Clinical and Experimental Obstetrics & Gynecology (CEOG) is published by IMR Press from Volume 47 Issue 1 (2020). Previous articles were published by another publisher on a subscription basis, and they are hosted by IMR Press on imrpress.com as a courtesy and upon agreement with S.O.G.
1 Department of Obstetrics and Gynaecology, Kanazawa Medical University, School of Medicin, Uchinada, Japan
Abstract
Purpose of Investigation: Dienogest is marketed in Japan as the first time in the world for novel treatment of endometriosis. This paper details the retrospective investigation how effective long-term use of dienogest is for the treatment of endometriosis. Materials and Methods: Data on 21 patients of diagnosed as endometriosis/adenomyosis and treated with dienogest for more than 12 months (ten patients with 60-month period) were collected. Results: Dienogest was effective in reducing pain and size of ovarian endometrioma, while the size of adenomyosis remained the same size as before treatment. The most frequent adverse effects were irregular bleeding. There was no reduction in bone mineral density after 60 months while plasma estradiol was maintained at a slightly lower level. Clinical parameters for did not change during a 60-month period. Conclusions: These results show that dienogest is safe to use for at least five years and is an effective treatment for endometriosis for long periods.
Keywords
- Dienogest
- Endometriosis
- Endometrioma
- Adenomyosis
- Irregular bleeding
Endometriosis is a common, chronic, and benign disease found in approximately 10% of women of reproductive age [1]. Symptoms typically include recurrent episodes of dysmenorrhea, dyspareunia, and pelvic pain [2]. Endometriosis is also frequently accompanied by sub-fertility or infertility [3, 4]. Surgical treatment provides substantial reduction in pain and infertility. However, due to the recurrent nature of endometriosis and a growing awareness of the risks of surgery on the ovarian reserve [5, 6], medical treatment is receiving greater attention for long-term treatment of endometriosis. Current medical therapies for endometriosis are mostly used to alleviate painful symptoms and improve quality of life. Several types of hormonal agents are currently used: combined oral contraceptives (COCs) have a relatively safer profile and are suitable for long-term use [7]. They are effective in reducing dysmenorrhea related to endometriosis [8]. However, due to estrogens contained in COCs, the use of COCs is not recommended for older women whose population is not small [9]. In addition, there is insufficient evidence that endometriotic lesions regress in the peritoneal cavity [10]. A review notes that COCs inhibit endometrial cell implantation but COCs also have a protective effect against endometrial lesion necrosis, thus recurrence after cessation is thought to be high [11]. GnRH agonists have been proven to be effective for both pain and endometriosis lesions, but use of these agents is restricted to a period of six months because a hypoestrogenic state leads to a reduction in bone mineral density [12]. Danazol is also effective but use of this drug is restricted to a period of four months because of androgenic side effects and an increase in the risk of thrombosis [13]. For these reasons, drugs which have comparable effectiveness to GnRH agonists but with safer profiles are awaited [14]. Dienogest, a novel therapy for endometriosis was launched in Japan in 2008 for the first time in the world to treat endometriosis. Dienogest is a fourth-generation of oral progestin. It uniquely combines the pharmacological advantages of 19-norprogestins and progesterone derivatives [15]. Preclinical studies have characterized the pronounced effect of dienogest on the endometrium without androgenic or glucocorticoid effects [16]. The mechanism of action includes inhibition of ovulation, inhibition of estrogen production, and direct inhibition of cell proliferation in endometriosis lesion. As there is only a slight report on the effects of dienogest when used for more than a year [17], there is concern about the long-term safety and efficacy profile of dienogest. The authors investigated long-term dienogest therapy to elucidate its efficacy and safety, by collecting and retrospectively analyzing data on patients.
A retrospective analysis was undertaken. Data was collected on patients visiting Kanazawa Medical University Hospital between April 2008 and September 2017 who were diagnosed with endometriosis or adenomyosis and were able to continue using dienogest (2 mg/day) for more than 12 months (ten patients with 60-month period). All patients provided informed consent at the Obstetrics and Gynaecology Department of Kanazawa Medical University Hospital (Kanazawa, Japan). This study was approved by the research ethics committee of Kanazawa Medical University. The degree of endometriosis/adenomyosis associated pelvic pain was assessed based on VAS. The volume of the ovarian cyst was determined by ultrasonography, and drug efficacy was categorized into: 1) complete regression (CR), 2) regression more than 30% (PR), and 3) enlargement more than 20% (PD), iv) or neither PR or PD (SD), according to RECIST criteria [18]. The degree of vaginal bleeding was recorded on a monthly basis by inquiry.
The degree of bleeding was categorized into four levels: 1) no bleeding, 2) spotting, 3) the same degree/less than menstruation, and 4) or more than menstruation. Other adverse reactions of the drug were also recorded on the basis of patient complaints. Ordinal clinical examination on blood count (hemoglobin, hematocrit), liver function (aspartate aminotransferase; AST, alanine aminotransferase; ALT), renal function (blood urea nitrogen; BUN, creatinine), lipid metabolism (total cholesterol, triglyceride), and coagulation (prothrombin time-international normalized ratio; PTINR, activated partial thromboplastin time; APTT) were also conducted. Plasma carbohydrate antigen 125 (CA125) level was determined as a parameter of clinical efficacy. Bone mineral density was determined by using dual X-ray abosorptiometry (DXA).
The data of 21 patients diagnosed with endometriosis or adenomyosis was collected. The age of patients ranged from 30 to 55 (average 41.0) years, and BMI ranged from 16.2 to 37.7 (average 24.0). Twelve patients had ovarian chocolate cyst only, four patients had uterine adenomyosis only, two patients had both uterine adenomyosis and uterine fibroid, two patients had peritoneal endometriosis only, and one patient had both uterine adenomyosis and ovarian chocolate cyst. The dienogest therapy was evaluated for efficacy and adverse effects on all patients. Hormonal status, clinical parameters, and bone mineral densitys of ten patients were observed for up to 60 months.
Dienogest effectively improved both dysmenorrhea and pelvic pain to the minimal level of less than 24 mm in VAS (Figure 1). Of the 13 patients who had endometrioma, eight patients were categorized into PR (regression more than 30%), five patients were categorized into SD. However, the size of uterus with adenomyosis remained almost the same size in all patients (Figure 2). The major side effect was irregular bleeding. More than 90% (19/21) of patients experienced irregular bleeding. Other side effects included headache, hot flash, depression, and fatigue (Figure 3). Episodes of individual irregular bleeding patterns are shown in Figure 4. The patients were categorized by the presence of leiomyoma or adenomyosis because patients with these complications should be treated with care according to manufacturer’s instruction.
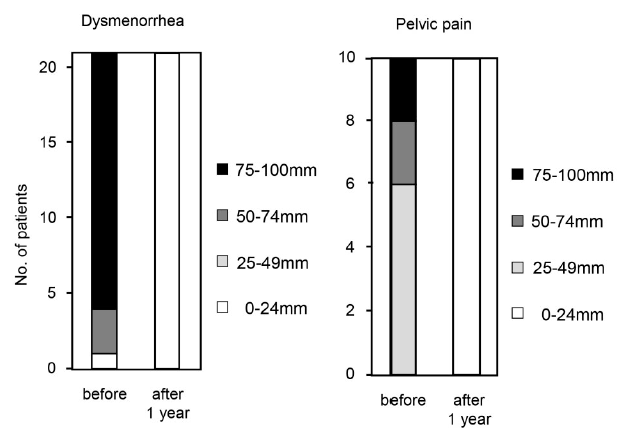 Figure 1.
Figure 1.— Effect of dienogest on endometriosis related pain. Degree of pain was evaluated at the end of treatment or after one year using visual analogue scale (VAS).
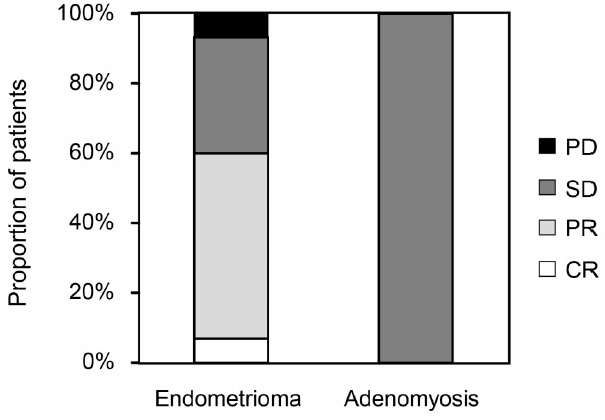 Figure 2.
Figure 2.— Effect of dienogest on endometrioma or adenomyoma. Effect of dienogest was evaluated at the end of treatment or after one year using ultrasonography. Effect of drug is categorized according to RECIST criteria14): CR: complete regression, PR: regression more than 30%, SD: neither PR or PD, PD: enlargement more than 20%.
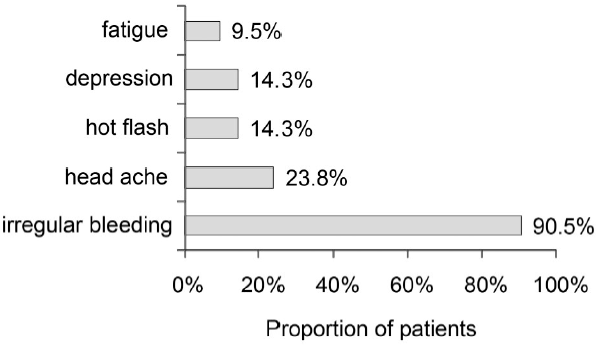 Figure 3.
Figure 3.— Adverse effects of dienogest.
 Figure 4.
Figure 4.— Episode of irregular bleeding. Degree of bleeding is presented as the height of the grey bar: level 0 = no bleeding, level 1 = spotting, level 2 = the same degree or less than menstruation, level 3 = more than menstruation. Treatment regimen was as follows: A: pretreatment with GnRH analogue; B: dienogest alone. a) Bleeding pattern of patients in the study period that were not applicable for the study due to early discontinuation of drug because of heavy bleeding are presented.
Two patients with adenomyosis stopped treatment due to heavy irregular bleeding. These two cases are shown in Figure 2 together with the other 21 cases. Although almost all patients experienced irregular bleeding, the episodes of irregular bleeding declined as the treatment continued.
Hormonal changes and other clinical parameters are shown in Table 1. There were no indicative changes in parameters for blood count (hemoglobin, hematocrit), liver function (AST and ALT), renal function (BUN and creatinine), lipid metabolism (total cholesterol and triglycerides) and blood clotting function (PT-INR and APTT). CA125 value decreased in patients that initially had high values, while the levels in patients with initially normal CA125 values remained within the normal range. Serum estradiol fluctuated within the range of 28.7-86.6 pg/mL. The mean estradiol level almost remained in the range designated as the therapeutic window (serum estradiol level between 30- 50 pg/mL) proposed by Barbier [19]. In response to lower estradiol levels, serum FSH level slightly increased. While serum estradiol was suppressed continuously for up to 60 months, mean bone mineral density (BMD) measured by DXA decreased slightly (-0.92%) at the 12-month point, and almost recovered to the pretreatment level (-0.55%) at the 60 months point (Figure 5).
| Laboratory data | Normal range | Pre-treatment | 12 months | 24 months | 36 months | 48 months | 60 months |
|---|---|---|---|---|---|---|---|
| Hb (g/dL) | 10.7-15.3 | 12.5±1.3 | 13.7±1.0 | 13.9±0.8 | 14.0±0.9 | 13.9±0.7 | 14.0±0.6 |
| HCT (%) | 33.6-45.1 | 38.3±3.0 | 40.5±3.3 | 41.4±3.1 | 41.7±2.7 | 41.2±2.3 | 41.5±1.9 |
| AST (U/L) | 13-33 | 23.4±14.3 | 16.2±2.7 | 16.6±2.5 | 17.3±3.4 | 19.5±4.7 | 17.1±3.4 |
| ALT (U/L) | 6-27 | 30.0±29.8 | 14.7±4.2 | 15.8±4.2 | 19.7±7.0 | 21.3±6.5 | 19.6±6.2 |
| BUN (mg/dL) | 8-22 | 11.4±2.9 | 10.6±2.2 | 11.6±3.0 | 10.7±2.6 | 10.6±2.2 | 10.7±1.9 |
| Creatinine (mg/dL) | 0.4-0.7 | 0.57±0.08 | 0.58±0.10 | 0.59±0.09 | 0.55±0.08 | 0.56±0.08 | 0.57±0.08 |
| Total cholesterol (mg/dL) | 128-219 | 183.3±22.0 | 182.8±17.0 | 179.8±26.2 | 177.2±22.0 | 180.3±26.6 | 199±26.9 |
| Triglyceride (mg/dL) | 30-149 | 77.0±27.7 | 70.8±25.1 | 97.6±77.5 | 85.1±44.5 | 93.1±63.2 | 83.2±50.6 |
| PT-INR | 0.9-1.1 | 1.16±0.15 | 0.97±0.04 | 1.01±0.05 | 1.00±0.03 | 1.02±0.05 | 1.00±0.05 |
| APTT (seconds) | 24-40 | 30.4±5.2 | 29.1±1.9 | 30.0±2.5 | 30.8±3.7 | 31.2±4.2 | 30.2±2.8 |
| Estradiol (pg/mL) | 24.5-195 | 86.6±85.1 | 55.9±32.8 | 35.9±27.9 | 38.1±31.5 | 46.3±40.4 | 28.7±18.4 |
| FSH (mIU/mL) | 3.5-12.5 | 10.4±7.2 | 9.3±6.9 | 8.9±6.3 | 11.1±8.9 | 9.6±7.7 | 13.6±10.3 |
| CA-125 (U/mL) | ≤35 | 50.9±48.7 | 12.4±4.3 | 12.1±4.7 | 11.1±3.5 | 12.9±7.5 | 12.3±5.4 |
Data are represented as mean and standard deviation (n=10).
Hb: hemoglobin, HCT; hematocrit, AST: aspartate aminotransferase, ALT: alanine aminotransferase, BUN: blood urea nitrogen, PT-INR: prothrombin time-international, APTT: activated partial thromboplastin time normalized ratio, FSH: follicle stimulating hormone, CA-125: carbohydrate antigen 125.
Normal range of estradiol and FSH represents follicular phase.
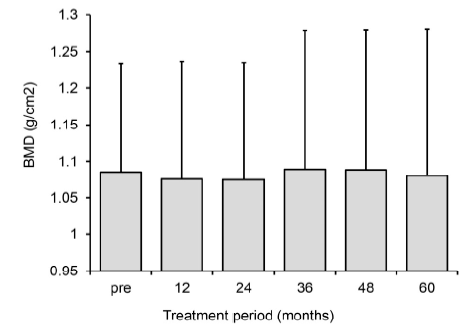 Figure 5.
Figure 5.— Changes in bone mineral density. Data are represented as mean ± SD (n=10).
Although there are many reports on the use of progestins for endometriosis, only a few progestins have established clinical efficacy and long-term safety. Among these, subcutaneous medroxyprogesterone acetate (MPA) and intrauterine levonorgestrel have been studied [20, 21]. Both drug formulations show efficacy, but MPA is reported to impair lipid metabolism and decrease bone density. The drug delivery of intrauterine levonorgestrel is almost limited to within the uterus [22] and therefore it is not a suitable therapy for ovarian endometrioma and peritoneal endometriosis. The use of dienogest to treat endometriosis in a study in Europe was first reported in 1987 [23]. After treatment with dienogest, endometriotic lesions had completely disappeared in 66.7% of patients, and a marked improvement was noted in 80.4% of patients, but no effect was visible in 19.6% of the implants. Eighty-four percent of patients reported symptomatic improvement. The same author also reported that dienogest had no or little effect on liver function [24]. Changes in Revised American Fertility Society classification (rAFS) score and the stage of endometriosis at laparoscopy were assessed in the 24-week, dose-range study. Dienogest at 2 and 4 mg daily dose produced significant reductions in the rAFS score between baseline and study end [25]. In a random trial, a dienogest group at 2 mg daily dose for 12 weeks showed a statistically significant decrease in mean visual analog scale (VAS), compared to the placebo group [26]. Dienogest 2 mg daily and leuprolide acetate at standard dose (3.75 mg, depot intramuscular injection every four weeks) provided comparable, continuous reductions in pelvic pain, as measured by VAS [27]. There was no reduction in bone mineral density of lumbar spine after 24 weeks of treatment. A follow-up study in which patients were treated for up to 15 months confirms that dienogest maintains a favorable safety, tolerability, and efficacy profile [17]. In Japan, dienogest 1 mg twice daily for six months showed a comparable effect to intranasal buserelin acetate 900 μg daily [28]. And in a 52-week study, dienogest therapy showed improvements of symptom in 72.5% of patients after 24 weeks and 90.6% of patients after 52 weeks [29]. Dienogest did not impair liver function or lipid metabolism in a 52-week study [29]. In the same study, dienogest slightly decreased BMD but the change was within the normal range. In the present institution, the authors treated 21 patients with dienogest for more than one year and ten patients for more than five years. Dienogest dramatically alleviated the painful conditions of these 21 patients. All the patients treated with dienogest showed VAS score less than 24 mm in both dysmenorrhea and pelvic pain. This result seems to show that the effectiveness of dienogest becomes more evident as the treatment period progresses. Most patients suffer irregular bleeding. Thus the authors believe initial management of bleeding is important. As pretreatment with GnRH agonist seemed to reduce initial bleeding episodes, this regimen may be useful for patients with adenomyosis whose bleeding tends to be heavy. Dienogest showed no changes in clinical parameters for blood count, liver function, renal function, lipid metabolism, and blood clotting function. Dienogest therapy resulted in slight bone loss at 12 months, however no cumulative bone loss was observed at 60 months. Thus, dienogest represents a treatment option for long-term treatment, although this study has a small sample size and there is still need for further examination.
These results show that dienogest is safe to use for at least five years and is an effective treatment for endometriosis for long periods.
The authors hope investigations will be conducted in the near future on whether dienogest is useful to protect against recurrences of endometriosis, and to avoid or postpone surgery to maintain ovarian functions.
The authors would like to express their deep and sincere gratitude to their advisor, Professor Satoru Makinoda.





