1 Department of Obstetrics and Gynecology, Umraniye Training and Research Hospital, 34764 Umraniye, Istanbul, Turkey
2 Department of Perinatology, Umraniye Training and Research Hospital, 34764 Umraniye, Istanbul, Turkey
Abstract
Background: Coronavirus disease 2019 (COVID-19) infection is associated
with increased perinatal morbidity and mortality. Despite this, vaccination
hesitancy among pregnant women remains to be a huge problem. In this study we
aimed to evaluate COVID-19 vaccination safety and uptake in pregnancy, and to
evaluate adverse perinatal outcomes associated with COVID-19 in pregnancy,
compared with non-infected pregnant women. Methods: This is a
retrospective single-institution cohort study of women who gave birth in 2021.
The primary outcomes of the study were the safety of COVID-19 vaccination during
pregnancy, and the comparison of perinatal outcomes in COVID-19 infected women
during pregnancy group vs non-infected. The secondary outcomes included
vaccine uptake rate, trimester evaluation of vaccination safety, and side
effects. Results: Among 3620 deliveries, 1943 individuals who were
vaccine eligible before delivery were included in the vaccine safety and uptake
analysis. Out of 1943 pregnant women, 212 (10.9%) women received at least one
dose of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) vaccine
during pregnancy. 189 (89.2%) of them received BNT162b2 (Pfizer/BioNTech) and 23
(10.8%) CoronaVac (Sinovac). Following propensity score matching, no significant
increase in emergency cesarean section (C-section) rates, intrapartum complications, stillbirth,
congenital anomalies, maternal or neonatal intensive care unit (NICU) admissions
were found in COVID-19 vaccinated group compared to the non-vaccinated
(p
Keywords
- COVID-19
- inactivated vaccine
- mRNA vaccines
- pregnancy outcome
- safety
- SARS-CoV-2
At the beginning of coronavirus disease 2019 (COVID-19) pandemic it was unclear whether pregnant women were more susceptible to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Recent studies show that COVID-19 infection is associated with increased perinatal morbidity and mortality. However, which negative perinatal outcome the infection is associated with is still controversial. In a study published in 2021, it was shown that the severe perinatal morbidity and mortality rate was more than twice as high in COVID-19 infected women [1]. Another study reported the risk of neonatal adverse outcome was 1.45 times increased, while rates of emergency cesarean section (C-section), preeclampsia, and preterm birth were found to be increased [2]. In a meta-analysis, preeclampsia rates were found to be increased 1.42 times, with neonatal intensive care unit (NICU) admissions being increased 1.86 times. No differences in maternal hemorrhage, placental abruption, or emergency C-section were noted [3]. Chmielewska et al. [4], found that maternal mortality and stillbirth were increased when interpreting data from low-middle income countries. However, they found no overall significant effect on other adverse perinatal outcomes in a meta-analysis in which they suggested that the increased rate of adverse outcomes might be driven by the inefficiency of the health-care system. Therefore, public health measures, including vaccination and disease management are crucial to protect both the pregnant women and their fetuses.
Although the long-term safety profile of COVID-19 vaccination regarding pregnant women is lacking, accumulated data suggest that the known risks of the infection outweigh the potential unknown risks of the vaccination [5]. Vaccination hesitancy among pregnant women is high, which is contradictory to the recommendations of healthcare professionals about susceptible populations [6, 7]. The American College of Obstetricians and Gynecologists (ACOG) and the Society for Maternal Fetal Medicine (SMFM) recommend that COVID-19 vaccines should not be withheld on the basis of pregnancy [8]. In a study including 1328 pregnant women eligible for a COVID-19 vaccine, it was shown that only 28.5% accepted vaccination during pregnancy [9]. Even in a large population-based cohort study including 97,590 individuals, vaccination in pregnancy was reported as low as 23% [10]. From a national study at the beginning of 2021, the intention of pregnant women to get vaccinated was reported to be around 37% if it was recommended during pregnancy [11].
In this study, we aimed to investigate the uptake rate and safety of COVID-19 vaccines, and to evaluate the perinatal outcomes of COVID-19 infection among pregnant women in comparison to non-infected pregnant women.
This was a retrospective cohort study of pregnant women who gave birth in Umraniye Training and Research Hospital, Department of Obstetrics and Gynecology, Turkey, between 01/01/2021 and 12/31/2021. Data was collected from the hospital’s electronic health information system. Women with unknown last menstrual period or no first trimester scanning were excluded.
Maternal age, ethnicity, parity, obstetric history, pregestational and gestational diseases, and obstetric outcomes were recorded. COVID-19 infection and vaccination status of the participants along with local and systemic side effects due to vaccination during pregnancy were obtained from the national public health management system. COVID-19 infection was confirmed with a polymerase chain reaction (PCR) test at the time of admission to labor and delivery ward or at the time of suspected infection. Vaccine types available in our country were messenger RNA (mRNA) BNT162b2 (Pfizer/BioNTech) and inactivated vaccine CoronaVac (Sinovac).
According to the national vaccination program in our country, by June 24, 2021, all pregnant women were eligible for COVID-19 vaccination of their own choice, including CoronaVac and BNT162b2. Therefore, pregnant women who gave birth after vaccine eligibility were included for statistical analysis of vaccination safety and side effects.
The safety of COVID-19 vaccination, and comparison of perinatal outcomes in
COVID-19 infected and non-infected groups during pregnancy, were the primary
outcomes of the study. Perinatal outcomes both to assess the safety of
vaccination and for the comparison of COVID-19 infection effect included
birthweight, mode of delivery, stillbirth (fetal death
Secondary outcomes of this study were vaccine uptake rates, trimester evaluation of the perinatal outcomes, and side-effects among the vaccinated group.
We performed normality tests of the distribution of continuous variables via Kolmogorov-Smirnov test. For the distribution of the quantitative variables which did not fit the normal distribution, the Mann-Whitney U test was used to determine the significance of the difference between two independent groups. For categorical variables, we used Pearson Chi-square, Continuity Correction (Yates Chi-square) or Fisher’s Exact test for the significance of the difference in the rates between two independent groups. Here, we made the selection according to the expected frequencies and we made evaluations for all variables separately.
For the comparison of perinatal outcomes between antenatally COVID-19 vaccinated
and non-vaccinated individuals, we used propensity score matching to eliminate
potential confounders. We determined potential covariates considering the factors
identified in group comparison with the p-values of
For trimester adverse effect outcome analysis, we performed One-Way Analysis of Variance (ANOVA) test for the significance of the difference between the means of three independent samples, which were normally distributed. For other variables, we applied non-parametric Kruskal Wallis H test.
We used R statistical analysis software (R Project 4.1.1, R Foundation for Statistical Computing, Vienna, Austria) for all statistical analyses.
A total of 3806 pregnant women were eligible for the study. The study flow diagram is provided in Fig. 1. 186 individuals were excluded due to missing data, vaccination before pregnancy, or history of COVID seropositivity before pregnancy. There were 3620 deliveries in 2021 which were included in the study, in Umraniye Training and Research Hospital, Department of Obstetrics and Gynecology. For the vaccine safety and uptake analysis, 1943 individuals who were vaccine eligible before delivery were included.
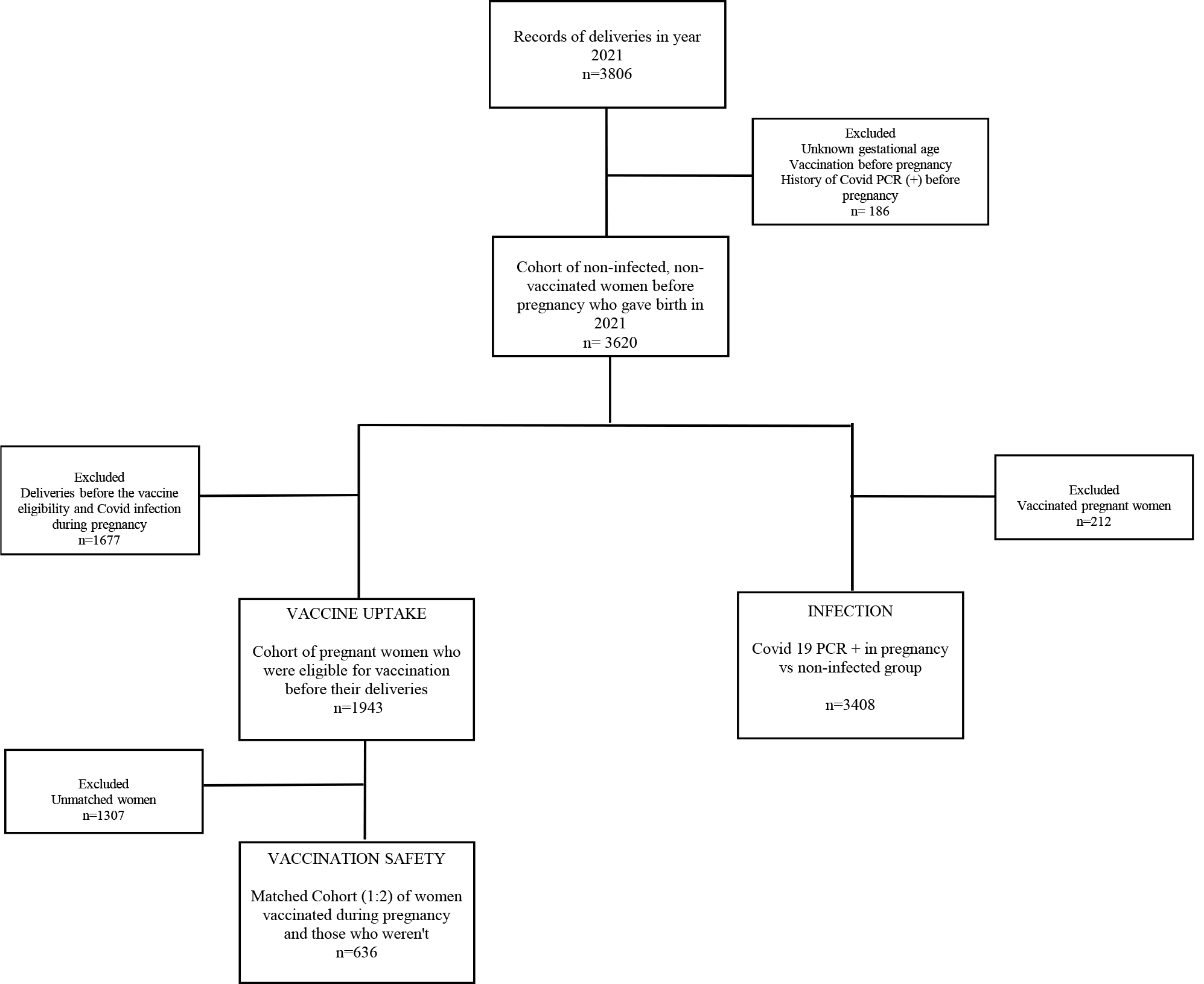 Fig. 1.
Fig. 1.Study flow diagram. COVID-19, coronavirus disease 2019; PCR, polymerase chain reaction.
Out of 1943 women, 212 (10.9%) received at least 1 dose of SARS-CoV-2 vaccine
during pregnancy. 1731 non-vaccinated pregnant women and 212 vaccinated women
were compared in terms of demographic characteristics and perinatal outcomes. The
baseline characteristics of the 2 groups are given in Table 1. Among the
vaccinated group, 189 pregnant women received BNT162b2 (89.2%), and 23 received
CoronaVac (10.8%). The mean age of the vaccinated group was higher, compared to
the unvaccinated group (29.7 vs 27.7, p
| Received |
Did not receive any COVID-19 vaccine | p-value | SMD | ||
| n (%) | n (%) | ||||
| n | 212 (10.9) | 1731 (89.1) | |||
| Vaccine Type | 1.78 | ||||
| BNT162b2 (mRNA) | 189 (89.2) | - | - | - | |
| CoronaVac (inactivated) | 23 (10.8) | - | - | - | |
| Maternal Age (years) (mean) | 29.7 (5.8) |
27.7 (5.7) |
0.35 | ||
| Ethnicity | 0.666 | ||||
| Turkish | 209 (98.6) | 1359 (78.5) | |||
| Syrian | 2 (0.9) | 324 (18.7) | |||
| Others | 1 (0.5) | 48 (2.8) | |||
| Multiparous | 166 (78.3) | 1283 (74.1) | 0.216 | 0.098 | |
| Pregestational Maternal Systemic Diseases | 52 (24.5) | 333 (19.2) | 0.083 | 0.128 | |
| Hypertension | 6 (2.8) | 14 (0.8) | 0.017 | 0.152 | |
| Diabetes Mellitus | 6 (2.8) | 20 (1.2) | 0.092 | 0.12 | |
| Hypothyroidism | 25 (11.8) | 153 (8.8) | 0.200 | 0.097 | |
| Hyperthyroidism | 0 (0.0) | 3 (0.2) | 0.059 | ||
| Cardiac Disease | 1 (0.5) | 20 (1.2) | 0.578 | 0.076 | |
| Pulmonary Disease | 2 (0.9) | 34 (2.0) | 0.441 | 0.085 | |
| Renal Disease | 0 (0.0) | 8 (0.5) | 0.672 | 0.096 | |
| Autoimmune Disease | 5 (2.4) | 19 (1.1) | 0.215 | 0.097 | |
| Neurological Disease | 2 (0.9) | 23 (1.3) | 0.883 | 0.036 | |
| Infectious Disease | 11 (5.2) | 63 (3.6) | 0.356 | 0.075 | |
| Smoking during pregnancy | 27 (12.7) | 179 (10.3) | 0.342 | 0.075 | |
| Antenatal anticoagulant use during pregnancy | 29 (13.7) | 171 (9.9) | 0.110 | 0.118 | |
| Aspirin | 8 (3.8) | 35 (2.0) | 0.165 | 0.105 | |
| LMWH |
4 (1.9) | 36 (2.1) | 0.014 | ||
| Both | 17 (8.0) | 100 (5.8) | 0.253 | 0.089 | |
| Multiple Pregnancy | 6 (2.8) | 19 (1.1) | 0.073 | 0.125 | |
| Antenatal Complications | 55 (25.9) | 372 (21.5) | 0.165 | 0.105 | |
| Gestational Diabetes | 23 (10.8) | 113 (6.5) | 0.029 | 0.154 | |
| Gestational Hypertension | 2 (0.9) | 32 (1.8) | 0.502 | 0.077 | |
| Preeclampsia | 10 (4.7) | 44 (2.5) | 0.110 | 0.116 | |
| Cholestasis | 1 (0.5) | 15 (0.9) | 0.843 | 0.048 | |
| IUGR |
7 (3.3) | 65 (3.8) | 0.891 | 0.025 | |
| Preterm birth | 6 (2.8) | 43 (2.5) | 0.943 | 0.022 | |
Uptake of COVID-19 vaccination was higher among Turkish individuals compared to Syrian ethnic and other ethnic groups (13.3%, 0.6% and 2%). Among COVID-19 vaccine-eligible pregnant individuals 385 (19.8%) had a systemic disease, and 52 (13.5%) of these pregnant individuals had received COVID-19 vaccination during pregnancy. Antenatal vaccine uptake was higher among women with pregestational diabetes compared to the overall vaccination rate (23% vs 10.9%). The vaccination rate among 178 individuals with hypothyroidism was 14%, 14.5% among 200 individuals who were using anticoagulants, and 20.8% among individuals with an autoimmune disease. 13.1% of pregnant individuals who were smoking during pregnancy had received vaccination.
Following 1:2 propensity score matching, standardized differences less than 0.1
were balanced, and the overlap in distributions was confirmed. 212 women who
received at least one dose of COVID-19 vaccine in pregnancy were matched with 424
women who did not. There was no significant difference in mode of delivery,
intrapartum complications, or NICU admissions (p
| Received |
Did not receive any COVID-19 vaccine | Effect magnitude (95% CI |
p-value | ||
| n (%) | n (%) | ||||
| n | 212 | 424 | |||
| Emergency C-section |
60 (28.3) | 125 (29.5) | 0.95 (0.68–1.36) | 0.773 | |
| Maternal ICU |
0 (0.0) | 1 (0.2) | NE |
NE | |
| Birthweight (g) | 3100.63 (700.51) |
3228.19 (713.62) |
–130.5 (–245.98– –15.03) | 0.027 | |
| Apgar Score 5th min | 9.32 (0.94) |
9.09 (1.25) |
0.24 (0.05–0.44) | 0.013 | |
| Intrapartum Complications | |||||
| Fetal Distress | 11 (5.2) | 38 (9.0) | 0.54 (0.22–1.09) | 0.090 | |
| Preeclampsia | 6 (2.8) | 7 (1.7) | 1.85 (0.46–6.45) | 0.269 | |
| Placental Abruption | 0 (0.0) | 9 (2.1) | NE |
NE | |
| Chorioamnionitis | 1 (0.5) | 0 (0.0) | NE |
NE | |
| Stillbirth | 3 (1.4) | 5 (1.2) | 1.44 (0–9.29) | 0.639 | |
| Congenital Anomalies | 15 (7.1) | 42 (9.9) | 0.68 (0.43–1.15) | 0.132 | |
| NICU |
57 (26.9) | 99 (23.3) | 1.21 (0.87–1.76) | 0.259 | |
Covariates: Maternal age, smoking, antenatal anticoagulant use (aspirin, LMWH), maternal systemic disease (hypertension, pulmonary disease).
To determine the effect of COVID-19 infection in pregnancy on perinatal
outcomes, 212 vaccinated pregnant women were excluded from the 3620 pregnant
women without a history of COVID-19 infection or vaccination before pregnancy.
The baseline characteristics of the 3408 women were divided into two groups:
COVID-19 seropositive and negative, presented in Table 3. In the COVID-19
seropositive group, 102 (23.4%) pregnant women had a pregestational chronic
disease. Compared to the seronegative group, the seropositive group had
significantly higher pregestational hypothyroidism (7.1% vs 12.2%,
p
| COVID-19 PCR positivity during pregnancy | COVID-19 PCR negative | p-value | ||
| n (%) | n (%) | |||
| n | 436 (12.8) | 2972 (87.2) | ||
| Maternal Age (years) (mean) | 28.3 (0.27) |
27.7 (0.12) |
0.062 | |
| Ethnicity | ||||
| Turkish | 395 (90.6) | 2322 (78.1) | ||
| Syrian | 35 (8.0) | 577 (19.4) | ||
| Others | 6 (1.4) | 73 (2.5) | 0.017 | |
| Multiparous | 324 (74.3) | 2254 (75.8) | 0.487 | |
| Pregestational Maternal Chronic Diseases | 102 (23.4) | 507 (17.1) | 0.002 | |
| Hypertension | 4 (0.9) | 29 (1.0) | ||
| Diabetes Mellitus | 4 (0.9) | 32 (1.2) | 0.685 | |
| Hypothyroidism | 53 (12.2) | 210 (7.1) | ||
| Hyperthyroidism | 2 (0.5) | 8 (0.3) | 0.678 | |
| Cardiac Disease | 5 (1.1) | 36 (1.2) | ||
| Pulmonary Disease | 6 (1.4) | 56 (1.9) | 0.496 | |
| Renal Disease | 1 (0.2) | 9 (0.3) | ||
| Autoimmune Disease | 6 (1.4) | 12 (0.4) | 0.073 | |
| Neurological Disease | 2 (0.5) | 27 (0.9) | 0.615 | |
| Infectious Disease | 19 (4.4) | 89 (3.0) | 0.129 | |
| Smoking during pregnancy | 26 (6.0) | 321 (10.8) | 0.002 | |
| Antenatal anticoagulant use during pregnancy | ||||
| Aspirin | 14 (3.2) | 67 (2.3) | 0.177 | |
| LMWH |
11 (2.5) | 52 (1.7) | 0.177 | |
| Both | 28 (6.4) | 142 (4.8) | 0.377 | |
| Multiple Pregnancy | 3 (0.7) | 38 (1.3) | 0.412 | |
| Gestational age at delivery (week) | 38.44 (0.11) |
38.46 (0.04) |
0.954 | |
| Mode of Delivery | ||||
| Unassisted Vaginal | 212 (48.6) | 1610 (54.2) | 0.051 | |
| Emergency C-section | 101 (23.2) | 734 (24.7) | 0.366 | |
| Elective C-section | 123 (28.2) | 628 (21.1) | 0.053 | |
| Maternal ICU |
3 (0.7) | 8 (0.3) | 0.157 | |
| Maternal Death | 1 (0) | 0 (0) | NE | |
| NICU |
88 (20.2) | 547 (18.4) | 0.373 | |
| Antenatal Complications | 105 (24.1) | 634 (21.3) | 0.192 | |
| Birthweight (g) | 3178.32 (29.43) |
3180.3 (11.16) |
0.485 | |
| Apgar Score 5th min | 9.23 (0.07) |
9.26 (0.03) |
0.559 | |
| Congenital Anomalies | 19 (4.4) | 197 (6.6) | 0.069 | |
| Intrapartum Complications | ||||
| Fetal Distress | 34 (7.8) | 256 (8.6) | 0.582 | |
| Preeclampsia, Eclampsia | 6 (1.4) | 33 (1.1) | 0.638 | |
| Placental Abruption | 1 (0.2) | 31 (1.0) | 0.258 | |
| Chorioamnionitis | 0 (0) | 1 (0) | NE | |
| COVID-19 related C-section | 0 (0) | 1 (0) | NE | |
| Stillbirth = 1 (%) | 3 (0.7) | 10 (0.3) | 0.48 | |
In the subgroup analysis of 212 pregnant women vaccinated during pregnancy,
vaccine 1st dose uptake according to trimester was determined to be 3.3% in the
first trimester, 38% in the second, and 58% in the third trimester. Gestational
age distribution of vaccinations is shown in Fig. 2, and shows a very low
vaccination rate in the first trimester. All pregnant women in the first
trimester group received BNT162b2, whereas the ratios were 84.1% in the second
trimester, and 92.6% in the third trimester. There was no difference in 3 groups
in terms of maternal age, ethnicity, and pregestational maternal chronic diseases
(p
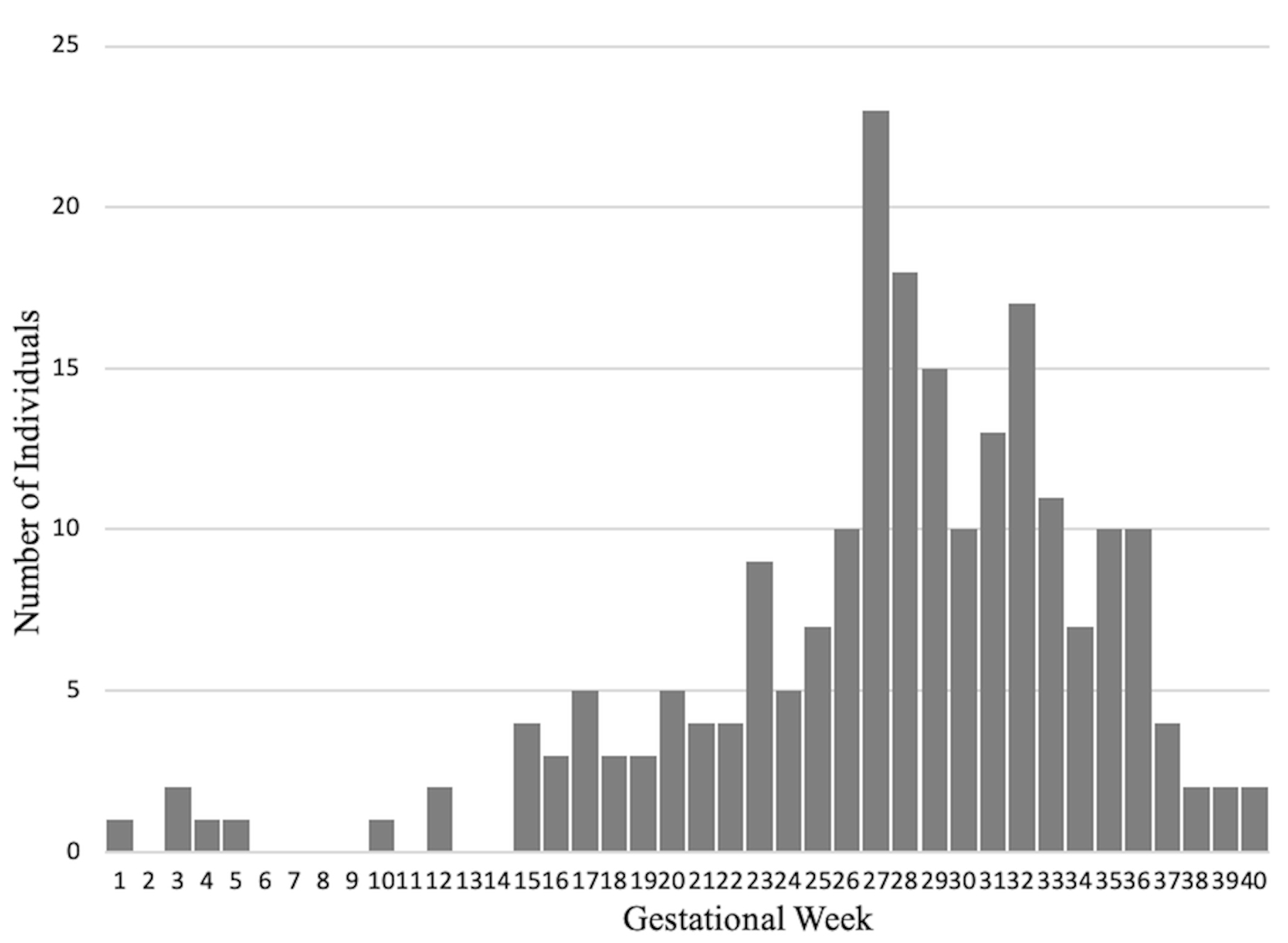 Fig. 2.
Fig. 2.Gestational age distribution at receipt of dose 1 during pregnancy.
| 1st Trimester | 2nd Trimester | 3rd Trimester | p-value | ||
| n (%) | n (%) | n (%) | |||
| n | 8 | 82 | 122 | ||
| Vaccine Type | 0.73 | ||||
| BNT162b2 (mRNA) | 8 (100) | 69 (84.1) | 113 (92.6) | ||
| CoronaVac (inactivated) | 0 (0) | 13 (15.9) | 9 (7.4) | ||
| Maternal Age (years) (mean) | 28.9 (3.87) |
30.3 (5.58) |
29.1 (5.94) |
0.34 | |
| Ethnicity | 0.91 | ||||
| Turkish | 8 (100) | 81 (98.8) | 120 (98.4) | ||
| Syrian | 0 (0) | 1 (1.2) | 1 (0.8) | ||
| Other | 0 (0) | 0 (0) | 1 (0.8) | ||
| Gestational age at vaccination 1st dose (week) | 8.73 (1.58) | 23.41 (0.37) | 32.04 (0.26) | ||
| Pregestational Maternal Chronic Diseases | 2 (25.0) | 23 (28.0) | 28 (23.0) | 0.63 | |
| Smoking during pregnancy | 3 (37.5) | 6 (7.3) | 15 (12.3) | 0.03 | |
| Birthweight (g) | 2305.45 (1109.40) |
3028.30 (793.30) |
3200.21 (537.92) |
0.02 | |
| Gestational age at birth (week) | 34.12 (7.47) | 37.26 (2.99) | 38.84 (1.69) | ||
| Congenital Anomalies | 1 (12.5) | 7 (8.5) | 4 (3.3) | 0.74 | |
| Maternal ICU |
0 (0) | 0 (0) | 0 (0) | NE | |
| Maternal Death | 0 (0) | 0 (0) | 0 (0) | NE | |
| NICU |
4 (50.0) | 27 (32.9) | 18 (16.4) | ||
| Local Side Effects after vaccination 1st dose | 3 (37.5) | 13 (15.9) | 20 (16.4) | 0.047 | |
| Systemic side effects after vaccination 1st dose | 2 (25) | 11 (13.4) | 15 (12.3) | 0.08 | |
| Fever | 1 (12.5) | 0 (0) | 2 (1.6) | 0.04 | |
| Fatigue | 1 (12.5) | 7 (8.5) | 6 (4.9) | 0.15 | |
| Myalgia-Arthralgia | 1 (12.5) | 5 (6.1) | 3 (2.5) | 0.04 | |
| Headache | 1 (12.5) | 0 (0) | 2 (1.6) | NE | |
| Vaginal Bleeding | 0 (0) | 0 (0) | 0 (0) | NE | |
| Uterine Contraction | 0 (0) | 0 (0) | 2 (1.6) | NE | |
Of the vaccinated women, 17% experienced local side effects and 13.2% systemic side effects from the vaccine. Local side effects included swelling and localized pain at the injection site and were reported to be similar between the second and third trimester groups (15.9% vs 16.4%). Fatigue and myalgia were the most common reported systemic side effects. Two pregnant individuals who received their first vaccine dose in third trimester described temporary uterine contractions. None of the women reported vaginal bleeding after vaccination.
In this study, BNT162b2 or CoronaVac vaccination during pregnancy was not associated with statistically significant adverse perinatal outcomes. These findings support the recommendation of vaccination in pregnancy, especially, during the second and third trimesters.
In a study conducted in Canada, Fell et al. [10] found that vaccination during pregnancy with Pfizer BioNTech BNT162b2, Moderna mRNA-1273, or AstraZeneca AZD1222 vaccines was not associated with an increased risk of post-partum adverse outcomes, such as postpartum hemorrhage, chorioamnionitis, cesarean delivery, admission to NICU, or low 5th minute Apgar score. In a different study, Magnus et al. [12] reported no significant increase in pregnancy complications among the vaccinated group compared to the unvaccinated group. The vaccine of choice among the vaccinated group was BNT162b2, and the majority of vaccinations were in the second and third trimesters. The findings of a meta-analysis by Prasad et al. [13] concluded that there was no increase in any adverse outcome examined for the mother or the baby. They stated that there was some evidence of benefit, such as a 15% decrease in stillbirths in the vaccinated group during pregnancy compared to the unvaccinated group [13]. These cumulative data are important to increase the vaccination rates in pregnancy throughout the world since the pregnant population is at a higher risk of severe COVID-19 infection [14]. Our findings are in concordance with these studies, which found no adverse perinatal outcomes following SARS-CoV-2 vaccines during pregnancy.
There are several well-known risk factors associated with mortality among COVID-19 infected individuals. Older age, diabetes mellitus and hypertension were reported to increase the risk of mortality [15, 16, 17]. In a meta-analysis including 33 country territories, pregnant women with comorbidities such as diabetes mellitus, hypertension, and cardiovascular disease were at increased risk for severe maternal and fetal COVID-19 related outcomes [3]. In our study, we found that pregnant individuals with comorbidities had received at least one dose of COVID-19 vaccine at a rate of 13.5%, higher than the overall vaccination rate, which was 10.9%. Pregnant women with pregestational diabetes, infectious diseases, autoimmune diseases, and who were smokers during pregnancy got vaccinated at a higher rate than the average. Whereas individuals with cardiac diseases had very lower vaccination rate in contrast to expectations.
COVID-19 vaccination campaigns have been carried out throughout the world, and in February 2022, according to United States data, it has been reported that 68% of pregnant individuals have completed their vaccine series [18]. A cross-sectional survey done in United States from August to December 2020, reported that only 41% of the pregnant participants were willing to get vaccinated for COVID-19 if they were eligible [19]. In a meta-analysis evaluating COVID-19 vaccine uptake rates and predictors of vaccination receipt, the overall COVID-19 uptake rate was found to be 27.5% [6]. The majority of the studies in this study were from high-income countries. The predictors of getting COVID-19 vaccination were reported to be older age, trust in COVID-19 vaccines, fear of COVID-19 infection during pregnancy, and pregestational diabetes [6]. A study including 623 pregnant women in Sudan found that only 2.7% of individuals were willing to receive COVID-19 vaccine during pregnancy [20]. In a meta-analysis including 17 studies in which 5 of them were from low-middle income countries, the overall vaccine acceptance rate was reported to be 49% [21]. In a global mapping study, including 519 articles, pregnant and breastfeeding individuals were reported to have the lowest vaccine acceptance and uptake rate of 54% and 7% respectively, compared to other population groups [22]. A study from our country, Turkey, found that 37% of pregnant women had an intention of receiving COVID-19 vaccine [11]. However, in our study we found a vaccine uptake rate of 10.9%.
Globally, there is a large gap between COVID-19 vaccine acceptance and uptake rates [22]. Vaccine hesitancy remains to be a huge problem. As Bhattacharya et al. [21] mentioned in their study, similar trends were observed in the uptake of influenza vaccines in the previous pandemic. In 2017, the most frequent barriers to pandemic influenza vaccine uptake for pregnant individuals included vaccine safety concerns, mistrust ineffectiveness of the vaccine and misconceptions about the vaccine [23]. A study from our country reported comparable reasons among pregnant women refusing to receive COVID-19 vaccines, including lack of data on the safety of the vaccine in pregnant population, possible harm to the fetus and mistrust in the efficacy of COVID-19 vaccines [11].
An understanding of the factors related to increased COVID-19 vaccine uptake in pregnant women is crucial to use appropriate communication tools. A national study pointed out that the ratio of pregnant women in the vaccine acceptance group who thought that they were informed adequately about the COVID-19 vaccine was significantly higher than in the vaccine refusal group [11]. Therefore, the main strategy to overcome vaccine hesitancy should be to inform target groups and provide necessary data regarding the safety and efficacy of COVID-19 vaccines in order to improve trust in the vaccines. Media sources and public information tools should be used effectively to increase the vaccination rates as these are the main channels where the vaccine acceptance group thought that they were informed adequately [11]. These highlight the need for persistent public education campaigns, promotion of the vaccine by health institutions, and a systematic monitorization of vaccine uptake among risk groups.
In a meta-analysis which included 111 studies, enrolling 42,754 COVID-19-positive pregnant women, the risk of low-birth weight, premature delivery, preeclampsia, and stillbirth was higher in COVID-19 seropositive pregnant women compared to non-infected. The authors concluded that there was no evidence of an increased maternal mortality in pregnant women infected with COVID-19 [24]. In parallel, we found no significant increased risk of preeclampsia, premature delivery, maternal or NICU admission in the COVID-19 infected group compared to the non-infected group.
A recent study with a large sample size of vaccinated pregnant women claimed that the women vaccinated in the second trimester were more likely to have a preterm birth compared to their nonvaccinated counterparts [25]. In contrast, in our trimester analysis we found out that the pregnant individuals vaccinated in the first trimester, delivered before term and had a lower birthweight. In a mega-cohort from Liu et al. [26], any women smoking during the three months prior to conception and who continued smoking into the first trimester of pregnancy was associated with increased preterm birth compared to nonsmokers. Accordingly, we believe that the high smoking rates we found in the first trimester group may have potentially contributed to the increased rates of preterm birth.
In a mini-review by Chen et al. [5], it was mentioned that higher placental antibody transfer ratio was associated with increasing duration between maternal vaccination and delivery. They summarized that maternal vaccination starting from the early second trimester might be an optimal time for newborn immunity against SARS-CoV-2 infection, since the placental antibody transfer begins from the 17th week of pregnancy. In contrast, a study evaluating maternal and cord blood antibody levels at birth after mRNA COVID-19 vaccination during the second trimester of pregnancy demonstrated that antibody levels fade in the maternal circulation as time passes, implying that the impact of antenatal vaccination timing may influence neonatal seroprotection. They concluded that a booster dose might be beneficial for those who completed the two-dose vaccine series before conception, or in the early gestational period [27]. In our study, we found that NICU admission was lower in the group who received the 1st dose of COVID-19 vaccine in the early third trimester compared to the groups who received the vaccine in the first or second trimester. Further studies on fading maternal antibody levels and its time course might be useful in order to interpret our findings.
To our knowledge this study is the first study that includes CoronaVac inactivated vaccine in the vaccinated pregnant group, comparing perinatal outcomes in two propensity score matched groups. Although CoronaVac is the first COVID-19 vaccine proposed in our country, by the time of eligibility for pregnant women, BNT162b2 and CoronaVac were the two COVID-19 vaccines available. We found out that the majority (89.2%) of women received at least one dose of BNT162b2 during their pregnancy. We believe that the scarcity of transparently published data on CoronaVac may have led to this result [28].
Data in our study are from a specific region, therefore generalizability may be limited compared to multicenter studies. Moreover, the sample size is small and although the rate of vaccination in second and third trimesters are similar, the rate in the first trimester remains too small for any further interpretation.
In this study, COVID-19 vaccination in pregnancy was not associated with significant adverse perinatal outcomes. Vaccine uptake rate was below the average global vaccine uptake rates. In order to increase COVID-19 vaccine uptake, policymakers and healthcare professionals should use effective media campaigns and public health messages to inform target populations about the safety of vaccination and reduce the mistrust in the efficacy of available vaccines. Overall, COVID-19 infection was not associated with increased adverse perinatal outcomes. Our results should be confirmed in a bigger cohort in order to draw more definite conclusions.
The data supporting the findings of this study is available via OSFHOME data repository https://osf.io/mjquk/ with DOI number 10.17605/OSF.IO/MJQUK.
MD, IK and MM designed the research question. AC, IU, ZS, SC, EY, CK joined MD and IK with drafting the manuscript and data acquisition. MD, IK and MM have done the critical review. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The Local Ethics Committee of Umraniye Training and Research Hospital, Istanbul, Turkey has approved this study (Ethics Committee Approval No: B.10.1.TKH.4.34.H.G.P.0.01/354). The study protocol was maintained in accordance with the Declaration of Helsinki, and informed consent was obtained from all the participants.
We would like to express our gratitude to all those who helped us during the writing of this manuscript. We’d like to thank Meral Yay Ph.D. from Mimar Sinan Fine Arts University, for her expertise and assistance in the statistical analysis of our study. Thanks to all the peer reviewers for their opinions and suggestions.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


