1 Beijing Key Laboratory of Printing & Packaging Materials and Technology, Beijing Institute of Graphic Communication, 102600 Beijing, China
2 College of Food Science and Technology, Shanghai Ocean University, 201306 Shanghai, China
Abstract
Strawberries is a fruit rich in various nutrients. Their thin skin structure and high respiratory activity make them susceptible to microbial contamination and rapid spoilage. Traditional preservation techniques still have limitations. Therefore, this study aims to investigate the effect of plasma-processed air (PPA) on strawberry preservation.
The efficacy of plasma-processed air generated via sliding arc discharge, with treatment times of 15 s, 30 s, and 60 s, was compared to untreated controls in terms of microbial suppression and quality preservation of fresh strawberries during cold storage.
PPA treatment significantly reduced surface microbial colonies, with a 60 s exposure achieving the lowest colony count (1.18 log colony-forming units (CFU)/g), representing approximately a 95% reduction compared to control (2.49 log CFU/g). While PPA exposure maintained nutritional parameters (total soluble solids (TSS), titratable acid (TA), pH, ascorbic acid (ASA)) at levels not significantly different from those of the untreated fruit (p > 0.05), PPA exposure effectively mitigated quality deterioration. PPA-treated strawberries were firmer, showed better color retention and less weight loss, maintained cell membrane integrity, and had lower ethylene production (p < 0.05). Notably, shelf-life extension was dose-dependent, reaching 15, 18, and 16 days at 4 °C for the treated strawberries (15 s, 30 s, and 60 s treatments, respectively) compared to 9 days for the untreated control fruit.
PPA can effectively extend strawberry shelf-life by maintaining fruit quality and inactivating microorganisms. Furthermore, this continuous industrialized green treatment technology could ultimately be used as a standard and applied linearly during the cold chain transportation of fresh produce.
Keywords
- plasma-activated gas treatment
- strawberry preservation
- shelf-life extension
- colony inactivation
Strawberry is a highly perishable fruit prized for their distinctive aroma, polyphenol-rich composition, and proven health benefits in chronic disease prevention [1, 2, 3]. However, their thin epidermal structure and high respiratory activity render them vulnerable to mechanical injury, microbial colonization (particularly Botrytis cinerea), and rapid postharvest deterioration [4]. While conventional preservation methods including thermal processing [5], edible coatings [6], and ozone treatment demonstrate partial efficacy [7], practical limitations persist regarding chemical residues, energy consumption, and operational complexity.
Emerging as a breakthrough in non-thermal food processing, low-temperature plasma technology has revolutionized food industry through its unique combination of microbial inactivation and quality preservation mechanisms and has been widely applied across multiple areas [8]. This technology generates reactive oxygen/nitrogen species (ROS/RNS), ultraviolet photons, and charged particles through gas ionization, which synergistically disrupt microbial cellular components while maintaining the bioactive compounds of the fruit to achieve a bactericidal effect [9, 10, 11]. Notably, plasma-activated water (PAW) applications can extend shelf-life of various produce including apple [12] and blueberry [13], yet this technology faces some challenges due to real-time production costs and inconsistent sterilization efficacy [14].
The present study introduces an innovative: plasma-processed air (PPA) technology under ambient instead of inert gases atmosphere. This approach significantly reduces operational costs while enabling continuous processing which brings a critical advantage for commercial scalability. Building upon recent advancements in dielectric barrier discharge systems, our investigation focuses on PPA’s dual capacity to suppress microbial proliferation (particularly total viable counts and Botrytis infection) and modulate physiological responses in stored strawberries. Through comprehensive evaluation of physicochemical parameters (texture, color stability, weight loss), biochemical markers (ethylene biosynthesis, membrane permeability), and nutritional retention (ascorbic acid), this work addresses a critical research gap in plasma application protocols for soft fruits. The findings hold significant implications for optimizing postharvest strategies, particularly in addressing the 20–35% annual loss of strawberry production attributed to inadequate preservation methods.
The implementation of active gas sterilization technology, characterized by short contact time and no chemical residues, without the use of any chemical agents, combined with modified atmosphere packaging (MAP) immediately after sterilization, eliminates the risk of secondary contamination and enables continuous online processing. This avoids the drawbacks of other non-thermal disinfection methods, such as the potential health hazards of ultraviolet (UV) disinfection and the limitations of pulsed electromagnetic field technology due to medium restrictions. The research apparatus uses electricity as the sole consumable energy source, with air as the medium, eliminating other energy consumption and significantly reducing costs. By establishing correlations between plasma treatment parameters and fruit quality indicators, our study provides actionable insights for implementing PPA technology in commercial cold chain systems.
Strawberry cv. Dandong (n = 576 fruit), were purchased from Beijing Agricultural Products Market, Beijing, China; LB Nutrient Agar, Beijing Aoboxing Biotechnology Co., Ltd., Beijing, China; Sodium Chloride (analytically pure), Phenolphthalein (98% mass fraction), Shanghai Aladdin Biochemical Science and Technology Co., Ltd., Shanghai, China; Sodium hydroxide standard solution, Shandong Keyuan Biochemistry Co., Ltd., Heze, Shandong, China; Potassium iodide (analytically pure), Tianjin Juhengda Chemical Co., Ltd., Tianjin, China; soluble starch (analytically pure), Tianjin Fuchen Chemical Reagent Co., Ltd., Tianjin, China.
The experiment employed the apparatus shown in Fig. 1. PPA was generated by an arc discharge system (Lab-V1 plasma preservation device), which ignited an air flow between two-point electrodes (spacing: 3 cm) via a 20–40 kV high-voltage electric field to form a plasma discharge. Using air as the gas source, the cold plasma device was activated to produce atmospheric pressure cold plasma (CAPP). The generated CAPP was introduced into a sealed chamber via a vacuum circulation pump (flow rate: 40 L/min), thereby producing plasma-activated gas (PPA). The discharge power was set at 220 W, with the high-voltage power supply operating at a center frequency of 50 kHz and a discharge volume of 30 dm3. Strawberries were processed in batches within the chamber for treatment durations of 15 seconds, 30 seconds, and 60 seconds. After preheating is complete, sample processing begins. Energy consumption during the experiment is the product of the equipment power and the processing time.
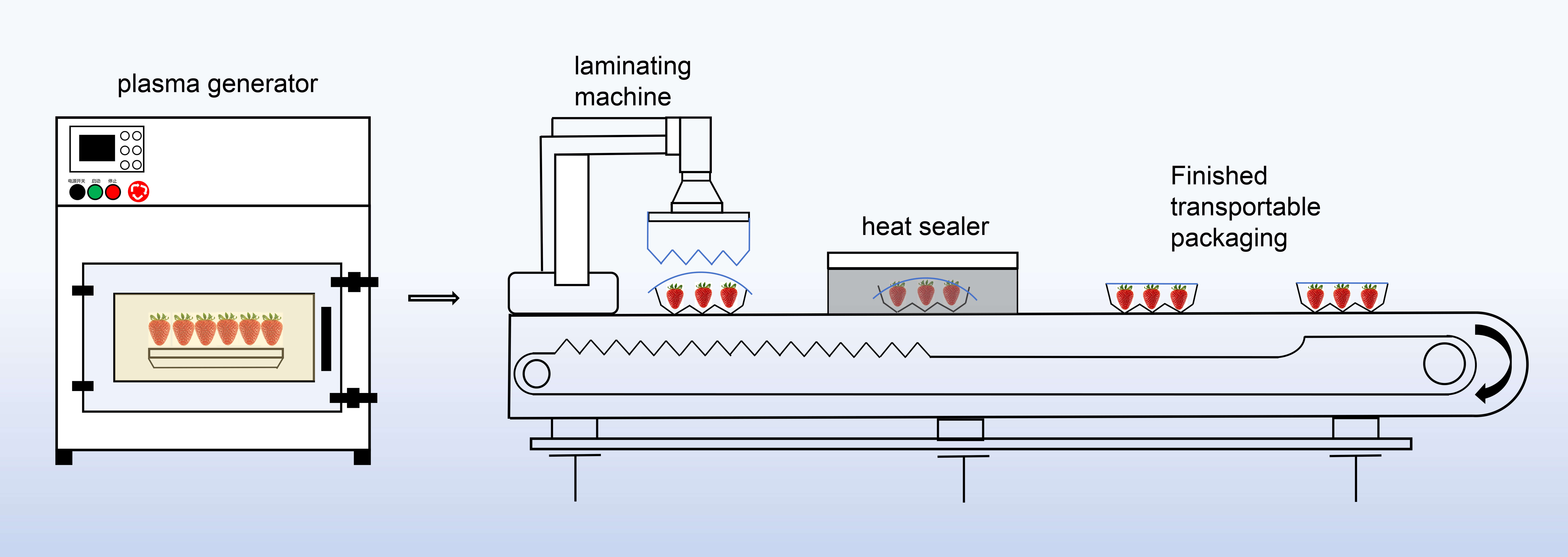 Fig. 1.
Fig. 1.
Atmospheric plasma equipment and flow chart used for processing strawberries.
Uniform strawberry samples with consistent size, color, and weight, and free of mechanical damage were selected. Remove the pedicel and place the fruit in a refrigerated device (model: YC-300L) at a constant temperature of 4 °C for 4–6 hours to achieve thermal equilibrium.
Strawberries were randomly assigned to three experimental treated and one
untreated experimental groups, PPA15, PPA30, PPA60 and CK, respectively (15 s PPA
treatment = PPA15; 30 s PPA treatment = PPA30; 60 s PPA treatment = PPA60;
untreated control = CK). Samples were packed in polypropylene trays (Model:
TQBC-0775; dimensions: 222
Fig. 1 shows the atmospheric plasma processing equipment, comprised by a plasma
generator, a conveyor belt, a laminated machine, and a heat sealer. After the air
was broken down in the discharge ceramic tube, plasma active substances were
generated, and used to treat the strawberries in the chamber. Each batch of
strawberries (6 fruit each) was processed at a time. After processing, all
samples were immediately transferred to the conveyor belt and heat sealed with
food-grade polyethylene (PE) film to complete the entire sterilization and
packaging process, and stored in a crisper under controlled temperature
conditions (4 °C
The Check point headspace MAP gas analyser (Shanghai AMETEK Trading Co., Ltd., Shanghai, China) was used to measure the levels of oxygen and carbon dioxide in the packages. The air indicator was calibrated and a sealed adhesive strip was attached to the top, middle and bottom of each package. One sample was taken from each strip and the average of the three data points was taken and the final value calculated.
The ethylene content (ppm) of the packages was measured using a gas chromatograph (Model GC7860, Shanghai Youke Instrumentation Co., Ltd., Shanghai, China), with samples taken from the top, middle and bottom positions of each package, and the average of the three data points was calculated as the final value.
The relative conductivity method was used to measure cell membrane permeability. Damage to the cell membrane under abiotic stress leads to electrolyte leakage, and changes in membrane permeability are quantified by measuring changes in solution conductivity.
Strawberry tissue (2 g) was mixed with 20 mL deionized water inside a beaker. Then, the strawberry-deionized water mixture was let to rest at constant room temperature for 1 h to determine the initial conductivity (L0) measured using a conductivity meter (AZ-8362, Hengxin Industrial Co., Ltd., Taichung, China). Afterward, the beaker containing the mixture was transferred to a water bath at 100 °C for 15 min. After that, it was left to cool at room temperature and conductivity measured again (L1). Relative conductivity was calculated using the following formula:
Based on the weight changes during storage, the weight loss rate a calculated using the weighing method described by Wang et al. (2019) [15]. The weight was measured using an electronic balance (model: MTB5000D, Shenzhen Meifu Electronics Co., Ltd., Shenzhen, Guangdong, China), and the calculated formula was as follows:
A G-type fruit hardness tester (Yueqing Aidebao Technology Co., Ltd., Wenzhou, Zhejiang, China) was used to test the hardness of strawberries according to the method described by Yang et al. (2023) [16]. After zeroing the device, the center of the strawberry was selected, and the reading was taken when the penetration depth reached the instrument scale (unit: kg/cm). The average hardness of six strawberries in each tray was taken as the final result for each treatment.
Using a DS-200 colour difference meter (Zhejiang Caipu Technology Co., Ltd., Hangzhou, Zhejiang, China), the CIE Lab method was used [17]. After calibrating the instrument, each group (6 strawberries) was sampled and tested. The sampling location was the equatorial part of the strawberry. Each strawberry was sampled three times at equidistant positions, and the L*, a*, and b* values were measured separately. The average value of each group of strawberries was taken as the final value. The formula for calculating color difference is as follows:
where
Mash the strawberries and filter them. The total soluble solids content was
measured using a PAL-
The titratable acid content was determined using the sodium hydroxide solution titration method [19]. Based on the acid-base neutralization reaction, the acidic substances in the sample were titrated with a standard sodium hydroxide solution, and the endpoint is determined by the color change of the indicator. The total acid content was calculated based on the volume of sodium hydroxide consumed.
The formula for calculating the titratable acid contents as follows (units: %):
Potassium iodate titration was used to determine the ascorbic acid content in
strawberries [19]. After grinding the strawberry juice, take 1.0 g of juice,
recorded as
The pH value of strawberry juice was measured using a FE28 pH (Mettler-Toledo Technology Co., Ltd., Shanghai, China) meter. The average value of three repeated measurements was used.
Total colony counts on strawberry surfaces were determined according to GB
4789.2-2022. A 10 g fruit sample was homogenized using sterile physiological
saline. Dilutions were prepared at different concentrations (–1 to –3). One
milliliter of each dilution was transferred to plate count agar (PCA) medium
cooled to 46 °C and mixed. The mixtures were cooled to room temperature
under a laminar flow hood until the agar solidified. The plates were incubated at
36 °C
Unpackaged strawberries were assessed by observing signs of decay on the surface of the fruit, characterized by softening of the injured area and the appearance of brown spots [20], and the total amount of decay was recorded. The decay rate was calculated using the following formula:
Following the method described by Fu et al. (2024) [21], each group of strawberries was evaluated for color, aroma, softness, and appearance-related factors (Table 1). Under constant environmental conditions (temperature 21 °C, relative humidity 60%, daylight, air circulation), three independently conducted assessments were performed by three uniformly trained evaluators. The final score was determined as the average of all individual scores.
| Value of a score | Colour | Fruit morphology | Odour |
| 9 |
Bright red, shiny | No deformation | Intense fruity aroma |
| 6 |
Dark red, slightly dull | Slight atrophy | Weak aroma and no odour |
| 3 |
Fuchsia, dull | Significantly shrivelled | No detectable aroma |
| 0 |
Brown, extremely dull | Extremely shrivelled mould | Off-odors present |
The data collected was analyzed by one-way ANOVA to determine the significant
differences between treatments (SPSS 20.0, IBM, New York, USA). The least significant
difference a p
Table 2 shows quality attributes of PPA-treated and untreated strawberries.
| Parameter of analysis | Type of processing | |||
| CK | PPA15 | PPA30 | PPA60 | |
| Weight loss (%) | 0.000 |
0.024 |
0.021 |
0.020 |
| L* | 31.27 |
31.25 |
31.39 |
31.07 |
| a* | 35.65 |
36.28 |
35.45 |
35.22 |
| Firmness (N) | 1.48 |
1.47 |
1.43 |
1.49 |
| TSS (%) | 11.17 |
11.23 |
11.60 |
11.42 |
| TA (%) | 0.80 |
0.77 |
0.83 |
0.84 |
| ASA (%) | 37.36 |
35.97 |
39.54 |
37.61 |
| pH | 3.44 |
3.44 |
3.45 |
3.43 |
| O2 (%) | 19.87 |
19.88 |
19.87 |
19.88 |
| CO2 (%) | 0.00 |
0.00 |
0.00 |
0.00 |
| Colony number (log CFU/g) | 2.49 |
2.04 |
1.47 |
1.18 |
*Different superscript letters (a, b, c and d) represent
significant differences in the data (same row); the same letter is a
non-significant difference (p
For the weight loss of strawberries at moment zero, the treatment group showed a slight reduction of less than 0.1% compared to the control sample, indicating that the PPA had a negligible effect on this quality parameter. The results of this study are in agreement with the results of Rana et al., 2020 [22], who studied whole fresh strawberries using in-package air discharges to generate plasma, as well as Giannoglou et al., 2021 [23] who showed that plasma treatment of strawberries did not significantly change their weight loss percentage score.
The color (L* and a*) of the PPA-treated strawberries was retained, and hardness
showed only minor changes, none of which were significant (p
It is worth noting that, compared with the control group, the total number of
bacterial colonies on strawberries significantly decreased after PPA treatment
(p
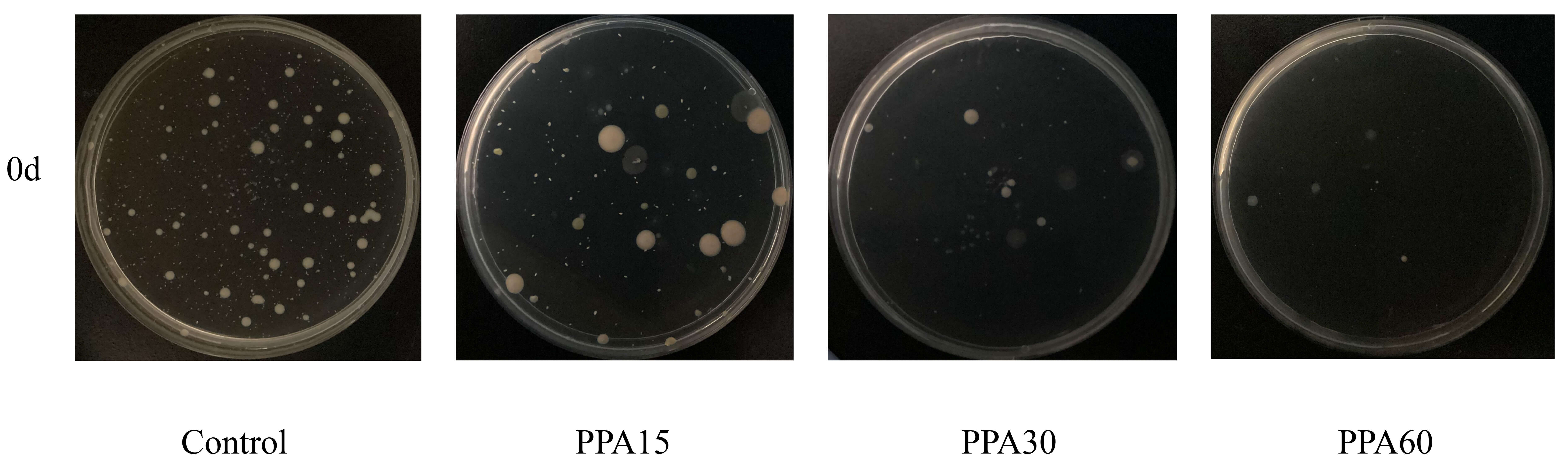 Fig. 2.
Fig. 2.
Number of strawberry surface colonies under different treatment conditions (before preservation).
As expected, the reactive oxygen and nitrogen species generated by surface discharge diffuse into the fruit, exerting a sterilizing effect on the strawberries and reducing the microbial content on their surfaces. The sterilization efficacy of PPA treatment increases with longer treatment times, as the active components within PPA come into more prolonged and thorough contact with the strawberry surfaces. This confirms that PPA, after diffusing to the surface of strawberries in the treatment chamber, can effectively inactivate microorganisms on the surface of strawberries [25], and its inactivation effect is proportional to the treatment time.
Moisture loss in strawberries, resulting from ripening, respiration, and
transpiration processes, leads to diminished surface gloss, wilting, and
shriveling. Weight loss rate serves as a critical indicator for evaluating the
efficacy of postharvest preservation treatments [26]. The weight loss rate
exhibited a progressive increase with prolonged storage duration (Fig. 3).
One-way ANOVA revealed statistically significant differences (p
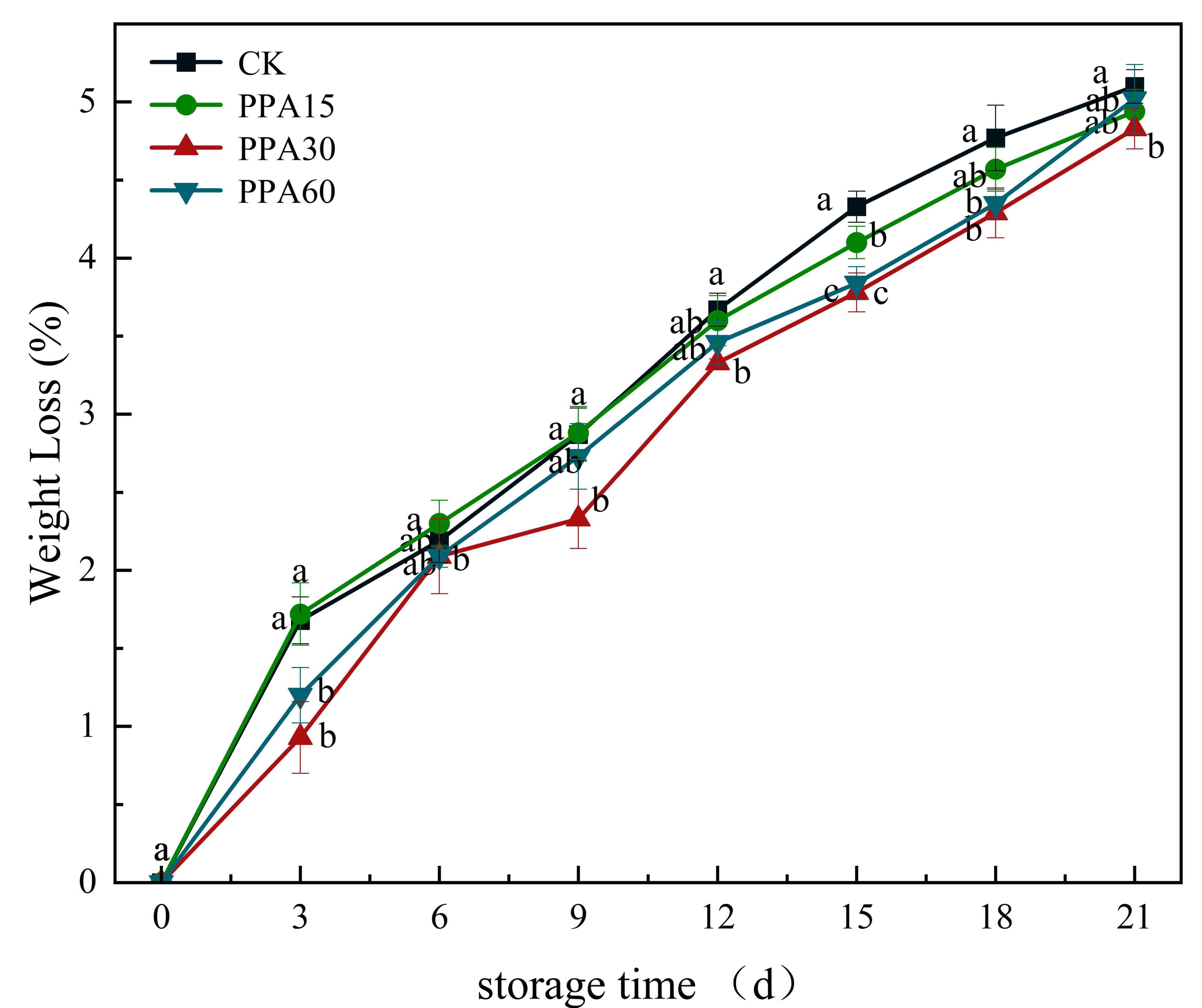 Fig. 3.
Fig. 3.
Effects of different PPA treatments on weight loss of
strawberries during cold storage. CK, control; PPA15, PPA treatment for 15 s;
PPA30, PPA treatment for 30 s; PPA60, PPA treatment for 60 s. Different letters
indicate significant differences (p
Firmness represents a critical quality attribute in strawberries, serving as a key indicator of both ripeness and postharvest storage quality [27]. All treatments exhibited a biphasic firmness pattern during storage, characterized by initial increase followed by gradual decline (Fig. 4). This phenomenon may be attributed to incomplete maturity at harvest, with subsequent postharvest ripening during storage, accompanied by progressive strengthening of cell wall integrity and pectin cross-linking [28], as well as moisture loss-induced tissue densification leading to increased firmness. Prolonged storage resulted in significant softening, likely due to enzymatic degradation of cell wall components, and based on the morphological characteristics of the decayed area (such as softening of the flesh) and the extent of decay, it can be inferred that this may be related to microbial activity.
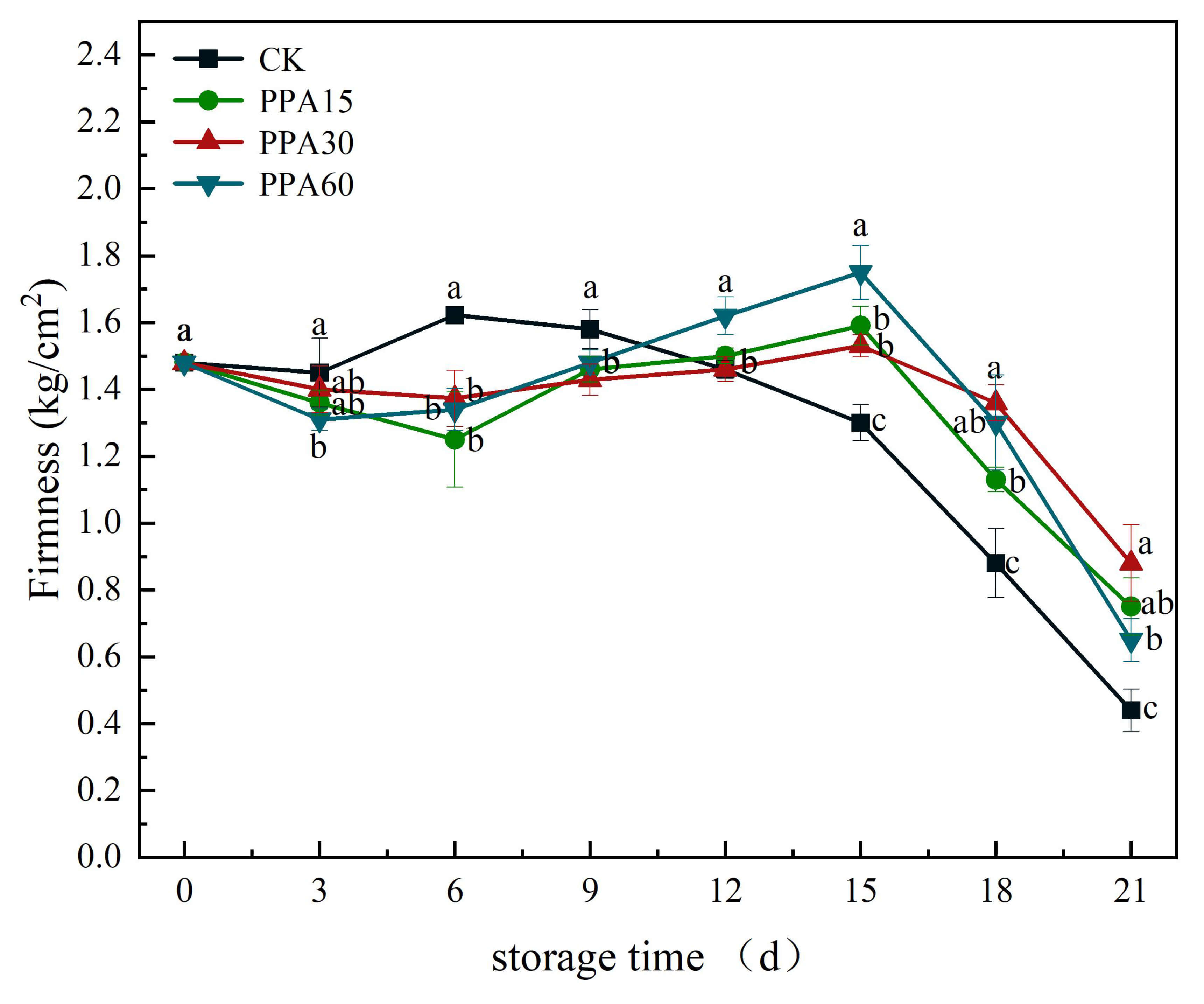 Fig. 4.
Fig. 4.
Effects of different PPA treatments on firmness of
strawberries during cold storage. CK, control; PPA15, PPA treatment for 15 s;
PPA30, PPA treatment for 30 s; PPA60, PPA treatment for 60 s. Different letters
indicate significant differences (p
PPA-treated strawberries showed the highest stability in firmness (p
The color parameters L*, a*, b*, and
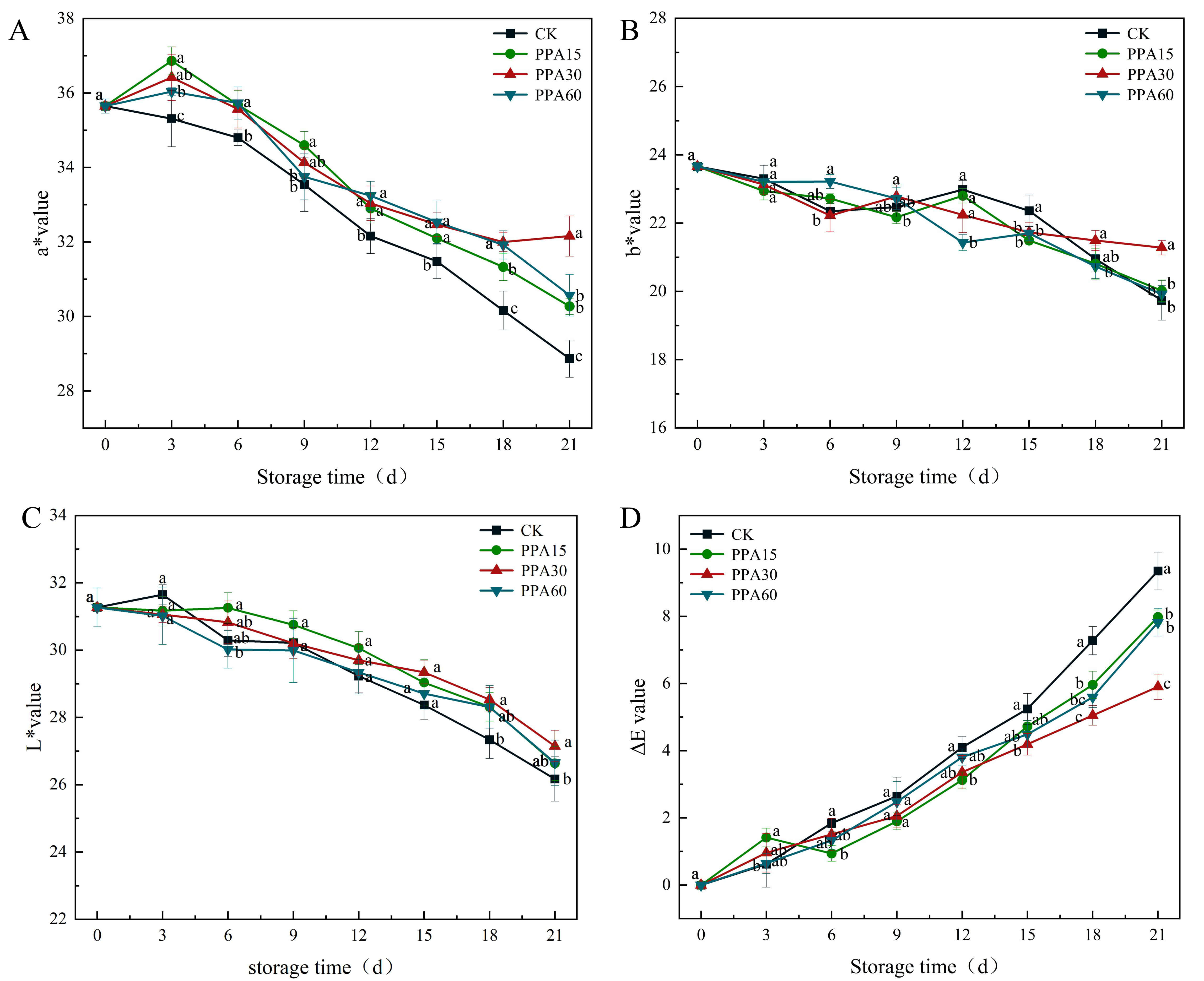 Fig. 5.
Fig. 5.
Effect of PPA treatments on a* (A), b* (B), L* (C) and
Total soluble solids (TSS) content, a key biochemical indicator for assessing strawberry sugar content [31], exhibited a continuous decline in both control and treated fruits during storage (Fig. 6A), with no significant difference observed between treatments and control throughout. This reduction can be attributed to the combined effects of respiratory metabolism and microbial activity [25], where soluble carbohydrates are metabolized to sustain cellular functions while microbial proliferation concurrently consumes available nutrients. Notably, the PPA30 treatment demonstrated superior efficacy in TSS preservation, suggesting an optimal treatment for maintaining fruit quality within the tested concentration range. These findings corroborate previous reports on low-temperature plasma applications in strawberry preservation [25], supporting the potential of plasma-based technologies in postharvest management.
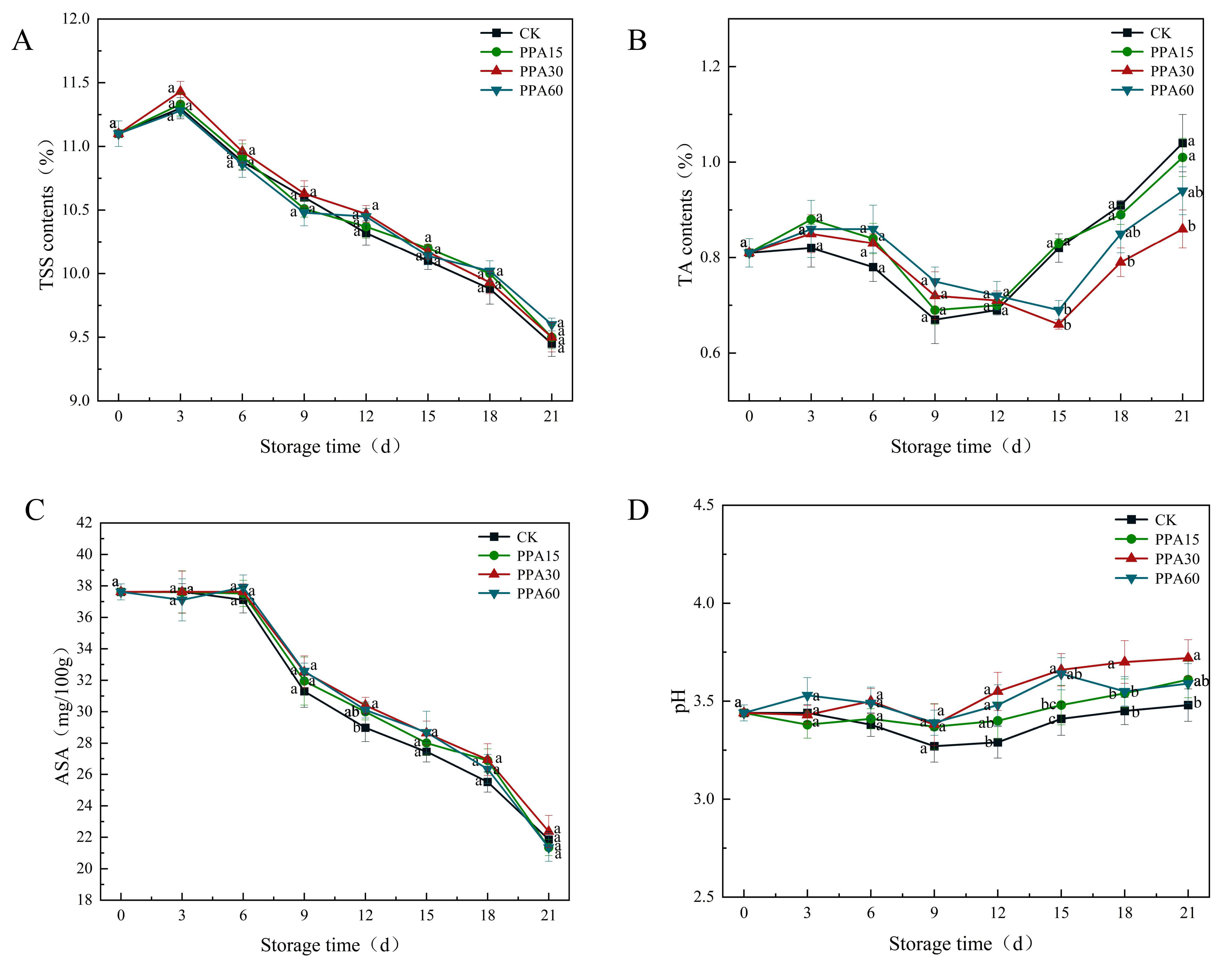 Fig. 6.
Fig. 6.
Effect of different PPA treatments on TSS (A), TA (B), ASA (C)
and pH (D) of strawberries during cold storage. CK, control; PPA15, PPA
treatment for 15 s; PPA30, PPA treatment for 30 s; PPA60, PPA treatment for 60 s.
Different letters indicate significant differences (p
The titratable acidity (TA) content, a key biochemical indicator of sugar-acid
balance and sensory quality in strawberries, exhibited a characteristic biphasic
pattern during storage (Fig. 6B), with an initial decline (0–12 days) followed
by a subsequent increase. This dynamic profile primarily reflects metabolic
regulation, particularly through respiratory pathways [32]. The initial TA
decrease corresponds to normal physiological maturation, where organic acids are
preferentially metabolized as respiratory substrates. PPA treatment had limited
impact on TA dynamics, with no statistically significant differences observed
between the 0–12 day treatment groups and the control group (p
The antioxidant properties of ASA, a water-soluble antioxidant with strong
reducing properties, helps reducing oxidative damage while enhancing fruit
resistance to abiotic and biotic stresses [26]. Our results showed a progressive
decline in ASA content during storage across all treatments (Fig. 6C),
attributable to the combined effects of exogenous oxidative stress, endogenous
respiratory metabolism, and microbial growth. However, no significant differences
in ASA content were observed between treated and control strawberries throughout
the entire storage period (0–21 days) (p
The pH value can affect the balance of organic acids and sugars to maintaining
the unique sweet and sour flavour of strawberries, and too high or too low of a
value may lead to an imbalance in flavour, which has an important effect on
storage quality of fruits and vegetables [33]. Results from our study showed
constrained pH fluctuations within a relatively narrow range throughout storage
(Fig. 6D), with the initial phase showing minimal divergence between treated and
control fruit. However, prolonged storage resulted in systematically higher pH
values in PPA-treated samples, potentially mediated through suppressed microbial
proliferation and consequent reduction in acid generation on fruit surfaces,
though statistical analysis indicated these differences were not significant
(p
Postharvest respiratory metabolism in strawberries, a critical physiological
process for maintaining cellular functions [30], was significantly modulated by
PPA treatment as evidenced by characteristic gas composition changes showing
progressive O2 depletion and CO2 accumulation (Fig. 7). These results
were consistent with established respiratory patterns in strawberry fruit [28].
Comparative gas analysis revealed significantly higher O2 and lower CO2levels in PPA-treated samples compared to control (p
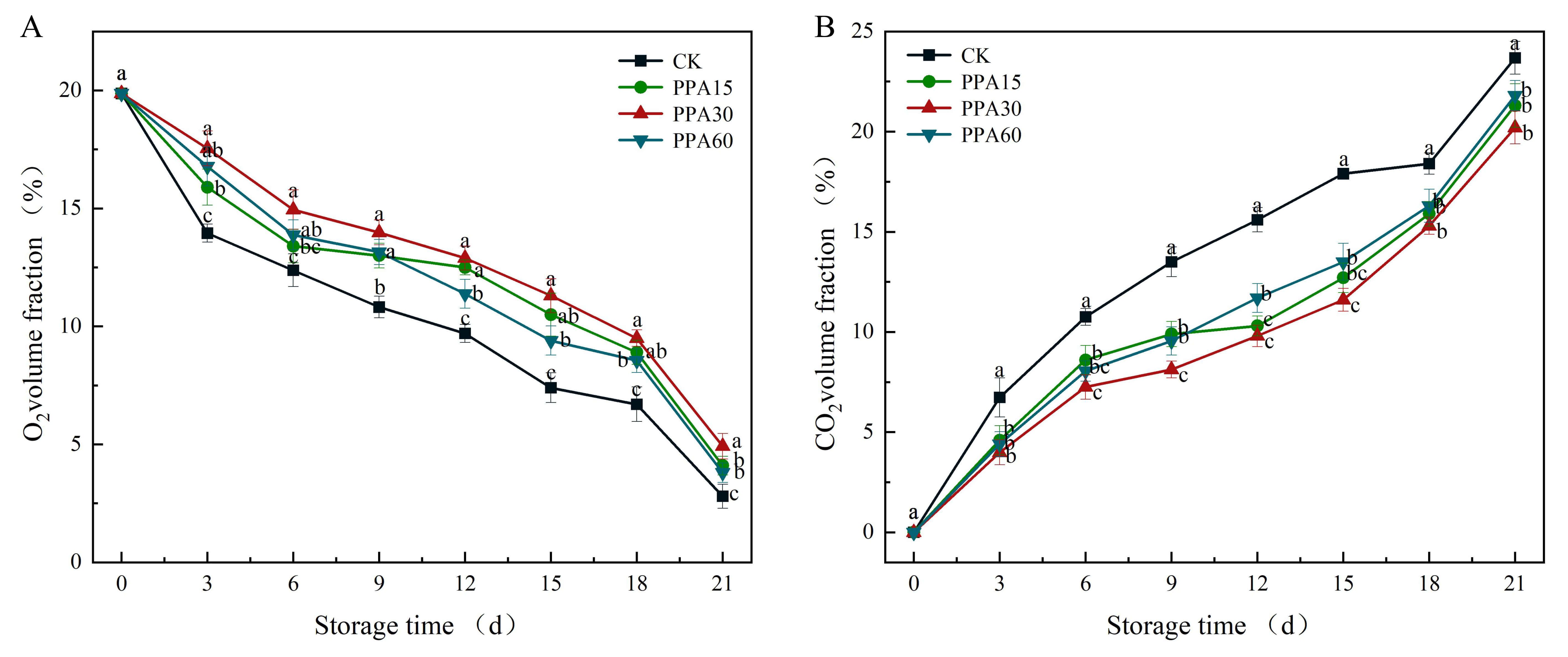 Fig. 7.
Fig. 7.
Effect of different PPA treatments on the O2 (A) and
CO2 (B) levels inside packages of strawberries during cold storage. CK,
control; PPA15, PPA treatment for 15 s; PPA30, PPA treatment for 30 s; PPA60, PPA
treatment for 60 s. Different letters indicate significant differences
(p
Ethylene, is a phytohormone that promotes the respiration of fruits and
vegetables, and accelerates ripening and senescence, affecting the quality of
fruits during storage [28]. During the storage period, the ethylene content of
strawberries showed an increasing trend, regardless of the treatment (Fig. 8A).
The PPA15 and PPA30 treated strawberries showed significantly lower (p
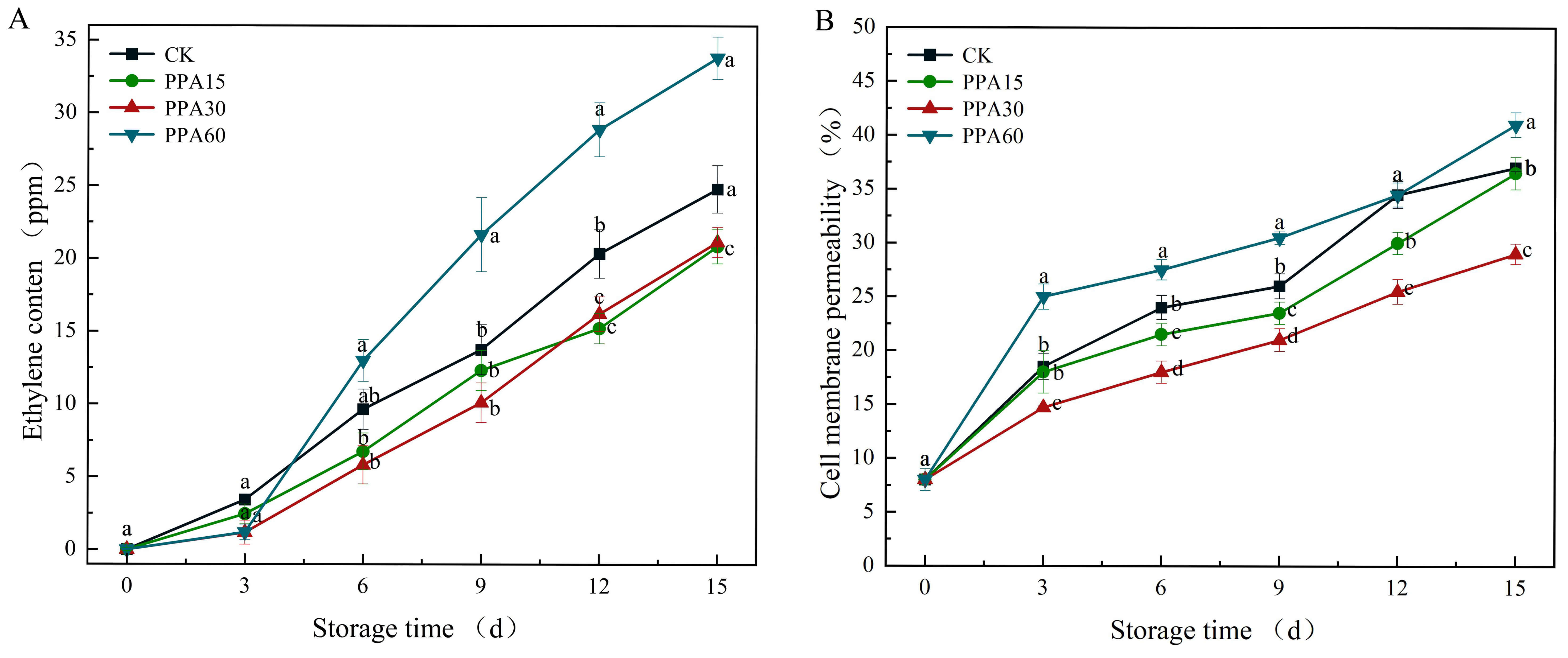 Fig. 8.
Fig. 8.
Effect of different PPA treatments on ethylene production (A)
and cell membrane permeability (B) of strawberries during cold storage. CK,
control; PPA15, PPA treatment for 15 s; PPA30, PPA treatment for 30 s; PPA60, PPA
treatment for 60 s. Different letters indicate significant differences
(p
Cell membrane permeability, which serves as a critical indicator of cellular
integrity and senescence progression in fruit, exhibited a characteristic
increase during storage (Fig. 8B), reflecting oxidative damage [35]. The
integrity of the cell wall provides a stable environment for the selective
permeability of the cell membrane. Therefore, our results suggest that the
integrity of the cell wall of strawberries was disrupted during storage.
Statistical analysis showed significant differences (p
The microbial load on strawberry surfaces, is a critical food safety concern and
primary contributor to postharvest decay [37]. Botrytis cinerea, also
known as gray mold, one of the primary fungus causing strawberry spoilage, and
responsible for most of postharvest losses. In this study, the number of colonies
on the surface of strawberries showed a gradual increase during storage
regardless of the treatment (Fig. 9). However, a significant growth suppressio
was observed in PPA-treated fruit compared to the control (p
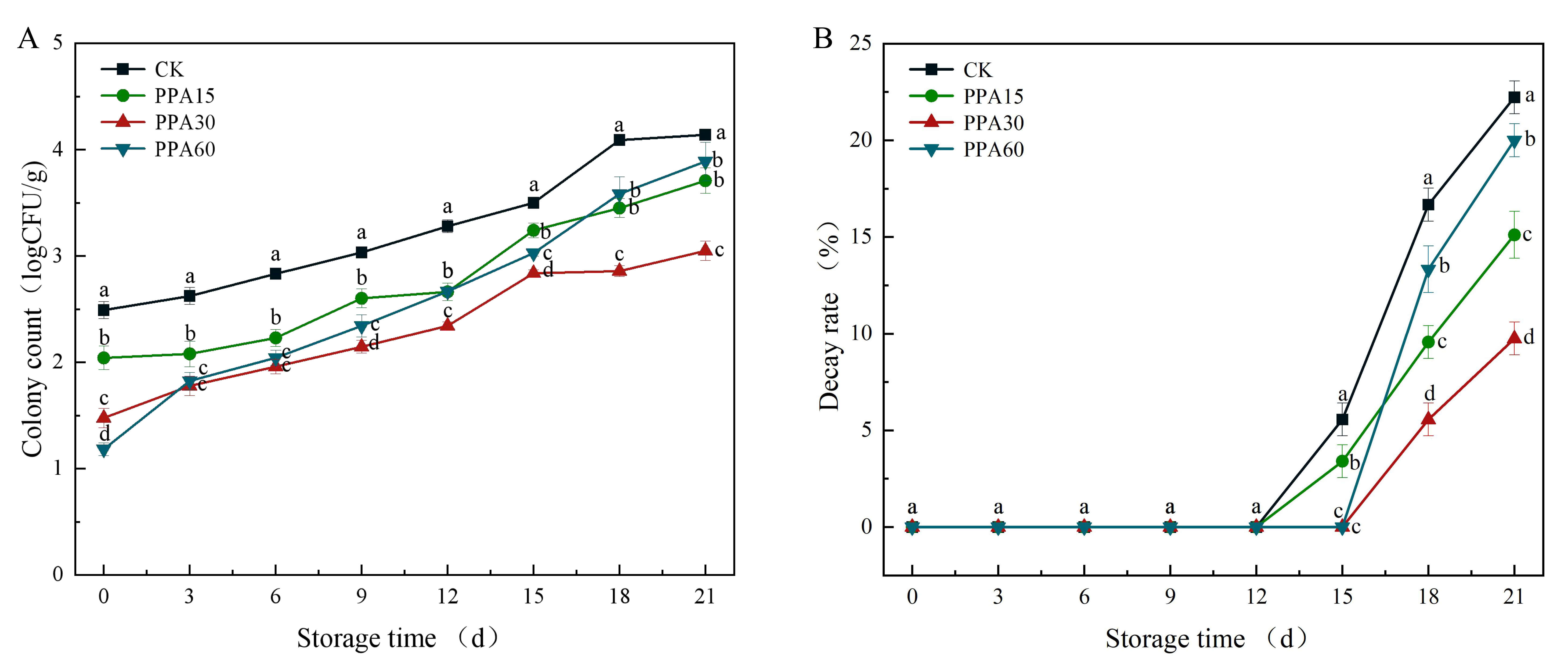 Fig. 9.
Fig. 9.
Effect of different PPA treatments on colony count (A) and decay
rate (B) of strawberries during cold storage. CK, control; PPA15, PPA treatment
for 15 s; PPA30, PPA treatment for 30 s; PPA60, PPA treatment for 60 s.
Different letters indicate significant differences (p
PPA30 reduced the total colony count by 1.09 log units (91.85% lower than the control group) (Fig. 9A), with a final microbial load of 3.04 log CFU/g. This indicates effective growth inhibition, but it did not meet sterilization standards, as the reduction did not reach the 6-log threshold required for sterilization. Reactive oxygen species/reactive nitrogen species (ROS/RNS) primarily damage cell membranes rather than causing complete cell disintegration. As shown by Giannoglou et al., 2021 [23], total bacterial counts decreased by 0.6 log levels after cold plasma treatment. The mechanism of action of PPA aligns with microbial inhibition rather than sterilization, consistent with the plasma-induced cell membrane damage mechanism. Rana et al., 2020 [22] also confirming the effectiveness of PPA in controlling microbial growth.
The decay rate analysis revealed significant preservation effects of PPA treatment, with treated strawberries exhibiting delayed onset and reduced severity of decay compared to the control (Fig. 9B, Fig. 10). While decay initiated on day 15 in both control and PPA15 treated fruit, PPA30 and PPA60 treatments extended the decay-free period to day 18, demonstrating temporal extension of fruit quality (Fig. 11). Quantitative assessment showed PPA30 achieved optimal efficacy with 56.1% decay reduction, surpassing PPA15 (32.0%) and PPA60 (10.0%), revealing a non-linear relationship between treatment duration and preservation outcomes. That is, insufficient exposure (PPA15) allowed for residual microbial proliferation, while excessive exposure time (PPA60) induced cellular damage compromising fruit defense mechanisms, as evidenced by accelerated late-stage decay. These findings align with established plasma research showing positive correlation between treatment duration and antimicrobial efficacy [39], while supporting observations of potential tissue damage from prolonged PPA exposure [36]. Results from our study highlight the critical importance of optimizing treatment parameters to balance immediate antimicrobial effects with preservation of host resistance mechanisms for maximal postharvest benefit.
 Fig. 10.
Fig. 10.
Strawberry samples rot with PPA treatment and storage time.
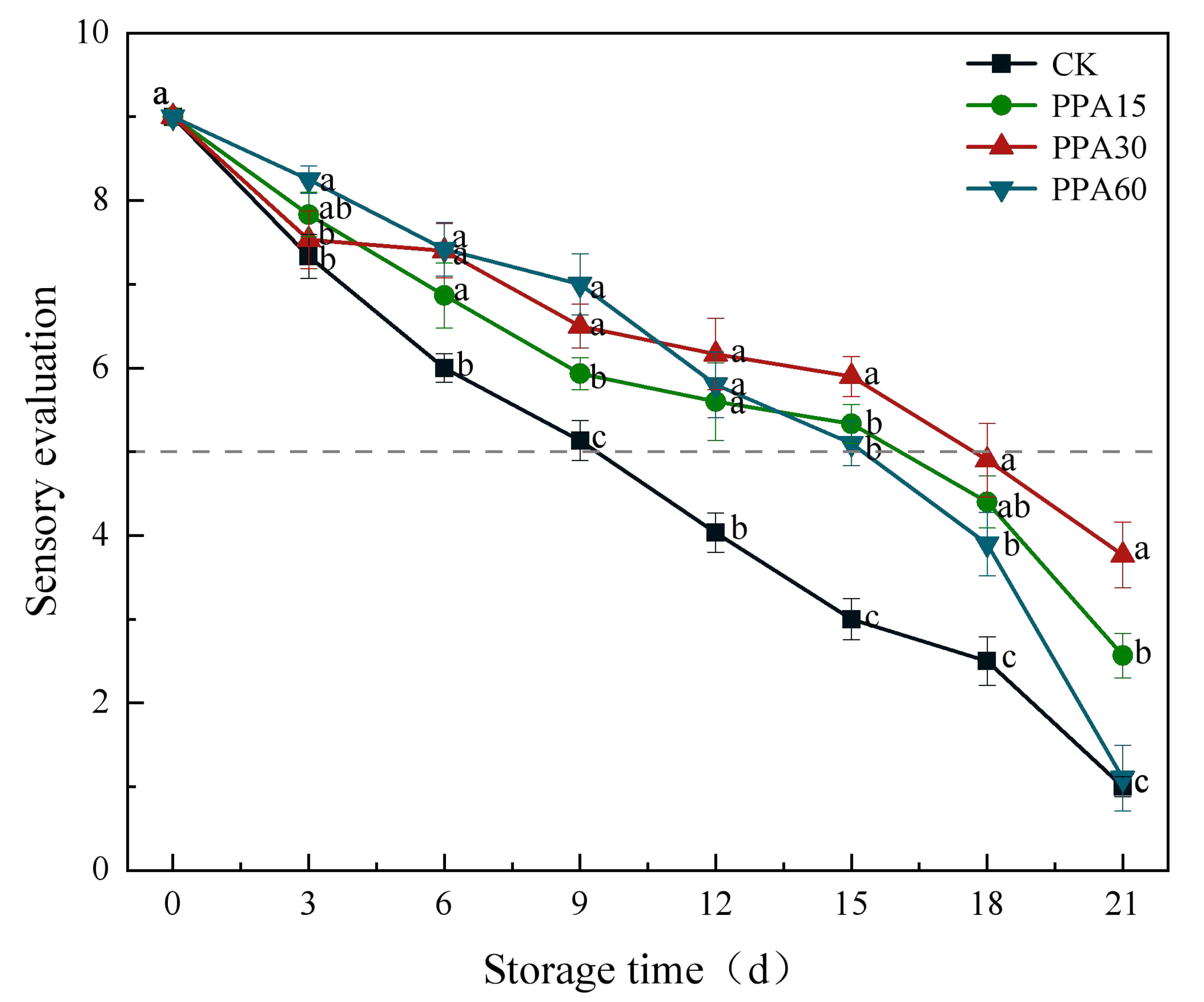 Fig. 11.
Fig. 11.
Effect of different PPA treatments on sensory quality of
strawberries during cold storage. Dash line (rating of 5) represents minimum
quality acceptability. CK, control; PPA15, PPA treatment for 15 s; PPA30, PPA
treatment for 30 s; PPA60, PPA treatment for 60 s. Different letters indicate
significant differences (p
Sensory evaluation, as a comprehensive assessment index, integrates a variety of
sensory experiences to reveal the overall quality of fruits and vegetables during
handling and storage, and has great significance to the commerciality of
strawberries [40]. Sensory evaluation revealed a progressive deterioration in
strawberry quality during storage (Fig. 11), with PPA-treated strawberries
demonstrating significantly (p
 Fig. 12.
Fig. 12.
Effect of different treatments on strawberry storage periods.
PPA treatments significantly inhibited microbial growth and extended the shelf
life of strawberries (p
Data are available from the author on request.
CYW and YBF and LL designed the research study. CYW and YBZ and YQL and YMZ performed the research. CYW and JZS and YBZ analyzed the data. CYW wrote the manuscript. All authors have contributed to the editorial changes made to the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research was funded by the Beijing Education Reform Innovation Project, grant number 20240227, Sub-Project of National Key R&D Program, grant number 10000200383 and Enterprise Collaborative Research Project grant number HXDK202450. The authors also express thanks to Dr. Sun for providing experimental equipment convenience.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.












