1 College & Hospital of Stomatology, Anhui Medical University, Anhui Provincial Key Laboratory of Oral Diseases Research, 230032 Hefei, Anhui, China
2 Stomatology Hospital, School of Stomatology, Zhejiang University School of Medicine, Zhejiang Provincial Clinical Research Center for Oral Diseases, Key Laboratory of Oral Biomedical Research of Zhejiang Province, Engineering Research Center of Oral Biomaterials and Devices of Zhejiang Province, 310000 Hangzhou, Zhejiang, China
3 School of Pharmaceutical Sciences, Anhui Medical University, 230032 Hefei, Anhui, China
†These authors contributed equally.
Abstract
The progression of periodontitis is accompanied by destruction of keratinized epithelium, while members of the tumor necrosis factor receptor superfamily (TNFRSF) play critical roles in epithelial repair. This study aimed to elucidate the role of TNFRSF in the pathogenesis and progression of periodontitis. Furthermore, we investigated the mechanisms underlying the repair of epithelial keratinization, with the ultimate aim of translating these insights into clinical therapeutic applications.
Single-cell RNA sequencing was used to investigate the TNFRSF expression profiles in the gingival epithelium of patients with severe periodontitis. Gingival tissues were collected from healthy individuals and those with periodontitis. An in vitro model was also established using retinoic acid to inhibit keratinization and BMS493 to promote keratinization. Bulk RNA sequencing was performed to further substantiate the model and validated the findings by gene knockdown and overexpression experiments. Protein–protein interaction (PPI) analysis and immunoprecipitation identified key protein interactions. In addition, a TNFRSF21 overexpression plasmid and a full-thickness dorsal skin wound mouse model were used to confirm regulatory processes during keratinization.
TNFRSF21 expression, along with epithelial keratinization-related genes were significantly reduced in clinical periodontitis tissues. However, TNFRSF21 increased significantly during epithelial repair following initial periodontal therapy for severe periodontitis, particularly in proliferative keratinocytes and basal layer cells. An in vitro keratinization model revealed that TNFRSF21, Keratin 8 (KRT8), and KRT18, were downregulated during the inhibition of keratinization and upregulated during its promotion. Importantly, the expression levels of KRT8, KRT18, and Claudin-1 were consistently downregulated in the TNFRSF21 knockdown group and upregulated in the TNFRSF21 overexpression group. Single-cell RNA sequencing combined with PPI analysis revealed a significant interaction between TNFRSF21 and amyloid precursor protein (APP). This was validated by STRING database analysis and immunoprecipitation. Mice treated with TNFRSF21 overexpression plasmids showed accelerated wound healing and increased keratin expression on dorsal skin.
Our findings indicate that TNFRSF21 is a pivotal regulator of epithelial keratinization and tight junction integrity in oral epithelial keratinocytes. Targeting TNFRSF21 may represent a novel therapeutic strategy to restore oral epithelial function.
Keywords
- periodontitis
- epithelium
- keratins
- TNFRSF21 protein
- single-cell analysis
Periodontitis is a major global health burden affecting billions of people. It is characterized by symptoms ranging from gingival bleeding and tooth mobility to severe tooth loss [1, 2]. Current clinical interventions for periodontitis primarily target the suppression of inflammation, but fail to restore the original periodontal architecture. The mechanistic understanding of disease progression is thus imperative for the development of regenerative therapeutic paradigms [3, 4].
Healthy gingiva is able to maintain normal keratinized epithelial attachment [5]. In periodontitis, the loss of gingival keratinization leads to gingival recession and clinical loss of attachment [6]. The standard treatment is periodontal debridement and appropriate application of mouthwash, such as those containing chlorhexidine. This removes subgingival calculus and plaque, thereby reducing gingival irritation and promoting epithelial reattachment. The adjunctive use of antimicrobial mouth rinses is supported by in vitro evidence demonstrating the antibacterial efficacy of agents like chlorhexidine against oral pathogens [7, 8]. Importantly, oral epithelial keratinocytes show upregulated expression of epithelial keratinization-related genes (KRTs) and exhibit high proliferative and differentiation capacities that drive tissue regeneration [9]. These cells maintain the oral mucosal barrier and produce keratin, which is essential for epithelial protection [10].
The tumor necrosis factor receptor superfamily (TNFRSF) comprises diverse transmembrane proteins that are prominently expressed on cell surfaces [11]. These receptors orchestrate various physiological and pathological processes, including cell signaling transduction, immune modulation, cell proliferation, differentiation, and apoptosis [12]. TNFRSF-ligand interactions are critical in immune regulation and cancer therapy [12, 13]. For instance, the binding of soluble Fas ligand (sFasL) to death receptor 5 exacerbates autoantibody-induced arthritis [14]. Such interactions modulate autoimmune responses and confer dual effects in cancer by either fostering tumor cell proliferation/survival, or triggering cancer cell apoptosis [15, 16, 17]. However, potential links between TNFRSF mechanisms and the development of periodontitis or gingival epithelium keratinization remain unexplored.
In the present study, our aim was to elucidate the role of TNFRSF in periodontitis-associated disorders of keratinization. Additionally, we explored the underlying mechanism for the repair of epithelial keratinization, with the goal of translating these findings into clinical periodontal therapy. Our hypothesis is that TNFRSF plays a pivotal role in maintaining gingival epithelial homeostasis, and that dysfunction of TNFRSF is a key mechanism underlying the impairment of keratinization and poor healing observed in periodontitis. We first evaluated post-treatment epithelial changes in patient samples, then investigated the mechanism of epithelial keratinization injury at the cellular level. Subsequently, we validated the findings in an animal model, with the ultimate aim of translating the results into the clinic for patient benefit. Our study found a novel molecular mechanism involving TNFRSF21 and which appears to underlie defective keratinization in periodontal tissues and keratinocyte differentiation. Overexpression of TNFRSF21 promotes the expression of keratin by oral epithelial keratinocytes, leading to wound keratinization and suggesting that it may be a promising therapeutic target. We also developed an in vitro keratinization gradient model using small molecule drugs, offering a new tool to study the loss of epithelial keratinization. These findings provide a new perspective on the pathogenesis of periodontitis and suggest that TNFRSF21-targeted strategies could be used to treat impaired keratinization in periodontal disease.
Human oral keratinocytes (HOKs) were purchased from Shanghai QuiCell Biotechnology Co., Ltd. The item number is QuiCell-Y1694. These were STR-certified and free of mycoplasma. Following resuscitation in cell culture flasks, the cells were incubated at 37 °C with 5% CO2 for 3–4 hours to stabilize their condition. The complete culture medium consisted of DMEM (11965092, Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 10% fetal bovine serum, 100 U/mL penicillin, and 0.1 mg/mL streptomycin. Upon reaching 70% confluency, the cells were detached using 0.25% trypsin and passaged at a 1:2 ratio [18].
Resuscitated HOKs were seeded into six-well culture plates, and 2 mL of complete medium (DMEM supplemented with 10% fetal bovine serum, penicillin, and streptomycin) was added to each well. The cells were cultured in an incubator at 37 °C and 5% CO2 until they reached 70%–80% confluence. Treatment groups were exposed to 0, 1, and 10 µM concentrations of retinoic acid (RA), or 0, 100, and 500 nM concentrations of BMS493. The drug-containing medium was replaced every two days to ensure stable drug concentrations. On day 9 of culture, cell samples were collected for experiments, including Western blotting to detect changes in the expression of keratinization-related markers.
An siRNA that targets TNFRSF21 was designed and synthesized by TsingKe Biological (Beijing, China). The adenovirus vector GV315 containing TNFRSF21 (NM_014452.5) was purchased from TsingKe Biological (Beijing, China). Transfection was performed using Lipofectamine 2000 (TsingKe Biological) in accordance with the manufacturer’s instructions. All sequence details are provided in Supplementary Table 1.
All animal work was performed in accordance with the ARRIVE guidelines 2.0. The experimental procedures were approved by the Laboratory Animal Care and Use Committee at Anhui Medical University (Approval No. LLSC20232087). Eight C57BL/6 mice aged 8 months were randomly and equally divided into two groups via a simple randomization method using a random number generator. The first group (negative control) received an empty vector plasmid (Vector), while the second group was treated with a plasmid for the overexpression of TNFRSF21 (OE-TNFRSF21). Under surgical anesthesia induced by 2% sodium pentobarbital (50 mg/kg), all mice underwent full-thickness excisional wounds on their dorsal sides. Following hair removal on the back, two 6-mm full-thickness wounds were made along the midline of the back of each mouse using a sterile biopsy punch. Immediately after wounding, the mice were given multiple subcutaneous injections of either the empty vector or the OE-TNFRSF21 plasmid (four injections per wound site), with each wound receiving a single treatment. Additionally, by adding the opioid tramadol (0.1 mg/mL) to the drinking water to alleviate pain related to wounds [19]. On day 9 postoperatively, mice were euthanized via cervical dislocation following induction with 5% isoflurane anesthesia. The dorsal wound tissues were harvested for histological examination and fixed in a 4% paraformaldehyde solution, along with visceral organs, including the heart, liver, spleen, lungs, and kidneys. Subsequently, quantitative histomorphometric analysis was conducted on these tissues to evaluate the progress of wound healing and the overall systemic biocompatibility associated with plasmid therapy.
Patients were classified based on clinical and radiographic (X-ray) criteria.
Periodontal tissues were collected during tooth extraction or ridge repair,
placed in sterile PBS tubes, and transported to the laboratory within 10 minutes
for single-cell preparation and RNA sequencing. The periodontal therapy (PDT)
patient group (n = 3) showed minimal gingival inflammation after treatment, but
had severe clinical attachment and bone loss (
Gingival tissues collected from clinical samples of healthy volunteers or
periodontitis patients were homogenized in RIPA lysis buffer (1
Protein A/G beads (50 µL per reaction, HY-K0202, MedChemExpress,
Monmouth Junction, NJ, USA) were washed with an appropriate amount of IP buffer,
then centrifuged, and the supernatant discarded. TNFRSF21 or IgG antibody (2
µg) was added before incubation for 4–6 h at 4 °C to couple the
antibody with the beads. The supernatant from lysed HOKs was then collected, and
100 µL was taken as the Input group. The remaining cell lysate was added to
the pre-treated beads and incubated at 4 °C overnight to allow the
TNFRSF21 protein to bind with the specific antibody. Next, the beads were washed
three times with lysis buffer to remove unbound proteins. Loading buffer
(5
Tissue sections (4 µm) were subjected to H&E staining using standardized protocols. Briefly, deparaffinized sections were stained with Mayer’s hematoxylin (4 min) and then differentiated (1% acid ethanol, 3 sec), blued (0.2% ammonia water), and counterstained with eosin Y (3 min). After dehydration and mounting, the slides were imaged using a Nikon Eclipse E100 microscope (Nikon Eclipse E100, Nikon Instruments Inc., Tokyo, Japan).
Tissue sections were deparaffinized in xylene (2
Tissue sections were encircled with a hydrophobic barrier, treated with an
autofluorescence quencher (G1221, Servicebio, Wuhan, Hubei, China) for 5 min, and
rinsed under running water (10 min). Non-specific binding was blocked with 3%
BSA/PBS (30 min, RT). Primary antibodies (diluted in PBS) were applied and
incubated overnight at 4 °C in a humidified chamber. After PBS washes (3
To investigate whether TNFRSF regulates the healing process after treatment for severe periodontitis, we analyzed single-cell RNAseq of gingival epithelial tissues collected after initial PDT [20]. t-SNE cluster analysis performed on the scRNA-seq data of oral epithelial cells revealed 7 distinct cell clusters (Fig. 1A). The differentiation trajectory was analyzed and visualized with partition-based graph abstraction (PAGA) (Fig. 1B). KEGG enrichment analysis revealed significant enrichment of the TNF signaling pathway within the Basal_V and Pro_KC cell clusters. In addition, signaling pathways related to tight junctions, inflammatory regulation, and interaction with extracellular mechanisms were also significantly enriched (Fig. 1C). Subsequently, we aggregated the expression data of TNFRSF across all cell clusters and observed that TNFRSF21 was prominently expressed in each cluster. This consistent expression pattern indicates that TNFRSF21 functions as a pivotal marker in modulating the healing processes of periodontitis (Fig. 1D).
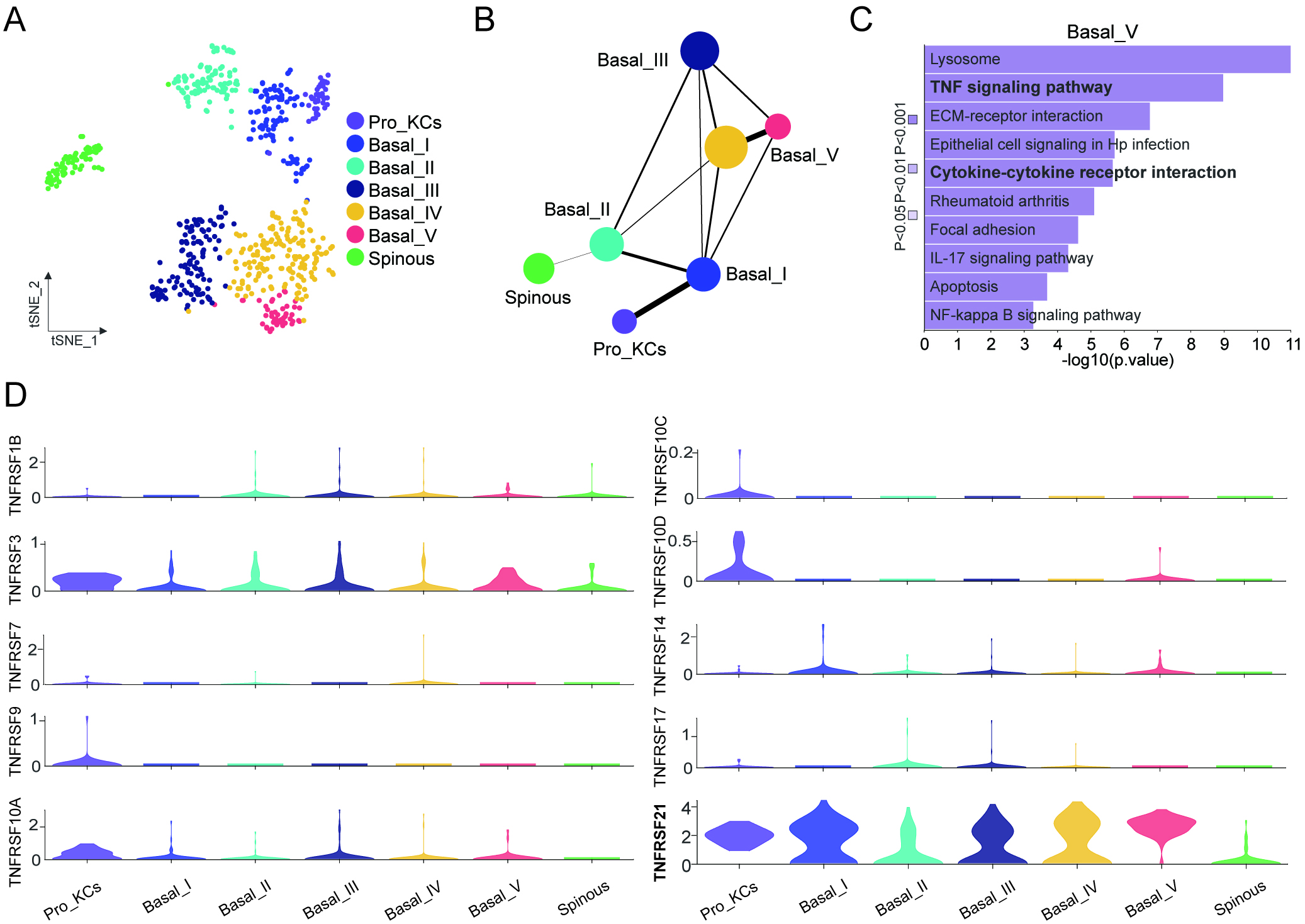 Fig. 1.
Fig. 1.
Tumor necrosis factor receptor superfamily 21 (TNFRSF21) is a key molecule during periodontal regeneration and repair. (A) The main cell types present in the gingival epithelium after periodontal treatment were identified by the Seurat R package and visualized by t-Distributed Stochastic Neighbor Embedding (t-SNE). (B) Partition-based graph abstraction (PAGA) was employed to analyze and visualize the differentiation trajectories. (C) Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis of Basal_V and Pro_KCs showed significant activation of the TNF signaling pathway. (D) Violin plot illustrating the expression levels of selected members of the TNFRSF family during the periodontitis healing process.
To further examine specific gene expression in periodontal epithelial tissues, we compared the expression of TNFRSF21 and KRTs between individuals with periodontitis and healthy controls. The expression levels of both TNFRSF21 and KRTs were found to be significantly reduced in the periodontitis group compared to the control group (Fig. 2A). Subsequently, we established an in vitro concentration gradient model for inhibiting and promoting keratinization in HOK cells using RA, a small molecule drug, and its inhibitor BMS493, respectively [29, 30, 31]. This model was designed to simulate the processes of incomplete keratinization in epithelial tissues during the onset of periodontitis, and of re-keratinization following treatment and recovery. Western blot analysis confirmed that expression levels for TNFRSF21 and KRTs (including KRT8 and KRT18) were significantly reduced in the keratinization inhibition group (RA), whereas they were significantly increased in the keratinization promotion group (BMS493; Fig. 2B). Differential gene analysis of bulk RNA sequencing showed that RA treatment reduced the expression of keratin proteins such as KRT9, KRT16, and KRT17, while increasing the expression of chemokines like C-C Motif Chemokine Ligand 2 (CCL2). Furthermore, bulk RNA sequencing data revealed excellent intra-group homogeneity and clear inter-group separation across the NC, RA, and BMS493 groups (Fig. 2C). Gene Ontology (GO) functional enrichment analysis revealed the DEGs were significantly correlated with cell differentiation, immune regulation, epithelial migration, and keratinization (Fig. 2D). These findings imply that epithelial regeneration, reduction of inflammation, and the critical regulatory role of keratinization are integral to both the pathogenesis of periodontitis and its therapeutic recovery.
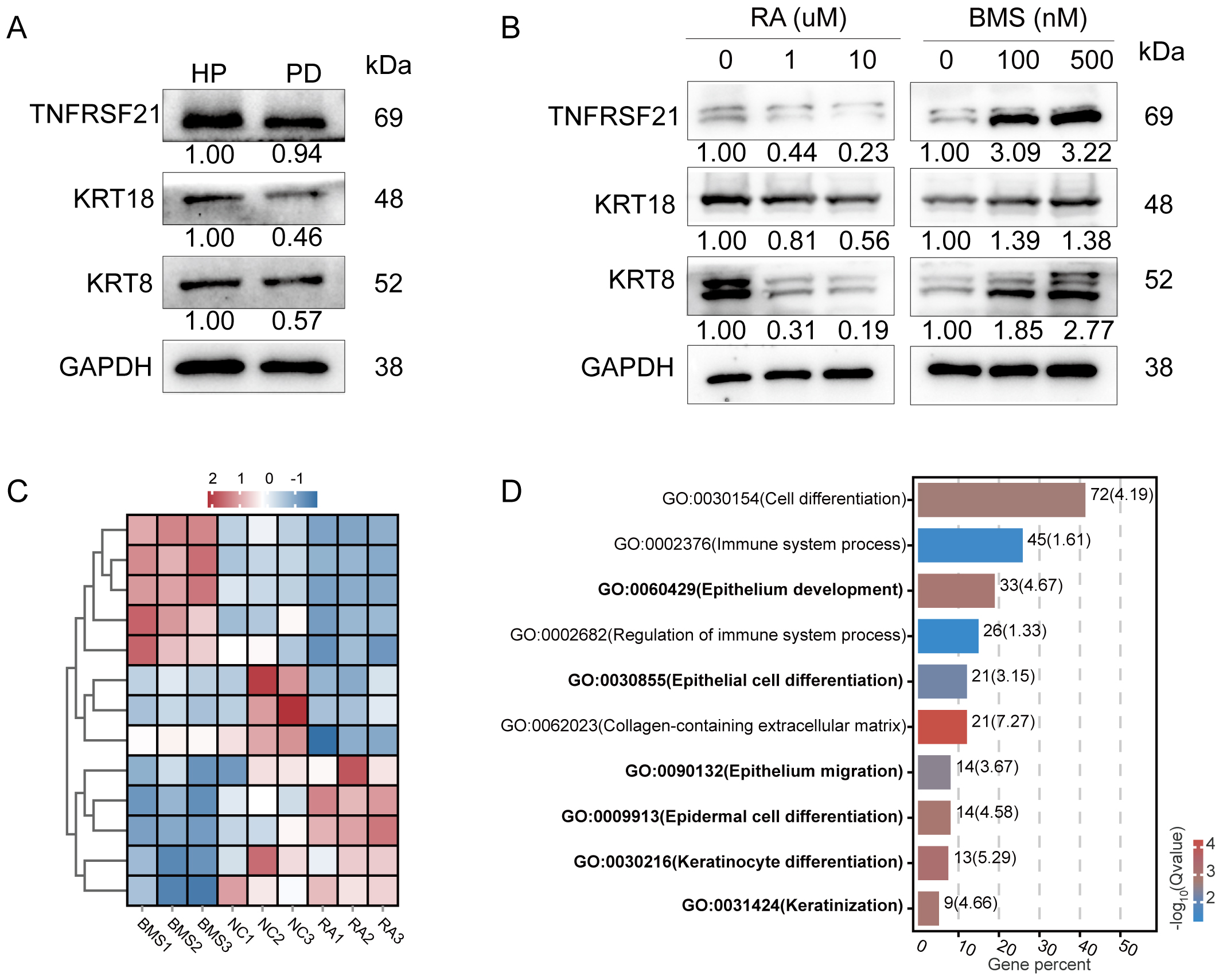 Fig. 2.
Fig. 2.
Changes in TNFRSF21 expression during the keratinization process, as revealed by the in vitro keratinization induction model. (A) Western blot analysis showed a significant reduction in the expression levels of keratin and TNFRSF21 in the gingival epithelial tissues of periodontitis patients (PD) compared to healthy controls (HP). (B) In vitro models showed decreased TNFRSF21 and keratin expression in the keratinization inhibition group (treated with retinoic acid (RA)), and increased expression in the keratinization promotion group (treated with BMS493). (C) Bulk RNA sequencing revealed high intra-group consistency and clear segregation between the negative control (NC), RA, and BMS493 groups. (D) GO enrichment analysis of differentially expressed genes identified by bulk-RNA sequencing.
To further investigate the potential regulatory role of TNFRSF21, we employed gene knockdown and overexpression techniques to suppress and enhance its expression. Validation of the knockdown efficiency revealed markedly decreased TNFRSF21 protein expression (Fig. 3A). We also examined alterations in the expression levels of target KRTs, including KRT8, KRT18, and Claudin-1 (Fig. 3B). Subsequently, we constructed a plasmid for TNFRSF21 overexpression and transfected it into HOK cells. This resulted in increased expression of TNFRSF21, as well as increased expression levels of KRT8, KRT18, and Claudin-1 (Fig. 3C). Together, these findings support a regulatory role of TNFRSF21 in keratin expression by HOK cells.
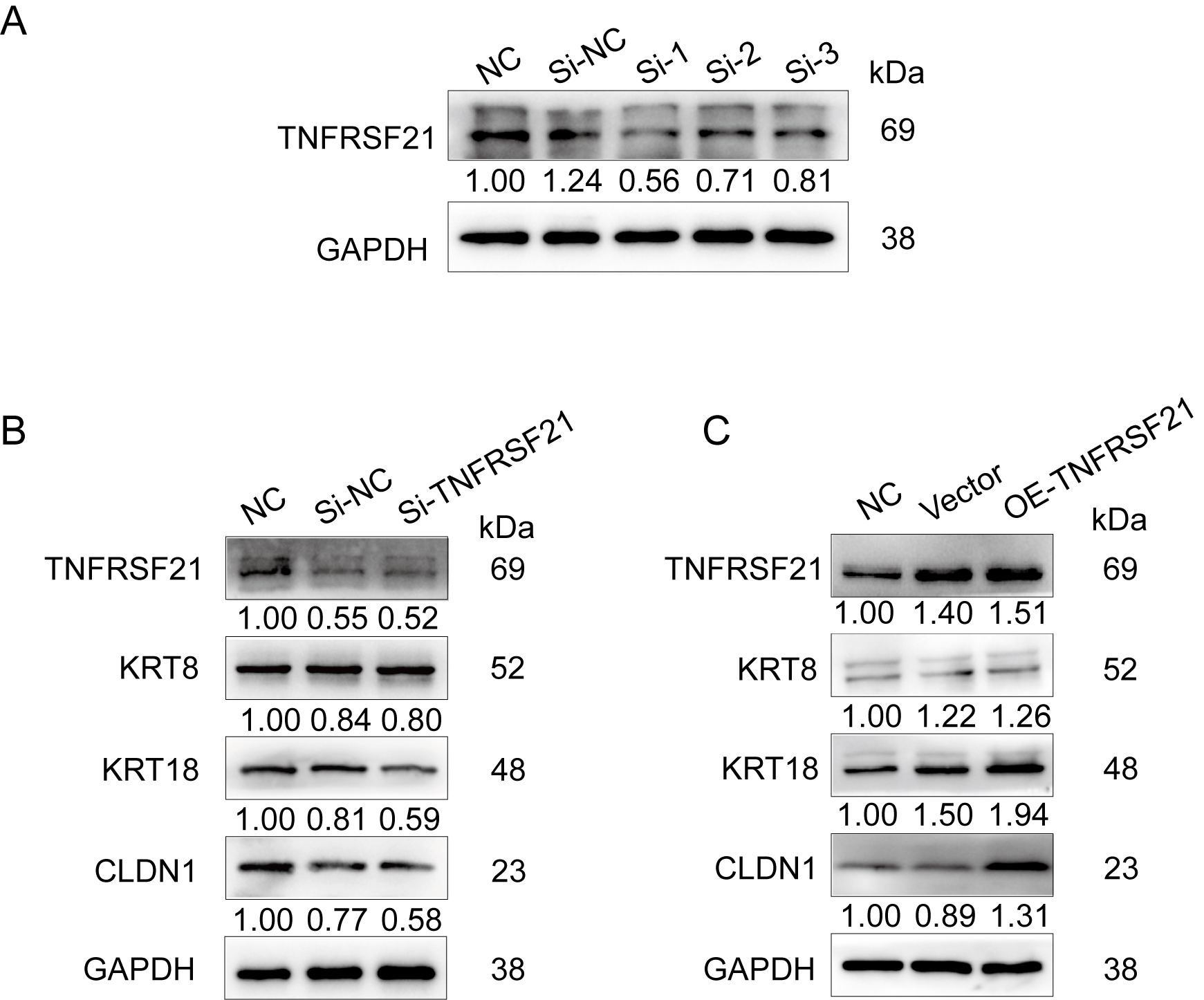 Fig. 3.
Fig. 3.
Knockdown and overexpression of TNFRSF21 demonstrate its regulatory role during keratinization. (A) Verification of TNFRSF21 knockdown efficiency with siRNA. (B) Knockdown of TNFRSF21 reduced the expression of KRTs. (C) Overexpression of TNFRSF21 significantly increased the expression of KRTs. KRTs, keratinization-related genes.
TNFRSF is a class of cell surface receptor that typically mediates physiological functions upon binding to its ligands [12]. We next investigated interactions between the TNFRSF family and their ligands during treatment for periodontitis and subsequent healing. Through protein–protein interaction (PPI) analysis of scRNA-seq data (Fig. 4A), we identified a significant interaction between TNFRSF21 and amyloid precursor protein (APP). Further validation via STRING database analysis of DEGs confirmed this interaction (Fig. 4B). Additionally, an immunoprecipitation (IP) assay provided direct evidence for the interaction between TNFRSF21 and APP in HOK cells (Fig. 4C). These observations confirm the strong interaction between TNFRSF21 and its ligand APP throughout the periodontitis recovery phase. Moreover, we speculate that the complex formation between TNFRSF21 and APP may play a synergistic role in modulating the functions of oral epithelial keratinocytes.
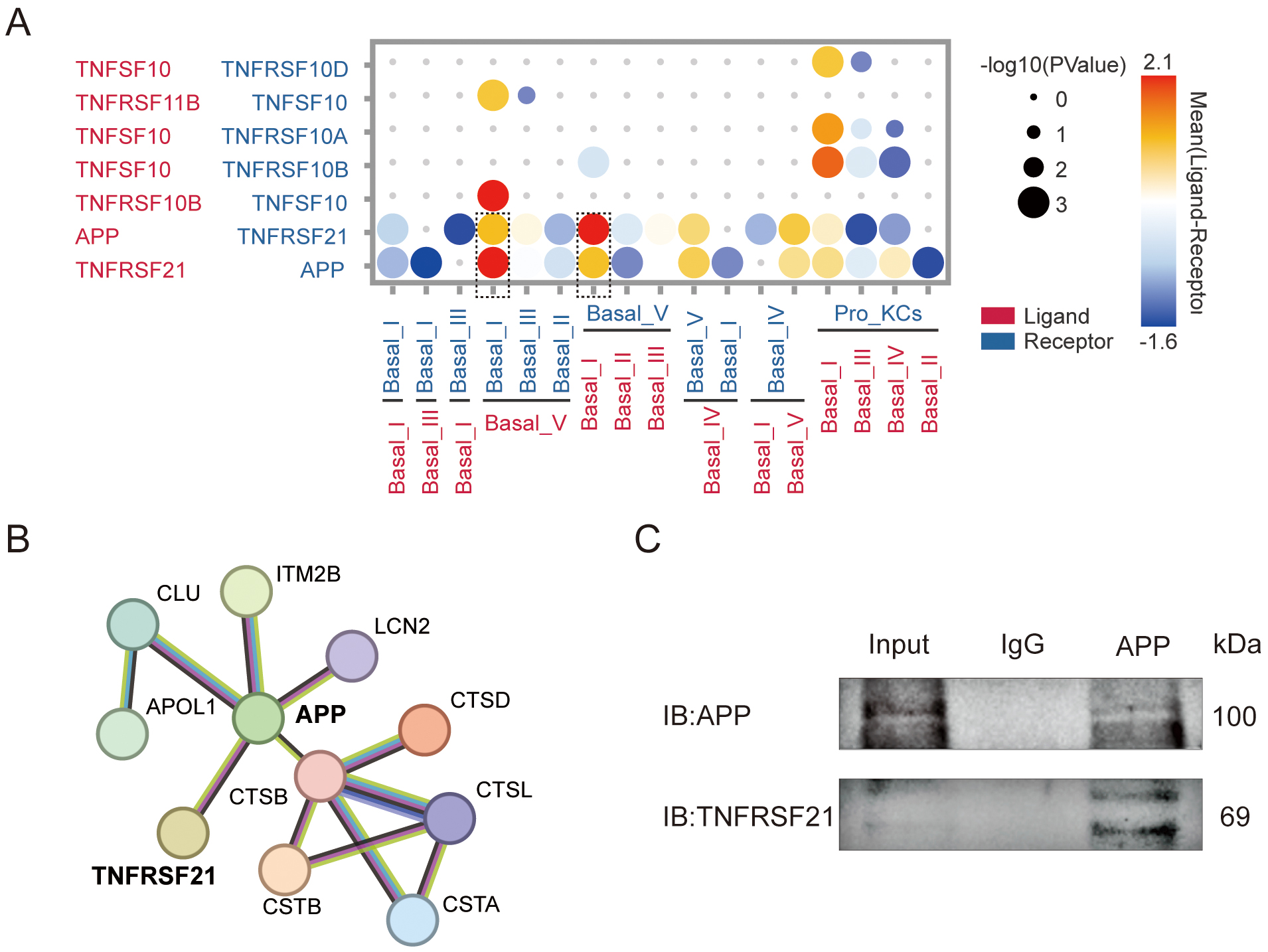 Fig. 4.
Fig. 4.
Interaction of TNFRSF21 and amyloid precursor protein (APP) during keratinization. (A) Protein–protein interaction (PPI) analysis revealed that TNFRSF21 and APP undergo significant interaction. The dashed box highlights that APP and TNFRSF21 have the strongest PPI within the TNFRSF family. (B) This interaction was further validated using the String database. (C) Western blot analysis confirmed the interaction between TNFRSF21 and APP in human oral keratinocytes (HOKs) cells.
We next conducted animal experiments to further investigate the role of TNFRSF21 in promoting keratinization and wound healing. Using a puncher, we created a 6-mm diameter wound on the back of C57BL/6 mice and subsequently injected plasmids into the wound site that contained either an empty vector, or a vector overexpressing TNFRSF21. Wound conditions were monitored and documented on days 1, 3, 5, 7, and 9 post-injection (Fig. 5A). On day 9, a biopsy of the wound tissue was performed, followed by hematoxylin and eosin (H&E) staining and immunofluorescence analysis. Our findings revealed that the plasmid overexpression group had approached full wound closure by day 9. In contrast, the control group still presented with a substantial wound area at day 9. Additionally, mice treated with the TNFRSF21-overexpressing plasmid showed enhanced wound healing, characterized by a more rapid decrease in wound size and elevated keratin expression levels compared to the control group (Fig. 5B,C,E). Systemic effects of the overexpressing plasmid on the heart, liver, spleen, lung, and kidney tissues were also investigated by H&E staining, but no evidence of toxicity was observed (Fig. 5D).
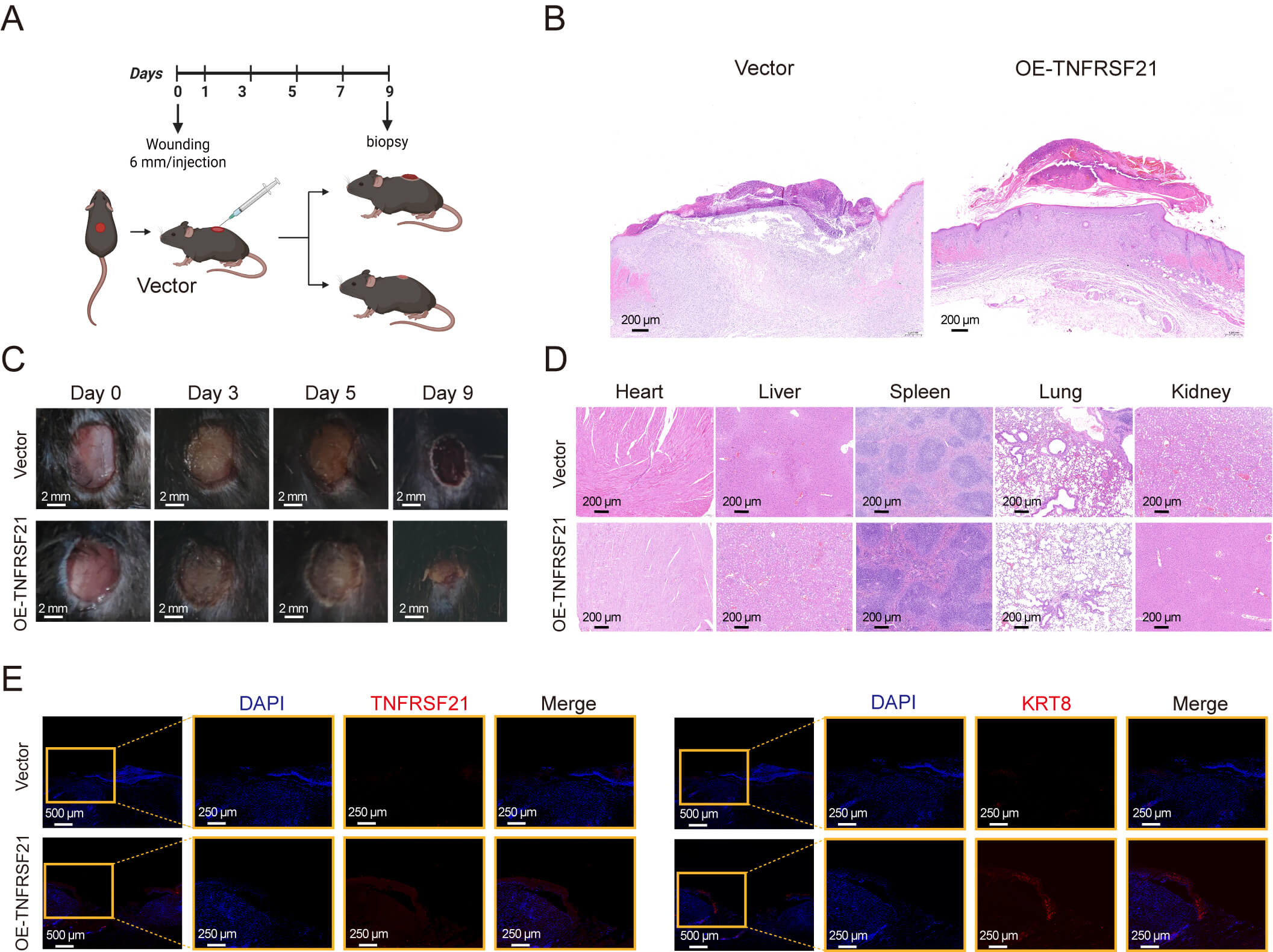 Fig. 5.
Fig. 5.
Overexpression of TNFRSF21 promotes keratinized wound healing in
mouse dorsal skin. (A) Diagram of the experimental design used to evaluate skin
wound healing in C57BL/6 mice. On day 0, a 6-mm full-thickness skin incision was
created using a biopsy punch as the wound model, followed by the injection of
either empty vector or overexpression plasmids into the wound site. (B)
Representative hematoxylin and eosin (H&E)-stained skin sections on day 9 after
the injection of empty vector or overexpression plasmid. Scale bar = 200 µm. (C)
Photographs of the wound area in C57BL/6 mice treated with either an empty vector
or an overexpression plasmid on days 0, 3, 5, and 9 during the wound healing
process. Scale bar = 2 mm. (D) Representative H&E-stained sections of the heart,
liver, spleen, lung, and kidney on day 9 after injection with empty vector or
overexpression plasmid. Scale bar = 200 µm. (E) Skin sections stained by
immunofluorescence on day 9 after injection of empty vector or overexpression
plasmid. The field of view is 5
Periodontitis is a chronic inflammatory disease that presents significant global
health challenges. It profoundly compromises oral health and has been linked to
systemic conditions, including diabetes and cardiovascular diseases [32, 33]. The
gingival epithelium serves as a critical protective barrier against pathogens and
toxins [34]. Bacterial toxins and inflammatory cytokines such as TNF-
In the initial stage of periodontitis, the plaque biofilm activates the host immune response, leading to the production of reactive oxygen species (ROS), which then attack biological macromolecules such as proteins, lipids, and DNA [36]. Furthermore, ROS impairs fibroblasts, reduces collagen synthesis, and activates matrix metalloproteinases (MMPs), resulting in degradation of the extracellular matrix [36]. Previous studies have reported a marked expansion of the TNFRSF21+ fibroblast population during the progression of periodontitis, leading to neutrophil infiltration [20, 37]. These fibroblasts secrete chemokines and cytokines such as C-X-C Motif Chemokine Ligand 1, which intensify inflammation in periodontal tissues and can also regulate osteocyte activity, affecting the metabolic balance and regeneration of periodontal bone [20]. In addition, the excessive accumulation of neutrophils in periodontitis may compromise the keratinization and barrier function of the gingival epithelium, which could in turn intensify the inflammatory response [6]. Similar to our study, previous research has shown that TNFRSF11A (also known as RANK) can significantly promote the proliferation and survival of thymic epithelial cells, thereby enhancing thymic regeneration through activation of the RANK-RANKL signaling pathway [38]. However, our study is the first to demonstrate that TNFRSF21 modulates keratin expression by HOK cells as periodontitis evolves. Moreover, we found that TNFRSF21 interacts with APP, with this interaction being particularly evident in PPI analysis of scRNA-seq data.
Previous research on APP has predominantly focused on its role in the
onset and progression of Alzheimer’s disease (AD) [39]. Accumulating evidence
indicates that periodontitis may indirectly aggravate AD pathology by enhancing
systemic inflammation and disrupting intestinal homeostasis [40, 41]. In AD,
APP is cleaved by
The current findings demonstrate that TNFRSF21 enhances wound healing
during periodontitis treatment via the regulation of keratinization and tight
junction integrity in oral epithelial cells. This potentially occurs through the
involvement of
The accession number of the single-cell RNA-seq reported in this paper comes from the Gene Expression Omnibus (GEO): GSE171213 (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE171213). Data supporting the results of this study can be provided at the reasonable request of the corresponding author.
Software, Investigation, Writing—original draft preparation: WZ, YZ, SJ. Writing—review and editing: HZ, WZ, WS, MW. Visualization: ZS, BL, FX, YL, YY. Supervision: HZ, WS, MW. Conceptualization, Funding acquisition: HZ, MW, WS. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All animal work was performed in accordance with the ARRIVE guidelines 2.0. The animal research project was approved by the Ethical Committee at Anhui Medical University on May 30, 2023, with the approval number LLSC20232087. Human Gingival Tissue Collection: Gingival tissues were collected from healthy volunteers or chronic periodontitis patients undergoing implant surgery at College & Hospital of Stomatology, Anhui Medical University. All subjects or their legal guardians signed an informed-consent form permitting the use of surplus surgical tissue for research. This study has been approved by College & Hospital of Stomatology, Anhui Medical University (Approval Number: T2021014). The study was carried out in accordance with the guidelines of the Declaration of Helsinki.
We thank Yi Zhang for providing technical assistance in single-cell RNA-Seq analysis.
This work was supported by the 2023 Disciplinary Construction Project in the School of Dentistry, Anhui Medical University (2023xkfyts01), the Key Project of Natural Science Research in Anhui Provincial Universities (2024AH050683).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/FBL48371.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





