1. Introduction
Cancer cells that develop as a result of chronic inflammation move to peripheral
tissues via blood vessels through angiogenesis. To metastasize to surrounding
tissues, cancer cells need to degrade the extracellular matrix. In particular,
the gelatinases such as matrix metalloproteinase (MMP)-2 and MMP-9 among MMPs
degrade collagen 4, the main component of the basement membrane [1], involved in
angiogenesis and metastasis. Therefore, it is crucial to regulate the expression
of MMPs to limit the metastatic ability of cancer cells. Therefore, treatment of
tumor cells with a substance that inhibits the expression of those inflammatory
cytokines involved in MMPs regulation is also expected to limit metastasis
formation.
While screening medicinal plants for anti-metastasis research, it was found that
the inhibitory effect of milk thistle (Cirsium japonicum) extract was
excellent. The silibinin employed in this study corresponds to 50–70% of the
three isomers of silymarin which makes up roughly 2% of milk thistle’s active
component [2]. It has been known to have a role in the anti-tumor drug
cisplatin’s hepatoprotection, antioxidation, anti-angiogenesis, inhibition of
inflammation, and nephrotoxicity [3]. The action mechanism of silibinin on
metastasis remains unclear. The great efficacy of silibinin to target cancer
cells’ migratory and invasive features as well as their capacity to metastasize
to distant organs has also been demonstrated in recent pre-clinical trials.
According to thorough mechanistic investigations, silibinin targets signaling
molecules that control the epithelial-to-mesenchymal transition (EMT), activation
of proteases, adhesion, motility, and invasiveness as well as the components of
the supporting tumor microenvironment, preventing metastasis [4]. Therefore, we
tried to investigate whether silibinin could inhibit cell invasion and MMPs in
the model of human fibrosarcoma cells (HT1080 cell line) widely used for the
study of metastasis. Moreover, the expression of proteins such as MAPKs and
IL-1 related to invasion and metastasis was examined to clarify the
mechanism by which silibinin inhibits metastasis.
2. Materials and Methods
2.1 Materials
Dulbecco’s Modified Eagle’s Medium (DMEM), fetal bovine serum (FBS),
Trypsin-EDTA, and antibiotics such as penicillin (10,000 U/mL)/streptomycin
(10,000 g/mL)/amphotericin (2500 g/mL) reagents for cell culture
were obtained from Life Technologies (Paisley, Scotland). MTT reagent, gelatin,
agarose, RIPA lysis buffer and other reagents are obtained from Sigma Chemical
Co., Ltd. (St. Louis, MO, USA). Silibinin samples were purchased from Sigma
Chemical Co. (St. Louis, MO, USA).
2.2 Cell Line and Culture
HT1080 cell line (ATCC No.CCL-12, Homo sapiens, fibroblast, lung) and IMR90 cell
line (ATCC No.CCL-186, Homo sapiens, fibroblast) purchased from ATCC (American
Type Culture Collection) were cultured using DMEM containing 10% of FBS and
subcultured with trypsin-EDTA. Antibiotics such as
penicillin/amphotericin/streptomycin were used to prevent cell culture from
bacterial contamination. Mycoplasma testing was performed to authenticate the
cell lines used in this study. This was accomplished using the
MycoAlert™ Mycoplasma Detection Kit (Lonza, Bend, OR, USA), which detects
enzymatic activity associated with viable mycoplasma in cell cultures. Briefly,
cells were harvested and lysed, and the resulting lysate was incubated with the
MycoAlert™ substrate for 10 minutes at 37 °C. The
fluorescence of each sample was then measured to check mycoplasma
contamination. The silibinin was freshly dissolved in dimethyl sulfoxide (DMSO) as
a solvent before use. Silibinin at 2.5, 5, 10, 15, 20, and 25 M was used
in all experiments. All doses, including blank and controls, were adjusted at
0.1% DMSO, which is the concentration contained in the highest silibinin dose
used, and shown to have no cytotoxic effects.
2.3 2,2-Diphenyl-1-picrylhydrazyl (DPPH) Radical Scavenging Assay
Silibinin (500 L) at 2.5, 5, 10, 15, 20, and 25 M was reacted with
DPPH (2,2-diphenyl-1-picrylhydrazyl) solution at 0.15 M (500 L),
for 1 h at 25 ℃. Vitamin C at 0.01% was used as a positive control. The optical
density (OD) of the product was measured at 532 nm using a visible
spectrophotometer (SpectraMax M3, Molecular Devices). The amount of
DPPH-generated radicals was represented as % [(OD of silibinin treatment
group/OD of the blank group) 100]. Blank means the group containing
DPPH spontaneously generated radicals and DMSO without silibinin.
2.4 Reducing Power Assay
Different doses of silibinin (6 L) at 2.5, 5, 10, 15, 20, and 25
M, 1% potassium ferricyanide (200 L), distilled water (194
L), and 200 mM phosphate buffer (200 L) at pH6.6 were reacted in a
microtube. After incubation for 20 min at 50 ℃, trichloroacetic acid solution at
10% (200 L) was added to the reaction product. After centrifugation at
2000 g for 10 min, the supernatant (250 L) was mixed with distilled water
(250 L). Next, after ferric chloride (50 L) was added, the optical
density was measured at 700 nm using a UV spectrophotometer (SpectraMaxM3,
Molecular Devices, San Jose, CA, USA). The blank group contained ferricyanide, FeCl, and DMSO
without silibinin, and the positive control contained 0.001% of vitamin C. The
level of antioxidant activity as a reducing power was displayed as % [(OD of
silibinin treatment group/OD of the blank group) 100].
2.5 MTT Assay
The growth inhibitory effect of silibinin on HT1080 cells was evaluated using
3-(4,5-Dimethyl-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) [5]. The cells at a
density of 5 10 cells/well were inoculated into 96-well plates.
After treatment with silibinin at 2.5, 5, 10, 15, 20, and 25 M for 24 h,
20 L of MTT (5 mg/mL) were added to each well for 4 h. DMSO (150 L)
was added to solubilize the formazan salts and measured the OD at 570 nm using a
visible spectrophotometer (SpectraMax M3, Molecular Devices). Relative survival
of cells was represented as a % compared to the blank group [(OD of the
silibinin treatment group/OD of the blank group) 100].
2.6 Gelatin Zymography
The activities of MMP-2 and MMP-9 were examined using gelatin zymography
according to a previous study [6]. HT1080 cells were cultured in the presence of
silibinin at 2.5, 5, 10, 15, 20, and 25 M for 1 h. Then, phorbol myristate
acetate (PMA) at 1 ng/mL was added for 3 days to induce the expression of MMP.
The conditioned medium collected, cleared by centrifugation, and used for the
analysis of gelatin zymography. The bands representing MMPs activity were
detected as clear zones, and the degree of the bands’ intensity was measured with
Davinch-ChemiTM. The MMP enzyme activity was expressed as % in comparison to the
blank group [(Silibinin treatment group / blank group 100)].
2.7 Western Blot Analysis
Western blot analysis was carried out according to standard procedures. HT1080
cells were exposed to silibinin at 2.5, 5, 10, 15, 20, and 25 M for 1 h.
Then, they were stimulated using 1 ng/mL PMA for 24 h in the presence of
silibinin. Next, cell lysis was performed with RIPA lysis buffer. The proteins
from cell lysates were transferred from a 10% polyacrylamide gel to a
nitrocellulose membrane. Thereafter, the membrane was treated with 5% of skim
milk. Next, after the treatment of primary antibodies against the target protein
such as MMP-2, TIMP-1, p-JNK, ERK-1/2, IL-1 and
-actin, secondary antibody treatment was performed. Target proteins were
determined using a chemiluminescent ECL kit (Amersham Pharmacia Biotech,
Piscataway, NJ, USA). The degree of band intensity was analyzed with a LAS3000
® image analyzer (Fujifilm Life Science, Tokyo, Japan).
2.8 Immunofluorescence Staining Assay
HT1080 cells were cultured in a slide chamber at 37 °C for 24 hours.
After treatment with each concentration of silibinin, for 1 h, PMA at 1 ng/mL was
added and incubated for 24 h. After fixing with 10% formalin for 15 min, the
cells were permeabilized with phosphate-buffered saline (PBS) containing 0.5% of
Tween 20 (PBS T-20) for 30 min and washed 3 times with 0.1% PBS T-20. After
blocking with 5% of donkey normal serum, primary antibodies (MMP-2) were added
for 2 h. Then, after washing 3 times for 5 min each with 0.1% PBS T-20,
secondary antibodies (donkey anti-goat conjugated CY3, donkey anti-mouse
conjugated CY3, donkey anti-rabbit conjugated FITC) were added for 1 h. Then,
after washing them, the slides were exposed to DAPI reagent for nuclei staining
and observed with the iRiS Digital Cell Imaging System (Logos Biosystems,
Gyeonggi-do, Korea). All reagents were purchased from Sigma Chemical Co. (St.
Louis, MO, USA).
2.9 Cell Invasion Assay
The invasion of HT1080 cells was carried out in accordance with the
Chemicon® methodology. The invasion chamber from the Cell
Invasion Assay Kit (ECM550) consists of a 24-well tissue culture plate and 12
cell culture inserts containing polycarbonate membrane (8.0 m pore size),
over which a thin layer of ECMatrix solution is applied. HT1080 cells in 300
L of serum-free media were added to each insert and 500 L of media
containing 10% fetal bovine serum (chemoattractant) was added to the lower
chamber. After the cells adhered to insert, they were treated with each
concentration of silibinin and stimulated with 1 ng/mL PMA. Chambers were then
incubated in 5% CO and at 37 °C for 72 hours. Non-invading cells
as well as the ECM gel layer were removed using a cotton-tipped swab and washed
with PBS. On the other side of the filter, invasive cells on the lower surface of
the membrane were stained by dipping inserts in the staining solution for 20 min.
After washing them with PBS, the stained cells were dissolved in 10% acetic
acid. Then, the optical density was measured at 560 nm using a UV
spectrophotometer (SpectraMaxM3, Molecular Devices). Relative invasion of cells
was represented as a % compared to the blank group [(OD of the silibinin
treatment group/OD of the blank group) 100].
2.10 Statistical Analysis
Data were analyzed using ANOVA and post hoc (Duncan) test as means of values
SD from three independent experiments (*, p 0.05, **,
p 0.01 and ***, p 0.001).
3. Results
3.1 Antioxidant Effect of Silibinin
The antioxidant activity of silibinin was examined using the DPPH radical
scavenging assay and the reducing power assay. Vitamin C (Vit. C) at 100
g/mL, used as a positive control, decreased by 65% of the radicals
generated by DPPH, whereas silibinin showed no radical scavenging activity (Fig. 1A). The difference between blank group and silibinin treatment groups above 10
M was significantly observed (Fig. 1B). When looking at the reducing power
(Fig. 1B), vitamin C at 10 g/mL (the highest dose of 100 g/mL gave results out
of scale) showed a 300% increase over the blank, and silibinin at doses of 10 M
and higher showed a modest but significant activity, which reached a 49%
increase at 25 M. These results indicate that silibinin has no radical
scavenging activity, although being endowed with some reducing power. Therefore,
its efficacy as an antioxidant is quite low.
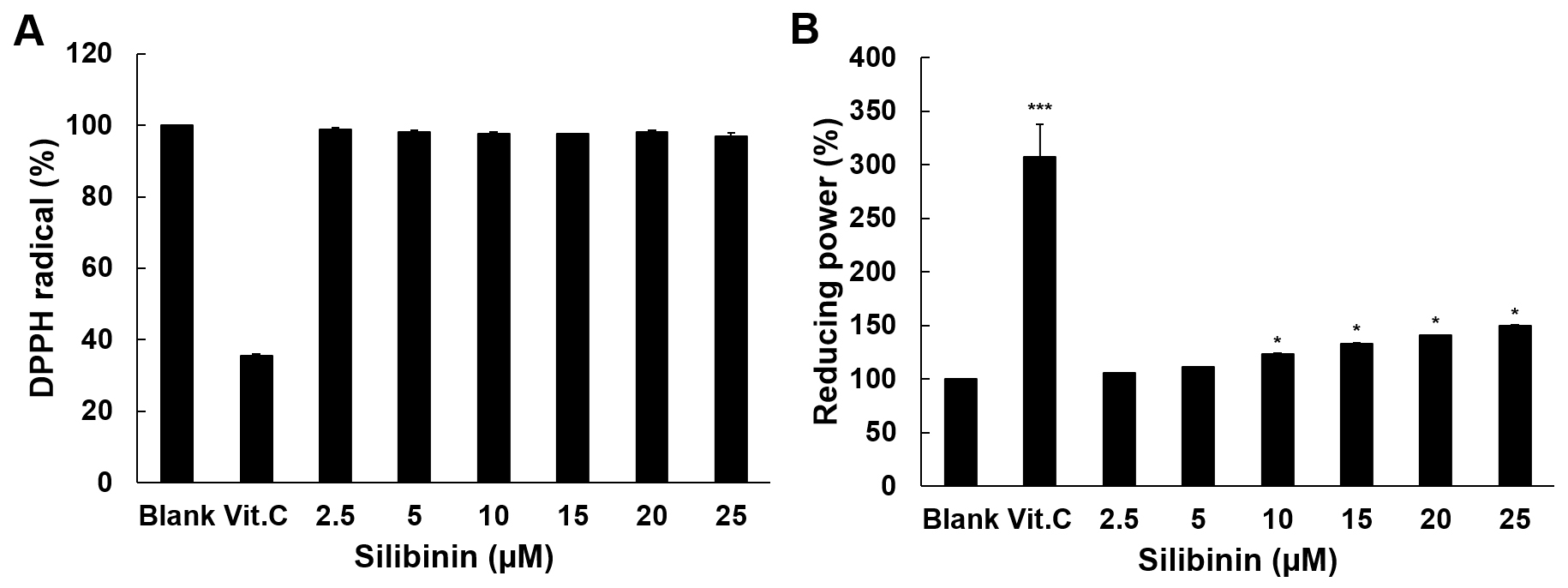 Fig. 1.
Fig. 1.
Antioxidant activity of silibinin. (A) The scavenging effect of
silibinin on DPPH radicals is shown in this experiment. Vitamin C was used as
positive control at 100 g/mL. (B) Reducing power of silibinin. Vitamin C
was used as a positive control at 10 g/mL. Data are shown as mean
SD from three independent experiments, all run in triplicates. The level of
significance between blank and silibinin treatment was evaluated statistically
(*, p 0.05; ***, p 0.001) using ANOVA and post hoc
(Duncan) test.
3.2 The Effect of Silibinin on Cell Viability in HT1080 Cells and
IMR-90 Cells
The effect of silibinin on cell viability was examined in HT1080 and IMR-90
cells. In HT1080, silibinin at low dose (2.5 M and 5 M) resulted in
a 37% and 38% increase in cell growth, respectively. On the contrary, at the
higher doses (20 M and 25 M), silibinin induced a 26% and 29%
reduction in cell viability, respectively (Fig. 2A). Silibinin effects on IMR-90
cells resulted in a milder growth inhibition (around 20%) at all doses, with no
evident dose-effect. Moreover, the growth inhibitory effect of silibinin above 20
M on HT1080 cells was significantly higher than that on IMR-90 cells,
indicating some specificity of the effect, likely due to a different growth
control in tumor versus normal cells.
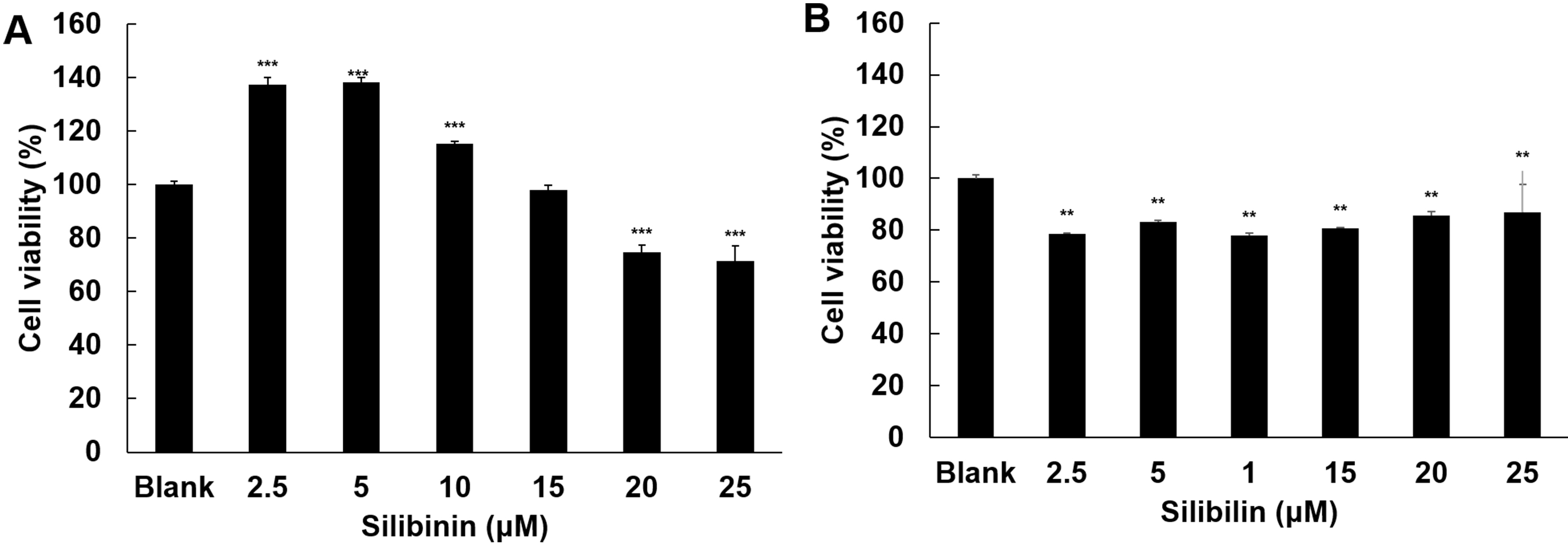 Fig. 2.
Fig. 2.
Effect of silibinin on cell viability. The effects of silibinin
on cell viability were examined in HT1080 cells (A) and IMR-90 cells (B),
respectively. The cells were treated with silibinin at 2.5, 5, 10, 15,
20, and 25 M. Cell viability was examined by MTT assay after 24 h
treatment. Data show mean SD from three independent experiments, each one
run in triplicate. The significance level between blank and silibinin treatment
was evaluated statistically (**, p 0.01; ***, p 0.001)
using ANOVA and post hoc (Duncan) test.
3.3 The Effect of Silibinin on MMPs Activation in HT1080 Cells
Stimulated with PMA
The effect of silibinin on MMPs activity was examined in HT1080 cells. Silibinin
at low doses had no inhibitory effect, and only at the two higher doses of 20
M and 25 M reduced MMP-9 activity by 188% and 943% compared to
the PMA group, respectively (Fig. 3). Silibinin effects on MMP2 were quite
different, because there was a stimulation of its activity at the lower doses
(5-10-15 M), whilst at only the highest concentration of 25 M it reduced
MMP-2 activity by 147%, compared to the PMA group.
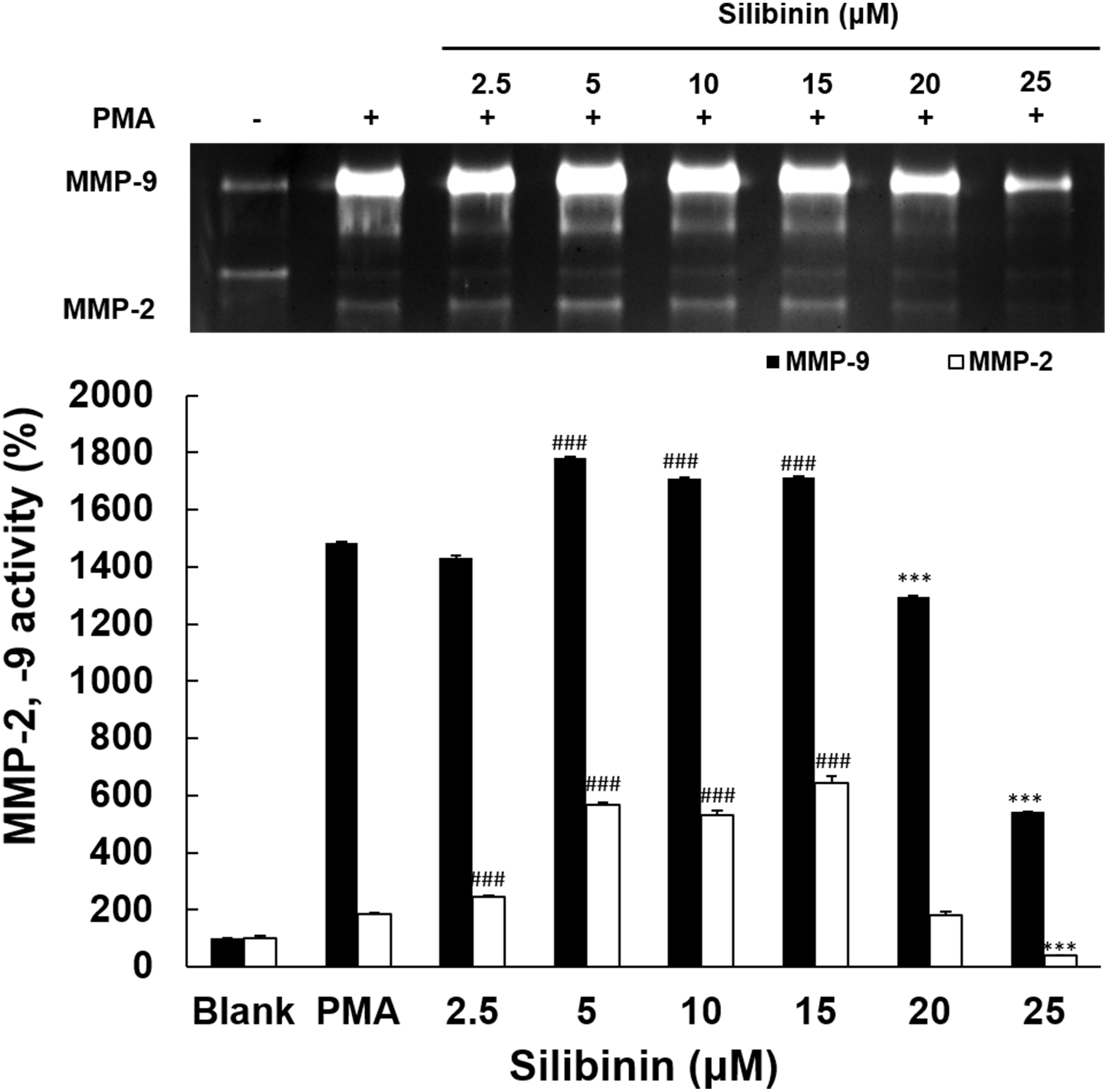 Fig. 3.
Fig. 3.
Effects of silibinin on MMPs activation. The inhibitory effects
of silibinin on the inhibitory activities of MMP-9 and MMP-2 was evaluated in
PMA-stimulated HT1080 cells to induce MMPs expression. The silibinin at 2.5, 5,
10, 15, 20, and 25 M was added under serum-free conditions for 72 h. MMP-9
and MMP-2 activities were analyzed using gelatin zymography assay. Data display
means values SD from triplicate experiments. The significance level
between PMA groups and silibinin treatment groups was determined statistically
(, p 0.001 for the activity increase of MMPs, ***,
p 0.001 for the activity decrease of MMPs) using ANOVA and post hoc
(Duncan) test.
3.4 Effect of Silibinin on the Protein Expression Associated with
Metastasis
The expressions of MAPK and other mediators regulating MMPs were examined to
elucidate how silibinin influences the regulation of MMP-2 and MMP-9 expression
and activity. The expression of ERK-1/2, p-p38, p-JNK (Fig. 4A), MMP-2, TIMP-1, and IL-1 (Fig. 4B) were analyzed in PMA-stimulated
cells with or without silibinin presence, by using western blot. The expression
of ERK-1/2 was diminished by 27% only in the presence of the highest dose (25
M) of silibinin, whereas p-p38 expression was inhibited at all
doses tested, with values between 23% and 50%. In particular, the expression
level of p-JNK was significantly higher than in the PMA-treated control
group in the presence of silibinin at 10 M, with values of 28%. Silibinin
at all doses progressively inhibited the expression of MMP2, TIMP1 and
IL-1 and at the highest dose of 25 M reduced the expression level
of MMP-2 by 64%, TIMP-1 by 149% and IL-1 by 91% compared to the
PMA-stimulated group.
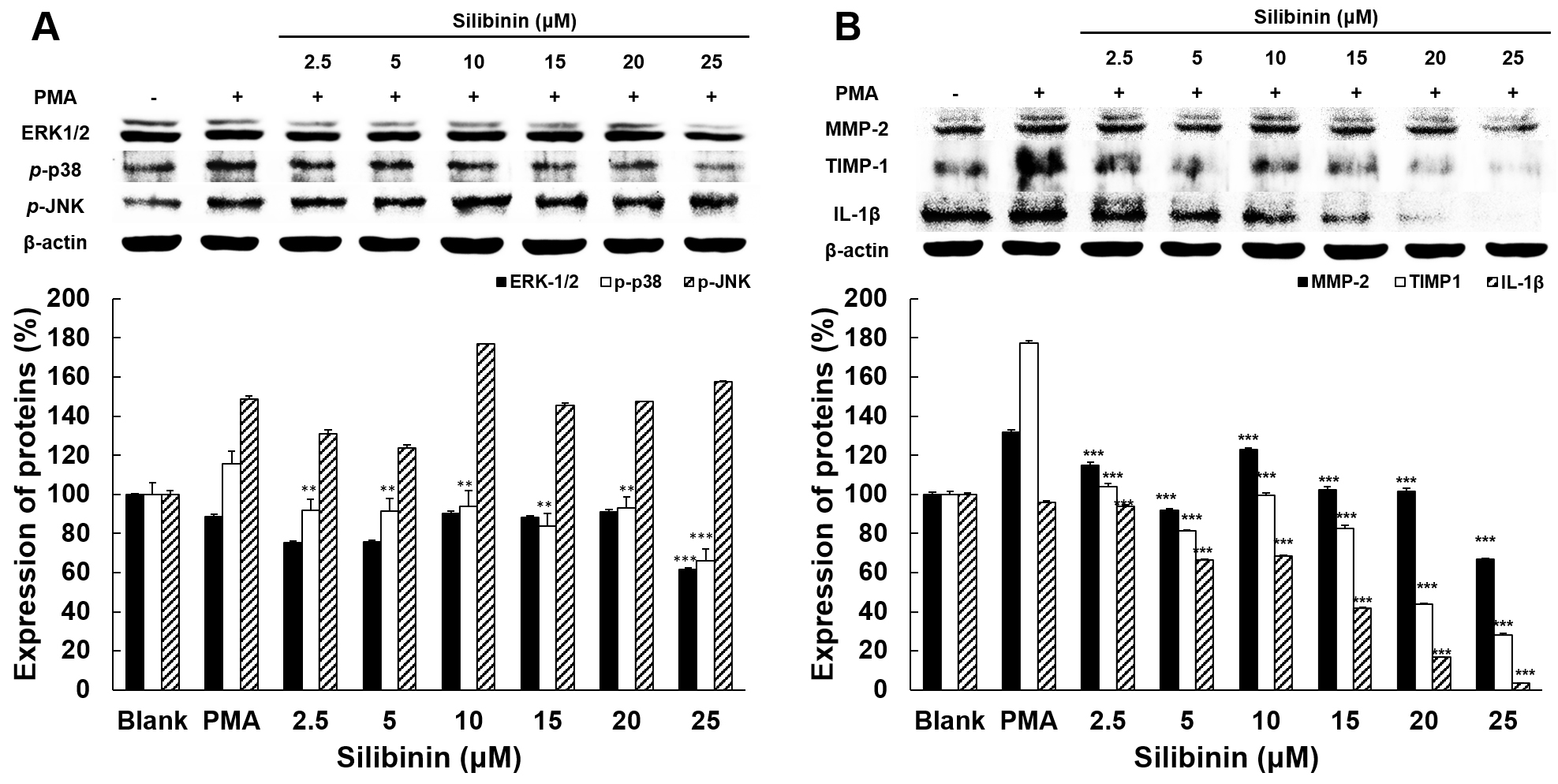 Fig. 4.
Fig. 4.
The effect of silibinin on the expression of proteins associated
with invasion and metastasis in HT1080 cells. (A) Effects of silibinin on the
expressions of ERK-1/2, p-p38, and p-JNK. (B) Effects of
silibinin on the expressions of MMP-2, TIMP-1, and IL-1. The level of
protein expression in cell lysates was determined by western blot analysis using
the indicated antibodies. Target proteins were normalized using the expression of
-actin as housekeeping reference. Data represent means SD from
triplicate experiments. The level of significance between PMA groups and
silibinin treatment groups was identified statistically (**p 0.01;
***, p 0.001) using ANOVA and post hoc (Duncan) test.
3.5 Immunofluorescence Staining of p-p38, IL-1 and MMP-2
Associated with Metastasis
To investigate the effect of silibinin on the expression of metastasis-related
proteins, immunofluorescence staining of MMP-2 was performed in PMA-stimulated
HT1080 cells, with or without silibinin treatment. The cell’s nuclei were
observed in blue color after being stained with DAPI. MMP-2 were stained with CY3
and displayed in red color. Silibinin treatment at 25 M decreased the
degrees of MMP-2 compared to the PMA-stimulated group (Fig. 5), thus confirming
that silibinin at 25 M could reduce MMP2 expression.
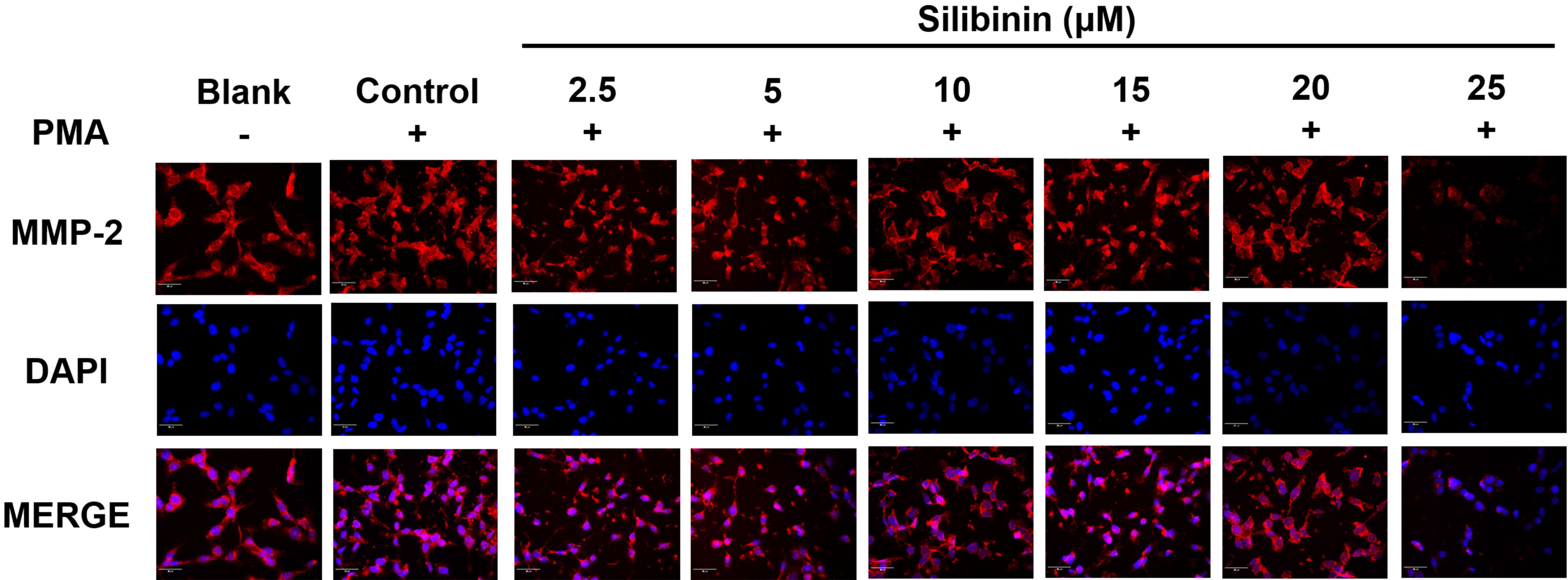 Fig. 5.
Fig. 5.
The immunofluorescence staining analysis of MMP-2. HT1080 cells
were treated with silibinin at 2.5, 5, 10, 15, 20, and 25 M in the
presence of PMA. Cells were detected with specific antibodies and counterstained
with DAPI. The cells were observed at 200 of magnification using the
iRiS™ Digital Cell Imaging System.
3.6 Effect of Silibinin on Cell Invasion Related to Metastasis
Tumor cells degrade collagen in the extracellular matrix to obtain more
nutrients and make space to move into other tissues through blood vessels.
Therefore, in this study, an invasion assay was performed using HT1080 cells
stimulated with PMA, in order to evaluate the efficacy of silibinin in the
inhibition of cell invasion. Paradoxically, though in line with data on cell
growth and effects on MMP2, silibinin treatment at low doses promoted invasion,
while at concentrations above 10 M progressively diminished cell invasion
(Fig. 6). Silibinin at 25 M inhibited cell invasion by 73% compared to
PMA-stimulated cells, thus supporting the effect of silibinin as inhibitor of
cancer metastasis.
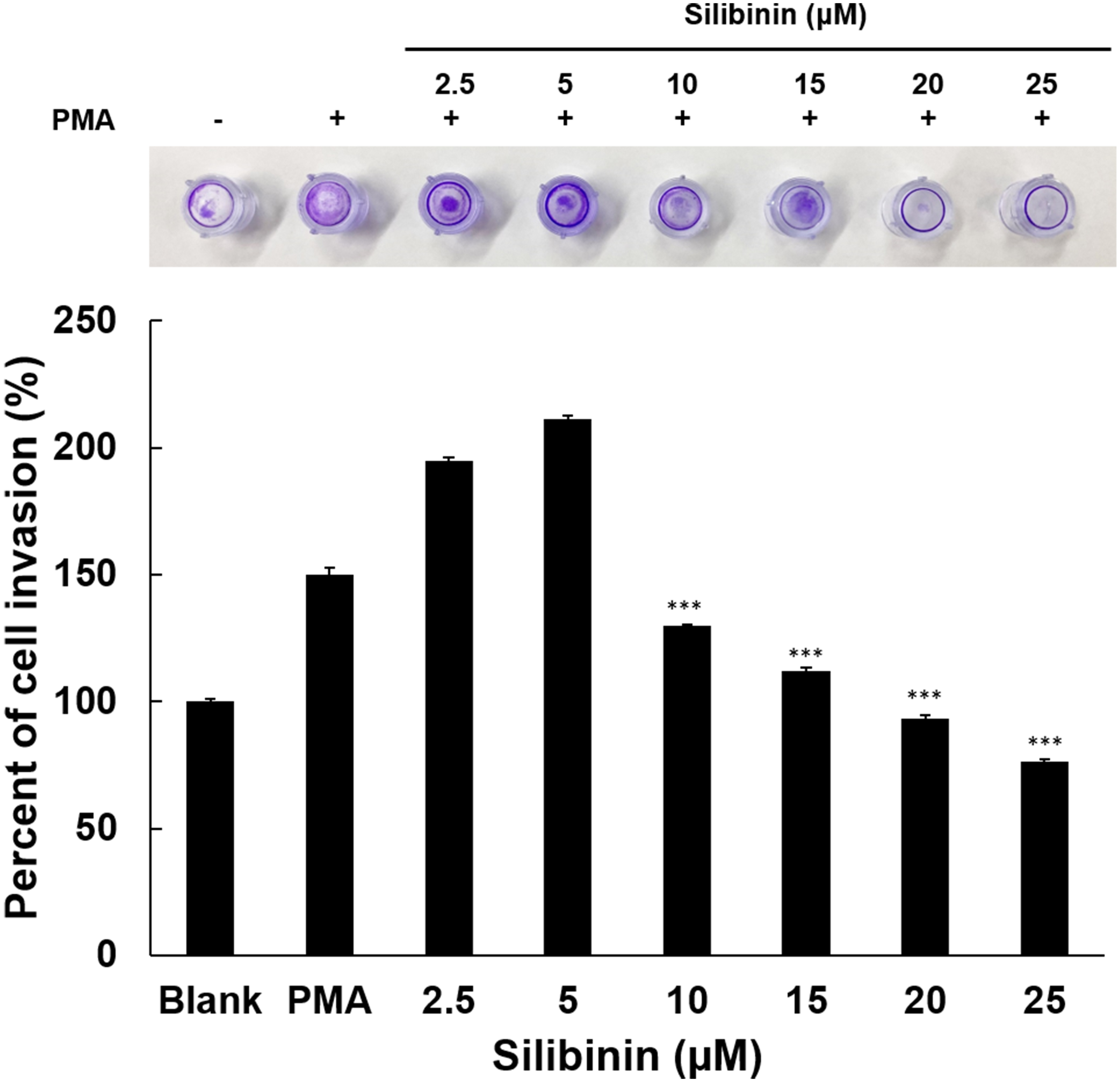 Fig. 6.
Fig. 6.
Effects of silibinin on HT1080 cell invasion. Cell penetration
into the ECM layer through the polycarbonate membrane was examined in the
presence of silibinin. Using a 24-well tissue culture plate with an insert and a
polycarbonate membrane with an 8 m pore size, an invasion assay was
carried out in the invasion chamber. To treat HT1080 cells, silibinin was used at
concentrations of 2.5, 5, 10, 15, and 25 M. Data display means values SD
from triplicate experiments. The level of significance between PMA groups and
silibinin treatment groups was identified (***, p 0.001) using ANOVA
and post hoc (Duncan) test.
4. Discussion
Chronic inflammation and the continuous release of inflammatory cytokines may
contribute to cancer development and its evolution into a malignancy that demands
oxygen and nutrition over time. As a result, matrix metalloproteinases (MMPs) can
be induced in growing cancers, when the surrounding nutrients are low. MMP-2 and
MMP-9 in particular digest collagen IV, a critical component of the extracellular
matrix. Cancer cells then may reach and travel through the bloodstream, finally
infiltrating metastatic cells into other permissive tissues. Therefore, blocking
cell invasion may play a key role in cancer therapy and the prevention of cancer
metastasis. In this study, we have shown that silibinin at the highest doses
tested may suppress MMP-9 and MMP-2 activity and expression, as well as
IL-1 and p-p38, with the final effect of preventing cell
invasion by human fibrosarcoma cells (HT1080), previously stimulated by the tumor
promoter PMA. This effect raises the intriguing possibility that silibinin could
be used as a therapeutic candidate to prevent cancer cells invasion and
metastasis. Previous study reported that silibinin possesses strong antioxidant
activity and also modulates many molecular changes caused by xenobiotics and
ultraviolet radiation to protect the skin [7]. Although we did not find an
antioxidant activity of silibinin at the doses here tested, and only a modest
reducing ability (Fig. 1), previous studies reported that silibinin was effective
in blocking the cell cycle [8, 9, 10, 11, 12].
However, the doses of silibinin here used could inhibit the activity of
gelatinases such as MMP-2 and MMP-9 in HT1080 cells previously stimulated with
PMA. This effect is consistent with previously published results showing that
silibinin inhibited the activity of MMP-2 and MMP-9 in osteoblasts [13].
During metastasis, AP-1 and NF-kB are the main mediators that promote the
transcription of tumor necrosis factor (TNF) , interleukin-1,
interleukin-6, growth factor, COX-2, and MMPs [14, 15]. PMA is a cancer promoter,
is an activator of protein kinase A and phosphorylates and activates ERK, JNK,
and p38, all playing a crucial role in cell division and apoptosis. The activated
MAPK migrates to the nucleus and regulates the production of MMPs by modulating
the expression of transcription factors AP-1 and NF-kB, finally resulting in cell
invasion and metastasis [16]. In this study, we have shown the inhibitory effects
of silibinin the effects of silibinin on the protein expression levels of
IL-1, NF-kB and AP-1 as well as MMP-2, MMP-9 and MAPKs such as ERK-1/2,
p-p38, and p-JNK were investigated in fibrosarcoma cells. It
was shown that the expression of IL-1, an inflammatory mediator, was
also remarkably reduced by silibinin. Accordingly, silibinin was reported to
inhibit the expression of the expression degree of IL-1 in HepG2 and RAW
264.7 cells [8, 9], thereby suppressing inflammation and inhibiting metastasis
[17, 18]. Therefore, these findings are consistent with the previous result,
suggesting that the inhibitory effect of silibinin on IL-1 could play a
key role in the prevention of metastasis [19]. In addition, silibinin increased
the activity of p-JNK, but decreased the expression of ERK-1/2 and
inhibited the activation of p38. Along the same line, previous studies reported
that oxymatrine and resveratrol inhibited the activity of p-p38 and
ERK-1/2, thereby reducing MMP expression and inhibiting cancer metastasis [20, 21]. Therefore, it is tempting to suggest that silibinin could suppress
metastasis by inhibiting p-p38 and ERK-1/2, thereby inhibiting MMPs
expression and activity, necessary for invasion and metastasis.
In addition, our results indicate that silibinin could modulate the MAPK
signaling pathway of ERK and p38 via IL-1, finally decreasing MMPs
expression. This resulted in the inhibition of invasive abilities of fibrosarcoma
cells, as shown by the invasion assay performed on HT1080 cells in the presence
of PMA and silibinin (Fig. 6). Similarly, in a previous study it was also
reported that metformin suppressed MMPs through AP-1 and NF-kB inhibition in
MCF-7 cells finally reducing their metastatic ability [22] (Fig. 7).
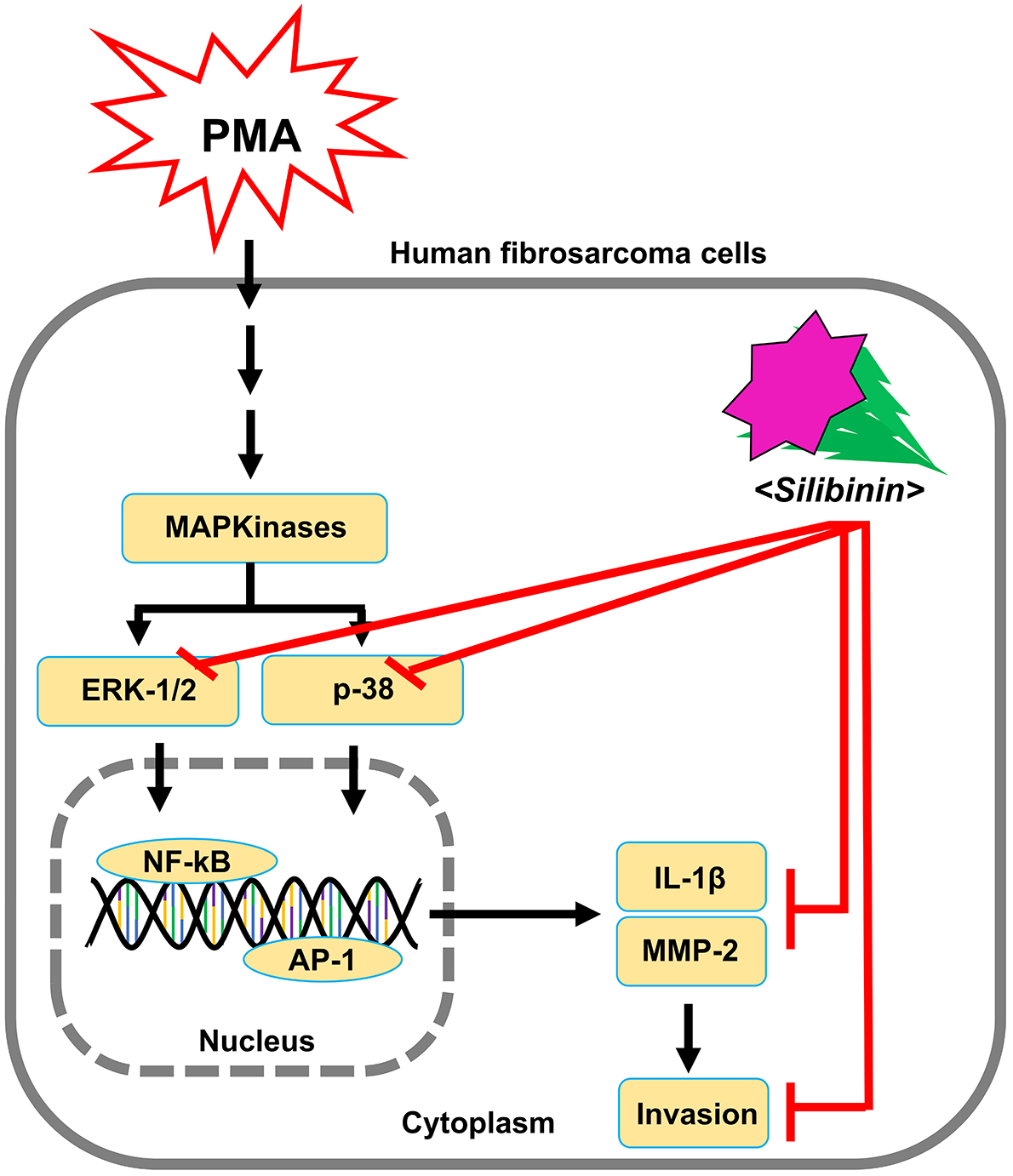 Fig. 7.
Fig. 7.
Schematic diagram for the effect of silibinin on invasion and
metastasis of HT1080 cells stimulated by PMA.
5. Conclusions
In the end, this study shows a paradoxical effect of silibinin on HT1080 cells.
At low doses, it appears to promote growth and MMPs expression, thus favoring
invasion. Only at the higher doses it shows inhibitory effects on these
parameters, and therefore it might be supposed to inhibit the metastatic ability
of cancer cells. In vivo, it is not predictable which dose will reach
tumor cells within the tumor mass, and so it remains also unpredictable which
effect is expected, whether promotion or inhibition of invasion and metastasis.
More studies in vivo, at different doses, will be necessary to elucidate
the pharmacokinetics and the pharmacodynamics of silibinin, in order to identify
a dose which will exert the desired effect of preventing cell invasion and
metastasis.
Abbreviations
ATCC, American Type Culture Collection; DMEM, Dulbecco’s Modified Eagle’s
Medium; DMSO, dimethyl sulfoxide; DPPH, 2,2-diphenyl-1-picrylhydrazyl; ERK,
extracellular signal-controlled kinases; FBS, fetal bovine serum; HT1080, human
fibrosarcoma cells; IL-1, Interleukin-1beta; MAPK, mitogen-activated
protein kinase; MMP-2, Matrix metalloproteinase 2; MMPs, matrix
metalloproteinase; MTT, 3-(4,5-Dimethyl-2-yl)-2,5-diphenyl tetrazolium bromide;
PMA, phorbol myristate acetate; p-JNK, phospho-c-Jun NH 2-terminal
kinases; TCA, trichloroacetic acid; TIMP-1, TIMP metallopeptidase inhibitor 1;
TNF, tumor necrosis factor; Vit. C, vitamin C.
Availability of Data and Materials
The data used to support the findings of this study are available from the
corresponding author upon request.
Author Contributions
AIJ performed the experiments, analyzed the data, and assisted in writing the
manuscript. Prof MMK proposed the concept, designed the experiment, analyzed the
data, and revised the manuscript. Both authors read and approved the final
manuscript and agree to be accountable for all aspects of the research.
Ethics Approval and Consent to Participate
Not applicable.
Acknowledgment
We wish to thank Sojeong Jeon for her kind advice and help with this study.
Funding
This research received no external funding.
Conflict of Interest
The authors declare no conflict of interest.
 Fig. 1.
Fig. 1. Fig. 2.
Fig. 2. Fig. 3.
Fig. 3. Fig. 4.
Fig. 4. Fig. 5.
Fig. 5. Fig. 6.
Fig. 6. Fig. 7.
Fig. 7.






