1 Department of Obstetrics and Gynecology, Faculty of Medicine, Alexandria University, 21521 Alexandria, Egypt
2 Department of Pathology, Faculty of Medicine, Alexandria University, 21521 Alexandria, Egypt
Academic Editor: Raffaele Tinelli
Abstract
Background: To test the accuracy of office hysteroscopy in the
diagnosis of chronic endometritis (CE) as compared to histopathological
diagnosis. Methods: This prospective cohort study was conducted in a
private hospital from July 2018 to January 2020. 220 infertile women were
scheduled for first attempt of intracytoplasmic sperm injection (ICSI). Patients
ranging in age from 20–40 with history of primary infertility scheduled for ICSI
for the first time were included, while patients with chronic diseases, severe
vaginal bleeding, previous failed ICSI, polycystic ovary syndrome (PCOS), or
endometriosis were excluded. All patients received postmenstrual office
hysteroscopy to rule out the presence of CE; at the same setting, endometrial
biopsy was taken and sent for histopathological examination. Results:
174 cases (79.1%) were diagnosed as CE at hysteroscopy, while 162 (73.6%) cases
were positive at histopathology. 99.4% of the cases presented with hyperaemia,
followed by oedema in 74.7% and micro-polypi in 58.6% of cases with CE. There
was a significant difference between the hysteroscopic and the histopathologic
diagnosis of CE (p
Keywords
- ICSI
- Office hysteroscopy
- Chronic endometritis
- CD138
- Plasma cells
Uterine factor infertility accounts for approximately 10–15% of infertile couples. Uterine anomalies may be either congenital or acquired [1]. Congenital uterine malformations vary from subtle anomalies such as arcuate uterus to a major anomaly such as Mullerian agenesis. Different classifications for uterine anomalies have been published such as those by the American Fertility Society (1988), European Society of Human Reproduction and Embryology (ESHRE) and the European Society of Gynaecologic Endoscopy (ESGE) in 2013, along with modifications such as American Society of Reproductive Medicine (ASRM) (2016) and Congenital Uterine Malformation by Experts (CUME) classification for arcuate and T-shaped uterus [2, 3, 4, 5, 6]. Acquired causes include myometrial lesions as myomas, adenomyosis, adenomyoma and sarcomas. Endometrium may be the cause of infertility as in cases of endometrial polyps, endometrial hyperplasia, chronic endometritis, and endometrial carcinoma. Specific entities in the acquired causes of uterine disease include intrauterine adhesions, abnormal caesarean scar, and scar niche [1].
Chronic endometritis (CE) is a chronic inflammation of the endometrium caused by bacterial infection of the cavity. The most common organisms responsible for this pathology are Enterococcus faecalis, Enterobacteriaceae, Gardnerella vaginalis, Streptococci, Staphylococcus species and Mycoplasma. Sexually transmitted infections, such as Chlamydia trachomatis, Ureaplasma urealyticum, and gonorrhoeae are potential causes of the infection [7, 8].
CE is asymptomatic in most cases, although it may present with pelvic pain, abnormal uterine bleeding, dyspareunia, and vaginal discharge [9]. While its actual prevalence in the general population is difficult to calculate, researchers estimate it as ranging from 0.8 to 19% [10]. Although CE is asymptomatic, it may account for lower pregnancy rates from both natural and in vitro fertilization conceptions; further, it may lead to poor obstetric outcome and neonatal complications [11, 12, 13, 14, 15, 16, 17]. CE has been diagnosed in 2.8–39% of women suffering from infertility [14, 18, 19]. However, prevalence may be as high as 60% in patients with recurrent pregnancy loss or 66% in patients with repeated implantation failure [20]. Diagnosis is difficult due to lack of specific symptoms as well as diagnostic ultrasound findings. Histopathology is diagnostic for CE by the presence of plasma cells in the endometrial stroma. Histopathology often has false positive results when the biopsy is taken in the late luteal or during menstruation [9].
Hysteroscopy is a diagnostic tool for CE [8, 20]. Hysteroscopic diagnosis depends on subjective findings observed such as stromal oedema, focal or diffuse hyperaemia, and/or the presence of micropolypi [20]. Treatment of CE may improve in vitro fertilization (IVF) success in women with recurrent implantation failure. Endometrial biopsy is recommended to confirm CE diagnosis before initiating IVF cycle [21].
The uterine cavity can be assessed through different techniques such as 3D transvaginal sonography, hysterosalpingogram, and hysteroscopy [22]. Hysterosalpingogram has low specificity, as well as high false-negative and false-positive rates [23]. Anomalies of the uterine cavity can be diagnosed by 3D ultrasound, a tool that exceeds diagnostic hysteroscopy, demonstrating accuracy as high as 90% in diagnosing uterine cavity abnormalities [24, 25]. Hysteroscopy enables visual assessment of the cervical canal and uterine cavity for any pathology such as intrauterine adhesions, polyps, grade zero myomas, chronic endometritis, or congenital uterine anomalies that may affect implantation. Hysteroscopy is also a good therapeutic tool in most conditions, while hysteroscopic guided biopsy is more informative about endometrial pathology [26, 27]. Two prospective and randomized controlled trials (RCTs) showed improvement in the outcome of assisted reproductive techniques (ART) cycles following office hysteroscopy [28, 29] despite lack of definitive pathological diagnosis [30]. On the other hand, a large, randomized study (INSIGHT) found no improvement in the live birth rate after office hysteroscopy before first IVF attempt in cases with normal transvaginal ultrasound [31]. Another study (TROPHY) found no improvement even in cases of previous ART failure [32].
The aim of this study was to test the accuracy of office hysteroscopy in the diagnosis of chronic endometritis as compared to histopathological diagnosis.
Prospective cohort study.
The study was conducted at a private hospital from July 2018 to January 2020.
220 infertile women scheduled for first attempt of intracytoplasmic sperm injection (ICSI).
We included patients (aged 20–40) with history of primary infertility scheduled for first ICSI. Those with chronic general diseases (immunological, cardiac, D.M, hepatic), severe vaginal bleeding, previous failed ICSI, PCOS women, and endometriosis were excluded.
This study was approved by the Ethics Committee, Faculty of Medicine, Alexandria University (number: 0304338). The study was conducted in accordance with the Code of Ethics of the World Medical Association (Declaration of Helsinki) for experiments involving humans. The manuscript is in line with the Recommendations for the Conduct, Reporting, Editing and Publication of Scholarly Work in Medical Journals. Written informed consent for participation in the study and publication of their clinical details and/or clinical images was obtained from each patient. A copy of the consent form is available for review by the Editor of this journal.
All women were subjected to complete history-taking and general examination,
vaginal speculum examination, routine laboratory investigations and transvaginal
ultrasound. Office hysteroscopy was done by Trophyscope (Storz)®, Tuttingen, Germany 30
An endometrial biopsy using a Novak’s curette was taken at the time of hysteroscopy and sent for histopathologic examination to confirm the diagnosis. The specimen was preserved in formalin 10%. Specimens were stained with haematoxylin and eosin and then examined for plasma cells. Immunohistochemistry (IHC) was performed for syndecan-1 (CD138) for cases with presence of plasma cells using streptividine-biotine peroxidase technique. Patients with positive chronic endometritis were treated with doxycycline 100 mg twice daily for 14 days. Endometrial biopsy was taken after one month and sent for histopathologic examination to confirm resolution of the condition before start of ART cycle.
Data were entered and analysed using IBM SPSS software package version 21.0, by SPSS Inc. Chicago, IL, USA [34]. Qualitative data were described using numbers and percentages. Quantitative data were described using range (minimum and maximum), mean, standard deviation and median. Significance of the obtained results was judged at the 5% level.
For categorical variables, in comparing different groups.
Correction for chi-square when more than 20% of the cells have expected count less than 5.
For normally quantitative variables. The test compares between more than two groups. Additionally, Post Hoc test (LSD) was used for pairwise comparisons.
For abnormally quantitative variables, to compare between two study groups.
The study was conducted on 220 women with complaints of primary infertility
scheduled for the first attempt of ICSI. There was no drop out as the two
procedures (office hysteroscopy and endometrial biopsy) were done in the same
setting. Patient age ranged from 20–40 years, mean 29.99
| Hysteroscopic findings | No | % |
| Negative | 46 | 20.9 |
| Positive (chronic endometritis) | 174 | 79.1 |
| Histopathological findings | No | % |
| Negative | 58 | 26.4 |
| Positive (chronic endometritis) | 162 | 73.6 |
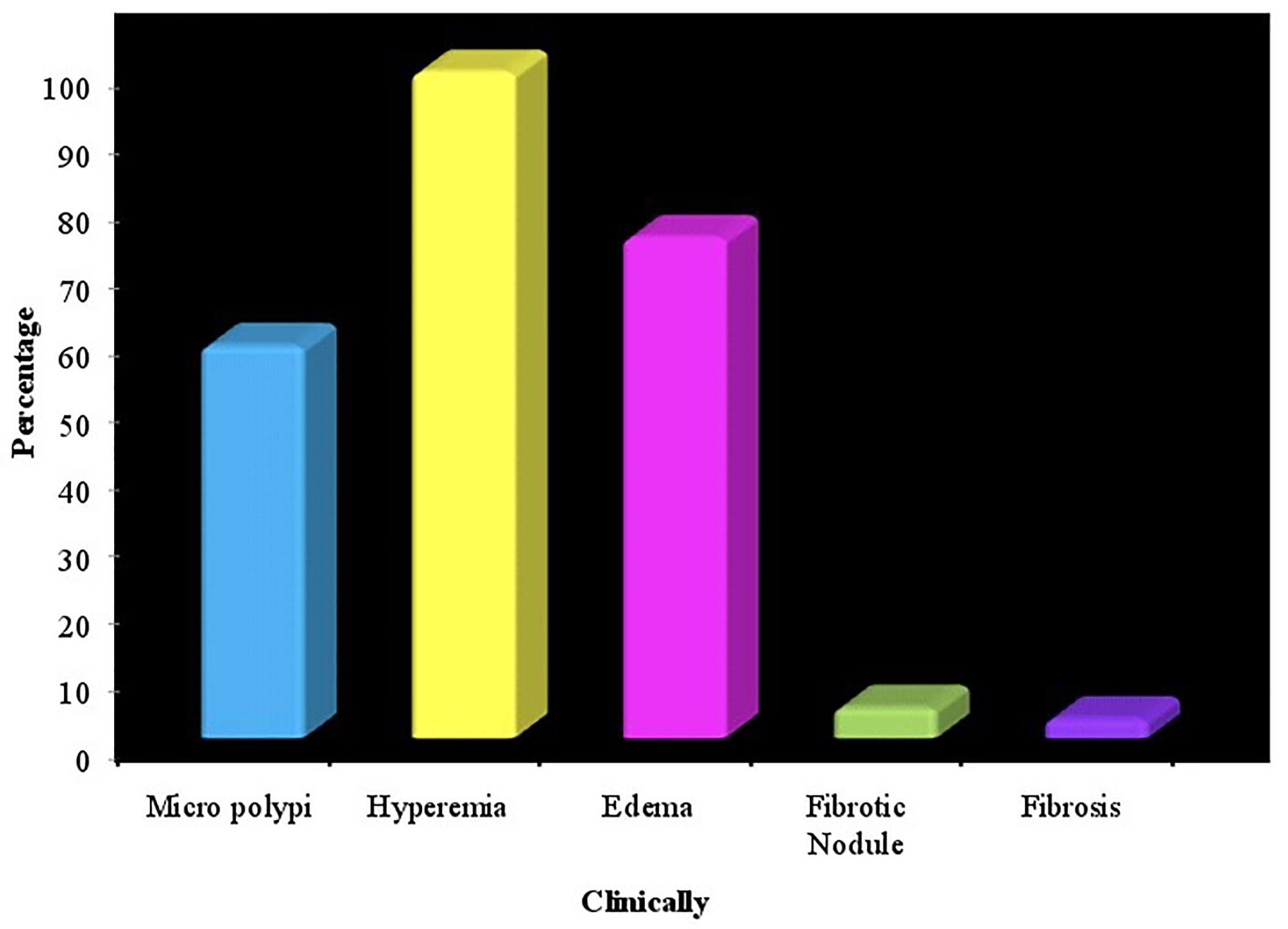 Fig. 1.
Fig. 1.Distribution of the hysteroscopic findings in cases of chronic endometritis (n = 220).
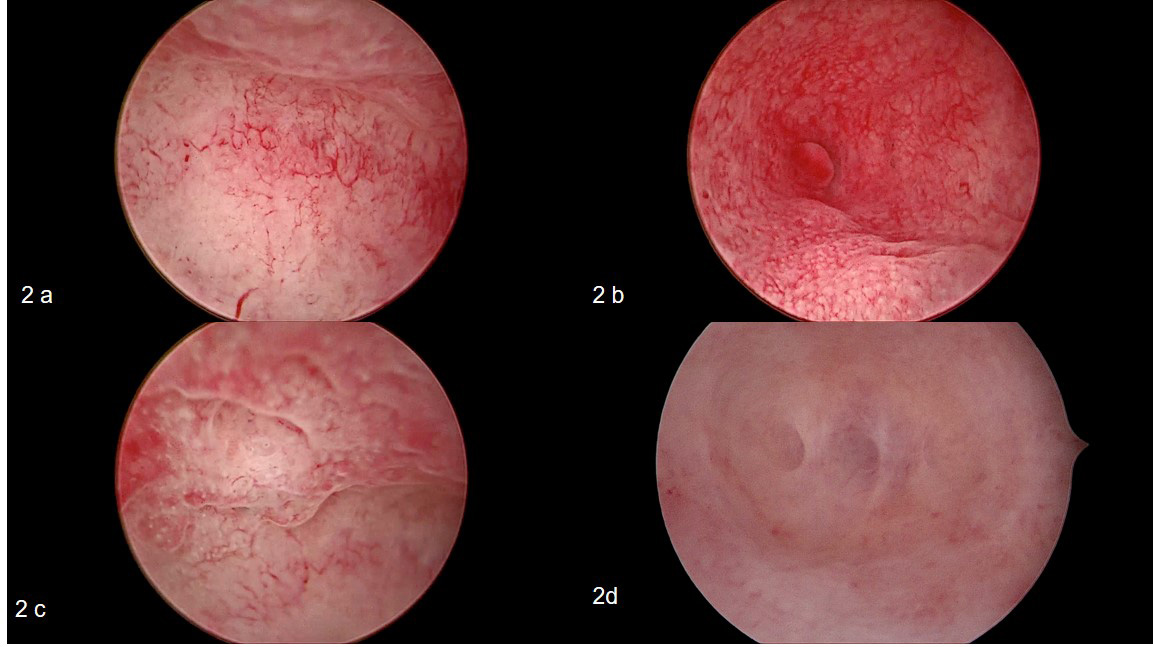 Fig. 2.
Fig. 2.Different appearance of chronic endometritis during office hysteroscopy. (a) hypervascularity, (b) micropolyp near right ostia, (c) endometrial oedema and hyperaemia, and (d) endometrial filmy adhesions separated by fluid gush.
| Histopathologic findings | Sensitivity | Specificity | PPV | NPV | ||||
| Negative (n = 58) | Positive (n = 162) | |||||||
| No | % | No | % | |||||
| Hysteroscopic findings | ||||||||
| Negative | 36 | 62.1 | 10 | 6.2 | 93.83 | 62.07 | 87.36 | 78.26 |
| Positive | 22 | 37.9 | 152 | 93.8 | ||||
IHC staining of the specimen for cases diagnosed as CE by histopathology showed that 16% of cases were negative for CD138 and 31.5% showed few CD138 cells while 52.5% showed considerable concentration of CD138 (Table 3). Repeating the endometrial biopsy after treatment showed that 22 cases (13.6%) still demonstrated chronic endometritis and 140 cases (86.4%) resolved (Table 3). The distribution of the studied cases according to IHC results is shown in (Table 4). It also shows the result of endometrial biopsy after treatment of endometritis. It was normal in 86.4% of treated cases and 13.6% in persistent cases.
| Histopathology (n = 162) | |||
| No | % | ||
| IHC | |||
| Few | 51 | 31.5 | |
| Negative | 26 | 16.0 | |
| Considerable | 85 | 52.5 | |
| Result after treatment | |||
| Normal endometrium | 140 | 86.4 | |
| Chronic endometritis | 22 | 13.6 | |
Chronic endometritis is a pathology that is hardly diagnosed via non-invasive approaches. TVS and MRI cannot confirm the diagnosis, while CE can be suspected only in patients with uterine synechia, hydrometra or pyometra. CE is asymptomatic in many cases or may cause mild symptoms such as vaginal spotting, bleeding and vague pelvic pain. The causative pathogen was found to be non-gonococcal, non-chlamydial infections [35].
Cicinelli et al. [35] established diagnostic criteria for CE using
hysteroscopy. Fluid hysteroscopy is more sensitive than CO
Hysteroscopy has the advantage of direct inspection of the uterine cavity and the detection of signs of inflammation; it may serve as a first choice for diagnosis as it can be done as an outpatient procedure [37]. Unsuspected uterine pathologies have been diagnosed in 11 while 45% of women received hysteroscopy before IVF [38]. Reports have concluded that hysteroscopy in the cycle preceding IVF could be helpful for patients with recurrent implantation failure (RIF) [39]. One of the pathologies that cannot be diagnosed with ultrasound and HSG is chronic endometritis. The gold standard for CE diagnosis is the presence of plasma cells in the endometrial stroma [40]. However, leukocytes that may be found in the endometrium, especially in the luteal phase, must be taken into consideration. Additional research has confirmed that fluid hysteroscopy can diagnose CE reliably based on the presence of diagnostic signs such as micropolyps, stromal oedema and focal or diffuse hyperaemia [7, 35].
The current study was conducted on 220 patients suffering from infertility,
selected from a private infertility clinic. Patient age was 20–40 years, mean
These results are in line with those of Gebauer et al. [37] who found presence of micropolyps in 51 patients in their study group (83 patients) by hysteroscopy. Polyps were diagnosed by endometrial curettage alone in only 43% of these cases. Our results were also supported by Garuti et al. [41] who found that hysteroscopy had a high accuracy for the diagnosis of micropolyps (95.3% sensitivity, 95.4% specificity, 98.9% NPV, and 81.7% PPV). The results of the present study demonstrate that diagnosis of absence of inflammation at hysteroscopy is very reliable, while the detection of endometrial hyperaemia, oedema and micropolyps strongly suggests the presence of CE. Our study carries acceptable negative predictive value (78.26%) and a high positive predictive value (87.36%).
Cicinelli et al. [35] found that 88.6% of patients with positive findings
at hysteroscopy were histologically positive for CE, but in our study, 93.48% of
them were positive histologically. Our results disagree with those of other
researchers who concluded that hysteroscopy is not useful for screening chronic
endometritis in asymptomatic infertile women. The rationale for their conclusion
assumed that incidence of chronic endometritis in the population is 10%, such
that a negative predictive value of 89.1% added no additional tools for risk
assessment for CE. However, these researchers used CO
Micropolypi characterized by the presence of inflammatory cells and stromal oedema are likely the expression of an active inflammatory reaction and release of interleukins and certain growth factors [36]. Micropolyps were demonstrated in 58.5% of cases with CE. Other researchers demonstrate that there is a clear correlation between different signs of CE at hysteroscopy and severity of inflammation at histopathology. Our research reveals that there is a good corelation between hysteroscopic and histological evaluation of CE. Previous studies demonstrate that the presence of micropolyps is a reliable sign of inflammation and that this sign has a high positive predictive value [35]. The presence of micropolyps is proportionate to the severity of inflammation, to the severity of mucosal injury and to an abnormal stimulus in the endometrium. Leucocytic infiltration in chronic endometritis is not only a quantitative but also a qualitative variation in infiltration by leucocytes. It exists as an abnormal distribution of endometrial lymphocyte [42].
Micropolypi are minute in size; detection of these minor lesions is easier when
saline is used for distention as it allows the micropolyps to float. On the
contrary, when using CO
Limitations of this study are that CE is a subtle pathology during hysteroscopic examination, and thus requires time to identify such cases. Some hysteroscopic pictures such as hyperaemia and oedema may be present in other pathologies like endometrial hyperplasia which may be seen in PCOS patients seeking fertility.
Office hysteroscopic evaluation is relatively accurate on its own but can be further confirmed by CD138 staining.
MA did the hysteroscopic examination and took the endometrial biopsy, and analysed and interpreted the patient data regarding the studied parameters. IG and MS contributed in the recruitment of the cases, as well as follow-up and data analysis. AA was responsible for histopathologic examination of the biopsies. MA and MS contributed in writing the manuscript. All authors read and approved the final manuscript and agree for publication.
This study was approved from the Ethics committee, faculty of medicine, Alexandria university (number 0304338). Informed consent was taken from all women recruited in the study, that they accept to go through the study and they agree publishing the medical data and the hysteroscopic images in medical journals.
The authors wish to acknowledge the gynaecology outpatient clinic staff for their assistance.
This research received no external funding.
The authors declare no conflict of interest.


