1 Department of Clinical Medicine and Surgery, University of Naples Federico II, School of Medicine, 80130 Naples, Italy
Abstract
The polyadenylated (polyA) tail of mRNA plays a crucial role in regulating mRNA stability and translation, and it may also contribute to genome integration through interactions with long interspersed nuclear element-1 (LINE-1, L1) retrotransposons. This interaction is particularly relevant for mRNA vaccines. Understanding how the polyA tail interacts with L1 proteins, especially open reading frame 2 protein (ORF2p), is critical for assessing these risks and developing strategies to enhance the safety of mRNA vaccines. We suggest conducting in vitro experiments to explore polyA tail modifications and their effects on L1 binding.
Keywords
- L1 retrotransposon
- mRNA vaccines
- mutation
The polyadenylated (polyA) tail of mRNA plays a crucial role not only in mRNA stability and translation [1] but also in its potential involvement in genome integration through interactions with retrotransposons, particularly long interspersed nuclear element-1 (LINE-1, L1) elements [2]. This interaction is particularly relevant in the context of mRNA vaccines, such as those developed for coronavirus disease 2019 (COVID-19), which utilize modified mRNA to induce an immune response against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [3]. Understanding the role of polyA tails in the cytoplasm remains a significant challenge. Since the discovery over 50 years ago that mRNAs are polyA at their 3′ end, the polyA tail has been recognized as a key regulator of mRNA translation and stability [4, 5]. Despite this progress, several questions remain, particularly in the context of mRNA vaccines. For instance, what role does the L1 protein play in the widely accepted “closed loop” model of translation? Can the open reading frame 2 protein (ORF2p) interfere with the “closed loop” structure, potentially leading to aberrant termination events? These questions are increasingly pertinent given the rapid development and deployment of mRNA vaccine technology. One of the significant advantages of mRNA-based vaccines is their rapid developmental timeline, which is crucial when new viral variants are discovered and the vaccine needs swift adjustment [3]. However, the biological nature of retrotransposons, such as L1 elements, necessitates a cautious approach to their widespread application, regardless of the safety and efficacy assurances of SARS-CoV-2 mRNA vaccines that have been reported as safe and effective for the vast majority of the population [4]. Although mRNA vaccines are designed to be non-integrative, the possibility that these exogenous mRNAs could be recognized and integrated into the cellular genome via L1 elements requires further investigation [6]. L1 elements are autonomous retrotransposons constituting approximately 17–20% of the human genome [7]. They can replicate themselves and insert new copies into the genome through retrotransposition. This process relies on two proteins encoded by L1, ORF1p and ORF2p, with ORF2p being particularly crucial due to its reverse transcriptase and endonuclease activities [8]. ORF2p binds strongly to the polyA tail of mRNA, initiating reverse transcription, which could potentially lead to the integration of complementary DNA (cDNA) into the host genome [9]. Although ORF2p shows a strong cis-preference for its own L1 mRNA, it is also capable of trans-recognition, i.e., binding to the polyA tails of other mRNAs, including those introduced by vaccines [10]. This suggests that the presence of a polyA tail in mRNA vaccines could theoretically make them susceptible to recognition by the L1 machinery, leading to reverse transcription and potential genome integration (Fig. 1). While this is considered a rare event, the potential implications are significant, further underscoring the need for comprehensive research.
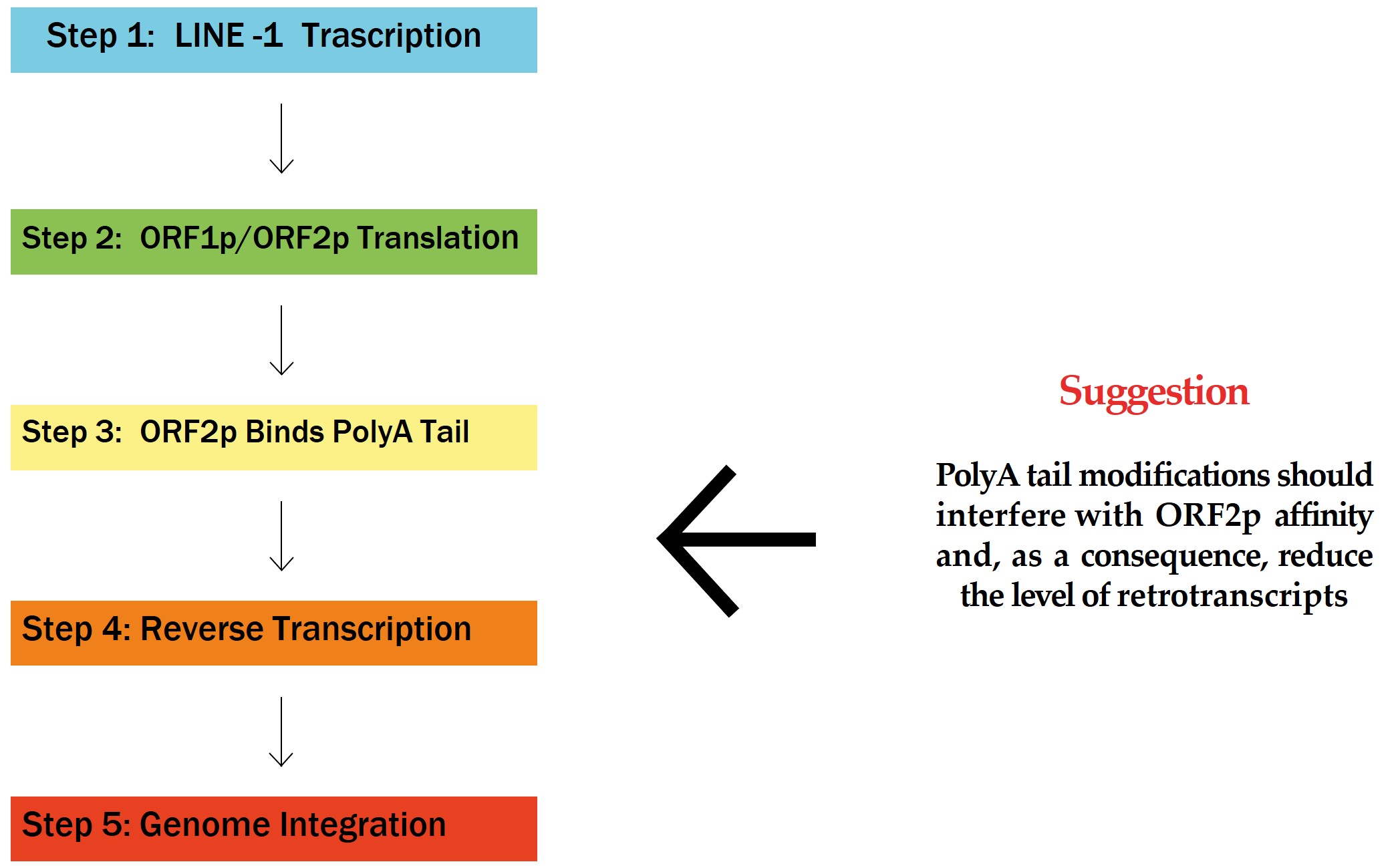 Fig. 1.
Fig. 1.
Flowchart of L1 Retrotransposition. ORF, Open Reading Frame; PolyA, polyadenylated; L1, LINE-1.
Our objective is to encourage the scientific community to undertake further research to better understand the mutagenic potential of mRNA vaccines, particularly because of their interaction with L1 elements. This research should include the design of in vitro experiments to explore how the polyA tail of vaccine mRNAs interacts with L1 proteins, such as ORF2p, and to identify molecular strategies to mitigate any potential risks of insertional mutations. One such strategy could involve genetically modifying the 3′ end of the polyA tail to evaluate, through in vitro experiments, the kinetics of RNA-L1 protein association and determine whether such modifications might reduce the likelihood of L1 binding and subsequent retrotransposition [11]. Furthermore, ORF2p expression has been observed to increase in certain aging tissues and neurodegenerative diseases, suggesting a role in the pathology of these conditions [12]. A recent study has also indicated that the 3′ end of the SARS-CoV-2 genome is more frequently integrated into cellular DNA than the sequences closer to the 5′ end, raising questions about the susceptibility of different mRNA regions to retrotransposition [13]. Understanding these dynamics could facilitate the design of mRNA vaccines with reduced potential for genome integration. The broader scientific inquiry should also explore how ORF2p’s RNA-binding and mobilization activities might influence the expression and stability of both endogenous and exogenous RNAs. This could provide deeper insights into how L1 elements impact genomic stability and gene regulation, with significant implications for understanding and potentially mitigating the risks associated with mRNA vaccines. In conclusion, while the polyA tail is essential for mRNA function, its role in potential genome integration via L1 retrotransposition mechanisms warrants careful investigation. By advancing our understanding of these processes, we can better assess the risks associated with mRNA vaccines and develop strategies to further enhance their safety.
The interaction between polyA tails in mRNA vaccines and L1 retrotransposons represents a theoretical high risk of genome instability, which clearly warrants further investigation. The evidence for genome integration is limited due to technical difficulties; however, the biological plausibility of such mutations underlines the importance of deeper investigations. We recommend biochemically focused studies to clarify the mechanisms of RNA-L1 interactions. These studies should help implement mRNA vaccines with reduced retrotranspositional risks. The scientific community is morally responsible to provide a safe mRNA vaccine technology for future global health challenges.
Conceptualization, writing and editing by IF.
Not applicable.
Not applicable.
This work was supported by the Italian Ministry of Education, University and Research (MIUR), fund for Investments on Basic Research (FFABR) 000015_FFABR_2017_FELICIELLO.ISIDORO.
The author declares no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

