels that fill interstitial spaces, enhance water binding, and stabilize the protein network [47]. Overall, the maintenance of cohesiveness during storage underscores chia seed extract’s structural and antioxidative roles, improving the texture, juiciness, and handling properties of beef burgers without significant degradation over time.
Chewiness, a function of hardness, cohesiveness, and springiness, reflects the
energy required to masticate the sample. Initially, chewiness values ranged from
61.18 N
Resilience measures the product’s ability to recover energy after deformation,
thus reflecting structural elasticity. Values ranged between 0.38 and 0.48 across
treatments. T2 displayed the highest resilience (0.48
Across all treatments, resilience values ranged narrowly between 0.38 and 0.48, with no statistically significant changes over time (same uppercase letter “A” in each column). Burgers supplemented with 1.0% CSE (T2) consistently exhibited slightly higher resilience values compared to the control and other treatments, suggesting that moderate levels of chia seed extract enhanced the elastic recovery of the meat structure. The 1.5% CSE (T3) group showed comparable resilience stability, indicating that higher chia inclusion did not compromise textural integrity.
This relative stability can be attributed to the hydrocolloid-forming polysaccharides present in chia mucilage, which interact with the meat matrix to strengthen the gel network and help retain water. Additionally, the antioxidant phenolic compounds identified in the extract—particularly gallic, ferulic, caffeic, and quercetin—may have contributed to maintaining protein functionality by limiting oxidative damage to myofibrillar proteins [49, 50, 51]. The combined hydrocolloid and antioxidant mechanisms likely prevented excessive protein cross-linking or degradation, thereby preserving textural elasticity during cold storage.
From a technological standpoint, the stability of resilience across all formulations indicates that chia seed extract effectively supports the mechanical structure of beef burgers without inducing textural deterioration. This is particularly relevant for refrigerated meat systems, where protein oxidation and moisture loss typically reduce elasticity over time. Therefore, the inclusion of CSE not only maintains the natural rebound capacity of the meat matrix but also contributes to the overall textural and structural quality of the product.
Collectively, these findings confirm that chia seed extract functions as a natural stabilizer, preserving resilience and ensuring that the final product retains desirable firmness and elasticity throughout storage. Such stability reinforces its potential as a clean-label antioxidant and texture-enhancing agent in reformulated meat products aimed at extending shelf-life while maintaining consumer-acceptable texture profiles.
Overall Evaluation: The incorporation of chia seed extracts effectively
enhanced or preserved the texture of beef burgers during refrigerated storage.
Treatments containing moderate concentrations (T2) achieved the most favorable
balance between firmness, elasticity, and cohesiveness, whereas high
concentrations (T3) resulted in slightly softer but juicier products. Chia’s
physicochemical properties—especially its soluble fiber and
mucilage—contribute to improved water-holding capacity and gel-forming
potential, preventing excessive hardening and moisture loss over time.
Furthermore, chia’s abundant phenolic antioxidants likely inhibited protein
oxidation and crosslinking, thus maintaining a stable texture profile during
storage. From a technological standpoint, these findings demonstrate that chia
seed extract functions as a natural texture stabilizer comparable to synthetic
antioxidants, with added benefits of water retention and oxidative protection.
This aligns with the growing demand for clean-label, plant-derived additives in
processed meat formulations. The ability of chia components to maintain
consistent hardness, chewiness, and cohesiveness over 15 days indicates their
synergistic action between hydrocolloid and antioxidant properties. Overall, the
inclusion of chia seed extracts, particularly at intermediate levels (T2),
provided significant texture preservation without compromising sensory
attributes. This suggests potential application in reformulated meat systems
aimed at improving shelf-life, juiciness, and structural stability using natural
alternatives to synthetic stabilizers. Future work should focus on correlating
these textural changes with microstructural and rheological analyses to elucidate
the mechanisms of chia–protein interactions responsible for the observed
improvements. These 3D RSM plots clearly demonstrate that chia seed extract (CSE)
not only enhances oxidative stability but also plays a pivotal role in preserving
the textural integrity of beef burgers during refrigerated storage. The
synergistic functionality of chia mucilage, dietary fiber, and polyphenolic
compounds underscores its value as a multifunctional, clean-label additive for
meat processing. Among the tested inclusion levels, 1.0% CSE (T2) provided the
most balanced texture profile, maintaining the highest resilience and overall
textural stability during storage, whereas 1.5% CSE (T3) exhibited slightly
lower resilience values but retained desirable firmness and structural integrity.
The T1 formulation produced softer textures, suitable for applications requiring
enhanced tenderness, while T3 imparted greater hardness and cohesiveness.
Cohesiveness and springiness remained largely stable (p
Lipid oxidation is one of the most critical factors affecting the quality,
safety, and shelf-life of meat products during storage. It leads to the
development of rancid flavors, discoloration, and nutritional deterioration. The
thiobarbituric acid reactive substances (TBARS) assay was used to assess the
extent of lipid oxidation in beef burgers formulated with chia seed extract (CSE)
at varying concentrations (T1–T3) and stored at 4 °C for 15 days. The
corresponding data are presented in Table 9. At the beginning of storage (day 0),
the malonaldehyde (MDA) content in the control burgers was 0.47 mg/kg, which
increased sharply to 2.52 mg/kg by day 10 before slightly declining to 1.27 mg/kg
at day 15. This pattern reflects the typical progression of lipid oxidation,
where primary oxidation products (hydroperoxides) accumulate and subsequently
decompose into secondary aldehydes such as MDA. The increase observed in the
control samples was statistically significant (p
| Treatment | 0th day | 5th day | 10th day | 15th day |
| Control | 0.47 |
0.51 |
2.52 |
1.27 |
| SET | 0.27 |
0.28 |
0.38 |
0.33 |
| T1 (Low CSE) | 0.27 |
0.40 |
0.48 |
0.62 |
| T2 (Med CSE) | 0.23 |
0.42 |
0.45 |
0.59 |
| T3 (High CSE) | 0.21 |
0.35 |
0.36 |
0.28 |
Notes:
In contrast, burgers containing chia seed extract (CSE) or synthetic antioxidant (SET) displayed markedly lower TBARS values across all time points, indicating effective oxidative stabilization. The SET-treated samples showed only minimal changes (0.27–0.38 mg/kg) over 15 days, consistent with its known antioxidant potency. Among the CSE formulations, the T3 treatment (high CSE) exhibited the lowest MDA concentrations throughout storage, ranging from 0.21 to 0.36 mg/kg.
The T2 formulation (medium CSE) also effectively delayed lipid peroxidation, showing only minor fluctuations from 0.23 mg/kg at day 0 to 0.59 mg/kg at day 15. Although there was a slight upward trend, the changes over time were not statistically significant until the final sampling point (A–B superscripts), suggesting that moderate chia extract concentrations are sufficient to retard oxidation over two weeks of refrigerated storage.
Importantly, the T1 formulation (low CSE) maintained statistically similar MDA
values throughout storage (all labeled “A”), indicating no significant increase
(p
Among all treatments, the control group exhibited the highest degree of
oxidation, while T3 (high CSE) and SET (synthetic antioxidant) performed best in
preserving oxidative stability. The low TBARS values in T3 samples across all
time points (
Mechanistically, the antioxidant activity of CSE can be attributed to both its
hydrophilic and lipophilic constituents. The hydrophilic phenolics quench
reactive oxygen species (ROS) in the aqueous phase, while the viscous chia
mucilage forms a network that restricts lipid mobility and oxygen penetration.
This dual mode of action results in a consistent reduction of oxidation products
and enhances the overall oxidative stability of the burgers. Additionally, chia
seed’s high content of
Overall, the results from Table 9 confirm that chia seed extract significantly inhibits lipid oxidation in beef burgers during refrigerated storage. The efficacy increases with extract concentration, with T3 providing the most robust antioxidative effect. The absence of significant differences over time in the T1 and T2 groups underscores the stability and sustained antioxidant potential of chia phenolics even at moderate dosages. From a formulation perspective, CSE offers a promising natural alternative to synthetic antioxidants, aligning with clean-label and functional food trends. The use of chia seed extract not only supports oxidative stability but also complements previous findings (Tables 6,7,8) showing improvements in cooking yield and textural quality.
Thus, chia seed extract can be considered a multifunctional ingredient—providing both antioxidant protection and technological benefits—making it highly suitable for the development of healthier, more stable meat products.
The observed antioxidant effect can be attributed to chia’s rich bioactive
composition, including polyphenols (such as gallic, caffeic, and ferulic acids),
flavonoids (notably quercetin and rutin), and
These findings are consistent with recent evidence showing that plant-derived antioxidants, when applied at effective concentrations, can rival or even surpass the activity of synthetic preservatives such as BHT and BHA [54, 55]. Moderate CSE inclusion levels (T1 and T2) provided intermediate protection, with TBARS values remaining below the rancidity threshold but increasing more gradually over storage. This outcome reinforces the dose-dependent antioxidant capacity of chia extracts. The steady rise in TBARS across all treatments—most pronounced in the control and lower CSE levels—reflects the inherent progression of oxidative reactions in refrigerated meat systems, primarily driven by the abundance of unsaturated fatty acids, oxygen exposure, and iron-catalyzed prooxidant activity [56].
Beyond direct oxidative control, CSE may also enhance water retention and structural stability by forming a hydrophilic fiber–mucilage network that limits oxygen diffusion and delays lipid degradation. This dual protective mechanism highlights CSE’s multifunctionality, simultaneously improving oxidative resistance and maintaining textural quality during storage. From a technological and commercial standpoint, these attributes are highly relevant: the incorporation of CSE not only prolongs the shelf-life of meat products but also aligns with the growing demand for clean-label, naturally preserved foods. Its dual role as a preservative and functional ingredient, coupled with its rich nutraceutical profile, positions chia extract as a promising additive for developing healthier meat formulations targeted at quality-driven and health-conscious consumer markets [56, 57, 58].
While TBARS analysis provided clear evidence of lipid stabilization by chia seed extract, it primarily reflects secondary oxidation products. Future studies should incorporate complementary endpoints such as protein carbonyl quantification, volatile aldehydes (e.g., hexanal by GC–MS), and metal chelation assays to capture a more comprehensive picture of oxidative processes and confirm the dual lipid–protein protective effects of the extract.
In addition to the observed antioxidative and physicochemical benefits, future research should also include systematic microbiological assessments (total viable counts, psychrotrophs, Enterobacteriaceae, and yeasts/molds) under different storage and packaging systems (aerobic, vacuum, and MAP). This would provide a more comprehensive evaluation of chia seed extract’s contribution to both chemical stability and microbiological safety in extending the shelf-life of beef burgers.
It should be noted that polyphenols in Salvia hispanica extract may chelate transition metals, potentially influencing non-heme iron bioaccessibility. Although the concentrations applied in this study align with clean-label practices, future research should evaluate iron bioavailability and heme stability to ensure that antioxidant benefits are not accompanied by unintended nutritional trade-offs.
Within the tested inclusion levels, chia seed extract (CSE) proved technologically effective in enhancing oxidative stability, color retention, and textural attributes of beef burgers while maintaining sensory acceptability. These results support its compatibility with clean-label objectives, providing a natural alternative to synthetic antioxidants such as BHT and BHA. Nevertheless, broader considerations are needed to consolidate its application in commercial systems. Standardization of the extract, including marker identification and batch-to-batch reproducibility, will be essential for industrial reliability. Additionally, further studies should define the optimal balance between antioxidant efficacy and sensory thresholds, supported by consumer acceptance testing. Expanding oxidative stability assessments to include protein oxidation and volatile compounds, alongside systematic microbiological monitoring under different packaging conditions, will provide a more complete picture of shelf-life extension. Safety aspects, including potential allergenicity, antinutritional factors, and effects on iron bioavailability, must also be evaluated to ensure nutritional adequacy. Finally, pilot-scale studies across different meat matrices (e.g., poultry, pork) and fat levels will help validate scalability and generalisability. Overall, the present findings highlight CSE as a promising candidate for clean-label meat preservation, with future work focused on regulatory compliance and industrial feasibility to support broader adoption.
This study provides strong evidence that chia seed extract (CSE) can improve oxidative stability, physicochemical properties, and shelf-life of beef burgers under refrigerated storage. Nonetheless, several limitations and safety considerations should be acknowledged to guide future research and facilitate potential industrial applications [59, 60, 61].
First, while thiobarbituric acid reactive substances (TBARS) effectively capture secondary lipid oxidation, this endpoint alone may not fully describe the oxidative dynamics within meat systems. Complementary measurements, such as protein carbonyls, volatile aldehydes (e.g., hexanal) by GC–MS, and metal chelation assays, would strengthen mechanistic interpretation by confirming both lipid and protein stabilization. Incorporating such broader oxidative endpoints across different food matrices will better elucidate the multifunctional role of chia-derived phenolics.
Second, microbiological quality remains a critical determinant of refrigerated shelf-life. Although CSE delayed lipid oxidation, it may not directly influence microbial proliferation. Systematic monitoring of total viable counts, psychrotrophs, Enterobacteriaceae, and yeasts/molds under various packaging systems (aerobic, vacuum, and modified-atmosphere packaging) is essential to define the extent to which CSE contributes to safe shelf-life extension [62, 63].
Third, extract preparation and standardization warrant further refinement. In
this study, aqueous extraction was employed for food-processing feasibility;
however, phenolic profiling by HPLC–DAD provided only tentative identification.
Confirmation of marker compounds via LC–MS/MS, alongside reporting of
batch-to-batch variation and establishing specifications for total phenolics (mg GAE g–1) and key marker ranges, would improve reproducibility and industrial
scalability. Such standardization is essential for ensuring consistent
antioxidant efficacy in commercial applications. Dose optimization is another
important consideration. While the 1.5% inclusion level yielded the greatest
antioxidant benefit, practical application must balance functional efficacy with
potential sensory thresholds such as bitterness or astringency. Estimating
phenolic intake per serving and conducting larger-scale consumer acceptance
studies (n
Safety and allergenicity are further important aspects. Although chia is generally recognized as safe (GRAS) in several jurisdictions, rare allergic reactions to chia proteins have been reported, with possible cross-reactivity to other seeds. Moreover, water-soluble antinutrients such as oxalates, phytates, and tannins may be partially retained in aqueous extracts. Targeted determination of these compounds and estimation of their contribution per burger serving will help refine the safety profile of CSE.
Finally, regulatory and labelling frameworks vary internationally. Chia seed and its derivatives are approved for use in specific categories under “novel food” regulations in the European Union and as GRAS in the United States; however, broader applications in processed meat products may require additional safety assessment and formal approval. Transparent labelling, including potential allergen statements, will be essential to ensure consumer trust and regulatory compliance. In summary, while CSE demonstrates clear promise as a multifunctional, clean-label preservative for meat systems, future research should address these limitations by integrating advanced analytical endpoints, microbiological monitoring, standardized extract specifications, consumer sensory testing, nutritional assessments, and regulatory alignment. Such efforts will support the safe, scalable, and market-ready adoption of chia-based antioxidants in the food industry.
This study confirmed the efficacy of chia seed extract (CSE) in enhancing the oxidative stability, texture, and shelf-life of beef burgers; however, certain limitations should be acknowledged. The findings are based on a single beef burger formulation under aerobic refrigerated storage, which may not fully capture the variability of commercial meat systems. Replication across species (poultry, pork), fat levels, and packaging types (vacuum, MAP), along with pilot-scale trials, is necessary to validate generalisability and industrial feasibility. From a regulatory perspective, chia-derived ingredients are classified as “novel foods” in some regions, with varying approval and labelling requirements, including allergen declarations due to rare cases of cross-reactivity. Standardization of extract composition and further toxicological validation will be essential to support regulatory acceptance. Addressing these aspects will help position CSE as a safe, effective, and clean-label antioxidant for commercial meat preservation.
Incorporation of chia seed (Salvia hispanica L.) extract significantly enhanced the physicochemical quality, oxidative stability, and shelf-life of beef burgers during refrigerated storage. The 1.5% extract level (T3) was most effective, reducing lipid oxidation, improving texture, and maintaining desirable color and moisture. These effects are attributed to the synergistic action of phenolic acids, flavonoids, and omega-3 fatty acids present in the extract. The findings demonstrate chia seed extract’s potential as a clean-label, natural alternative to synthetic antioxidants such as BHT and BHA, supporting its use in the development of functional and sustainable meat products.
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
EHA—Conceptualization, methodology, formal analysis, investigation, data curation, writing – original draft, and review & editing. The author read and approved the final manuscript. The author has participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
The authors would like to acknowledge the Deanship of Graduate Studies and Scientific Research, Taif University, for funding this work.
This research received no external funding.
The author declares no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



