1 Department of Dairy Technology, Faculty of Agriculture, Ege University, 35100 Bornova-İzmir, Turkiye
2 Tire-Kutsan Vocational Training School, Ege University, 35900 Tire-İzmir, Turkiye
Abstract
Kefir is a traditional fermented beverage enriched with microorganisms, predominantly lactic acid bacteria (LAB) and yeasts, and is conventionally produced from animal-derived milks. However, the rising prevalence of lactose intolerance, the growing adoption of vegan diets, and increasing environmental sustainability concerns have stimulated interest in the utilization of plant-based milk alternatives for kefir production. Plant-based milks, such as those derived from soy, rice, oats, almonds, hemp, coconuts, and legume-based milks, present diverse nutritional compositions, bioactive compound profiles, and fermentation behaviors. Their protein, carbohydrate, and lipid contents directly influence the metabolic activity and growth of the symbiotic microbiota in kefir grains. Plant-based kefirs are of particular interest as functional foods, owing to their cholesterol-free composition, presence of phytochemicals such as phenolic compounds and phytosterols, and lactose-free nature. Nevertheless, challenges remain in achieving desirable sensory attributes, ensuring microbial adaptation, extending shelf life, and standardizing industrial-scale production processes. This review critically evaluates the suitability of plant-based milks for kefir production by integrating findings from recent studies and highlights their potential to align with sustainable food manufacturing, food safety considerations, shelf-life requirements, and evolving consumer health preferences.
Keywords
- kefir
- plant-based milks
- vegan diets
- food safety
- legume-based milks
- microbiological stability
Fermented dairy products have become essential components of the food landscape, reflecting a historical evolution deeply embedded in human dietary practices. Ancient civilizations recognized fermentation processes, even if they did not fully understand the underlying mechanisms. The spontaneous fermentation of milk marked the beginning of this culinary tradition, while the reuse of fermentation vessels improved the process’s repeatability and stability over time [1, 2].
Kefir, one of the most renowned fermented milk beverages, has its origins in the traditional practices of the Northern Caucasus Mountain tribes of Russia, located between the Caspian and the Black Sea. The longevity attributed to Caucasian populations has long been linked to their regular consumption of fermented dairy products, particularly kefir [3]. However, historical records concerning the precise emergence of kefir grains or the beginning of kefir production remain elusive. Traditionally, kefir was produced in sheepskin bags through a process of continuous fermentation under uncontrolled environmental conditions, in which fresh milk was replenished as fermented milk was withdrawn, thereby maintaining a perpetual fermentation cycle [4].
Kefir is a traditionally fermented beverage produced by inoculating animal-derived milks with kefir grains, which consist of a symbiotic consortium of lactic acid bacteria (LAB), acetic acid bacteria, and yeasts. The fermentation process results in a slightly acidic flavor, mild effervescence, and a complex microbial and biochemical profile characteristic of kefir [5]. Since kefir is made from cow milk, it contains all the nutrients in milk, such as fat, lactose, minerals, and vitamins. In fact, the synthesis of some vitamins during their formation and the partial breakdown of protein and lactose increase the nutritional value of kefir. The substances formed as a result of the changes in lactose and proteins under the influence of microorganisms cause a cooling, appetizing feature, a popular taste, and aroma to form. During the fermentation and storage of kefir, some changes are observed in the chemical properties of milk (lactose, fat, protein ratios, acidity, organic acids, free fatty acids, and volatile compounds) [6, 7].
The chemical profile of kefir is intricately influenced by both the milk substrate used and the production methods employed, whether traditional or industrial. Throughout the fermentation process, significant changes in ingredient composition and nutrient content have been observed. The primary fermentation byproducts include acetoin, acetaldehyde, ethanol, diacetyl, CO2, lactic acid, and acetic acid. Additionally, fermentation leads to a dynamic transformation in the vitamin content of kefir, with notable increases recorded in vitamins such as B1 and B12, as well as soluble amino acids, calcium, vitamin K, and folic acid. These findings emphasize the multifaceted effects of fermentation on the functional and nutritional components of kefir, highlighting its potential as a functional food product [8].
The nutritional value of kefir, which contains all the nutrients in milk, is quite high. Changes during fermentation cause differences in the composition of milk. Kefir is rich in vitamins B12, B1, and K and folic acid. Since the yeasts and acetic acid bacteria in its microflora show high antibiotic activity, it becomes a product rich in free amino acids, other protein hydrolysis products, and B-group vitamins as a result of their proteolytic activities [9]. The microflora of kefir ferments the milk and forms lactic acid, CO2, small amounts of acetone, alcohol, acetaldehyde, and diacetyl, contributing to the formation of kefir’s unique aroma and consistency. Diacetyl is produced by Lactococcus lactis subsp. diacetylactis and Leuconostoc species. Along with its high calcium and magnesium content, kefir is also a good source of phosphorus, which helps the use of carbohydrates, fat, and protein. A good kefir should contain 0.6–0.9% lactic acid, 0.6–0.8% alcohol, and 50% CO2, 0.6–0.8% ethyl alcohol, different aldehydes, and acetone [10, 11]. The microbiological profile of kefir is profoundly influenced by the diverse array of microorganisms found within the kefir grains. Kefir, resulting from the fermentation of milk with either kefir grains or bulk cultures derived from these grains, exhibits distinct microbial characteristics [12].
The kefir microbiota, particularly the complex community of bacteria and yeasts, is a key factor that determines the functional properties of kefir. The microbial composition differs between milk kefir and water kefir. In milk kefir, LAB and yeasts are the dominant microorganisms present in the kefir grains. These microorganisms play a crucial role in fermentation during the production process and contribute to the development of probiotic properties [13, 14].
Researchers conducted a comprehensive investigation to identify the bacterial and yeast cultures present in kefir, revealing a diverse microbial consortium. The bacterial species identified included Lactobacillus kefir, Lactobacillus delbrueckii subsp. delbrueckii, Enterococcus faecalis, Enterococcus faecium, Lactococcus lactis subsp. cremoris, Lacticaseibacillus casei, Lactobacillus brevis, Lactobacillus acidophilus, Streptococcus thermophilus, Lactobacillus helveticus, Lactobacillus fermentum, Lactobacillus kefiranofaciens, Leuconostoc spp., and Lactococcus spp., along with Lactobacillus delbrueckii subsp. bulgaricus. The identified yeast strains included Zygosaccharomyces spp., Kluyveromyces spp., Pichia spp., Torula spp., Candida spp., and Saccharomyces spp., with Kluyveromyces lactis, Kluyveromyces marxianus, and Saccharomyces cerevisiae predominating among the yeast microflora. Additionally, Torulaspora delbrueckii, Zygosaccharomyces rouxii, Debaryomyces hansenii, Saccharomyces unisporus, and Torulaspora elbrus were identified in both kefir beverage and kefir grain microflora. This thorough microbial characterization provides insights into the intricate microbial dynamics within kefir, enhancing our understanding of its fermentation process and potential health benefits [15, 16, 17, 18, 19, 20, 21].
Traditional and industrial kefir differ primarily in their production methods, microbial composition, and overall quality. Traditional kefir is typically made through natural fermentation using a combination of kefir grains, which contain a diverse community of bacteria and yeasts. This process, which often occurs on a smaller scale in home environments or small dairies, enables a more diverse microbial profile and typically results in a richer, more complex flavor and texture. In contrast, industrial kefir is produced on a larger scale using standardized fermentation processes, typically with selected starter cultures that may lack the diverse microbial diversity found in traditional kefir. The industrial process is optimized for consistency, longer shelf life, and high-volume production, which can sometimes lead to a more uniform taste but may not capture the same depth of microbial complexity or health benefits. Furthermore, traditional kefir tends to retain more of the natural nutritional content due to minimal processing, while industrial kefir may undergo pasteurization and additional processing steps that could reduce its probiotic potential and nutrient levels [22, 23].
The preference for plant-based beverages over dairy milk is primarily driven by nutritional, health, and environmental considerations. Individual sensitivities such as lactose intolerance or milk protein allergies steer consumers away from animal-based products toward plant-based alternatives [24]. Additionally, vegan or plant-based dietary choices, often motivated by ethical and environmental concerns, further encourage the consumption of plant-derived beverages [25]. Plant-based milks offer advantages for cardiovascular health due to their low saturated fat content and absence of cholesterol. Furthermore, soy, almond, oat, and other plant-based milks can be fortified with vitamins and minerals to enhance nutritional value and provide functional food properties [26]. From a sustainability perspective, the production of plant-based beverages typically requires less water and generates lower greenhouse gas emissions compared to dairy milk production [27]. Collectively, these factors play a significant role in guiding consumers toward plant-based beverages, taking into account both their health benefits and environmental responsibility. In this context, plant-based milk alternatives have begun to be used in the production of many dairy products, such as kefir, and this demand continues to increase (Table 1, Ref. [28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39]).
| Study (Authors, publication) | Plant-based milk/substrate | Fermented product type | Microorganisms/starters | Main findings/notes |
| Harper et al. [30] | General (soy, oat, almond, coconut) | General (yogurt-like drink, kefir) | LAB and yeasts | Fermentation properties vary across plant matrices; starter adaptation required. |
| Ruiz de la Bastida et al. [31] | Soy | Fermented soy beverage | Probiotic Lactobacillus spp. | Soy products are suitable as probiotic carriers. |
| Liu et al. [32] | Oat milk | Fermented oat milk | Yogurt bacteria | Showed immunomodulatory functional effects. |
| Li et al. [33] | Soy | Soy yogurt | Four LAB strains | Strain selection critically affected quality attributes. |
| Ziarno et al. [34] | Soy and oat | Yogurt-type beverages | Commercial starters | Some starters are successful; texture/syneresis issues may occur. |
| Benmeziane and Belleili [35] | Oat milk | Fermented oat milk | Yogurt starters | Enzymatic pretreatment improved product stability. |
| Xu et al. [36] | Oat | Set-type oat yogurt | LAB + enzymatic hydrolysis | Enzymatic pretreatment enhanced quality. |
| Ceylan and Öncül [28] | Oat, soy, coconut | Plant-based kefir | Kefir cultures | Observed pH reduction, probiotic viability, and antimicrobial activity. |
| Sahin et al. [37] | Almond milk (+ almond skin) | Plant-based kefir | Kefir grains | Almond skin enhanced phenolic and antioxidant content. |
| Tajmousavilangerudi et al. [38] | Various plant substrates | Water kefir | Water kefir grains | Mixed substrates yielded successful kefir products. |
| La Torre et al. [29] | Almond | Almond kefir | Milk & water kefir grains | Bioactive content varied depending on kefir grain type. |
| Luca and Oroian [39] | Oat | Oat yogurt + synbiotic microcapsules | Probiotic microcapsules | Improved probiotic stability and texture. |
LAB, Lactic Acid Bacteria.
This review aims to systematically examine plant-based milk alternatives used in kefir production. The primary objective of the study is to analyze the effects of different plant-based milk types (soy, almond, oat, rice, coconut, etc.) on fermentation properties, microbial adaptation, probiotic stability, and sensory quality, thereby guiding future research and industrial applications. The scope of the study includes evaluation of the nutritional composition of plant-based milks, the performance of kefir grains or starter cultures, functional ingredient production, and shelf-life parameters. The article focuses on which plant-based milk alternatives are most suitable for kefir production, how microbial diversity and probiotic stability vary across different plant-based milks, which strategies may be effective for improving sensory and functional quality, and how industrial-scale production and standardization processes can be optimized. This approach aims to identify knowledge gaps in plant-based kefir production and contribute to the development of sustainable, nutritious, and consumer-friendly products.
In response to increasing lactose intolerance, the growing adoption of vegan diets, and the demand for more sustainable food systems, kefir is now produced not only from bovine, caprine, or ovine milks but also from a wide range of plant-based milk alternatives. These include soy, almond, oat, rice, coconut, legume-derived, and cereal-based milks [28]. The varying protein, carbohydrate, and lipid compositions of plant-based milks directly influence the metabolic activity of kefir grain microbiota, thereby affecting fermentation dynamics, nutritional properties, and sensory attributes. Consequently, plant-based kefir has emerged as a promising functional and sustainable fermented product with evolving industrial and consumer relevance [40].
Plant-based fermented dairy alternatives exhibit shelf-life characteristics that are strongly influenced by their intrinsic composition, starter culture dynamics, and post-fermentation storage conditions. Unlike cow milk, plant matrices such as soy, oat, almond, and coconut differ substantially in carbohydrate profile, protein structure, buffering capacity, and phenolic content, all of which modulate acidification kinetics and microbial stability during fermentation. These products typically rely on selected LAB or probiotic strains that lower pH and produce antimicrobial metabolites (e.g., organic acids, bacteriocins), contributing to pathogen inhibition and shelf-life extension. However, the lower protein content and variable fat composition of plant substrates may reduce microbial robustness and accelerate physicochemical deterioration, including phase separation, syneresis, and oxidation-driven off-flavors. Shelf life is additionally shaped by packaging atmosphere, cold-chain integrity, and potential post-processing steps such as heat treatment or high-pressure processing. Overall, achieving optimal shelf stability in plant-based fermented products requires a tailored approach that combines substrate optimization, targeted culture selection, and controlled processing to maintain microbial viability, sensory quality, and safety throughout storage [41, 42].
Food safety, microbial risks, and shelf life are critical quality and safety parameters in the industrial production of plant-based fermented products. Due to the agricultural origin of raw materials, chemical hazards such as pesticide residues, heavy metals, mycotoxins (particularly aflatoxins and ochratoxins), as well as allergens from soy and nuts, represent major safety concerns [43, 44]. From a microbiological perspective, pathogens including Escherichia coli O157:H7, Salmonella spp., Listeria monocytogenes, and Bacillus cereus are considered the primary risks [45]. The use of Generally Recognised as Safe/Qualified Presumption of Safety (GRAS/QPS)-status LAB in controlled fermentation enhances safety through pH reduction, production of organic acids, and bacteriocins; however, in low-acid substrates, fermentation kinetics are generally slower compared to dairy matrices, thereby limiting product shelf life [46]. Typically, the shelf life of plant-based fermented products ranges between 21–30 days under refrigeration, yet factors such as lipid oxidation, flavor deterioration, and microbial spoilage may reduce stability. To address these limitations, the industry implements strict cold-chain management, hygienic equipment design, and oxygen-barrier packaging solutions, while novel non-thermal technologies such as pulsed electric fields and high-pressure processing are increasingly explored to extend shelf life and ensure microbiological stability [47, 48].
Cow milk and plant-based milks exhibit fundamental differences in their native microbiota, chemical composition, and fermentation behavior, which collectively result in markedly distinct microbial dynamics during fermentation. Cow milk inherently contains a diverse and well-characterized microbial community—including LAB, coryneform bacteria, micrococci, pseudomonads, and low levels of yeasts and molds—originating primarily from the animal, the milking environment, and post-harvest handling conditions. The natural abundance of LAB renders cow milk highly conducive to fermentation. In contrast, plant-based milks generally harbor environmental microorganisms such as Bacillus, Pseudomonas, and various wild yeasts derived from plant surfaces or processing equipment, while LAB are typically absent or present only at negligible levels. The differences in carbohydrate profiles further modulate microbial metabolism: lactose in cow milk serves as an optimal carbon source for LAB, whereas the glucose, sucrose, starch, or maltodextrin fractions characteristic of plant-based milks often constitute suboptimal substrates, necessitating sugar supplementation or enzymatic pre-treatment to support adequate fermentation. Moreover, plant-based milks contain phytochemicals—including phenolic compounds, saponins, and phytic acid—that may exert inhibitory effects on kefir-associated microorganisms, in contrast to cow milk, whose principal antimicrobial defenses consist of lactoferrin, lysozyme, and immunoglobulins. The strong buffering capacity of cow milk, attributed to its casein–phosphate system, facilitates controlled acidification, whereas the comparatively low buffering capacity of plant-based milks leads to rapid pH decline and premature inhibition of bacterial growth. As a result, kefir grains readily adapt to cow milk and maintain stable bacterial–yeast interactions, while plant-based substrates often require adaptation phases and display more variable microbial performance. Collectively, these microbiological and compositional disparities profoundly influence the fermentation efficiency, sensory profile, and overall stability of plant-based kefir products [49, 50, 51].
Although kefir can generally be produced from cow, sheep, or goat milk, studies have also explored kefir made from soy, rice, coconut, and other plant-based milks (Fig. 1). Beyond changing the type of raw material used in production, recent research has investigated the effects of incorporating various ingredients, such as prebiotics, minerals, and functional components, on the physical, chemical, microbiological, textural, sensory, and functional properties of kefir [52, 53]. This section also summarizes studies on the enrichment and functional properties of plant-based kefirs.
 Fig. 1.
Fig. 1.
General flow diagram in the production of plant-based milk alternatives. In some processes (e.g., oats) heat treatment may be applied before milling or straining; the order varies according to the manufacturer’s recipe.
Several processing steps are involved in the production of plant-based milk alternatives (Fig. 2). Depending on whether these beverages are intended for direct consumption or further processing into other food products, different procedures may be applied. Raw material selection and quality control include the choice of seeds, nuts, grains, or bran and involve checks for moisture, mold, foreign matter, and pesticide residues [54]. Cleaning operations such as washing, stone or gravel removal, and elimination of hardened fragments are performed to ensure safety and quality. In some cases (e.g., legumes and nuts), soaking is required to enhance digestibility and nutrient bioavailability. Typically, almonds or soybeans are soaked for 6–12 hours in cold water or 1–2 hours in hot water, with a solid-to-water ratio ranging from 1:3 to 1:4 (w/v) [25]. The soaked material is then finely ground with water using high-speed mills, blenders, or industrial stone/conical grinders. The degree of grinding directly affects the texture and viscosity of the resulting milk. Additional water is incorporated to form the crude milk, with the water-to-solid ratio depending on the raw material (e.g., almond 1:3–1:5, soy 1:8–1:10, oat 1:3–1:5) [55]. To separate the solid fraction, straining/filtration is performed using cloth, sieves, centrifuges, or membrane filters, such as ultrafiltration. The filtration degree determines creaminess and fiber content [56]. To ensure microbiological stability, thermal processing is commonly applied, such as high-temperature short-time (HTST) or ultra-high temperature (UHT) treatments. UHT processing is especially used for shelf-stable products and may also serve to inactivate starch- and polyphenol-degrading enzymes in oats and soy [54, 56]. To achieve a stable oil-in-water emulsion, emulsification/homogenization is carried out using high-pressure homogenizers (e.g., 100–200 bar) [25]. For direct consumption, stabilizers and flavoring agents may be added, such as salt, sugar, natural flavors (e.g., vanilla), ions, emulsifiers (e.g., lecithin), and stabilizers (e.g., pectin, carboxymethyl cellulose, gum arabic). Stabilizers help reduce sedimentation and phase separation [24]. Fortification is often achieved through the addition of calcium carbonate or calcium phosphate, as well as vitamins such as D2/D3, B12, and riboflavin [57]. After pasteurization, rapid cooling to 4–6 °C helps to slow microbial growth. Filling is conducted aseptically or under chilled conditions, using packaging materials such as cartons, PET bottles, or glass containers. Finally, storage and distribution conditions are defined according to the applied thermal treatment, and shelf-life is determined before labeling [55].
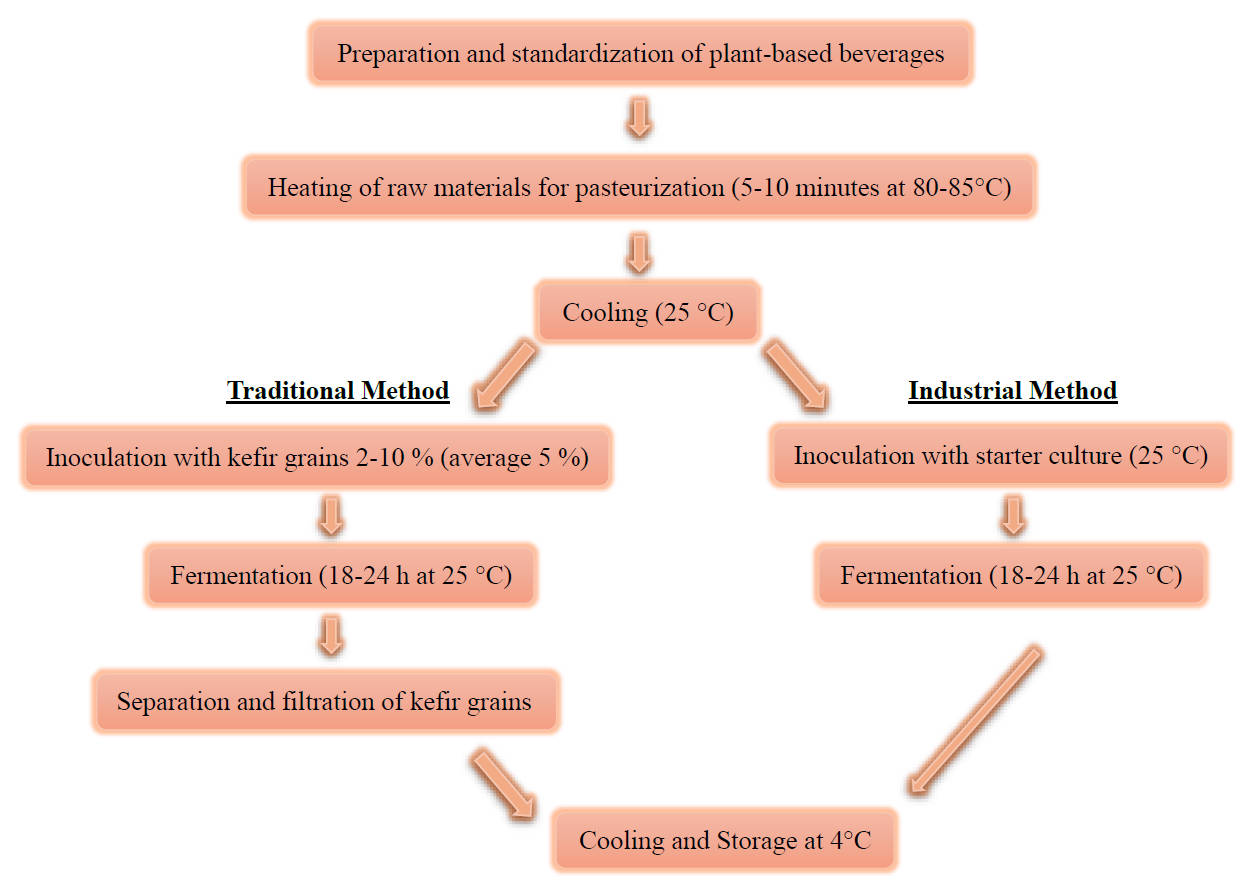 Fig. 2.
Fig. 2.
Schematization of traditional and industrial plant-based kefir production stages.
While it is used as an alternative to many cow milk products, soy milk is the most commonly used substitute for cow milk in kefir production. From this perspective, most studies have been on fermented dairy products produced using soy milk. Liu et al. [58] demonstrated that both cow and soy milk kefirs exhibit significantly higher antimutagenic activity and enhanced antioxidant properties—including increased DPPH (1,1-Diphenyl-2-picrylhydrazyl) radical scavenging, inhibition of linoleic acid peroxidation, and reducing power—compared to their unfermented counterparts. However, kefir samples showed lower glutathione peroxidase activity, while superoxide dismutase activity and iron ion chelation remained unaffected. These results underscore the potential of milk and soy milk kefirs as functional foods with antimutagenic and antioxidant benefits.
Gamba et al. [59] evaluated cow and soy milk kefirs in terms of chemical, microbial, and functional properties. Fermentation increased free amino acids (20.92 mg/100 mL in cow milk and 36.20 mg/100 mL in soy milk kefir), with glutamic acid predominating, indicating microbial proteolysis. Cow milk kefir contained higher yeast and acetic acid bacteria counts, while soy milk kefir showed lower levels. Common microorganisms included Lactococcus lactis subsp. lactis, Saccharomyces cerevisiae, and Kazachstania unispora, with Acetobacter orientalis detected only in cow milk kefir. Both kefirs exhibited enhanced antibacterial activities, antioxidant, and ACE (Angiotensin-Converting Enzyme)-inhibitory, demonstrating their functional and health-promoting potential.
In addition to soy milk, important studies have also been conducted on some other milk alternatives. Sirirat and Jelena [60] produced kefir from cow and rice milk, investigating their bacterial inhibition and antimicrobial activity using the disk diffusion method. The inhibitory effects were tested against Staphylococcus aureus, Bacillus subtilis, Escherichia coli, and Pseudomonas fluorescens. Kefir demonstrated activity against all the microorganisms tested. Notably, rice milk kefir exhibited significantly higher antioxidant activity compared to cow milk kefir. The researchers suggested that rice milk kefir could be considered a promising food ingredient for preventing oxidative stress.
Carullo et al. [61] developed a novel symbiotic kefir by fortifying probiotic milk kefir with inulin-based polymers enriched with ultrasound-assisted white grape seed pulp extract, rich in catechins and glycosylated flavonoids. This post-fermentation fortification enhanced antioxidant capacity, with 50 g of fortified kefir providing antioxidant activity equivalent to 300 mg of ascorbic acid. In vitro assays confirmed no adverse effects on intestinal barrier integrity, highlighting the potential of this formulation as a functional food with improved health-promoting properties.
As in the studies summarized above, although research on plant-based kefir production has increased in recent years, several gaps remain in this field. First, the comparative evaluation of different plant-based milk alternatives in terms of their nutrient composition and suitability for fermentation is still insufficient. Moreover, the long-term adaptation of kefir grains or starter cultures to plant-based substrates, the preservation of microbial diversity, and the stability of probiotic populations have not been adequately explored. Efforts to improve sensory attributes such as taste, aroma, and texture through innovative biotechnological approaches are also at an early stage. In addition, systematic investigations into the production and bioavailability of functional compounds (e.g., bioactive peptides, vitamins, antioxidants) are lacking. Industrial-scale challenges such as process standardization, shelf-life optimization, and consumer acceptance have also received limited attention. These gaps highlight the need for innovative strategies to transform plant-based kefir into not only an alternative product but also a sustainable food with enhanced nutritional and functional value [62, 63].
Plant-based milks present unique challenges in terms of pathogen persistence, safety control strategies, shelf-life extension, and overall food safety due to their highly variable compositions and the absence of intrinsic antimicrobial components typically found in animal-derived milk. Unlike cow milk—which contains lactoferrin, lysozyme, immunoglobulins, and a robust buffering system that can inhibit or slow the growth of pathogens—plant-based milks rely primarily on the antimicrobial properties of phytochemicals such as phenolics, saponins, and organic acids, whose effectiveness varies considerably among raw materials [64, 65]. Numerous studies indicate that pathogens such as Salmonella spp., Listeria monocytogenes, Bacillus cereus, and Escherichia coli O157:H7 may survive or even proliferate in plant-based milks, particularly those with higher pH values, low buffering capacity, and simple sugars that support microbial metabolism. The stability of spores, especially from B. cereus and related species commonly associated with cereals and legumes, poses an additional concern. Effective safety control strategies therefore, require a combination of stringent thermal or non-thermal processing (e.g., HTST pasteurization, UHT treatment, high-pressure processing, pulsed electric fields), hygienic production practices, and the use of protective cultures or bio-preservatives to inhibit pathogenic growth. Furthermore, shelf-life is strongly influenced by initial microbial load, water activity, oxygen permeability of packaging, and susceptibility to post-processing contamination [66, 67]. Advanced packaging technologies such as aseptic filling, oxygen-barrier multilayer materials, and modified atmosphere packaging can significantly reduce spoilage and pathogen risk. Comprehensive hazard analysis, routine microbial testing, and the integration of predictive microbiology models are essential to ensure food safety throughout distribution and storage. Collectively, managing pathogen survival and ensuring microbiological stability in plant-based milks requires an optimized combination of formulation adjustments, processing technologies, and robust safety monitoring systems [68, 69].
The use of plant-based milks in the production of kefir and other fermented dairy products has emerged as a significant research area in recent years, driven by the increasing prevalence of vegan dietary preferences, lactose intolerance, and allergies to milk proteins. Plant-derived substrates such as soy, almond, oat, coconut, rice, chickpea, and flaxseed are being employed as alternatives to bovine milk, enabling the development of fermented products with high probiotic content and functional properties [70]. During the fermentation of these beverages, traditional kefir grains or specific starter cultures (Lactobacillus spp., Lactococcus spp., acetic acid bacteria, Lactobacillus acidophilus, Bifidobacterium spp.) are used, resulting in a typical pH reduction to the range of 4.6–4.9, an increase in total titratable acidity, and the maintenance of probiotic populations at levels of 106–107 CFU/mL throughout storage [28, 71]. Furthermore, fermentation has been shown to markedly enhance the concentrations of phenolic compounds, flavonoids, and total antioxidant capacity [29, 72, 73]. However, the microbial ecology of plant-based kefir may differ from that of dairy-based kefir, and certain pathogens (Escherichia coli O157:H7, Salmonella Typhimurium) have been reported to exhibit greater persistence in specific plant matrices [28]. From a sensory perspective, kefirs produced from certain plant-based milks, such as those derived from nuts, have been found to possess a richer aroma profile, improved fatty acid composition, and pseudoplastic rheological characteristics, whereas oat- and rice-based kefirs tend to offer a lighter flavor and a creamier texture [74, 75]. Nevertheless, the low buffering capacity, differences in protein composition, and absence of lactose in plant-based beverages can influence fermentation kinetics and product stability; therefore, the achievement of optimal pH reduction, texture, flavor balance, and microbial viability requires careful optimization of starter culture selection, pre-treatment methods (e.g., enzymatic hydrolysis, heat treatment), and fermentation conditions [71]. When comparing studies using plant materials as alternatives to cow milk;
Kesenkas et al. [76] produced kefir using mixtures of cow and soy milk, investigating the physicochemical, microbiological, and sensorial characteristics of the kefir samples during refrigeration. The overall composition of the samples was closely related, with the exception of lactose content. The levels of tyrosine in the kefir were also quite similar. As the proportion of soy milk increased, the leucine content also increased. Serum separation was observed to increase during storage for all samples. The lowest viscosity value was recorded when soy milk was mixed with cow milk in a 50:50 ratio. Lactic acid levels were the highest among the organic acids present. The addition of soy milk did not significantly impact the microbiological population. However, sensory scores generally decreased with higher soy milk content.
Egea et al. [77] evaluated the physicochemical and rheological properties of soy milk fermented with kefir. Titratable acidity ranged from 0.25% to 1.47%, and pH values varied between 5.06 and 5.89%. The flow behavior of the fermented soy milk was described using the Herschel-Bulkley model, indicating a non-linear relationship between shear rate and shear stress. Consequently, all samples were classified as non-Newtonian, exhibiting pseudoplastic behavior.
Bensmira and Jiang [78] investigated the antioxidant activity and total phenolic content of a novel peanut-based kefir. The composition of the kefir extract was characterized using HPLC (High-performance liquid chromatography), while antioxidant activity was evaluated via three complementary assays. Results demonstrated that peanut-milk kefir exhibited enhanced antioxidant properties compared to the original milk, and fermentation with kefir culture increased the soluble phenolic content. The extract contained two main classes of phenolics: phenolic acids and flavonoids.
Cui et al. [79] conducted a study evaluating the use of kefir grains as an inoculum for producing walnut milk beverages. Their investigation focused on assessing the individual effects of fermentation time, temperature, sucrose concentration, and inoculum size on the fermentation process of walnut milk. The results underscored the significant influence of each factor on fermentation. Specifically, fermentation time, temperature, and sucrose concentration exhibited highly significant single-factor effects, while inoculum size also demonstrated a notable impact. Based on their findings, the optimal fermentation conditions for walnut milk beverage production were identified as a temperature of 30 °C, a fermentation time of 12 hours, an inoculum size of 3 grams of kefir grains (wet weight), and a sucrose concentration of 8 grams per 100 mL. Furthermore, the analysis of the resulting beverages revealed the presence of viable cells of lactococci, lactobacilli, and yeast, with concentrations of 7.91, 8.24, and 6.00 log CFU/mL, respectively. These findings provide valuable insights into the optimal conditions for fermenting walnut milk beverages using kefir grains as an inoculum, as well as the resulting microbial populations within the beverage.
Sabokbar and Khodaiyan [80] conducted a study to evaluate the feasibility of using a mixture of pomegranate juice and whey as a substrate for creating a novel probiotic beverage with kefir grains. They explored various fermentation conditions, including different temperatures (ranging from 19 °C–25 °C) and two levels of kefir grain infusion (5% and 8% w/v). The results indicated that kefir grains effectively metabolized lactose, resulting in a reduction in pH, an increase in acidity, and the production of lactic and acetic acids, while the level of citric acid decreased. Importantly, these changes depended on both the fermentation temperature and the concentration of kefir grains, with the most significant effects observed at 25 °C and with an 8% (w/v) kefir grain infusion. These findings suggested that the combination of pomegranate juice and whey was a suitable substrate for producing a novel probiotic milk-juice beverage using kefir grains. Additionally, sensory evaluations indicated favorable results regarding the sensory properties of the resulting beverage, suggesting its potential as an acceptable and probiotic-rich product.
Abadl et al. [81] investigated the feasibility of producing coconut-based kefir beverages using low- and high-fat coconut milk. The study evaluated multiple parameters, including pH, titratable acidity, peptide content, antibacterial and antioxidant activities, as well as the rheological properties of the resulting beverages. The results demonstrated a significant increase in the populations of LAB and yeasts after 24 h of fermentation. Notably, fermentation with kefir grains enhanced the peptide content in both low-fat coconut milk (0.7 mg/L) and high-fat coconut milk (0.571 mg/L). Furthermore, the study revealed that low-fat coconut kefir demonstrated strong antibacterial growth inhibition and antioxidant activity. Rheological analysis indicated that the apparent viscosity of the kefir beverage decreased with increasing shear rate, while exhibiting higher viscosity at lower temperatures. Based on these findings, it was concluded that low-fat coconut milk serves as a viable alternative to cow milk in the production of kefir-based beverages. This research highlights the potential of coconut-based kefir as a functional beverage with promising health benefits, providing a valuable option for individuals seeking alternatives to traditional dairy-based kefir beverages.
Wulansari [82] evaluated kefir quality with the addition of oat milk (8.12% and 16% w/v) and Lactobacillus casei AP (2% and 4%, v/v), focusing on physicochemical properties, including nutrient content. The findings revealed that increasing the oat milk concentration reduced water content and enhanced viscosity. Notably, the combination of 16% oat milk and 4% (v/v) Lactobacillus casei AP yielded the highest viscosity and water retention, resulting in the most favorable acceptability of the kefir products. This study underscores the potential to optimize kefir quality by adjusting oat milk levels and probiotic addition, highlighting the role of these factors in improving sensory characteristics and overall product acceptance. However, increasing the Lactobacillus casei AP inoculum and oat milk concentrations did not affect microbiological quality. In conclusion, the addition of 16% oat milk and 4% Lactobacillus casei AP improved the physical and sensory qualities of kefir products.
Gocer and Koptagel [83] explored the use of hazelnut, cashew, peanut, walnut, and almond-based beverages in kefir production. The hazelnut beverages had the highest energy content (73.71 kcal/100 g), followed by kefir made from hazelnut products (74.89 kcal/100 g). Over the storage period, total sugar, total organic acids, and polyunsaturated fatty acids levels in the kefir samples decreased, while saturated fatty acid content and pH increased. Kefir made from hazelnut beverages had a higher unsaturated fatty acid content and a lower saturated fatty acid content than kefir made from cow milk. Among flavor components, ethanol and ethyl acetate, key contributors to flavor, were found in the highest concentrations in kefir made from cow milk.
As can be seen, kefir production has been carried out using many plant-based milk alternatives to cow milk. However, much research is needed to improve the quality, stability, and sensory properties of these products. Furthermore, studies on microbiological risks, shelf life, and pathogen persistence of kefir produced using plant-based milk alternatives are insufficient. Therefore, further studies on these topics are necessary.
The utilization of plant-based beverages in the production of kefir and other fermented milk analogues has emerged as a rapidly expanding domain within functional food science, driven by increasing consumer demand for dairy-free, lactose-free, and allergen-friendly products. Plant-derived substrates have been successfully fermented using traditional kefir grains or selected probiotic starter cultures, yielding products with desirable microbial counts, appropriate pH and acidity profiles, and enriched levels of bioactive compounds. These beverages not only diversify the probiotic food market but also offer additional nutritional and phytochemical benefits, including phenolic compounds, flavonoids, and enhanced antioxidant capacity. Enzymatic pretreatments of different plant-based milk substrates, such as protein hydrolysis or starch modification, could enhance fermentation suitability, supporting microbial growth and probiotic stability. Biotechnological approaches, including strain selection and adaptive laboratory evolution, may be applied to ensure long-term adaptation of kefir grains or starter cultures to plant-based matrices. Sensory attributes can be improved through co-culture fermentations with aroma-producing bacteria or enrichment with natural aroma precursors. Additionally, the bioavailability of functional compounds could be enhanced via microencapsulation, combination with prebiotics, and the use of active packaging technologies to extend shelf-life at an industrial scale.
Industrial-scale kefir production using plant-based milks continues to present substantial research gaps despite growing scientific interest. The distinct protein composition, buffering capacity, and carbohydrate profile of plant matrices markedly alter microbial fermentation kinetics compared to traditional dairy kefir, creating unresolved challenges related to kefir grain adaptation, maintenance of a stable microbial consortium, and consistent product quality. From an industrial perspective, process standardization, achieving uniform viscosity, preventing post-fermentation phase separation, and optimizing thermal and cold-chain stability remain major technological hurdles. Furthermore, matrix components such as phytates, tannins, and dietary fibers may inhibit microbial metabolism and reduce the bioavailability of key bioactive compounds produced during fermentation—including short-chain fatty acids, bioactive peptides, and exopolysaccharides. The gastrointestinal stability, absorption mechanisms, and targeted physiological effects of these compounds are still insufficiently characterized, demanding more comprehensive in vivo evidence. Focus on elucidating microbial ecology dynamics through multi-omics approaches, modeling matrix–microbe interactions, and developing advanced processing technologies to enhance product stability and improve the bioactive profile of plant-based kefir systems are important issues. Future research should focus on optimizing the selection of starter cultures tailored to specific food matrices, as well as pre-treatment techniques (e.g., enzymatic hydrolysis, fortification, controlled heat treatment) and fermentation parameters, to improve both functional and sensory attributes. Moreover, comprehensive shelf-life and safety assessments are essential to ensure the microbiological safety of products in commercial applications.
OY, HEO & YB: Writing–review & editing, Writing–original draft, Visualization, Validation, Supervision, Resource, Methodology, Investigation, Funding acquisition, Conceptualization. All authors have contributed to the editorial changes made to the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We are grateful to Ege University Planning and Monitoring Coordination of Organizational Development, Article Writing and Publication Support Office, and Directorate of Library and Documentation for their contribution in editing and proofreading services of this study.
This research received no external funding.
The authors declare no conflict of interest.
AI and AI-Assisted Technologies were also utilized for the editing. After using these tools, the authors reviewed and edited the content as needed and takes full responsibility for the content of the publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


