1 Laboratory of Immunobiology, Department of Experimental Therapy, Hirszfeld Institute of Immunology and Experimental Therapy, Polish Academy of Sciences, 53-114 Wrocław, Poland
2 Department of Biostructure and Animal Physiology, Division of Histology and Embryology, Wrocław University of Environmental and Life Sciences, 50-375 Wrocław, Poland
3 Department of Immunology, Pathophysiology and Veterinary Preventive Medicine, Wrocław University of Environmental and Life Sciences, 50-375 Wrocław, Poland
4 Department of Functional Food Products Development, Wrocław University of Environmental and Life Sciences, 51-630 Wrocław, Poland
Abstract
Prolonged psychic stress leads to immune suppression, which preferentially affects the cellular immune response. Yolkin is an egg-derived protein with firmly established immunoregulatory activities in rodent models.
The aim of this work was to evaluate the effects of oral administration of yolkin, when mice experience prolonged exposure to immobilization stress, on in vivo and in vitro parameters of contact sensitivity (CS) to oxazolone (OXA).
BALB/c mice were exposed to 5-day immobilization stress, followed by immunization with OXA. Yolkin was applied in drinking water during stress. Ear thickness and auricle histology, concanavalin A (Con A)- and lipopolysaccharide (LPS)-stimulated production of interferon-gamma (IFN γ) and interleukin 6 (IL-6) in splenocyte cultures, Con A-induced splenocyte proliferation, as well as serum corticosterone levels and thymocyte number were analyzed.
We showed that the treatment with stress alone led to a lower thymocyte number and a decreased thickness of the auricle skin. Control mice that were stressed also exhibited an increase in the number and area of pustules in the epidermis. The treatment of stressed mice with yolkin resulted in a partial normalization of several parameters that were lowered in stressed mice, such as auricle thickness, thymus cell content, Con A-induced splenocyte proliferation, production of IFN γ and IL-6. The serum levels of corticosterone were correlated with several histological parameters.
Oral administration of yolkin normalizes the antigen-specific and nonspecific parameters of the immune system altered by chronic psychic stress in mice.
Keywords
- yolkin
- contact sensitivity to oxazolone
- chronic psychic stress
- corticosterone
- interleukin-6
- interferon gamma
Stress represents an organism’s response to various harmful external stimuli that leads to biochemical and physiological homeostasis being disturbed. Depending on duration and intensity, stress leads to profitable or undesirable consequences, including immunological status [1, 2, 3]. The most devastating is strong and long-term stress, lowering efficiency of the immune system, including the ability to generate antibodies following vaccination [4], and increasing susceptibility to developing autoimmune diseases such as multiple sclerosis (MS) [5], gastric mucosal damage [6] or irritable bowel disease [7]. The association of the central nervous system (CNS) with the hormonal and immune systems plays a fundamental role in the interpretation of effects of stress on the immune response [3, 8, 9, 10]. It has also been documented that chronic stress affects the immune system through the toll like receptor 4 (TLR4) signaling pathway [11]. Generally, the T cell lymphopoiesis during acute psychic stress is much more affected than that of B cells [9, 12, 13] and delayed type hypersensitivity reactions are preferentially inhibited in comparison to the humoral immune response [10, 14, 15, 16, 17]. Short acute stress, applied before immunization stimulates, and prolonged stress suppress the immune response [9, 10, 18]. The effects of psychic stress are also associated with accumulation of myeloid-derived suppressor cells in bone marrow [19], alterations in skin dendritic cell morphology suggesting a reduction in the antigen presenting function [16], decreased natural killer (NK) cell activity in the spleen and lower phagocytic function of macrophages [15]. In addition, restraint stress delays wound healing by enhancing infiltration of neutrophils to wounds, thus extending its inflammatory phase, and decreases macrophage markers associated with wound healing as well as phagocytic activities of phagocytic cells and cell surface expression of adhesion molecules on blood monocytes [20].
The hitherto studies demonstrated that immunoregulatory proteins and peptides from colostrums, such as colostrinin (proline-rich polypeptide, PRP) and lactoferrin (LF) affect CNS. Colostrinin improves cognitive functions [21] and LF ameliorates stress reactions [22], pain [23] and lowers the adverse effect of psychic stress on the immune response [10, 24].
The egg yolk of birds contains numerous constructive, regulatory and protective constituents, essential for proper embryo development [25, 26, 27]. The research of the last decade has led to identification, isolation and description of one of its constituents—yolkin, a vitellogenin-derived protein [28]. Immunoregulatory, antiviral, precognitive, neuroprotective and antioxidant properties of yolkin were described [29]. The abilities of yolkin to stimulate the humoral immune response in mice [30] and contact sensitivity in juvenile mice [31] were also recently demonstrated. A precursor of yolkin was also obtained in a recombinant form which indicates a possibility for application of this protein in health-promoting nutraceutics, for example in prophylaxis of neurodegenerative disorders and immune deficiencies [28, 29]. Of interest, an egg yolk preparation, in mice subjected to physical stress, restored lipopolysaccharide (LPS)-induced B cell proliferation, levels of interleukin (IL)-2 and IL-10, normalized production of IgA and IgG and improved NK cell cytotoxic activity [32].
The aim of this study was to evaluate if yolkin, administered to mice per os during long-term immobilization stress, would alter the immune parameters of contact sensitivity (CS) to oxazolone (OXA). In particular, the production of cytokines in splenocyte cultures was determined, as well as the serum level of corticosterone and thymocyte cell number. In addition, the thickness and histological picture of the skin of the inflamed auricle was analyzed by histologists to identify immune cell engagement in the inflammatory processes.
8 week-old female BALB/c mice were purchased from Envigo, Netherlands. The Local Ethics Committee of the Hirszfeld Institute of Immunology and Experimental Therapy in Wrocław, Poland gave consent # 039/2022 to perform the experiments. The mice were housed in cages at 21–22 °C with a 12/12-h light/dark cycle and were fed a commercial, pellet food as well as water ad libitum.
Hanks’ balanced salt solution (HBSS, L0606), phosphate buffered saline (PBS, L0615) were from Biowest (Nuaillé, France), bovine serum albumin (BSA, A7030), oxazolone (OXA, E0753) and 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT, M2128) were from Sigma-Aldrich (St. Louis, MO, USA), May-Grünwald (1030.2) and Giemsa stains (1020.2) from Aqua-Med, Łódź, Poland. The reagents used in the histological and histochemical investigations were as follows: formalin (114321729), xylene (115208603) from Chempur, Piekary śląskie, Poland; hematoxylin (497-03971), eosin (17372-87-1), toluidine blue (89640), Meyer’s hematoxylin (MHS16) from Merck KGaA, Darmstadt, Germany; Ly6G antibody (clone RB6-8C5, MA1-10401), T lymphocytes antibody (clone SP7, MA5-14524) from Thermo Fisher Scientific, Waltham, MA, USA; Langerin/CD207 antibody (clone 929F3.01, DDX0362P-100) from Novus Biologicals, Littleton, CO, USA; ImmPRESS Reagent Kit anti-rabbit Ig (MP-7451) and ImmPRESS Reagent Kit anti-rat IgG (MP-7444), chromogens: ImmPACT™ DAB (SK-4105) and ImmPACT™ NovaRED™ (SK-4805) from Vector Laboratories Inc, Newark, CA, USA.
Yolkin was prepared from hen egg yolks as previously described [30]. The yolkin preparation contained three main proteins with a molecular weight below 45 kDa.
The mice were divided into four groups: (1) control, unstressed mice (Ctrl), (2) unstressed mice drinking yolkin solution in tap water (Yolkin), (3) stressed mice (Ctrl Stress) and (4) stressed mice drinking yolkin (Yolkin/Stress). The control, stressed mice underwent stress immobilization, i.e., they were kept in 50 mL perforated tubes, 4 h daily, for 5 consecutive days and had access during period 20 h/day to standard filtered tap water. Other stressed mice had access during this period to filtered tap water containing 5 µg/mL of yolkin. Unstressed mice from the Ctrl and Yolkin groups during the stress period remained in their cage, but similarly to stressed mice, without access to water and food for 4 h daily. 24 h later the mice were administered buprenorphine and the auricle edema was measured. Then, the mice were anaesthetized with 3% of isoflurane, bled and sacrificed by cervical dislocation. The auricles were cut off for histological examinations and thymocytes and splenocytes were isolated for immunological analysis. The experimental design is presented in Fig. 1.
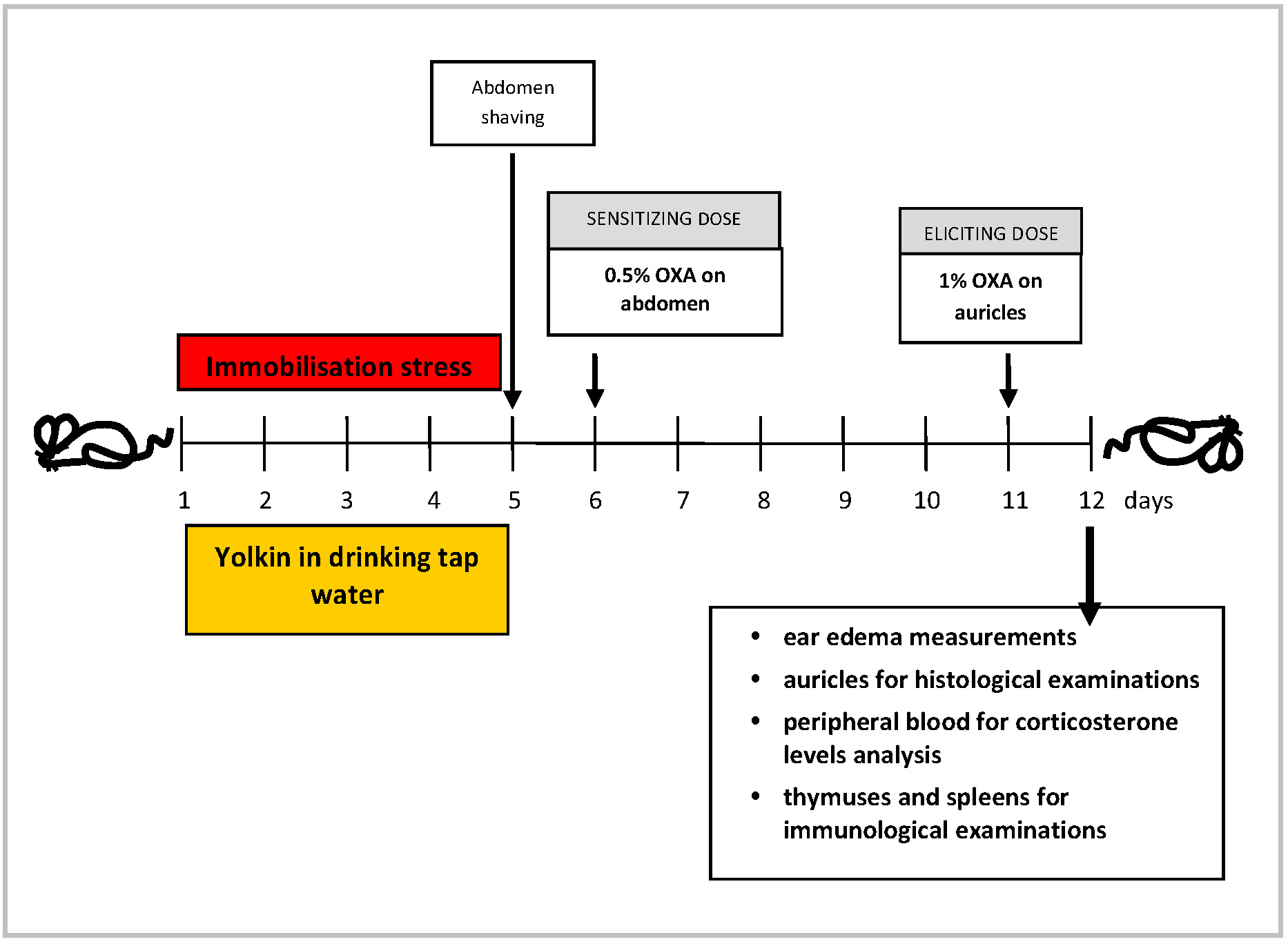 Fig. 1.
Fig. 1.
The experimental design. OXA, oxazolone.
After buprenorphine (Bupaq Multidose) administration and measurement of the ear thickness, the mice were anaesthetized with isoflurane (Isoflurin, Vetpharma Animal Health, SL, Barcelona, Spain) bled and sacrificed by cervical dislocation. The thymuses and auricular lymph nodes were isolated and disintegrated to single cell suspension in cold PBS, using syringe piston and plastic screen.
The splenocytes were obtained as above using 0.83% NH4Cl solution, instead of PBS, to lyze erythrocytes for 5 min incubation at room temperature (RT). The cells were subsequently washed twice with Hanks’ solution, passed through cotton wool columns to dispose debris, and re-suspended in the culture medium consisting of RPMI-1640 (Roswell Park Memorial Institute 1640 medium, L0500, Biowest, Nuaillé, France), supplemented with 10% fetal calf serum (FCS, S181B, Biowest, Nuaillé, France), sodium pyruvate (L0642, Biowest, Nuaillé, France), 2-mercaptoethanol (M3148, Sigma-Aldrich, St. Louis, MO, USA) and antibiotics (L0022, Biowest Nuaillé, France). For enumeration of cells in the lymphoid organs Türk’s solution (109277, Sigma-Aldrich, St. Louis, MO, USA) was used. The numbers of viable cells were counted in a hemocytometer using 0.1% Trypan blue reagent (T6146, Sigma-Aldrich, St. Louis, MO, USA) in PBS.
The pooled splenocytes (5
Circulating blood from the retroorbital plexus was obtained in isoflurane anesthesia on the day of elicitation of contact sensitivity. The sera were isolated and kept at –80 °C until corticosterone determination using a Demeditec Diagnostics GmbH kit (DEV9922, Kiel, German).
The originally described test [33] was used with some modifications. 100 µL of 0.5% OXA dissolved in acetone was applied on the shaved abdomen. After 5 days, 50 µL of 1% OXA was administered to both sides of the auricles. The ear thickness was measured by means of a spring caliper after 24 h following application of the second dose of antigen. The results were shown as an antigen-specific increase of the ear thickness, i.e., background (BG) values (mice given only the eliciting dose of the antigen) were subtracted.
The paraffin auricle sections were prepared in a standard procedure and stained
with hematoxylin and eosin (H&E). The staining of cells with respective
antibodies was performed according to the manufacturers’ instructions
(Immunohistochemistry kits; ImmPRESS Reagent Kit anti-rabbit Ig (MP-7451) and
ImmPRESS Reagent Kit anti-rat IgG (MP-7444), Vector Laboratories Inc, Newark, CA,
USA). Cells were counted in 3 random fields for each animal. In the morphometric
analysis a cross-sectional area of the pustules in the epidermis (expressed in
µm2) was measured. Quantitative and morphometric measurements of
pustules were performed on both sides of the auricles, from the base toward the
apex. Nikon Eclipse 80i microscope (Nikon Corporation, Tokyo, Japan), with
imagine software NIS Elements (Nikon Corporation Tokyo, Japan), at 100
The calculations and statistical analysis were accomplished using
Microsoft® Excel XP (Microsoft, Warszawa, Poland) and
STATISTICA® 7.0 programs for Windows (StatSoft Polska,
Kraków, Poland). Eight mice per group were used in this study. The
statistical significance of differences between the mean values was tested by
means of variance analysis (ANOVA) for the one-factor classification with post
hoc analysis (multi comparisons) by RIR Tukey’s test. The evaluation of normality
of the variable distribution was performed with the W Shapiro-Wilk test, and the
evaluation of the homogenous variance assumption with Brown-Forsyth’s test.
Moreover, the existence of dependencies between group means and variances were
verified. Fulfillment of the above assumptions (normal or symmetric distribution
of variables, homogeneity of variance, lack of dependencies between means and
variances) made it possible to use the variance analysis for testing the mean
equality hypothesis. If these assumptions were not fulfilled, a non-symmetrical
Kruskal-Wallis’ test was used. The significance was set at p
The mice were subjected to immobilization stress for 5 days followed by immunization with OXA. One group of mice was administered yolkin in their drinking water throughout the stress period. Fig. 2 shows the results of the measurement of the auricle edema after 24 h following elicitation of the contact sensitivity reaction. Yolkin alone had no effect on the magnitude of the CS reaction, but the psychic stress led to a significant (more than 50%) decrease of the ear edema. Although the treatment with yolkin did not completely restore the CS response to control levels, its reconstituting effect on the ear edema was statistically significant.
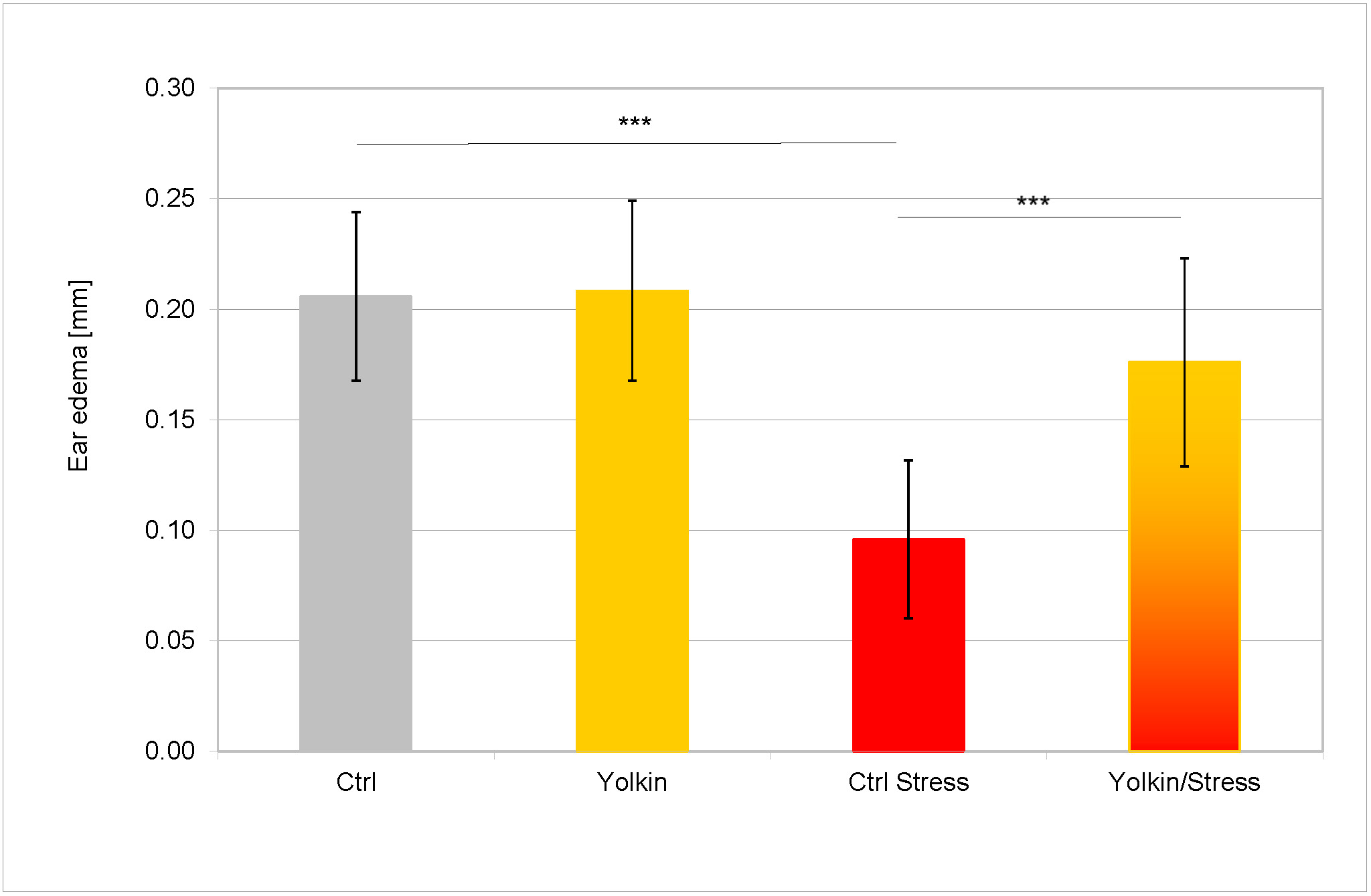 Fig. 2.
Fig. 2.
The effects of stress and yolkin on the antigen specific
thickness of auricles of mice with fully developed CS to oxazolone. The exposure
of mice to stress and the performance of the contact sensitivity test are
described in sections 2.4 and 2.8. The experimental design is presented in the
Materials and Methods section. Briefly, mice drank water containing yolkin (5
µg/mL) for 5 days during stress. Next day 0.5% OXA was applied on
the skin of their abdomens. The CS effectual reaction was evoked after 5 days by
application of 1% OXA on both sides of auricles. The thickness of auricles was
determined after 24 h with a spring caliper. The antigen specific increase of ear
thickness (BG values are subtracted from the responses of sensitized mice) is
presented as a mean value of ear thickness measured in 8 mice (16 determinations)
in mm
As shown in Fig. 3A, at the time of elicitation of CS, the IFN
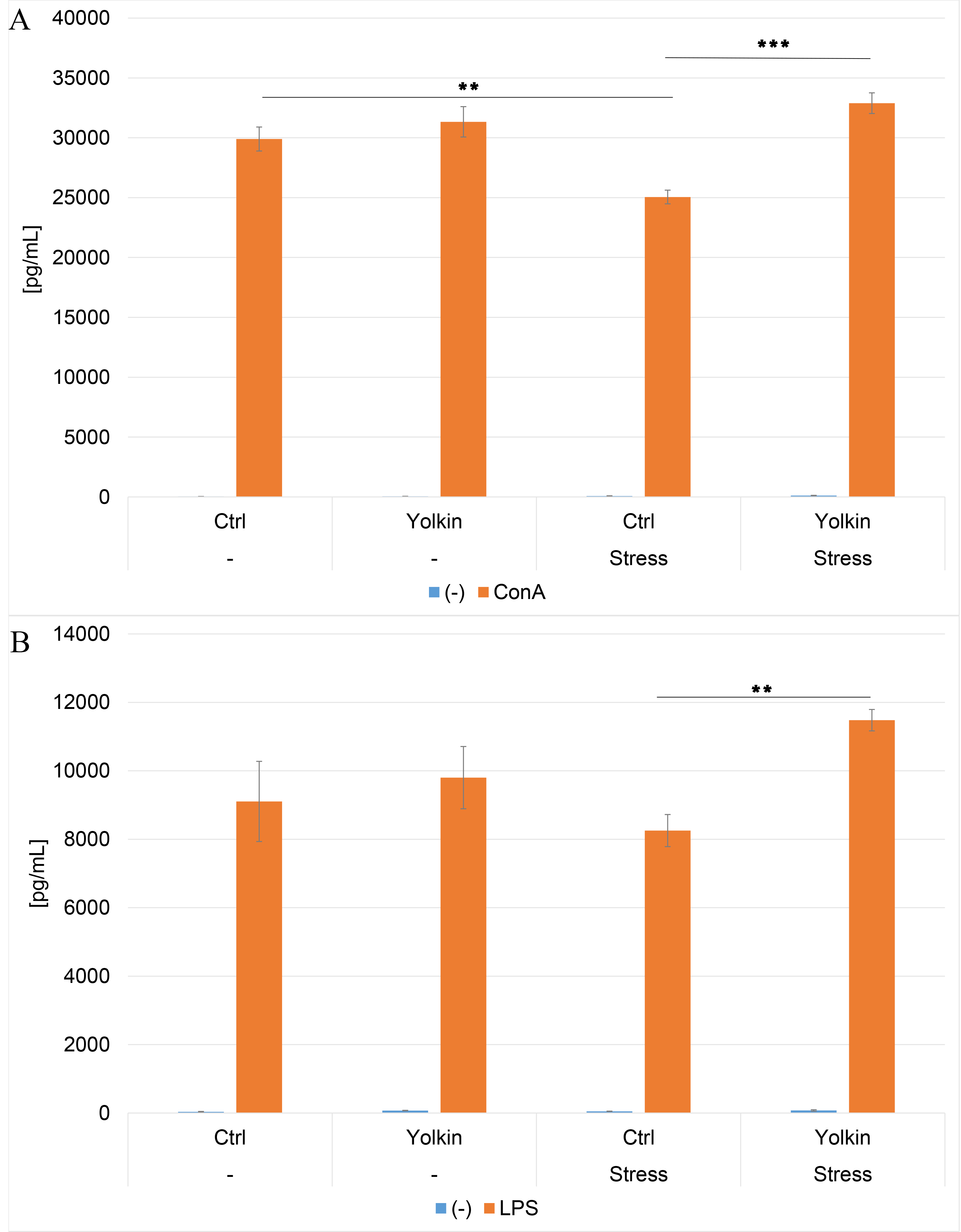 Fig. 3.
Fig. 3.
The ex vivo effects of oral yolkin administration
during restraint stress and development of contact sensitivity on Con A- and
LPS-induced IFN
Similar results were obtained in the splenocyte cultures stimulated with LPS
(Fig. 3B) although the upregulatory effect of yolkin on IFN
A similar inhibitory effect of stress was observed in IL-6 production by Con A-induced splenocytes (Fig. 4). The effects of yolkin alone or stress were not significantly inhibitory. On the other hand, the treatment of the stressed mice with yolkin resulted in the complete recovery of IL-6 production over the control values.
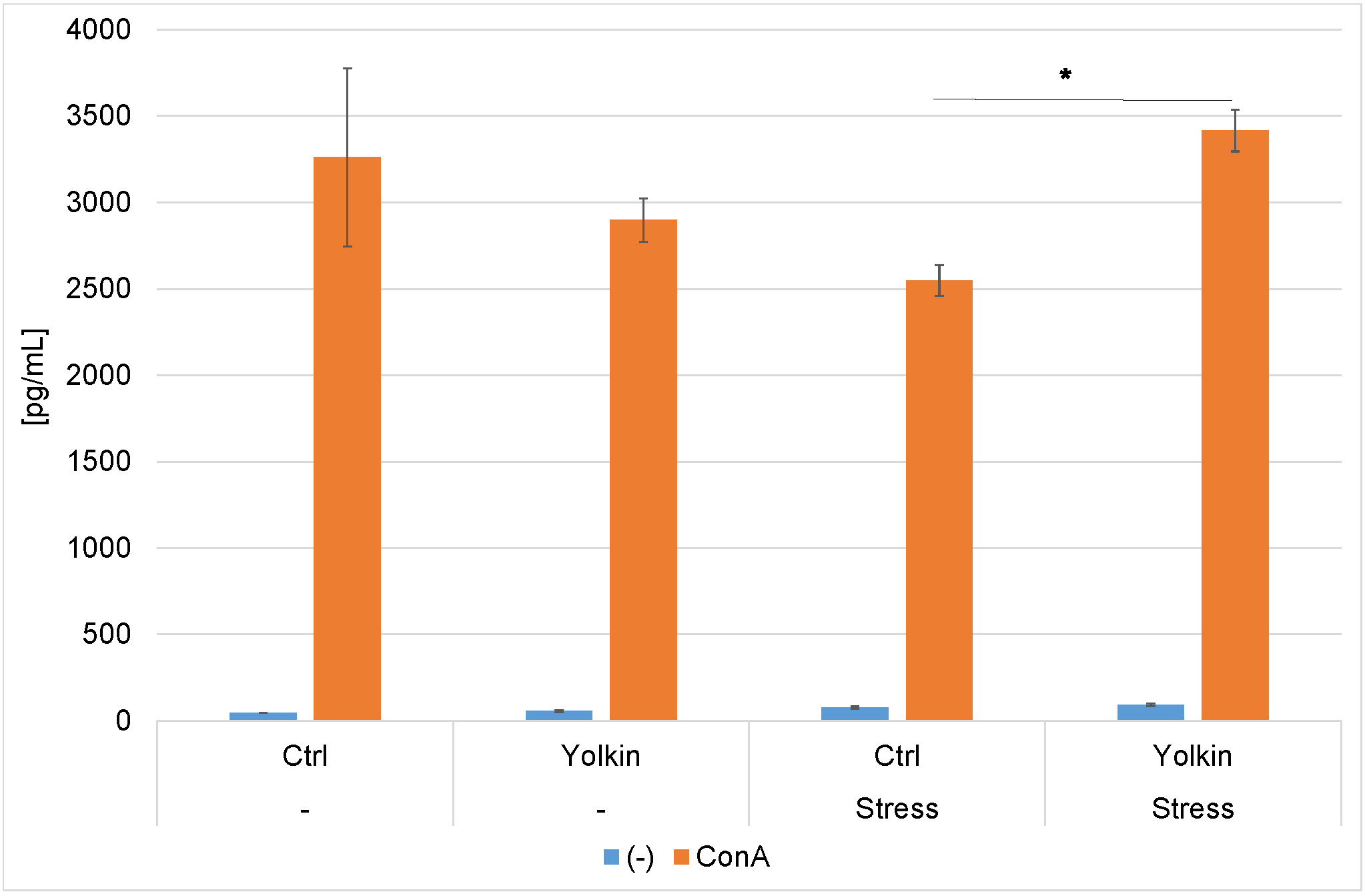 Fig. 4.
Fig. 4.
The ex vivo effects of oral yolkin administration
during restraint stress and development of contact sensitivity on Con A-induced
IL-6 production by splenocyte cultures. The treatment of mice was described in
the legend to Fig. 2. The pooled splenocytes from four mice were isolated 24 h
after elicitation of the CS. The results are presented as a mean value from
triplicate measurements in pg/mL
As shown in Fig. 5A, at the time of CS induction, the levels of proliferation in Con A-stimulated splenocyte cultures were comparable in control and yolkin-treated mice. A lower level of proliferation of splenocytes from stressed mice was found. However, this effect was reversed in the stressed mice drinking the yolkin solution. In the case of LPS-induced splenocyte proliferation, there was no reduction in proliferation (Fig. 5B).
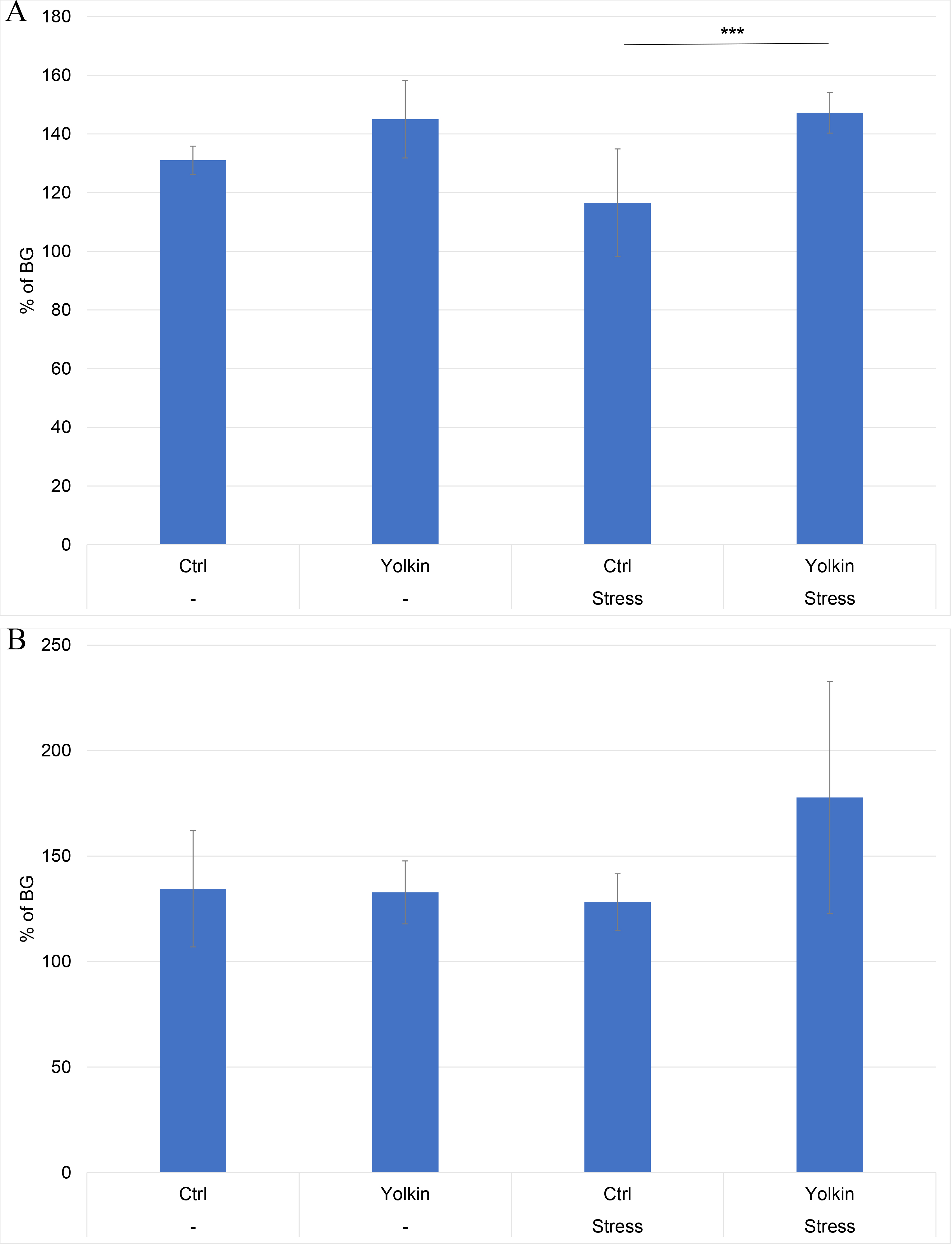 Fig. 5.
Fig. 5.
The ex vivo effects of oral yolkin administration
during restraint stress and development of contact sensitivity on Con A-induced
(A) and LPS-induced (B) splenocyte proliferation. The treatment of mice was
described in the legend to Fig. 2. The pooled splenocytes from four mice
(repeated 2 times) were isolated at 24 h after elicitation of the CS. The
percentage of proliferation was calculated relative to the proliferation
determined in the background group of mice. The results are presented as mean
values of ten measurements
The serum corticosterone levels in mice upon elicitation of the CS reaction are shown in Fig. 6. Its concentration was significantly elevated in the mice treated with yolkin alone but not in the stressed mice. However, the corticosterone levels were lower in the mice subjected to stress and treated with yolkin.
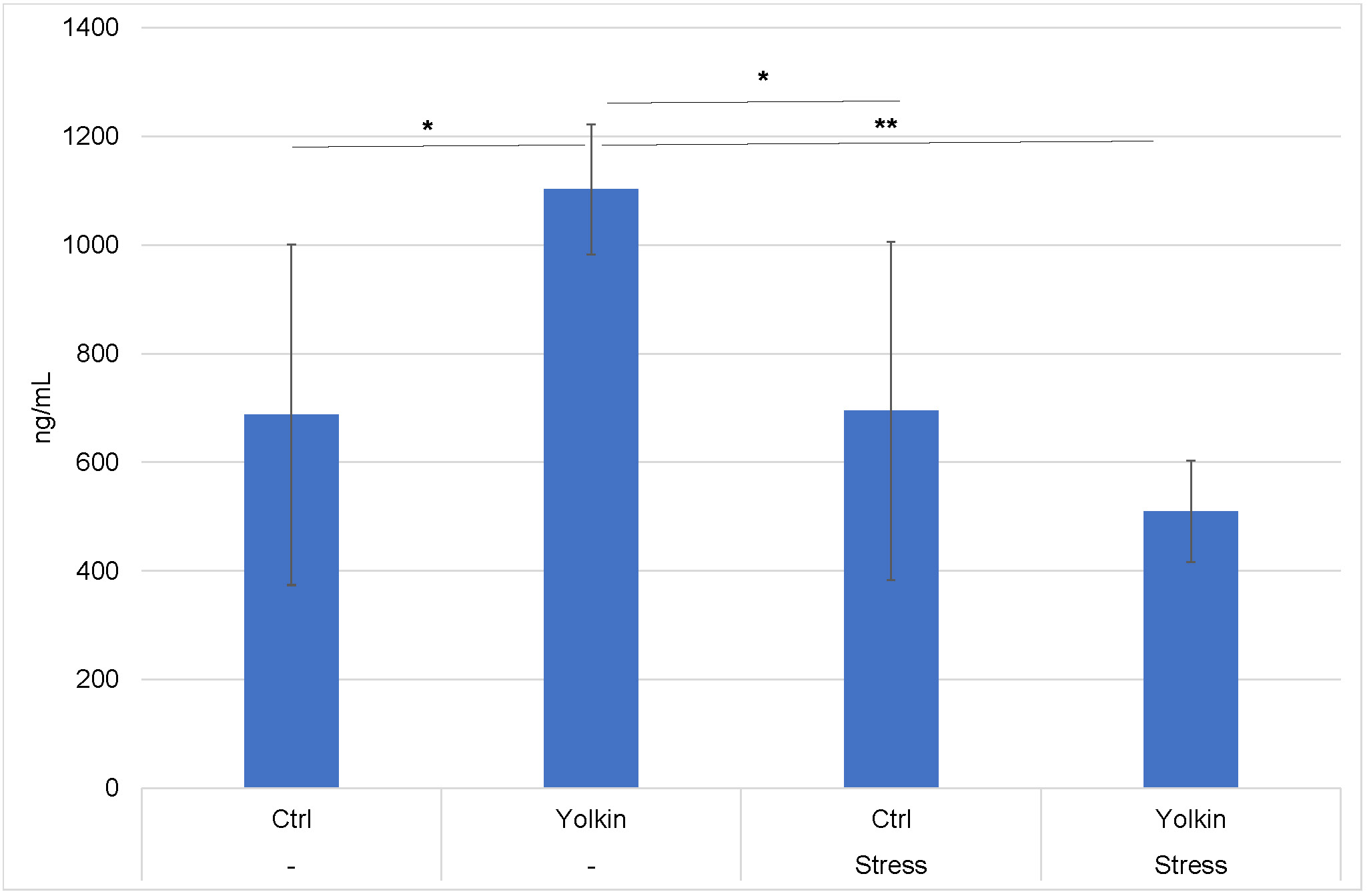 Fig. 6.
Fig. 6.
Serum corticosterone levels in stressed mice with
developed contact sensitivity. The treatment of mice was described in the legend
to Fig. 2. The peripheral blood was isolated from mice 24 h after elicitation of
the CS. The results are presented as mean values in ng/mL
As shown in Fig. 7 the number of thymocytes, calculated as a ratio of cell number per body weight, in mice subjected to the immobilization stress, was decreased. The administration of yolkin to the unstressed mice did not change the thymocyte number. However, the treatment of stressed mice with yolkin caused a significant recovery of the thymocyte numbers.
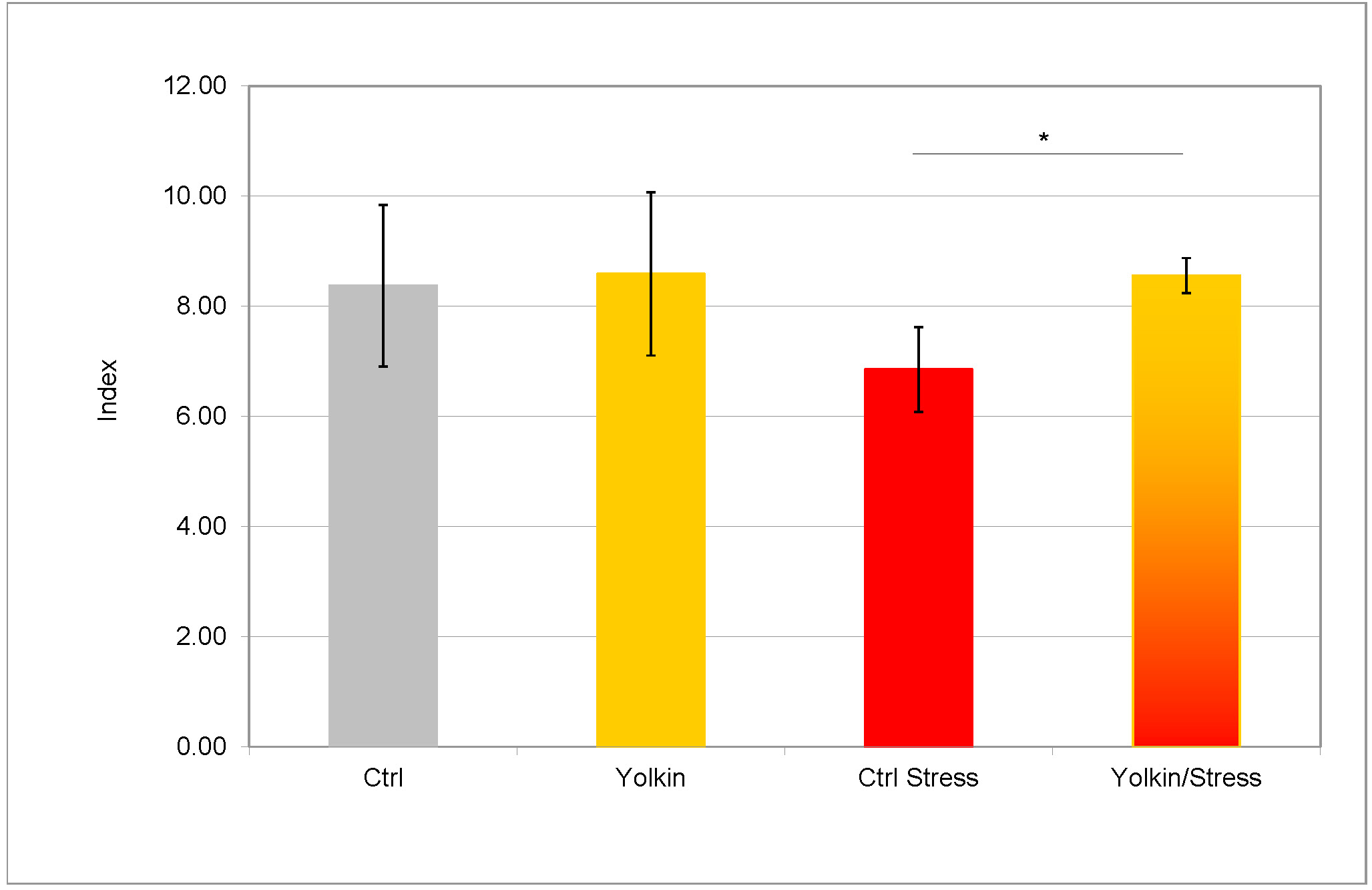 Fig. 7.
Fig. 7.
The effects of immobilization stress on the number of
thymocytes. The treatment of mice was described in the legend to Fig. 2.
The thymuses were isolated from mice 24 h after elicitation of the CS.
The results are presented as mean values of the thymocyte number index (thymocyte
number
Figs. 8,9 illustrate the histological picture of auricles. Background (BG) group of mice represented mice sensitized to OXA, but instead receiving the eliciting dose of antigen they were treated only with acetone (vehiculum) onto the auricles. Therefore, they represent a group with nonspecific skin irritation. In the auricles from the BG group of mice (Fig. 8A and Fig. 9A), focal and weak inflammatory infiltrates, mostly composed of Gr-1 positive cells (neutrophils), were found. The infiltrates were present mainly near the base of the auricles. The inflammatory process occurred in the perivascular zone of the dermis and in the epidermis in the form of pustules, which were also Gr-1 positive in the immunohistochemical analysis. The morphometrical analysis revealed that pustules were in the early stage of formation, with the number of pustules and the area of lesions being the lowest in comparison to other groups (Table 1). Only very small subcorneal pustules were observed. Immunohistochemical staining revealed that the number of T lymphocytes was the smallest, but the number of mast cells was the highest of all the studied groups. The number of Langerhans cells counted in the high power field was comparable to other tested groups. Because of lack of significant differences in the contents of lymphocytes, mast cells and Langerhans cells between the studied groups, the results were not shown.
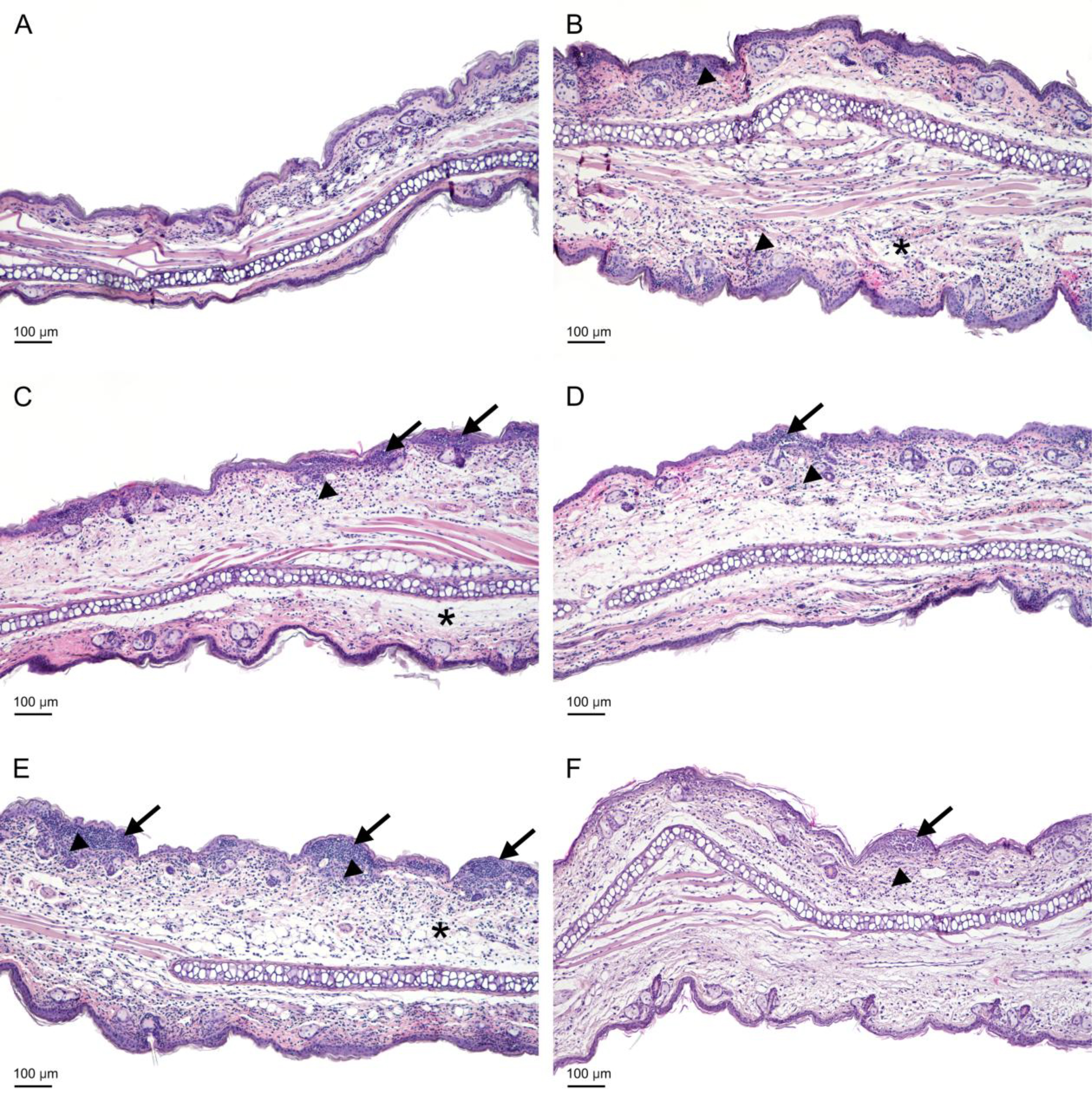 Fig. 8.
Fig. 8.
The histological examination of the auricles of the experimental mice. Mice were administered yolkin in their drinking tap water, at a concentration of 5 µg/mL, for 5 consecutive days when they were stressed. Then the mice were treated with OXA to develop the CS (as described in the legend to Fig. 2). The auricles were isolated from mice 24 h after elicitation of the CS. (A) BG group (weak, focal infiltrate). (B) Ctrl group (edema and extensive neutrophilic infiltration in the dermis). (C) Ctrl group (edema and infiltration in the dermis, medium-sized pustules in the epidermis). (D) Yolkin group (moderate infiltration in the dermis and small pustules in the epidermis). (E) Ctrl Stress group (severe infiltration and edema in the dermis, large pustules in the epidermis). (F) Yolkin/Stress group (moderate infiltration in the dermis, small and medium pustules in the epidermis). Arrows – pustules, arrowheads – infiltration, asterisks – edema; scale bar = 100 µm. BG, background group (mice given only the eliciting dose of the antigen).
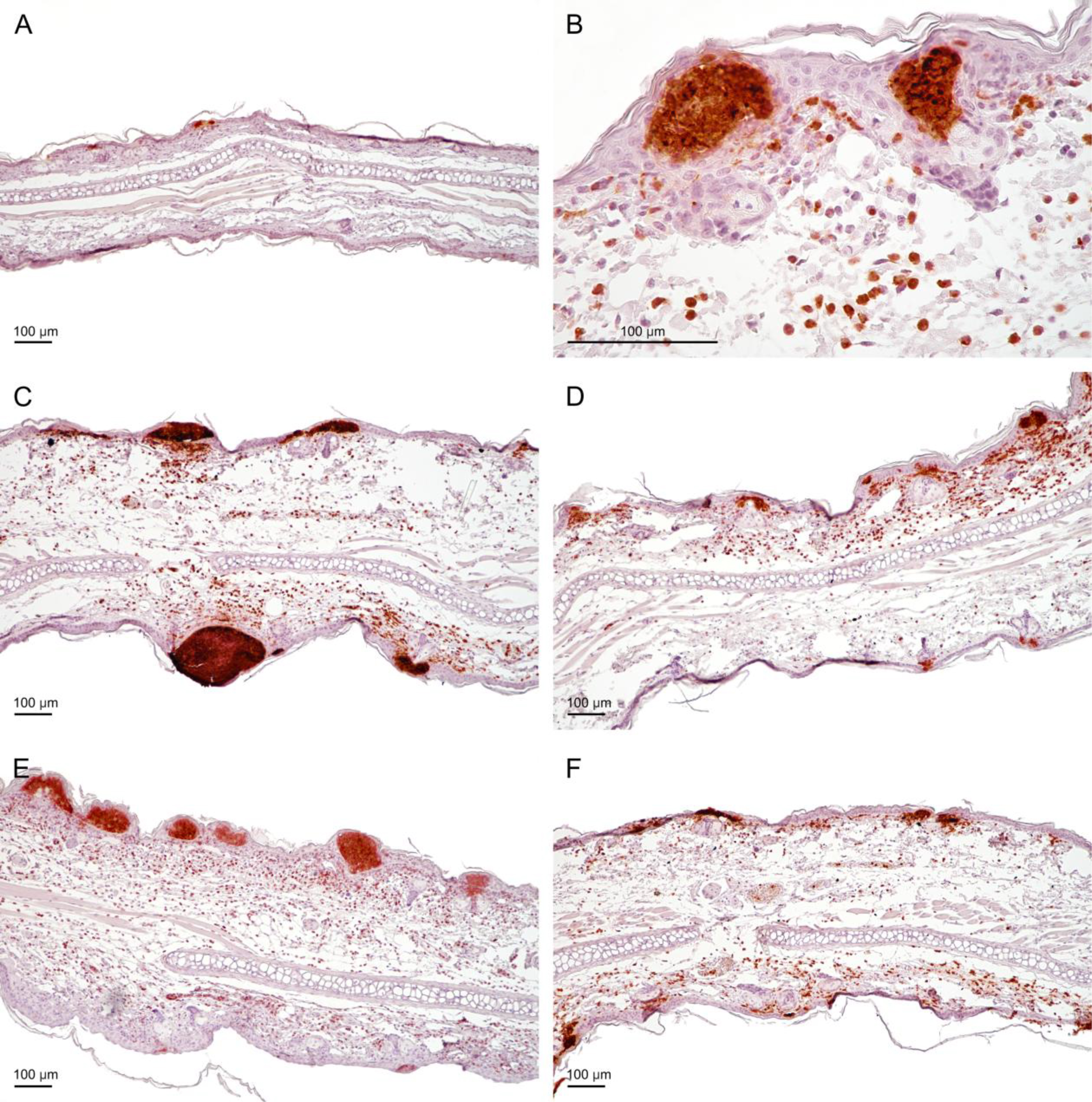 Fig. 9.
Fig. 9.
The presence of neutrophils in the auricles of the experimental mice. Mice were administered yolkin in their drinking tap water, at a concentration of 5 µg/mL, for 5 consecutive days when they were stressed. Then the mice were treated with OXA to develop the CS (as described in the legend to Fig. 2). The auricles were isolated from mice 24 h after elicitation of the CS. Neutrophils were detected with rat anti-mouse Ly6G antibody (component of antigen Gr-1, clone RB6-8C5). (A) BG group, receiving only the eliciting dose of the antigen (very small subcorneal pustules). (B) Ctrl group, higher magnification of mouse auricle (strong infiltration in the subepidermal region of the dermis, and pustules in the epidermis). (C) Ctrl group (visible infiltration and medium sized and large pustules). (D) Yolkin group (moderate infiltration in the dermis and small pustules). (E) Ctrl Stress group (extensive infiltrate in the dermis and numerous large pustules). (F) Yolkin/Stress group (moderate infiltration in the dermis and small pustules in the epidermis); scale bar = 100 µm. BG, background group (mice given only the eliciting dose of the antigen).
| Group | Number and pustule area [µm2] | |||||||
| 5 × 102 | 103 | 2 × 103 | 5 × 103 | 104 | 1.5 × 104 | 2 × 104 | ||
| BG | 10 | 3 | 2 | 2 | 1 | 0 | 0 | 0 |
| Control | 18 | 13 | 10 | 14 | 8 | 1 | 1 | 3 |
| Yolkin | 14 | 17 | 19 | 20 | 4 | 1 | 0 | 0 |
| Control stress | 36 | 26 | 21 | 16 | 7 | 5 | 3 | 4 |
| Yolkin/stress | 17 | 18 | 13 | 12 | 2 | 0 | 0 | 0 |
The treatment of mice was described in the legend to Fig. 2. The auricles were isolated from mice 24 h after elicitation of the CS. BG, background group (mice given only the eliciting dose of the antigen).
In the control, immunized and non manipulated group (Fig. 8B,C and Fig. 9B,C), strong inflammatory infiltrates in the dermis and very numerous pustules (small, medium and large) in the epidermis, along the entire auricles, were registered (Table 1). Because of the intensification of the inflammatory process, the number of neutrophils increased twofold in comparison to the background group. The number of lymphocytes increased, compared to the background group, but the number of Langerhans cells and mast cells did not change. The morphometrical analysis revealed an increase in the thickness of auricles in the control group. On the H&E stained sections, a loosening of collagen fibers, indicating edema, was observed.
In the group of stressed mice, the inflammatory process in the dermis was comparable to the control group, with extensive, neutrophilic infiltrations in the dermis (Fig. 8E and Fig. 9E). The inflammatory process in the epidermis of stressed mice was more profound than in the unstressed group regarding area and number of pustules (Table 1). The auricle thickness was still significant and swelling of the connective tissue was visible, indicating edema. The numbers of T lymphocytes (CD3+), Langerhans (CD207+) and mast cells, were similar to those in the control group (data not shown).
In the group of mice treated with yolkin and yolkin-treated stressed mice, a diminution of the inflammatory process was observed (Fig. 8D,F and Fig. 9D,F). There were still moderate neutrophilic infiltrates in the auricle dermis, but was expressed less than in the control group and mice subjected to the stress. The number of neutrophils also decreased. The formation of neutrophilic pustules was limited to small and medium lesions (Table 1). In both groups the morphometric measurements demonstrated a decrease in the auricle thickness which indicates a reduction of edema. The number of lymphocytes, mast cells and Langerhans cells did not change in comparison to other groups.
The quantitative parameters of the inflamed auricles regarding the size and distribution of pustules is presented in Table 1.
Preliminary studies on the biological activity of yolk protein water extracts [32] strongly suggested that yolkin may have a normalizing effect on neurologic pathologies and physiological processes modified by psychic stress. In this respect yolkin bears some similarities to colostrinin [21] and particularly to lactoferrin [10]. In this work, contact sensitivity to oxazolone was applied [33], as a model of delayed type hypersensitivity, which is more susceptible to suppression by prolonged psychic stress than the humoral immune response [17]. Yolkin suppresses delayed type hypersensitivity in adult mice, probably due to the ability to induce high levels of serum corticosterone [34] and preferential suppression of Th1-type activity [30]. When discussing the regulatory effects of yolkin on CS modified by psychic, restraint stress, a release of corticosterone upon stress should be also considered in the final interpretation of the results. Hence, the changes in the investigated in vitro, in vivo and histological parameters in this model may result from the individual or combined actions of the applied factors (long-term stress and yolkin), which are both known to suppress cellular immune response. It should be mentioned that both yolkin [35] and restraint stress [11] engage toll like receptor 4 (TLR4) and the phosphoinositide 3-kinases/protein kinase B (PI3K/Akt) signaling pathway, although its activation is suppressed in the latter case.
The proliferative response of splenocytes, induced by ConA and LPS is suppressed
by steroids [36]. Likewise, both Th1- and Th2-derived cytokines are inhibited by
corticosterone [37]. In our model, both Con A- and LPS-induced IFN
The results suggest that yolkin restores the impairment of IFN
In our study we found some drop in the proliferative response of splenocytes from stressed mice to Con A (Fig. 5A), which was reversed by yolkin. Although a similar effect was not found by others [17], an inhibition of T cell proliferative response to mitogens in stressed mice was reported in another study [39]. On the other hand, immobilization stress had no effect on LPS-induced splenocyte proliferation (Fig. 5B), which is consistent with B cells’ resistance to steroids as described by others [40, 41]. The stimulation of LPS-induced proliferation in mice subjected to stress and treated with yolkin may be associated with the lowest serum level of corticosterone with the immune suppressive property among these mouse groups (Fig. 6).
Another characteristic feature of stress-induced consequences is the decline in thymocyte content [42], also registered in this work. The loss of the thymocyte number was prevented by yolkin, and may be, in part, explained by its strong T cell tropic activities [30]. The serum level of corticosterone in stressed mice, most probably responsible for the loss of thymocytes, was not higher in the stressed mice. However, it was determined upon elicitation of the CS reaction and not immediately following stress, which is a limitation of this study.
Prolonged, psychic stress suppresses immune response via TLR4 signaling [11]. It is likely that yolkin, as a TLR4 agonist, may down regulate TLR4 expression, similar to LF being a TLR4 agonist [43], which was shown to ameliorate psychic stress-induced suppression of the delayed type hypersensitivity [10, 24].
The histological analyses revealed significant differences in the number and area of pustules in the auricles between the experimental groups. Other morphometric studies (not shown) did not reveal significant changes in the content of mast cells, CD3 cells and CD207 Langerhans cells in the auricles of the studied experimental groups. Apparently, the differences between the number of effectual inflammatory cell content (neutrophils), as well as their infiltration range, regarded only the inflamed auricles. In mice treated with yolkin, the decreased pustule area and neutrophil content were probably associated with the already described suppressive action of yolkin on CS [30]. In the stressed mice, the increased number of pustules may result from the corticosterone action promoting myelopoiesis and subsequent translocation of neutrophils from the circulation to the inflamed auricles. This is analogous to the enhancing effect of stress on neutrophil infiltration to wounds [20]. A similar phenomenon was reported in lungs of stressed mice, additionally treated with LPS [44]. We are aware of a discrepancy between the effect of the stress on the histological picture of the investigated auricles and their thickness measured with the caliper. Although this discrepancy is difficult to explain, all remaining ex vivo parameters reveal suppressed immune reactivity of the stressed mice. On the other hand, in the stressed mice treated with yolkin, preventative mechanisms could occur, possibly associated with the downregulation of TLR expression and the below described effects of yolkin on the prostanoid metabolism. The histological parameters correlate with serum corticosterone levels. For example, high corticosterone levels in mice, treated only with yolkin, corresponded to decreases in the thickness of native and fixed auricles. In addition, low corticosterone levels in the mice subjected to stress and yolkin were correlated with a low pustule number and area as well as a low number of infiltrating neutrophils. In conclusion, treatment of the stressed mice with yolkin leads to resolution of the histopathological changes in the auricles.
The ability of yolkin to strongly induce cyclooxygenase 1 (COX-1) production in mouse splenocyte cultures (manuscript submitted) may also play a role in ameliorating the consequences of acute restraint stress. The significance of a protective role of COX-1 derived prostaglandin E2 (PGE2) was established in the models demonstrating deleterious effects of restraint stress on endothelial function [45] and gastric mucosa [46, 47].
The available data on biological activities of yolkin in in vivo and in vitro models indicate that the protective action of yolkin is not restricted to only one type of stressful conditions. Yolkin lowered anxiety of tested rats in behavioral tests [48] and oxidative stress induced by LPS in mouse macrophage cultures [29]. Our present study (to be published) also indicates amelioration of mouse endotoxemia symptoms by yolkin.
A very recent study [28] provided more data on composition, glycosylation, thermal stability and storage conditions of yolkin preparations. The authors concluded that 4-week storage in a lyophilized form at temperature range from –20 °C to 25 °C does not affect biological properties of yolkin. Nevertheless, a long term storage at 4 °C was recommended for its application as a potential nutraceutical. Potential benefits of yolkin as a neutraceutical for human health was also anticipated in another study [29], in particular for patients with cognitive deficits.
In summary, our findings on the amelioration of consequences of the psychic stress on the antigen specific and nonspecific cellular immune response contribute to the already established beneficial actions of yolkin on the central nervous system, such as enhancement of pro-cognitive functions in old rats. Therefore, yolkin preparations may have potential in the prevention and treatment of neurological disorders and stressful conditions that adversely affect the function of the immune system.
Con A, concanavalin A; COX-1, cyclooxygenase 1; CS, contact sensitivity; IFN
Data are available from the corresponding author on request.
MZ designed the experiments and wrote the manuscript; JA performed the experiments, statistics and final correction; MK performed the experiments, figures, statistics; EZ performed the experiments; KK-K performed the histological analysis and its description; JPM performed the histological analysis; PK performed the histological analysis; AZ isolated and characterized the yolkin; ŁB isolated the yolkin. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and have agreed to be accountable for all aspects of the work.
The Local Ethics Committee of the Hirszfeld Institute of Immunology and Experimental Therapy in Wrocław, Poland gave a consent # 039/2022 to perform the experiments. The principle of 3Rs (replacement, reduction and refinement) was included in the experiment.
The authors thank Elżbieta Krasowska for her excellent technical assistance.
This research was funded in whole by National Science Centre, Poland, no. DEC-2021/41/B/NZ6/01167.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.









