1 Centro Nacional de Ciencia y Tecnología de Alimentos (CITA), Universidad de Costa Rica (UCR), Ciudad Universitaria Rodrigo Facio, 11501-2060 San José, Costa Rica
2 Escuela de Química, Facultad de Ciencias Básicas, Universidad de Costa Rica (UCR), Ciudad Universitaria Rodrigo Facio, 11501-2060 San José, Costa Rica
Abstract
The study’s main objective was to quantify the acrylamide (ACR) content in potato, plantain, and cassava crisps—products widely consumed in Costa Rica. ACR, a possible carcinogen according to various global organizations, is generated during the Maillard reaction when foods rich in asparagine (ASN) and reducing sugars are subjected to temperatures above 120 °C.
Using GC-MS analysis on n = 54 samples (24 potatoes, 18 plantains, and 12 cassavas), it was determined that ACR levels were within the ranges established by international organizations such as Codex Alimentarius (CODEX) and the Food and Drug Administration (FDA). The reducing sugra and ASN content of the raw materials was quantified to correlate them with the ACR in the final product.
One potato product was identified with an ACR concentration that significantly exceeded the 750 μg kg-1 limit stipulated in Recommendation (EU) 2019/1888. For plantain and cassava chips, which currently have no specific regulations, the results showed ACR content to be significantly lower compared to potato crisps. The findings demonstrated a significant correlation between the initial asparagine content and ACR formation in potato crisps, a relationship not observed with reducing sugars. In contrast, no direct correlation was found between precursors and ACR in plantain chips. The analysis also revealed that, in addition to asparagine concentration, the crisps’s surface-to-volume ratio is a crucial physical parameter for minimizing ACR formation.
The data obtained on daily ACR intake will serve as a valuable input for future risk studies in the Costa Rican population, suggesting that plantain and cassava chips are a safer alternative due to their lower ACR content.
Keywords
- Costa Rica
- snack foods
- acrylamide
- asparagine
Plant-based crisps are revolutionizing the snack industry by providing consumers with a healthier and more sustainable alternative to traditional potato chips [1, 2]. As a relevant food commodity, snack consumption has been estimated worldwide, including in the United States [3], Iran [4], Germany, and the United Kingdom [5]. It is an industry projected to grow. For example, in Costa Rica alone, the snack food market is expected to grow at an annual rate of 4.32% [6]. It is expected to represent a total revenue of up to USD 275.15 million in 2025, from which salty snacks made from potatoes, plantains, and cassava account for 9% of the trade items [7]. However, despite their popularity, findings suggest that fried snack overeating is linked to an increase in the number of diseases such as cancer, heart disease, obesity, diabetes, and high blood pressure [8]. Additionally, high temperatures increase the risk of nutrient loss, oil oxidation, and acrylamide (ACR) formation during the frying process [9, 10], a process primarily used to make crispy snacks.
Heat treatments, such as drying or frying, are often used in the food industry to process and preserve plant-based foods [11]. Frying enables physicochemical changes, such as starch gelatinization (resulting in larger starch granules), crust formation (due to the drying of the fried product), and the formation of taste and aroma components [8]. When heated above 120 °C, plant-based foods are particularly susceptible to the Maillard Reaction [12]. They are likely to produce a significant amount of ACR due to the presence of reducing sugars and the amino acid asparagine (ASN) [13, 14]. In addition, ACR formation is susceptible to frying temperature, time, the size of the contact surface with oil, and the interaction between raw materials and oil. As a result, there has been significant concern about the ACR incidence in fried foods due to the mounting evidence of its adverse health effects.
The International Agency for Research on Cancer [15] classified ACR as a probable human carcinogen (Group 2A). It is an already familiar hazardous compound with reported carcinogenicity, genotoxicity, neurotoxicity, and reproductive toxicity in animal studies [16]. In addition, several recent studies have focused on the levels of ACR in vegetable crisps [17, 18, 19, 20]. Additionally, several studies have examined the impact of various cooking parameters on ACR formation in fried foods [21]. Although researchers have explored strategies to mitigate ACR formation during high-temperature cooking processes [16, 22, 23], few studies are comprehensive and seldom include non-traditional food ingredients used in snack production, as well as their impact on ACR levels in the final product [23, 24]. This research provides an overview of the current ACR content in three fried snacks popular in the Costa Rican industry, and, especially, how the chemical nature of the different plant-based raw materials used in the preparation of such foods directly determines ACR levels in the crisp products.
The analysis of the incidence of ACR content in these three fried crisps produced in Costa Rica was carried out with the collaboration of three companies (coded as A, B, and C) specialized in the production of fried potato (companies A, B, and C), plantain (companies A and B), and cassava crisps (company A). Each company provided three different batches (1 kg per batch) of raw materials and their corresponding finished products, along with information on the frying process. The crop varieties used by the companies were the same: for potatoes (Solanum tuberosum L.), the Floresta and Única varieties; for plantains (Musa paradisiaca L.), the Curare variety; and for cassava (Manihot esculenta Crantz), the Valencia and Señorita varieties. The samples were delivered to the laboratory frozen in coolers and were kept at –20 °C until processing.
For the frying treatment, all three companies used the immersion method in palm olein. The frying temperatures vary depending on the vegetable type and shape: for corrugated potato crisps, 165 °C; for sliced potatoes, 170 °C; and for potato sticks, 180 °C for 4–5 minutes. For plantains (“patacón”, unripe and ripe slices), a pre-frying step was performed for 1–1.5 minutes at 160–170 °C, followed by a final frying step at 175–185 °C for 3.5–4.0 minutes. In the case of cassava (“patacón” and slice), the frying lasted 2–3 minutes at 170–180 °C. Following the frying process, the products were cooled on conveyor belts with air circulation before packaging. Samples from each batch were delivered to the laboratory in their respective packaging and are stored at 25 °C until processing.
The analysis of ACR content in commercially available crisps was conducted on a total of 54 crisp-type products sampled from various local markets in the Central Metropolitan Area. The samples were classified based on their matrix: potato (n = 24), plantain (n = 18), and cassava (n = 12). Each lot was subjected to three replicates, which were then homogenized using a food mill (Knife Mill Grindomix, Retsch GM 200, Haan, Germany) to create a composite sample. This composite sample was placed in vacuum-sealed, high-density polyethylene bags and stored at –20 °C until the ACR analysis could be performed.
Analytical standards as: ACR (catalog number 23701), ACR-2,3,3-d3
(catalog number 636568, 98 atom % D, D2C=CDCONH2, used as internal
standard), Fructose (catalog number PHR1002), dextrose (catalog number PHR1000),
sucrose (catalog number PHR1001-16), ASN (catalog number PHR1001-16) were
purchased from Millipore Sigma (Saint Louis, MO, USA). Sodium dihydrogen
phosphate monohydrate (NaH2PO4, catalog number 1.06346)
o-phthalaldehyde (OPA,
The raw samples (potato, plantain, and cassava) were homogenized in a food mill (Retsch GM 200, Germany). A portion of the fresh sample was immediately analyzed for water activity (aw) using AQUA LAB 4TE (Decagon Devices, Pullman, WA, USA). Moisture content was determined according to AOAC 964.22 [25], and pH was determined according to AOAC 981.12 [25]. The remaining portion was frozen (–20 °C) and then lyophilized (FreeZone 6, Labconco, Kansas, MO, USA).
The physicochemical analysis performed on freeze-dried material matter was total starch according to AOAC 996.11 [25] using the Total Starch Assay Kit purchased from Megazyme International Ireland Ltd. (Wicklow, Ireland), moisture content was determined using TGA701 (LECO, Saint Joseph, MI, USA) at 100 °C, and the sugar profile and free ASN content.
Each company sent ca. 250 g of crisps (potato, plantain, and cassava). Three replicates were taken from each batch and homogenized in a food mill (Retsch GM 200, Germany) to obtain a composite sample. The sample was placed in vacuum-packed high-density polyethylene bags and stored at –20 °C until further analysis (including aw, moisture, fat content, sugar profile, free ASN, and ACR content). Physicochemical analyses were performed: aw using AQUA LAB 4TE (Decagon Devices, WA, USA). Moisture content was determined using TGA701 (LECO, Saint Joseph, MI, USA) at 100 °C, and fat content was determined according to AOAC 920.85 [25].
A sugar profile was analyzed according to Sullivan and Carpenter [26] with the following modifications: a 3.0 g subsample of freeze-dried material was extracted with 30 g type I water. The mixture was stirred for 20 min, centrifuged (Sorvall™, ST16R, catalog number 75007203, ThermoFisher Scientific, Waltham, MA, USA) for 10 min at 4000 rpm. A 2 mL aliquot was filtered through a syringe filter (catalog number 1751AQ, Minisart®, Polytetrafluorethylene (PTFE), Pore Size 0.2 µm, Göttingen, Germany) and finally transferred to a HPLC vial (2 mL, Type 1 borosilicate amber glass, PTFE/silicone screw cap and septa, catalog number 5182-0716, Santa Clara, CA, USA).
For the crisps, five grams were extracted with 30–50 mL hexane on an orbital shaker (KS 130 Basic, IKA™, Staufen, Germany) for at least 10 min. The sample was centrifuged for 10 min at 4000 rpmand the organic phase was discarded. The process was repeated once more. Hexane remnant was evaporated in a convection oven at 60 °C until the solvent odor was no longer perceptible. To the fat-free residue, 30 g type I water was added and shaken for 5 min at 300 rpm. Later, the mixture was centrifuged for 10 min at 4000 rpm. A 2 mL aliquot of the supernatant was filtered through a 0.2 µm pore size syringe filter into an HPLC vial.
The HPLC analysis was performed with a Shimadzu HPLC system (Shimadzu, Kyoto,
Japan) equipped with a refractive index detector (RID-10A), column compartment
(CTO-20A), autosampler (SIL-20A HT), and a quaternary pump (LC-20AT). An amino
column (Zorbax Carbohydrate 5 µm, 150 mm
The method was based on Žilić and coworkers [27] with the following modifications: Two grams of the previously homogenized crisps or a subsample of freeze-dried material was weighed in centrifuge tubes (50 mL, CLS430829, polypropylene, Corning®, Corning, NY, USA), 20 mL of a phosphate buffer (NaH2PO4, 40 mmol L-1, pH = 7.8) was added and was stirred for 20 minutes at room temperature in an orbital shaker. Then the mixture was centrifuged at 4500 rpm for 10 minutes. A 5 mL sample of the supernatant was filtered through a 0.20 µm PTFE micropore, 10 µL was taken into a 2 mL vial, 100 µL of o-phthalaldehyde reagent (prepared as follows: 10 mg OPA, 20 µL of mercaptoethanol in 10 mL of borate buffer 50 mmol L-1, pH = 10) was added, along with 130 µL of borate buffer (50 mmol L-1, pH = 10) and 260 µL of water type I.
The analytical determination for free ASN was performed using HPLC system
(Agilent 1260 infinity, Agilent Technologies) equipped with a quaternary pump
(61311C), a column compartment (kept at 25 °C during analysis, G1316A),
a fluorescence detector (G1321C) and an automatic liquid sampler module
(injection volume set at one µL, ALS, G7129A) prepared with a reverse phase
chromatographic column (Zorbax Eclipse AAA column 75 mm
Two grams of the previously homogenized crisp samples were weighed in a centrifuge tube (50 mL, polypropylene, conical bottom, Corning®). Afterward, 10 mL of hexane was added to the mixture, which was then shaken for 5 min using a vortex (SI™ Vortex-Genie™ 2, Scientific Industries Inc., Bohemia, NY, USA). The mixture was centrifuged for 10 min at 4000 rpm, and the final hexane supernatant was discarded. This same defatting process was performed once more. To the residue, 10 mL of distilled water and 100 µL of the internal standard solution were added. A 10 mL sample of acetonitrile was added, and the solution was stirred for 30 min in an orbital shaker (KS 130 Basic, IKA™, Staufen, Germany). 4 g of anhydrous Na2SO4 and 0.5 g of NaCl were added, and the solution was stirred for 10 more min. The solution was centrifuged at 4 °C for 8 min at 5500 rpm, 1.00 mL of the upper liquid phase was taken and filtered through a 0.20 µm micropore (regenerated cellulose, catalog 18407, Sartorius, Gotinga, Germany) into a GC/MS-ready vial (2 mL vials and 11 mm aluminum crimp vial caps, red silicone/clear PTFE seal, catalog number 5190-9045, Agilent Technologies)
The ACR analysis was performed using an Agilent 7820A gas chromatograph (Agilent
Technologies) coupled with a 5977B mass spectrometry detector and equipped with a
30 m
The Kruskal-Wallis test was used to assess significant differences between types
of crisp within each category. ANOVA post hoc Dunnett’s test was performed to
compare potato crisps ACR values to the benchmark value of 750 µg
kg-1. Spearman’s rank order test was used to evaluate the association
between ACR (µg kg-1 dry basis) and their respective
precursors (ASN and reducing sugars on a dry basis) found in the raw materials
when compared to the crisp products; in this case, the pair of variables with
positive correlation coefficients and p values below 0.05 tend to
increase together. All statistical tests were performed using a threshold of
The Latin American Nutrition and Health Study (ELANS) [28] provided food consumption data for the Costa Rican population. The dataset comprised n = 798 individuals aged 15 to 65 years, consisting of n = 404 women and n = 394 men. For analysis, the data were stratified by gender and then by age group. ADE from crisp product consumption in Costa Rica was estimated using the following equation,
with results expressed in micrograms per kilogram of body weight per day (µg kg-1 bw day-1) for each person. RI represents the daily intake (g day-1) of crisp products, averaged over a two-day consumption period. C means the average ACR concentration in each crisp product (µg kg-1). Bw is the individual’s body weight (kg). The average dietary exposure to ACR was calculated for each gender and age group.
The three raw vegetable samples exhibited an optimal pH range of 4–6. Following
the frying process, the final product exhibited an ideal water activity (aw)
of below 0.3. The precursor content within the potato varieties utilized by the
Costa Rican industry is consistent with findings reported by several authors
[29, 30, 31]. These studies showed that reducing sugar content is considerably lower
than asparagine content. Asparagine, specifically, is recognized as the primary
precursor for the formation of induced ACR in fried potato products, and its
formation can be mitigated by reducing the initial ASN concentration [32]. A
positive correlation was observed between the formation of ASN and ACR after
frying the raw potato samples. Significant relationships were found between
precursor ASN and ACR content in potato products. A Spearman’s rho of 0.829
(p
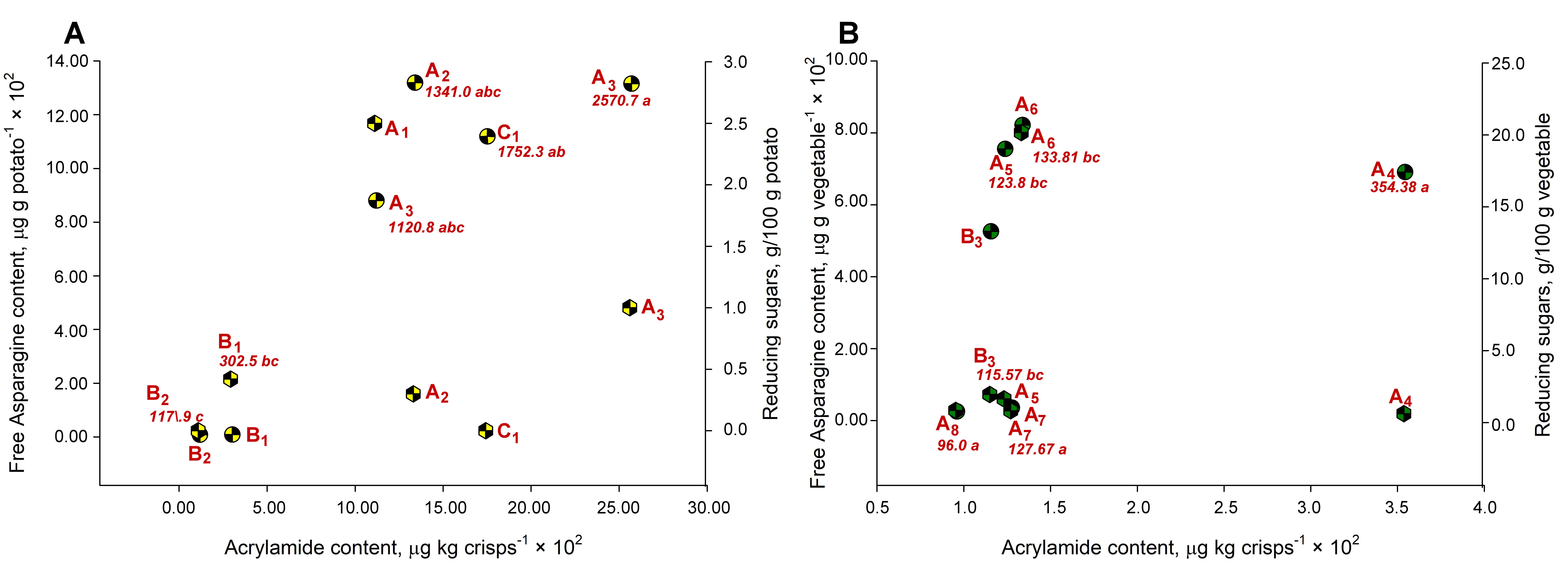 Fig. 1.
Fig. 1.
Multiple-axis chart showing the spatial distribution among three simultaneous variables, acrylamide (ACR) and two different precursors in fried crisps. Correlation between the acrylamide (ACR) content in fried chips
(A: potato; B: plantain and cassava) and the content of precursors asparagine
(asparagine (ASN), hourglass circles) and reducing sugars (
Bassama and coworkers [33] reported undetectable levels of up to 321.5 mg
ASN/100 g (dry basis) in a wide variety of plantain cultivars, noting that this
precursor decreases with increasing postharvest time. In the case of the plantain
chips evaluated, the asparagine content of the Curare variety was very similar to
that of the Harton variety [32]. There are no significant differences in the ASN
values between potatoes and plantains (p
Cassava slices presented significantly lower ASN content (p
No significant relationship between fructose or glucose content and ACR in any
of the vegetable crisps evaluated (Tables 1,2,3 and Fig. 1A,B, p
| Parameter | Material | Potato crisp presentation | |||||
| Corrugated | Slice | Stick | |||||
| A1 | B1 | A2 | B2 | C1 | A3 | ||
| pH | Raw | 5.80 |
5.81 |
5.59 |
5.83 |
5.86 |
5.88 |
| Starch (g 100 g–1) | Raw | 11.0 |
10.1 |
10.88 |
11.2 |
13.8 |
10.9 |
| Asparagine (µg g–1) | Raw | 153 |
1.57 |
217 |
1.75 |
225 |
224 |
| Moisture (g 100 g–1) | Raw | 82.3 |
81.7 |
84.7 |
81.67 |
80.67 |
83.7 |
| Crisps | 2.45 |
1.27 |
2.98 |
1.92 |
2.05 |
3.1 | |
| aw | Raw | 0.9960 |
0.9943 |
0.9982 |
0.9948 |
0.9967 |
0.9955 |
| Crisps | 0.239 |
0.0847 |
0.241 |
0.139 |
0.074 |
0.23 | |
| Glucose (g 100 g–1) | Raw | (ND |
ND |
(ND |
ND |
0.155 |
(ND |
| Crisps | (ND |
ND |
ND |
ND |
(ND |
ND | |
| Fructose (g 100 g–1) | Raw | (ND |
0.079 |
ND |
ND |
0.040 |
(ND |
| Crisps | (ND |
ND |
ND |
ND |
ND |
ND | |
| Sucrose (g 100 g–1) | Raw | (ND |
0.41 |
(ND |
0.31 |
0.036 |
(ND |
| Crisps | (NC |
ND |
(ND |
ND |
(NC |
ND | |
| Fat (g 100 g–1) | Crisps | 36.1 |
41.0 |
29.5 |
30.8 |
27.4 |
34.83 |
| Acrylamide (µg kg–1) | Crisps | 1093 |
298 |
1301 |
116 |
1716 |
2491 |
ND, not detectable; NC, not quantifiable. Three different Costa Rican companies coded as A, B and C.
| Parameter | Material | Plantain crisp presentation | |||
| “Patacón” | Unripe slice plantain | Ripe Slice plantain | |||
| A4 | B3 | A5 | A6 | ||
| pH | Raw | 5.63 |
6.05 |
5.23 |
4.78 |
| Starch (g 100 g–1) | Raw | 20.1 |
23.8 |
22.5 |
19.7 |
| Asparagine (µg g–1) | Raw | 268 |
216 |
298 |
323 |
| Moisture (g 100 g–1) | Raw | 62.7 |
60.4 |
61.2 |
62.1 |
| Crisps | 3.21 |
1.36 |
3.88 |
4.02 | |
| aw | Raw | 0.9937 |
0.9931 |
0.9932 |
0.9761 |
| Crisps | 0.199 |
0.0775 |
0.2406 |
0.2713 | |
| Glucose (g 100 g–1) | Raw | 0.157 |
0.57 |
0.38 |
4.09 |
| Crisps | ND |
ND |
ND |
(ND | |
| Fructose (g 100 g–1) | Raw | 0.39 |
0.229 |
0.51 |
3.77 |
| Crisps | ND |
ND |
ND |
(ND | |
| Sucrose (g 100 g–1) | Raw | 0.71 |
0.65 |
0.79 |
0.93 |
| Crisps | NC |
ND |
(ND |
9.61 | |
| Fat (g 100 g–1) | Crisps | 36.03 |
26.4 |
19.2 |
24.5 |
| Acrylamide (µg kg–1) | Crisps | 343 |
114 |
120 |
128 |
ND, not detectable; NC, not quantifiable. Three different Costa Rican companies coded as A, B and C.
| Parameter | Material | Cassava crisp presentation | |
| “Patacón” | Slice | ||
| A7 | A8 | ||
| pH | Raw | 5.49 |
5.32 |
| Starch (g 100 g–1) | Raw | 33.9 |
33.7 |
| Asparagine (µg g–1) | Raw | 13.9 |
9.93 |
| Moisture (g 100 g–1) | Raw | 60.8 |
60.6 |
| Crisps | 3.66 |
2.08 | |
| aw | Raw | 0.9945 |
0.9943 |
| Crisps | 0.2287 |
0.0848 | |
| Glucose (g 100 g–1) | Raw | 0.37 |
0.23 |
| Crisps | ND |
ND | |
| Fructose (g 100 g–1) | Raw | 0.48 |
0.51 |
| Crisps | ND |
ND | |
| Sucrose (g 100 g–1) | Raw | 1.72 |
1.67 |
| Crisps | 1.33 |
(NC | |
| Fat (g 100 g–1) | Crisps | 27.63 |
21.8 |
| Acrylamide (µg kg–1) | Crisps | 123 |
94 |
ND, not detectable; NC, not quantifiable.
The surface-to-volume ratio (SVR) seems to play a crucial role in ACR formation.
The potato product with the highest ACR values (p
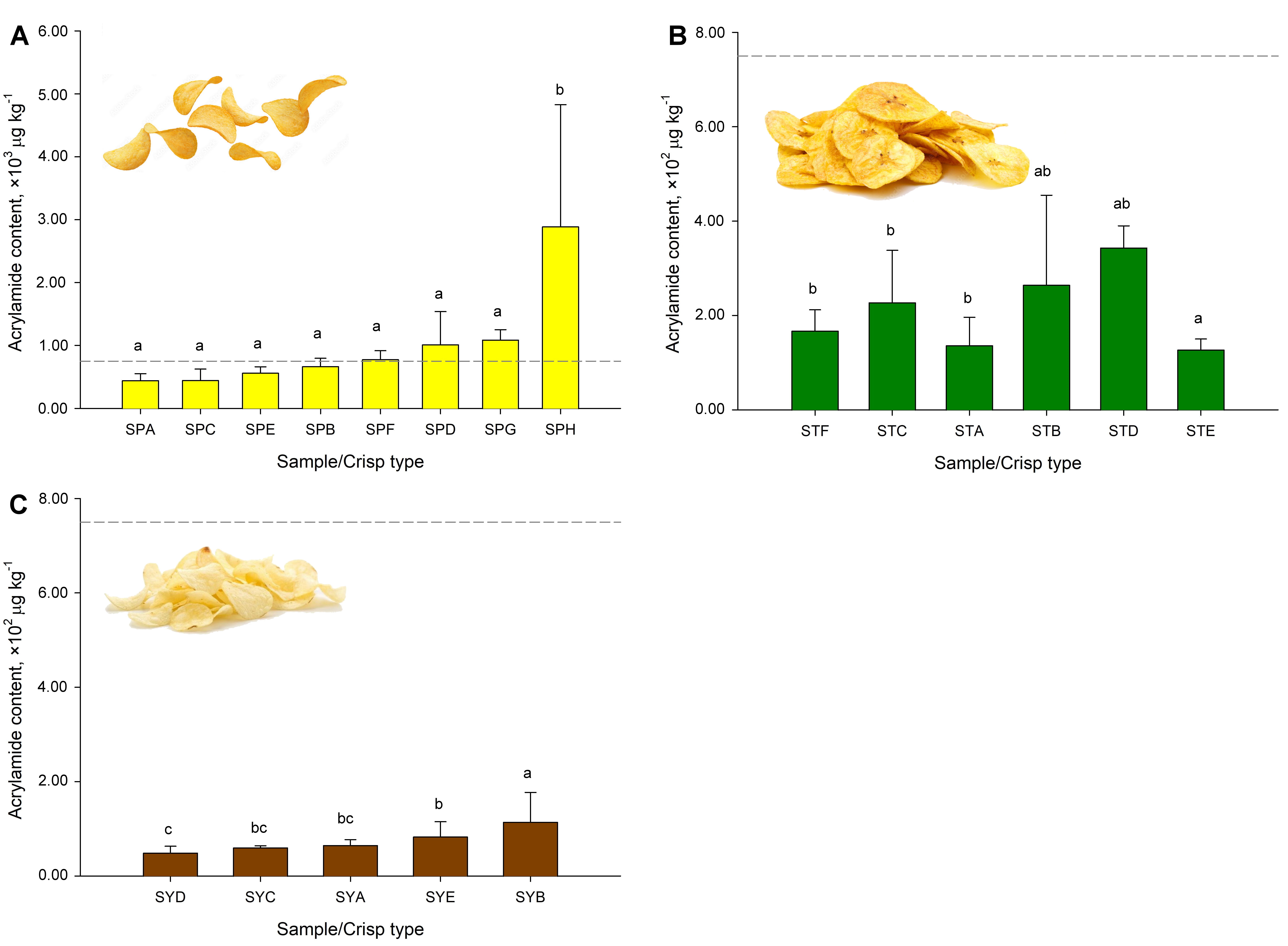 Fig. 2.
Fig. 2.
Acrylamide (ACR) formation during the frying process of different snacks, each colored bar represents a distinct matrix or sample presentation. Acrylamide (ACR) content in (A) sliced potato crisps (SPA, SPB,
SPC, SPE, SPF), potato corrugated crisps (SPG) and stick-cut potato crisps (SPH),
(B) unripe-sliced plantain crisps (STA, STB, STC, STD), unripe plantain
“patacón” (STE), ripe-sliced plantain crisps (STF), (C) sliced cassava
crisps (SYA, SYC, SYD, SYE), cassava “patacón” (SYB), commercially
available in Costa Rica with respect to the benchmark of 750 µg
kg-1. Dissimilar lowercase letters (a, b, c) represent significant
differences at p
In the case of plantain crisps, it was observed that product “patacón”
(Table 2 A4, Fig. 2B STE), shaped (larger surface area and volume),
presented a higher amount of fat, and it’s the product with the highest ACR
values (p
It was observed (Fig. 2A) that the maximum ACR values for potato crisps are
ten-fold higher (p
The ACR content in potato crisps produced in Costa Rica is similar to, or even
lower than, that found in other locations. According to Mesias and coworkers [21]
although the average ACR content found in 2019 in potato snacks marketed in Spain
(664 µg kg-1, range 89–1930 µg kg-1) has
decreased over the years (55.3% lower than in 2004 and 10.3% lower than in
2008), 27% of the samples still exhibited concentrations above the reference
level established in the Regulation (750 µg kg-1), suggesting
that efforts to reduce ACR formation in this industry should be continued.
Data from Ethiopia showed maximum values of ACR reported in potato crisps as high
as 3515 µg kg-1 [42], Pakistan´s products
reported 2649.8
Unlike potato crisps, the “alternative” tubers used in this experiment
(plantain and cassava) showed relatively the lowest ACR production (Fig. 2B,C).
The unripe plantain-based products (STA, STB, STC, STD) had ACR levels ranging
from 137 µg kg-1 to 265 µg kg-1, which are
comparable to previous findings in Malaysian (28–243 µg ACR
kg-1) [46] and Colombian products (130.4 µg ACR kg-1)
[47]. In ripe plantain, the average ACR level was 128
The average ACR content in the cassava products processed in this study was 75.2 µg/kg, a value similar to the 59.42 µg/kg reported by Díaz-Ávila and coworkers [48]. A survey by González-Cuello and coworkers [51] found a significantly higher maximum ACR content of 101.10 mg/kg when cassava chips were fried at 180 °C for 5 minutes. These types of products are a good alternative for consumers who prefer fried chips and have a low ACR content [52].
The synthesis of ACR in foods is dependent on a myriad of factors (including cooking temperature, time, pH, surface/area ratio, moisture content, cooking method, use of fertilizer, harvesting, cultivar, and storage conditions, to name a few), which may or may not be easy to manipulate or even assess. Hence, ACR behavior is complex, and even toxin bioavailability comes into play when considering exposure [18]. Interestingly, recent findings support a deleterious impact of ACR within the gastrointestinal tract [53].
As reported in a consumption survey conducted by the Latin American Nutrition and Health Study (ELANS) [28], among n = 798 individuals aged 15 to 65 years. Only 19.55% of this population consumes fried crisps (Fig. 3A,B, Ref. [28]). The results revealed that corn-based crisps are the most consumed (61.53%), followed by potato-based crisps (19.23%), then plantain-based crisps (12.82%), and finally cassava-based crisps (6.41%). It was observed that the portion of crisps varies considerably by gender and age (Fig. 3A,B). In the case of potato crisps, both men and women consume a daily portion of 30 g, whereas for plantains, the average daily intake increases to 70 g per day (Fig. 3A,B). However, men consume a larger portion (77 g per day) than women (64 g per day). Unlike cassava crisps, women consume more (92 g per day) than men (55 g per day; Fig. 3A,B).
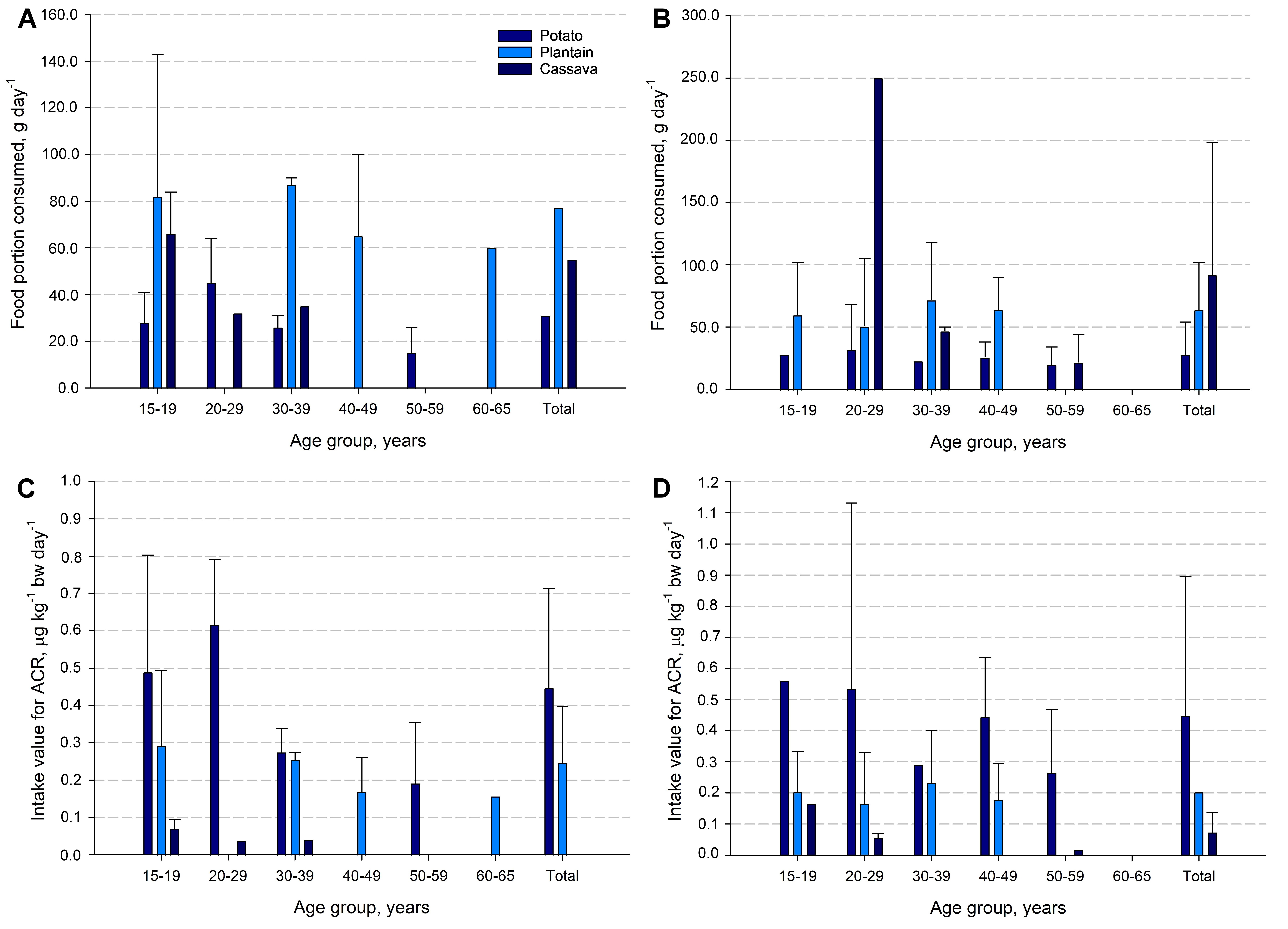 Fig. 3.
Fig. 3.
General population consumption practices in Costa Rica regarding snack foods. Crisps product consumption by gender and age (A: men and B: women) according to the study Latin American Nutrition and Health Study (ELANS) [28], and estimated acrylamide exposure value (ng kg-1 bw day-1) by crisps products consumption trends among the local population for Costa Rican (C: men and D: women).
The exposure value of ACR (ng kg-1 bw day-1) in the population
consuming the products under study was obtained taking into account gender and
age, and the results are presented in Fig. 3C,D. An average of 447
This preliminary investigation analyzed the chemical safety of fried snacks on the Costa Rican market. Our findings indicate that although potato chips produced the highest quantities of ACR upon frying, only one of the products examined statistically surpassed the EU-established maximum levels. This was likely attributable to its high surface-to-volume ratio. These results underscore the importance of advising manufacturers to implement mitigation strategies, such as controlling the physical properties of the raw ingredients, to reduce ACR formation and guarantee product safety. Moreover, plantain and cassava present promising alternatives for fried snacks. Their inherent asparagine content and processing methods result in significantly lower ACR levels compared to those found in potato-based products. Despite this, further research is warranted to optimize production conditions. The estimated daily ACR intake from these snacks alone suggests a potential health risk for the Costa Rican population, given their consumption habits and typical portion sizes.
The datasets generated and analyzed during the present study are available upon reasonable request. The data is not publicly available due to privacy and ethical restrictions.
Conceptualization, CCH and MQV; Data curation, MQV, CCH, and FGC; Formal analysis, MQV, CCH, FGC, and GA; Funding acquisition, CCH and GA; Investigation, MQV, CCH, FGC, and GA; Methodology, CCH, FGC, and GA; Resources, CCH and GA; Supervision, CCH, FGC, and GA; Writing—original draft, CCH and FGC. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
The authors wish to express their profound gratitude to all institutions and individuals who provided essential support for this research project. Specifically, we acknowledge the Chemistry Laboratory of the National Center for Food Science and Technology (CITA), UCR, for their invaluable technical support in chemical analysis. We also extend our sincere thanks to the companies involved in this study, whose cooperation was crucial in providing necessary information on the frying process, as well as the raw materials and final products utilized in the research. Furthermore, we recognize the support of Laura Arroyo in the development of student Quesada’s final graduation project. Finally, we would like to thank Georgina Gómez-Salas for providing us with access to information on food consumption among the Costa Rican population according to the ELANS study.
This work was supported by the Vicerrectoría de Investigación of the Universidad de Costa Rica [grant number C0023].
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



