1 Clinical Center for Biotherapy, Central Laboratory, Zhongshan Hospital (Xiamen), Fudan University, 361015 Xiamen, Fujian, China
2 Department of Cardiology, The Second Clinical Medical College, Jinan University (Shenzhen People's Hospital), 518020 Shenzhen, Guangdong, China
3 Integrated Chinese and Western Medicine Postdoctoral Research Station, Jinan University, 510632 Guangzhou, Guangdong, China
4 Xiamen Clinical Research Center for Cancer Therapy, 361015 Xiamen, Fujian, China
5 College of Pharmacy, Shenzhen Technology University, 518118 Shenzhen, Guangdong, China
Abstract
Background: The number of older people in the world is increasing year by year; studies have shown that more than 90% of cardiovascular disease occurs in the older people population, indicating that aging is one of the major risks involved in the development of cardiovascular disease. Therefore, retarding the development of cardiac aging is an important strategy to prevent aging-related cardiovascular diseases. Methods: In the current study, we examined the anti-cardiovascular aging potential of canthaxanthin in vitro and in vivo experiments. For this, a model of cardiomyocyte senescence induced by D-galactose was established, which was used to investigate the canthaxanthin’s effect on cardiac premature aging. Results: We found that canthaxanthin obviously mitigated the cardiomyocyte senescence in vitro. Further mechanistic studies revealed that canthaxanthin seems to alleviate cardiomyocyte senescence by regulating the autophagy process. Furthermore, the effects of canthaxanthin on cardiovascular senescence were further evaluated. We also observed that canthaxanthin mitigated cardiac aging and fibrosis in the aged mice model. Conclusions: To sum up, the current work showed that canthaxanthin could obviously alleviate cardiac premature aging, indicating that canthaxanthin could be used as a biologically active molecule for the treatment of cardiac aging and fibrosis.
Keywords
- cardiovascular aging
- canthaxanthin
- fibrosis
- p16
- autophagy
At present, the proportion of older people in the world is increasing year by year, and the proportion of the older people population will increase from 12% in 2015 to 22% in 2050 [1]. Therefore, the aging society will face huge economic, social, and health challenges in the next few decades. Studies have found that more than 90% of cardiovascular disease occurs in the older people population [2], indicating that aging is one of the major risks involved in the development of cardiovascular disease. Aging is a complex physiological process [3]. Cardiac aging refers to age-dependent degenerative changes, which in turn make the heart more sensitive to stress damage [4]. The risk of age-related cardiovascular disease in older people increases significantly year by year. Therefore, delaying the occurrence and retarding the development of cardiac aging is an important strategy in the prevention of aging-related cardiovascular diseases.
Canthaxanthin (ß, ß-carotene-4, 4
Currently, the effect of canthaxanthin on cardiovascular premature aging has not been studied. To this end, in the present study, we established a model of cardiomyocyte senescence induced by D-galactose, which was used to further explore the effect of canthaxanthin on oxidative stress-induced aging of cardiomyoblasts and arterial endothelial cells. The experimental observations revealed that CX might mitigate cardiomyoblast senescence and cardiac premature aging. Further mechanistic studies indicated that canthaxanthin might alleviate cardiomyoblast senescence by regulating the autophagy process. To sum up, this work showed that canthaxanthin obviously mitigates cardiomyoblast senescence and cardiac premature aging, suggesting that canthaxanthin has the potential application value for treating premature aging of the heart.
Bicinchoninic Acid Assay (BCA) protein concentration assay kit, Reactive oxygen species detection kits,
RIPA lysate, and
H9c2 (rat embryonic cardiomyoblast-derived cell line) was purchased from the
ATCC. HCAES (Human Primary Coronary Artery Endothelial Cells, Cat.no. PCS-100-020) was from ATCC. H9c2 and
HCAES cells were cultured in the DMEM medium supplemented with 10% FBS, 100 U/mL
penicillin, and 100 µg/mL streptomycin at 37 °C and 5% CO
After the cell culture medium was discarded, paraformaldehyde (4%) was added to
the cells, and cells were fixed for 0.5 h. After washing, freshly prepared
Sa-
Cells were processed using cell lysate; the cells were then centrifuged to harvest at 12,000 g for 30 min at 4 °C. After centrifugation, the supernatant was collected. A BCA kit was used to determine protein concentration. Proteins were separated by SDS-PAGE and transferred to PVDF membranes. The PVDF membrane was placed in a 5% BSA blocking solution for 2 h at room temperature. PVDF membranes were then placed into the diluted primary antibody solution for 12 h at 4 °C. After washing, PVDF membranes were placed in the secondary antibody solution for 120 min at room temperature (RT). After three washes in TBST (10 min each time), the blots were detected by the enhanced chemiluminescence (ECL) kit. The protein band images were analyzed using image J software (developed by National Institutes of Health, Bethesda, MD, USA).
H9c2/HCAES cells were seeded into 96-well plates at a density of 5000 cells per well and cultured for 24 h, after which the cells were cultured with fresh medium containing D-gal at different concentrations (5, 10, 20, 40, 60 g/L), the cells were cultured for 48 h. After washing, 10 µL of CCK-8 solution was added into each well and incubated for 1.5 h. The absorbance of cell samples was detected using a microplate reader at a wavelength of 450 nm.
The cells were washed twice with PBS, and 0.25% trypsin was added to digest the cells. When the cells were obviously round, and then the cell suspension was collected. The cells were treated with 5 µL of Annexin V-FITC (for 10 min) and 5 µL of PI solution (for 10 min), respectively. The cell samples were subsequently detected using a Flow cytometer (BD). Data were analyzed using FlowJ software (developed by Flow Cytometry Software Inc, version: 10.8.1, Sunnyvale, CA, USA).
The cells were digested with trypsin. The cells were then centrifuged at 1000 rpm for 5 min. Cells were washed three times with PBS, 1 mL of complete medium was added, then JC-1 staining solution was added and incubated in the dark for 20 min. The JC-1 staining solution was discarded. After 2 washes with pre-cooled PBS buffer, cells were detected via a Flow cytometer.
The medium was discarded, and the cells were washed once with sterile PBS. ROS fluorescent probe (DCFH-DA, 1:1000 dilution) was added. After incubation for 20 min, the medium was discarded, and the cells were washed three times with a serum-free medium. The cells were checked under a fluorescence microscope within 30 min.
The reactive oxygen species detection kit (Solarbio, Beijing, China) was used to detect the ROS level. The cells were seeded in 6-well plates. When the cells reached 70–80% confluence, the cells were treated with CX. After washing, the cells have been digested. The cells were collected by centrifugation and rinsed once with a serum-free medium. DCFH-DA was then added and incubated for 20 min. After washing three times with PBS, the cell samples were analyzed by Flow cytometry.
Malondialdehyde content was determined using an MDA detection kit. After canthaxanthin administration, the cells were digested and prepared into a single-cell suspension. The cells were then centrifuged for 5 min. The cell samples were centrifuged at 8000 g/min for 10 min. Cell supernatants were assayed by a microplate reader.
Experimental animals (C57 mice, weight 28–30 g, 18-month-old) were purchased from Huafu Kang Biotechnology Co (Beijing, China). We treated mice with canthaxanthin (15 mg/kg) by gavage for 6 weeks, and the control group was treated with normal saline by gavage. All animal experiments were approved by the animal ethics committee of Jinan University (approval number: 20200326).
Paraffin-embedded myocardial tissue was sectioned into a thickness of approximately 4 µm. After the sections were deparaffinized, they were placed in xylene I (20 min), xylene II (20 min), absolute ethanol I (5 min), absolute ethanol II (5 min), and 75% alcohol (5 min), then sections were washed and stained with Hematoxylin and Eosin. After sealing with neutral gum, samples were observed using a microscope. The images were analyzed by ImageJ software.
Masson staining was performed to analyze the degree of tissue fibrosis. Cardiac tissue was dehydrated with different concentrations of ethanol and then embedded in paraffin. Subsequent processing of tissue samples was performed according to Masson staining kit instructions.
After H9c2 cells were treated with canthaxanthin, PBS was added to rinse the cell slides 3 times (3 min each). Cells were fixed using pre-chilled acetone for 15 min. After the samples were washed 3 times, 0.5% Triton X-100 was added to the cells. After incubation for 20 min, cell samples were washed three times. After blocking with BSA, the cells were placed in the primary antibody solutions for 12 h. After 3 washes with PBS, fluorescently labeled secondary antibody was added and incubated at 37 °C for 1 h. After the cell samples were washed three times with PBS, DAPI staining solution was added and incubated in the dark for 10 min. After washing three times with PBS, the samples were treated with 50% glycerol and observed under a confocal microscope (Olympus, FV3000, Tokyo, Japan).
The data were expressed as mean
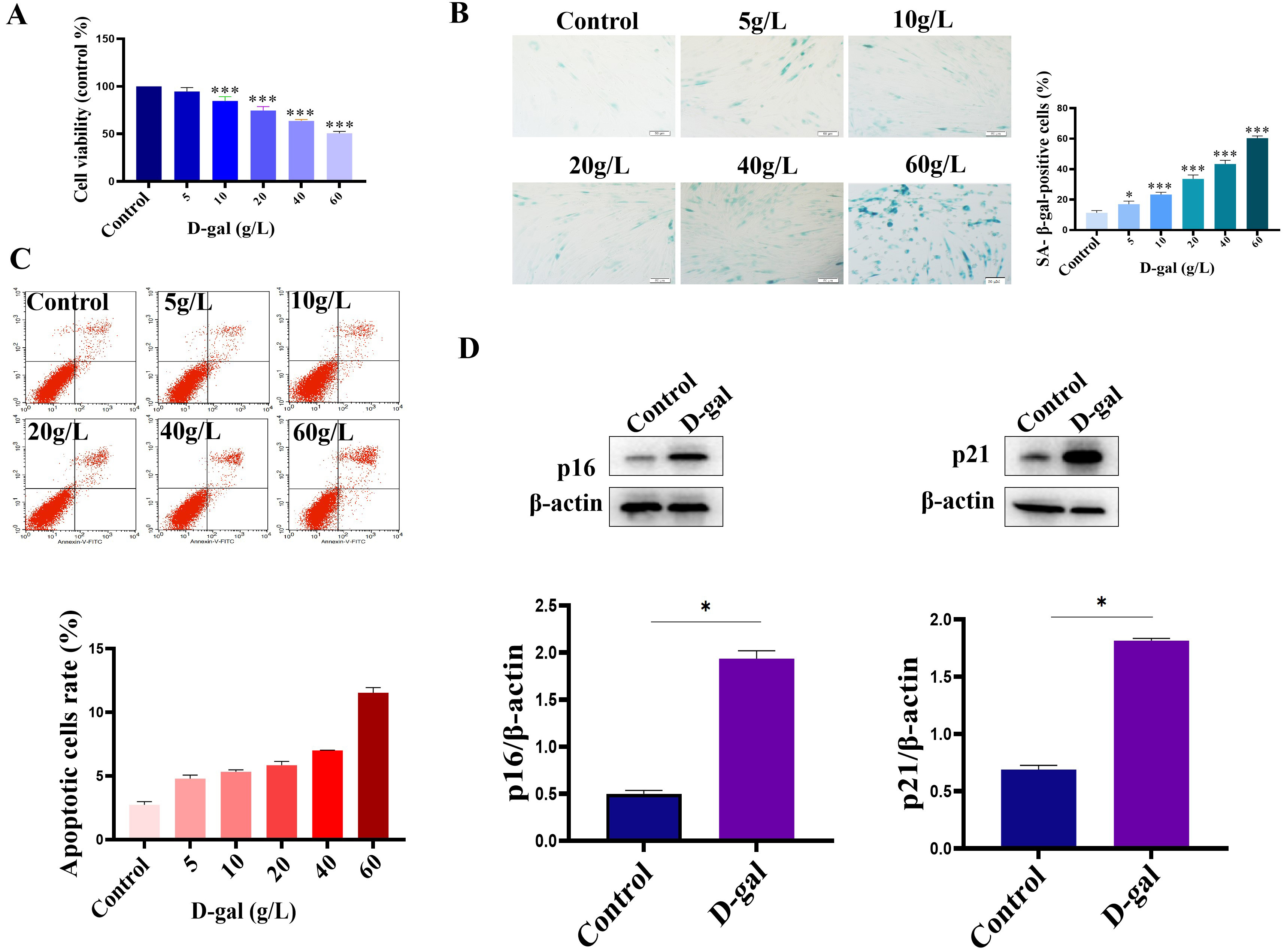
D-gal was used to establish a model of cardiomyoblast senescence. For this, we first analyzed the effect of D-gal on the viability of H9c2 cells. In the per-experiment, the cells were cultured for 48 h in a complete medium containing 5 g/L, 10 g/L, 20 g/L,40 g/L, and 60 g/L D-gal, respectively. The results showed that the cell viability of H9c2 cells decreased after treatment with different concentrations of D-gal (Fig. 1A). Furthermore, we analyzed the effect of different concentrations of D-gal on the H9c2 senescence. As shown in Fig. 1B, compared with the control group, the different concentrations of D-gal induced H9c2 cell senescence. The number of H9c2 senescence was increased in a dose-dependent manner. When the concentration of D-gal was at 60 g/L, the cell morphology changed significantly, and some cells died (Fig. 1B). These findings suggest that D-gal could induce the H9c2 cells senescence in a dose-dependent manner. Furthermore, Flow cytometry was conducted to test the apoptosis of H9c2 cells after treatment with different concentrations of D-gal. As shown in Fig. 1C, the H9c2 cell apoptosis rate was significantly increased when the concentration of D-gal reached 60 g/L. In addition, the expressions of p16 and p21 (the markers of cellular senescence) were also increased (Fig. 1D). Taken together, in the current study, D-gal at a concentration of 40 g/L was chosen to establish the H9c2 senescence model.
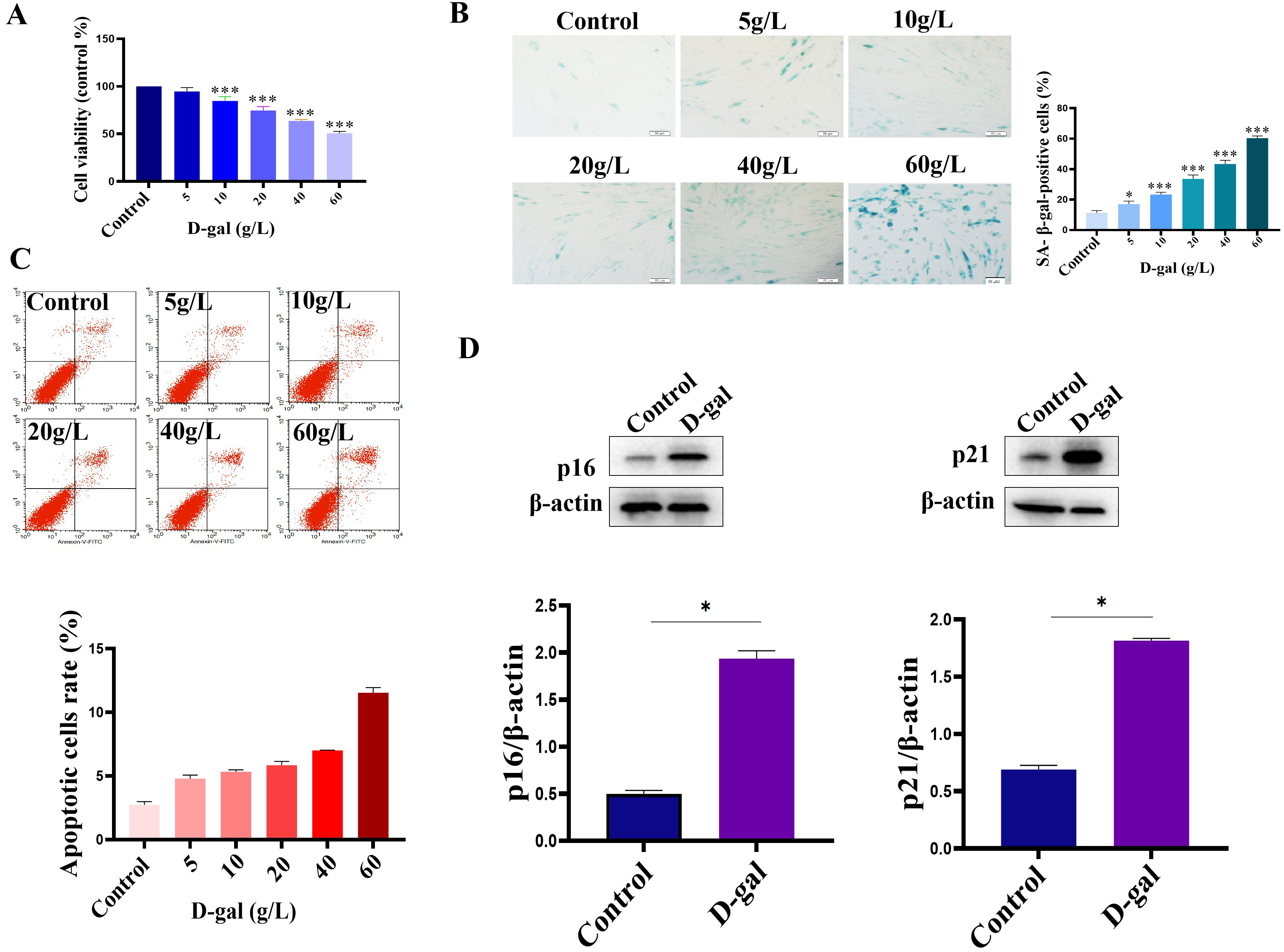 Fig. 1.
Fig. 1.Establishment of H9c2 senescent cell model. (A) The cell
viability of H9c2 cells was decreased after D-gal treatment. (B) D-gal induced
the senescence of H9c2 cells. (C) Analysis of cell apoptosis by Flow analysis.
Cells were treated with different concentrations of D-gal, and then the cell
apoptosis was analyzed using an Annexin V-PE/7-AAD double staining kit according
to the manufacturer’s instructions. (D) The expression of p16 and p21 was
up-regulated in the D-gal treatment group. n = 3 from 3 independent experiments.
*p
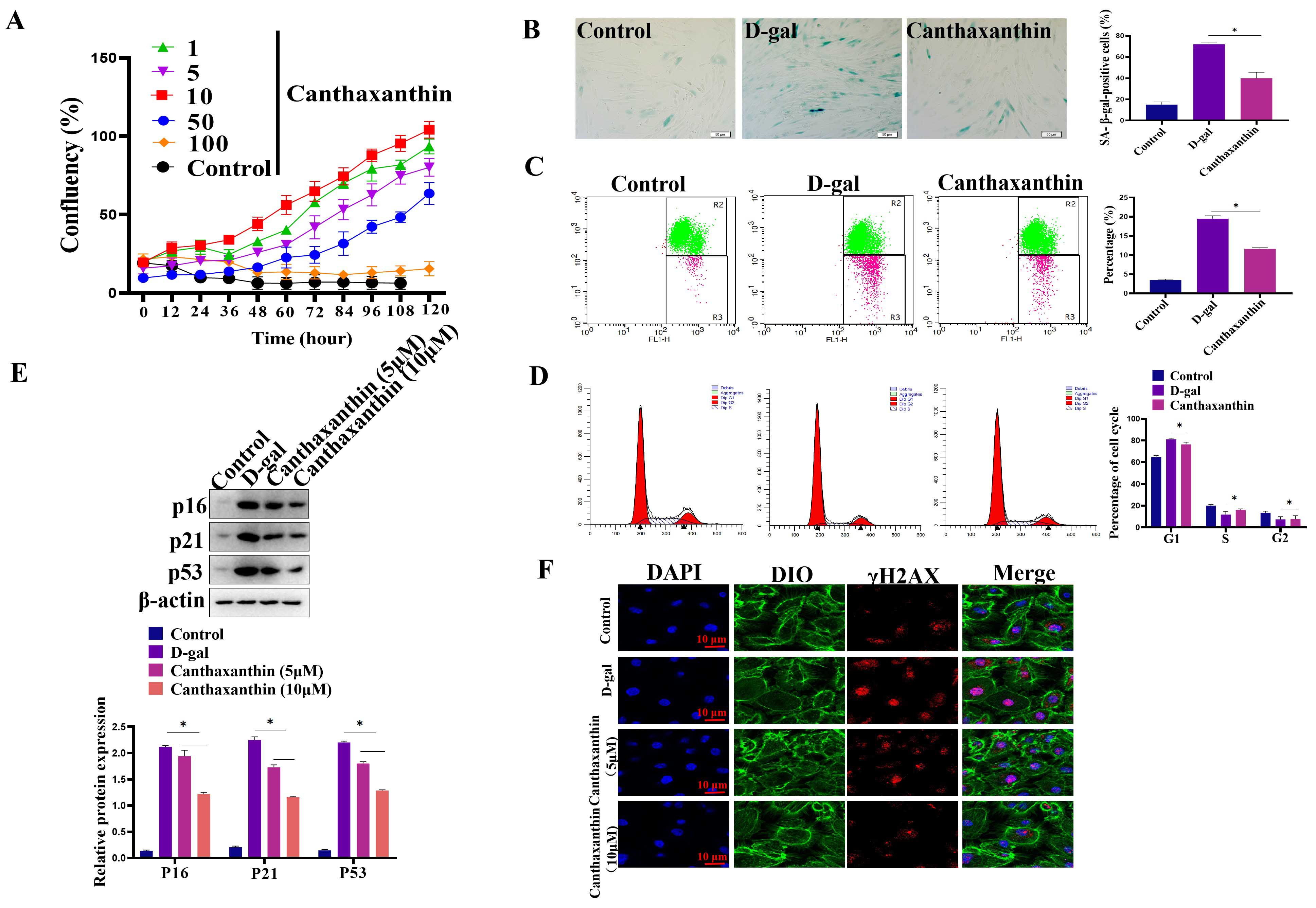
In order to study the effect of canthaxanthin on H9c2 senescence, we first
determined the concentration of canthaxanthin used in the current work by CCK8
assays. The results showed that 10 µM canthaxanthin showed a good effect on
promoting cell proliferation (Fig. 2A). Therefore, we used 10 µM
canthaxanthin for the following experiments. To analyze the effect of
canthaxanthin on the senescence of H9c2 cells, SA-
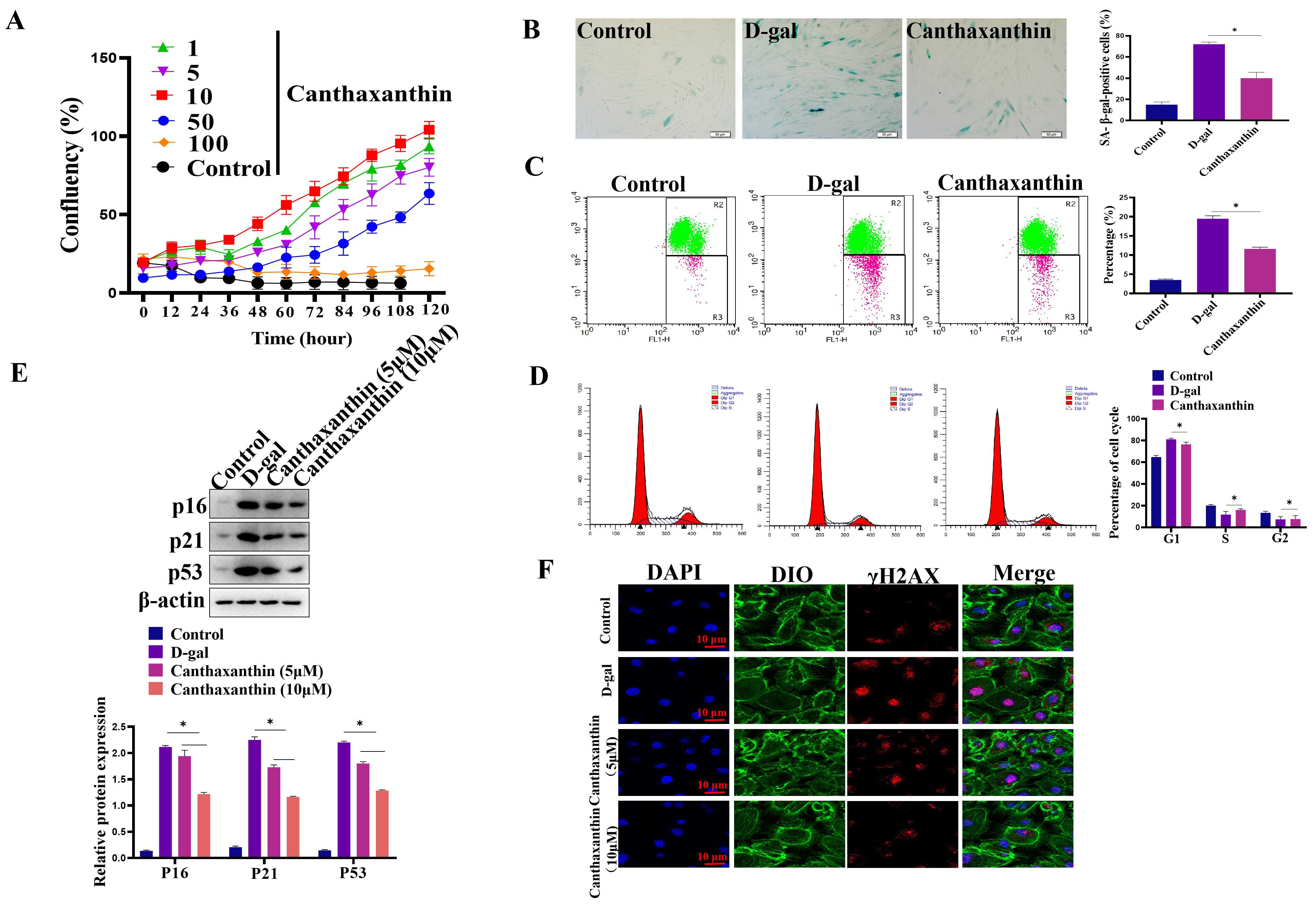 Fig. 2.
Fig. 2.Canthaxanthin alleviated the H9c2 senescence. (A) Effects of
different concentrations of canthaxanthin on the proliferation of H9c2 cells. (B)
The effect of canthaxanthin on the senescence of H9c2 cells by Sa-
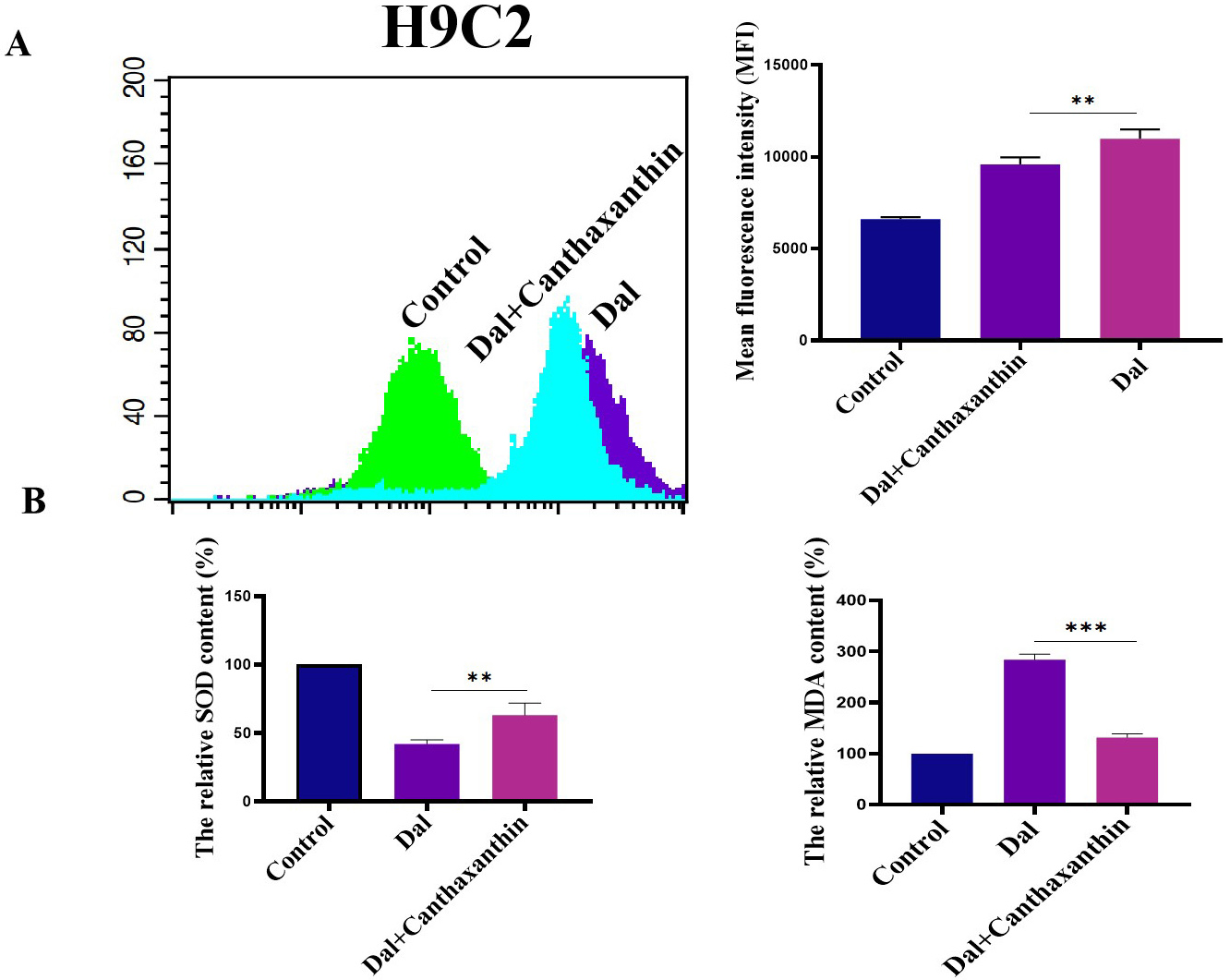
We investigated the effect of canthaxanthin on D-gal-induced oxidative stress in H9c2 cardiomyoblasts. Flow cytometry analyses were performed to detect ROS content. As shown in Fig. 3A, the mean fluorescence intensity of the D-gal group was enhanced compared with the control group, indicating that the ROS content was significantly increased; compared with the D-gal group, the ROS level in H9c2 cardiomyoblasts was significantly decreased after 48 h of canthaxanthin treatment. Furthermore, the effect of canthaxanthin on the levels of Malondialdehyde (MDA) and Superoxide Dismutase (SOD) was also detected. As shown in Fig. 3B, SOD in the D-gal treatment group was significantly decreased, but MDA content was significantly increased. Compared with the D-gal group, the SOD in the canthaxanthin treatment group was increased, and the MDA content was obviously decreased. These results suggest that canthaxanthin could alleviate the oxidative stress of the senescent cardiomyocytes.
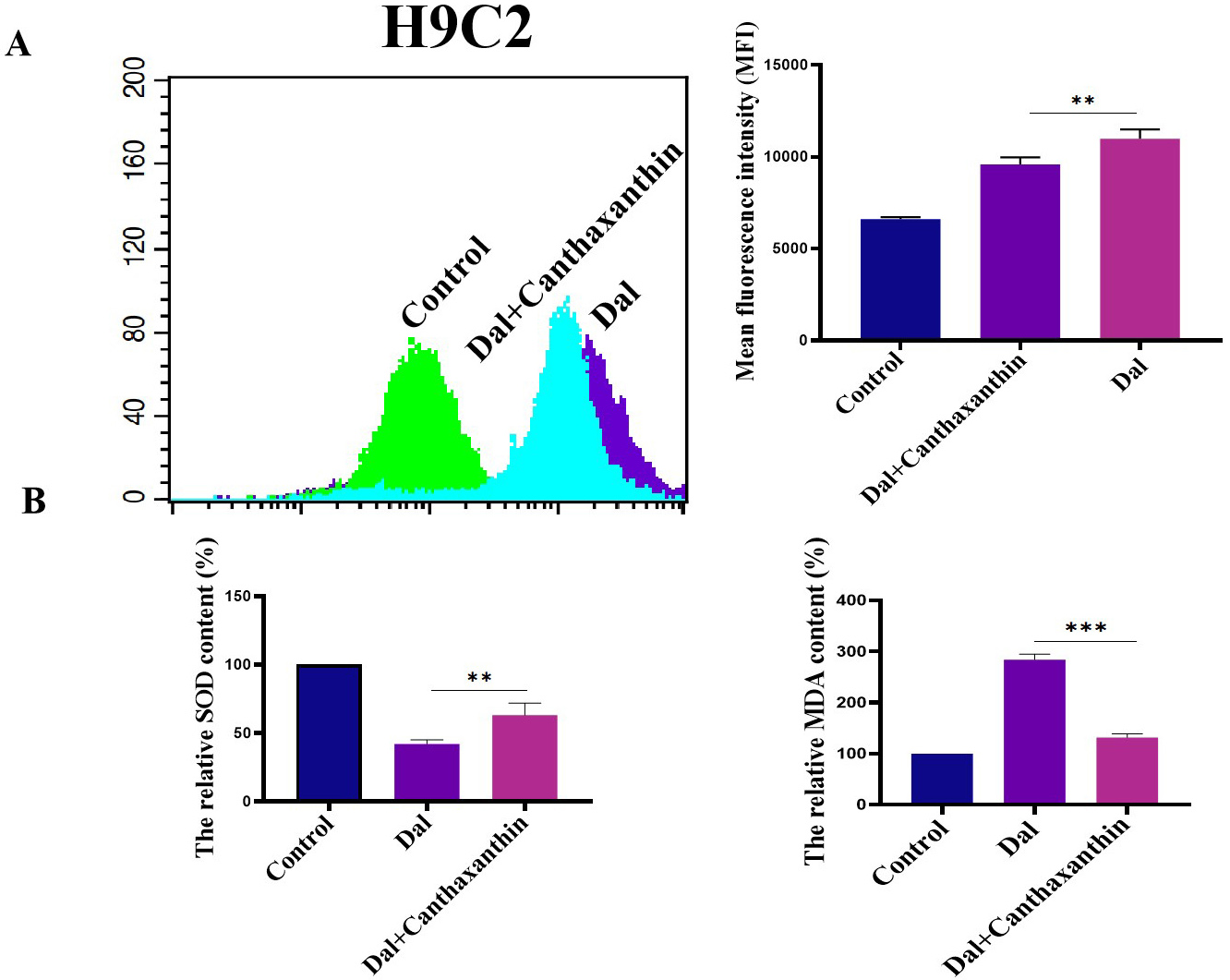 Fig. 3.
Fig. 3.Effect of canthaxanthin on oxidative stress. (A) The reactive oxygen species (ROS) content was significantly decreased by canthaxanthin treatment. (B) The effect of
canthaxanthin on the levels of Malondialdehyde (MDA) and Superoxide Dismutase
(SOD). n = 3 from 3 independent experiments. **p
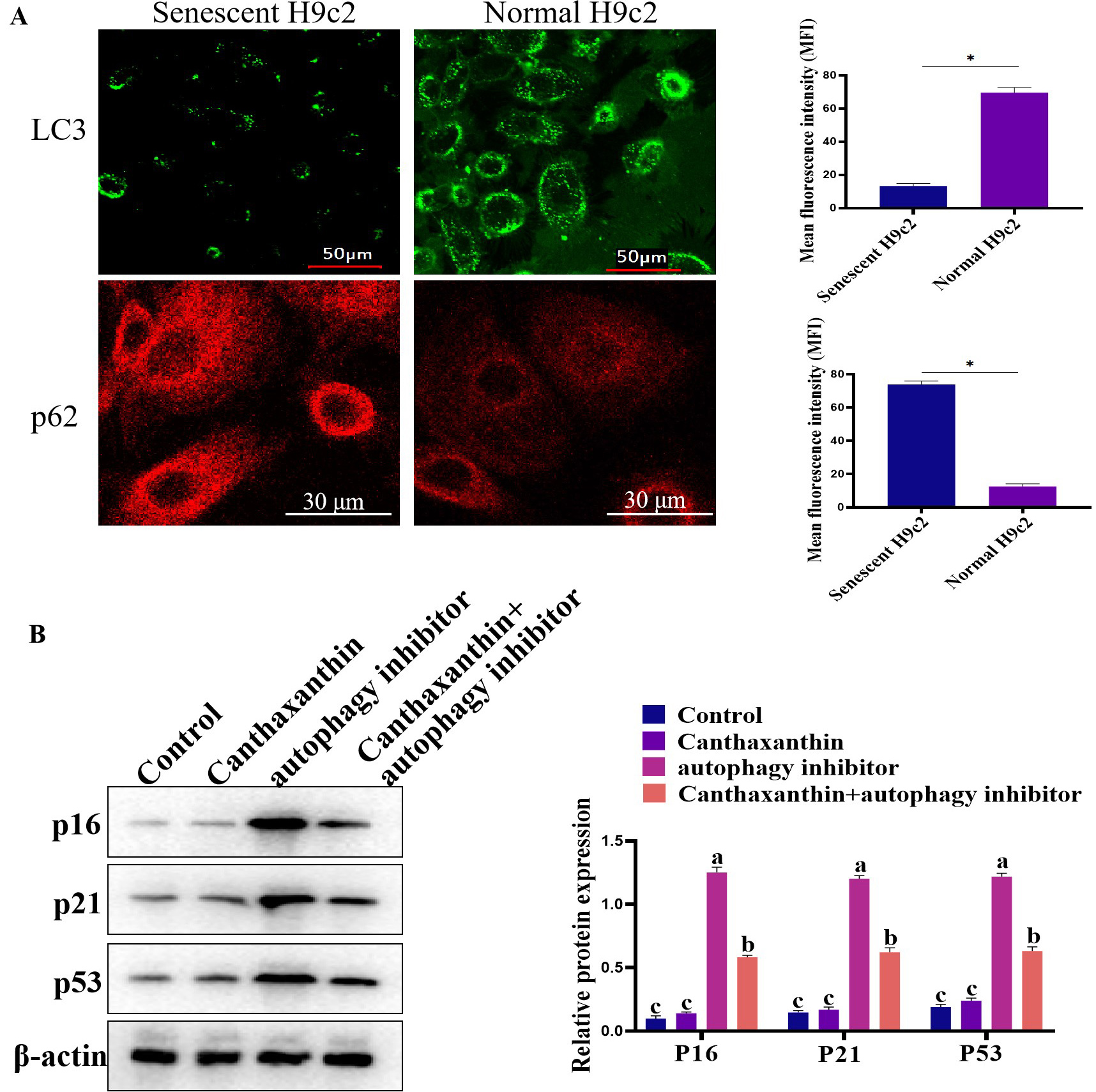
Oxidative stress is closely related to aging, and it has been reported that canthaxanthin has antioxidant effects. Here, we further analyzed whether canthaxanthin could alleviate H9c2 cell senescence through other molecular mechanisms. Previous studies have shown that cardiac aging is closely related to autophagy [13]. In the senescent cells, autophagy levels were significantly reduced [14]. In the current study, we found that in the senescent H2c9 cells, LC3 expression was significantly decreased and p62 expression was increased (Fig. 4A). In contrast, the level of autophagy was significantly increased in the canthaxanthin-treated group by detecting LC3 (Supplementary Fig. 1). These results suggest that canthaxanthin mitigates cardiac premature aging via regulating autophagy. In order to further verify whether canthaxanthin inhibits myocardial aging by regulating autophagy, we conducted a rescue experiment; canthaxanthin was added into the cell medium, at the same time, an inhibitor of autophagy (chloroquine diphosphate, ab142116, 50 µM) was also added. The experimental results showed that the autophagy inhibitor neutralized the anti-aging effects of canthaxanthin (Fig. 4B), which suggests that the anti-aging effect of canthaxanthin may be through the regulation of autophagy (at least in part).
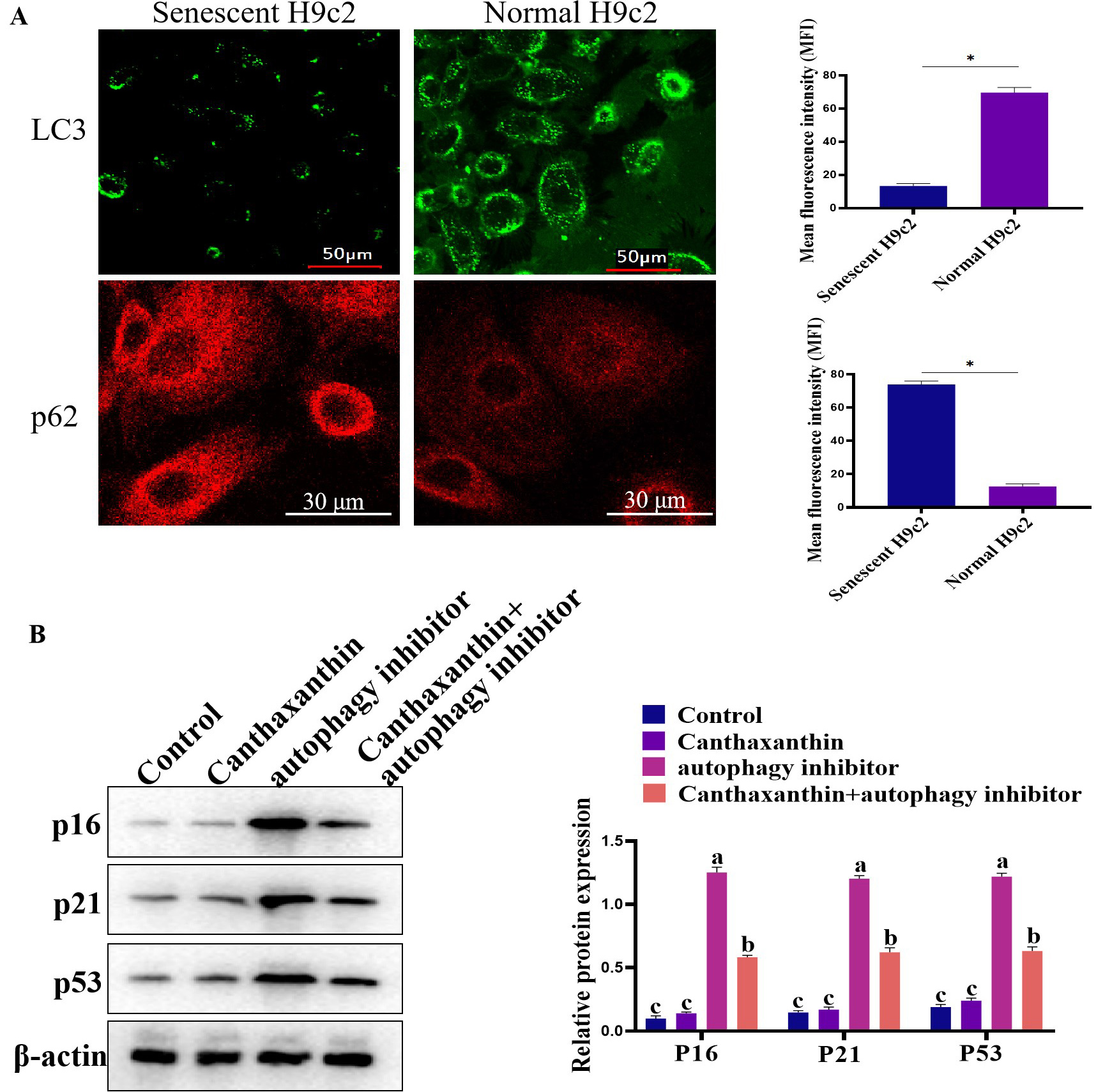 Fig. 4.
Fig. 4.Exploring the molecular mechanism of cantaxanthin in alleviating
cellular aging. (A) Autophagy levels were elevated by canthaxanthin treatment.
(B) Canthaxanthin exhibited an anti-aging effect via regulating autophagy. n = 3
from 3 independent experiments. *p
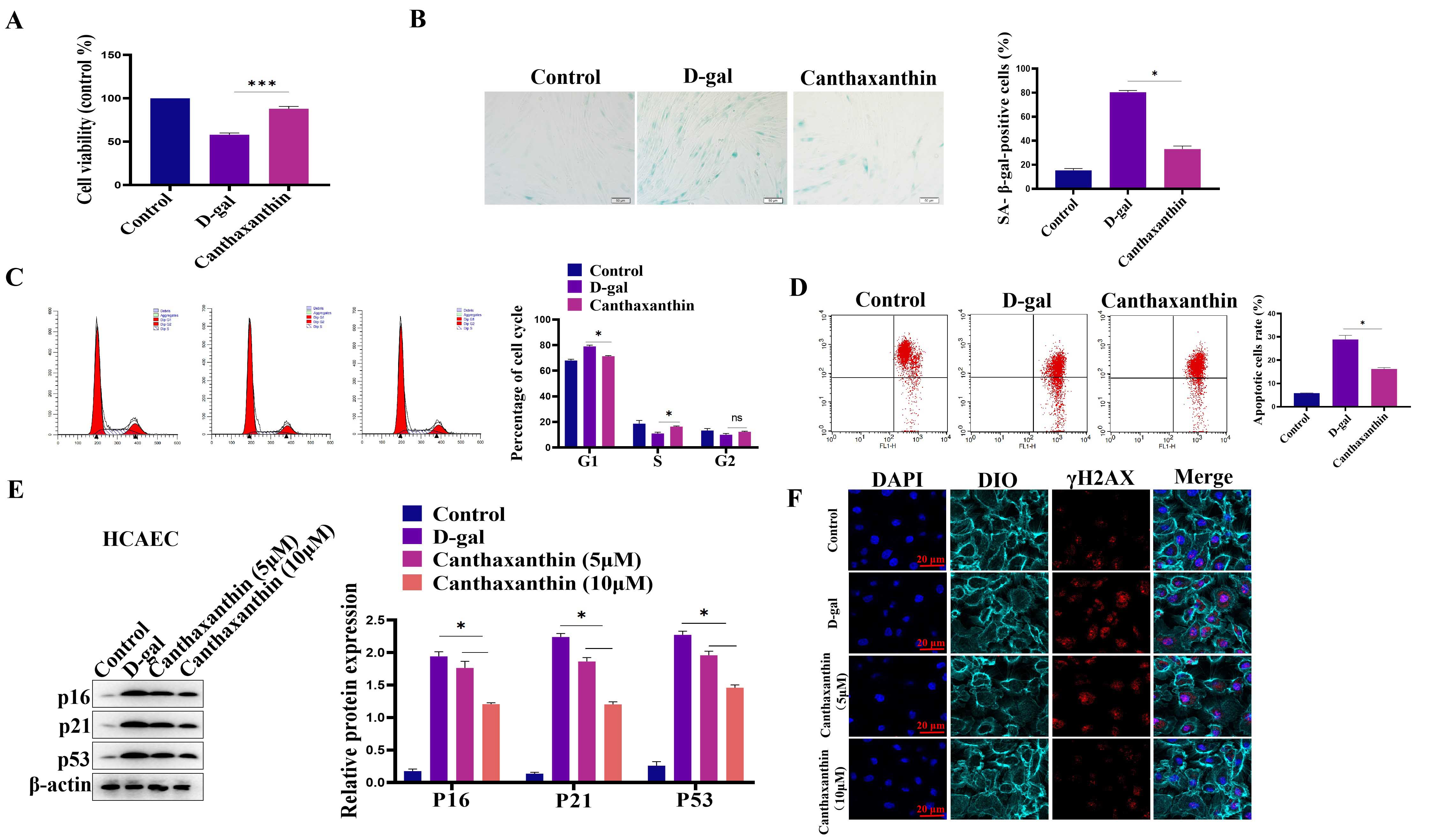
We also established the model of D-gal-induced HCAEC senescence. We first
analyzed the effect of D-gal on HCAEC viability. Different concentrations of
D-gal (5–80 g/L) were used in the pre-experiment, and results showed that 30 g/L
D-gal could successfully induce HCAEC senescence. Fig. 5A showed that D-gal
treatment decreased the viability of HCAEC compared with the control group. In
contrast, canthaxanthin treatment could significantly increase the viability of
HCAEC. Furthermore, D-gal treatment led to a significantly higher number of
senescent cells compared to the control group. However, canthaxanthin
significantly reduced the senescence of HCAEC cells (Fig. 5B). Furthermore, cell
cycle experiments showed that canthaxanthin significantly increased the
proportion of cells in S phase (Fig. 5C). JC-1 analysis shown that canthaxanthin
could increase the mitochondrial membrane potential (Fig. 5D). In addition, the
effect of canthaxanthin on the expression of the senescence-related markers was
also tested, and results were that D-gal treatment increased the protein
expressions of p16, p21 and p53 in HCAEC cells.
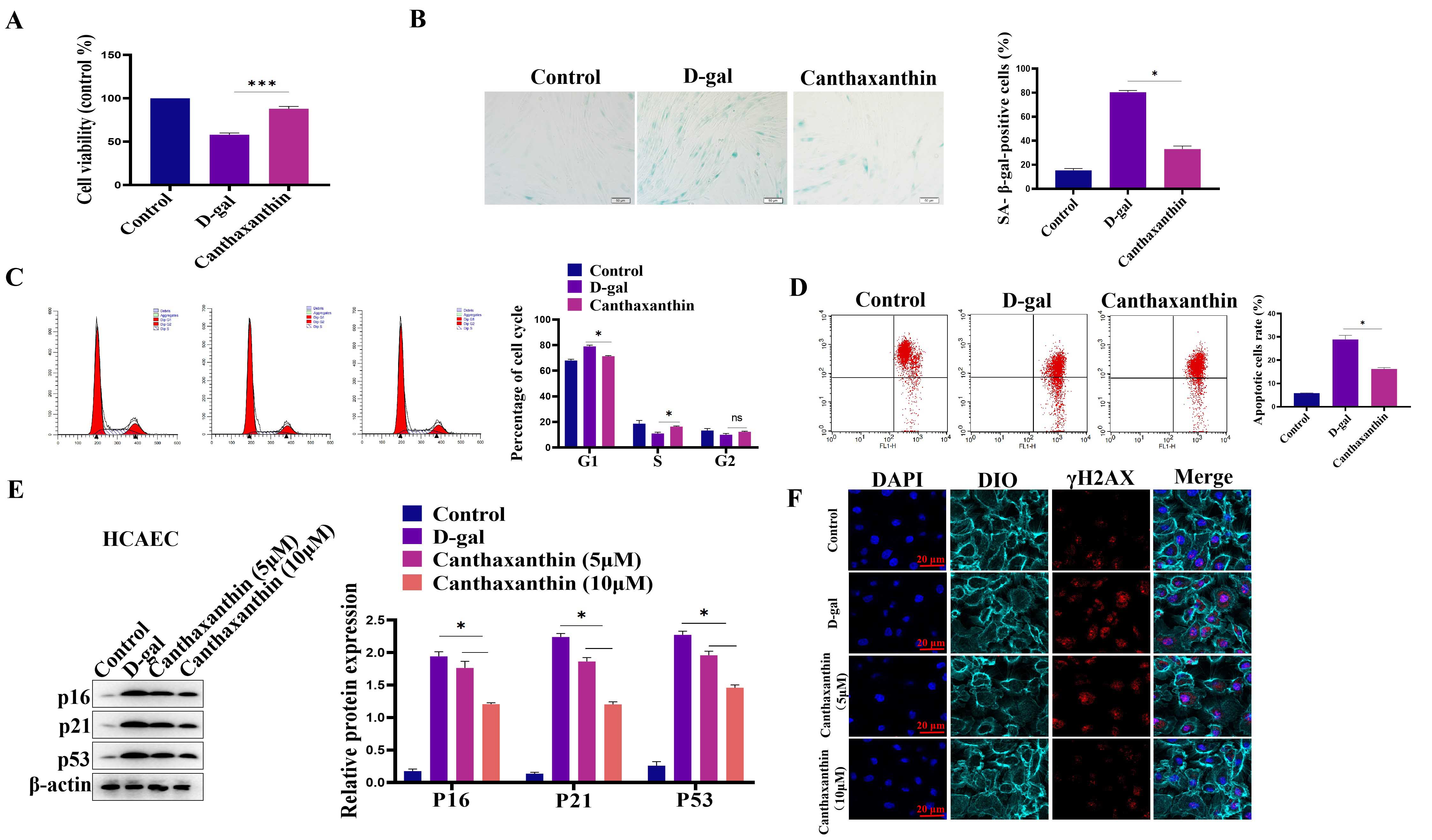 Fig. 5.
Fig. 5.Canthaxanthin alleviated the human primary coronary artery endothelial cells (HCAEC) senescence. (A) D-gal
treatment decreased the viability of HCAEC cells compared with the control group.
(B) Canthaxanthin reduced the senescence of HCAEC cells. (C) Canthaxanthin
increased the proportion of cells in S phase. (D) Canthaxanthin could increase
the mitochondrial membrane potential of the senescent cells. (E) Canthaxanthin
treatment down-regulated the expression of p53, p21 and p16. (F)
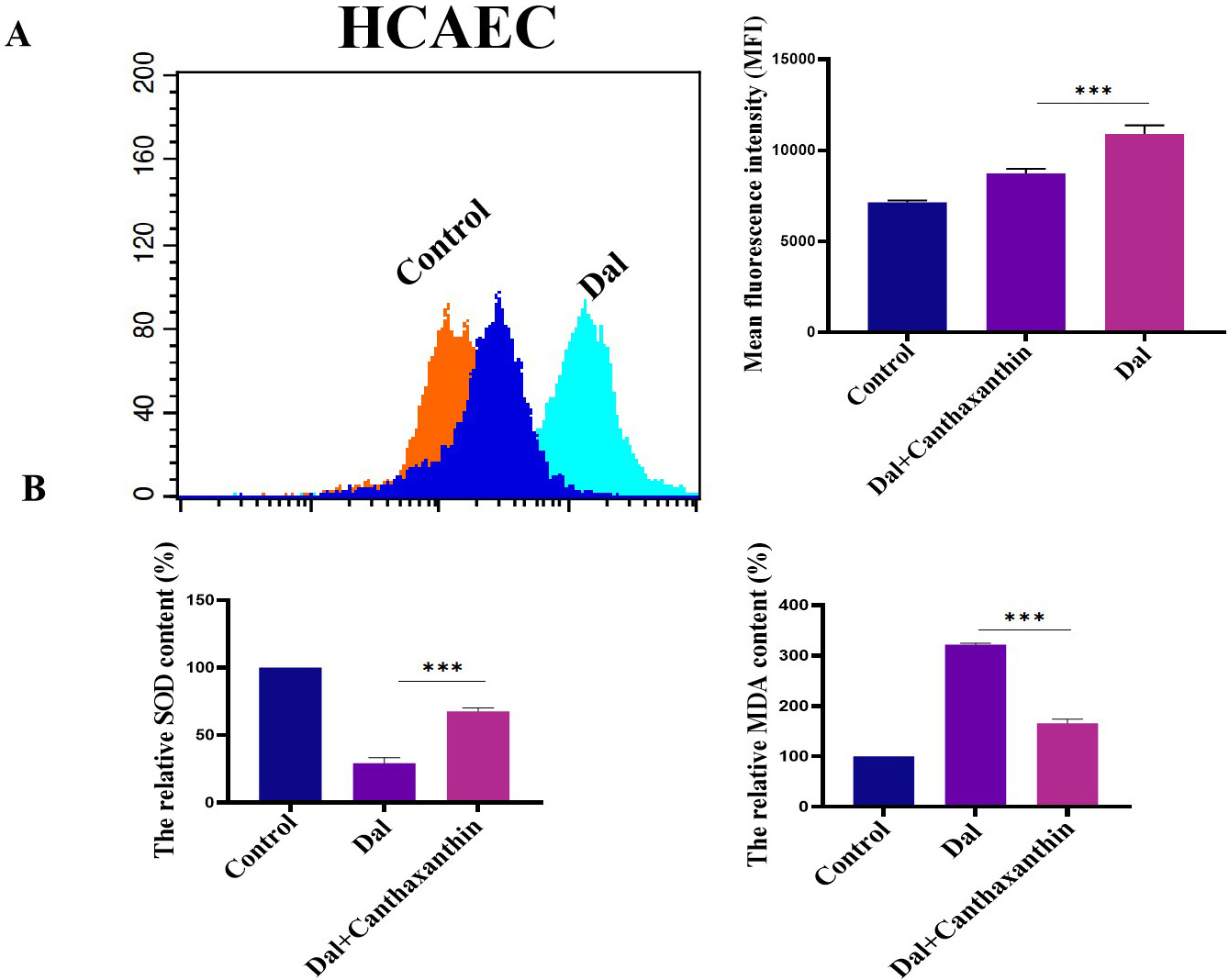
We investigated the effect of canthaxanthin on D-gal-induced oxidative stress in HCAEC. Flow cytometry was used to analyze the ROS content. The results are shown in Fig. 6A; the mean fluorescence intensity of the D-gal treatment group was obviously enhanced. Compared to the D-gal group, the level of ROS was significantly decreased after the H9c2 cells were treated with canthaxanthin for 48 h. Furthermore, the effect of canthaxanthin on the expression levels of MDA and SOD in the HCAEC cells was also analyzed, and the results showed that the SOD in the D-gal group was significantly lower than that of the control group, but the MDA content was significantly higher. Compared to the D-gal group, the SOD in the canthaxanthin treatment group was increased, and the MDA content was decreased (Fig. 6B). These findings indicated that canthaxanthin can relieve the oxidative stress of HCAEC cells.
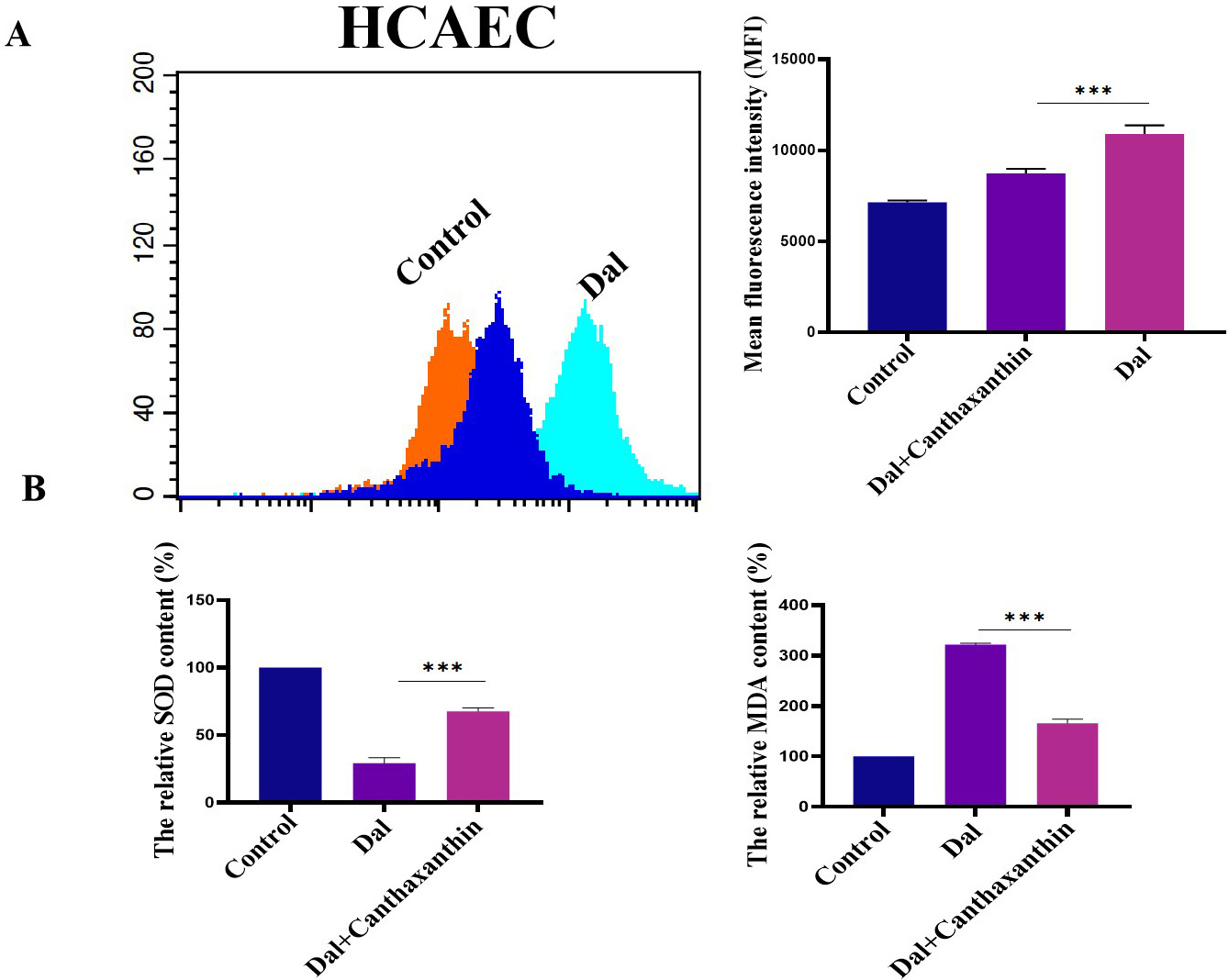 Fig. 6.
Fig. 6.Effects of canthaxanthin on oxidative stress in HCAEC. (A) The
level of ROS in .cells was significantly decreased after canthaxanthin treatment.
(B) The SOD in the canthaxanthin treatment group was increased, and the MDA
content was decreased. n = 3 from 3 independent experiments. ***p
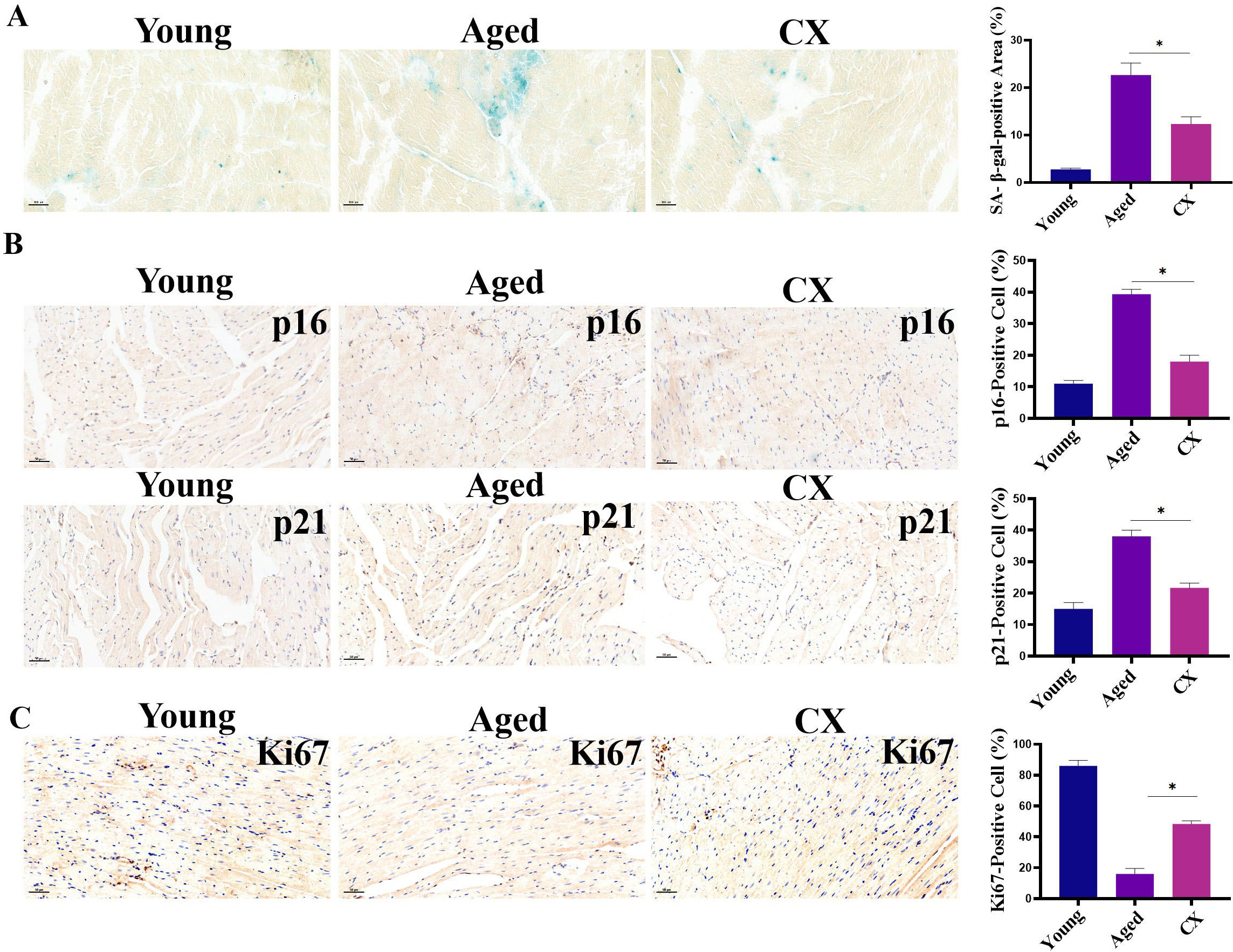
In in vitro experiments, we evaluated the effect of canthaxanthin on
heart aging. For an in vivo experiment, we selected 18-month-old mice as
research subjects. We treated the experimental mice with canthaxanthin (15 mg/kg)
by gavage and the control mice with normal saline by gavage. Heart tissues were
extracted for analysis of aging markers. Canthaxanthin treatment reduced the
SA-
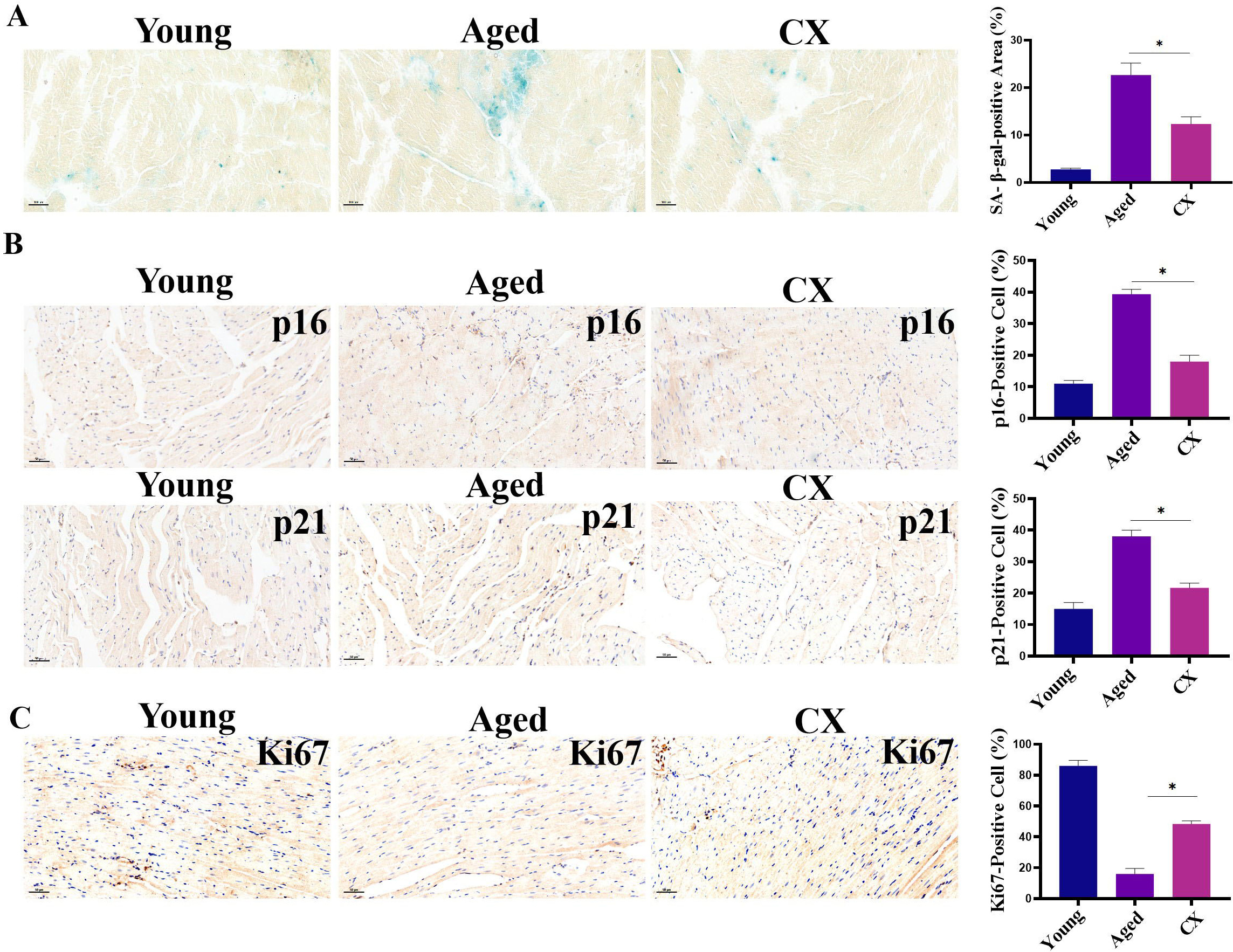 Fig. 7.
Fig. 7.Evaluation of anti-aging effects of canthaxanthin in
vivo. (A) Canthaxanthin reduced the SA-
Heart aging is closely related to fibrosis; therefore, we also analyzed the
effect of CX on myocardial fibrosis in vivo. Masson staining showed that
the level of myocardial fibrosis was significantly lower in the CX-treated group
(Fig. 8A). Furthermore, CX also inhibited the expression of Col-1 in the cardiac
tissue (Fig. 8B), and the expression of
 Fig. 8.
Fig. 8.The effect of CX on myocardial fibrosis in vivo. (A)
The level of myocardial fibrosis was down-regulated in the canthaxanthin-treated
group. (B) Canthaxanthin inhibited the expression of Col-1 in cardiac tissue. (C)
The expression of
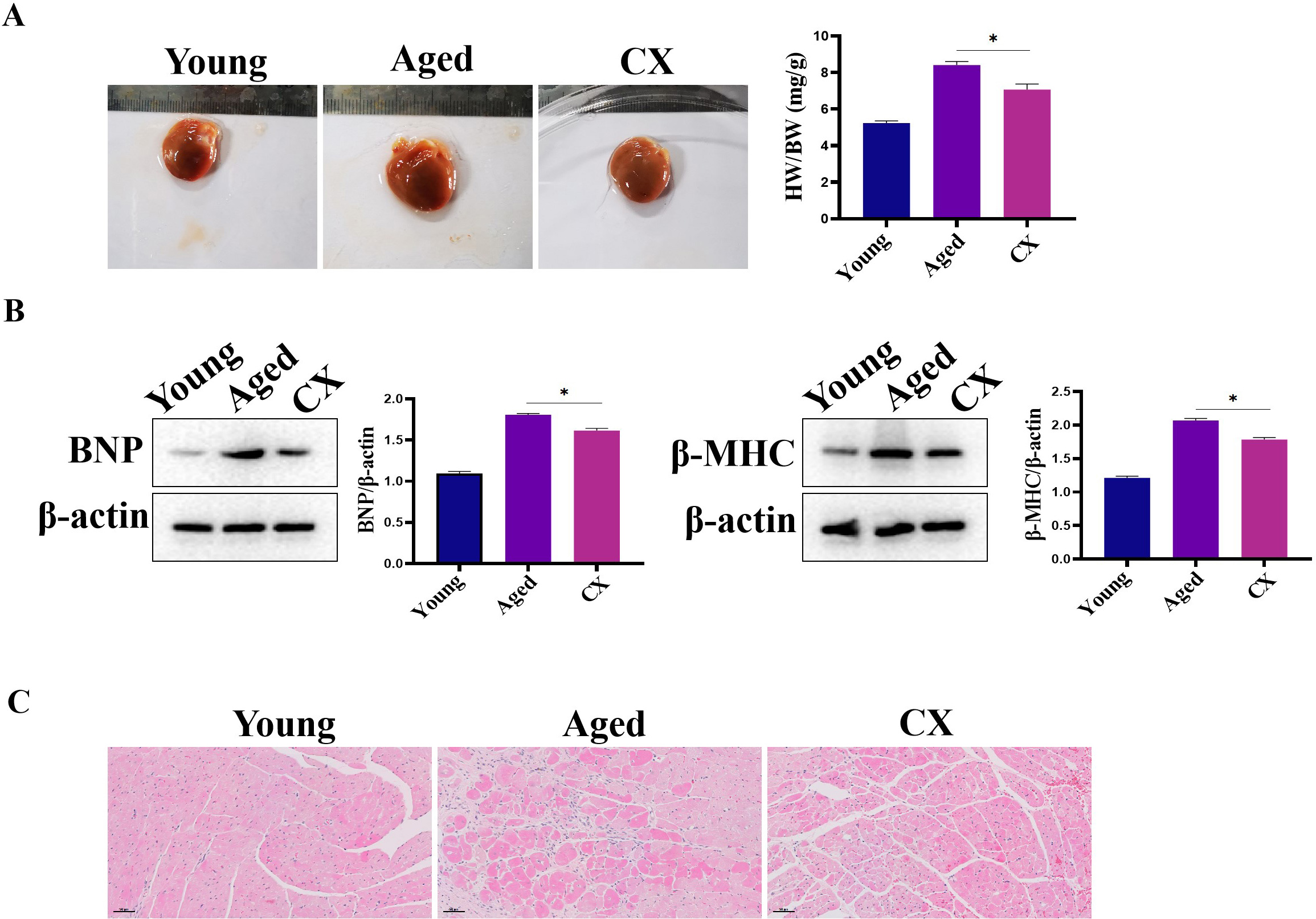
Compared with the hearts of young mice, the heart/body-weight ratio (HW/BW) of
the 18-month-old mice was abnormal; the ratio was significantly higher in the
aged heart (Fig. 9A), suggesting that the myocardium may be hypertrophic. The
expression of brain natriuretic peptide (BNP) and
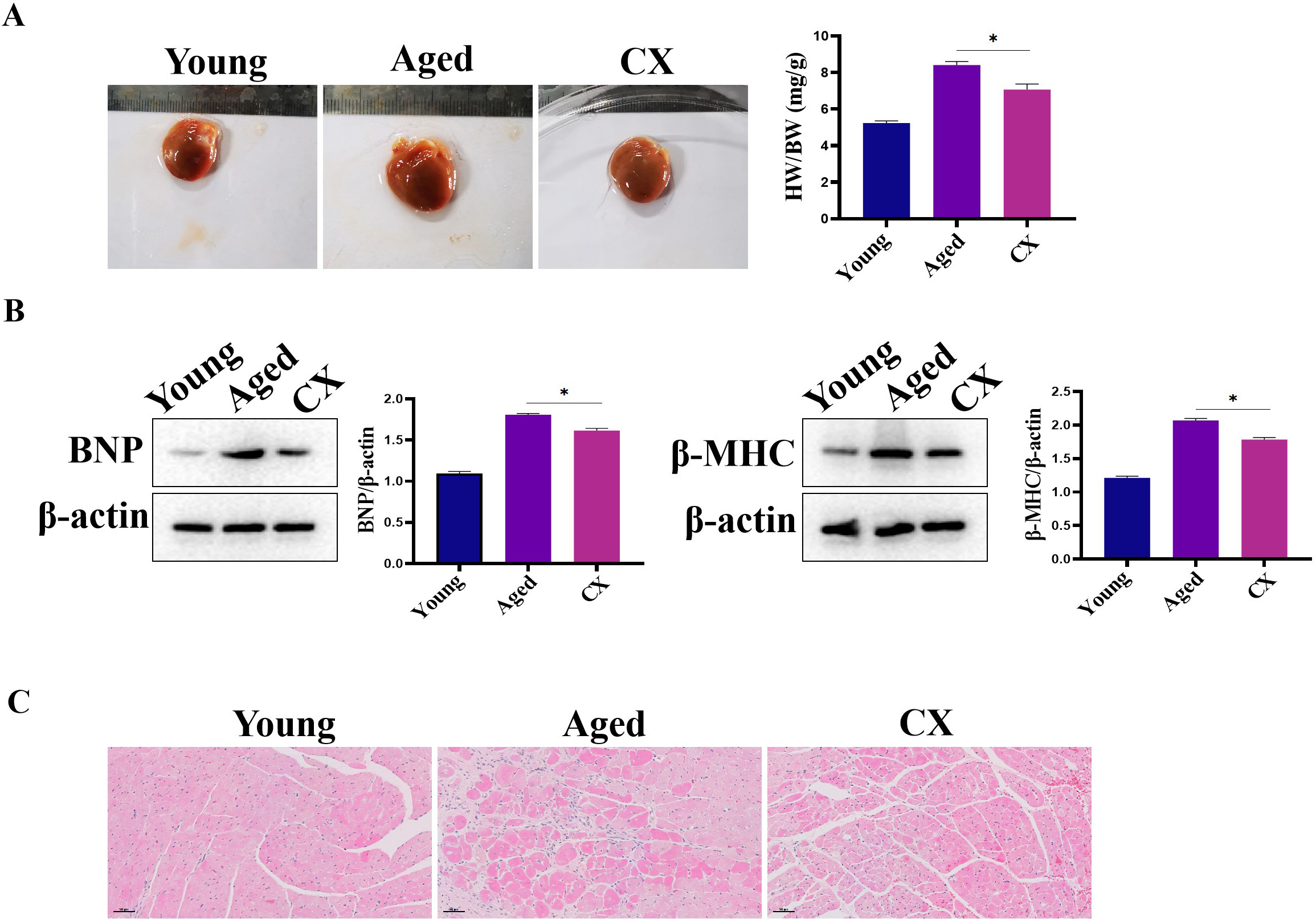 Fig. 9.
Fig. 9.The effect of CX on heart in vivo. (A) The ratio the
HW/BW was down-regulated by Canthaxanthin treatment. (B) The expressions of BNP
and MHC were down-regulated after canthaxanthin treatment. (C) H&E analysis of
the effect of canthaxanthin on myocardial tissue. *p
Population aging has become one of the major social problems in the world [15]. This brings great challenges to limited social medical resources [16]. The aging population has brought unprecedented challenges to medical and health services [17]. Aging is a process of chronic accumulation of biological changes in the body over time. Because of the importance of the heart, cardiac aging is one of the important pathological factors in the aging of the body [18]. Symptoms of cardiac aging include left ventricular wall thickening, myocardial fibrosis, and decreased diastolic function [19]. The expression of aging-related markers p16 and p21 has been found to increase in aging cardiomyocytes of humans and animals. Cardiomyocyte aging is closely related to the occurrence of cardiovascular diseases such as heart failure, atherosclerosis, and arrhythmia [19]. Therefore, there is an urgent need for us to further elucidate the underlying molecular mechanisms of cardiac aging and to find effective treatments or interventions to delay or retard cardiac aging. In the present study, we found that CX has a promising anti-aging effect on the heart.
In the present study, H9c2 was used as a cell model to establish a senescent
cardiomyoblast model. In in vitro experiments, we found that CX could
alleviate the senescence of H9c2. The proportion of Sa-
Autophagy is closely related to cardiomyocyte aging, which is responsible for degrading the damaged organelles and macromolecules [24]. Autophagy can be activated under stress. However, studies have shown that autophagy gradually decreases with aging. Therefore, the reduction of autophagy is one of the important reasons for cardiac aging [25]. In the current study, we showed that CX can increase autophagy in the senescent cardiomyoblast, which may be one of the potential mechanisms by which CX exhibits an anti-aging effect.
The incidence of age-related cardiovascular diseases increases exponentially with the global aging trend [26, 27]. Studies have shown that vascular aging plays a key role in the occurrence and development of aging-related diseases [26, 27]. Senescent endothelial cells accumulate continuously in the vessel wall, leading to the decline of endothelial function [28]. Previous studies have shown that senescent endothelial cells accumulate in atherosclerotic plaques, leading to vascular dysfunction [29]. Therefore, endothelial cells can be used as a tool for evaluating vascular aging and function. In the present study, we found that CX could significantly alleviate the senescence of vascular endothelial cells. These results suggest that CX can relieve vascular endothelial cell senescence.
Cardiac aging is closely related to oxidative stress [30]. Oxidative stress occurs when ROS exceeds the buffering capacity of the antioxidant defense system. ROS-mediated oxidative damage plays an important role in vascular aging. In endothelial cells, nitric oxide (NO) is considered to be the main substance maintaining endothelial-cell function. In the present study, we found that CX can significantly increase vascular NO content.
CX has many advantages over the anti-aging substances that have been mentioned in previous reports. CX can be eaten as food or applied as a medication (because anti-aging is a long-term process). An interesting question is, how much CX does the body get from food every day? As of now, there is no data on how much CX can be obtained from food every day because inter-individual variability is very large. However, studies have shown that CX can be used in a very wide range of safe concentrations.
Of course, current research also has certain limitations. H9c2 cells are cardiomyoblast-derived immortal cells that are fundamentally different from terminally differentiated cardiomyocytes with respect to cell cycle progression. Since cell cycle arrest is a major determinant of cellular senescence, H9c2 cells are not the best model for cardiomyocyte senescence. Theoretically, primary cardiomyocytes should be the best cellular model, but under our experimental system, primary cardiomyocytes cannot meet the current experimental requirements. Therefore, we finally chose H9c2 as a model to evaluate the anti-aging potential of canthaxanthin.
In the current study, we showed that CX can alleviate cardiomyoblast senescence via regulating the autophagy process, indicating that canthaxanthin may be an important bioactive molecule that has shown potential for alleviating cardiac aging and fibrosis.
CX, Canthaxanthin; MHC,
The data used during the current study are available from the corresponding author on reasonable request.
ZW, KZ and SD designed the research; WL, MZ, JY performed the research; JF and KZ provided help and advice on the conclusions; MZ and JF analyzed the data; ZW wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All animal experiments were approved by the animal ethics committee of Jinan University (approval number: 20200326).
Not applicable.
This study was supported by the National Natural Science Foundation of China (Grant No. 82000254), the China Postdoctoral Science Foundation (Grant No. 2019TQ0121), the Natural Science Foundation of Top Talent of SZTU (Grant No. 20200215).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/j.fbl2902070.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.