1 Department of Obstetrics and Gynecology, Division of Reproductive Endocrinology and Infertility, McGill University, 845 Rue Sherbrooke O, Montréal, QC H3A 0G4, Canada
2 MUHC Reproductive Center, McGill University, 888 Boul de Maisonneuve E #200, Montréal, QC H2L 4S8, Canada
Abstract
Purpose: To determine which factors predict pregnancy outcome in women
aged 40 years and above who underwent in-vitro fertilization. Method: We
conducted a retrospective case-control study of 631 women aged 40–46 years, who
underwent a total of 904 IVF cycles with autologous gametes. We used stepwise
logistic regression analysis to develop predictors of pregnancy, clinical
pregnancy and live birth outcomes. Data are presented as mean
Keywords
- Advanced maternal age
- Advanced paternal age
- Biologic clock
- IVF
- In-vitro fertilization
The proportion of women postponing pregnancy beyond age 40 has greatly increased over the last decades. On average, over 50% of these women will have difficulty attempting conception [1]. According to natural fertility studies, by age 40, one-third of women will be infertile [2]. In the United States, between 2006–2010, around 15% of women aged 40–44 required artificial reproductive technologies (ART) to conceive, compared to 1.3% of women aged 15–24 years [3]. In 2016, women over 40 represented approximately 21% of the treated patients at infertility centers in the USA [4]. When undergoing IVF with their own gametes, studies have reported a live birth rate (LBR) of 12.7% per IVF cycle in women between the ages of 40–42 years, falling to 5.1% for ages 43–44 years, and decreasing further to 1.5% for women aged 45 years and above [5, 6]. Similarly, older women who conceived with their own eggs have a high risk of miscarriage, ranging from 33% at 40 years of age and increased to almost 100% by age 45 [7, 8, 9]. These miscarriages are attributed mainly to embryonic aneuploidy [7, 8, 9]. Even with normal ovarian reserve, embryos generated from older women have a low pregnancy potential [10]. Numerous studies have demonstrated that the female age is the most important factor in predicting IVF success [11, 12].
The effect of paternal age on reproduction is still not well understood. While, there is evidence that advanced paternal age increases chromosomal abnormalities [13], especially beyond the age of 50 [14]. Other studies found that there is insufficient evidence to demonstrate an unfavorable effect of paternal age [15].
Finding predictors of a positive outcome in women of advanced maternal age undergoing IVF is central to achieving success in ART. In this study, we aimed to determine the predictors for positive clinical outcomes in women over 40 undergoing IVF.
This is a retrospective cohort study. Data was celected from patient charts. The inclusion criteria used included all females aged 40–46 years old going through IVF (In-Vitro Fertilization), with or without intra-cytoplasmic sperm injection (ICSI) cycles at the MCGill University Health Center Reproductive Center (A university affiliated reproductive center) from August 2010 to June 2012. All cycles during this time period were included with either cleavage or blastocyst transfer. Exclusion criteria were cycles where oocyte collection was not performed, oocyte donation or sperm donation cycles, and any intra-uterine pathology including submucosal fibroids or polyps that were not corrected pre-transfer. All subjects had normal serum TSH levels (0.4–2.5 IU/L), and prolactin per assay guidelines (on 1 out of 2 specimens). Couples requiring surgical sperm extraction or severe male factor defined as less than 5 million total motile sperm count were also excluded. All subjects had a normal uterine cavity on imaging—without uterine anomalies, including septum, unicornuate or bicornuate uteri. None of the subjects had intra-mural fibroids without an intra-cavitary component of more than 1.5 cm in maximum diameter, and these polyps and small fibroids were removed before inclusion in the study. None of the subjects were canceled for too few follicles developing. Even subjects with a single follicle were continued to the collection. If no embryos developed or no oocytes were collected (less than 5% of subjects), they could not be included in the analysis because a lack of embryos would have dropped that subject from the analysis by stepwise logistic regression. None of the subjects had evidence of endometriosis or hydrosalpinges. All subjects had a diagnosis of advanced maternal age causing infertility. None of the subjects had anovulation or thin endometrium at the time of transfer. The stimulation protocol and doses of gonadotropins were prescribed on a case-by-case basis according to patient basic characteristics and clinician preferences. The starting dose of gonadotropin was individualized for each patient based on age, basal FSH (Follicular stimulating hormone) levels, AFC (Antral follicular count), body mass index (BMI), and previous response to ovarian stimulation. Adjustment of the dose was performed according to the ovarian response, which was monitored by vaginal scans and estradiol (E2) determinations.
All treatments were conducted as previously described [16]. Ovarian stimulation
was performed by one of the following protocols: the majority were treated with
the micro dose-flare protocol (491 cycles) [17]. The rest were treated with
either a fixed GnRH (Gonadotropin-releasing hormone) antagonist protocol (332
cycles) [18] or the GnRH-ag long protocol (81 cycles) [19]. Once the primary
follicle had an average diameter
Oocyte retrieval was performed 36-hours after hCG triggering. All visible follicles, regardless of diameter, were aspirated. Fertilization of retrieved oocytes was carried out by IVF or ICSI. All embryos were cultured in cleavage medium (Cook Medical, Sydney, Australia) until day 3 and graded as previously described [20], and subsequently transferred to blastocyst medium (Cook Medical, Sydney, Australia) for culture to the blastocyst stage and then graded as previously described by Gardner et al. [21].
Luteal support was given by Estradiol (Estradiol Valerate, Actavis Pharma, USA) 2 mg orally three times daily and progestin supplements (Prometrium 200 mg vaginally, three times daily, Merck Germany; Endometrin 200 mg vaginally, twice daily, Ferring USA; Crinone 8% vaginally twice daily, Actavis USA or intramuscular progesterone 100 mg daily, Actavis USA) were started on the day after oocyte collection and continued until 12 weeks of pregnancy.
Embryo transfer was performed on day 2, day 3, or day 5, based on embryo quality, a prior history of failed IVF cycles, and the number of embryos available for transfer. Three embryos were the maximum number allowed to be transferred, based on the Quebec regulatory law. The quality of the embryos was assessed morphologically, based on previous publications [22, 23]. Ultrasound-guided trans-cervical embryo transfer was performed using a Wallace catheter (Smiths Medical, USA) under transabdominal ultrasound guidance, with a full urinary bladder. None of the patients had embryos to vitrify.
A positive pregnancy test was defined as a serum
An analysis of a retrospectively collected dataset, which consisted of 631 women who underwent a total of 904 IVF cycles from August 2010 to June 2012, was performed. Only cycles resulting in fresh embryo transfer with autologous oocytes and sperm were included. The following variables were analyzed: maternal & paternal age, number of IVF cycle, treatment protocol, number of follicles less, or at least 14 mm on the day of trigger, number of oocyte(s) retrieved, viable oocytes, metaphase two (MII) oocyte(s), two pronuclei (2PN) embryo(s) number, number of cells and grade of the embryo(s) transferred. Patients were classified based on achieving a pregnancy, clinical pregnancy, and live birth.
Approval from the Research Ethics Board was obtained (IRB#13-053-SDR).
Statistical analysis
Statistical analysis was done using SPSS 11
A total of 631 women over 40 years attempted IVF/ICSI cycles resulting in 904
autologous embryo transfers. According to the patients’ age at the start of
treatment, there were 233, 245, 219, 127, 62, 16, and 2 cycles performed for
women at 40, 41, 42, 43, 44, 45, and 46 years old, respectively. Two hundred and
fourteen women (33.9%) had more than one IVF cycle. The mean number (
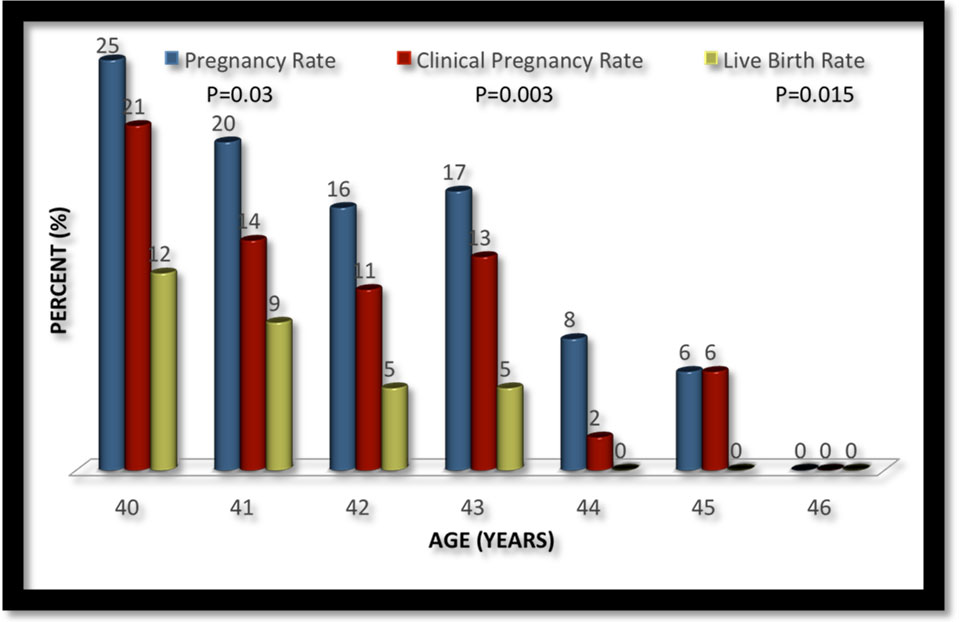 Fig. 1.
Fig. 1.Pregnancy outcome as a function of female age.
Predictors of pregnancy as determined by stepwise binomial regression analysis are presented in Table 1. As expected, maternal age was a predictor of pregnancy. However, the other significant predictors of pregnancy were: number of follicles stimulated, number of oocytes collected, number of resulting embryos, grade of best embryo transferred, and day of embryo transfer. The group with a higher percentage of blastocysts transfers, when compared to the cleavage stage, was more likely to conceive. It should also be highlighted that paternal age, the previous number of IVF attempts, and stimulation protocol used were not predictors of pregnancy in this group.
| Conceived | Not Conceived | P-value | CI | |
| (n = 170) | (n = 734) | |||
| Maternal Age (Years) | 41.2 |
41.6 |
0.01 | 0.58–0.92 |
| Paternal Age (Years) | 43.3 |
43.4 |
0.81 | NS |
| Protocol Used | 47% MDF | 56% MDF | 0.55 | NS |
| 38% ANT | 37% ANT | |||
| 15% LONG | 7% LONG | |||
| Attempt number | 2.0 |
2.1 |
0.58 | NS |
| Follicles |
6.2 |
4.5 |
0.05 | 1.01–3.8 |
| Follicles |
5.3 |
4.2 |
0.004 | 1.3–2.9 |
| Oocytes Collected | 9.1 |
6.5 |
0.0001 | 1.7–2.6 |
| Viable Oocytes | 9.0 |
6.5 |
0.0001 | 1.4–2.4 |
| MII Oocytes | 7.0 |
4.8 |
0.0001 | 2.0–2.9 |
| 2PN Embryos | 5.0 |
3.3 |
0.0001 | 1.9–3.0 |
| Cleavage Stage Embryos | 4.9 |
3.2 |
0.0001 | 1.7–2.5 |
| Number of Embryos Transferred | 2.0 |
1.9 |
0.004 | 1.1–2.0 |
| Number cells of best embryo transferred | 5.5 |
4.6 |
0.001 | 1.6–2.4 |
| Grade best Embryo Transferred | 82% grade 2 | 65% grade 2 | 0.01 | 0.37–0.95 |
| 18% grade 3 | 33% grade 3 | |||
| 3% grade 4 | ||||
| Day of Transfer | 40% day 2 | 66% day 2 | 0.008 | 1.5–2.7 |
| 26% day 3 | 22% day 3 | |||
| 4% day 4 | 2% day 4 | |||
| 29% day 5 | 11% day 5 | |||
| CI, Confidence interval; MII, Metaphase II; MDF, Micro-dose flare protocol; ANT, GnRH antagonist protocol; LONG, Long protocol; NS, Not significant. | ||||
Predictors of clinical pregnancy are presented in Table 2. These predictors were very similar to the predictors of pregnancy. Although, it should be noted that the grade of the embryo transferred and percent of blastocyst transfers, were no longer significant predictors of clinical pregnancy.
| Clinical Pregnancy | No Clinical Pregnancy | P-value | CI | |
| (n = 126) | (n = 778) | |||
| Maternal Age (Years) | 41.1 |
41.6 |
0.001 | 0.50–0.89 |
| Paternal Age (Years) | 42.9 |
43.4 |
0.67 | NS |
| Protocol Used | 47% MDF | 55% MDF | 0.99 | NS |
| 39% ANT | 37% ANT | |||
| 14% LONG | 8% LONG | |||
| Attempt number | 2.0 |
2.1 |
0.34 | NS |
| Follicles |
6.8 |
4.5 |
0.012 | 1.3–3.2 |
| Follicles |
5.4 |
4.3 |
0.005 | 1.6–2.7 |
| Oocytes Collected | 9.3 |
6.6 |
0.0001 | 1.7–2.5 |
| Viable Oocytes | 9.0 |
6.7 |
0.0001 | 1.3–2.1 |
| MII Oocytes | 7.2 |
4.9 |
0.0001 | 1.9–2.9 |
| 2PN Embryos | 5.2 |
3.3 |
0.0001 | 1.6–2.4 |
| Cleavage Stage Embryos | 5.2 |
3.2 |
0.0001 | 1.8–2.2 |
| Number of Embryos Transferred | 2.0 |
1.9 |
0.012 | 1.1–2.9 |
| Number cells of best embryo transferred | 5.6 |
4.6 |
0.009 | 1.4–2.6 |
| Grade best Embryo Transferred | 80% grade 2 | 66% grade 2 | 0.17 | NS |
| 20% grade 3 | 32% grade 3 | |||
| 2% grade 4 | ||||
| Day of Transfer | 39% day 2 | 64% day 2 | 0.29 | NS |
| 27% day 3 | 22% day 3 | |||
| 5% day 4 | 2% day 4 | |||
| 30% day 5 | 12% day 5 | |||
| CI, Confidence interval; MII, Metaphase II; MDF, Micro-dose flare protocol; ANT, GnRH antagonist protocol; LONG, Long protocol; 2PN, 2 Pronuclei; NS, Not significant. | ||||
Predictors of live birth are presented in Table 3. Most predictors were similar to those presented in the other two previously presented tables (one and two), with those predictors related to a more vigorous number of follicles stimulated and maternal age remaining important. However, it should be highlighted that paternal age became an important predictor of live birth in this age group. The male age range was 25 to 73 years, with 95% falling below 53 years and the median was 41 years. However, when it comes to live birth, the number of embryos transferred and the number of cells in the best embryo transferred were no longer significant predictors. Interestingly, even though a greater percentage of patients with a live birth had a blastocyst transfer as compared to the clinical pregnancy and pregnancy groups, when controlling for confounding effects this was no longer a predictor of live birth.
| Live birth | No Live birth | P-value | CI | |
| (n = 67) | (n = 837) | |||
| Maternal Age (Years) | 41.0 |
41.6 |
0.002 | 0.36–0.78 |
| Paternal Age (Years) | 42.1 |
43.5 |
0.04 | 0.62–0.94 |
| Protocol Used | 43% MDF | 55% MDF | 0.85 | NS |
| 40% ANT | 37% ANT | |||
| 16% LONG | 8% LONG | |||
| Attempt number | 2.0 |
2.1 |
0.5 | NS |
| Follicles |
7.8 |
4.6 |
0.03 | 1.1–3.4 |
| Follicles |
5.9 |
4.3 |
0.03 | 1.2–3.2 |
| Oocytes Collected | 10.2 |
6.7 |
0.005 | 1.3–2.9 |
| Viable Oocytes | 10.2 |
6.7 |
0.005 | 1.2–2.8 |
| MII Oocytes | 8.0 |
5.0 |
0.001 | 1.3–2.4 |
| 2PN Embryos | 5.7 |
3.4 |
0.0001 | 1.8–2.6 |
| Cleavage Stage Embryos | 5.7 |
3.4 |
0.0001 | 1.8–2.6 |
| Number of Embryos Transferred | 2.0 |
1.9 |
0.07 | NS |
| Number cells of best embryo transferred | 5.4 |
4.7 |
0.30 | NS |
| Grade best Embryo Transferred | 88% grade 2 | 66% grade 2 | 0.10 | NS |
| 12% grade 3 | 32% grade 3 | |||
| 2% grade 4 | ||||
| Day of Transfer | 37% day 2 | 63% day 2 | 0.29 | NS |
| 24% day 3 | 23% day 3 | |||
| 1% day 4 | 2% day 4 | |||
| 37% day 5 | 12% day 5 | |||
| CI, Confidence interval; MII, Metaphase II; MDF, Micro-dose flare protocol; ANT, GnRH antagonist protocol; LONG, Long protocol; 2PN, 2 Pronuclei. | ||||
To determine whether the finding of male age as a significant predictor of live
birth was specific to our clinic’s patient population, a control group of 1292
IVF cycles among women less than 40 years of age, was collected during the same
time period as the other women above 40 years of age. The analysis for live birth
in this population was repeated (using multivaraite logistic regression). Male
age was not a significant predictor of live birth among women less than 40 years
of age (P = 0.42). We subsequently performed an interaction on male age,
female age grouping (less than 40 or greater than 40 years) for live birth using
the logistic regression analysis. This interaction component was statistically
significant (P = 0.01), suggesting that women at least 40 years of age
behave differently than do women less than 40 years of age in response to the
male age as a predictor of live birth. Interactions between the other factors
measured were not significant, which demonstrated that women under 40 vs. 40
years of age and above responded in similar manners to maternal age, number of
follicles stimulated, number of oocytes collected, number of resulting embryos,
grade of best embryo transferred, and day of embryo transfer, the previous number
of IVF attempts and stimulation protocol used. This means that among women less
than 40 years of age, maternal age, number of follicles stimulated, number of
oocytes collected, number of resulting embryos, and day of embryo transfer were
all significant predictors of live birth (P
This study was performed to fill the current gap in knowledge on stimulation and laboratory predictors of a positive clinical outcome in women at least 40 years of age being treated with IVF. Our data confirmed that maternal age is an important predictor of pregnancy, clinical pregnancy, and live birth [24, 25, 26], as is an improuved ovarian stimulation. However, in women at 40 years of age, male partners age was found to be a predictor of live birth.
Another finding was that paternal age was a significant predictor of live birth in this age group of women. The effect of advanced paternal age on reproductive outcomes has not been clear in all studies. Several publications have shown that advanced male age was found to be associated with increased risk of infertility [27], decrease in blastocyst formation [28], increased risk of miscarriage among women of different age groups, increased risk of fetal death [29], schizophrenia [30, 31] bipolar disorders [32], autism spectrum and attention deficit disorders as well as autosomal dominant diseases [33]. As opposed to the above, other studies shows that increased paternal age has no effect on implantation rate and pregnancy outcomes in IVF cycles nor significantly affect IVF/ICSI outcome in oocyte donation (OD) cycles [34, 35, 36, 37]. It was hypothesized that these findings reflect male genomic activation within the embryo [1, 15, 27, 38, 39, 40, 41, 42, 43, 44].
There is no clear definition of advanced paternal age for reproduction [45]. Nonetheless, the significant impact of paternal age on live birth rates noted in this study may dictate a different approach to the advanced paternal age phenomenon that parallels advanced maternal age. Therefore, in couples where the woman is at least 40 years old, they should undergo counseling about the implications of male age on the likelihood of live birth. Accordingly, it would seem logical that when selecting donor sperm, women above the age of 40 years should use male age as selection criteria, as younger woman’s oocytes may be able to repair defects in the sperm of older men. This ability may be lost in women at least 40 years of age. However, another publication claims that until the age of 45 years, the sperm donor age does not affect pregnancy rate [36], which is in contrast with what we found. Clearly, female age may play a role in the effect of male age on pregnacy outcomes with IVF.
In line with other studies, a greater number of follicles, oocytes collected, viable oocyte, more mature oocytes, and particularly the number of embryos available for transfer, were statistically significant predictors of clinical pregnancy and live birth [46]; in fact, they were more important than maternal age in this age group. This is likely due to the relatively small age range involved and the more vigorous stimulation, resulting in a positive outcome. The fact that better stimulation and collection performance improves LBR, might be explained by a better chance of getting the euploid embryo. Based on a previous publication, given the low oocyte to live birth rate during ART, which decreases with female age, achieving more follicles, oocytes and embryos might explain the increased LBR [47]. This might be compensated in multiple IVF cycles by creating a bank of embryos or by preimplantation genetic screening (PGD).
The influence of the number of embryos transferred on clinical outcomes remains
to be identified, but few studies have focused on the predictive value of the
number of embryos available in patients aged
As expected, stimulation protocol was not a predictor of pregnancy, clinical pregnancy, or live birth and the data remains inconsistent on the superiority of one protocol over the other, in this age group [53, 54, 55, 56].
A practical implication of this study is relevant for women who want to conceive from sperm donation. The findings of this study imply that the LBR might be higher if one chose a younger donor. Few studies have addressed the role of male age on pregnancy outcomes although this litterature is expanding and is relatively consistent. However larger prospective studies would help validate results.
The strengths of this study are that it confirms the role of male age and other factors on outcomes in older women undergoing IVF. It also demonstrates that this role is related to older oocytes and loses importance when younger women have children with males of older age. Another strength is that all subjects had a uniform diagnosis of advanced maternal age. None of the subjects had a severe male factor, tubal factor, endometriosis, anovulation, or a uterine factor. Lastly, the inclusion of live birth data is another strength. It should be noted form cycles from 2012 live birth data only became available at the end of 2015. The data then had to be collected analyzed and written which is why the collection period is long ago. This is an inherent drawback to live birth data.
Likewise, the limitations of this study are numerous and worth mentioning. This is a retrospective and single-center study, which may mask undetected bias. Another limitation includes the analysis of these cycles without other factors that might affect outcomes, including duration and type (primary or secondary) of infertility, and lifestyle habits, as well as ovarian reserve tests. This study reflects the role of patient age and stimulation on outcomes. Unfortunately, anti-mullerian hormone (AMH) levels were not routinely drawn on patients at this time period in our clinic.
It should be noted that there is no habile method to obtain a power analysis for multivariate logistic regression analysis. However, this was a study of over 900 IVF cycles and as such the data is robust and well powered.
As expected, live birth was related to maternal age, improved ovarian
stimulation, and embryo yield. Male age was also noted to be a significant
individual predictor of live birth at a relatively young mean paternal age (42.1
GS, NMA and MHD were responsible for the study design, data collection, statistical analyses, and manuscript writing. JB and AVP were involved in the study design, or statistical analysis, and manuscript review. WYS analyzed the samples and reviewed the manuscript. MHD acted as a senior/consulting author.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments.
Thank S. Yehuda Dahan for assistance with this article.
This research received no external funding.
The authors declare no conflicts of interest. The authors also declare that they have had full control of all primary data and that they agree to allow the Journal to review their data if requested.

