Clinical and Experimental Obstetrics & Gynecology (CEOG) is published by IMR Press from Volume 47 Issue 1 (2020). Previous articles were published by another publisher on a subscription basis, and they are hosted by IMR Press on imrpress.com as a courtesy and upon agreement with S.O.G.
1 Department of Women and Children’s Health, Unit of Gynecology and Obstetrics, University of Padua, Padua, Italy
2 Gynecologic and Obstetric Clinic, Department of Medical, Surgical and Experimental Sciences, University of Sassari, Sassari, Italy
3 University of Ferrara, Ferrara, Italy
Abstract
Background: Rectovaginal endometriosis (RVE) is a severe form of deep pelvic endometriosis associated with dysmenorrhoea, pelvic pain, and dyspareunia. Diagnosis of RVE is a challenge for clinicians. Aim of outhe present study was to compare the diagnostic accuracy of sonovaginography with MRI on a consecutive cohort of women referred to this institution due to RVE suspect. Materials and Methods: The authors performed a retrospective study on consecutive patients undergoing surgery at this Unit due to a suspicion of RVE. All women were subject to MRI and sonovaginography. Primary endpoint was to compare the diagnostic accuracy of sonovaginography and MRI in the detection of RVE. Results: RVE was diagnosed (with surgery plus histology) in 60 women. In all cases, the surgical procedure was completed without complications. Sonovaginography and MRI showed high sensitivity (95% vs. 81.7%) and similar specificity (93.8% and 91.2%, respectively) in the diagnosis of RVE. Conclusions: Sonovaginography, when performed by an expert sonographer, may represent a valid alternative to MRI for the diagnosis of RVE, with lower costs and minimal time consumption.
Keywords
- Rectovaginal endometriosis (RVE)
- Deep pelvic endometriosis
- Sonovaginography
Deep endometriosis (DPE) is arbitrarily defined as an endometriotic lesion that infiltrates the peritoneum by more than 5 mm [1, 2]. Rectovaginal endometriosis (RVE) is a severe form of deep pelvic endometriosis which affects between 3.8% and 31.4% of all women suffering from endometriosis. RVE infiltrates the rectovaginal septum and can obliterate the pouch of Douglas [3, 4].
RVE often presents with symptoms including dysmenorrhoea, pelvic pain, and dyspareunia. Nevertheless, it can cause also non-specific clinical manifestations like back pain, change in posture and bowel symptoms, resulting in a delayed diagnosis [5, 6].
Therapy for RVE comprises different drugs (i.e. progestogens, estro-progestin, androgens, GnRH analogues) and surgery. Medical therapy is temporarily effective in controlling pain, while surgery (when a complete excision of endometriotic nodule is achieved) is associated with a long-term pain resolution and a significant improvement in patient’s quality of life [7, 8].
Diagnosis of RVE is a challenge for clinicians [9]. Despite several diagnostic instruments have been investigated to date (i.e. transvaginal ultrasound, rectal ultrasound, MRI, colongraphy, CT), the gold standard is still surgical excision (laparoscopic or laparotomic) with histopathological confirmation [10, 11].
In a previous study [12], the current authors evaluated the accuracy of sonovaginography, namely a new ultrasound technique for the diagnosis of RVE, on a cohort of patients with a suspect of RVE. Surprisingly, sonovaginography showed high sensitivity (90.6%) and specificity (85.7%) in the diagnosis of RVE, considerably higher than transvaginal ultrasound examination (TVS).
In this present study, the authors aimed to compare the diagnostic accuracy of sonovaginography with MRI on a consecutive cohort of women referred to this institution due to RVE suspect.
The authors performed a retrospective study on a cohort of consecutive patients referred at this Unit (Gynecology and Obstetrics Clinic, University of Sassari, Italy) from January 2011 to November 2016 due to chronic pelvic pain, in whom RVE was suspected. Exclusion criteria were: previous abdominal surgery (except for appendicectomy), previous diagnosis of endometriosis, and history of malignant tumours. For the purpose of the study, patients were included only if RVE was confirmed through both surgical excision (laparoscopic/laparotomic) and histological examination. The aim of this study was to compare the sensitivity and specificity of vaginosonography with MRI for the diagnosis of RVE.
Vaginosonography is an instrumental method that allows the study of the vagina and adjacent structures by applying a means of contrast between the vaginal walls and the crystals of the transvaginal probe. It is a method that can be applied in patients who previously had sex, having to be performed using the transvaginal probe. The technique consists in introducing through a single-path catheter into the vaginal cavity 250-500 ml of physiological solution following the positioning of the ultrasound probe.
With the non-dominant hand, a slight pressure is exerted between the labia and the transvaginal probe with the index thumb and the middle, in order to avoid the outflow of the physiological solution outside. The physiological solution extends the vaginal walls by highlighting the anterior and posterior spaces, better defining the anatomical regions called the anterior compartment and posterior compartment. The distension of the walls allows a clearer visualization of the thickness of the vaginal wall anteriorly, posteriorly, and laterally at the level of the arches. The method allows a good vision of the bladder-uterine space, of the urethral course, of the Douglas cord and of uterosacral ligaments. The technique involves causing the probe to float inside the vagina without placing the ultrasound probe in direct contact with the vaginal walls. The door is followed as an anatomical repair and translational movements are performed horizontally and obliquely anteriorly, posteriorly, and laterally. The examination does not require intestinal preparation and no prophylactic antibiotic therapy is suggested unless there is an obvious indication as a manifest clinical infection. The method has allowed classifying the various types of lesions of the posterior compartment in four sub-categories. There are exophytic lesions (Figure 1) that protrude into cavities, in which the nodule is covered by the vaginal mucosa. Plaque injuries (Figure 2) of increased consistency and well defined in their contours. Injuries with a characteristic infiltrating appearance (Figure 3) in which the ectopic tissue shows off traces and indentations that creep into adjacent tissues. Mixed lesions showing the various types mentioned above can be seen (Figure 4) .
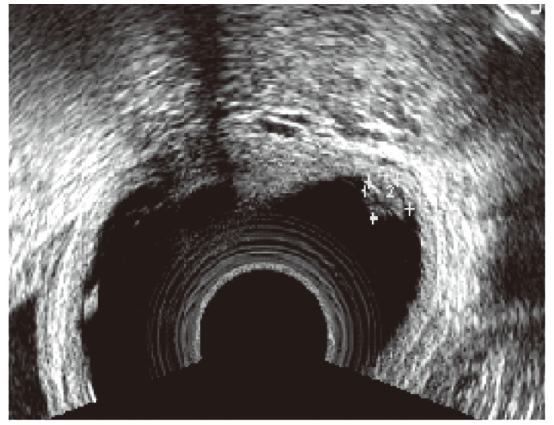 Figure 1.
Figure 1.— Ultrasound image of a rectovaginal endometriosis nodule appearing as an exophytic lesion covered by vaginal mucosa.
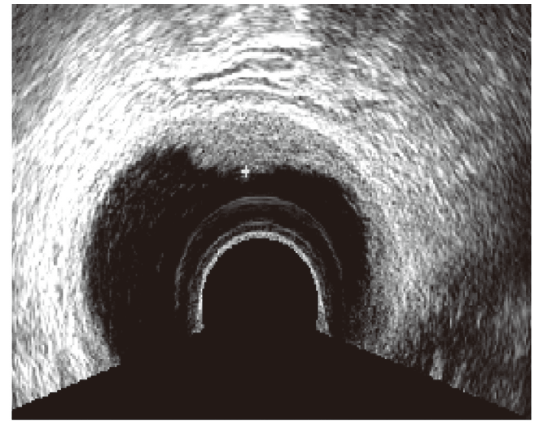 Figure 2.
Figure 2.— Ultrasound image of a rectovaginal endometriosis lesion appearing as a plaque with well-defined margins.
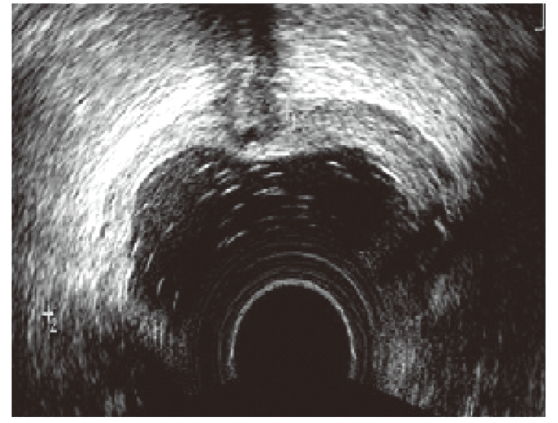 Figure 3.
Figure 3.— Ultrasound image of a rectovaginal endometriosis lesion with infiltrating appearance, in which the ectopic tissue shows off traces and indentations that creep into adjacent tissues.
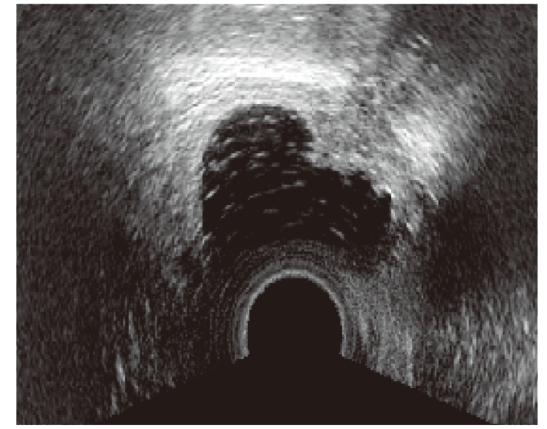 Figure 4.
Figure 4.— Ultrasound image of rectovaginal endometriosis with mixed aspect.
A total number of 189 women were referred to the present center due to chronic pelvic pain and/or dyspareunia. General features of patients are reported in Table 1.
| Age (years) | 37.1 + 6.4 |
|---|---|
| BMI (kg/m2) | 21.5 + 5.2 |
| Parity | 0.8 + 0.6 |
| Nodule size (mm) | 1.4 + 6.2 |
TVS, sonovaginography, and MRI diagnosed RVE in 37.04% of cases (60 patients). Surgery plus histology confirmed the presence of RVE in all 60 women. In all cases, the procedure was completed with minimal patients’ discomfort.
Both sonovaginography and magnetic resonance imaging showed a high sensitivity, 95% and 81.7%, respectively, and an almost overlapping specificity of 93.8% and 91.2% in the diagnosis of RVE (Table 2).
| Sonovaginography | RMI | |
|---|---|---|
| Sensitivity | 95% | 81.7% |
| Specificity | 93,8% | 91.2% |
Endometriosis is as estrogen-dependent disease characterized by the presence of endometrial cells and stroma outside the uterine cavity [13, 14].
RVE is a specific form of deep pelvic endometriosis, in which endometriotic nodules are located within the rectovaginal septum, often infiltrating the surrounding structures [15]. Due to its specific localization, RVE is associated with chronic pelvic pain, dyspareunia, and apareunia. As RVE is associated with a chronic inflammatory status (with an increase in the systemic oxidative stress and a reduction in the antioxidative defence of the human blood cells [16-18]), some novel targeted therapies have been proposed in order to improve endometriosis-related symptoms, but the gold standard is still the surgical excision of the nodules [19-21].
The diagnosis of RVE is a challenge for clinicians [22]. In this study, starting from the authors’previous experience [12], they aimed to compare the diagnostic accuracy of sonovaginography with MRI for RVE. Interestingly, among a large cohort of patients, the sensitivity and specificity of this technique for the detection of RVE were considerably higher in comparison to MRI (respectively 95% vs. 81.7% in sensitivity and 93.8% vs. 91.2% in specificity). The overall accuracy of sonovaginography was superior in respect to the previous results (i.e. sensitivity of 90.6% and specificity of 85.7), perhaps due to an improvement in the skill of the sonographers throughout the period from our previous study.
What is important is the overall number of patients in whom RVE diagnosis was missed by sonovaginography, which was only 5% (n=3/60) and all the procedures were well-tolerated by patients. It is seemingly suggesting that sonovaginography may be considered as a valuable option in the diagnostic algorithm of women suffering from RVE.
The rationale of using sonovaginography relies on the acoustic window which is created between the transvaginal probe and the surrounding structures of the vaginal channel (i.e. vaginal fornix, the vaginal walls, rectovaginal septum, the uterosacral ligaments, and vesicovaginal septum) by the instillation of a saline solution into the vaginal channel [12, 23, 24].
The major advantages of this technique are correlated with the low costs, easy access and minimal patients’ discomfort [24]. Nevertheless, it is an operator-dependent technique, thus its overall accuracy is expected to vary according to the experience of the operator. At this regard, other Authors reported a considerably lower diagnostic power for sonovaginography in comparison to MRI [24, 25].
MRI is considered as the gold standard pre-operative approach for DPE, due to high diagnostic power and the need of minimal operator’s expertise. For the evaluation of endometriotic lesions, pelvis is evaluated at the sequences in T1 and T2 SPAIR with suppression of the adipose tissue signal followed by sequences with paramagnetic contrast medium (Gadolinium). In the present study the overall diagnostic accuracy of MRI was acceptable, but lower in comparison to other authors’ experience [26, 27].
Sonovaginography, when performed by an expert sonographer, may represent a valid alternative to MRI for the diagnosis of RVE, with lower costs and minimal time consumption.




