1 Department of Experimental Medicine, University of Tor Vergata, 00133 Rome, Italy
2 Department of Laboratory Medicine, Tor Vergata University Hospital, 00133 Rome, Italy
3 Immunology and Allergology Laboratory Unit, S. Giovanni di Dio Hospital, 50143 Florence, Italy
4 Department of Experimental and Clinical Medicine, University of Florence, 50121 Florence, Italy
5 Rheumatology Unit, S. Giovanni di Dio Hospital, 50143 Florence, Italy
6 Clinical Pathology Laboratory Unit, S. Giovanni di Dio Hospital, 50143 Florence, Italy
7 Department of Health Science, University of Florence, 50121 Florence, Italy
8 Laboratory of Virology, National Institute for Infectious Diseases Lazzaro Spallanzani, IRCCS, 00149 Rome, Italy
9 Emerging Technologies Division, International Federation Clinical Chemistry and Laboratory Medicine, 20159 Milan, Italy
†These authors contributed equally.
Academic Editor: Alessandro Poggi
Abstract
Background: Several commercial surrogate Virus Neutralization Tests
(sVNTs) have been developed in the last year. Neutralizing anti-SARS-CoV-2
antibodies through interaction with Spike protein Receptor Binding Domain (S-RBD)
can block the virus from entering and infecting host cells. However, there is a
lack of information about the functional activity of SARS-CoV-2 antibodies that
may be associated with protective responses. For these reasons, to counteract
viral infection, the conventional virus neutralization test (VNT) is still
considered the gold standard. The aim of this study was to contribute more and
detailed information about sVNTs’ performance, by determining in vitro
the anti-SARS-CoV-2 neutralizing antibody concentration using four different
commercial assays and then comparing the obtained data to VNT. Methods:
Eighty-eight samples were tested using two chemiluminescence assays (Snibe and
Mindray) and two ELISA assays (Euroimmun and Diesse). The antibody titers were
subsequently detected and quantified by VNT. Results: The overall
agreement between each sVNT and VNT was 95.45% for Euroimmun and 98.86% for
Diesse, Mindray and Snibe. Additionally, we investigated whether the sVNTs were
closer to the gold standard than traditional anti-SARS-CoV-2 antibody assays
S-RBD or S1 based, finding a higher agreement mean value for sVNTs (98.01
Keywords
- surrogate Virus Neutralization Tests
- live virus neutralization test
- anti-SARS-CoV-2 antibodies
- immunoassays
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and resulting COVID-19 disease have presented a relevant challenge in laboratory diagnostics [1, 2]. During the current pandemic, several SARS-CoV-2 diagnostic platforms have been developed and tested for clinical use. Serological assays are essential in the detection of anti-SARS-CoV-2 antibodies and in monitoring convalescent and vaccine-induced immunity [3]. Mainly, they play a supplementary but indispensable role in diagnosing suspected cases with negative swab, in asymptomatic or past SARS-CoV-2 infection patients, for epidemiological assessment, assuring the contact-tracing of positive cases and identifying new foci, and in evaluating vaccine efficacy and therapeutic antibody development [4, 5, 6, 7, 8].
During the last 18 months, different serologic tests detecting specific SARS-CoV-2 antibodies were developed, some of which give quantitative results; however, these tests may detect all binding antibodies to a specific target, but they do not provide correct information on the potency of functional antibodies that may be associated with protective responses. In general, when analysing the correlation between antibody titer and neutralizing capacity it is necessary to distinguish between humoral immunity induced by natural infection and humoral immunity induced by vaccination. In fact, only in the latter do the amount of antibodies and the neutralizing capacity correlate very well [9, 10, 11, 12]. Therefore, in parallel to the development of reliable quantitative serological tests, there is an urgent need for fast and sensible functional assays to improve the understanding of SARS-CoV-2 immune responses, their kinetics over time and the knowledge of antibody persistence [13]. It is unclear how long SARS-CoV-2 neutralizing antibodies last and, as a result, whether they can provide long-term protection [14]. Although, some studies have shown a correlation between binding and neutralizing antibodies, neutralizing antibody assays are still a gold standard to judge the immunity tone of a patient [15, 16].
Notably, the Virus Neutralization Test (VNT), and more specifically the Plaque Reduction Neutralization Test (PRNT) [17], is considered the gold standard for serological detection measuring neutralizing antibodies. It detects functional antibodies and its results demonstrate blocking virus infection, thus representing a strong correlative indicator of protection from disease. As PRNT variants, some Neutralization Tests (NTs) were developed by observing the Focus Reduction (FRNT) or the CytoPathic Effect (CPE) [18]. The principles are basically identical to those of PRNT, and are based on the single microscopy observation of infected foci or CPE reduction, instead of counting plaques.
All these assays use SARS-CoV-2 replicating virus. VNTs are inconvenient in that they require BioSafety Level (BSL)-3 laboratories and often require intense and time consuming (3–7 days) work as well as not being readily amenable to automatization [19].
Alternatively, pseudotype-based neutralization assays represent an improvement towards VNT with the advantage of being able to work at a lower containment level (BSL-2), in which case infectivity can be measured by marker gene expression using ELISA plate readers. However, they still require 1–2 days, skilled operators, and are affected by variations between laboratories and different preparations of viral pseudotypes.
Many efforts have been made to develop other methods to test sera for the presence of neutralizing antibodies that are more practical, simple, and at the same time, reliable.
These are commercial assays that detect SARS-CoV-2-specific antibodies directed against the Spike Receptor Binding Domain (S-RBD) protein and are capable of detecting antibodies showing neutralizing activity [20, 21]. This year, several surrogate Virus Neutralization Tests (sVNTs) have appeared on the market that, based on the manufacturers’ description, claim excellent correlation with the neutralizing titer.
Neutralizing antibodies, in most cases act by interacting with the binding between the S-RBD protein on the viral surface and its receptor, the angiotensin-2 converting enzyme (ACE-2) receptor, expressed on the target cell. Neutralizing antibodies can block virus entry and infection in the host cell through interaction with S-RBD [22, 23]. This mechanism of virus entry inhibition into the target cell reduces viral replication and consequently viral load. Indeed, the appearance of neutralizing antibodies correlates with host immunity and protects, at least temporarily, against future reinfection [24, 25]. Recent studies have shown that the level of neutralizing antibodies can vary markedly between patients with severe and mild-to-moderate symptoms [26, 27].
However, there are no current data available regarding the titer of neutralizing antibodies providing patients protection from reinfection or from disease. Whilst the mechanism of protection certainly involves cellular immunity, there are supporting data which suggest neutralizing antibodies may be a correlate of protection [24, 27, 28].
To increase comparability of the results of serologic tests detecting neutralizing antibodies to support the identification of a possible protective correlate, it is crucial that all tests are calibrated and developed using a well-characterized and globally traceable reference standard. In this regard, an International Standard (IS) containing a known amount of anti-SARS-CoV-2 immunoglobulins with well-characterized neutralizing activity was made available by the WHO in December 2020 with the aim of allowing the accurate calibration of assays to the International Unit (IU) for neutralising antibodies or Binding Antibody Units (BAU) for binding antibodies, thereby reducing interlaboratory variation and creating a common language for reporting data [29]. In light of these premises, comparative performance data and proper validation are essential to guide the appropriate use of serology in COVID-19 diagnostics [13].
Our principal aim was to compare four different assay systems to determine in vitro SARS-CoV-2 neutralizing antibody concentrations and correlating the antibody levels with the gold standard VNT. Finally, we investigated whether the sVNTs performances were closer to the gold standard, comparing them with the traditional anti-SARS-CoV-2 S-RBD or S1 based antibody assays.
Eighty-eight consecutive serum samples (F:M ratio 1.4:1, mean age = 57.0
| Manufacturer | Kit assay | Method | Cut-off | Conversion factor | Dynamic range | Assay principle |
| Mindray | SARS-CoV-2 Neutralizing Antibodies | CLIA | 10 AU/mL | 1 AU = 3.6547 IU/mL | 2 AU/mL to 400 AU/mL | Neutralizing antibody in the sample competes with ACE2-ALP conjugate for binding sites of SARS-CoV-2 S-RBD immobilized in the paramagnetic microparticles |
| Snibe | Maglumi SARS-CoV-2 Neutralizing Antibody | CLIA | 0.05 µg/mL | 1 µg/mL = 405 IU/mL | 0.050–30 µg/mL | Neutralizing antibody in the sample competes with recombinant SARS-CoV-2 S-RBD antigen labeled with ABEI for binding ACE2 antigen immobilized on magnetic microbeads |
| Euroimmun | SARS-CoV-2 NeutraLISA | ELISA | % IH |
n.a. | % IH 0–100 | Neutralizing antibody in the sample competes with the receptor biotinylated ACE2 (sample buffer) for the binding sites of the SARS-CoV-2 S1/RBD proteins immobilized on the plate |
| % IH | ||||||
| Diesse | Chorus SARS-CoV-2 “Neutralizing” Ab | ELISA | n.a. | 20–1500 BAU/mL | Neutralizing antibody in the sample competes with the peroxidase-conjugated SARS-CoV-2 anti-S1 therapeutic monoclonal antibodies to bind S1 subunit fixed on the solid phase support | |
| ABEI, N-(aminobutyl)-N-(ethylisoluminol); ALP, alkaline phosphatase; CLIA, chemiluminescence immunoassay; ELISA, enzyme-linked immunosorbent assay; IH, percentage of inhibition; n.a., not available. | ||||||
The “Maglumi SARS-CoV-2 Neutralizing antibody” assay (Snibe Diagnostic,
Shenzhen, China) for the detection of anti-SARS-CoV-2 IgG/IgA/IgM neutralizing
antibodies was performed on the fully automated Maglumi analytical system (Snibe
Diagnostic, Shenzhen, China). This assay is based on the competition between
neutralizing antibodies and S-RBD antigens for the binding to ACE2. The linearity
range is 0.050–30
The “SARS-CoV-2 Neutralizing Antibody” chemiluminescent assay (Mindray Medical, Shenzhen, China) for the detection of anti-SARS-CoV-2 neutralizing antibodies was performed on the fully automated Mindray CL-1200i analytical system (Mindray Medical, Shenzhen, China). Neutralizing antibody in the sample competes with ACE2-alkaline phosphatase conjugate for binding sites of SARS-CoV-2 antigens. The linearity range is 2 AU/mL to 400 AU/mL, cut-off value is 10 AU/mL. As declared by the manufacturer, the conversion factor to transform AU/mL in IU/mL is 0.2734; sensitivity and specificity are 95.7% and 99.9%, respectively.
The “SARS-CoV-2 NeutraLISA” enzyme-linked immunosorbent assay (ELISA) (Euroimmun, Lübeck, Germany) provides semiquantitative in vitro determination of neutralizing anti-SARS-CoV-2 antibodies. The assay is based on the competition between neutralizing antibodies and S-RBD antigens for the binding to ACE2. Briefly, during the first incubation, samples and controls are diluted with a sample buffer containing biotinylated ACE2. If neutralizing antibodies are present in the sample, they compete with the ACE2 receptor for the binding sites of the SARS-CoV-2 S1/RBD proteins. A second incubation step is performed to detect the bound ACE2 by the addition of peroxidase-labelled streptavidin, which catalyses a colour reaction. The intensity of the products absorbance is inversely proportional to the concentration of neutralizing antibodies in the sample.
The percentage of inhibition (% IH) is calculated with the following formula:
% IH = 100% – (Extinction of patient sample
Results below 20% IH are considered negative;
The “Chorus SARS-CoV-2 “Neutralizing” Ab” (Diesse Diagnostica Senese, Siena, Italy) for the quantitative determination of total anti-S1 SARS-CoV-2 neutralizing antibodies was performed on the automated Chorus TRIO instrument (Diesse Diagnostica Senese, Siena, Italy).
The detection method is based on the principle of competitive testing using SARS-CoV-2 anti-S1 therapeutic monoclonal antibodies.
The SARS-CoV-2 anti-S1 antibodies present in the sample compete with a peroxidase-conjugated anti-S1 monoclonal antibody to bind the Spike protein S1 subunit Recombinant Binding Domain (RBD) site fixed on the solid phase support and available in limited numbers. The higher the concentration of antibodies present in the patient’s serum, the lower the possibility of binding peroxidase-conjugated SARS-CoV-2 anti-S1 monoclonal antibody to the fixed antigen.
The results are expressed in BAU/mL, as declared by the manufacturer.
The samples are considered positive for values
The live virus neutralization test (VNT) is a specialized type of immunoassay to detect antibodies able to inhibit virus replication in vitro. The live VNT was performed to establish the lower serum cut-off value to protect against SARS-CoV-2 infection.
The neutralizing antibodies titer was determined using the following 4 step-protocol: epithelial cell line VERO E6 culture, viral growth in cell culture and SARS-CoV-2 virus titration and micro-neutralization assay with subsequent cytopathic effect (CPE)-read out.
Epithelial cell line VERO E6, derived from the kidney of African green monkey Cercopithecus aethiops, was acquired from the American Type Culture Collection (ATCC-CRL 1586). Adherent sub-confluent VERO E6 cells monolayers were prepared in DMEM medium containing 10% FBS.
SARS-CoV-2 virus 2019-nCov/Italy-INMI1-strain (GISAID accession ID: EPI_ISL_412974) was purchased from Lazzaro Spallanzani Institute (Rome, Italy) via the European Virus Archive Global (EVAg).
The virus was propagated on VERO E6 using DMEM, supplemented with 2% FBS, cells were daily observed, and the virus was harvested until an 80–90% CPE was observed. This viral preparation was titrated in serial 1 Log dilutions to obtain a 50% tissue culture infective dose (TCID50) on 96-well culture plates of VERO E6 cells. The end-point titer was calculated according to the Reed & Muench method based on eight replicates for each titration.
Sera were heat-inactivated for 30 minutes at 56
Results were expressed as the mean value with Standard Deviation (SD) when data were normally distributed. Otherwise, results were expressed as the median value with range. The Student t-test and the correlation coefficient (in the case of normally distributed data), the Wilcoxon’s matched pairs t-test and the Spearman’s coefficient of rank correlation (both in the case of non-normally distributed data) were used when required. p values less than 0.05 were considered statistically significant. Statistical analysis was performed using MedCalc Statistical Software version 14.8.1 (MedCalc Software bvba, Ostend, Belgium; http://www.medcalc.org; 2014).
In this study, all samples have been tested with four different commercial sVNTs for anti-SARS-CoV-2 neutralizing antibodies detection and compared with the VNT titer.
In addition, a qualitative test was used to evaluate the overall agreement rate
between each sVNT and VNT. As shown in Table 2, a high agreement rate was
demonstrated for all tests, ranging from 95.45% (Euroimmun) to 98.86% (Diesse,
Mindray and Snibe). For each sVNT overall agreement, it has been associated a 2
| sVNT | % |
| Diesse sVNT | 98.86 |
| Mindray sVNT | 98.86 |
| Snibe sVNT | 98.86 |
| Euroimmun sVNT | 95.45 |
| Anti-SARS-CoV-2 | % |
| Mindray S RBD IgG | 94.32 |
| Roche S RBD tAb | 97.73 |
| Snibe S RBD IgG | 96.59 |
| DiaSorin Trimeric S IgG | 94.32 |
| Thermo Fisher S1 IgG | 93.18 |
| Beckman S1 IgG | 94.32 |
| Euroimmun S1 IgG | 96.59 |
| Vircell VIRCLIA S1 IgG | 93.18 |
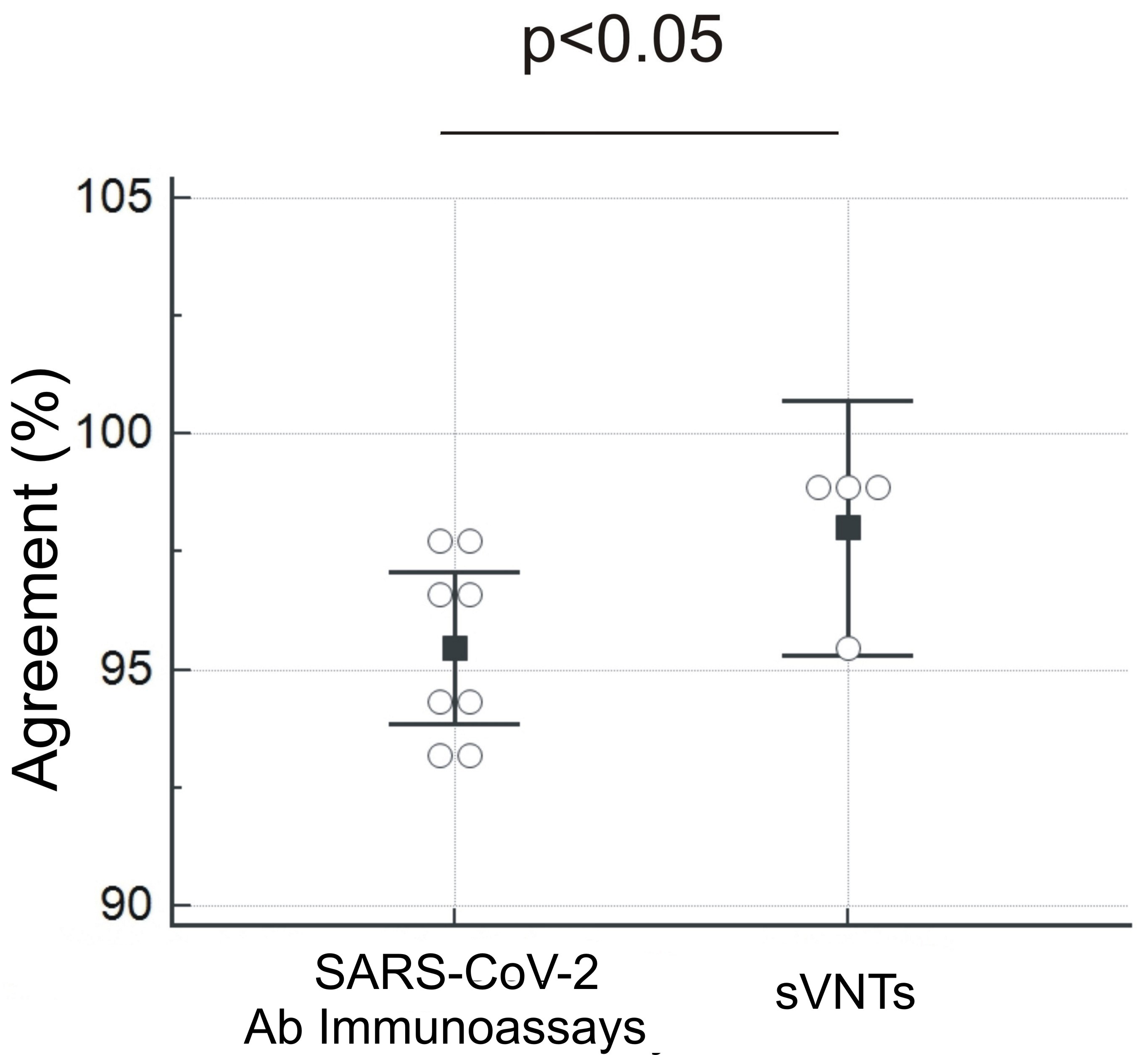 Fig. 1.
Fig. 1. Overall agreement of eight traditional anti-SARS-CoV-2 antibody
assays (S-RBD or S1 based) vs sVNTs. The sVNTs mean value (98.01
In Table 4, the discordant results for each sVNT and VNT assay were compared to the patients’ history: Euroimmun displayed four discordant cases; Diesse, Mindray and Snibe only one case. The discordances were possible false positive sVNT results except one possible false negative VNT case.
| VNT (titer) | sVNT | Patient’s history | Comment |
| Euroimmun sVNT | |||
| 1/160 | Negative | HCW, history of COVID-19 in 2020, unvaccinated | false negative? |
| 1/40 | Negative | HCW, history of COVID-19 in 2020, unvaccinated | false negative? |
| 1/40 | Negative | Patient in treatment with Certolizumab for reumatoid arthritis, never had COVID19, unvaccinated, negative swab | asymptomatic COVID-19? |
| 1/20 | Negative | HCW, history of COVID-19 in 2020, unvaccinated | false negative? |
| Diesse sVNT | |||
| 1/40 | Negative | HCW, history of COVID-19 in 2020, unvaccinated | false negative? |
| Mindray sVNT | |||
| Positive | HCW, history of COVID-19 in 2020, 1 vaccine dose given 15 days prior to the present blood test | true positive? | |
| higher sensitivity of the test compared to the VNT? | |||
| Snibe sVNT | |||
| 1/160 | Negative | HCW, history of COVID-19 in 2020, two months since 2nd dose of the vaccine to the present blood test | false negative? |
All quantified sVNTs results were graphically correlated to VNT in Fig. 2. The
dot plot graph illustrates on the vertical axis the sVNT antibody values and on
the horizontal axis the neutralization titers, expressed as the reciprocal of the
serum dilution, showing an increase in VNT titers correlated to the antibody
concentrations. Spearman’s statistical analysis was used in order to find the
correlation of neutralizing antibodies levels versus VNT. The highest coefficient
(r = 0.810) was obtained with Euroimmun sVNT (%) (Fig. 2D) and the lowest (r =
0.666) with Mindray sVNT (IU/mL) (Fig. 2B). Diesse sVNT (BAU/mL) achieved an r =
0.696 (Fig. 2A), while Maglumi sVNT (IU/mL) achieved an r = 0.779 (Fig. 2C). The
statistical significance level estimated was p
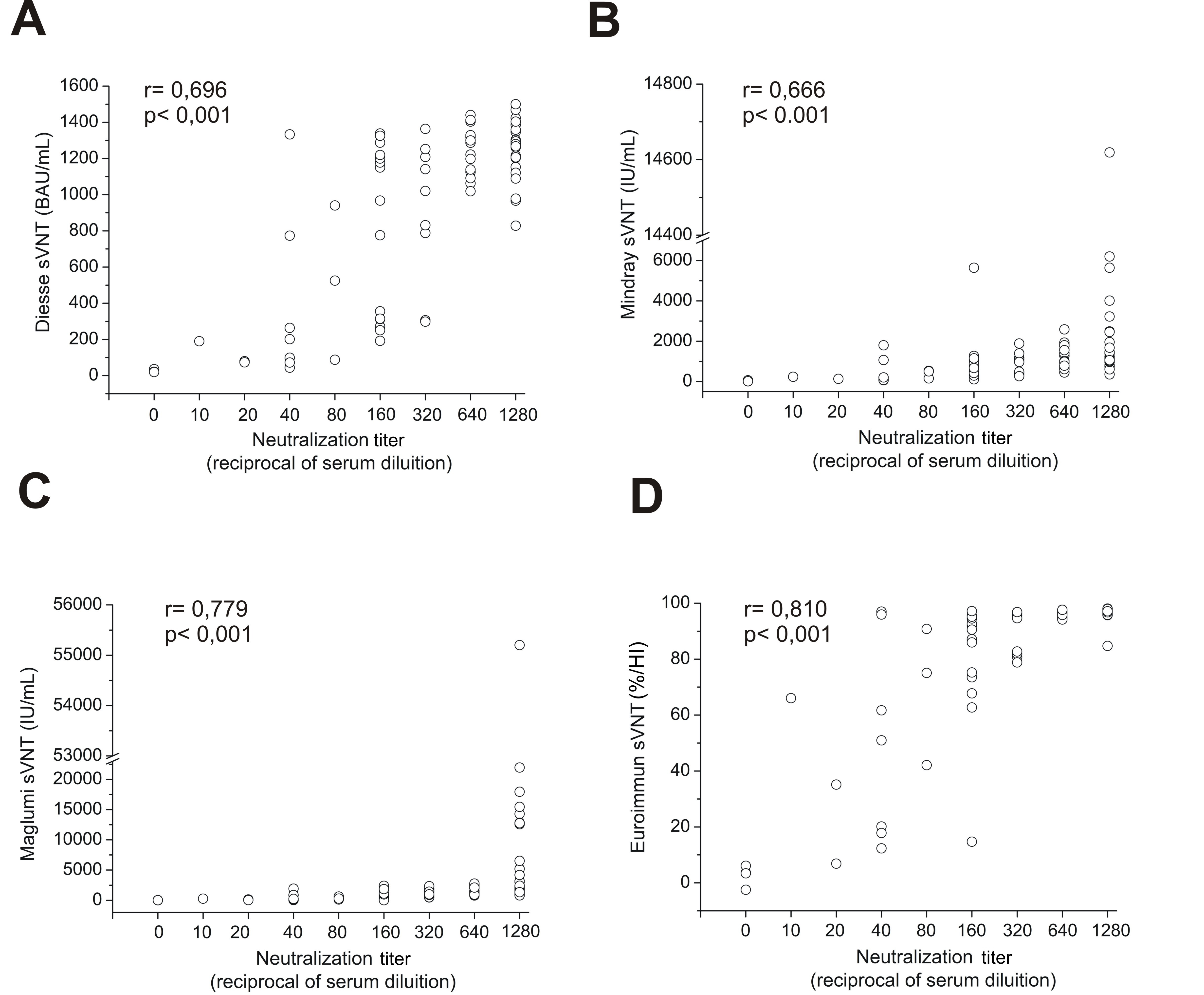 Fig. 2.
Fig. 2.Four graphs of correlation between sVNTs and VNT: (A) Diesse sVNT (BAU/mL) with r = 0.696. (B) Mindray sVNT (IU/mL) with r = 0.666. (C) Maglumi sVNT (IU/mL) with r = 0.779. (D) Euroimmun sVNT (% IH) with r = 0.819. Spearman’s test was used for correlation analysis.
After the first year of the COVID-19 outbreak, the role of serology shifted its focus from the acute diagnostic strategy of seroprevalence and protective herd immunity to (i) controlling the pandemic, to (ii) the critical measurements of individuals’ neutralizing antibody levels for vaccine clinical trials, and finally (iii) research studies and disease prevention.
Traditionally SARS-CoV-2 antibody assays detect the isotypes of different antibodies and use different antigenic proteins. However, since competition with ACE2 may reveal the presence of potential neutralizing antibodies for RBD binding, recently various assays mainly based on the ACE2-RBD competition have spread widely and are known as sVNT.
Despite WHO efforts, only some of the commercial kits available today use the IU suggested for the neutralization assays. In fact, in our study only two out of the four sVNTs tested express their results as IU (Mindray and Maglumi), one as a percentage of HI (Euroimmun) and one as BAU/mL (Diesse). Also, VNT uses NTs instead of IU. Conversely, the harmonization process for anti-SARS-CoV-2 antibody assays has been achieved since most commercial assays use BAU/mL. Since the minimal neutralizing antibody threshold of protection from infection is still unknown, as is how long this protection might last after natural infection or vaccination, there is a great need for assays profiling patient responses to infection, and to establish acquired immunity against SARS-CoV-2 [30, 31, 32].
The sVNTs, given their high throughput nature and no need for biosafety level 3 containment, make them accessible to the wider community, enabling greater availability for effective mass testing.
Our data confirm its potential routine use as a screening test of neutralizing antibodies in a diagnostic laboratory scenario, given the high percentage of agreement with the gold standard test (95.45%–98.86%) [10]. In addition, agreement between sVNT values and VNT was higher than SARS-CoV-2 immunoassays agreement (98.01% vs 95.02%). Regarding the association between NTs and quantitative results, we observed a higher but not significant Spearman coefficient (sVNTs: r = 0.738 vs. SARS-CoV-2 immunoassays: r = 0.711).
However, the moderate correlation observed suggests that VNT cannot be replaced by the two examined assays groups, with the purpose of assessing functional antibody activity. Interestingly, the Euroimmun sVNT is the test with the highest number of qualitative discrepancies but with the strongest quantitative correlation, meaning the different aspects of the test. Our results are partially in agreement with a recent study [10] which showed an average correlation of 0.473, assessed by Spearman’s test, between VNT and three different SARS-CoV-2 immunoassays. In detail, the correlation assessed by Matusali et al. [10] was lower compared to our results (0.473 vs. 0.724), possibly due to the different population studied (COVID-19 patients’ sera vs routine sera) and to the assays examined. In fact, the tests used in our study were all anti-SARS-CoV-2 antibody assays S-RBD or S1 based, excluding nucleocapsid based assays with a lower neutralizing capacity activity [10]. Despite great efforts in recent months to develop reliable assays, the VNT still remains the gold standard, given its intrinsic characteristics. Indeed, the sVNTs may reveal only a portion of neutralizing antibodies and not measure the total neutralizing activity directed against epitopes outside the RBD, such as the N-terminal domain of the S protein [33, 34, 35, 36].
Our correlation data between sVNT vs VNT show significant Spearman coefficients,
with a statistical significance level p
Most evaluation studies have mainly compared serological assays with VNT results [37, 38, 39, 40, 41, 42, 43]. but, to the best of our knowledge, there are no studies that have considered the two different assay families separately, anti-SARS-CoV-2 antibody assays (S-RBD or S1 based) and sVNTs, compared with each other and against the gold standard.
The main limitation of the study is the small number of negative patients included due to the fact that we studied a routine population during the pandemic and the vaccination campaign, consequently scarce of negative samples. Therefore the bias resulting by a higher number of negative samples might alter the overall agreement. A second limitation of this study was that we did not use the Genscript (cPass) [44] as the sVNT reference method. Indeed, cPass is widely accepted as a reference in the literature and it is fully approved by the FDA. Unfortunately, at this stage it was not possible to perform further comparisons.
Finally, we calculated the median value for all commercial assays at 1/160 VNT titer. Mindray, Maglumi and Diesse had similar median values while Euroimmun could not be compared because it has a percentage unit of measurement. However, all tests show that as the concentration of serum antibodies increases, so does the VNT titer.
However, the strength of our work is that it is a so-called “real-life” research study typically designed to better reflect aspects of routine care.
Starting from these premises, our comparison study aims to contribute to the refinement of anti-SARS-CoV-2 antibody testing strategies for public health use.
Further studies on larger sample numbers are required to better understand the strengths and limitations of commercially available serological assays and for the flow chart optimization of COVID-19 serological tests, including both families of tests at different levels.
Serological studies are an aid to questions about the duration of immune protection and vaccine effectiveness. Although VNT still remains the gold standard, given the good agreement between VNT and sVNT, sVNT has proved to be a potential valuable tool for screening larger populations, due to shorter response times, lower costs, and good performance. In the next future sVNT could be used to personalize decision on vaccination shedule or disease therapy.
These should be presented as follows: MP, MI, MM, MN designed the research study. MI, MP, MN, VG, FT, SS performed the research. CC, AA, MM, PC, MB and SB provided critical revision of the manuscript and valuable comments. MI, MP, MN, LS, ER, FT, SS, BL analyzed the data. MI, MP, MM, MN, ER wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
The study was performed according to local ethical approval protocol no. R.S.44.20. Informed consent was obtained from all subjects enrolled in the study. The study was in accordance with the Helsinki Declaration, as revised in 2013.
The Authors would like to thank Diesse (Diagnostica Senese, Siena, Italy), Snibe (Diagnostic, Shenzhen, China), Mindray (Medical, Shenzhen, China), Euroimmun, (Lübeck, Germany) for kindly having provided kits and Diesse for having performed live virus neutralization test (VNT) for this study, and Giada Mattiuzzo (National Institute for Biological Standards and Control, UK) for discussion and critical reading of the manuscript.
This research received no external funding.
The authors declare that they have no conflict of interest. AA, ER are serving as the Editorial Board members of this journal. We declare that AA and ER had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to AP.
Supplementary material associated with this article can be found, in the online version, at https://www.imrpress.com/journal/FBL/27/2/10.31083/j.fbl2702074.


