Frontiers in Bioscience-Landmark (FBL) is published by IMR Press from Volume 26 Issue 5 (2021). Previous articles were published by another publisher on a subscription basis, and they are hosted by IMR Press on imrpress.com as a courtesy and upon agreement with Frontiers in Bioscience.
1 Laboratorio de Especiacion Quimica de Aerosoles Organicos Atmosfericos y Desarrollo de Tecnologias Verdes, Departamento de Ciencias Ambientales, Centro de Ciencias de la Atmosfera, Universidad Nacional Autonoma de Mexico, CP 04510, Ciudad de Mexico, Mexico
2 Departamento de Inmunologia, Instituto de Investigaciones Biomedicas, Universidad Nacional Autonoma de Mexico, AP 70228, Ciudad de Mexico, CP, 04510, Mexico
3 Laboratorio de Genotoxicologia y Mutagenesis Ambientales, Departamento de Ciencias Ambientales. Centro de Ciencias de la Atmosfera, Universidad Nacional Autonoma de Mexico, CP 04510, Ciudad de Mexico, Mexico
Abstract
Phthalates are endocrine disrupting compounds (EDCs) used as plasticizers in a wide array of daily-use products, from flooring and automotive parts to medical devices and are even present in the children´s toys. Since these compounds are not covalently bound other molecules, they leach from these synthetic products, causing a high level of human exposure to them. EDCs exert several endocrine effects, most typically, reduced biosynthesis of the male hormone, testosterone and disturbances in estrogen, androgen, PPAR-gamma and AhR that control complex immunoendocrine regulatory networks. Besides impacting the developmental processes and long-term adverse effects, since cells of the immune system express endocrine receptors, and synthetize and respond to several hormones and other endocrine ligands, phthalates also cause dysregulation of immune system.
Keywords
- Phthalates
- Immunomodulation
- Immune
- Endocrine
- Disruptor
- EDC
- Review
Phthalates are considered endocrine disrupting compounds (EDCs), which are molecules that interfere with hormonal homeostasis. Particularly, phthalates have been shown to interact with several receptors involved in estrogenic and androgenic pathways (1). It is important to note that even though the main target of EDCs is the endocrine system, they may affect other tissues. One of such tissues are cells from the immune system, which are greatly susceptible to endocrine modulation. This relationship was first recognized with epidemiological studies that found a higher incidence of some autoimmune diseases -such as rheumatoid arthritis and systemic erythematous lupus- in the female population (2, 3). Later, it was demonstrated that sex steroids regulate multiple immunological functions like lymphocyte maturation, cellular migration, expression of MHC molecules, and cytokine production and that this regulation is mediated by hormonal receptors expressed by immune cell populations (4).
Though some immunological effects of EDCs have been revised previously (5), this review aims to summarize the current evidence of immune dysregulation caused specifically by phthalates; and linking them with the acknowledged molecular mechanisms under an immunoendocrine perspective.
Phthalates are alkyl diesters of phthalic acid and are used as plasticizers in PVC products, as solvents and fixatives in personal care products and as additives in the enteric coating of some drug tablets. Thus, phthalates are present in a wide array of daily-use products, from flooring and automotive parts to medical devices and even children´s toys. Since these compounds are not covalently bound to the products, they easily migrate from them (6), leading to high human exposure.
Phthalate exposure has been estimated by biomonitoring studies and ranges between 1 and 2 ug/kg bw/day of a single compound for adults (7, 8) and up to 4 ug/kg bw/day for children (9). While oral consumption of contaminated food and dermal absorption from personal care products are considered the main exposure routes (10–12), phthalates are ubiquitously found in environmental matrices such as in the atmosphere, soil and water bodies (13). Of particular interest is the presence of phthalates in the atmospheric aerosol, forming part of the organic content of fine particulate matter (PM2.5). Phthalate concentrations can range from 1 to 100 ng/m3 outdoors (14, 15) and up to 1,000 ng/m3 indoors (16, 17), being Bis-(2-ethylhexyl) phthalate (DEHP) and Di-n-Butyl phthalate (DBP) the most common.
An Endocrine disruptor compounds is defined by the United States Environmental Protection Agency (EPA) as an “exogenous agent that interferes with synthesis, secretion, transport, metabolism, binding action, or elimination of natural blood-borne hormones that are present in the body and are responsible for homeostasis, reproduction, and developmental process” (18). EDCs mimic endogenous hormones and bind to their receptors inducing functional and regulatory activities of gene expression.
Phthalates exert sexually dimorphic effects, affecting in different ways to male and female individuals. Bases on epidemiological data, elevated levels of DEHP urinary metabolites were linked to female patients experiencing premature thelarche (19). On the other hand, phthalates effects on males are grouped into the so called “phthalate syndrome”, which is a type of testicular dysgenesis featuring cryptorquidism hypospadias, incomplete testicular descend, reduced ano-genital distance, low sperm count and/or quality, elevated risk of infertility and testicular cancer (6, 20–22), caused by a reduced testosterone synthesis.
Phthalates are readily metabolized once in the organism, leading to the urinary and fecal excretion of their metabolites. The first metabolic phase is an enzymatic hydrolysis of one of the side chains, which turns the original diester into a monoester (e.g. DEHP into MEHP). Phase I is catalyzed by esterases, which are present in blood plasma and most tissues (23). It is important to note that phthalate monoesters can also exert biological effects and maintain binding potential to some receptors (24–27). After this first phase, the monoesters can be further transformed by hydroxylation and/or carboxylation of the remaining side chain, before being further metabolized to glucuronide conjugates in phase II (23).
The biological action induced by phthalates differ from the target cell in which they have an effect. The best described mechanisms involve signaling through the hormone receptors: estrogen (ER) and androgen (AR) receptors; and by the peroxisome proliferator-activated receptor gamma (PPAR-gamma), although other non-transcriptional mechanisms have been described, such as the activation of G protein-coupled receptors (Figure 1).
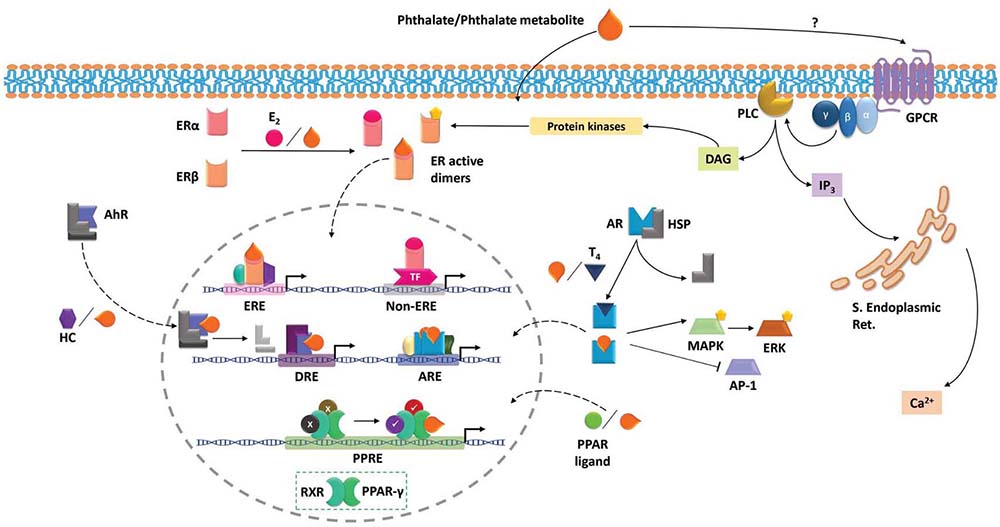 Figure 1
Figure 1Molecular signaling induced by phthalates. Phthalates and some phthalate metabolites may exert fast non-transcriptional effects by engaging and activating G protein-coupled receptors (GPCR), which activate downstream pathways that lead to intracellular calcium increases and activation of diverse protein kinases. Phthalates and their metabolites readily cross cell membranes, entering the cytoplasm. Once in the cytoplasm, they may bind to estrogen receptors (ER) and induce their dimerization and therefore their activation, leading to transcriptional activity, which may vary depending on the existing coregulators. Phthalates may also bind androgen receptor (AR), triggering transcriptional effects and/or regulating the activation of kinases and transcription factors. Aryl hydrocarbon receptor (AhR) may also bind phthalates, enter the nucleus and regulate transcription of responsive genes. Upon nucleus localization, phthalates may also act as Peroxisome proliferation-activated receptor gamma (PPARγ) ligands, leading to transcriptional activity.
Classical nuclear estrogen receptors (ER-alpha and ER-beta) belong to the ligand-activated transcription factor family. These receptors are widely expressed in mammal tissues, some of them expressing ER-alpha (uterus, liver, kidney and heart) or ER-beta (ovary, prostate, hematopoietic and central nervous systems) and some others expressing both (mammary gland, thyroid and adrenal glands and some brain regions). Even though both receptors are highly homologous and display a similar affinity for their natural ligand 17-beta-estradiol and for estrogen response elements (EREs) in the DNA, (28, 29), ligand binding to one receptor or another triggers different responses. The key to such differential responses lays in the signaling pathways of ERs.
Estrogen receptors reside in the cytoplasm and form homo- or heterodimers upon ligand binding, translocate to the nucleus and bind to the EREs in the DNA, typically located near the promoter regions of target genes. This activation leads to the recruitment of coregulators, which associate to the ER dimers, forming the initial transcriptional complex (30). It is important to note that coregulators can either increase a gene’s transcription (coactivator) or inhibit it (corepressor) and that recruitment of one or another depends on the type of ER dimer (ER-alpha or ER-beta homodimers or heterodimer) and, of course, on which coregulators are present in a particular moment in a particular cell type.
The activity of estrogen receptors is not limited to target genes containing EREs. Ligand-activated ERs can modulate gene expression by attaching to other transcription factors which in turn act on their respective response elements (30). In addition to the former, estrogen receptors may also be embedded into the plasma membrane, where they can bind extracellular ligands. Moreover, some non-classical membrane estrogen receptors have been described, such as GPER (or Gpr30), which is a G protein-coupled receptor. These membrane-bound receptors trigger rapid effects such as calcium flux, second messenger production (cAMP, DAG, IP3) and kinase activation (31, 32).
Considering the former, estrogens effects depend not only on the dose, but on the homo- or heterodimers formation, their target (either nucleus or cytoplasmic proteins), the coregulators and active transcription factors existing and all of it varies according to the context and specific cell type.
Similar to estrogen receptors, androgen receptor (AR) has a widespread distribution, from reproductive tissues to muscle, bone, adipose tissue, and cardiovascular, immune and nervous system (33). Endogenous ligands for AR are testosterone and dihydrotestosterone (DHT), a metabolite of the former with a greater agonistic activity.
AR resides in cytoplasm, attached to the cytoskeleton by associating with heat shock proteins or other chaperones. Upon androgen binding, AR suffers conformational changes that dissociate it from the chaperones, which allows it to interact with coregulators and translocate to the nucleus, where it can dimerize and bind to DNA regions denominated androgen response elements (AREs). Once there, AR together with coregulators forms an initial transcription factor that recruits the whole transcription machinery (33, 34).
Androgens can also indirectly modulate genetic expression without binding to AREs. The ligand-AR complex can activate other signaling pathways such as ERK and MAPK or even sequestrate other transcription factors (e.g. AP-1), repressing in this way the expression of their target genes (33).
Phthalates have been also described as peroxisome proliferators, inducing hepatocyte peroxisome and cellular proliferation, which in some studies in mice and rats, have an impact in the incidence of hepatocellular adenomas. This effect is dependent on the activation of the nuclear receptor peroxisome proliferator activated receptor gamma (PPAR-gamma). This receptor belongs to the nuclear receptor subfamily of ligand-inducible transcription factors, which were originally described as key transcriptional regulator in the metabolism of glucose and fatty acids, lipid storage and insulin sensitivity (35–37). In fact, it is one of the main targets in the treatment of metabolic syndrome, insulin resistance and type 2 diabetes (38), since it is highly expressed in white and brown adipose tissues.
Constitutively, PPAR-gamma forms a heterodimer with the retinoid X receptor (RXR) and they bind to specific DNA sequences termed PPAR response elements (PPRE). In basal conditions, PPAR-gamma is bound to corepressor molecules such as the silencing mediator of retinoid and thyroid hormone receptors (SMRT), the nuclear receptor corepressor (NCoR) and some histone deacetylases (HDACs). Upon ligand binding, there is a conformational change in the PPAR-gamma protein that allows the dissociation of its repressors and the recruitment of its coactivators. Among these are the CREB-binding protein (CBP), the histone acetyl transferase (p300) and the PPAR-binding protein 4 (PBP). During adipocyte differentiation, changes in the recruitment of coactivators allow the formation of different transcription complexes that modify the responses to metabolic stimuli and ensure the activation of specific signaling pathways (39).
The AhR was initially described when analyzing the effects of the herbicide 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) induced in exposed chemical workers. This receptor belongs to the family of the bHLH Pas domain transcription factors that are involved in sensing environmental changes. The canonical signaling pathway is induced when its ligand reaches the cytoplasm and bind to the AhR –which in the absence of ligand, is attached to actin filaments by the interaction with the chaperon proteins HSP90, AIP and p23. The ligand-AhR/HSP90/p23 complex then translocate to the nucleus, where it is released from this complex and heterodimerizes with the ARNT protein. This dimer binds to genomic regions containing the dioxin response element (DRE) and induce the transcription of genes such as CYP1A1, CYP1A2, CYP1B1 and the AhR repressor (AhRR). The expression and function of these proteins regulate negatively this signaling pathway by three mechanisms: 1) AhR proteosomal degradation – mediated by the covalently union of ubiquitin-; 2) Ligand degradation by CYP1A1 and 3) AhR/ARNT complex disruption by the repressor AhRR. This repressor is a protein similar to AhR but differs in two characteristics: it lacks its ligand-binding domain and contains a transactivation domain in the C-terminal domain that recruit other co-repressors (40, 41).
In addition to this canonical pathway, the AhR/ARNT complex can recruit other proteins and regulate either transcription or chromatin remodeling. Among these proteins, the SW1/SNF remodeling complex and the steroid receptor coactivator-1 (SRC-1), CBP/p300, p160/bHLH-PAS, NCoA2/GRIP1/TIF2, pCIP (p300/CPB/CoIntegrator associated protein) and RIP140 or the ATP-dependent chromatin remodeling components such as BRG-1 (40, 41).
There are other signaling pathways related to AhR activation. For example, the ligand TCDD increase Ca2+ concentration by activating the Src tyrosine kinase and the Focal adhesion kinase (FAK). These two pathways converge in MAPK activation and therefore, there is also an induced migration, adhesion and inflammation-related functions such as prostaglandin and arachidonic production.
The immune and endocrine systems are part of the neuroimmunoendocrine network, a recently recognized multisystem circuitry that regulates homeostasis. Regarding the immune system, its cellular components not only express endocrine receptors, but synthetize and respond to several hormones and other endocrine ligands. Some molecular pathways typically associated to the endocrine system play a developmental and regulatory role on the immune system, such as the thymus involution through age, the sexual dimorphism in immune response and the tolerogenic character of immune response during pregnancy.
Estrogen receptors are expressed in most immune cell populations, from hematopoietic precursors to mature B and T lymphocytes, NK cells, monocytes and dendritic cells (42). However, expression patterns are different, with some cells expressing preferentially ER-alpha (e.g. CD4+ T cells) and other expressing higher levels of ER-beta (e.g. B cells) (43, 44). In homeostatic conditions, ER-alpha acts on myeloid and lymphoid precursors, inducing developmental pathways (42).
The developmental importance of ER-alpha is depicted by the thymic and splenic hypoplasia observed in ER-alpha-deficient mice (45). ER has a profound effect on T lymphocytes which is sensitive to the dose or physiological levels of estradiol, the natural ligand. A good example is the shift from Th1 response towards Th2 and Treg expansion due to high levels of estrogen, as occurs during pregnancy. B cells are deeply modulated by estrogen, both ER-alpha and ER-beta pathways are involved in maturation, function and survival of this population (42), so that females tend to display a greater humoral response than males (46). Differentiation and maturation of dendritic cells, as well as macrophage effector functions are also modulated by estrogen receptor pathways (44).
AR is expressed in several cell populations from both myeloid and lymphoid origin and has been involved in sexual immunological dimorphism and immunosuppression (47). The majority of the studies regard androgens and and androgen receptor as suppressive or regulatory. AR pathways downregulate T cell and B cell proliferation, as well as the inflammatory response (48). Androgens also modulate the functions of dendritic cells, downregulating expression levels of MHC, HLA and costimulatory molecules, which further modulates T cell response (47). One exception to the predominantly suppressive role of androgens is the neutrophil subpopulation, since androgens (via AR) promotes their differentiation and maturation, probably by enhancing Granulocyte colony-stimulating factor (G-CSF) signaling (48). Though histamine release by mast cells appears to be downregulated by androgens, testosterone has been shown to induce IL-33 expression, which drives the generation of innate lymphoid cells and basophils, which in turn produce Th2 cytokines and drive IgE antibody switch (47). A very interesting observation is that IgE is found to be higher in young males over females affected by allergic rhinitis (49).
PPAR-gamma is expressed in lower amounts in some immune cells such as macrophages, eosinophils, dendritic cells, T and B lymphocytes; however, it has a critical role in both differentiation and activation of these cells in diseases that involve chronic inflammation such as atherosclerosis, inflammatory bowel disease and rheumatoid arthritis (50).
Expression of PPAR-gamma is differentially expressed in different subtypes of macrophages. Resting bone-marrow-derived macrophages express low levels of PPAR-gamma mRNA, whereas activated peritoneal macrophages express high levels of PPAR-gamma (51). However, peritoneal macrophages incubated with 15-dPGJ2 –a PPAR-gamma agonist- showed morphological features typical of resting cells, even in the presence of IFN-gamma. In addition, it also downregulates iNOS expression and inhibit IFN-gamma-dependent nitrite production. This suggests that might inhibit the expression of genes that become upregulated during macrophage differentiation and activation (51). In contrast, other studies report that activation of PPAR-gama primes primary human monocytes into M2 differentiation, but it is not able to modify marker expression of differentiated M1 or M2 macrophages (52).
Eosinophils, an immune cell population related to allergic responses and asthma, express PPAR-alpha, PPAR-gamma and PPAR-delta. In fact, the administration of PPAR-gamma agonists (rosiglitazone or pioglitazone) in a murine model of allergic asthma, induce a reduction in the number and activation state of eosinophils in the airways, determined by the expression of IL-4, IL-5 and ovalbumin-specific IgE, which suggest that PPAR-gamma may be protective factor in the pathogenesis of the asthma (53). This correlate with in vitro experiments in which the presence of high concentrations of PPAR-gamma agonists reduce the eotaxin-dependent eosinophil migration; while low concentrations of this compounds increase migration (54). Interestingly, this effect is mediated by changes in calcium influx, but not by the expression of CCR3 or phosphorylation of p38 or ERK (55).
As mentioned for eosinophils, PPAR-gamma expression in dendritic cells have been related to allergic responses. Particularly, PPAR-gamma deficient CD11b+ dendritic cells fail to migrate to the lung draining lymph nodes and therefore a reduced capacity to polarize naïve CD4+ T cells toward a Th2 phenotype (56, 57). Interestingly, Tuna and collaborators mention that the effect of PPAR-gamma activation depend on the tissue microenvironment. While in the lung it may induce a mucosal phenotype in mDCs and that loss of PPAR-gamma promotes an inflammatory phenotype (determined by the ability of BM-DC to polarize CD4 T cells toward iTregs and to induce CCR9 – a mucosal homing receptor- on T cells); deficiency of this receptor showed no change in the frequency or phenotype of mDC in the colon. This suggest that the intestinal microenvironment can maintain the mucosal DC phenotype of via PPAR-gamma-independent mechanisms (58).
T cells express PPAR-gamma 1, but not PPAR-gamma 2 and it is upregulated on activated T cells. Interestingly, its activation through agonists (such as 15-deoxy-Δ12, 14-prostaglandin J2 or troglitazone) inhibits PMA- mediated proliferation and induce a decrement in cell viability (59). This effect was also described in human and B cell lines. Incubation of B lineage cells with PPAR-gamma against but not with PPAR-alpha agonists, induce apoptosis as demonstrated by the increase of AnnexinV staining and the evaluation of DNA fragmentation in TUNEL assays (60).
Because of its function as an environmental sensor, AhR is widely expressed in the body and the immune system. However, it is highly expressed at the barrier sites such as the skin, lung and gut. In the immune system, the expression of AhR has been described in dendritic cells, B cells and some subpopulations of T cells such as Th17, and in lesser amount, in regulatory T cells (61).
In the T cell subpopulations, AhR is highly expressed in Th17 and in a lesser amount in regulatory T cells (Tregs), but it has not been found in naïve, Th1 or Th2 cells (41). The role of this receptor in Th17 cells remains mostly elusive, but some studies suggest that AhR interacts with NF-κB and STAT1, inhibiting those proinflammatory pathways (62) and promoting the Th17 phenotype by enabling cells to produce high levels of IL-17 and IL-22 (63). Regarding B cells, it has been reported that AhR is expressed in several subpopulations, although at different levels (41). AhR role in B cell function is still not well understood, but an in vitro study reported B cell maturation impairment and antibody production suppression when LPS stimulation occurred in presence of a potent AhR ligand (64). AhR is also expressed by antigen presenting cells, such as dendritic cells, whose differentiation and maturation can be affected by AhR activation. Furthermore, activation of AhR decreases the expression of class II MHC, as well as costimulatory molecules, promoting regulatory T cells differentiation (65).
As summarized in Table 1, most studies about the impact of phthalates in the immune system are developed in murine macrophages either cell lines or in primary cultures of peritoneal exudates. In these cells, it was found that some phthalates such as DEHP, DEP and MEHP promoted a pro-inflammatory state characterized by an increase in the expression and secretion of IL-1, IL-6, TNF-alpha, and the chemokine CXCL1 and ROS (66). Other studies found that the secretion of IL-6, IL-10 and the chemokine CXCL8 was enhanced, while TNF-alpha was impaired by DEP and DnBP presence during stimulation (67). They are also capable of increase the phagocytic index of rabbit lung macrophages, which is accompanied by an increment in the release of lysosomal hydrolases (68). However, Shertzer found that even when the phagocytic index after the infection with S. aureous was increased, there was a reduction in the rate of pathogen destruction; which may impact as a deficient response in subsequent exposures (69). On the contrary, other phthalates such as the DBP, BzBP, DEP and DPrP inhibit the production of IL-1beta, IL-6 and IFN-beta (70, 71), apparently by the inactivation of the IFN-beta promoter (70).
| Country | Cellular type | Immune system component | Phthalate | Findings | Reference |
|---|---|---|---|---|---|
| USA | Primary culture of rat testicle germ cell | Macrophages, IL-6, TNF-alpha and CXCL1 | DEHP and DEP |
The expression of proinflammatory cytokines (IL-6, TNF-alpha and CXCL1) are increased | (66) |
| USA | Rabbit alveolar macrophages | Macrophages | DEHP |
Significant stimulation of phagocytic activity in macrophages pre-exposed to DEHP as well as release of lysosomal hydrolases. | (68) |
| USA | Rabbit lung macrophages | Macrophages | DEHP |
The cultures previously exposed to DEHP, after infection with S. Aureus, show an increase in the phagocytic index and a decrease between 2 and 14 times of the rate of destruction of the pathogen. | (69) |
| Japan | RAW 264 | IFN-beta | BBP, DBP, DOP, DEP, DHP, DPP and DPrP |
Inhibition of IL-1beta and IL-6. Only DEP and DPrP inhibit the LPS-induced activation of IFN-beta promoter. | (70) |
| Japan | Mice (abdominal macrophages) and RAW 264 cell line | TNF-alpha | BBP |
Inhibits the production of TNF-alpha in RAW 264. In abdominal macrophages no response was seen. | (71) |
| Japan | Bone marrow-derived dendritic cells (BMDC) and splenocytes from mouse | Dendritic cells, splenocytes, IL-4 and IFN-gamma | DEHP |
Increases BMDC differentiation but not the activation. Higher IL-4 and IFN-gamma expression on T lymphocytes in response to BMDC differentiation. Increased Th2 response in splenocytes. | (72) |
| China | Primary cultures of mouse splenocytes | IL-4, IL-6 and IFN-gamma | DEHP (1-50 uM) and MEHP (5-80 uM) | DEHP: Increases IL-4 and IFN-γ, decreases the Th1/Th2 ratio (IFN-gamma / IL-4). MEHP: decreases the Th1/Th2 ratio (IFN-gamma / IL-6. | (73) |
| Korea | Primary cultures of mouse splenocytes | B lymphocytes |
DEHP |
Increases B cell proliferation by inducing Ca2+ flux, PKC, ERK1/2 and NF-kappa B activation. | (74) |
| Japan | Mouse thymocytes and splenocytes | Thymocytes (IL-3, IL-4 and IFN-gamma) and splenocytes |
DEHP |
Stimulates the proliferative response in thymocytes and splenocytes. Increases the production of IL-3 in thymocytes, IL-4 and IFN-gamma in Con A-treated thymocytes | (75) |
| USA | Mouse bone marrow-derived B cells and BU-11 cell line | B lymphocytes | MEHP |
Induces apoptosis and suppresses B cell proliferation in developing bone marrow | (76) |
| USA | PD31 cell line (murine pre-B cells) and primary cultures of rainbow trout kidney | B cells, Plasmatic cells and IgM | DEHP |
DEHP: Inhibits B cells proliferation and reduces the number of plasmatic cells secretory of IgM. Accelerates the terminal differentiation of B cells | (77) |
| Japan | Rat basophils RBL-2H3 | Mast cells | DBP, DIBP and DEHP |
Without Ag stimulation, the phthalates don´t cause a significant increase in degranulation. In cultures sensitized with Anti IgE, the three induce degranulation: DIBP > DBP > DEHP. | (78) |
| Korea | RAW 264.7 cell line (murine macrophages) | iNOS, TNF-α and IL-1beta | DCHP, DEP and DBP |
DCHP, DEP, DBP: changes in iNOS mRNA expression. DBP: Suppression of TNF-alpha and IL-1beta transcription | (80) |
| Korea | RAW 264.7 | Macrophages, iNOS | BBP |
Reduces the production of NO via regulation of iNOS mRNA expression | (81) |
| China | Macrophages of murine peritoneal exudate | IL-1β, IL-6, IL-10, IL-12 and TNF-alpha | DBP |
Inhibition of IL-1beta, IL-6 and TNF-alpha production. No change in IL-10 and IL-12 synthesis | (82) |
| Norway | (RAW 264.7 | TNF-alpha | MEHP |
Increases TNF-alpha and changes the cellular morphology of macrophages to a more elongated form, reminiscent of differentiated M2 macrophages | (83) |
| Japan | Peripheral blood mononuclear cells (PBMC) of mice | Mature and immature DCs | DEHP and MEHP |
DEHP inhibits the expression of DC differentiation and maturation markers. MEHP has no effect. | (84) |
| Norway | Primary cultures of rat alveolar macrophages | TNF-alpha, ROS, CXCL2 and leukotriene B4 (LTB4) | MEHP |
MEHP increases the production TNF-alpha, ROS, CXCL2 and (LTB4) | (85) |
| Finland | RAW 264.7 | ROS production and cell death | DEHP, DBP, BBP and DIBP |
Increased ROS production. Only DBP and DBIP caused cell death due to necrosis at their highest doses (effect>with DIBP). | (86) |
| USA | primary cultures of murine cells | IL-4, IFN-gamma and IgE | DEHP and DINP |
By themselves they don´t induce the production of IL-4 and IFN-gamma in mouse lymph nodes. In stimulated cells with PMA or metalloproteins an increase in IL-4 is observed. | (87) |
| UK | Human and rat neutrophils | CD11b (lymphocyte activation marker) | DEHP |
DEHP initiates inflammatory response in human and rat neutrophils (by expression of CD11b) | (88) |
| Japan | Carp leukocytes | Leukocytes and kidney macrophages | DEHP and DBP |
Both regulate the function of carp phagocytic cells | (89) |
| UK | Rat peritoneal leukocytes | Leukocytes | DEHP, MEHP and DBP |
Inhibition of the formation of prostaglandins and leukotrienes (PGE-2 y LTB4) in leukocytes. | (90) |
In T cells, phthalates seem to promote the polarization to the Th2 phenotype. Hansen et al. showed that 100 uM of DEP and DnBP were capable to suppress the secretion of IL-2 and IL-4, TNF-alpha and IFN-gamma, with no impact in cell viability. It was found that DEHP stimulation might also increase the expression of TCR and CD3 molecules and antigen-induced proliferation (72). In total splenocyte cultures, DEHP significantly increased IL-4 and IFN-gamma concentrations, while MEHP increased IL-6 in supernatants. Interestingly, the effect of DEHP was mediated by the activation of the Ca2+/CaN/NF-AT signaling pathway, which was not observed with MEHP. These data suggest that both phthalates may induce the same outcome but by altering different cellular mechanisms (73). DEHP also induces B cell proliferation; activate Ca2+ flux, membrane PKC translocation, ERK1/2 phosphorylation and NF-kappa B activation after 2 hrs (74).
In murine thymocytes, DEHP also increases B cell proliferation as demonstrated by Yamashita et al. (75). In contrast, MEHP was found to suppress B cell ((3)H) thymidine incorporation in both bone marrow-derived B cells and the B cell line BU-11 at doses of 25 or 100 uM. This was explained by an increase in B-cell apoptosis at high doses and an apparent arrest without death at low doses (76).
Interestingly, these alterations are also seen in other organisms such as the rainbow trout, in which, in vitro DEHP exposition of B cells inhibited in a dose-dependent way the proliferation of these cells. Also, authors found that there were changes in the differentiation that generates deficient plasmablast expansion and a reduced number of IgM-secreting plasma cells (77).
Regarding to other immune cell types, there is also evidence in which the effect of some phthalates modifies the function of bone marrow-derived dendritic cells (BM-DC) and basophils. In DC’s, the presence of 10 uM of DEHP during the differentiation of BM-derived macrophages increased the expression of maturation markers such as MHC-II, CD80/86, CD11c and DEC205. It also increased their capacity to induce secretion of IFN-gamma, IL-4 and IL-10 by T cells. This effect was also observed when DC’s were differentiated in normal conditions and DEHP was added during the activation. Interestingly, at 100 uM of DEHP during activation, the percentage of maturation/activations markers were reduced and their antigen presenting activity is not affected as observed by the reduced proliferation of co-cultured T cells. Authors suggest that the enhancement on these markers and function might contribute to the aggravating effect of DEHP on allergic disorders, which also may be useful for the treatment in this kind of pathologies (72).
Finally, since air pollutants have been related to airway diseases, the study of immune cell populations related to allergies has gained relevance. The RBL-2H3 cells, a basophil-derived cell line, which has been described also as a mast cell model, was exposed to different phthalates -DPB, DiPB and DEHP- at concentrations of 50-500 uM. Authors described that in the absence of antigen stimulation, none of those compounds induce β-hexosaminidase release. However, when cells were pre-sensitized and then stimulated with an antigen, all of the phthalates increased the degranulation of these cells (78). Another report found an interest correlation with the number of carbon atoms present in the phthalates and the histamine-release of human basophils. In this work, PMBC’s were incubated in the presence different phthalates and the stimulated with any of the following stimuli: anti-IgE, the bacterial derived peptide fMLP, a calcium Ionophore or an allergen (cat hair extract). As described before, there was no effect on the histamine release in the absence of activation. Interestingly, only the phthalates and monophthalates with an 8-carbon atoms alkyl chain length were the stronger histamine-release potentiators, when there were no differences for the 4-, 9- or 10-carbon chain (79).
Some studies have also assessed the effect of phthalates on human cell lines and primary cultures (Table 2). As in the former studies, heterogeneity in exposure times and concentrations is observed, but some general features can be distinguished. It is interesting to note that, in general terms, a more reactive or proinflammatory response is observed in human cells in vitro exposed to different phthalates, regardless of the heterogeneity of models and exposure schemes.
| Country | Cellular type | Immune system component | Phthalate | Findings | Reference |
|---|---|---|---|---|---|
| Denmark | Monocytes/Macrophages and T cell (peripheral blood mononuclear cells) | Monocytes/Macrophages | DEP, DnBP and MnBP |
DEP and DnBP increase IL-6, IL-10 and CXCL8. They decrease the synthesis of TNF-α. T cells: decrease in IL-2, IL-4, TNF-alpha and IFN-gamma expression. | (67) |
| Denmark | THP-1 and peripheral blood mononuclear cells (PBMC) of allergic and healthy individuals | THP-1: IL-1β, IL-6, IL-12α PBMC: IL-4, IL-5 and IFN-alpha | MBUP, MBEP, MEHP, MOP, MINP and MIDP. |
Mono-phthalates don´t increase the cytokine production in TPH-1 alone but increase it upon LPS stimulation. There seems to be a relationship between the length of the side chains and the toxicity. MBUP tends to increase the levels of IL-4 in PBMC of allergic patients | (79) |
| UK | Human and rat neutrophils | CD11b (lymphocyte activation marker) | DEHP |
DEHP initiates inflammatory response in neutrophils (by expression of CD11b) of humans and rats | (88) |
| Slovak Republic | THP-1 | TNF-alpha, IL-1beta, IL-8 | DiP |
At high doses (10 uM), increases IL-8 secretion | (91) |
| France | THP-1cell line (differentiated macrophages) | IL-1β, TNF-α and IL-8 (secretion of cytokines induced by LPS) | DEHP and DBP |
DBP: increased expression of TNF-alpha | (92) |
| USA | THP-1 and placental macrophages (PM) | COX-2 and prostaglandin E2 | MEHP |
MEHP: Increases the release of PGE2 via increased expression of COX2 | (93) |
| Portugal | Peripheral blood-derived M1 and M2 macrophages | IL-1beta, IL-6 and IL-10 | DEHP and DBP |
M1 macrophages: DEHP—>Increase the expression of IL-1beta, IL-10 and decrease IL-6. DBP—> Increase the expression of IL-1β is not affected. IL-10 and IL-6 the same DEHP. M2 macrophages: DEHP—>IL-1beta is not affected. IL-10 and IL-6 decrease. DBP—>Increase the expression of IL-1beta. IL-10 and IL-6 decrease. | (94) |
| Japan | Peripheral blood leukocytes | Granulocytes | DEHP |
Dose-dependent reduction of chemotaxis and bactericidal. Cell count and phagocytic activity aren´t modified. | (95) |
| Italy | Primary cultures of granulocytes isolated from blood | Chemotherapy and ROS production | DEHP |
Stimulates chemotaxis and ROS production | (96) |
| Puerto Rico | TK6 cell line (Lymphoblasts) | Mitochondrial membrane permeability, ROS, Caspases 3 y 7 | DEHP and MEHP |
Both compounds affect the viability of lymphoblasts by increasing the permeability of the mitochondrial membrane, generating ROS and activating Caspases 3 and 7. | (97) |
| Norway | Peripheral blood mononuclear leukocytes | Leukocytes | TBEP and DEHP |
Affects the binding of beta-adrenergic ligand in mononuclear leukocytes (interacts with specific and nonspecific binding sites). | (98) |
| Germany | Peripheral blood leukocytes | Leukocytes | DBP and DiBP |
Genotoxicity: DiBP >DEHP | (99) |
| Germany | Peripheral blood lymphocytes | Lymphocytes | MEHP |
Genotoxicity | (100) |
| USA | Primary cultures of umbilical cord blood | IL-1beta, IL-8, IL-6 and VEGF | MEHP |
Inhibits neutrophil apoptosis (neonatal cells are more sensitive than adults). In adults and neonates increases the production of VEGF and IL-1beta, only increases IL-8 in adult neutrophils. IL-6 is not affected. | (101) |
| Korea | HMC-1 cell line (mast cells) | TNF-alpha and IL-4 | DEHP |
Increases the expression of TNF-alpha and IL-4 | (102) |
| Japan | THP-1 | TNF-alpha, IL-1beta, IL-8 and IL-6 | DEHP |
Increases TNF-alpha, IL-1b, IL-8 and IL-6 secretion. Increases the mRNA expression levels of IL-8, CXCL1, CXCL2, CXCL3, CXCL6, CCL3, MMP10, CSF2, TNF-a, IL-1beta and IL-6. Induces p65 NF-kappa B translocation to the nucleus. | (103) |
| Taiwan | pDCs (Plasmacytoid dendritic cells), serum | IFN-alpha, IFN-beta, IFN-gamma and IL-13 | DEHP and BBP |
DEHP and BBP: suppress the expression of IFN-alpha, and IFN-beta. BBP: Suppresses the expression of IFN-gamma but increases the IL-13 (for T cell CD4+) | (104) |
| Taiwan | MDA-MB-231 cell line (adenocarcinoma) | mdDCs and TADCs (CXCL1) | BBP (1 and 10 uM) | BBP stimulates the production of CXCL1 by the TADCs | (105) |
Studies with THP, a macrophage cell line seem to concur in that phthalates and their metabolites increase the cytokine and chemokine production (79, 91, 92). Tetz and cols. Also observed a proinflammatory effect of phthalate exposure, with activation of COX pathway and increased prostaglandin production (93). An interesting study by Teixeira noted that the effect of phthalate exposure showed differential features, depending on the macrophage phenotype, either M1 or M2 (94).
One of the first studies that documented the effect of phthalates on immune cells was derived from the concern of donated blood being storage in plastic bags; this study found decreased chemotaxis and bactericidal capacity of neutrophils (95). However, more recent studies using human granulocytes have found rather an increase in chemotaxis, IL-8 (96, 97), and promotion of inflammatory response (88).
Mast cells are of particular interest, given their role in allergic disease. Lee and cols. Reported an increase in IFN-gamma and IL-4 production upon DEHP exposure, which is linked to a more reactive phenotype. However, more studies regarding mast cells differentiation, migration and function are desirable.
Given the complex multidirectional regulation that occurs between immune and endocrine system, it is clear that, although they offer valuable information, in vitro studies don´t fully represent what occurs in a whole organism, and it is why in vivo studies are of such importance. Data from the full literature review is detailed in Table 3.
| Country | Organism | Immune system component | Phthalate | Findings | Reference | |||
|---|---|---|---|---|---|---|---|---|
| China | Mice | IgE, IgG1, Tfh (follicular T helper), Plasmatic cells, IL-21 and IL-4 | DEHP |
DEHP acts on Thf cells by increasing its production of IL-21 and IL-4, which leads an increase of IgE and IgG1 secretion. | (106) | |||
| Denmark | WT and PPAR-α-deficient mice | IgG1, IgE and IgG2a | DEHP |
It induces increased levels of IgG and IgG2a in both strains of mice | (107) | |||
| Denmark | Mice | IgG1, IgE and IgG2a | DEHP |
DEHP is capable of increasing serum IgG1 levels and inflammatory cell levels in the lungs but only at very high concentrations | (108) | |||
| Denmark | Mice | IgG1, IgE, IgG2a, eosinophils and lymphocytes (serum, bronchoalveolar fluid and draining lymph nodes) | MEHP |
Increase in IgG1. > number of eosinophils and lymphocytes in bronchoalveolar fluid | (109) | |||
| Korea | Mouse | Cytokines, immunoglubulines, lung inflammation | DINP |
DINP suppresses Th1 polarization and promotes Th2. In asthmatic mice, DINP increases IL-4, IL-5, IgE, IgG1, decreases IgG2a and IFN-gamma (in serum) and induces lung inflammation s (Caspases 1 and 3 increased) | (110) | |||
| Sweden | Mice | IgE, IL-4, IFN-gamma | DEHP |
In sensitized mice, serum IgE increases in a dose-dependent manner. IL-4 in lung tissue increases in sensitized mice exposed to the highest DEHP dose | (111) | |||
| USA | Salmon | B lymphocytes | DEHP, DMP, DBP, BBP, DEP, and DnOP (from plastic-polluted lake) | DEHP accelerates the differentiation towards the plasmatic cells (secretory of Ab) | (112) | |||
| China | Mice | IgE, IFN-gamma (Th1), IL-4 (Th2), IL-17A (Th17), TNF-alpha, IL-5 and IL-13 | DBP |
In lesions due to atopic dermatitis, the exposure to DBP increases Th2 and Th17 cytokines | (113) | |||
| Japan | Mice with induced allergic rhinitis | IL-5, IL-6, IL-12 and IL-13 | DEHP |
Increased levels of nasal IL-13 in mice treated with the allergen +DEHP | (114) | |||
| Japan | Mice | CCL17, CCL22, IL-4 | DINP |
DINP increases the production of CCL17, CCL22 and IL-4 in induced atopic dermatitis | (115) | |||
| Japan | Mice | IL-4 | DBP and DPP (80 ul, together with acetone, as dermal vehicle) | Increased FITC+ DC in lymph nodes. Increased IL-4 in draining lymph nodes after skin sensitization. | (116) | |||
| Japan | Mice with induced contact sensitivity | TSLP (Th2 response cytokine) and IL-4 | DBP |
Increases the expression of TSLP in sensitized skin. DBP is essential for sensitization with FITC. DBP induces migration of Ag presenting cells | (117) | |||
| USA | Mice | TSLP (cytokine related to IL-7) produced by epithelial cells | DBP |
The epicutaneous application of DBP induced the expression of TSLP in the skin | (118) | |||
| Japan | Mice | Macrophages and DC | DBP and DPP |
In the presence of these phthalates, a number of antigen-presenting DCs are observed in the lymph nodes (they migrate from the skin, presenting FITC) | (119) | |||
| Taiwan | Mice | Macrophages, TNF-alpha, IL-1β and IL-6 | DEHP |
Promotes inflammation by increasing serum of TNF-alpha, IL-1β and IL-6. It also promotes the infiltration of macrophages in white adipose tissue. | (120) | |||
| USA | Rats | Macrophages | MEHP |
Induces the infiltration of CD11b+ macrophages in testis of immature peripubertal rats, triggering germ cell apoptosis. No infiltration observed in adult rats. | (121) | |||
| Japan | Mice | Lymphocytes, macrophages, IL-10 and IFN-gamma | DEHP |
Greater presence of lymphocytes and macrophages in testis, along with increased expression of IL-10 and IFN-gamma mRNA. | (122) | |||
| China | Rats | IL-1beta (of testicular macrophages) | DBP |
Increases the expression of IL-1beta mRNA (and suppresses the production of testosterone) | (123) | |||
| Japan | Mice | IFN-gamma | DEHP |
Increases the expression of IFN-gamma and the infiltration of leukocytes in autoimmune testicular inflammation. | (124) | |||
| New Zealand | Mice | Dendritic cells | DBP |
Stimulation of Th2 response | (125) | |||
| China | Zebrafish | Macrophages | DBP |
Decrease in macrophage formation. | (126) |
|||
| Japan | Mice with induced peritonitis | Macrophages, Neutrophils and IL-5 | DEHP |
Exaggerates inflammation by increasing macrophages and neutrophils and increases IL-5 and eotaxin production. | (127) | |||
| Netherlands | Rats | TNF-alpha | DEHP |
The exposure to DEHP increases TNF-alpha levels in splenocytes of adult rats (stimulated with LPS) | (128) | |||
| Japan | Epicuta-neously sensitized mice | IL-4, IFN-gamma (from draining lymph nodes) | DEHP and DBP |
DBP: Reduction of IL-4. DEHP: Reduction of IL-4 and increase of IFN-gamma | (129) | |||
| Japan | OVA-immunized mice | Splenocytes and thymocytes | DEHP |
It has no effect on the production of Ab. Stimulates the proliferation of splenocytes treated with Con A (mitogen for T cells) and of the thymus cells treated with Con A. | (130) | |||
| USA | Rats | Kupffer cells | DEHP |
An increase in free radicals in the bile of rats is observed after intragastric administration of DEHP. The increase in free radicals is due to the activation of Kupffer cells. | (131) | |||
In general terms, phthalate exposure leads to higher immunoglobulin levels. In some models, an increase of IgG -mainly IgG, has been observed (106–110), while in sensitized animals is the IgE class which is augmented (111). For one of the most studied phthalates – DEHP, the increase in IgE seems to be mediated by the promotion of differentiation of B cells into plasma cells (112) and by the stimulation of follicular T helper cells (Thf), producing higher IL-21 and IL-4 (106).
In diverse pathologies that are characterized by hypersensitivity, such as asthma, dermatitis and rhinitis, phthalate exposure has been shown to increase Th2 (DBP, DPP, DEHP and DINP) and Th17 cytokines (DBP) (110, 113, 114). In animal asthma models, the most studied phthalates -DEHP and DINP- have been shown to increase pulmonary inflammation, antibody production (specially IgE), and IL-4 production. This Th2 polarization has also been observed in an allergic rhinitis model, where DEHP exposure leads to higher IL-13 levels in the nasal epithelium (114). In cutaneous hypersensitivity models, DBP, DPP and DINP have been shown to promote Th2 response, as denoted by a high IL-4 production (115, 116).
Moreover, phthalate exposure also stimulates the production of chemoattractant molecules, such as TSLP, CCL17 and CCL22 (115, 117, 118), which coincides with another common finding: a higher recruitment of antigen presenting cells like dendritic cells and macrophages. This phenomenon, known as the adjuvant effect, has been observed in skin and other tissues, including adipose tissue, and for various phthalates (119, 120).
One of the most typical target organs regarding phthalate toxicity is the male gonad. The effect of phthalates on this tissue has been mainly attributed to pure endocrine mechanisms, but it seems that immunomodulatory mechanisms may as well play an important role. It has been observed that exposure to some phthalates, fundamentally DEHP, causes an increased infiltration of macrophages and other leukocytes in the testis, together with a higher expression of proinflammatory cytokines, such as IL-1β and IFN-gamma, which leads to germinal cell apoptosis and lower testosterone levels (121–124).
Epidemiological studies, albeit heterogeneous, support the immunomodulatory effects of phthalates, particularly due to prenatal exposure. Data from the full literature review is detailed in Table 4. A 70% higher risk of asthma diagnosis at age 5-11 was observed in children born to women with higher urinary levels of BBzP and DnBP metabolites during their pregnancies (132). Another study, carried out in a Spaniard cohort, also found an association between asthma diagnosis at age 7 and maternal urinary levels of MBzP and the sum of DEHP metabolites (133). In a similar fashion, a study in Taiwan reported an association between high maternal DEHP exposition and asthma incidence, particularly in boys (134). In a French cohort, a study found a correlation between maternal urinary concentrations of DiBP and DiNP metabolites and eczema occurrence; interestingly, this association was stronger for boys (135).
| Country | Biological sample | Study cohort | Immune system component | Phthalate | Findings | Reference |
|---|---|---|---|---|---|---|
| USA | Urine of pregnant inner-city women | CCCEH (1998-2006) | Asthma diagnosis in children between 5 and 11 years old | BBzP, DnBP, DEHP, DEP | Risk of asthma was > 70% higher among children with maternal prenatal BBzP and DnBP metabolite concentrations in the third versus the first tertile. | (132) |
| Spain | Urine of pregnant women | INMA (2004-2008) | Atopy and asthma in progeny | Phthalate metabolites | Sum of DEHP metabolites, as well as MBzP during pregnancy increase the risk of asthma at age 7 | (133) |
| Taiwan | Urine of pregnant women and children | Taiwan Maternal and Infant Cohort Study (2000-2009) | Asthma occurrence in progeny | Phthalate metabolites | Prenatal and postnatal exposure to phthalate was associated with the occurrence of asthma in children, particularly for boys | (134) |
| France | Urine of pregnant women | EDEN (2003-2006) | Development of Eczema in progeny | DiBP and DiNP metabolites | Occurrence of eczema in childhood is probably influenced by prenatal exposure to phthalates, in boys | (135) |
| Canada | Umbilical cord serum | MIREC (2008-2011) | IgE and IL-33 | 11 phthalate metabolites | Inverse non-linear associations between maternal urinary MCPP levels and elevated of IgE and IL-33 levels | (136) |
| Taiwan | Serum for children at 2 and 5 years | Ad hoc: from medical centers, regional hospitals, local hospitals, and clinics in Taiwan in 2004 | IgE | MEP, MEHP, MBP and MBzP | Phthalate levels positively correlated with serum IgE levels only in boys. | (137) |
| USA | Urine | NHANES (2005-2006) | Allergic symptoms and sensitization | Phthalate metabolites | MBzP positively associated with allergic symptoms in adults. Mono-(3-carboxypropyl) phthalate and the sum of DEHP metabolites positively associated with allergic sensitization in adults. Phthalate metabolites inversely associated with asthma and hay fever in children. | (138) |
| Austria | Serum of healthy and allergic to home dust individuals | 16 healthy and 16 HDM-allergic subjects | G-CSF, IL-5 and IL-6 | DEHP | On allergic subjects, low concentrations of DEHP in the dust increased G-CSF, IL-5 and IL-6, while high concentrations of DEHP decreased G-CSF and IL-6. | (139) |
At the molecular level, Ashley-Martin didn’t find an association between maternal MCPP (a nonspecific secondary metabolite) and levels of IgE, nor IL-33 in neonates (136). However, another study where MEP, MEHP, MBP and MBzP were measured, found a positive association between maternal urinary levels of those metabolites and IgE levels of children at age 2 and 5 (137). Interestingly, this association was only significative in boys.
Regarding current exposure in adults, the NHANES 2005-2006 study found an association between urinary levels of high molecular weight phthalate metabolites and allergic sensitization (defined by the response to at least 19 specific IgE antigens) (138). Furthermore, MBzP concentration correlated with the presence of allergic symptoms at the moment of the sampling. However, this same study found that in children there was an inverse relationship between urinary concentrations of phthalate metabolites and the incidence of asthma and hay fever. In another study, in adults allergic to dust mite, inhalation of DEHP-containing dust modified the expression of G-CSF, IL-5 and IL-6 in a differential fashion, depending on the concentration (139). At low levels, those cytokines increased, while at high concentrations both G-CSF and IL-6 decreased. It is interesting to note that, similarly to what is observed with some hormones, the dose-response relationship seems to be non-monotonic.
The immune system is regulated by a complex crosstalk between its own soluble mediators and several endocrine pathways. Immune cells not only express endocrine receptors but synthetize and respond to several hormones and other endocrine ligands. In this context, perturbations in the endocrine homeostasis may lead to immune dysregulation. Regarded as EDCs, phthalates and their metabolites interact with estrogen, androgen, PPAR-gamma and AhR pathways, and there is vast in vitro evidence of the effects these compounds exert on immune cells. Furthermore, reports on animal models confirm these phenomena occur in a whole organism setting. However, epidemiological evidence is the one that raises the more concerns, since it points out two relevant aspects of immune dysregulation associated to phthalates: developmental and long-term effects.
Taken together, the evidence suggests more consciousness should arise regarding the use of phthalates, especially in daily use personal care items intended for its use by vulnerable populations. We hope the recognition of the immunoendocrine modulation network and the role it plays from early developmental stages will lead a more integrative perspective in toxicological studies. Regarding phthalates, more studies on immunological imprinting and potential neurological effects are desirable, since this system is also profoundly modulated by endocrine signaling.
This work was supported by Grant No. IA202919 from Programa de Apoyo a Proyectos de Investigación e Innovación Tecnológica (PAPIIT) and Grant No. FC-2016-2125 from Consejo Nacional de Ciencia y Tecnología. Margarita Isabel Palacios-Arreola is a postdoctoral fellow from Dirección General de Asuntos del Personal Académico, UNAM.
EDCs
Endocrine disrupting compounds
Peroxisome proliferator-activated receptor gamma
Aryl hidrocarbon receptor
Major histocompatibility complex
Polyvinyl chloride
micrograms
body weight
Fine particulate matter (of less than 2.5 micrometers)
Bis-(2-ethylhexyl) phthalate
Di-n-butyl phthalate
Environmental Protection Agency
Mono(2-ethylhexyl) phthalate
Estrogen receptor
Androgen receptor
Estrogen response elements
Deoxyribonucleic acid
Estrogen receptors
G protein receptor-coupled estrogen receptor
Cyclic adenosine monophosphate
Diacylglycerol
Inositol triphosphate
Dihydrotestosterone
Androgen response elements
Extracellular signal-regulated kinase
Mitogen-activated protein kinase
Retinoid X receptor
PPAR response element
silencing mediator of retinoid and thyroid hormone receptors
nuclear receptor corepressor
histone deacetylases
CREB-binding protein
PPAR-binding protein 4
2,3,7,8-tetrachlorodibenzo-p-dioxin
basic helix-loop-helix
heat-shock protein 90
Aryl hydrocarbon receptor-interacting protein
Aryl hydrocarbon nuclear translocator
Dioxin response element
Cytochrome P450, family 1, subfamily A, polypeptide 1
AhR receptor
SWItch/Sucrose Non-Fermentable remodeling complex
steroid receptor coactivator-1
focal adhesion kinase
natural killer cells
T helper cells
Regulatory T cells
human leukocyte antigen
Granulocyte colony stimulating factor
interleukin
immunoglobulin E
15-deoxy-Δ12,14-prostaglandin J2
Interferon
Inducible Nitric oxide synthase
macrophage phenotype 1 or 2
cluster of differentiation
Bone marrow-derived dendritic cells
Induced Tregs
C-C chemokine receptor type 9
mature dendritic cells
phorbol myristate acetate
Terminal deoxynucleotidyl transferase dUTP nick end labeling assay
Diethyl phthalate
tumor necrosis factor
C-X-C chemokine ligand 8
Di-n-butyl phthalate
Benzylbutyl phthalate
Dipropyl phthalate
T cell receptor
calcium
calmodulin
Nuclear factor of activated T-cells
protein kinase C
nuclear factor kappa B
Diisobutyl phthalate
peripheral blood mononuclear cells
N-formyl-methionyl-leucyl-phenylalanine
cyclooxygenase
follicular T helper cells
D-n-propyl phthalate
Diisononyl phthalate
Thymic stromal lymphopoietin
Benzylbutyl phthalate
Monobenzyl phthalate
Diisononyl phthalate
Mono (3-carboxypropyl) phthalate
Monoetyl phthalate
Monobutyl phthalate
concanavalin A
Diisobutyl phthalate
Carboxi-monoisodecyl phthalate
Diallyl phthalate
Dicyclohexyl phthalate
Di-n-hexyl phthalate
Diisoheptyl phthalate
Diisohexyl phthalate
Diisononyl phthalate
Diisopentyl phthalate
Di-n-octyl phthalate
Di-n-pentyl phthalate
Di-n-pentyl phthalate
Ditridecyl phthalate
Monobenzyl phthalate
Mono-n-butyl phthalte
Monoisodecyl phthalate
Monoisodecyl phthalate
Monoisononyl phthalate
Mono-n-butyl phthalate
Mono-n-octyl phthalate
Monotridecyl phthalate
Reactive oxygen species
leukotriene B4
prostaglandin 2
Signal transducer and activator of transcription
Tris-(2-butoxyethyl)-phosphate

