1 Department of Soil Science and Agricultural Chemistry, Tamil Nadu Agricultural University, 641003 Coimbatore, Tamil Nadu, India
2 Department of Microbiology, Tamil Nadu Agricultural University, 641003 Coimbatore, Tamil Nadu, India
3 Department of Veterinary & Animal Science, Tamil Nadu Agricultural University, 641003 Coimbatore, Tamil Nadu, India
4 Department of Agronomy, Tamil Nadu Agricultural University, 641003 Coimbatore, Tamil Nadu, India
Abstract
The notable increase in chicken waste resulting from the rapid expansion of the chicken industry represents a major concern and danger to public health and the environment. Therefore, this varied waste stream in the chicken industry, including bedding materials, dung, feathers, and mortalities, requires efficient management techniques. Improper chicken waste disposal can lead to nutrient leakage and water and soil contamination, which can cause eutrophication and aid in spreading harmful bacteria such as Escherichia coli and Salmonella. Moreover, untreated waste exacerbates climate change by increasing greenhouse gas emissions. Thus, in response to these challenges, this review analyses many treatment techniques that might convert this complicated waste stream into a useful resource to support environmental sustainability in the chicken industry and enhance soil health. Furthermore, this study evaluates gasification, pyrolysis, anaerobic digestion, and composting as viable methods to reduce pollution from chicken waste while producing useful byproducts. Anaerobic digestion uses bacteria to produce biogas, a sustainable energy source; pyrolysis produces biochar and bio-oil; composting converts waste into fertilizer; gasification produces syngas for fertilizer production. However, choosing the most efficient treatment approach necessitates thoroughly assessing waste properties, intended end products, and economic factors. This review aims to expand the understanding of these treatment procedures and their related advantages to assist in developing sustainable and effective strategies for dealing with chicken waste. These strategies, which prioritize value development, environmental preservation, and public health, have the potential to pave the way for a more responsible and sustainable future for the chicken industry.
Keywords
- chicken waste
- public health
- sustainable waste treatment
- sustainable agriculture
- circular economy and waste to wealth
The chicken sector provides vital nutrition and a living for billions of people, making it a pillar of global food security and economic growth. The rapid expansion of the sector, however, has also resulted in an increase in chicken waste, which poses a serious threat to the environment and public health. A survey conducted in 2023 predicted that the global market for chicken will increase further and reach an estimated $ 487.39 billion by 2027. Ensuring efficient waste management across the whole production chain has become a recurring concern in the chicken industry because of the growing worldwide demand for chickens. This problem affects both industrialized and developing countries, cutting over the limits of geography and economic status. The largest chicken producers worldwide are the United States, Brazil, China, the European Union, India, and Russia. Europe, particularly Poland, also plays a significant role in chicken production and export. The management of the large amounts of chicken waste produced each year is a constant challenge for major chicken producers such as those in the United Kingdom, Bangladesh, India, Australia and the USA [1] (Table 1, Ref. [1, 2, 3, 4, 5, 6]).
| Country | Chicken waste generation (Mt/year) | References |
| UK | 4.55 | [2] |
| Bangladesh | 4.52 | [1] |
| India | 3.30 | [3] |
| Australia | 1.60 | [4] |
| USA | 14 | [5] |
| Poland | 3.6 | [6] |
Mt, metric ton.
Although chicken waste requires careful handling, it is a useful source of nutrients for plants. It is a complex mixture of macronutrients that are necessary for plant development, including potassium (2.02–2.32%), phosphorus (2.46–2.82%), and nitrogen (4.55–5.46%) [7]. It also provides important micronutrients, including manganese, iron, zinc, and copper. A number of contributing elements cause considerable heterogeneity in the nutritional composition of chicken waste. These variables include the type of chicken, chicken feed, bedding used in the housing system, and methods of storage. Chicken manure is a viable substitute for inorganic fertilizers due to its organic matter content and the presence of beneficial microbial communities. This might improve soil health by improving nutrient cycling and replenishing organic matter.
Chicken waste has a complex composition that includes potentially harmful elements such as heavy metals and pathogens. Improper disposal threatens soil and water integrity and is a major cause of hazardous eutrophication, which disrupts aquatic ecosystems. As a result, managing chicken waste requires strict standards to prevent serious effects on the environment and public health [1]. The volume of waste generated by bedding materials has increased significantly, highlighting the necessity for effective disposal techniques [8]. The improper handling of chicken waste poses serious risks to the environment and public health. Inadequate containment and disposal of food leftovers provide ideal conditions for the growth of insects that spread illness. The danger of disease spread is further increased by improperly disposing of deceased birds and eggshells. Chicken feathers need to be handled carefully to reduce fire threats because of their high keratin content [1]. Chicken waste that has not been treated can include dangerous pathogens such as Salmonella and Escherichia coli (E. coli) [9]. These pathogens pose a serious risk to the health of people and animals. Methane is another strong greenhouse gas that contributes to climate change and is released during the breakdown of untreated waste. Inhalable particles from poorly managed trash can include harmful germs, causing respiratory illnesses in surrounding communities [10]. Furthermore, improper composting of waste might increase the danger to public health by facilitating the spread of zoonotic diseases such as avian influenza [1]. As a result, implementing effective and sustainable waste management procedures is critical for protecting the environment and public health and addressing the issues connected with this complex issue.
Fortunately, there are several effective methods for turning this waste stream into useful resources that support environmental sustainability, such as gasification, pyrolysis, anaerobic digestion, and composting. These techniques efficiently reduce environmental hazards while producing useful goods, including power, fertilizer, and biogas [9, 11, 12, 13, 14]. The constantly expanding waste stream in the chicken industry presents a serious hazard to human health and the environment because of its diverse composition and potential for improper handling. The best approach must be chosen after a thorough analysis of the waste properties, target end goods, and financial factors. A solid grasp of these treatment techniques is essential for implementing value-driven and environmentally responsible waste management strategies in chicken businesses. By focusing on value creation and environmental conservation, this review hopes to improve our understanding of both strategies and aid in the establishment of workable, long-term solutions.
The growing importance of the chicken industry as a major economic force has led to a greater emphasis on managing waste from chicken farms. This waste stream is produced at different phases of development and can contain bedding material, excrement, feathers, eggshells, dead embryos, mortalities, and decomposing tissues. It can also contain both liquid and solid components (Fig. 1). Chicken droppings, recognized as unparalleled reservoirs of nutritional richness within the spectrum of chicken waste, contain high concentrations of essential elements, including nitrogen, phosphorus, potassium, calcium, and magnesium (Table 2, Ref. [7, 15, 16, 17, 18, 19]). In addition to these primary nutrients, chicken waste contains a diverse array of micronutrients, including copper, zinc, iron, manganese, and others. Delving deeper into the composition of chicken droppings reveal additional components integral to their nutritional wealth, such as cellulose (2.26–3.62%) and hemicellulose (1.89–2.77%), which form an organic treasure trove. Additionally, the presence of lignin, with concentrations ranging from 1.07–2.16%, further increased the overall nutrient content, emphasizing the intricate and multifaceted composition of chicken droppings. The diverse compositions of chicken droppings highlight their potential as strong organic fertilizers and valuable resources for sustainable farming practices [20, 21]. In addition, chicken waste is a unique source of essential plant micronutrients [22], which reinforces its importance in the field of organic farming [23].
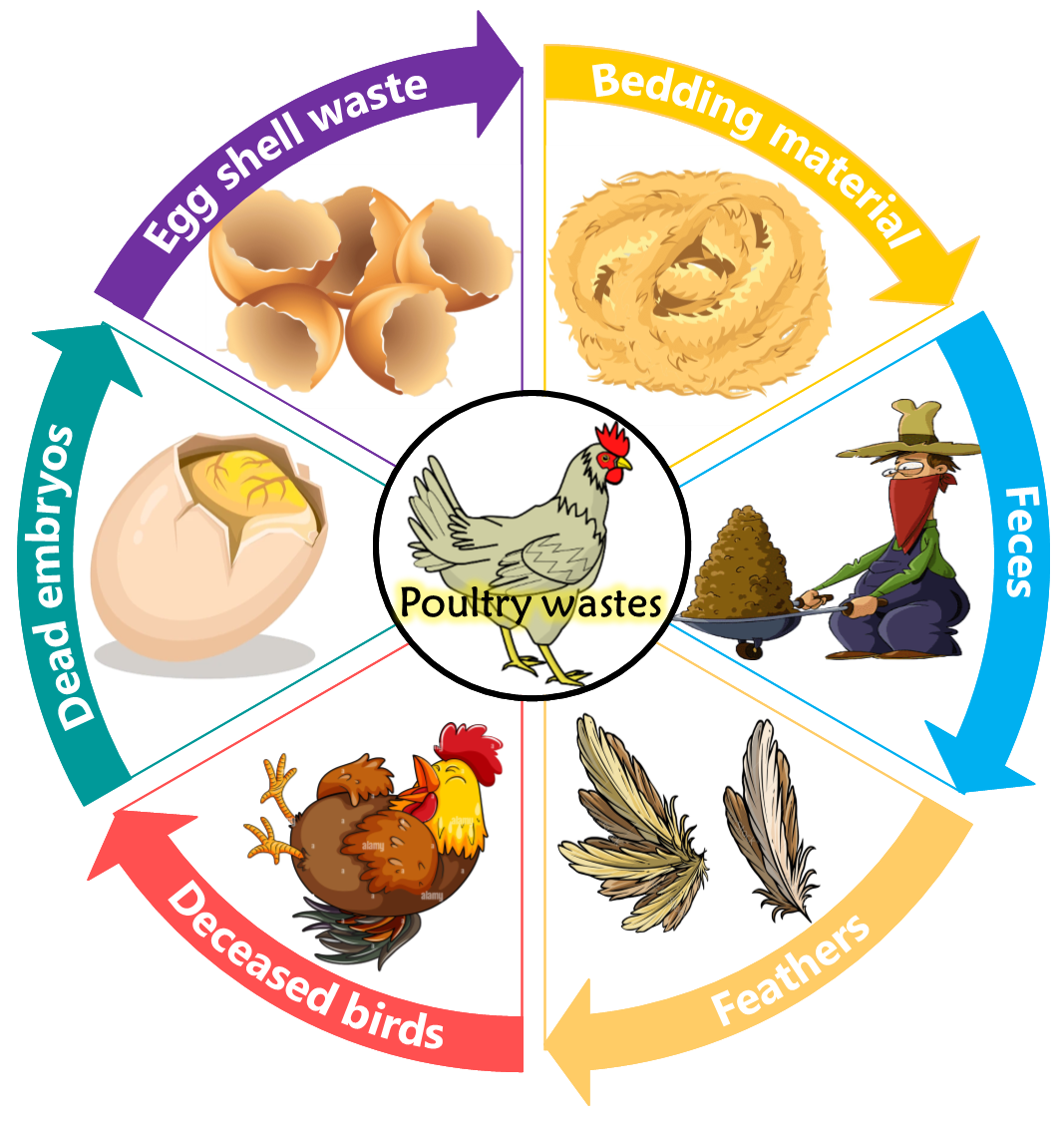 Fig. 1.
Fig. 1.
Chicken waste product.
| Nutrient concentration (g/kg) | References | |||||||
| C | N | P | K | Ca | Mg | Cl | S | |
| 323 | 24.9 | 18.3 | 29.9 | NA | NA | NA | 0.16 | [15] |
| 283 | 22 | 30 | 29 | NA | NA | NA | NA | [16] |
| 640 | NA | NA | 14.97 | 3.0 | 0.59 | NA | NA | [17] |
| 387 | 36.2 | NA | NA | NA | NA | NA | 5.3 | [18] |
| 433 | 57 | NA | NA | NA | NA | NA | 12 | [19] |
| NA | 54.6 | 28.2 | 23.2 | 8.15 | 0.73 | NA | NA | [7] |
Chicken producers can effectively use composting techniques to turn waste components into high-quality compost by leveraging the complex microbial community that is present in chicken waste. This community is made up of a variety of bacteria, actinobacteria and fungi, and it is essential for organic matter breakdown and subsequent nutrient cycling in the waste stream [24]. When paired with proper organic additions, this strategy enables nitrogen retention in the waste stream, resulting in a beneficial and sustainable waste management solution.
Chicken waste offers a plethora of alternatives for resource recovery beyond composting. Owing to its high phosphorus content, chicken waste may be used on farms in place of mineral phosphorus fertilizers to increase crop yield and growth. Furthermore, properly utilizing dry chicken waste or litter can drastically lessen the need for artificial fertilizers. Chicken litter has excellent fertilization value, but because it is less concentrated than chemical fertilizers are its value per unit mass is lower. However, by utilizing sustainable waste management practices, chicken waste may be strategically utilized to improve agriculture, the environment, and energy generation. Three goals are accomplished by applying treated chicken waste to soil: increasing soil productivity, resolving issues with waste disposal, and promoting soil quality through improved physical characteristics, chemical composition, and biological activity [25].
There are serious risks to the environment and public health when untreated chicken waste is used. According to Duan et al. [26], its disagreeable smell draws on flies, insects, and rodents and raises the possibility of infection and the development of antibiotic resistance. These strong arguments require a rigorous assessment of various approaches for treating and using chicken waste with the goal of being helpful. This study explores cost-effective and ethical methods for managing chicken waste to minimize risks to the environment and public health while simultaneously optimizing its potential as a valuable resource.
The increasing number of microorganisms in chicken excrement poses a serious threat to animal and human health and exacerbates the state of the environment. A multitude of causes can be contributed to the rise of potentially dangerous bacteria in chicken farming. Bacilli and gram-positive cocci, among other related bacterial taxa, are examples of aerobic mesophilic bacterial communities that are a significant contributing component [27]. Excessive amounts of these bacteria aid in the production of harmful pollutants such as endotoxins, and bioaerosols, among other unpleasant gases. These contaminants can have a negative impact on the operational efficiency and economic sustainability of chicken farms and can also pose occupational health concerns to farm workers. Moreover, the increase in the microbial population releases greenhouse gases such as CO2, CH4, and N2O, which exacerbates environmental pollution [28]. This emphasizes how crucial it is to put into practice efficient methods to control the microbial population in chicken waste to reduce its detrimental effects on a range of factors, including the health of people and animals, the quality of the environment, and the feasibility of farming.
Chicken farms harbor diverse microbial populations, with established minimum thresholds of 3.3 log10 cfu/m3. However, research has revealed significant variability, with documented instances exceeding 8.3 log10 cfu/m3 [27]. This elevated microbial density, often linked to high animal concentrations, creates conditions conducive to the emergence and spread of various diseases within the flock, including H1N1, H5N1 influenza, and brucellosis. Recent outbreaks of avian influenza underscore the critical need for effective microbial population management within chicken farms. Notably, Europe in 2019 and the United States in 2023 experienced significant outbreaks, highlighting the global reach and potential consequences impacting both animal health and global food security [29].
The extensive bacterial contamination of chicken litter poses a threat to public health and the environment worldwide. Actinobacillus, which is present throughout the whole litter spectrum in the Australian environment, serves as a prime example of this phenomenon. Moreover, study has shown that in Australia, 68% of non-reused litter and 83% of reused litter are contaminated with Salmonella [30]. Similar results have been reported in US, where 80%–100% of the fecal matter from chicken is dominated by Actinobacillus and Campylobacter [31, 32]. The distribution Campylobacter spp. becomes homogeneous throughout the capture, transport, and processing of chicken. Fecal contamination and consequent increases in Campylobacter spp. levels occur during the transportation of broilers from producing farms to processing facilities [32]. The digestive tracts of chickens may contain up to 107 CFU of Campylobacter spp. per gram of intestinal contents without causing any pathogenic effects on the avian host. During chicken processing, leakage or cross-contamination of intestinal contents can contaminate chicken carcasses. Fecal droppings offer a non-invasive method of determining the presence and levels of Campylobacter spp. in broiler flocks [31].
Furthermore, the presence of Salmonella varies; in Canada, it has been found in 0%–100% of chicken litter [33]. In addition to these major bacteria, additional pathogens of concern have occasionally been found in chicken litter and its compost at quantities higher than the safe thresholds advised for manure as a soil supplement. These include, notably, Bordetella, Corynebacterium, Mycobacterium, Campylobacter, E. coli, Listeria, and Globicatella.
In chicken manure, the density and abundance of such microorganisms are strikingly high. It reached up to 1010 cfu/g. Gram-positive bacteria, which primarily include Actinomycetes sp. and Bacillus sp., constitute 90% of the population. The robust and active presence of Actinomycetes sp. and Lactobacillus sp. not only played a substantial role but also proved important in preventing pathogen growth and multiplication. This robust defensive mechanism has unquestionably imparted a significant and remarkable benefit in sustaining a pathogen-resistant environment. The aerobic bacteria count in the manure was approximately 109 cfu/g. This count was dominated by Enterococcus. On the other hand, the bacterial plate count/density of aerobic bacteria, which are essentially digestible in nature, was approximately 108 cfu/g in chicken manure. In contrast, three orders of magnitude fewer aerobic bacteria, i.e., 105 cfu/g, were observed in the broiler manure. However, in both broiler and hen manure, the same order of magnitude, i.e., 103 cfu/g of most likely number of microorganisms, was detected [29].
Careful reassessment and possible prevention of the reuse of chicken litter or the addition of new flocks to existing litter are needed because of the significant association of this practice with increased Salmonella infection compared with that of non-reused litter [30]. The presence of this pathogen is associated with a variety of concerns, including substantial financial losses in addition to direct dangers to the health of humans and animals. Salmonella spp. may spread widely and have an enormous worldwide impact because of their ability to transmit both horizontally (through infected feed, workers, and equipment) and vertically (from parent flocks to offspring). Moreover, a variety of microorganisms resistant to antibiotics are present in chicken litter, which makes successful treatment plans more complicated and expensive. Extensive US investigations have shown that collected grill litter contains widespread multiresistant E. coli. According to Zhang et al. [34], this is directly related to the widespread use of antibiotics in industrial chicken operations, which promotes the evolution of resistant diseases through mobile genetic elements (MGEs) such as integrons, transposons, and plasmids. The emergence of these resistant strains highlights the necessity for the chicken industry to enact strict antibiotic stewardship policies [35]. This will help to prevent deaths, minimize the financial impact of antibiotic resistance [36], mitigate outbreaks, and lower hospitalization rates.
A wide variety of potentially dangerous microbes, including helminths (parasitic worms), fungi, viruses, and protozoa found in chicken litter [30]. Compared with fresh broiler litter, aged litter has a greater level of fungal complexity. Aspergillus, Alternaria, Cladosporium, and Penicillium spp. predominated in the fresh broiler litter. Penicillium species are still common in older litter, along with Scopulariopsis spp., Trichosporon spp., and Aspergillus spp. (Table 3) [30, 37]. Fungi such as Fusarium and Scopulariopsis which break down structural proteins necessary for strong hair and nails, are especially dangerous. Furthermore, species of Fusarium, Penicillium, and Aspergillus can act as reservoirs for aflatoxins that cause cancer in chickens [38]. The risk of histoplasmosis, particularly in immunocompromised humans, is highlighted by the presence of Histoplasma capsulatum in chicken litter. This might result in serious respiratory difficulties. Personal protective equipment (PPE), such as gumboots, dungarees, and nose masks, is essential for reducing the risk of transmission to anyone handling chicken litter [30].
| Broiler litter | Fungal species | Percentage (%) |
| Fresh broiler litter | Penicillin spp. | 59.9 |
| Alternaria | 17.8 | |
| Cladosporium | 7.1 | |
| Aspergillus | 5.7 | |
| Aged broiler litter | Penicillin spp. | 42.3 |
| Scopulariopsis spp. | 38.3 | |
| Trichospporon spp. | 8.8 | |
| Aspergillus spp. | 5.5 |
Chicken litter may also include parasitic helminths, such as nematodes and cestodes, which may impede blood flow and nutrient absorption. Ascaridia galli, Heterakis sp., and Raillietina sp. are common large-scale chicken parasites; young birds are especially vulnerable [39]. Strict cleanliness regulations are necessary since unhygienic behaviors and the reuse of litter might worsen parasite infections [40].
Giardia and Cryptosporidium spp. are examples of protozoans that cause zoonotic danger to humans and the environment at large because they can spread through polluted water sources [41]. The potential of Cryptosporidium to infect a variety of animals, including fish, cattle, chicken, and people, highlights the complicated problems associated with this disease [42].
The serious public health and economic consequences of microbial contamination demand strict laws on organic waste, including compost and biosolids. However, there is a major regulatory vacuum regarding the proper disposal of chicken litter. The upper bounds of acceptable microbial loads for soil amendments are defined by safety thresholds; values greater than these indicate a threshold for danger. According to Kyakuwaire et al. [30], the quantities of total coliform (104–106 cfu g-1) and E. coli (105–1010, average 109 cfu g-1) in chicken litter the maximum permitted limits for safe use as a soil amendment.
Furthermore, there are notable discrepancies in the guidelines proposed by
different organizations and regulatory agencies. Interestingly, several
acceptable limits for Salmonella are defined by Australian criteria,
ranging from
An extensive analysis of the microbiome of chicken manure revealed that different samples of the microbial community included bacteria, fungi, algae, and actinomycetes. Important functional groupings related to the cycling of carbon, nitrogen, and sulfur were identified. Chicken waste has a fairly high microbial density and is dominated by stringent or facultative anaerobic bacteria. Actinomycetes and fungi were also abundant, with the latter mostly found in the form of dormant spores. Although there were algae, they were fewer in number. Remarkably, several aerobic bacteria were found to exhibit acidophilic (acid-tolerant) traits, but the remaining algae presented spore-forming traits. The lack of cyanobacteria was likewise verified by this preliminary investigation [45].
The population of microorganisms detected in chicken waste has numerous capabilities, including proteolytic and ammonifying capabilities. Anaerobic cellulolytic, denitrifying, and anaerobic nitrogen-fixing capacities were among the other traits seen in the population [46]. Then, a number of residual activities were noted, including sulfate-reducing, anaerobic sulfur-mineralizing, amylolytic, and pectinolytic activities. Relatively low numbers of bacteria that prefer aerobic conditions were found. No sulfide oxidizers were found [47].
Detailed documentation of the beginning of antibiotic use in chickens started in 1946 [48]. Starr and Reynolds [49] produced groundbreaking research that led to the discovery of antibiotic resistance in food animals and discussed the possibility of resistance developing early and persuasively as early as 1969 [50]. Comprehensive guidelines were provided in 1969 in response to increasing concerns about human antibiotic-resistant bacterial infections [51]. These recommendations strongly opposed the use of antibiotics as growth promoters, particularly for drugs such as sulfonamides, penicillins and tetracyclines, which are also prescribed to humans. As defined by the World Health Organization, “an increase in the minimum inhibitory concentration of a compound for a previously sensitive strain” is known as antimicrobial resistance. Antibiotic resistance arises from four basic processes: drug inactivation, decreased permeability, increased efflux, and altered targets [52]. It is still unclear whether resistance genes arise from horizontal gene transfer, the transmission of genetic materials from one organism to another or from adaptation through chromosomal mutation, often known as gene shuffling. This process is not the same as typical vertical DNA transfer from parent to child [53]. A total of 106–109 bacterial cells or 103–106 different bacterial species can be found in one gram of soil. Thus, soils operate as a vast reservoir of microbiological variety [54, 55]. Interestingly, soil is more than just a passive medium; it is the source of many antimicrobial substances that are essential to animal health care. These substances are particularly obtained from bioactive substances produced by fungi or bacteria (such as Streptomyces spp.) [56]. The environment plays a critical role in this ecological dilemma, and a thorough analysis of the many interactions that take place in the environment is necessary to understand the complex dynamics of antimicrobial resistance.
In the United States, E. coli was detected in all (100%) of the broiler litter samples that were tested. The genes in these samples were resistant to more than seven antibiotics, such as ampicillin, sulfonamide, tetracycline, and ceftiofur [57]. Ampicillin and tetracycline resistance were present in 63% and 50%, respectively, of the E. coli-contaminated chicken litter compost [57]. All (100%) of the broiler litter samples examined in the US included coliform bacteria, which are typically resistant to sarafloxacin and nalidixic acid. Unfortunately, information about the presence of antibiotic-resistance genes in chicken litter is lacking in developing countries, where the use of chicken litter and the chicken industry are expanding rapidly. The US and Australia are two developed nations where this information is accessible. Within the Enterobacteriaceae family of facultative anaerobic gram-negative bacteria, E. coli is commonly found in the gastrointestinal tracts of warm-blooded mammals. Because of its widespread nature in this environment, E. coli has been extensively used for detecting antimicrobial resistance in food animals, notably chickens. Furthermore, some strains of E. coli carried by chicken may be dangerous because they include genes linked to antimicrobial resistance that may be transferred to humans [58].
Specifically, strains of E. coli known as “avian pathogenic E. coli” (APEC) cause colibacillosis, which is a major cause of illness and death in chicken worldwide [59]. A comprehensive study conducted in China revealed that avian pathogenic E. coli (APEC) strains possess the florfenicol resistance gene (floR), chloramphenicol acetyltransferase-like gene a (cmlA), chloramphenicol acetyltransferase 1 gene (cat1), chloramphenicol acetyltransferase 2 gene (cat2) and chloramphenicol acetyltransferase 3 gene (cat3) which are specifically associated with resistance to florfenicol and chloramphenicol [60]. Concurrently, a concomitant study in Egypt revealed that a significant 29.3% of the isolates had integrons, primarily class 1. These integrons are intricately linked to a variety of resistance-inducing genes, such as those that confer resistance to trimethoprim (dihydrofolate reductase A1 gene (dfrA1), dihydrofolate reductase A5 gene (dfrA5), dihydrofolate reductase A7 gene (dfrA7) and dihydrofolate reductase A12 gene (dfrA12), resistance to streptomycin/spectinomycin aminoglycoside adenyltransferase A1 gene (aadA1), aminoglycoside adenyltransferase A2 gene (aadA2), aminoglycoside adenyltransferase A5 gene (aadA5), and aminoglycoside adenyltransferase A23 gene (aadA23) and resistance to streptothricin (sat2). A significant contribution to a thorough understanding of the resistome was made by further elucidation of the identification of non-integron-associated resistance genes, such as those for tetracycline (tetracycline resistance gene A (tetA) and tetracycline resistance gene B (tetB) ), ampicillin (beta-lactamase TEM gene (blaTEM)), kanamycin (aminoglycoside phosphotransferase a1 gene (aphA1)), sulfonamide (sulfonamide resistance gene 1 (sul1) and sulfonamide resistance gene 2 (sul2), and chloramphenicol (chloramphenicol acetyltransferase 1 gene (cat1) [61]. The Enterobacteriaceae family contains the genus Salmonella pullorum or Salmonella gallinarum, which is responsible for two septicemic diseases: pullorum disease (S. pullorum) and chicken typhoid (S. gallinarum). While these illnesses were common in the past, they are now uncommon in commercial chicken operations in industrialized nations [62]. All isolates obtained in 1995 were completely sensitive to twelve antimicrobials, with the exception of tetracyclines, which had resistance rates greater than 83%. Between 1995 and 2001, there was a considerable rise in the incidence of strains resistant to ampicillin (87.0%), gentamicin (56.6%), kanamycin (30.4%), enrofloxacin (93.5%), ciprofloxacin (89.1%), norfloxacin (47.5%), and ofloxacin (17.4%). Moreover, the frequency of gyrase subunit A Gene (gyrA) mutation increased (from 5.6% to 89.1%), and the Minimum Inhibitory Concentrations of enrofloxacin, ciprofloxacin, norfloxacin, and ofloxacin significantly increased [63]. A similar increase in resistance was observed between 1962 and 2010 in a separate investigation encompassing 337 strains of Salmonella pullorum from China. Interestingly, compared with the int1(–) strains, bacteria expressing int1(+) presented noticeably greater resistance to 11 of the 16 tested antimicrobial agents. Furthermore, in contrast to their biofilm-negative counterparts, subtypes that exhibited biofilm formation had noticeably greater levels of resistance to cefamandole, trimethoprim, and cotrimoxazole [64]. Advances in whole-genome sequencing have shown that a Chinese strain of S. pullorum is resistant to many drugs. It has two prophages, prophage-4 (Fels2) and ST104, which were previously found in E. coli [65].
Mobile genetic elements, namely, interon transposons, and plasmids, allow the storage of genetic information in bacterial species [30]. These constituents have the capacity to transmit genetic material from a donor bacterium of a comparable strain or species to a receiver bacterium. This technique of horizontal transmission considerably facilitates the propagation of resistance features against a wide range of antibiotics, many of which are routinely utilized in both human and animal healthcare. As a result, managing infections becomes complicated and expensive [66]. Genetic material is mostly transferred in the animal gastrointestinal system through interactions between commensal and pathogenic bacteria [67]. Moreover, these gene transfer processes take place on environmental substrates [68]. This genetic exchange has far-reaching effects, including the evolution of multidrug-resistant bacterial infections. This phenomenon has economic ramifications, including direct expenditures related to hospital stays, medical treatment, test results, and consultations, as well as indirect costs resulting from inefficient work and missed workdays [69]. Despite the financial implications, the development of multidrug resistance can have catastrophic consequences.
Antibiotic resistance is intimately linked to resistance plasmids, which may
rapidly transmit across bacteria via conjugation. These plasmids contribute
significantly to the global spread of resistance, particularly multidrug
resistance [70]. Identifying and researching R-plasmids and their bacterial hosts
is critical for understanding how antibiotic resistance spreads. However, there
are still few well described plasmids from chicken waste isolates. Aminoglycoside
resistance plasmids (arr-3 and aacA) pRKZ3 (IncQ) and pKANJ7 (IncX) have been
discovered in pig and chicken waste, respectively [71]. This investigation
revealed two IncC R-plasmids, pECmdr13.2 and pECmdr14.1, which carried genes for
resistance to tetracyclines, aminoglycosides,
The numerous bacterial illnesses discovered in chicken litter are highly likely to spread through water, air, and agricultural land. One pathogen that is particularly significant is Staphylococcus aureus, which can infect humans and spread through the air, water, and contaminated crops [72]. Clostridium botulinum poses a much greater threat to cattle because it can poison them when it swallows spores found in soil or decaying bird corpses found in chicken litter [73]. Simultaneously, Clostridium perfringens, which may be obtained from polluted soils, human and animal waste, or products such as feed components and bedding, adds another range of possible risks. Listeria monocytogenes, the cause of listeriosis, spreads mostly through the consumption of crops infected with chicken litter that has been sprayed on the soil [74]. Because L. monocytogenes sticks to plant surfaces and roots, even if it is not absorbed by plants, it can infect fruits, vegetables, and root crops in areas treated with chicken litter [75]. The possibility of bacteria leaking into subterranean drinking water systems increases the risk. In places such as Uganda, where vegetable growers frequently place raw chicken litter in wetlands, especially during dry seasons, the danger of disease transmission to public water sources, such as rivers, streams, lakes, and ponds, is significantly increased [76].
Composting, the aerobic decomposition of biodegradable organic waste, occurs within a span of 4–6 weeks, yielding a stable end product characterized by low moisture, fine texture, and an absence of odor qualities, which make it an excellent choice for use as organic fertilizer. The biodegradation of agricultural waste into compost and its incorporation into soil may increase nutrient recycling and maintain soil fertility [77]. This natural and straightforward process involves the decomposition of organic matter, particularly in the case of composted chicken litter, where minimal odors contribute to a more pleasant and environmentally friendly waste management solution. The resulting composted material surpasses fresh litter in terms of stability and consistency. Composting chicken manure has been shown to be environmentally beneficial, safeguarding ground and surface waterways from nutrient overload [13].
As a nonpathogenic waste product, composted chicken litter is easily managed, despite several drawbacks, such as nutrient loss, labor and equipment expenses, persistent odor, and the need for ample land for composting operations [78]. Challenges related to chicken manure, such as low porosity, high moisture content, low C/N ratio, and elevated pH, can be effectively addressed by incorporating high C/N ratio bulking agents such as rice husk, wood chips, and sawdust. These additions optimize the composting process by improving the C/N ratio, pile porosity, and aeration channels while reducing the water content [79]. Maintaining optimal conditions is crucial for successful composting, and the recommended parameters include a moisture level ranging from 50–60%, a C/N ratio between 25 and 30, a pH ranging from 5.5 to 9, a temperature ranging from 55–63 °C, and an oxygen content exceeding 5%. A careful balance of these composting parameters ensures an efficient decomposition process [12].
As microbial activity intensifies, the compost temperature increases to 65–68 °C, resulting in transition from mesophilic bacteria to thermophilic microorganisms. The high temperatures in this phase play a crucial role in effectively eliminating pathogens, resulting in a sanitized and safe end product [80]. Following exposure to elevated temperatures, the compost temperature subsequently decreases, allowing fungi to proliferate and effectively decompose organic matter over three to six months. The end product is a stable humic material that is rich in nutrients and safe for use as fertilizer [81]. Composting is increasingly preferred for managing solid waste because of its effectiveness in promoting environmentally sustainable practices and minimizing landfill impacts [82]. The incorporation of compost into soil enhances soil organic matter, structure, agricultural productivity, and overall plant development, contributing to increased crop yield and improved soil health, thus playing a pivotal role in enhancing food security [83]. The optimization of compost over synthetic fertilizers also contributes to increased soil biodiversity and reduces the risk of nutrient leakage into the environment, highlighting its sustainable and eco-friendly aspects in agriculture [84].
Additionally, compared with stable compost, composting raw organic waste, such as raw chicken manure, is more manageable for storage and transportation [85]. The incorporation of cereal straw with easily decomposable carbon can effectively minimize nitrogen loss in aerobic composting processes. Applying this technique to chicken waste, combined with phosphate rock and elemental sulfur, produces a nutrient-rich and environmentally friendly fertilizer suitable for supporting organic farming techniques.
The use of chicken litter in long-term no-tillage systems has been found to increase soil organic carbon and fertility [86]. Chicken litter can supplement crop production and support a diverse soil microbial community when used sparingly to fulfilled plant needs for phosphate and nitrogen [87]. Continuous chicken litter application can increase soil P levels, leading to runoff and eutrophication of aquatic bodies. The application of chicken litter may have little effect on surface soil quality, but it has no effect on subsurface soil quality [88]. The use of chicken litter as an organic fertilizer resulted in a considerable increase in soil organic matter, which improved the soil physicochemical qualities, notably pH and base saturation. As Soil Organic Matter (SOM) decomposes, it becomes the dominant source of negative charges in most soils, affecting the ability of the soil to hold and make cations and nutrients including calcium, potassium, magnesium, and ammonium accessible. As a result, continual application of chicken litter increased both pH and base saturation because higher pH values release negative charges in the soil, which are subsequently filled by base cations, resulting in a rise in base saturation owing to a decrease in hydrogen ions in the solid phase of the soil [89]. The use of a higher dosage of chicken litter resulted in a considerable increase in microbial biomass nitrogen. This improvement can be attributed to the high nitrogen concentration of organic fertilizers obtained from animal excreta [90] which not only increases soil nitrogen retention but also enables temporary immobilization by microbial biomass. The effects of chicken litter compost on soil physical and chemical properties are explained in the Table 4 (Ref. [91, 92, 93]), Table 5 (Ref. [94, 95, 96]).
| Physical property | Description | Significance for plant growth and soil health | References |
| Bulk density | Reduced | Improved soil aeration, root penetration, and drainage. | [91, 92] |
| Water holding capacity | Increased | Enhanced plant resilience to drought stress and reduced irrigation requirements. | [91] |
| Aggregate stability | Enhanced | Improved soil structure and reduced susceptibility to erosion, particularly on sloping land. | |
| Oxygen diffusion | Improved | Facilitates optimal root respiration and soil microbial activity. | |
| Total porosity | Increased | Enhances air and water movement within the soil, crucial for plant growth and microbial activity. | [93] |
| Field capacity moisture content | Increased | Increased soil water storage capacity. |
| Chemical property | Description | Significance for plant growth and soil health | References |
| pH | Slight Increase (Variable) | Some plants may be able to get more nutrients, but this might affect microbial activity. Numerous variables, including the starting pH and kind of soil, influence the degree of change and impact. | [94] |
| Organic Carbon Content | Increased | Increases soil structure, water retention ability, and nutrient cycling to improve soil fertility and give plants a steady supply of nutrients. | [95] |
| Total Nitrogen Content | Increased | Increases the amount of nitrogen that is accessible to plants, which is an essential nutrient for the growth and development of plants. | [95] |
| Available Phosphorus Content | Increased | Increases the amount of easily absorbed phosphorus that is available for plant absorption, which is necessary for a number of physiological functions in plants. | [95] |
| Nitrogen Mineralization | Slower Release (Composted Manure) | Chicken dung that has been composted releases nitrogen more slowly than fresh manure, which may lessen the chance of excessive nitrogen leaking into the environment and encourage steady plant development. | [96] |
The addition of a small amount of elemental sulfur to the composting process mitigates ammonia volatilization loss and enhances compost with essential phosphorus and sulfur nutrients, emphasizing the importance of precise nutrient management in optimizing compost quality and effectiveness [97, 98]. When supplements such as alum and zeolite are added, ammonia volatilization decreases, and phosphorus in chicken litter becomes less soluble. These additions not only address environmental issues but also improve the quality of life for workers and birds by lowering ammonia levels and optimizing the nitrogen to phosphorus (N:P) ratio for crop uptake, making it a financially and environmentally sound option for chicken producers [99]. The composting process is explained in Fig. 2.
 Fig. 2.
Fig. 2.
Composting of chicken waste.
In anaerobic digestion, a diverse array of bacteria and substrates are utilized under strict anaerobic conditions, characterized by an oxidation-reduction potential below 200 mV. This intricate process converts organic materials into biogas, which is primarily composed of methane and carbon dioxide, demonstrating the potential of anaerobic digestion as an environmentally sustainable method for organic waste management and renewable energy production [9]. The anaerobic digestion process comprises four essential steps: hydrolysis, acidogenesis, acetogenesis, and methanogenesis with specific microbes playing roles in syntrophic interrelationships under varying environmental conditions [100].
During hydrolysis, complex organic compounds such as lipids, polysaccharides, proteins, and nucleic acids are broken down into water-soluble monomers, facilitated by exoenzymes from hydrolytic bacteria such as cellulase, cellobiase, xylanase, and lipase. During acidogenesis, facultative and obligatory anaerobic bacteria further degrade monomers into short-chain organic acids, alcohols, hydrogen, carbon dioxide, and volatile fatty acids [100]. In the anaerobic digestion process, the concentration of hydrogen ions becomes pivotal, influencing the diversity of generated products. The acidogenic stage, which is a precursor to the acetogenic phase, involves the continuous conversion of the exergonic combination of hydrogen and carbon dioxide into acetic acid by homo-acetogenic bacteria [101]. This stage is notable for the production of acetic acid and the transformation of organic acids and alcohols into acetate, a vital substrate for methane-forming bacteria. Acetogenic bacteria engage in symbiotic interactions with methane-forming bacteria, gaining heightened importance in the final stage of anaerobic digestion, known as methanogenesis. Two types of bacteria play essential roles: acetotrophic bacteria reduce acetate to methane and carbon dioxide, whereas hydrogenotrophic methanogens consume hydrogen to produce methane (Fig. 3). This intricate succession of stages underscores the dynamic and interconnected nature of anaerobic digestion, emphasizing the collaboration of various microbial communities in converting organic matter into valuable end products.
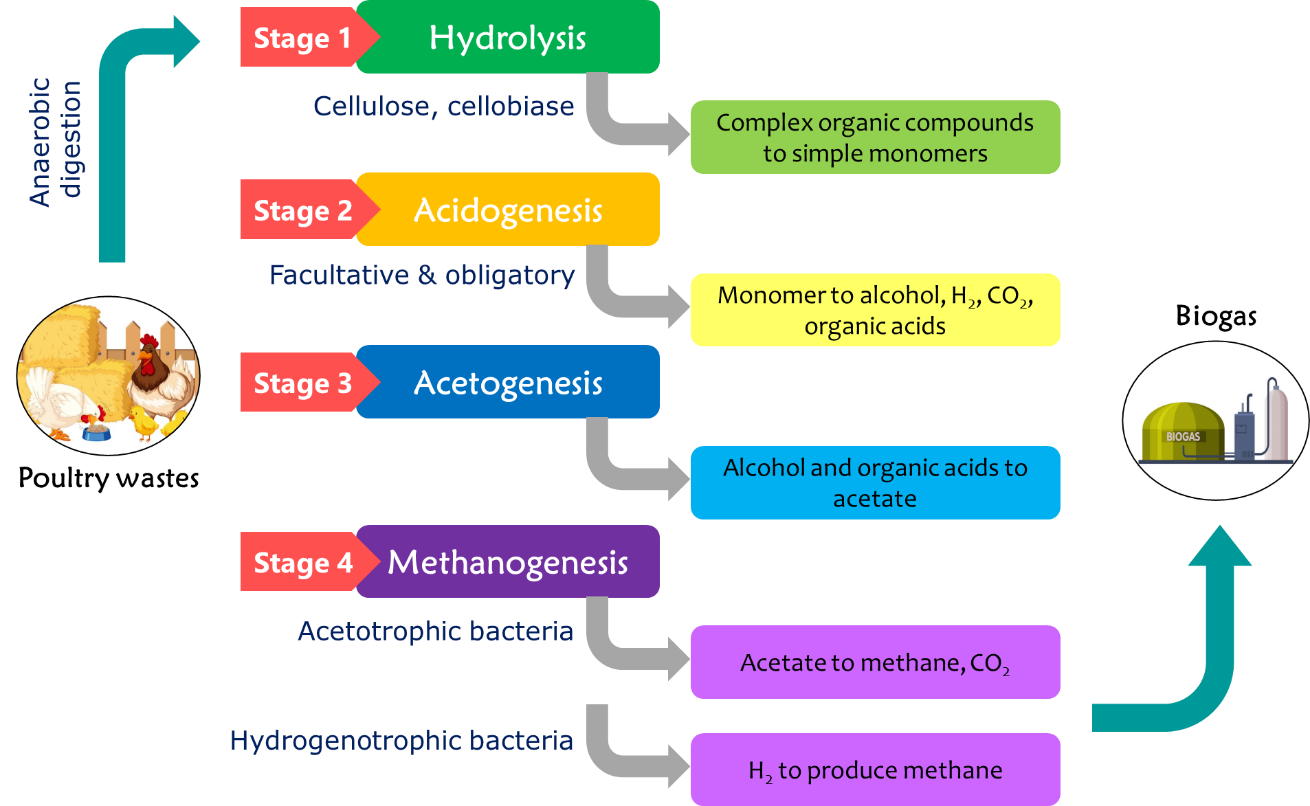 Fig. 3.
Fig. 3.
Anaerobic digestion of chicken waste.
Psychrophilic temperature conditions in anaerobic digestion have been shown to inhibit microbial growth and biogas production, whereas thermophilic settings may result in the production of less biogas due to methanogenic processes caused by volatile gases such as ammonia. Mesophilic temperatures are preferred for ensuring the stability of biogas generation, especially when chicken waste is utilized as the primary feedstock for anaerobic digestion [102, 103]. The specific pH requirements are crucial throughout the anaerobic digestion phase, with hydrolysis requiring a pH range of 4.0–5.0 and acidogenesis and methanogenesis requiring pH ranges of 5.0–6.5 and 6.8–7.5, respectively. The optimal pH range for anaerobic digestion in the context of chicken waste is 6.5–7.5, necessitating proactive pH modifications to chicken waste before the anaerobic digestion process can be started for effective biogas production [104, 105, 106]. The Organic Loading Rates (OLR) is a crucial metric for assessing the biological capacity of an anaerobic digestion system and represents the quantity of raw material fed to the digester per unit volume in a day. Excessive loading can disrupt the digestion process due to acid accumulation, impacting biogas production efficiency. While higher OLRs offer cost and space advantages, careful calibration is necessary to prevent issues associated with insufficient retention durations, maintaining a balance critical for optimizing the economic and operational elements of anaerobic digestion systems [107, 108].
Retention time, intricately linked to microbial growth rates, is significantly affected by factors such as OLR, substrate composition, and process temperature. Compared with other bioenergy sources, anaerobic digestion is considered a more environmentally friendly and energy-efficient process [109, 110]. The study indicated that higher OLRs in anaerobic digestion systems exclusively fed chicken manure under thermophilic conditions produced significantly less methane due to ammonia inhibitory effects [111]. Maintaining an appropriate carbon-to-nitrogen ratio of 20–30 or 20–35, with 25 as the optimum, is recommended for effective anaerobic digestion. The introduction of carbon adjuvants enhances the C/N ratio, increases protein solubility, reduces total ammonium nitrogen and free ammonia nitrogen concentrations in the system [112, 113]. Anaerobic digestion, a microbial conversion process, efficiently treats chicken waste, especially waste with a high water content, without the need for pre-treatment steps [114]. Compared with individual waste digestion, Co-digestion of chicken waste with other manures can increase biogas production, demonstrating the viability and benefits of waste digestion [115].
Pyrolysis is defined as the thermochemical breakdown of biomass in an oxygen-free environment and consist of three critical steps: (1) The biomass is warmed by heat from a heat source; (2) the biomass undergoes pyrolysis as a result of the high temperature; and (3) vapor phase products are produced [116].The influencing factors of the heating rate, pressure, residence duration, peak temperature, heat transfer rate, and vapor-solid interactions all play critical roles in shaping the outcome of the pyrolysis process [117]. Pyrolysis processes have the ability to reduce the solid waste volume by 80–95% while also converting it into diverse fractions, such as bio-oil, charcoal, and pyrolysis gas. Bio-oil, a liquid product that includes both aqueous and organic phases, is an important by-product of the pyrolysis process.
There are two primary types of pyrolysis processes: slow and quick pyrolysis. Slow pyrolysis is characterized by minimal heating rates (25–120 °C/min) and prolonged residence times (15–36 min), which mostly produce biochar as the main product [118]. Fast pyrolysis, on the other hand, produces more liquid and non-condensable gases since it works at faster heating rates (600 °C/s) and shorter residence times (0.5–5 s) (Fig. 4) [119]. Biochar is the principal product of both slow and quick pyrolysis of chicken manure; this is mainly explained by the high ash content of both the manure (14–22 weight percent) and the resulting biochar (42–66 weight percent) [120]. Slow pyrolysis is frequently chosen as the best technique for turning agricultural waste into biochar, as it typically produces a relatively large volume of biochar [121]. The impacts of chicken litter derived biochar on soil nutrients are explained in Table 6 (Ref. [122, 123, 124, 125, 126, 127, 128, 129, 130, 131]). The oil from fast pyrolysis of chicken litter did not meet biodiesel engine specifications because of the high water content (26 wt.%) that hinders ignition (upper limit 0.5 wt.%) and the very high total acidity of 38–46 mg of KOH/g (maximum limit 0.5 mg of KOH/g). Similarly, the total amount of inorganic trace elements (ash) in chicken litter pyrolysis oil was 10 times greater than the top permitted limit (0.02 wt.%) [132].
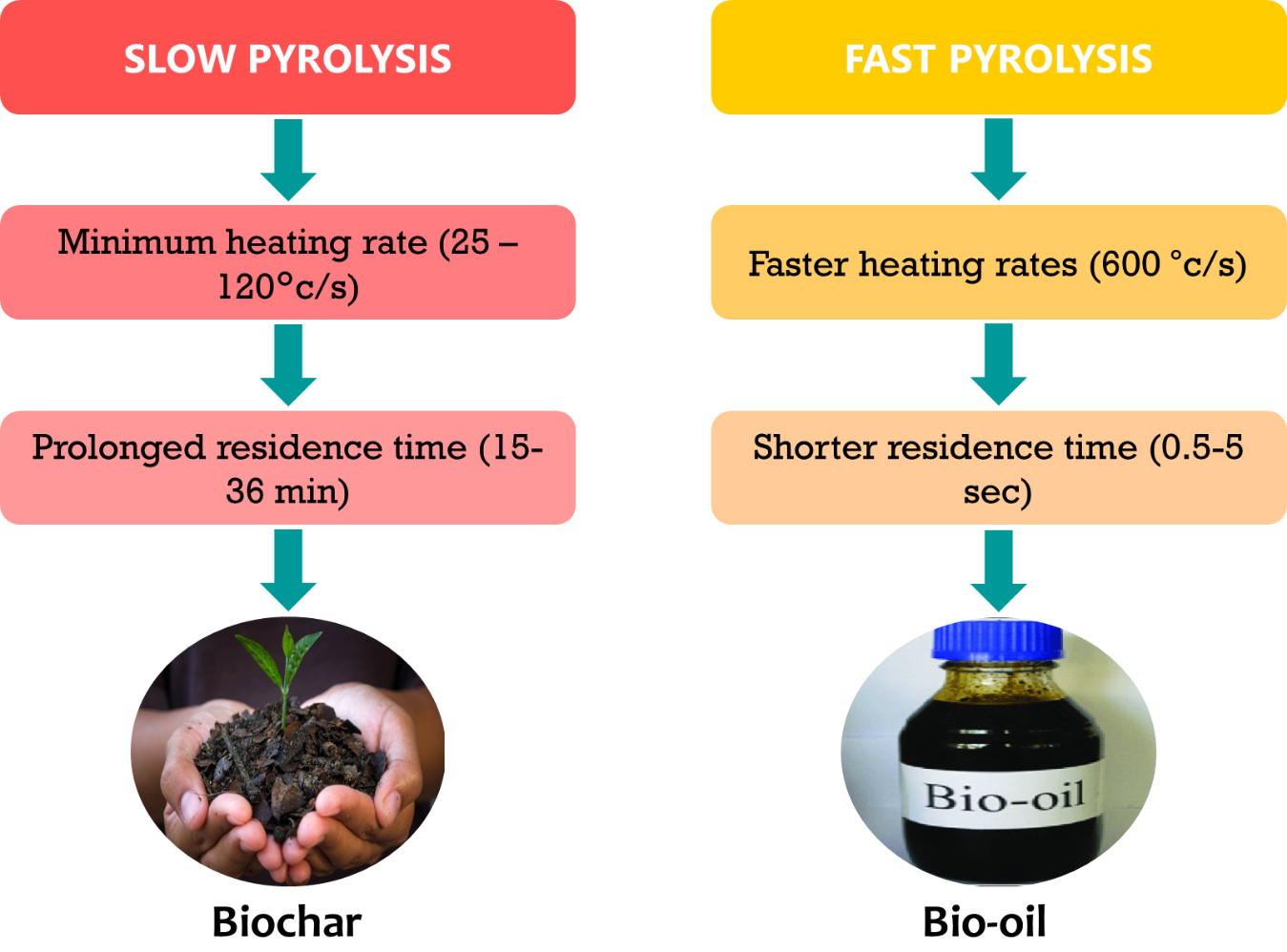 Fig. 4.
Fig. 4.
Pyrolysis of chicken waste.
| Element | Impact of Chicken Litter Biochar | Reference |
| Nitrogen | May be not a major direct contributor to the available N. Through the priming action, it increases the mineralization of native soil N, but it also loses N during pyrolysis because of volatilization at higher temperatures. | [122, 123, 124] |
| Organic Matter | Soil organic matter increases with high levels of organic carbon. A higher SOM helps to increase the water-holding capacity and fertility of the soil. | [125] |
| Phosphorus | When compared to raw chicken litter, the concentration of P is higher. Pyrolysis may increase the amount of plant P that is accessible. | [126, 127] |
| Potassium and Calcium | Enhances the possible availability of K and Ca in plants. | [128, 129] |
| Carbon Sequestration | Increased soil C sequestration results from inherent resistance to breakdown. Moreover, by eliminating atmospheric CO2, it lessens the effects of climate change. | [130] |
| Sulfur and Iron | Pyrolysis changes S and Fe into less soluble forms, which may have an effect on the availability of plants. | [131] |
SOM, Soil Organic Matter.
Biochar generated from chicken waste at 460 °C has a unique composition which is rich in phosphate chemical species due to its excellent availability of phosphorus [14]. This type of biochar is useful in agriculture because it acts as a slow-release fertilizer that ensures regulated and extended nutrient delivery. Its specific characteristics make it suitable for improving soil fertility, particularly under slightly acidic conditions, while also functioning as a phosphorus storage alternative. Compared with higher associated with burning, lower pyrolysis temperatures of 350–500 °C have a more pronounced positive impact on nutrient availability, making low-temperature pyrolysis a favorable and practical approach for the thermal treatment of chicken manure due to its improved nutrient preservation and overall resource efficiency [133].
Gasification is a thermochemical process in which gasifying agents such as carbon dioxide are used to convert carbon-rich feedstock into a combustible product gas. This complex process uses chemical reactions driven by gasifying agents to convert carbonaceous material into a gaseous mixture containing flammable components [134]. A carbon-based feedstock undergoes partial oxidation at elevated temperatures ranging from 700 to 1200 °C during the gasification process, which is aided by the presence of an oxidant, which can be air, steam, oxygen, or a combination of both. Under substoichiometric circumstances, this thermochemical process produces a flammable gas known as a “producer gas”, “product gas”, or “syngas”. This gas is mostly composed of carbon monoxide, hydrogen, carbon dioxide, methane and trace chemicals. However, contaminants such as tiny particles, tars, and alkali metals may be present [135].
Syngas, an abbreviation for synthesis gas. The generation of syngas, which is primarily composed of hydrogen, carbon monoxide, carbon dioxide, and methane with trace amounts of low molecular weight hydrocarbons, makes it a flexible fuel source that can provide both heat and power [136]. Through catalytic conversion or anaerobic fermentation, syngas may be processed to yield important biofuels and chemicals. Significant syngas reforming processes, such as the steam reforming of methane and heavier hydrocarbons or water-gas shift reactions catalyzed by iron or nickel catalysts at temperatures between 200 and 1100 °C and pressures between 1 and 30 bar, can notably increase the generation of hydrogen [137].
This gas is essential as a building block in the production of many fuels and chemicals. Because of its versatility and ability to undergo further chemical transformations, syngas is an important intermediate in the production of a diverse range of synthetic materials. These include fuels such as synthetic natural gas and methanol, as well as a variety of chemical compounds widely used in industrial applications [11]. Gasifying biomass is a more ecologically friendly process than traditional combustion, with a significant reduction in gaseous pollutants such as sulfur oxides, nitrogen oxides, heavy metals, and particle emissions. This cleaner technique not only complies with existing emission limits but also has potential as a more sustainable and environmentally friendly option for harvesting biomass resources. The first stage in the gasification process involves drying the feedstock, which requires the evaporation of both free and bound water. This drying phase is crucial because it sets the stage for later reactions, and the requisite heat is frequently provided by exothermic reactions in the next phases. The temperature during this drying cycle normally varies between 100 °C and 200 °C, which is insufficient to thermally breakdown the feedstock. This temperature range is mostly used to evaporate water from the feedstock. However, it is vital to note that the temperature at this point is insufficient to perform more difficult activities [138]. Drying is essential for high-moisture feedstocks because it reduces agglomerate formation and jamming, which are common issues with wet feedstocks like chicken manure. Without it, the product gas heating value decreases, resulting in a significant increase in tar content [139].
The steps of gasification continue from drying to devolatilization, where high temperatures without oxygen further degrade feedstock particles into volatile matter and carbonaceous solid residue known as “biochar” [140]. The process subsequently enters the combustion phase, which involves the oxidation of carbonaceous material, creating byproducts such as H2O, CO2, CO, and H2, which are required for subsequent processes [138]. This phase, which occurs at temperatures ranging from 700 to 1500 °C, is critical because it impacts the endothermic drying and pyrolysis phases, as well as the subsequent reduction process. Following the combustion stage, the process progresses to the reduction step where the temperature ranges from 800 to 1000 °C. This step is critical because it converts the remaining combustion products into the final combustible gas, which is mostly composed of hydrogen and carbon monoxide [141]. Biochar interacts with water, carbon dioxide, and hydrogen from the previous stage more easily in the reduction stage, producing a combustible gas mixture that includes carbon monoxide, carbon dioxide, hydrogen, methane, and light hydrocarbons such as ethylene and acetylene. Comparative reactivity analysis, revealed that biochar has a greater affinity for water than for carbon dioxide [142].
Gasification study in the United States has revealed that chicken litter with a moisture content of 25% can have an energy value of approximately 6.75 MJ/kg. The amount of gas produced is approximately 1.15 Nm3/kg of litter, with a caloric value of 5.4 MJ/kg [143]. The electricity generated from chicken litter is estimated to be approximately 0.5 kWh/kg. Chicken litter also contains high levels of vital nutrients. Although there has been little investigation into the potential value of char created during gasification, it is still a possible consequence. However, the use of char as a feedstock may decrease the overall energy efficiency of the gasification process. Additional study is needed to investigate the characteristics and economic sustainability of char generation from chicken litter [144].
Combustion is the simplest form of thermochemical conversion. For centuries, direct biomass combustion has been utilized to generate electricity and heat, however, the overall efficiency of this process is low, and combustion can produce gaseous pollutants [145]. To define the combustion of agricultural waste, including chicken litter, basic information such as the substance composition, especially its moisture level, is needed [146].
The combustion process involves several steps: the drying process removes residual moisture from chicken litter at 100 °C. Torrefaction subsequently begins at 300 °C, devolatilizing the litter and releasing organic compounds. As the temperature increases, cellulose and lignin are fractionated. Gasification occurs at 800 °C, converting solid char into a syngas of carbon monoxide and hydrogen. Char combustion follows, transforming oxidized char into carbon dioxide and carbon monoxide gases. Finally, above 800 °C, the oxidation process converts volatile gases and tar into heat energy [144].
The low ash fusion temperature of chicken litter might cause operational issues for typical grate combustion systems. To ensure maximum efficiency, careful management of combustion parameters such as temperature, air mixture, and moisture content is required within prescribed limits, which may vary depending on the unique combustion design [8]. The method produces an ash residue containing a large amount of the phosphate and potassium contained in the original chicken litter. While the initial nitrogen content may differ, nitrogen loss to the environment as NOx during combustion is typically not a substantial problem [8]. The ash created is stable, sterile, and easier to maintain and transport than typical chicken litter. Furthermore, it is more marketable as organic fertilizer. Efficient chicken litter combustion is dependent on the moisture content, volatile matter content, ash content, and mineral composition of the fuel feed [147]. While incineration plants can help limit trash buildup, the physicochemical qualities of litter, bedding materials, and incineration environments all affect the stability and completeness of chicken litter combustion [148].
A combustion system originally built for coal fuel may be adjusted to use chicken litter as a biomass alternative. By injecting chicken litter into a furnace and burning it at high temperatures, a boiler can efficiently transform water into steam via combustion heat. This steam may then be used to create other energy, such electricity [149]. However, the high chloride concentration of chicken litter can cause corrosion in boilers, particularly in systems that are not expressly designed for chicken litter, resulting in significant increases in maintenance costs [150]. While the combustion process has the potential to assist the environment, there are certain problems. Emissions of gases including NOx, SO2, CO, and CO2 can contribute to acid rain and greenhouse gas emissions. Furthermore, particulate matter and odours have a negative influence on air quality [151]. To counteract these environmental consequences, air pollution management techniques such as particle control systems, moist gas systems, and CO2 capture systems must be implemented [152]. On a positive side, the ash created may be used as fertilizer owing to its phosphorus and potassium concentrations [153].
There is doubt that chicken farming affects the physical and chemical properties of soil to a great extent. Owing to physical changes in the soil, exposure to chicken waste potentially increases the strength of the soil by acting as a cement soil aggregate. Additionally, the presence of hydrogen sulfide and ammonia in chicken waste makes the soil decomposable in nature and improves the suitability of the soil for sowing as it increases the water holding capacity [154]. Moreover, the exposure of soil to chicken waste can reduce soil erosion, weathering and other environmental effects, which greatly affect the physical properties of the soil.
Studies have shown that exposure to different kinds of waste affects soil properties in different manners. The most important effect of chicken waste on the chemical properties of the soil is that it drastically increases the concentrations of nitrogen, calcium, phosphorous, magnesium, potassium and other organic matter, which not only increases the quality of the soil but also helps in the production and yield of crops [155]. Moreover, the use of chicken waste on soil sometimes becomes a concern because such waste could also contain metals such as zinc, which not only have environmental effects but also reduce the chemical fertility of the soil. Interestingly, chicken manure, which forms part of chicken waste, is alkaline; thus, chicken manure can be used to treat the acidic nature of soil, which can ultimately help increase its fertility [156].
When evaluating soil quality, crop yield is frequently the only factor taken into account. As a result, soil chemical characteristics have historically received too much attention since they were frequently determinants that limited crop productivity [157]. For many years, chicken farming waste has been used as fertilizer in fields because of its high potassium, ammonia, phosphorous, nitrogen and many other organic matter contents, which enhances the fertility of the soil and potentially aids in the growth of crops. Furthermore, appropriate use of chicken waste increases the water retention of the soil. In addition, chicken waste also contains various kinds of microorganisms that are beneficial for the growth of crops [155].
Moreover, if the use of chicken waste on farms is not managed properly, then it could negatively affect the production and yield of crops. The overuse of waste can increase the level of nutrients in excess, which not only contributes to harmful environmental effects but also adversely affects the growth of plants. In addition, the overuse of chicken waste in the field could also increase the number of microorganisms, which may sometimes cause the growth of harmful parasites and various plant-based diseases [158]. Thus, before chicken waste is used in farming, it is essential to ensure that the organic content of such waste, such as chicken manure and other organic matter, is utilized on farms to increase the concentration of elements such as phosphorous and nitrogen in the soil, which ultimately helps in crop production.
The application of chicken manure has a variety of beneficial effects on soil properties. Adsorption capacity is accompanied by an increase in soluble P and phosphorus desorption [159]. This dual action not only increases the availability of nutrients but also highlights the potential contribution of chicken manure to the nutrient cycle of the soil ecosystem. Scientists have repeatedly emphasized the effectiveness of chicken manure in increasing available nitrogen levels [160, 161]. In addition, wheat and rice have shown a subtle improvement in their absorption of vital micronutrients, particularly zinc and iron. These findings are further supported by the observations of More and Ghonsikar [162], who explicitly reported a significant increase in wheat phosphorus content as a consequence of the prudent use of superphosphate and chicken manure. Chicken manure appears to be a useful resource for maximizing crop yields in larger agricultural settings. The concentrations of nitrogen, phosphorus, potassium, iron, manganese, and copper in faba beans significantly increase when chicken manure is applied [163]. The favorable effects of chicken manure on a variety of crops further demonstrate its adaptability as an agricultural input. The use of chicken manure resulted in increased onion bulb yields exceeding 35 t ha-1 and the combined use of mineral fertilizers and chicken manure produced the best tomato yields in terms of quantity and marketability [164].
Chicken and swine sparingly produce impressive tomato yields of 10 t ha-1 in the unique environment of Nigerian ferralitic soils [165]. These findings emphasize how chicken manure can adapt to a wide range of soil conditions and greatly increase crop output. Furthermore, a mixture that consists of 75% bio-digested chicken manure slurry and 25% recommended commercial fertilizer is effective [166]. This highlights the possibility of chicken manure being used as a substitute or complement to commercial fertilizer in the cultivation of sweet corn, which has important ramifications for economic and ecological farming methods. With respect to the use of recycled and composted chicken manure, the benefits of using sustainable agriculture techniques are important. This leads to increased rice grain yields, demonstrating the function of chicken manure as a source of nutrients as well as a factor in soil fertility and crop production. Obi and Ebo [167] provided strong evidence for the beneficial effects of using chicken manure in southern Nigeria. According to their research, the use of 100% chicken manure significantly increases the average grain yield of maize. Moreover, applying 50% inorganic fertilizer and 50% chicken manure to damaged soils increases crop output even more. The combined data from several studies highlight the extensive benefits of chicken manure on crop yields, nutrient availability, and soil physical characteristics. Chicken manure is a great resource for sustainable and effective agriculture since it can be applied to different types of soil and may either replace or augment commercial fertilizers.
Among the various waste treatment alternatives, composting stands out as the most sustainable, accessible, and cost-effective technique for transforming chicken litter from a threat to the environment to a beneficial resource. The composting of chicken waste is an economical and environmentally friendly way to handle chicken waste. It is a financially appealing choice because of its cheap operating costs, lower disposal costs, and possibilities for revenue generation through compost sales. Its sustainability is further enhanced by the long-term advantages of better soil health and less dependency on chemical fertilizers. The resulting compost greatly improves soil health and agricultural output since it is rich in organic matter and vital nutrients. Because of its natural composition, which is in line with modern sustainable farming methods, it is an important resource for both chicken farmers and the larger agricultural community. It increases agricultural output by supplying vital nutrients, strengthening the soil structure, encouraging microbial activity, improving water retention, reducing soil compaction, and inhibiting plant diseases. A reduced need for synthetic fertilizers, improved crop yields, and better soils can all be achieved by using this organic fertilizer in farming techniques. Together, these elements make composting chicken waste an advantageous option for chicken farmers. By promoting composting as a key component of environmental stewardship, the sector not only increases agricultural output but also promotes long-term ecological well-being. This comprehensive strategy not only improves waste management but also demonstrates the industry’s commitment to sustainability and its role in creating a healthier environment for current and future generations. Owing to the development of new techniques, the future of chicken waste management is bright. Techniques such as precise nutrient recovery, thermochemical conversion, and improved composting have great promise. Integrated systems incorporating pre-treatment, cascading usage, and circular agricultural concepts are necessary to accomplish efficient waste management. Transparent policies that include carbon market participation, subsidies, and tougher restrictions are likely to encourage the use of these techniques. Furthermore, the diffusion of information via cooperative research, farmer education, and consumer education is crucial. The chicken industry may efficiently turn trash into a useful resource and ensure a sustainable future by adopting this holistic strategy.
MPH — Conceptualized the idea and writing of the original draft; ST, DS, RA — Conceptualization, Supervision, review, revision and editing; MT, KS and DJ — Conceptualization and Review. All authors contributed to editorial changes in the manuscript. All authors reviewed and approved the final version of the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
The authors acknowledge the Tamil Nadu Agricultural University, Coimbatore, Tamil Nadu, India for providing library and web source facilities for the review paper.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




