1 Institute of Animal Reproduction and Food Research, Polish Academy of Sciences, 10-683 Olsztyn, Poland
The activity of phenolic compounds makes this group of molecules very important components in plant materials and plant-derived foods. Phenolic compounds constitute a class of chemical compounds with a highly diverse structure, which are characterized by the presence of benzene rings with hydroxy moieties attached.
Among known phenolic compounds, condensed tannins (proanthocyanidins, namely PACs) are particularly important and relevant to food technologists and chemists. From a chemical standpoint, PACs are biopolymers with a flavan-3-ol backbone structure (Fig. 1). PACs represent a vast family of polymers with varying chain lengths and linkage types, which contribute to the associated diverse properties. Under specific conditions (e.g., alcohol solutions of organic acids and high temperature), anthocyanidins and catechins are released from the PACs as terminal end groups [1].
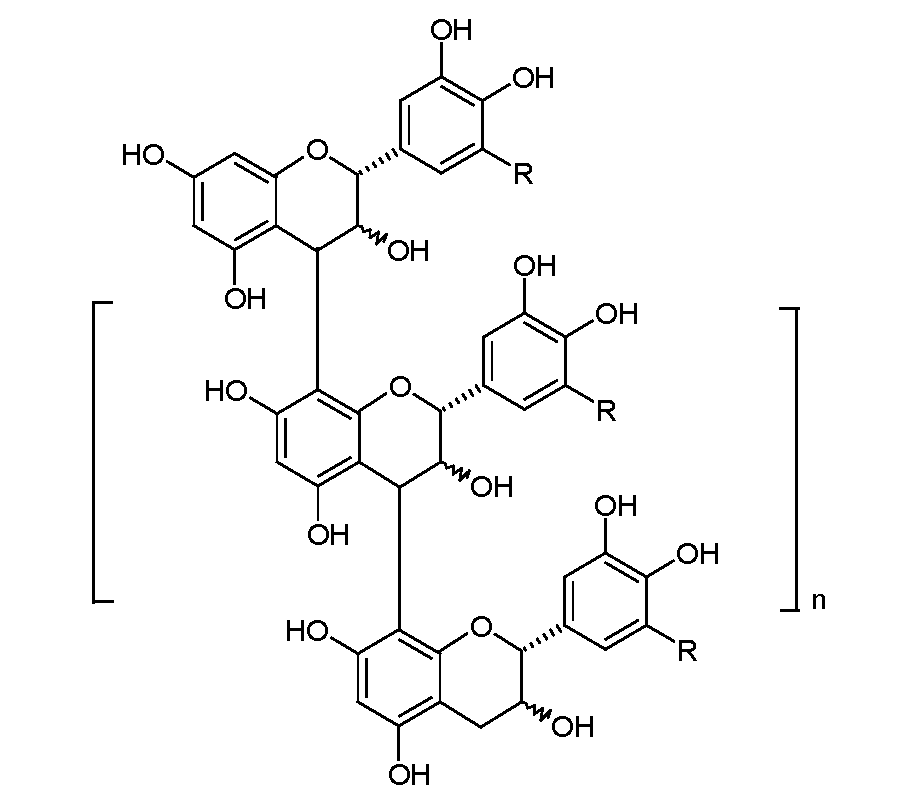 Fig. 1.
Fig. 1.
Chemical structure of condensed tannins.
Many pharmaceutical agents exhibit pharmacological properties that enable various applications. Nonetheless, this editorial focuses on the reduction in reactive oxygen species (ROS) and on the antimicrobial, antifungal, anti-obesity, anti-diabetes, nephroprotective, and antiviral activities of PACs. Moreover, PACs have shown potential to positively inhibit or amend the onset of Alzheimer’s disease, certain cancers, and aging [2].
For food consumers, the antibacterial activity of PACs is very important. PACs have shown bacteriostatic activity against a broad spectrum of microorganisms, including significant foodborne pathogens such as Listeria monocytogenes, Salmonella typhimurium, and Escherichia coli O157:H7, as well as spoilage bacteria such as Brochothrix thermosphacta [1].
Several mechanisms have been identified to explain the antibacterial activity of PACs. These include interactions between PACs and bacterial structures, as well as interference with bacterial cellular systems by binding to certain enzymes and cell wall structures. Consequently, the phenomena mentioned above can inhibit bacterial proliferation and development [3].
Since these compounds can donate hydrogen atoms or electrons to radical species,
PACs are considered primary antioxidants [4, 5]. Furthermore, PACs can act as
secondary antioxidants via chelation. The chelation of the pro-oxidant Fe(II) in
foods and the inhibition of cyclooxygenases by PACs have been reported [5, 6].
Notably, the tannin-rich fraction from leguminous seed extracts exhibited
stronger antioxidant activity in the reducing power assay, as measured by
DPPH• and ABTS•+ radical scavenging, and in the
As protease inhibitors, PACs can reduce the biological value of plant proteins. Therefore, in food chemistry books, PACs have been classified as antinutrients. The presence of PACs in plant-derived foods is responsible for astringency, which manifests as dryness, tightening, and puckering sensations in the oral cavity [7]. Astringency is caused by the interaction of tannins with proline-rich proteins from salivary glands [8, 9]. Other pharmacological activities of PACs have been attributed to their ability to chelate metal ions, thereby lowering metal ion toxicity in food by reducing PAC accumulation [2]. It is worth emphasizing that the chelation of iron ions actually reduces the availability of this element from the diet, while also having a positive effect by inhibiting the process of lipid autoxidation in the Fenton reaction:
Fe2+ + H2O2
Fe3+ + H2O2
Due to the adverse effects of PACs, regulating PAC content in plant-derived foods is sometimes necessary. A reduction in tannin content in plants can be achieved through heating (boiling, autoclaving, extrusion) and germination. Several authors have reported reductions in PAC levels during domestic processing (e.g., soaking and cooking) [1].
To a degree, the astringency of PACs can be masked by the addition of food gums (e.g., guar, xanthan gum, Arabic) and carboxymethylcellulose. This has been confirmed for tannin-rich extracts from black chokeberry fruit, green tea leaves, and walnut nutmeat [10]. The reason for masking the astringency is the competition between polysaccharides and tannins for binding salivary proteins and the formation of ternary complexes [11]. Future sensory studies on PACs should also consider interactions between PACs and other food matrix components (proteins, lipids, other polysaccharides) to develop functional foods with optimized sensory profiles.
Considering both the positive and negative aspects of PACs, food technologists must control PAC content in food to preserve the associated health-promoting properties while avoiding impairing the sensory properties of the product or the biological value of the contained protein. For food technologists, the current challenge is applying novel technologies such as encapsulation, enzymatic modification, and precision fermentation to tailor PAC functionality. The current challenge for food chemists is to investigate the specific structure–function relationships of PACs. It is important, from a basic and practical point of view, to know how the degree of PACs polymerization or the type of interflavan linkage (e.g., A-type vs. B-type) affects both bioactivity and astringency.
RA made substantial contributions to the conception and design of the work, drafted the manuscript, read and gave final approval of the version to be published, and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
Given his role as an Editorial Board member, Ryszard Amarowicz had no involvement in the peer-review of this article and has no access to information regarding its peer-review. Full responsibility for the editorial process for this article was delegated to Corinna Kehrenberg. The author declares no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

