1 Department of Life Science and Engineering, Foshan University, 528231 Foshan, Guangdong, China
2 Department of Animal BioSciences, University of Guelph, Guelph, ON N1G 2W1, Canada
Academic Editor: Graham Pawelec
Abstract
Background: Female infertility is a health issue for both humans and
animals and despite developments in medical interventions, there are still some
conditions that cannot be treated successfully. It is important to explore other
potential therapies or remedies that could improve reproductive health. Choline
is an over-the-counter supplement and essential nutrient that has many health
benefits. It has been suggested to be
beneficial in various aspects of fertility, including fetal development and
endocrine disorders like polycystic ovarian syndrome (PCOS). However, choline’s
impact on ovarian function has not been explored. Methods: To study the
effects of choline on ovarian development, 36 female Yorkshire
Keywords
- Infertility
- Ovarian development
- Female reproductive
- Choline
- Follicle maturation
Infertility is a global issue that affects approximately 8–12% of reproductively mature individuals, with 70% of cases being female [1]. Infertility is a disorder characterized by failure to become or remain pregnant after at least 12 months of unprotected sexual intercourse in humans, which may stem from ovulatory or hormonal disorders, diminished ovarian reserve (DOR), and anatomical, genetic or immunological abnormalities of the reproductive system [2]. In the livestock industry, infertility also causes productivity concerns and economic losses. The annual economic losses of infertility in beef cattle range from 700 to 1100 USD per cow, while seasonal infertility in swine results in approximately 420 million USD losses in the United States annually [3]. Therefore, addressing infertility and increasing reproductive capability is of great significance to both human health and livestock industries.
Research efforts have been focused on the relationship between diet and human fertility over the last decade. Specifically, “healthy” diets are associated with better fertility and higher live birth rate in assisted reproductive technology studies [4]. The majority of infertility cases caused by ovulation-related disorders may be preventable or improved by modifications in diet and lifestyle [5]. Long-chain Omega-3 fatty acids are one of the “healthy” dietary components that appear to improve female fertility [4, 6]. Specifically, lower dietary omega-6-to-Omega-3 ratio is more desirable in decreasing the risk of many diseases which may affect female fertility [7]. In a murine model, the lifelong consumption of an Omega-3 rich diet postponed ovarian aging and improved oocyte quality at advanced maternal age in comparison to the consumption of an Omega-6 rich diet [8]. Furthermore, women consuming more Omega-3 fatty acids are less likely to experience anovulation [9], which may be partly due to the alteration of sexual hormone production by Omega-3 fatty acids [9] and anti-inflammatory properties of the Omega-3 fatty acids, which modulate the immune response during ovarian follicular development and oocyte maturation [10]. Omega-3 fatty acids are not produced at a high enough quantity in the body and need to be obtained from dietary sources [11], similar to other essential nutrients. Hence, optimizing dietary composition is an ideal way to regulate female reproductive capacity, while being extremely cost-effective and safe for wide-spread application.
Choline is a lipotropic amine that carries out various functions in the body and has been described as an essential nutrient for humans [12]. The importance of choline in normal physiological function is noted in most mammals, where choline deficiency has been observed to lead to disorders of the liver, kidneys, pancreas, memory, and growth [13]. Choline is also important in the formation of acetylcholine, which is a neurotransmitter mainly involved in controlling smooth muscle contractions. It can be metabolized through the same pathway as folate and vitamin B12 that are involved in DNA methylation [14]. DNA methylation is important for regulating gene expression, as methylation in the promoter region and CpG islands of genes can influence transcription and subsequent cellular function [15]. Choline is also associated with fertility and fetal development. In utero, choline is pumped against a concentration gradient into the fetus, with the amniotic fluid containing ten times the concentration of choline compared to maternal blood [16]. Further, neural tube development defects are four times more likely to occur in fetuses of women who are in the lowest quartile of choline intake compared to the uppermost quartile [17]. Mother rats receiving choline-deficient diets in late pregnancy had offspring with lower numbers of progenitor cells and higher rates of apoptosis in the hippocampus, indicating poor fetal development [18]. Moreover, choline was significantly decreased in both plasma and follicular fluid of patients with PCOS [19, 20], a metabolic disorder that alters serum levels of a variety of metabolites, ultimately reducing the fertility of affected women [19]. This suggests that choline might have an overarching role in regulating ovarian function. However, the effects of dietary choline on ovarian development and ovulation are not known. Sus scrofa is a widely-used medical model due to its anatomical, physiological and metabolic similarities to humans [21]. Furthermore, studies show that the oocyte development patten, menstrual cycle length and hormonal cycle of female pigs are more similar to women compare to that of rodent species [22, 23], which makes it an idea model for human reproductive performance research. The objective of this study was to assess the effect of choline supplementation on ovarian follicular development and ovulation compared to the basic and Omega-3-supplemented diets. We hypothesized that, in a pig model, choline supplementation improves ovarian follicular development through regulation of genes and miRNA expression and altering levels of serum metabolites.
Thirty-six Yorkshire
| Grower phase* | Finisher phase* | |||||||
| Ingredient (%) | Control | Choline | Omega-3 | Choline + Omega-3 | Control | Choline | Omega-3 | Choline + Omega-3 |
| Corn | 56.87 | 56.75 | 56.87 | 56.75 | 52.67 | 52.56 | 52.67 | 52.56 |
| Wheat | 6.89 | 6.89 | 6.89 | 6.89 | 20.00 | 20.00 | 20.00 | 20.00 |
| Soybean meal | 19.90 | 19.90 | 19.90 | 19.90 | 12.00 | 12.00 | 12.00 | 12.00 |
| Canola meal | 10.00 | 10.00 | 10.00 | 10.00 | 10.00 | 10.00 | 10.00 | 10.00 |
| AV fat (Corn oil) | 2.50 | 2.50 | 0.00 | 0.00 | 2.00 | 2.00 | 0.00 | 0.00 |
| Vitamin and mineral premix | 0.50 | 0.50 | 0.50 | 0.50 | 0.50 | 0.50 | 0.50 | 0.50 |
| Limestone | 1.05 | 1.05 | 1.05 | 1.05 | 1.00 | 1.00 | 1.00 | 1.00 |
| Mono-Ca phosphate | 1.01 | 1.01 | 1.01 | 1.01 | 0.70 | 0.70 | 0.70 | 0.70 |
| Sodium chloride | 0.63 | 0.63 | 0.63 | 0.63 | 0.63 | 0.63 | 0.63 | 0.63 |
| Lys-HCl | 0.49 | 0.49 | 0.49 | 0.49 | 0.39 | 0.39 | 0.39 | 0.39 |
| Met | 0.06 | 0.06 | 0.06 | 0.06 | 0.01 | 0.01 | 0.01 | 0.01 |
| Thr | 0.09 | 0.09 | 0.09 | 0.09 | 0.08 | 0.08 | 0.08 | 0.08 |
| Vit B4 Choline Cl, 60% | 0.00 | 0.11 | 0.00 | 0.11 | 0.00 | 0.11 | 0.00 | 0.11 |
| Fish oil | 0.00 | 0.00 | 2.50 | 2.50 | 0.00 | 0.00 | 2.00 | 2.00 |
| Vitamin E | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 |
| SUM (above) | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| Net energy (kcal/kg) | 2447.4 | 2447.4 | 2447.4 | 2447.4 | 2452.69 | 2452.69 | 2452.69 | 2452.69 |
| Protein (%) | 19.51 | 19.51 | 19.51 | 19.51 | 17.16 | 17.16 | 17.16 | 17.16 |
| SID-lysine (%) | 1.16 | 1.16 | 1.16 | 1.16 | 0.99 | 0.99 | 0.99 | 0.99 |
| Choline (mg/kg) | 500 | 1000 | 500 | 1000 | 500 | 1000 | 500 | 1000 |
| Omega-6 (%) | 1.61 | 1.61 | 1.13 | 1.13 | 1.49 | 1.49 | 1.11 | 1.11 |
| Omega-3 (%) | 0.099 | 0.099 | 0.716 | 0.716 | 0.088 | 0.088 | 0.582 | 0.582 |
| Omega-6: Omega-3 | 16.3 | 16.3 | 1.58 | 1.58 | 17 | 17 | 1.9 | 1.9 |
| *Grower phase and finisher phase were fed between day 90 and 139, 140 and 186,
respectively. *Provided per kg of premix: vitamin A, 2,000,000 IU as retinyl acetate; vitamin D3, 200,000 IU as cholecalciferol; vitamin E, 8000 IU as dl- *SID-lysine: standardize ileal digestible (SID) lysine. | ||||||||
Experimental feed was provided to pigs ad libitum between 90 and 186 days of age. Starting at 90 days of age, pigs were fed the grower diet. At 140 days of age, the pigs were administered PG600 (Intramuscular injection of 400 IU of Equine chorionic gonadotropin and 200 IU of Human chorionic gonadotropin) and boar exposure to synchronize estrous cycles. Feed was also switched to the experimental finisher diet at 140 days of age. Five days later, the pigs entered heat cycle 1, indicating that heat cycle 3 would occur at 187 days of age based on the approximate 21-day estrous cycle. During the grower stage, one of the pigs was culled due to lameness and 35 pigs successfully completed the trial.
Pigs were sacrificed at 186 days of age. Just prior to sacrifice, gilts were
weighed, backfat was measured ultrasonically, and
10 mL of blood sample was collected from the
orbital sinus. After collection of the whole blood in a 15 mL sterile centrifuge
tube, blood was allowed to clot for 30 min at room temperature and the clot was
removed by centrifuging at 3000
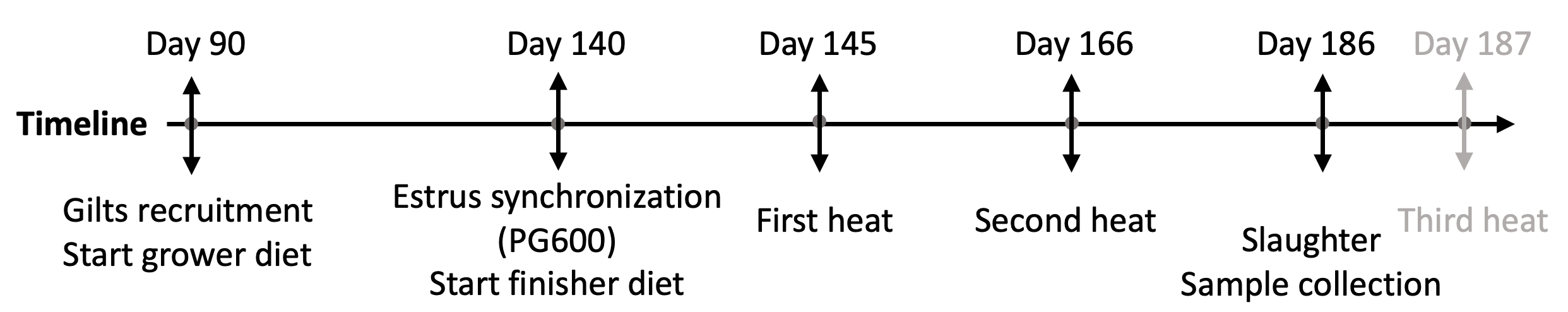 Fig. 1.
Fig. 1.Timeline of animal trial. Animals were recruited at 90 days of age and fed with experimental grower diet. At 140 days of age, PG600 was administrated for estrus cycle synchronization and feed was switched to the experimental finisher diet. At 186 days of age, one day before the expected third heat, gilts were sacrificed, and samples were collected for future analysis.
The ovary samples were fixed for 24 h in 4% formaldehyde and then switched to
70% ethanol for long-term storage. Fixed ovarian samples were dehydrated and
then processed by paraffin block embedding using standard techniques [26]. The
embedded samples were trimmed and sliced into 5
Total RNA was isolated from both ovarian and serum samples using the Norgen
Total RNA purification kit (Norgen Biotek,
Cat#: 17200, Thorold, ON, Canada). For the ovary samples, 10 mg of ovarian
tissue from each pig was thawed, homogenized in the provided lysis buffer, and
total RNA was extracted per manufacturer’s directions. For serum samples, 750
| Gene | Product size | Forward 5′-3′ | Reverse 5′-3′ |
| GAPDH | 147 | TCGGAGTGAACGGATTTGGC | TGCCGTGGGTGGAATCATAC |
| UBB | 127 | GGTGGCTGCTAATTCTCCAG | TTTTGGACAGGTTCAGCTATTAC |
| FSHR | 199 | ATCACACATGCCATGCAACT | GTACGAGGAGGGCCATAACA |
| CYP11A1 | 501 | TTCCAGAAGTATGGTCCCATTTA | TGAGCATGGGGACACTAGTGTGG |
| LHR | 279 | TGCTTTCCAAGGGATGAATAACG | ATCCTATTCTCTGAAAAAACTGCC |
| GAPDH, glyceraldehyde-3-phosphate dehydrogenase; UBB, Ubiquitin B; FSHR, Follicle Stimulating Hormone Receptor; CYP11A1, cytochrome P450 family 11 subfamily A member 1; LHR, luteinizing hormone receptor. | |||
The total RNA was reverse transcribed into complementary DNA for miRNA detection
using the miRCURY LNA RT kit (Qiagen, Cat#: 339340, Hilden, Germany) following
manufacturer instructions and incubated in a Thermocycler at 95
| miRNAs | GeneGlobe ID | Forward 5′-3′ |
| Ssc-miR-21-5p | YP00204230 | UAGCUUAUCAGACUGAUGUUGA |
| Ssc-miR-378-3p | YP00205946 | ACUGGACUUGGAGUCAGAAGGC |
| Ssc-miR-574-3p | YP00206011 | CACGCUCAUGCACACACCCACA |
A solution composed of 1:1 acetonitrile: methanol by volume was added to pig
serum to create an 80% organic solvent by mixing 800
Statistical analysis was performed using SAS version 9.1 (SAS Institute, Cary,
NC, USA) with each pig as the experimental unit, dietary treatment as the fixed
effect, and block as the random effect. Statistical significance was measured
with either an ANOVA post hoc Tukey test or independent two sample
t-test. Differences at p
For analysis of metabolomic data, the HILIC data was analyzed using
MetaboAnalyst 4.0 (McGill University, https://www.metaboanalyst.ca/). A one-way
ANOVA was performed at the p
To study the influence of choline supplementation on growth performance and
reproductive tract development, backfat thickness, oviduct length, vagina-cervix
length and the ovary weight data were collected and analyzed. Initial backfat
thickness was not different among treatment groups, nor were feed intake in the
growing and finishing phases, or final bodyweight (data not shown). Oviduct
length and ovary weight had no significant differences among treatment groups
(Table 4, Ref. [25]). Overall backfat gain was lowest in the control group, with the Omega-3
group having significantly higher backfat gain compared to the control group
(p
| Item | Control | Choline | Omega-3 | Choline + Omega-3 | SEM |
p value |
| N = 9 | N = 8 | N = 9 | N = 9 | |||
| Overall backfat gain (mm) | 7.43 |
8.65 |
9.47 |
9.12 |
0.63 | p = 0.019 |
| Oviduct length (L, cm) | 27.78 | 30.13 | 30.83 | 30.10 | 4.72 | p = 0.250 |
| Oviduct length (R, cm) | 32.16 | 34.43 | 34.28 | 33.34 | 5.43 | p = 0.398 |
| Vagina-cervix-length (cm) |
40.56 |
46.08 |
44.97 |
42.56 |
1.79 | p = 0.027 |
| Ovary weight (g) | 6.94 | 7.26 | 7.72 | 7.87 | 2.35 | p = 0.808 |
*Significance was measured using a one-way ANOVA post hoc Tukey test. Means assigned different letters (a, b) within a factor of are significantly different (p | ||||||
To investigate the effects of a choline-supplemented diet on ovarian
development, the number of ovarian follicles at different stages and corpus
luteum were counted. As shown in Fig. 2, the percentage of antral follicles
(percentage of antral follicles = number of antral follicles/number of total
follicles
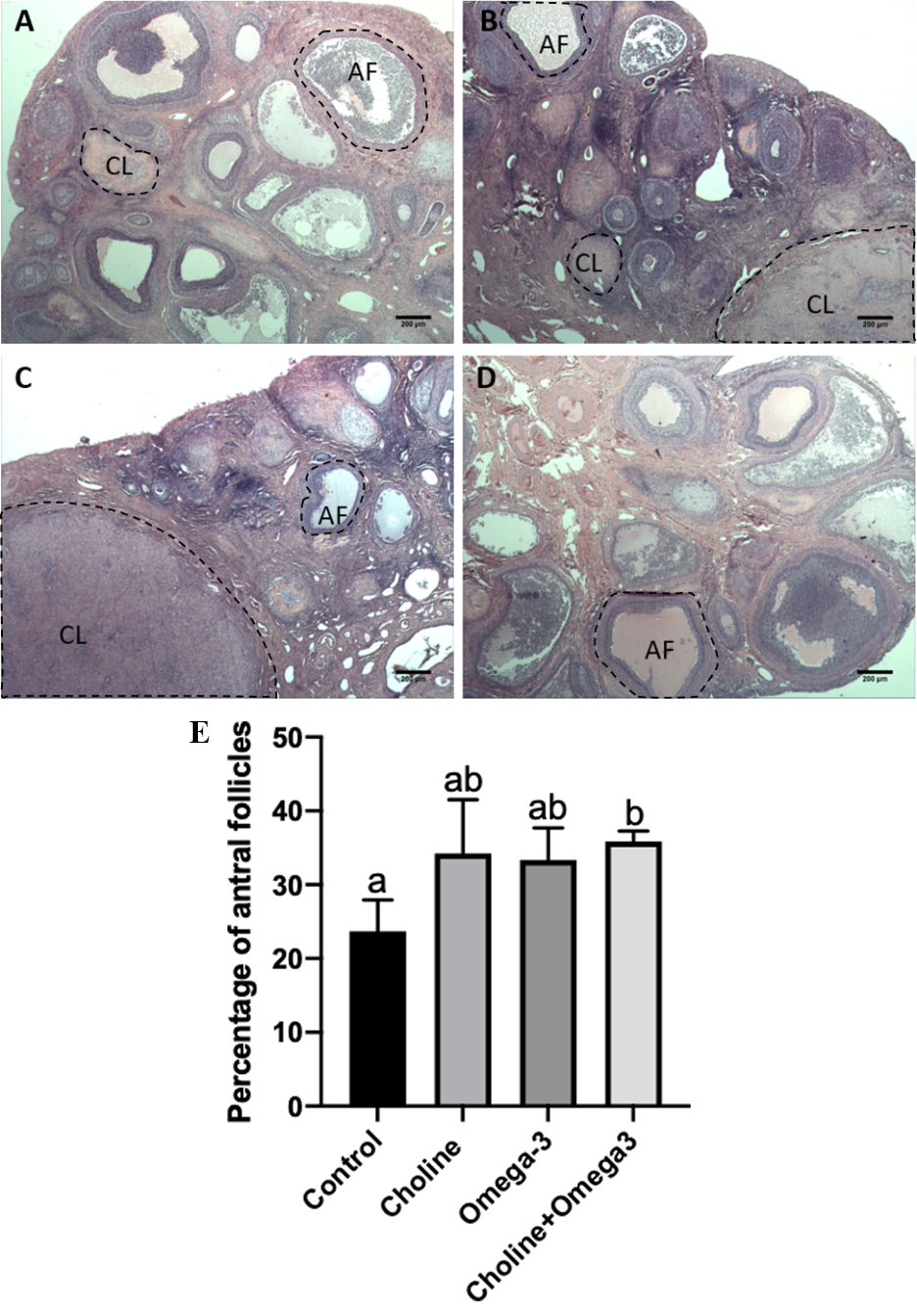 Fig. 2.
Fig. 2.The representative histological section of the ovary in each
group (A–D, H&E staining, 25
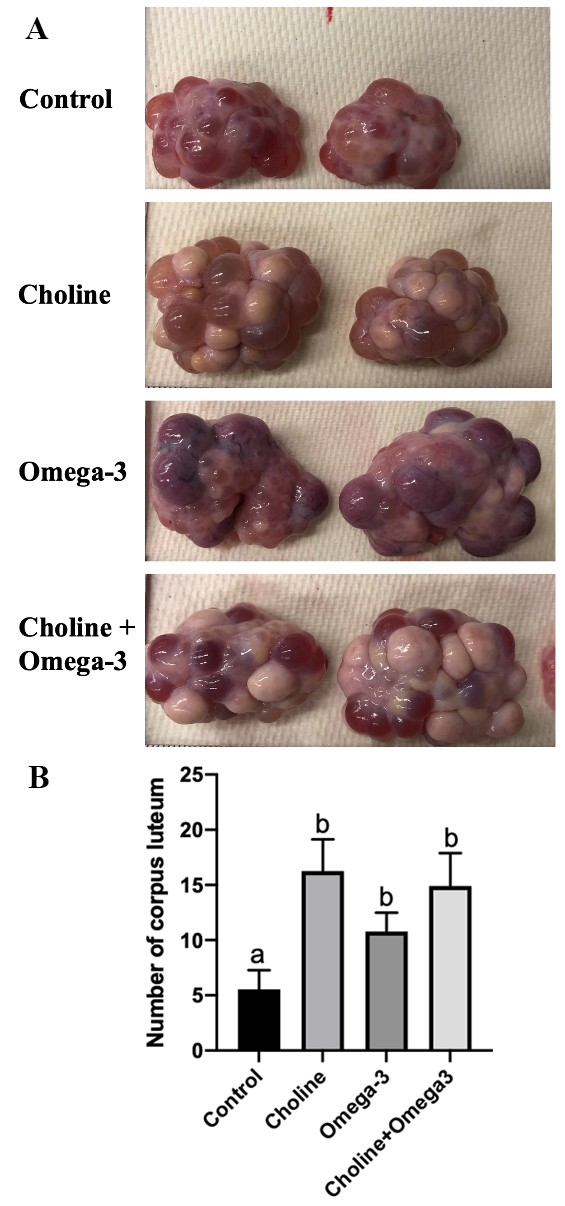 Fig. 3.
Fig. 3.Representative ovarian picture of each group (A) and The number
of visible corpus lutea (B). Values are means
To test the hypothesis that choline influences ovarian function through the
regulation of the expression of genes involved in ovarian development, qPCR
assays were conducted on the steroidogenesis related genes: LHR, FSHR
and CYP11A1. Out of all the measured genes, CYP11A1 and
LHR expression were significantly increased in the Choline-supplemented group in
comparison to the control group (p
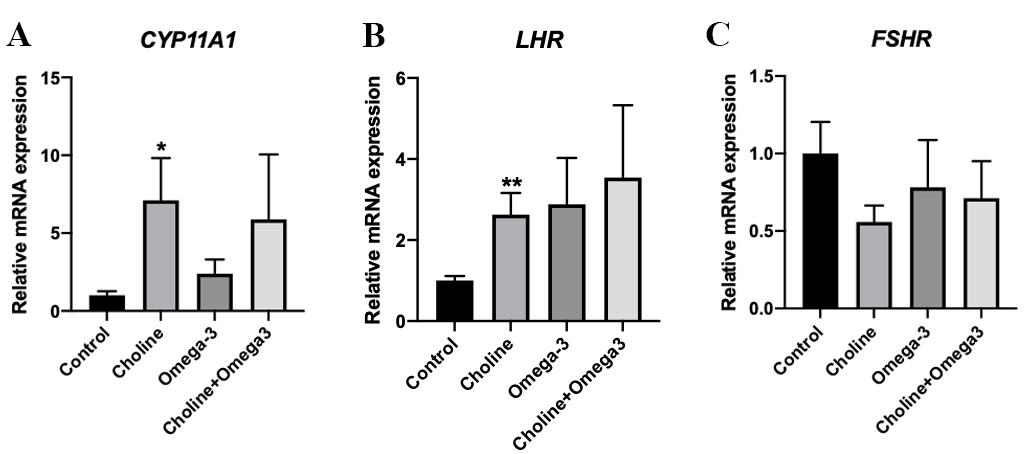 Fig. 4.
Fig. 4.Relative mRNA expression of steroidogenesis related genes in
ovarian tissues. (A) CYP11A1. (B) LHR. (C) FSHR.
Values are means
To examine if choline supplementation alters the expression of miRNAs known to
play an important role in ovarian function, the expression level of miR-21, -378,
-574 in both ovarian tissue and serum were detected via qPCR. As shown in Fig. 5,
miR-21 expression was significantly increased in the Omega-3 group, miR-378
expression was significantly higher in the Choline + Omega-3 group, and miR-574
expression was significantly higher in both Choline and Omega-3 groups, all in
comparison to the control group (p
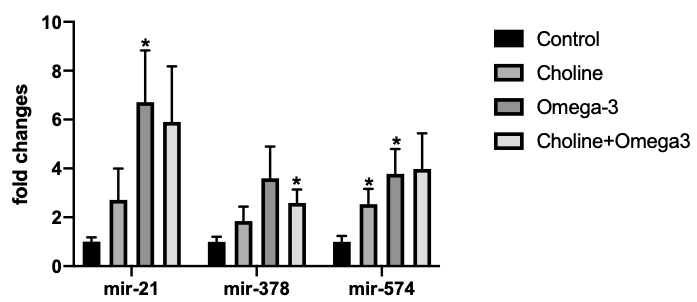 Fig. 5.
Fig. 5.miRNAs expression level in the ovarian tissues. Values are means
To study if choline supplementation influence serum metabolic profiles,
Hydrophilic Interaction Chromatography analysis was performed. Of the 22 targeted
(Supplementary Table 1) and 159 untargeted metabolites
(Supplementary Table 2), only the levels of the untargeted compound,
1-(5Z, 8Z, 11Z, 14Z, 17Z-eicosapentaenoyl)-sn-glycerol-3-phosphocholine, a
phosphatidylcholine, was significantly increased in the Choline group compared to
control group (p
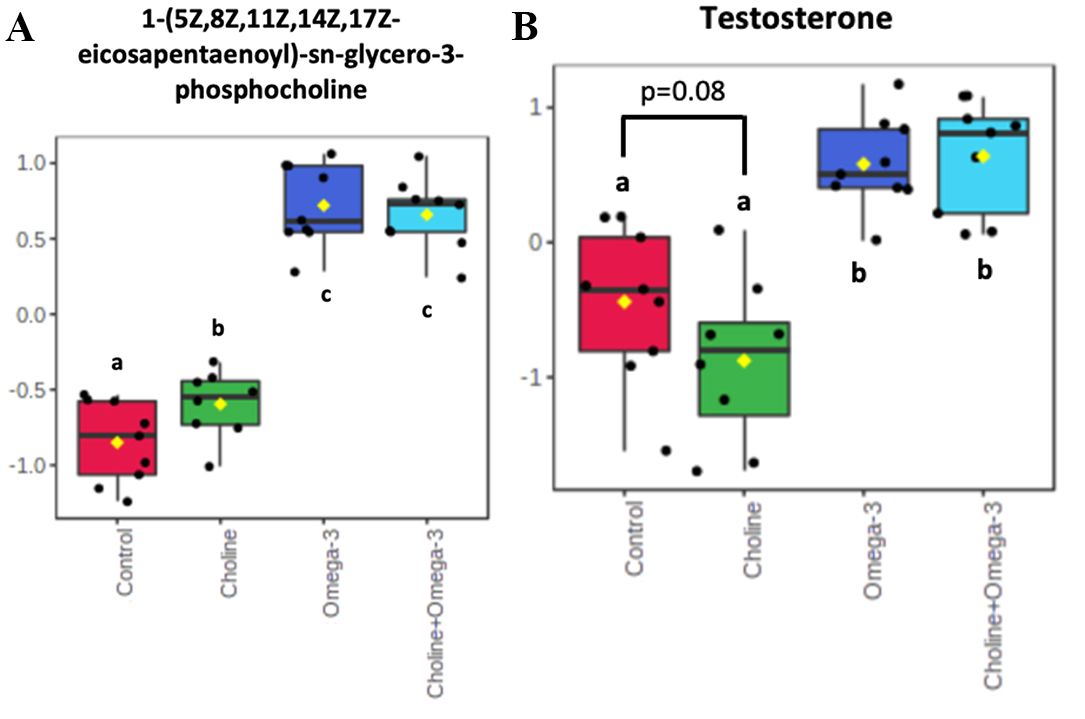 Fig. 6.
Fig. 6.Normalized concentrations
of 1-(5Z, 8Z, 11Z, 14Z, 17Z-eicosapentaenoyl)-sn-glycero-3-phosphocholine (A) and
testosterone (B) in serum. Only the levels of 1-(5Z, 8Z, 11Z, 14Z,
17Z-eicosapentaenoyl)-sn-glycero-3-phosphocholine were significantly increased
(p
Although choline is known for its health benefits in adults, fetal development
and other aspects of fertility [29], its specific role in the female reproductive
tract and ovarian development has not yet been explored. Our findings demonstrate
that choline increases reproductive tract development, which could increase
future litter size, the number of live fetuses and the lifetime piglet production
of female pigs [25, 30]. In humans, the
cervical length is inversely proportional to
the risk of spontaneous preterm births in which those with the shortest cervical
length have the highest risk of prematurity [31]. How the length of the
vagina-cervix is increased by choline supplementation is currently unclear. It is
possible that increased levels of phosphatidylcholine, the dominant phospholipid
component of eukaryotic membranes, was synthesized from the choline, which may
have contributed to the further growth of the reproductive tract tissues in the
Choline group [32]. It has been described that different tissues express
different levels of enzymes that catalyse phosphatidylcholine formation [33], and
the results of the present study could suggest that the reproductive tract
expresses higher levels of these enzymes relative to other tissues. Backfat
thickness has also been described to positively correlate to reproductive
potential in pigs [34, 35]. During reproductive periods, more nutrients are
required for maintenance and lactation, leading to the mobilization of fat
reserves [36]. Therefore, prenatal backfat reserves of the sows may contribute to
later reproductive success. In our study, backfat thickness was numerically
higher in all the supplementation groups, although the difference in comparison
to the control was only significant in the Omega-3 group (p
Ovarian developmental status and oocyte quality are the most important factors that affect female fertility [39]. During unassisted pregnancy in females, follicular development results in the release of oocytes from the ovary to the fallopian tube, where the oocytes can be fertilized by sperm. Moreover, ovulation rate is positively related to fecundity in various species such as sheep [40], cow [41], mice [42] and pig [43]. In this study, a higher percentage of antral follicles were found in Choline + Omega-3 supplemented group in comparison to controls, indicating that more follicles were recruited to form the antral follicle cohort. Follicle recruitment rate is positively correlated with reproductive ability and the oocyte quality in humans [44]. Furthermore, both Choline and Omega-3 groups had a significantly increased number of corpus luteum per ovary in comparison to the control, meaning more follicles had ovulated. Choline is also a substrate for the synthesis of betaine via choline dehydrogenase during oocyte in vitro maturation, and this betaine accumulation is reported to be required for oocyte meiotic maturation [45]. Taken together, this suggests that choline and/or Omega-3 supplementation promotes and enhances ovulation for successful fertilization and implantation. Additionally, choline is an essential component in forming phosphatidylcholines, a class of phospholipids that are a major element in forming the cellular membrane. Phosphatidylcholines accumulate during follicle growth, and their depletion significantly affects follicular development and antrum formation [46]. Thus, choline supplementation may have contributed to oocyte maturation and ovulation by providing additional substrate for the synthesis of these biomolecules.
During the estrus cycle, the level of luteinizing hormone acutely rises to trigger ovulation and generate corpus luteum. In response to the changes of the concentration of this gonadotropin, the density of its receptor, LHR, and related downstream catalytic enzyme expression in the ovary changes to ensure that the menstrual cycle progresses in an orderly manner [47, 48, 49]. Our finding that a significant increase of LHR expression in the ovary of the Choline-supplemented group is consistent with this notion, and agrees with the fact that more corpus luteum were observed. Furthermore, the expression of CYP11A1, a gene coding for the P450scc mitochondrial enzyme that is the first and rate-limiting catalytic enzyme in steroidogenesis for the synthesis of pregnenolone from cholesterol [48], was upregulated in the ovaries of pigs in the Choline group. This result is consistent with a previous finding that P450scc expression increases along with luteinizing hormone surge and corpus luteum formation [48]. These steroidogenesis-related gene expression results suggest that choline potentially plays a role in the regulation of the ovarian menstrual cycle and steroid hormone production.
MiR-21, -378, -574 are all important regulators of ovarian function. MiR-21 promotes cumulus expansion and oocyte maturation in vitro via targeting tissue inhibitor of metalloproteinase 3(TIMP3) [50], and miR-574 modulates phosphorylated extracellular-signal-regulated kinase (ERK) 1/2 level and increases estradiol production. MiR-378 regulates estradiol production and apoptosis rate in the ovary [51, 52]. Our findings that miR-21 expression was significantly increased in the Omega-3 group, and miR-574 expression was significantly increased in both the Choline and Omega-3-supplemented groups is consistent with the observed enhanced follicular development and ovulation observed. Interestingly, others have also reported that miR-21 is highly expressed in the female goats with high ovulation rate [53]. Ma et al. [54] identified that miR-378 as the most differentially expressed miRNA in which its expression level increased over fivefold during luteal maturation. In our study, miR-378 expression was significantly higher in Choline + Omega-3 group, which is consistent with the previous results that more corpus luteum were formed in this group in comparison to the control group. These results suggest that Omega-3 and choline supplementation promote positive regulators of folliculogenesis and ovulation, possibly enhancing reproductive potential of the individual.
Lastly, serum analysis of metabolites taken from the pigs revealed a significant change in phosphatidylcholine phospholipids and trending changes in testosterone. Testosterone is an androgen that coordinates a variety of bodily processes including energy metabolism, muscle and bone mass, libido, and especially, fertility [55]. Polycystic ovarian syndrome (PCOS) is a reproductive metabolic syndrome particularly characterized by hyperandrogenism, a state of excess androgen production in which testosterone is included [56]. Currently, it is unknown how choline administration may impact metabolites related to infertility and the PCOS phenotype. However, the trending lower level of testosterone suggests that choline may have potential in the treatment or prevention of PCOS. Further, the metabolic analysis of pig serum in the Choline group showed a significant increase in the levels of 1-(5Z, 8Z, 11Z, 14Z, 17Z-eicosapentaenoyl)-sn-glycero-3-phosphocholine, a form of phosphocholine, which is reasonable as choline is a major element of its components, however, its specific role in follicular development remains unknown. It is reported that choline and phosphocholine were reduced in the follicular fluid from ovarian follicles where the fertilized oocyte failed to cleave [57], suggesting that phosphocholine may play a role in the oocyte competence development in which 1-(5Z, 8Z, 11Z, 14Z, 17Z-eicosapentaenoyl)-sn-glycero-3-phosphocholine may be involved.
In summary, we report that choline supplementation promotes reproductive tract development and ovulation which is accompanied by enhanced expression of steroidogenesis-related genes and miRNAs related to ovarian regulation. To our knowledge, this is the first in vivo examination of the influence of choline supplementation on ovarian development and function. Our findings support that the supplementation of this nutrient may have fertility enhancement benefits and offer insights into how choline supplementation could be applied to improve reproductive function.
XZ, BW, LAH and JL conceived and designed the experiments; XZ, LF, SD and EB performed the experiments; XZ, LF and EB analyzed the data; XZ, LF, EB, LAH and JL contributed manuscript writing.
All experimental procedures were approved by the University of Guelph’s Animal Care Committee (AUP #4068) and followed the Canadian Council of Animal Care Guidelines (CCAC, 2009).
We thank Douglas Wey and the staff at Arkell Swine Research Station for their assistance in the animal trial, Renee Hilker for her assistance in collecting animal samples.
This work was supported by the Natural Sciences and Engineering Research Council of Canada (NSERC, grant number: 400259), and the Food from Thought Research program (grant number: 499093).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://www.imrpress.com/journal/FBL/26/12/10.52586/5046.






