1 Department of Cardiology, Zhongshan Hospital of Xiamen University, School of Medicine, Xiamen University, 361000 Xiamen, Fujian, China
2 Central Clinical Laboratory, Zhongshan Hospital of Xiamen University, School of Medicine, Xiamen University, 361000 Xiamen, Fujian, China
Academic Editor: Salvatore De Rosa
Abstract
Background: The role of soluble interleukin-1 receptor type 2 (sIL-1R2)
in acute myocardial infarction (AMI) remains undocumented. In the present study,
we aimed to evaluate the possible associations of sIL-1R2 with left ventricular
(LV) function, remodeling and future clinical events in the setting of AMI.
Methods: Circulating sIL-1R2 levels were quantified after percutaneous
coronary intervention (PCI) on day 1 of hospital admission for 204 AMI patients,
and upon enrollment of 204 healthy controls. Echocardiography was conducted in
the acute phase and at 12-month follow-up. Adverse clinical events were
registered after 12 months. Results: Circulating sIL-1R2 levels were
significantly higher in AMI patients than in healthy controls (medians
respectively 6652.81 pg/mL, 3799.13 pg/mL, p
Keywords
- acute myocardial infarction
- inflammation
- interleukin-1 receptor type 2
Acute myocardial infarction (AMI) is a leading contributor to morbidity and mortality worldwide [1]. Inflammation plays a pivotal role in the development of atherosclerotic plaques, as well as acceleration of plaque rupture and local thrombosis [2]. Inflammation is a double-edged sword. Although the post-AMI inflammatory response is prerequisite for normal healing of damaged heart tissue, excessive inflammation is associated with maladaptive left ventricular (LV) remodeling, progressive heart failure, and ultimately adverse clinical outcomes [3]. Thus, inflammation in AMI has potential as a therapeutic target.
Interleukin-1 (IL-1) plays a central role as a mediator propagating the
inflammatory response and is considered the main target in atherosclerotic
thromboprotection [4]. Two proteins, IL-1
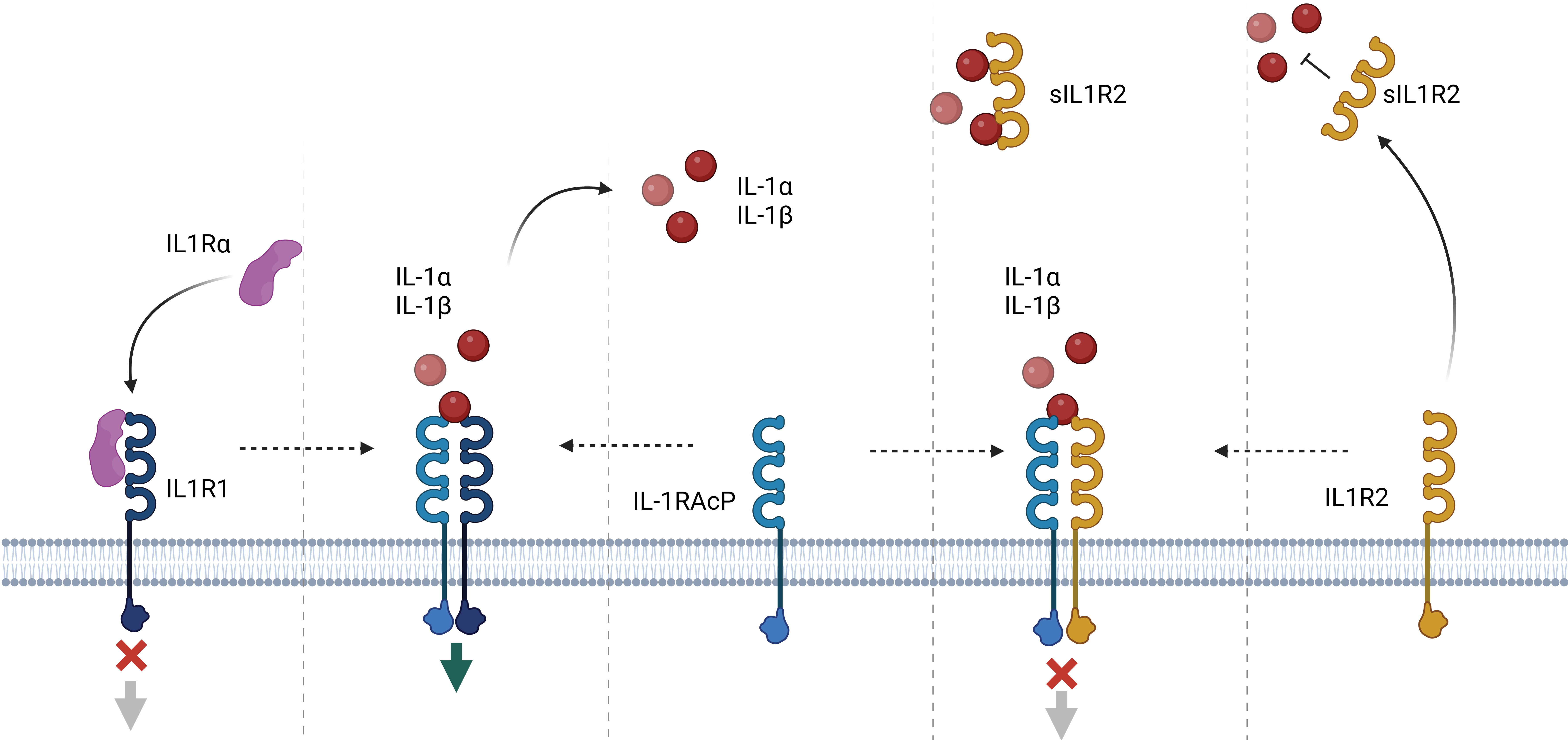 Fig. 1.
Fig. 1.The IL-1 system. Activation. The main extracellular
soluble activators IL-1
We hypothesize that sIL-1R2 may be an inflammatory indicator associated with LV remodeling after AMI. Using blood sampling and repeated echocardiography, we aim to assess the possible associations between sIL-1R2 and LVEF, ventricular remodeling and adverse clinical events.
Patients with AMI symptom duration
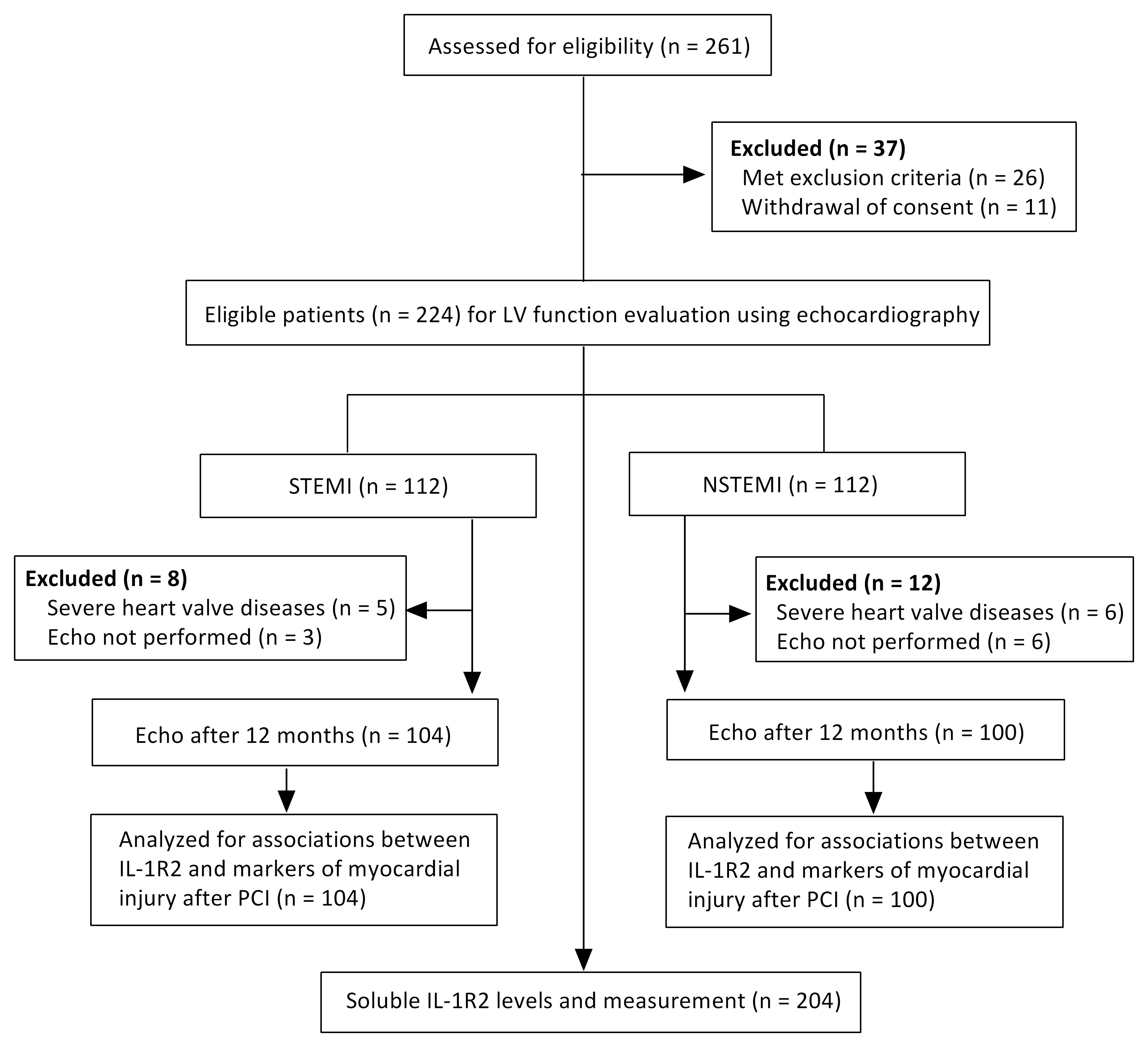 Fig. 2.
Fig. 2.Study flow diagram. Echo, echocardiography; PCI, percutaneous coronary intervention.
Blood samples for the analysis of sIL-1R2 were drawn from healthy controls (n = 204) upon enrollment and from AMI patients at day 1 following the PCI procedure, median 23.8 h after the onset of AMI (n = 204). All samples were centrifuged at 4000 rpm for 10 min at 4 °C to obtain plasma samples and then stored at –80 °C pending further analysis.
To determine plasma levels of sIL-1R2, we used human IL-1R2 enzymelinked
immunosorbent assay (ELISA) kits from R&D Systems (DY263, Minneapolis, MN, USA).
Levels of N-terminal pro-B-type natriuretic peptide (NT-proBNP) on hospital
admission were measured using an enzyme immunoassay (Roche Diagnostics, Mannheim,
Germany), while high-sensitivity cardiac troponin T (TnT) was determined on an
Elecsys 2010 analyzer (also from Roche Diagnostics). C-reactive protein (CRP)
tests and other routine biochemical analyses were performed by routine laboratory
assays daily or every other day. The maximum values of CRP and TnT measured
during hospitalization were defined as the peak CRP and the peak TnT,
respectively. The intra- and inter-assay coefficients of variation were
We performed echocardiography within 2 days after PCI and after 12 months using a Vivid E9 scanner with a phased-array transducer (M5S) (GE Ultrasound, Horten, Norway). Endocardial boundaries were outlined in the four-chamber and two-chamber sections to calculate the volume parameters, including LV end-systolic volume (LVESV) and left ventricular end-diastolic volume (LVEDV). The biplane Simpson method was adopted to calculate LVEF. Echocardiographic images were collected by 2 experienced radiologists who were unaware of the patient’s clinical data. Radiologists checked each other and made decisions together to improve diagnostic consistency.
The Kolmogorov-Smirnov test was used to analyze the distribution of data. For
continuous variables, the median (interquartile range) was used for statistical
description, and the Mann-Whitney U test and Kruskal-Wallis
tests were used for intergroup comparison. Categorical variables were described
in the form of counts (%), and their intergroup comparisons were assessed by the
chi-square test. Associations between sIL-1R2 and clinical variables were tested
by Spearman’s correlation. Levels of sIL-1R2 were analyzed in
logistic regression analyses with adverse LV remodeling, worsened LVEF and
reduced LVEF as binary responses. Adverse LV remodeling was defined as LV
dilatation (LVEDV increase of
A total of 261 AMI patients and 204 healthy controls were evaluated (Fig. 2). Samples from patients were acquired on day 1 after PCI (median 23.8 h after the onset of AMI). Patients were dichotomized by the median expression value for sIL-1R2. Samples from controls were acquired upon enrollment. Clinical characteristics are presented in Table 1. AMI patients had a greater proportion of smokers, drinkers, essential hypertension, hypercholesterolemia and diabetes. Patients with low sIL-1R2 levels had significantly lower admission neutrophil levels than patients with high sIL-1R2 levels.
| Healthy controls (N = 204) | All Patients (N = 204) | sIL-1R2 |
sIL-1R2 | ||
| Baseline characteristics | |||||
| Age, years | 62 (29–92) | 62 (32–94) | 63 (36–88) | 62 (32–94) | |
| Gender, male, n (%) | 157 (77.5) | 157 (77.5) | 77 (75.5) | 80 (79.4) | |
| BMI, kg/m |
24.5 (16.6–31.7) | 24.5 (17.6–32.9) | 24.3 (18.8–29.4) | 24.7 (17.6–32.9) | |
| Essential hypertension, n (%) | 88 (43.1) | 107 (53.4) |
58 (57.8) | 49 (49) | |
| Hypercholesterolemia, n (%) | 28 (13.7) | 130 (63.7) |
64 (62.7) | 66 (64.7) | |
| Diabetes mellitus, n (%) | 25 (12.3) | 79 (39.7) |
42 (42.1) | 37 (37.3) | |
| Current smoker, n (%) | 76 (37.3) | 103 (51) |
50 (49.0) | 53 (52.9) | |
| Alcohol consumption, n (%) | 37 (18.1) | 65 (32.4) |
32 (31.4) | 33 (33.3) | |
| Clinical characteristics | |||||
| STEMI, n (%) | ̵̶ | 104 (51.0) | 53 (52.0) | 51 (50.0) | |
| Triple vessel lesion, n (%) | ̵̶ | 29 (17.3) | 14 (17.0) | 14 (17.6) | |
| Biochemical analyses | |||||
| Peak troponin T, ng/L | n.d. | 3649.4 (126.1–10,000) | 3221.8 (126.1–10,000) | 4077 (134.0–10,000) | |
| NT-proBNP, nmol/L | 122.3 (5–496) | 2747.9 (10–35,000) |
3021.1 (23.1–29,571) | 2474.7 (10–35,000) | |
| CRP, mg/L | 3.6 (0.2–38.4) | 30.1 (0.2–194.2) |
28.9 (0.2–194.2) | 31.3 (0.4–179.5) | |
| WBC, 10 |
6.8 (3.1–11.9) | 10.2 (3.1–24.6) |
10.1 (4.1–24.6) | 10.4 (3.1–24.6) | |
| Neutrophil, 10 |
4.31 (1.6–8.6) | 7.64 (1.8–21.5) |
6.9 (3.1–21.5) | 8.43 (1.8–21.5) | |
| Lymphocyte, 10 |
1.9 (0.4–4.1) | 1.7 (0.3–5.4) | 1.6 (0.3–5.4) | 1.7 (0.5–3.5) | |
| PLT, 10 |
230.9 (79–427) | 231.3 (25–449) | 228.3 (25–422) | 234 (77–449) | |
| NLR | 2.7 (0.9–15.7) | 6.0 (0.6–32.5) |
6.0 (0.6–32.5) | 6.1 (1.1–19.8) | |
| PLR | 135.7 (52.2–337.3) | 165.7 (23.1–627.7) |
171.4 (23.1–627.7) | 160 (46.4–542.1) | |
| NPR | 0.019 (0.006–0.092) | 0.047 (0.009–0.31) |
0.051 (0.01–0.31) | 0.044 (0.009–0.088) | |
| Hemoglobin, g/dL | 139.2 (57–175) | 133.4 (58–169) | 130.7 (58–167) | 136 (64–169) | |
| Creatinine, µmol/L | 79.1 (31.5–185.2) | 83.6 (38.6–198.7) |
82.8 (40.9–194.4) | 84.4 (38.6–198.7) | |
| UA, µmol/L | 402.1 (198–578.2) | 409.2 (98.7–815) | 423.2 (173–760.2) | 395.2 (98.7–815) | |
| TG, mmol/L | 2.0 (0.5–8.7) | 1.7 (0.4–9.6) | 1.6 (0.4–5.9) | 1.9 (0.4–9.6) | |
| TC, mmol/L | 4.8 (1.4–10) | 5.1 (2.0–11.6) | 5.1 (2.0–11.6) | 5.1 (2.3–10.2) | |
| HDL-C, mmol/L | 1.2 (0.6–2.1) | 1.1 (0.4–2.2) | 1.1 (0.4–2.2) | 1.1 (0.6–1.8) | |
| LDL-C, mmol/L | 3.1 (0.8–7.4) | 3.4 (1.2–8.6) |
3.5 (1.2–8.6) | 3.4 (1.3–7.4) | |
| Glucose, mmol/L | 6.3 (3.8–11.8) | 9.4 (4.2–27.8) |
8.9 (4.2–24.4) | 9.9 (4.3–27.8) | |
| HbA1c, % | 5.9 (4.8–10.5) | 6.7 (4.8–17.3) |
6.5 (5.0–11.5) | 6.9 (4.8–17.3) | |
| BMI, body mass index; STEMI, ST-segment elevation myocardial infarction;
NT-proBNP, N-terminal pro–B-type natriuretic peptide; WBC, white blood cell;
CRP, C-reactive protein; NLR, neutrophil-to-lymphocyte ratio; PLR,
platelet-to-lymphocyte ratio; NPR, neutrophil-to-platelet ratio; UA, uric acid;
TG, triglyceride; TC, total cholesterol; HDL-C, high-density lipoprotein
cholesterol; LDL-C, low-density lipoprotein cholesterol; HbA1c, glycosylated
hemoglobin; n.d., no data. Values are medians (interquartile ranges), means | |||||
STEMI and NSTEMI populations were characterized by increased levels of sIL-1R2 when compared to healthy controls (Fig. 3A). There were no statistically significant differences in sIL-1R2 between STEMI and NSTEMI patients. To minimize the chance that elevated sIL-1R2 was due to metabolic diseases (overweight/obesity, diabetes and metabolic syndrome), we compared controls with and without metabolic diseases, finding no statistically significant differences (Fig. 3B).
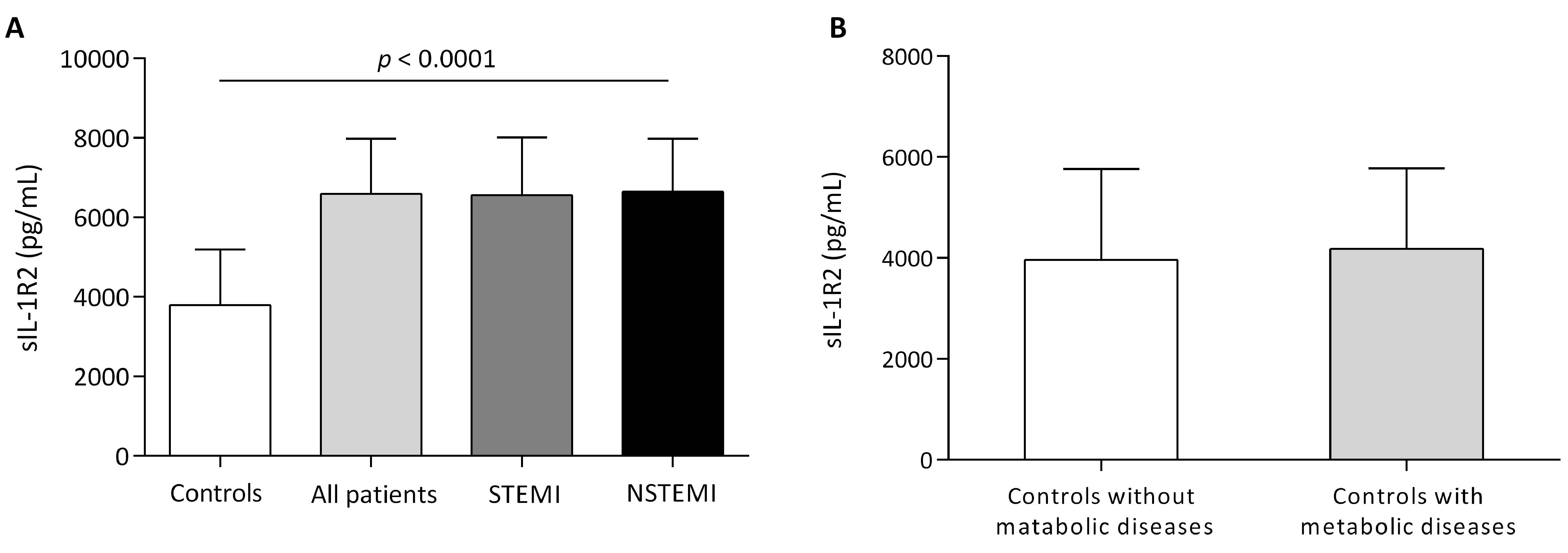 Fig. 3.
Fig. 3.Circulating levels of sIL-1R2. (A) Circulating sIL-1R2 levels in AMI patients (104 STEMI, 100 NSTEMI) and controls. (B) Circulating sIL-1R2 levels in controls with (n = 76) and without (n = 128) metabolic disease. Metabolic disease included overweight, obesity, diabetes and metabolic syndrome. Values are medians (interquartile ranges), p values for difference between groups of sIL-1R2.
We then studied the associations between sIL-1R2 and LV remodeling, LVEF. There
was no significant association between sIL-1R2 levels and LVEF values determined
by echocardiography in the acute phase or at 12-month follow-up (Table 2).
However, compared with patients who had worsened LVEF (LVEF decreased ˃10%) or
reduced LVEF (final LVEF
| sIL-1R2 |
sIL-1R2 ˃Median | p value | ||
| Acute phase | ||||
| LVEF, % | 56.7 (23.0–79.0) | 53.4 (21.0–77.0) | 0.060 | |
| LVEDV, mL | 104.7 (74.2–141.3) | 105.8 (73.5–147.4) | 0.801 | |
| LVESV, mL | 40.2 (27–54.2) | 41.1 (28–58.1) | 0.462 | |
| After 12 months | ||||
| LVEF, % | 56.1 (30.0–75.0) | 57.1 (23.0–79.0) | 0.516 | |
| LVEDV, mL | 103.5 (74.0–135.3) | 104.0 (74.2–135.3) | 0.711 | |
| LVESV, mL | 38.7 (26–50.9) | 37.3 (24.6–50.9) | 0.218 | |
| LVEDV, left ventricular end-diastolic volume; LVESV, left ventricular end-systolic volume. | ||||
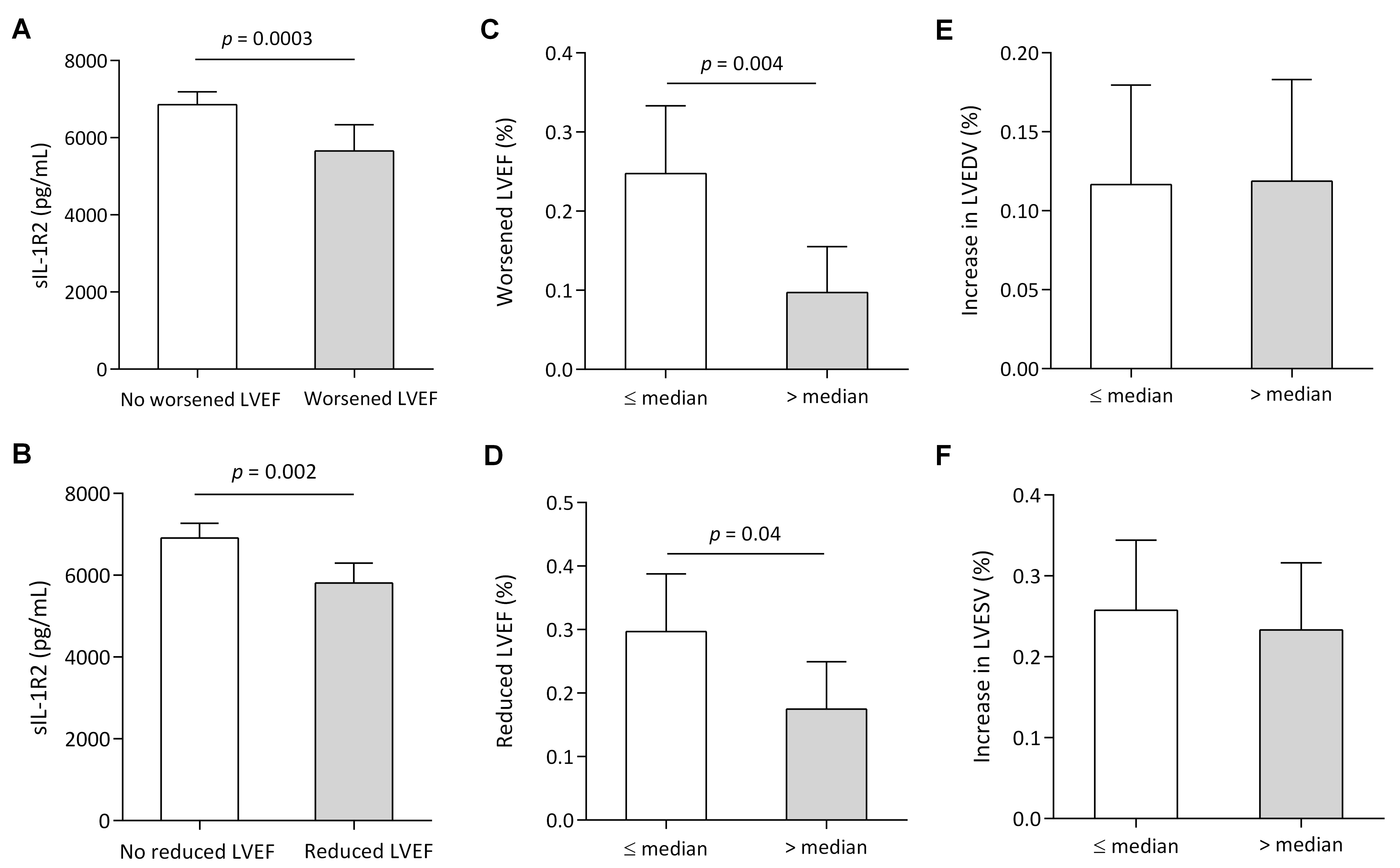 Fig. 4.
Fig. 4.Myocardial function according to sIL-1R2 levels. (A)
Circulating sIL-1R2 levels in patients with or without worsened LVEF. (B)
Circulating sIL-1R2 levels in patients with or without reduced LVEF. (C)
Proportion of worsened LVEF (LVEF decreased ˃10%) from the acute stage to
12-month follow-up, according to high or low sIL-1R2 levels: median: 6652.81
pg/mL. (D) Presence of reduced LVEF (final LVEF
The associations between sIL-1R2 levels and impaired recovery of LV function in AMI patients were investigated using univariate and multivariate logistic regression analyses (Table 3). Low sIL-1R2 levels (less than the median) were associated with increased odds of having worsened LVEF [odds ratio (OR): 3.1, 95% CI: 1.6–8.0, p = 0.006]. After adjustment for age, admission NT-proBNP, peak CRP and TnT in multivariable logistic regression analyses, low sIL-1R2 levels remained associated with worsened LVEF after 12 months (OR: 3.7, 95% CI: 1.6–8.5, p = 0.002). Patients with low sIL-1R2 levels were more likely to have reduced LVEF (unadjusted OR: 2.0, 95% CI: 1.0–3.9, p = 0.041). After adjustment for relevant clinical variables and age, this association remained significant (adjusted OR: 2.1, 95% CI: 1.1–4.3, p = 0.035).
| Worsened LVEF | Reduced LVEF | |||||||||
| OR | 95% CI | p value | OR | 95% CI | p value | |||||
| Univariable analysis | ||||||||||
| Low IL-1R2 | 1.118 | 3.1 | 1.6–8.0 | 0.006 | 0.691 | 2.0 | 1.0–3.9 | 0.041 | ||
| Age | 0.000 | 1.0 | 0.97–1.03 | 0.988 | 0.029 | 1.0 | 1.0–1.1 | 0.031 | ||
| Sex | 0.182 | 1.2 | 0.5–3.0 | 0.692 | –0.027 | 0.97 | 0.5–2.1 | 0.994 | ||
| STEMI | –0.116 | 0.9 | 0.4–1.8 | 0.754 | –0.051 | 0.95 | 0.5–1.8 | 0.877 | ||
| Triple vessel lesion | –0.739 | 2.1 | 0.6–7.4 | 0.252 | –0.143 | 0.9 | 0.3–2.3 | 0.775 | ||
| Peak TnT, per SD | 0.375 | 1.5 | 1.0–2.1 | 0.034 | 0.355 | 1.4 | 1.0–2.0 | 0.026 | ||
| Admission NT-proBNP, per SD | 0.405 | 1.4 | 1.1–1.9 | 0.020 | 0.399 | 1.5 | 1.1–2.0 | 0.007 | ||
| Admission neutrophil, per SD | 0.168 | 1.2 | 0.8–1.7 | 0.356 | –0.247 | 0.8 | 0.5–1.1 | 0.188 | ||
| Peak CRP, per SD | 0.393 | 1.5 | 1.1–2.0 | 0.014 | 0.378 | 1.5 | 1.1–2.0 | 0.013 | ||
| NLR, per SD | 0.261 | 1.3 | 0.9–1.8 | 0.129 | 0.058 | 1.1 | 0.8–1.5 | 0.731 | ||
| PLR, per SD | 0.195 | 1.2 | 0.9–1.7 | 0.245 | 0.195 | 1.2 | 0.9–1.7 | 0.207 | ||
| NPR, per SD | 0.280 | 1.3 | 0.9–1.8 | 0.087 | 0.106 | 1.1 | 0.8–1.5 | 0.506 | ||
| Multivariable analysis | ||||||||||
| Model 1 | ||||||||||
| Low IL-1R2 | 1.270 | 3.6 | 1.5–8.3 | 0.003 | 0.778 | 2.2 | 1.1–4.5 | 0.033 | ||
| Peak TnT, per SD | 0.498 | 1.7 | 1.1–2.4 | 0.011 | 0.450 | 1.6 | 1.1–2.2 | 0.010 | ||
| Admission NT-proBNP, per SD | 0.120 | 1.1 | 0.8–1.6 | 0.501 | 0.337 | 1.3 | 0.97–1.8 | 0.077 | ||
| Peak CRP, per SD | 0.378 | 1.5 | 1.0–2.1 | 0.029 | 0.290 | 1.4 | 0.99–1.9 | 0.062 | ||
| Age | ̵̶ | ̵̶ | ̵̶ | ̵̶ | 0.022 | 1.0 | 0.99–1.1 | 0.126 | ||
| Model 2 | ||||||||||
| Low IL-1R2 | 1.302 | 3.7 | 1.6–8.5 | 0.002 | 0.760 | 2.1 | 1.1–4.3 | 0.035 | ||
| Peak TnT, per SD | 0.502 | 1.7 | 1.1–32.4 | 0.010 | 0.441 | 1.6 | 1.1–2.2 | 0.011 | ||
| Admission NT-proBNP, per SD | ̵̶ | ̵̶ | ̵̶ | ̵̶ | 0.337 | 1.4 | 1.0–1.9 | 0.035 | ||
| Peak CRP, per SD | 0.403 | 1.5 | 1.1–2.1 | 0.017 | 0.317 | 1.4 | 1.0–1.9 | 0.048 | ||
| Worsened LVEF was defined as a decrease in LVEF ˃10%, and reduced LVEF was
defined as a final LVEF | ||||||||||
During 12 months of follow-up, 24 (11.8%) patients experienced an adverse
clinical event (9 reinfarctions, 10 hospitalizations for heart failure, 3 strokes
and 2 deaths). In patients who experienced adverse clinical events compared with
patients without, sIL-1R2 levels were significantly lower (Fig. 5). Patients with
low sIL-1R2 levels had lower freedom from major adverse cardiac events (MACEs)
during the first 12 months (Fig. 6). After adjustment for admission NT-proBNP
levels, low levels of sIL-1R2 remained associated with an increased risk of
experiencing an adverse clinical event during the first 12 months (HR 2.5; 95%
CI: 1.0–6.1; p = 0.039) (Fig. 7). The ability of sIL-1R2 to
discriminate between patients with or without the adverse clinical event was also
assessed by the area under the ROC curve, presented in Fig. 8. In all patients,
the area under the curve (AUC) was 0.721 (95% CI: 0.617–0.824), and the sIL-1R2
cutoff value of 5022.97 pg/mL had 0.542 sensitivity and 0.806 specificity for
detecting AMI. Comparison of AUC between NSTEMI patients and STEMI patients
showed that there was an overlap between 95% of the confidence intervals under
the ROC curve (p = 0.739), suggesting that there was no significant
difference in the AUC between the two different groups (Fig. 8A). Soluble IL-1R2
had the highest predictive value for the incidence of an adverse clinical event,
with an AUC of 0.721 (95% CI: 0.628–0.896, p
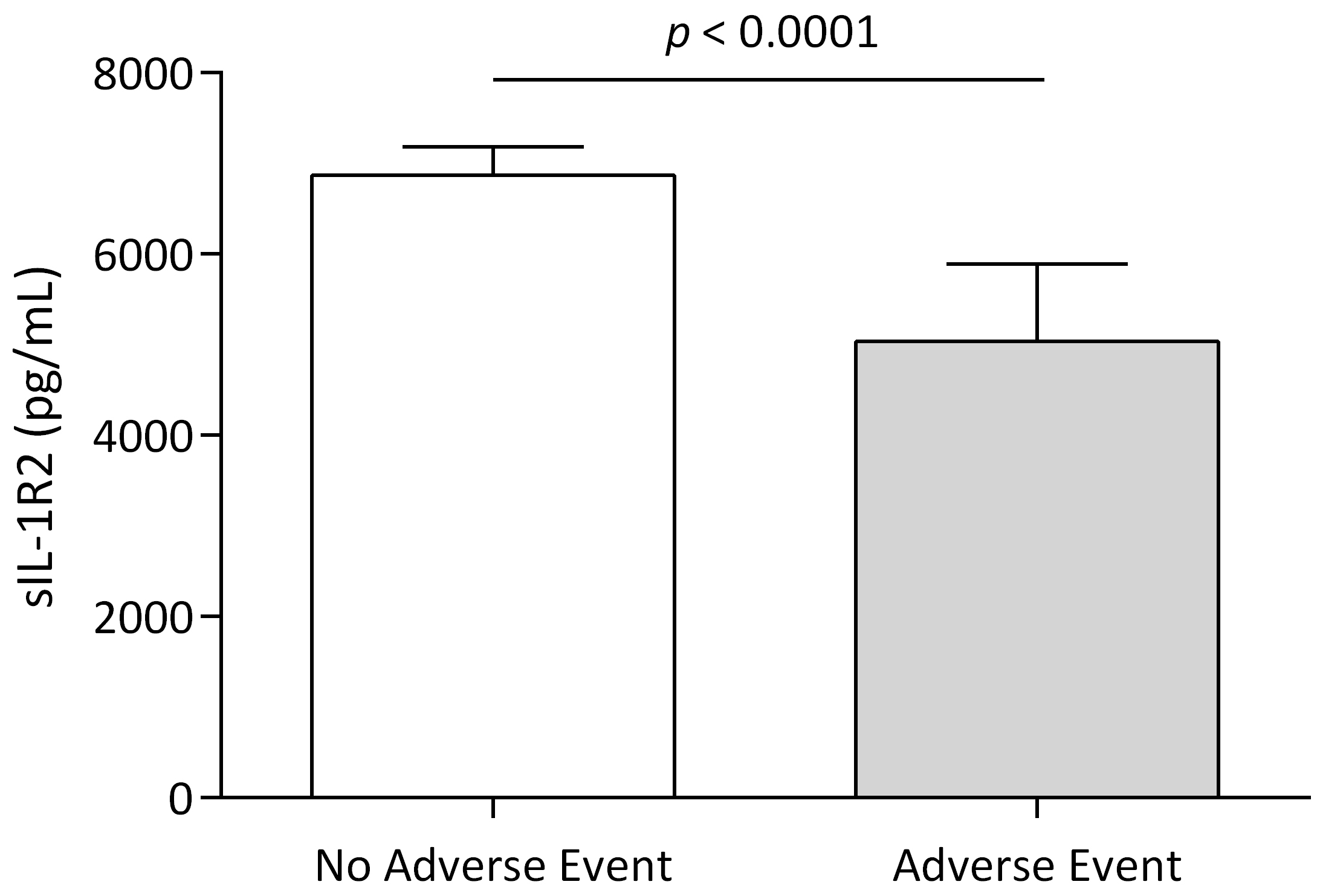 Fig. 5.
Fig. 5.Adverse clinical events according to sIL-1R2 levels. Levels of sIL-1R2 in patients with or without adverse clinical events during the first 12 months. Data are presented as medians (interquartile ranges), p-values for differences in sIL-1R2 between groups.
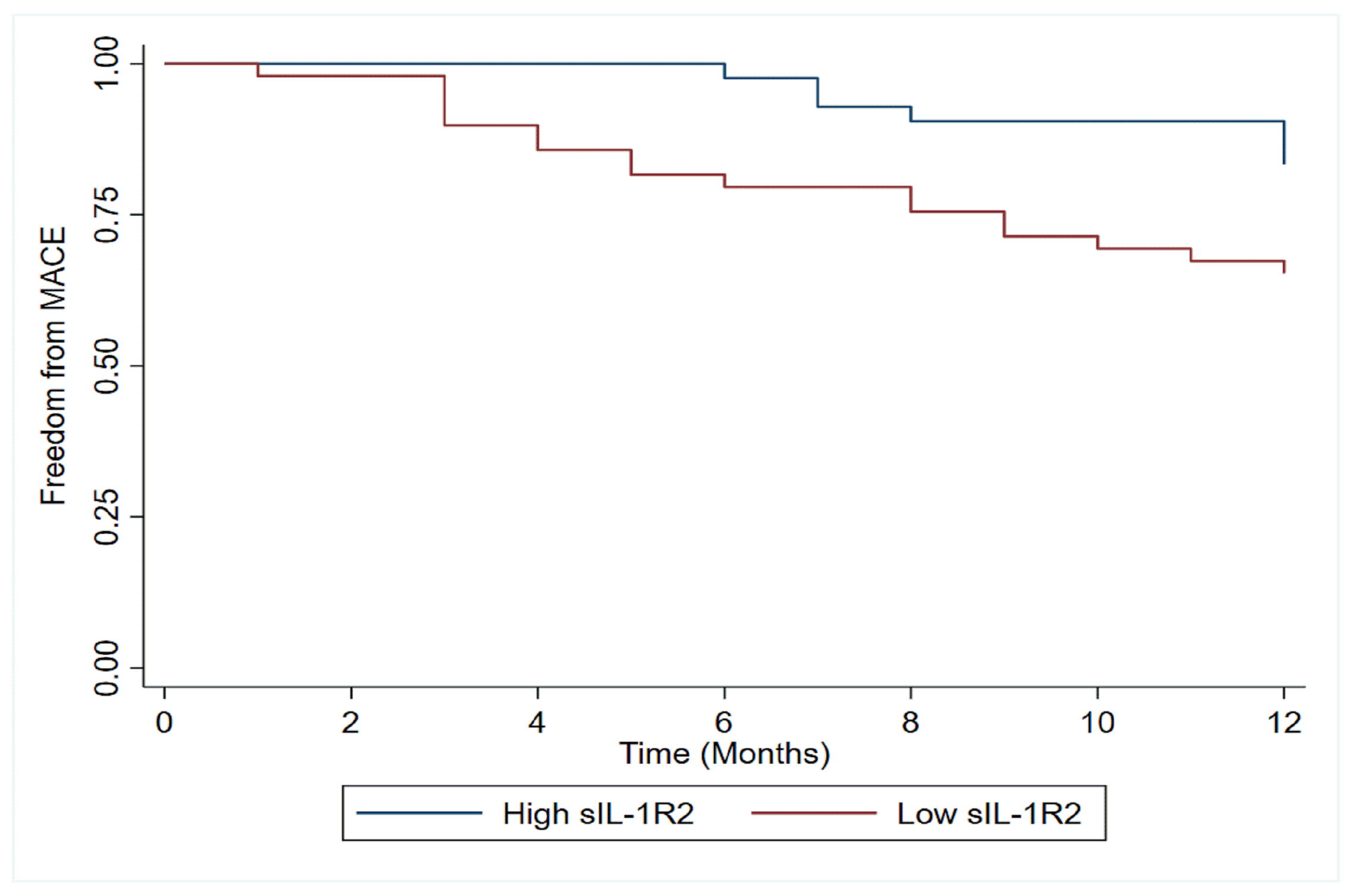 Fig. 6.
Fig. 6.Kaplan-Meier plots of adverse clinical cardiac events according to sIL-1R2 levels in acute myocardial infarction. Kaplan-Meier plots of adverse clinical events during the 12-month follow-up according to high or low sIL-1R2. MACE, major adverse cardiac events.
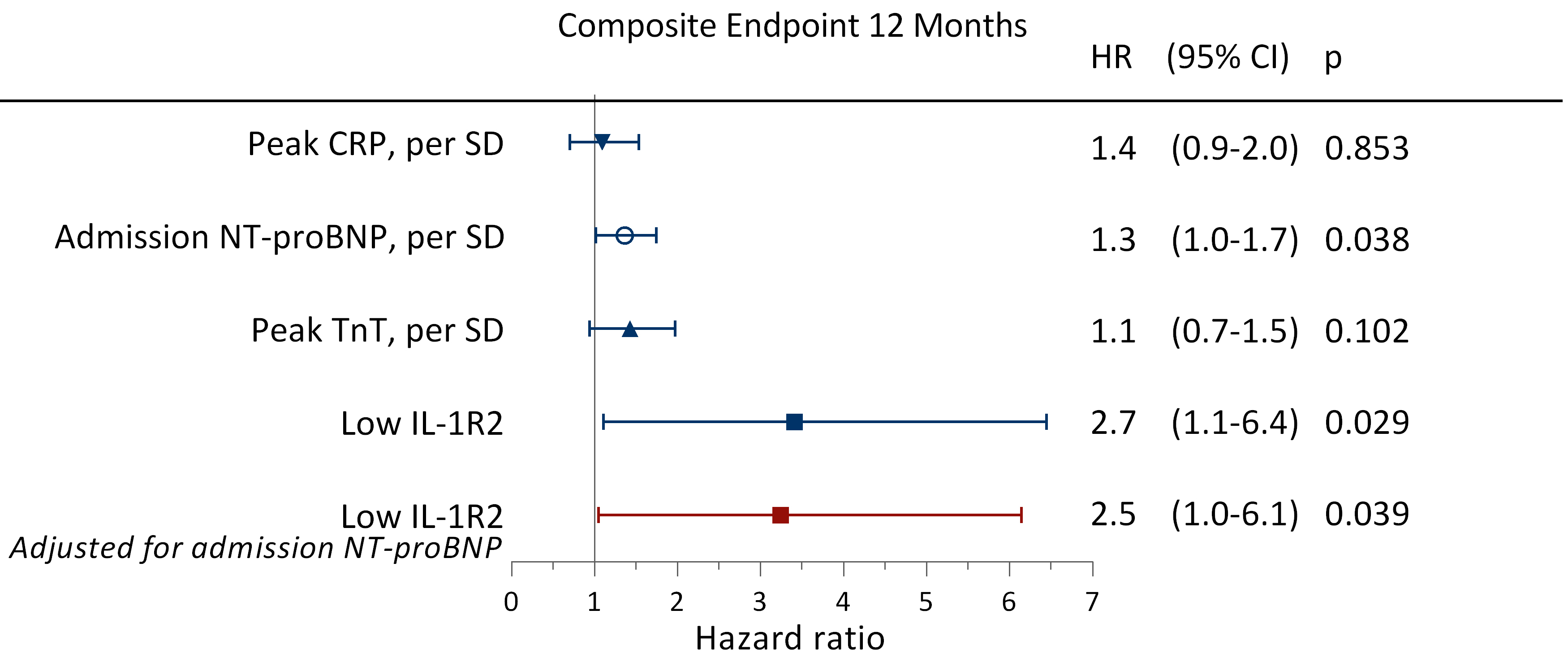 Fig. 7.
Fig. 7.Hazard ratios for experiencing adverse clinical events. Unadjusted and adjusted HRs obtained by Cox regression analyses for experiencing an adverse clinical event during the first 12 months of follow-up when having low sIL-1R2 levels (less than the median) during hospitalization. Adverse clinical events were defined as all-cause mortality, reinfarction, rehospitalization for heart failure, or stroke. HR, hazard ratio; CI, confidence interval; other abbreviations as in Fig. 1.
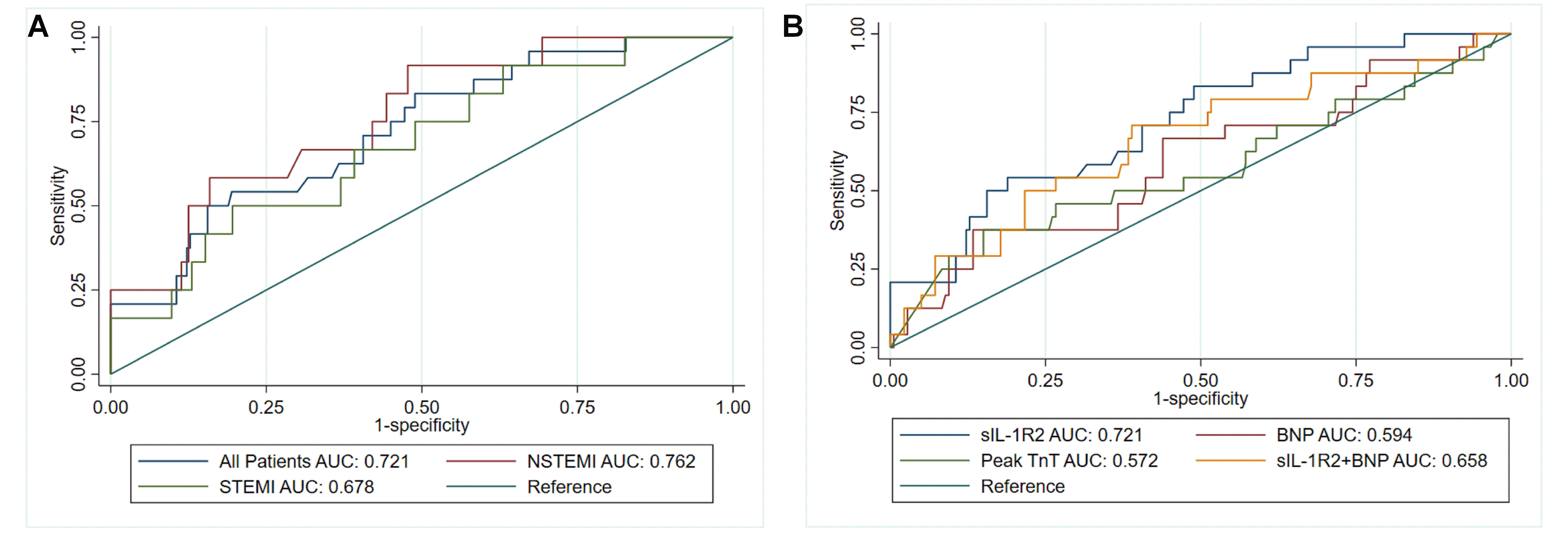 Fig. 8.
Fig. 8.The discriminative value of sIL-1R2 for adverse clinical events. (A) ROC curves for an adverse clinical event during the first 12 months after AMI according to sIL-1R2 levels for STEMI, NSTEMI and all patients. (B) ROC curves for detecting an adverse clinical event during the first 12 months after AMI according to sIL-1R2 levels and other biomarkers. Adverse clinical events were defined as all-cause mortality, reinfarction, rehospitalization for heart failure, or stroke. AUC, area under the receiver operating characteristic curve.
Circulating levels of sIL-1R2 were elevated in AMI patients as compared to healthy controls. Circulating sIL-1R2 measured early in AMI patients was associated with recovery of LV function and with clinical events during 12 months of follow-up. These findings support the notion that sIL-1R2 may play a crucial role in postinfarction inflammation, and that sIL-1R2 may be clinically useful as a predictor of increased risk of new events in AMI patients.
IL-1 signaling disorders after myocardial infarction can affect infarction
healing, cause collateral damage, deteriorate cardiac function and lead to
adverse clinical outcomes [18]. IL-1 activity is controlled by the activation of
the receptors [4]. Soluble IL-1R2 can competitively bind to IL-1 with high
affinity in the circulation and exclude it from the signal transduction complex,
so that excessive IL-1 cannot exert its biological function [7]. Moreover,
intracellular sIL-1R2 can directly bind to the IL-1
Recently, IL-1R2 has been identified as a pivotal mediator of a broad spectrum of inflammatory cytokines involved in the development of coronary atherosclerosis [24]. In animal models of arthritis [25], IL-1-induced inflammation [26] and cardiac allograft surgery [27], overexpression of IL-1R2 has anti-inflammatory profiles. In transgenic mice, phorbol ester-induced dermal and epidermal inflammation is ameliorated by overexpressing IL-1R2 in the epidermis [28]. The conventional view holds that IL-1R2 is mainly expressed in neutrophils, monocytes and macrophages [5]. Recent research has indicated that there is release of soluble IL-1R2 from injured cardiomyocytes subjected to ischemia/reperfusion conditions. In addition, myocardial ischemia/reperfusion-induced apoptosis is abrogated by IL-1R2 overexpression in cardiomyocytes [29]. Some studies have suggested that IL-1R2 plays a role in regulating monocyte accumulation during myocardial ischemia/reperfusion injury [17]. These findings suggest that IL-1R2 is probably more than redundant in endogenous IL-1 antagonist systems and could be a promising mediator of the inflammatory response in AMI.
Persistent and excessive inflammation unrelated to infarct size has been
considered an important contributor to increased risk of ventricular remodeling
and adverse clinical events following MI [30]. Abnormal inflammatory status after
myocardial infarction is associated with adverse LV remodeling and underlies
heart failure pathogenesis [31]. We excluded patients with infectious diseases,
chronic inflammatory diseases or cancer to eliminate the effects of other disease
processes on the association between low levels of sIL-1R2 and adverse outcomes.
The major finding in this study was that low levels of sIL-1R2 during the acute
phase of AMI were significantly associated with impaired LV contractibility
defined as a decrease in LVEF ˃10% from hospitalization to 12 months and LVEF
Randomized trial data have consistently demonstrated persistent inflammation to
be as important a potential therapeutic target for atheroprotection [32, 33]. The
CANTOS trial provided proof of concept that attenuating IL-1 inflammation reduces
the risk for acute cardiovascular events [8]. Due to the clinical usage of
anakinra, the recombinant human IL-1Ra analog, the beneficial effects of IL-1Ra
during MI are well documented [34]. In contrast, the role of IL-1R2 in AMI has
not been well elucidated. Although anakinra is a valuable therapeutic tool, it
has a short in vivo half-life, necessitating daily injection
[35]. IL-1R2 has a longer half-life, low affinity for IL-1Ra and high affinity
for IL-1
The limitations of this study should be acknowledged. The results of this study
provide no evidence of a causal relationship involving sIL-1R2 and LV function or
adverse clinical outcomes. The reported number of adverse events in the present
cohort was relatively small. Moreover, we used echocardiography to assess adverse
remodeling in the present study, which needs to be considered when interpreting
the results. Furthermore, we lacked follow up measurements and the temporal
profile of sIL-1R2 on the AMI patients between days 1 and 12 months. Other
inflammatory markers, such as IL-1
The present study demonstrated that low levels of sIL-1R2 in the acute phase of AMI patients were associated with impaired recovery of LV function and increased future adverse clinical events. The results indicated that sIL-1R2 may be a clinically useful biomarker for risk prediction in AMI patients, and sIL-1R2 itself may be a novel target for atherothrombotic protection.
SFL and FG designed the study and drafted the manuscript. SL and QTY collected the data. TGG and YZ analyzed the data. SFL, JYC and SL contributed to patient management. CWJ and YNZ performed coronary angiography and percutaneous coronary intervention. SFL and FG performed the statistical analysis. All authors revised the manuscript critically for important intellectual content and approved the submitted manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
The study was conducted in accordance with the Declaration of Helsinki and the Ethics Committee of Zhongshan Hospital of Xiamen University (No. 2021-141). Written informed consent was obtained from all patients prior to inclusion.
We thank Haiqiang Chen for valuable assistance with the statistical analyses. We appreciate the help and support of study nurses, echocardiography technicians, and the staff at the Intensive Coronary Care Unit, Zhongshan Hospital of Xiamen University.
This research was funded by the Xiamen Science and Technology Program Project (3502Z20209023 and 3502Z20214ZD1049), Science and Technology Plan Project of Fujian Provincial Health Commission (2022GG01010075).
The authors declare no conflict of interest.
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.








