1 Center for Immunology and Cellular Biotechnology, Immanuel Kant Baltic Federal University, 236001 Kaliningrad, Russia
2 Higher School of Living Systems, Immanuel Kant Baltic Federal University, 236001 Kaliningrad, Russia
Abstract
Obese individuals are at high risk for developing type 2 diabetes mellitus, cardiovascular diseases, and nonalcoholic fatty liver disease. The aim of this review was to analyze the scientific literature and databases to reveal the fundamental role of neuregulin 4 (NRG4) and its receptors in the development of obesity-associated metabolic disorders. This review demonstrates that NRG4 and its receptors are promising therapeutic targets for the treatment of socially significant obesity-associated pathologies. The review contains nine chapters. Information on the structure of ERBB4 and NRG4 splice isoforms and subsequent activation of downstream targets is presented. The tissue-specific features of the NRG4 and ERBB4 genes and protein production are also highlighted. The role of NRG4 and ERBB3/4 in the pathophysiological mechanisms of the development of metabolic disorders in obesity is discussed in detail. The final chapter of the review is devoted to the miRNA-dependent regulation of NRG4 and ERBB4. Recent studies have shown that several miRNAs regulate ERBB4 expression, but no information was found on the interaction of NRG4 with miRNAs. We now demonstrate the putative relationships between NRG4 and let-7a-5p, let-7c-5p, miR-423-5p, miR-93-5p, miR-23a-3p, and miR-15b-5p for the first time. In addition, we found SNP mutations affecting the interaction of NRG4 and ERBB4 with miRNA in these genes as well as in miRNAs. In summary, this review provides a detailed and comprehensive overview of the role of NRG4 in obesity-associated metabolic disorders. The review summarizes all current studies on this topic and opens perspectives for future research.
Keywords
- ERBB signaling
- NRG4
- ERBB4
- obesity
- type 2 DM
- NAFLD
- miRNA
- SNP
Obese individuals are at high risk for developing type 2 diabetes mellitus (type 2 DM), cardiovascular diseases, non-alcoholic fatty liver disease (NAFLD), and other obesity-associated diseases. The presence of these pathologies significantly reduces quality of life and causes enormous socio-economic losses.
Neuregulins are a group of growth factors that transmit signals through a group of receptor tyrosine kinases, the human epidermal growth factor (EGF) receptors (ERBB/HER)—ERBB 1, 2, 3, 4—all of which regulate various biological processes [1]. Neuregulin family proteins (NRG), NRG1 and NRG2, bind to both ERBB3 (HER3) and ERBB4 (HER4), while NRG3 and neuregulin 4 (NRG4) bind preferentially to ERBB4. Of particular interest is the ERBB4 ligand NRG4, which is expressed in liver [2], lung, pancreas [3], and most actively in adipose tissue [4]. In a recent review, Blüher (2019) [5] and Tutunchi et al. (2019) [6] demonstrated the important role of pharmacological activation of brown adipose tissue in metabolic health. Therefore, it is necessary to understand the mechanisms of action and secretion of batokines, including NRG4, in the pathogenesis of obesity and metabolic disorders [5, 6].
NRG4 acts as an autocrine, paracrine and endocrine factor through proteolytic release of an EGF-like domain. ERBB4 has several salient features compared to other members of the ERBB family, making it a potentially unique and selective target in terms of signal transduction. Deletion of ERBB 4 in mice was found to lead to dysregulation of carbohydrate and lipid metabolism [7] and to accelerate the development and progression of renal fibrosis after kidney injury [8].
However, scientific data on the involvement of NRG4 and ERBB4 in various diseases are contradictory. In a study, ERBB4 gene polymorphisms (rs10932374, rs13003941, rs1595064, rs1595065) were associated with the risk, severity, and prognosis of heart failure in the Northern Han population [9].
Over the past two decades, compelling evidence has accumulated for the involvement of microRNAs in the development of several obesity-associated metabolic diseases. However, data on miRNA-dependent regulation of this pathway in metabolic disorders is extremely limited.
The aim of this review was to comprehensively assess the data on the fundamental role of NRG4 and its receptors in the development of obesity-related metabolic disorders. Before examining the role of NRG4 and its ERBB4 receptor in metabolic diseases, we address the structural and genetic features of these proteins. Structural variations of ERBB4 and NRG4 proteins influence their functions as well as their role in the pathogenesis of socially significant diseases.
ERBB4 (a.k.a. HER4), a 147 kDa glycoprotein, belongs to the ERBB receptor family, which also includes ERBB1 (EGFR/HER1), ERBB2 (HER2/NEU), and ERBB3 (HER3). ERBB receptors are structurally similar: they consist of a glycosylated extracellular region (ectodomain), a small transmembrane region, and an intracellular cytoplasmic domain (Fig. 1). The ectodomain of the receptor consists of 4 subdomains and is responsible for binding of the ligand, one of the growth factors, and for dimerization of the receptor. Ligand binding to the extracellular domain leads to homo- or heterotypic receptor dimerization and activation of the intracellular tyrosine kinase domain. The intracellular region of ERBB receptors consists mainly of three subdomains: a small juxtamembrane, a tyrosine kinase, and a C-terminal domain. In ERBB4, the juxtamembrane is located in the ectodomain [10].
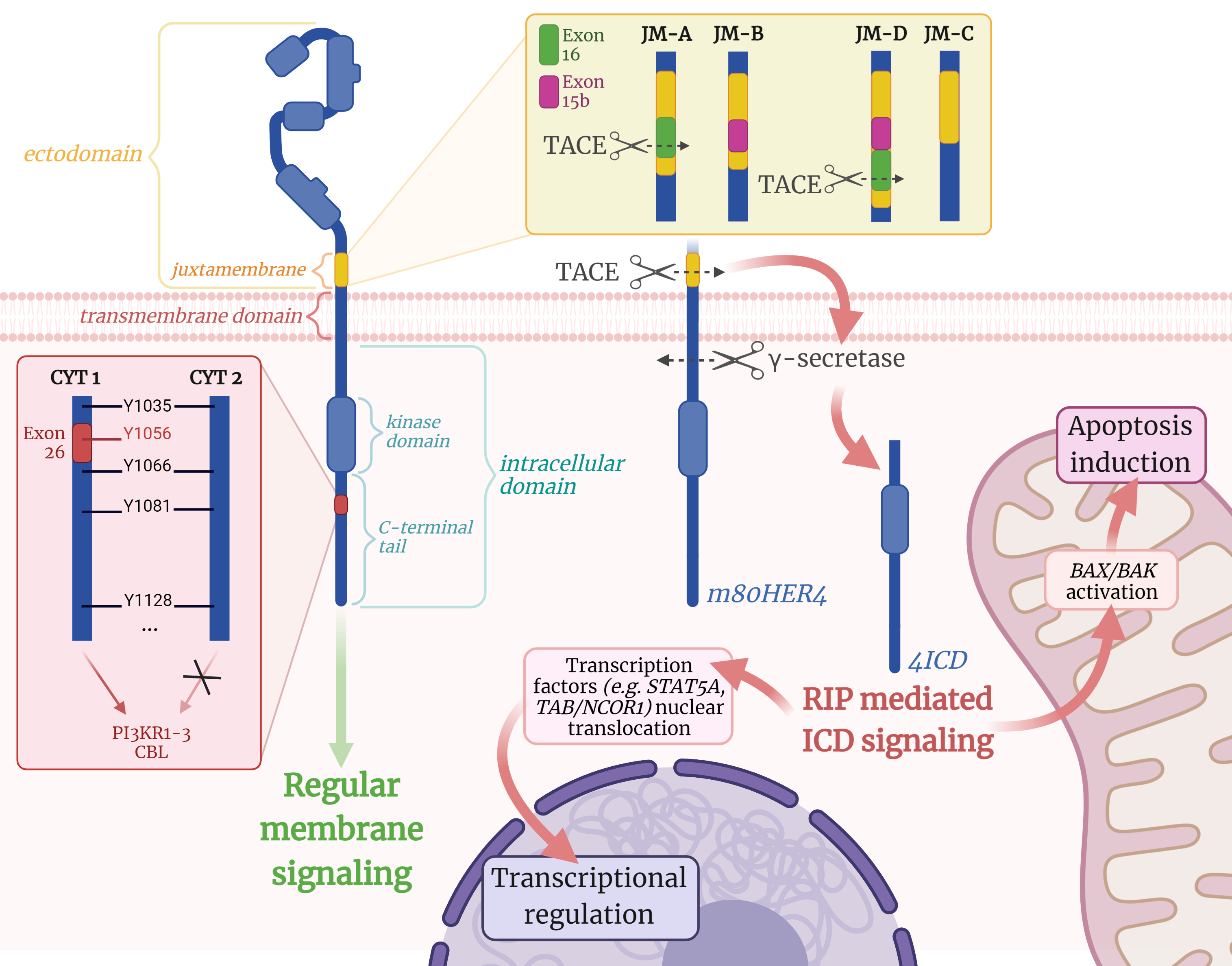 Fig. 1.
Fig. 1.The structure and splice isoforms of the ERBB4 receptor and
subsequent activation of downstream targets. CBL, Cbl Proto-Oncogene; CYT,
cytoplasmic; m80HER4, ERBB4 membrane-associated fragment of 80 kDa; 4ICD, ERBB4
intracellular domain; ICD, intracellular domain; JM, juxtamembrane; PIK3R1,
phosphatidylinositol 3-kinase regulatory subunit alpha; RIP, regulated
intramembrane proteolysis; TACE, tumor necrosis factor-
Ligand-activated receptors phosphorylate each other in a series of tyrosine residues that serve as docking sites for downstream enzymes or adapter proteins in the signaling pathway [11]. ERBB4 is activated by the binding of one of seven ligands: heparin-binding EGF-like growth factor, epiregulin, betacellulin, and the neuregulin family of proteins: NRG1, NRG2, NRG3, and NRG4 [12]. It is important to note that members of the ERBB family (particularly ERBB4) are capable of autophosphorylation even in the absence of the ligand when overexpressed [11].
In 2008, Kaushansky et al. [11] demonstrated that ERBB4 (CYT1 isoform) has 19 tyrosine residues in the C-terminal domain, 18 of which can be phosphorylated (pTyr). They noted that ERBB4 has more intracellular tyrosine residues capable of phosphorylation than most other tyrosine kinase receptors. However, some of these 18 tyrosine sites are thought to be phosphorylated only in response to specific ligands or only in certain cells. In addition, some of the identified pTyr sites might be phosphorylated only when the receptor is auto-activated by overexpression. In total, there are 14 proteins with Src homology 2 (SH2) domains that are recruited by ERBB4 kinase activity, namely PI3KR1-3, ABL2, SRC, SYK, PLCG2, CRK, CRKL, RASA1, VAV2, SHC1, STAT1, and CBL.
The ERBB4 gene, located on human chromosome 2, is alternatively spliced in two specific regions of the primary transcript: the C-terminal domain (CYT1 and CYT2 variants) [13] and the juxtamembrane region (JM-A, JM-B, JM-C, JM-D) [14] (Fig. 1). The CYT-2 isoform has a deletion of 16 amino acids in exon 26 corresponding to positions 1046-1061 in the human ERBB4 protein sequence. As a result, this isoform loses its ability to directly activate the downstream signaling proteins phosphatidylinositol 3-kinase-related kinase 1-3 (PI3KR1-3) and Cbl Proto-Oncogene (CBL) since its potentiation occurs via pTyr at position 1056 [9]. Alternative splicing of exons 16 (encoding 23 amino acids) and 15b (encoding 13 amino acids) of the ERBB4 gene creates isoforms JM-A and JM-B [13]. The functional difference between the JM-A and JM-B isoforms lies in their different resistance to proteolytic cleavage. It was discovered that the JM-A isoform loses its entire extracellular domain upon exposure to phorbol esters, resulting in loss of the ability to bind ligands. At the same time, the JM-B isoform is resistant to phorbol esters. In malignant medulloblastoma cells, two additional isoforms have been found that either completely lack the JM-C or are elongated by the contents of both exons 16 and 15b (JM-D) [14].
The ability for proteolytic cleavage is definitely not a “defect” of the JM-A
isoform, since this isoform is involved in the so-called regulated intramembrane
proteolysis (RIP). Proteolytic cleavage of the JM-A ERBB4 isoform occurs in
response to tumor necrosis factor alpha converting enzyme (TACE) metalloprotease
in the juxtamembrane region between His-651 and Ser-652, resulting in the
formation of an 80-kDa membrane-bound fragment of ERBB4 (m80HER4) [15, 16].
m80HER4 is in turn further processed by a presenilin-dependent
In 1999, during analysis of mouse DNA, Harari et al. [21] discovered a fourth member of the neuregulin family, NRG4 (later the open transcript was designated NRG4A1 [22]), which they hypothesized to be a ligand exclusively for ERBB4. NRG4 was later shown to also cause a dose-dependent increase in phosphorylation of ERBB3 tyrosine residues in cultured HEK293 cells. However, ERBB3 has no intrinsic kinase activity, and its increased phosphorylation is likely due to ligand-dependent heterodimerization with ERBB4 in response to NRG4 [23].
NRG4 is a transmembrane protein with a unique EGF-like motif and a short cytoplasmic domain. After cleavage by the metalloprotease TACE, the extracellular region of NRG4 containing the EGF-like domain enters the intercellular space and acts as an ERBB4 ligand [21]. Analysis of the nucleotide sequence of mouse NRG4 showed that it has relatively low homology with other sequences of the Nrg genes, especially near the transmembrane domain [22]. In addition, sequence analysis in the Ensembl database has shown that the human NRG4 protein is much shorter than other members of the neuregulin family: its length is 110–115 amino acid residues, while NRG1–3 has a length starting at about 500 amino acid residues [24].
In 2007, a total of five NRG4 splice isoforms were discovered: A1-2 and B1-3. NRG4A2 differs from the original NRG4A1 by having a shorter intracellular domain. However, unlike NRG4A1, the NRG4A2 isoform has a PDZ binding motif and a different cellular translocation [21]. The NRG4B1–3 isoforms were discovered by bioinformatics analysis and their expression was confirmed in human prostate cell lines. NRG4B1–3 are highly truncated compared to the A isoforms (36, 35, and 56 amino acid residues, respectively). They all lack transmembrane and intracellular segments as well as the exon encoding cysteine residues 5 and 6 of the EGF-like domain. Therefore, they possess only part of the EGF-like domain and appear unable to activate ERBB4, as demonstrated in the rat pheochromocytoma cell line [25].
All ERBB4 isoforms possess two tyrosine residues that serve as binding sites for SHC Adaptor Protein 1 (SHC1) [11]. Upon activation, ERBB4 acquires tyrosine kinase activity and phosphorylates SHC1, leading to recruitment of growth factor receptor-bound protein 2 (GRB2) and Son of Sevenless (SOS1) protein. This event in turn activates the mitogen-activated protein kinase (MAPK) signaling pathway, which leads to increased cell survival by inducing proliferation and cell differentiation through the activation of proteins such as nuclear factor kappa B (NF-kB), and inhibition of forkhead proteins (FOXO) (Fig. 2, Ref. [23, 26, 27, 28]).
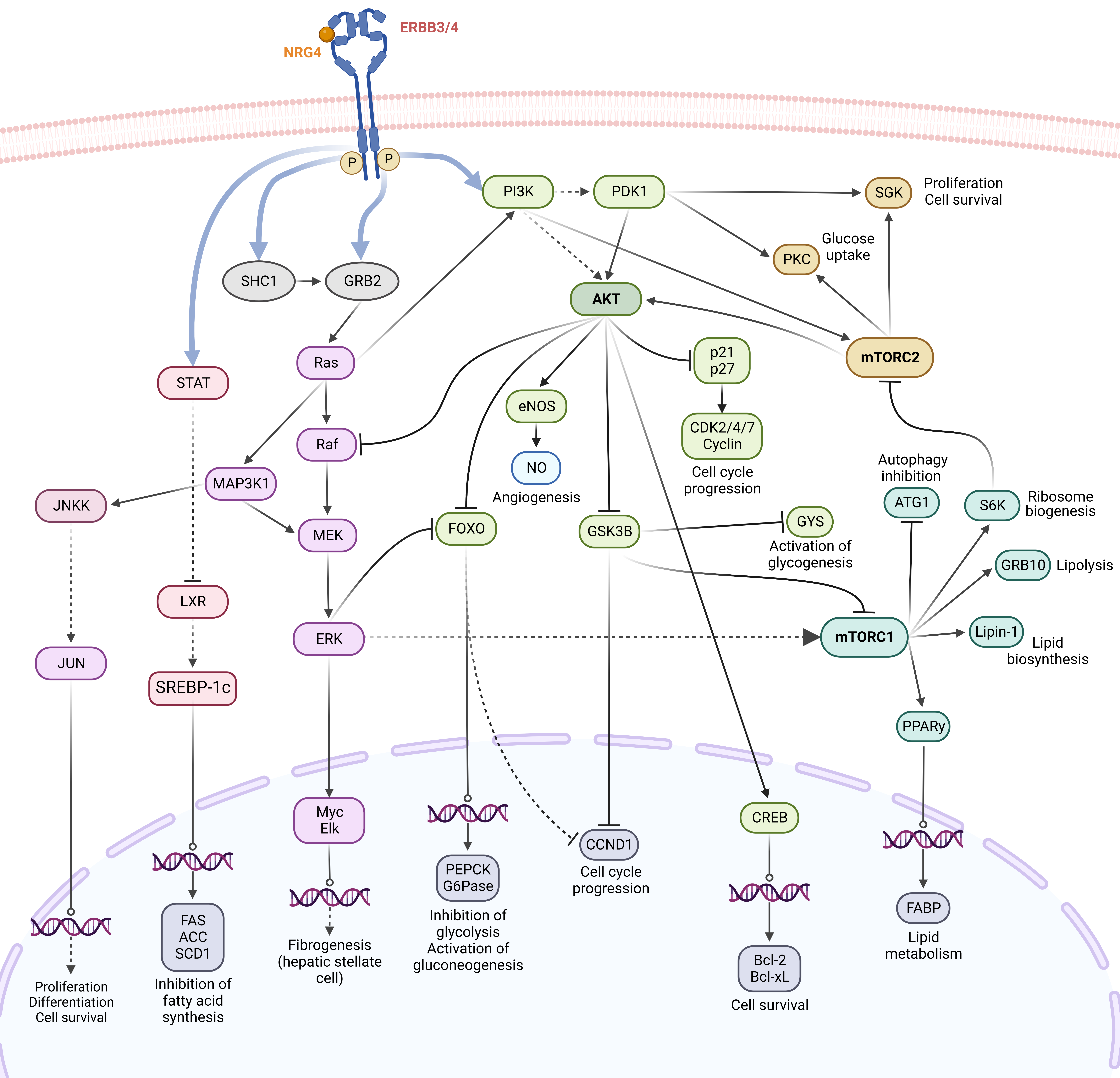 Fig. 2.
Fig. 2.Regular ERBB4 membrane signaling and resulting metabolism-related processes. Activation of ERBB receptors by autophosphorylation of their tyrosine residues in intracellular domains leads to mobilization of a variety of metabolic pathways, of which the following are particularly noteworthy: mitogen-activated protein kinase (MAPK) signaling pathways, phosphoinositide-3-kinase/protein kinase B (PI3K/AKT), and mammalian target of rapamycin (mTOR). Based on open data from the Kyoto Encyclopedia of Genes and Genomes [26, 27, 28] and adapted from [23]. Created with BioRender.com.
The CYT1-ERBB4 isoform can directly trigger PI3K/AKT signaling due to the presence of phosphorylated tyrosine residue 1056 in the intracellular C-terminal domain. Of the four members of the ERBB protein family, ERBB3, unlike EGFR and ERBB2, is also capable of directly activating phosphoinositide 3-kinases (PI3K). Thus, the PI3K/AKT pathway can be recruited by dimers containing ERBB-3/4. Activation of PI3K catalyzes the conversion of phosphatidylinositol-3,4-bisphosphate (PIP2) to phosphatidylinositol-3,4,5-triphosphate (PIP3). PIP3 on the cell membrane activates protein kinase B (AKT) directly or indirectly via phosphoinositide-dependent kinase-1 (PDK1). Phosphorylated AKT mediates numerous cellular functions, including angiogenesis (via endothelial nitric oxide synthase), metabolism (via FOXO proteins, glycogen synthase kinase (GSK3), protein kinase C (PKC)), growth, proliferation, survival (via proteins FOXO, cAMP response element-binding protein (CREB), cyclin-dependent kinases, and cyclins) (Fig. 2) [26, 27, 28].
The mammalian target of rapamycin (mTOR) is a highly conserved serine-threonine protein kinase that exists in two complexes, mTORC1 and mTORC2. mTORC1 is activated indirectly via AKT in the ERBB pathway and plays an important role in regulating lipid metabolism (activates growth factor receptor-bound protein 10 (GRB10) and lipin-1) and autophagy (inhibits autophagy-related protein 1 (ATG1). In contrast, mTORC2, which is also activated via the PI3K/AKT pathway, controls cytoskeletal organization and metabolism (via PKC) and cell survival (via serine-threonine protein kinase (SGK)) (Fig. 2) [26, 27, 28].
According to RNA-seq data from the Human Protein Atlas (HPA) [4], in which the transcriptomes of 27 different organs and tissues from a total of 95 individuals were examined by next-generation sequencing, ERBB4 expression in humans is highest in the brain (particularly in the midbrain, basal ganglia, hypothalamus, and cerebral cortex). In addition to brain structures, the fallopian tubes, breast, and kidneys also have relatively high ERBB4 gene expression [29]. It should be noted that ERBB4 protein levels are somewhat inconsistent with transcriptome analysis data, as shown in Fig. 3 (Ref. [30]).
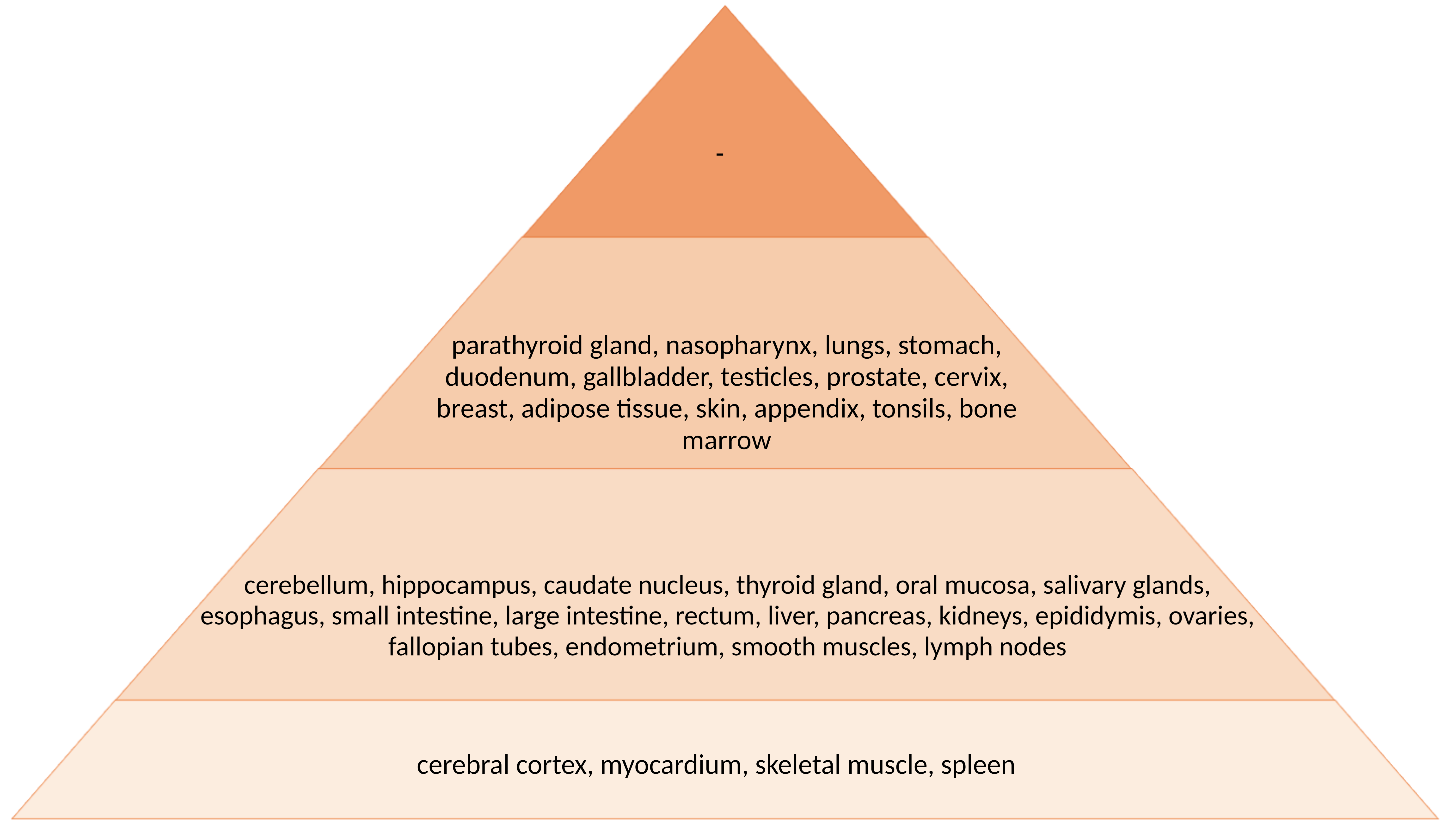 Fig. 3.
Fig. 3.Intensity of histological staining of human body tissues with antibodies against ERBB4 protein according to the HPA study [30]. The intensity of the staining (the concentration of the protein) corresponds to the color in the figure.
NRG4 has the highest expression in brown adipose tissue [23]. Of note, NRG4 gene expression in mouse white adipose tissue is significantly increased upon exposure to cold or differentiation into brown adipose tissue [31]. One of the main differences between brown and white adipose tissue is the degree of sympathetic innervation. Rosell et al. (2014) [31] showed that NRG4 is secreted into the culture medium of brown mouse adipocytes and this medium can promote the growth of axons in neurons. This suggests that NRG4 is a novel adipokine of brown adipose tissue with important autocrine/paracrine functions in the development of adipose tissue innervation [31]. According to HPA data, the highest expression level of the NRG4 gene in humans is found in the cerebellum [29].
Analysis of the production levels of NRG4A1 and NRG4A2 isoforms by histological staining yielded the following results, which are summarized in Fig. 4 (Ref. [32]).
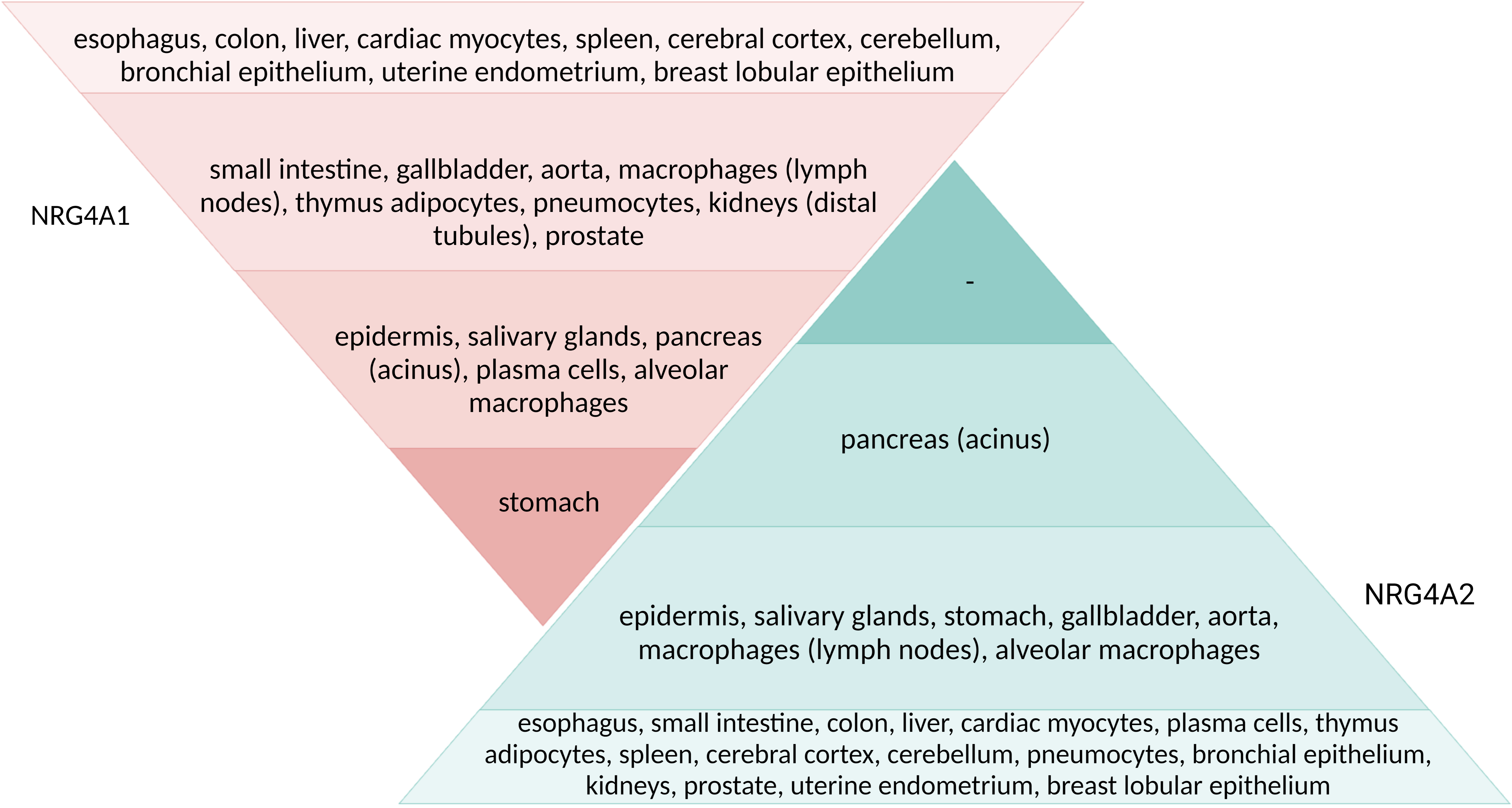 Fig. 4.
Fig. 4.Intensity of histological staining of human body tissues with antibodies against two isoforms of NRG4 protein. Adapted from [32]. The intensity of the staining (the concentration of the protein) corresponds to the color in the figure.
In the presence of pro-inflammatory (IFN
Inhibition of
Obesity results from an imbalance between energy intake and expenditure that leads to excessive accumulation of adipose tissue [6]. Excess body fat is associated with the development of type 2 diabetes, hypertension, dyslipidemia, and chronic inflammation, leading to the development of cardiovascular disease. Adipose tissue is considered a highly active metabolic organ that secretes a number of biologically active compounds called adipokines, which can regulate energy homeostasis [35].
In 2014, two studies reported that NRG4 has a high production profile in adipose
tissue, particularly brown adipose tissue (BAT), and is considered an adipokine
[23, 31]. It has been previously mentioned that although NRG4 is abundant in BAT,
NRG4 expression is upregulated in white adipose tissue (WAT) upon exposure to
cold [31]. Therefore, NRG4 may be a key factor in the acquisition of BAT
functions in the WAT depot, such as more advanced innervation. In addition, NRG4
is able to regulate adipose tissue vascularization. It is assumed that in
obesity, an increase in the volume of adipose tissue is associated with
vasoconstriction and the development of hypoxia, which in turn is associated with
an inflammatory phenotype (activation of HIF-1
In obese adults and children, NRG4 gene expression decreases in adipose tissue [38]. A cross-sectional study of 1212 obese adult subjects showed that patients with metabolic syndrome had lower circulating NRG4 levels than patients without metabolic syndrome and that, in turn, metabolic syndrome was diagnosed more frequently in subjects with reduced circulating NRG4 levels. In addition, patients with low circulating NRG4 levels are more likely to have significant increases in fasting blood glucose and blood pressure [39]. NRG4 expression in subcutaneous adipose tissue of children has also been shown to correlate negatively with body mass index and standard deviation score. A large study from China found an association between NRG4 and metabolic syndrome in patients with newly diagnosed type 2 diabetes mellitus [40]. However, these authors stress that their results may not be generalizable to other populations; and it was not possible to determine the correlation between NRG4 levels and IR, HOMA-IR, apoB/apoA, and the TyG index. Plasma NRG4 has been shown to be a useful biomarker for metabolic and cardiovascular disorders associated with obesity [41].
It should be noted that physical exercise leads to an increase in blood NRG4 levels. Thus, resistance training, especially interval training, increased plasma levels of NRG4 and adiponectin and decreased leptin levels in obese men [42]. It is important to point out that blood NRG4 levels also seem to depend on hormonal background. For example, obese girls diagnosed with polycystic ovary syndrome (PCOS) were found to have elevated NRG4 levels compared with control subjects without PCOS. In contrast, circulating NRG4 levels do not increase in girls with PCOS but decrease after weight loss [43]. This should be further investigated and considered when evaluating NRG4 expression profiles.
Thus, a decrease in NRG4 expression in adipose tissue is observed in obesity in an animal model, in adults, and in children. The decrease in NRG4 levels is influenced by pro-inflammatory activity, but increased physical activity and exposure to cold can stimulate NRG4 expression.
In 2016, Kang et al. [44] first found a positive correlation between
NRG4 levels and glucose, HOMA-IR, and fasting triglyceride levels. Subsequently,
in several studies NRG4 has been proposed as a marker for type 2 DM [45, 46].
Kocak et al. [46] found a significant increase in serum NRG4 in type 2
DM that correlated positively with glucose levels compared to the control group.
A comparative analysis of patients with well-controlled type 2 DM (glycated
hemoglobin (HbA1c)
In obese patients, serum NRG4 levels correlate negatively with insulin sensitivity (hyperinsulinemic-euglycemic clamp method) and positively with the high-sensitivity inflammatory marker C-reactive protein (hsCRP) [47]. These results suggest that pro-inflammatory and anti-inflammatory signals have opposing effects on NRG4 expression in adipocytes. Inflammation of adipose tissue reduces NRG4 expression in adipose tissue and which likely explains the decrease in NRG4 expression in patients with persistent type 2 DM.
The development of hyperglycemia in type 2 DM occurs through an increase in endogenous glucose levels due to increased gluconeogenesis in the liver. The role of NRG4 in the liver in humans and animal models is controversial.
Zhang et al. (2019) [47] showed that NRG4 in the liver plays a critical
role in the regulation of gluconeogenesis. Knockdown of NRG4 in the liver of
C57BL/6 and db/db mice inhibited the expression of peroxisome
proliferator-activated receptor gamma coactivator 1-alpha (PGC-1
These authors suggested that NRG4 may be a good predictor of one or more
diabetic microvascular complications (DME): retinopathy, neuropathy and
nephropathy [45]. NRG4 levels in patients with type 2 DM were 1.23 (0.02–5.1)
ng/mL in groups with DME and 2.5 (0.21–6.01) ng/mL in groups without DME
(p
A recent Chinese study (2021) [51] found an association between NRG4 and NRG1 levels in the development of GDM and a high risk of transition to type 2 DM. Low NRG4 levels and high NRG1 levels in the circulation are considered risk factors for GDM [51]. Therefore, determination of NRG4 levels in the bloodstream is of clinical importance for the diagnosis of type 2 diabetes. The negative effect of NRG4 on the liver is manifested by alteration of metabolic and mitochondrial functions, whereas NRG4 in adipose tissue protects against obesity-induced insulin resistance. Thus, in a culture of 3T3-L1 cells with NRG4 knockdown (2021) [52], an increase in the expression of pro-inflammatory cytokines and a violation of insulin signaling due to a decrease in the expression of insulin and GLUT4 receptors were detected. However, we have not yet found any publications on the effects of drug treatment of type 2 DM and its complications on serum levels of NRG4 in patients with this disease. In 2022, the first study was published showing that 6 months of metformin therapy improved NRG4 levels and glycemic control in children with type 1 diabetes [53].
In view of these findings, the multidirectional dynamics of blood NRG4 levels could be caused by the experimental design, the specifics of the analytical methods, and to an even greater extent, by the characteristics of the population that was studied. In addition, serum levels of NRG4 in patients with type 2 diabetes may be affected by drug therapy. A meta-analysis of observational studies has shown that circulating NRG4 levels are positively associated with glucose levels, but the exact role of circulating NRG4 in the pathogenesis of T2DM is still unclear [54].
NAFLD is emerging as one of the most common chronic liver diseases worldwide [55, 56]. Simple steatosis may progress to steatohepatitis, fibrosis, and hepatocellular carcinoma or manifest over a long period of time as only a small accumulation of lipids in the liver. Therefore, timely diagnosis and identification of new targets for the treatment of NAFLD and its complications are particularly important.
Specific binding assays on histological sections allowed the identification of the liver as the main target tissue of NRG4 [23]. The functions of NRG4 in the liver include regulation of metabolism and protection of hepatocytes. The protective effect of NRG4 was confirmed in a study in which NRG4-overexpressed adipose-derived MSCs (ADSCs) were transplanted into mice [57]. These mice had low plasma and liver triglyceride and total cholesterol levels compared with mice treated with regular ADSCs. Recently, recombinant NRG4-Fc protein was shown to inhibit pathological reprogramming of the liver microenvironment in NASH and prevent hepatocellular carcinoma (HCC) formation in mice [58]. In addition, transplantation of NRG4-ADSCs decreased the expression of SREBP1c and fatty acid synthase [57].
Li et al. (2021) [59] identified two rare missense mutations, p.R44H and p.E47Q, in the EGF-like domain in the NRG4 gene in Chinese patients with metabolic syndrome and obesity. Examination of these mutations showed that NRG4 has a protective effect in NAFLD. Experiments with recombinant mice showed that E47Q is a gain-of-function mutation. NRG4 with this amino acid substitution more potently activated ERBB4 phosphorylation and suppressed de novo lipogenesis in hepatocytes via the ERBB4-STAT5-SREBP-1C pathway compared with WT-NRG4. The R44H mutation is a loss-of-function, and this NRG4 does not bind to ERBB4. Thus, genetic variations in NRG4 may result in mutant proteins with aberrant functions. Impaired or enhanced NRG4 function may be either a risk or protective factor in NAFLD and associated metabolic disorders.
In addition to the mechanisms described above, NRG4 is able to activate autophagy via the AMPK/mTOR-dependent signaling pathway [60] (Fig. 2). Studies in aged C57BL/6 mice fed a high-fat diet and L-02 cells treated with palmitic acid confirmed that NRG4 administration activates autophagy through activation of the microtubule-associated light chain 3B-II (LC3BII) gene and a decrease in p62 [61]. AMPK/mTOR-dependent autophagy is able to reduce lipid accumulation and inflammation in hepatocytes [62]. Since hepatocyte apoptosis is the main event in the initiation and maintenance of fibrosis, autophagy can be considered an anti-fibrogenic process required for liver cell survival.
A study by De Munck et al. (2021) [63] failed to confirm the involvement of NRG4 in the pathogenesis of NAFLD in humans. In another study, decreased plasma Nrg4 levels are associated with increased oxidative stress, inflammation, and dyslipidemia [64]. Decreased plasma levels of NRG4 have been found in children with NAFLD [65]. Tutunchi et al. [66] also showed a correlation between decreased NRG4 levels and BMI, waist circumference, TG, cholesterol and HDL levels, and the HOMA-IR index. In addition, NRG4 is an independent predictor of NAFLD [67].
Thus, NRG4 possesses several mechanisms that inhibit the progression of NAFLD including reducing the accumulation of lipids in liver cells, activating autophagy, and suppressing apoptosis and necroptosis. However, these mechanisms have only been confirmed in cell lines or animal models. The role of NRG4 in the pathogenesis of NAFLD is still uncertain and requires further investigation.
The role of NRG/ERBB signaling in the cardiovascular system and understanding how it responds to physiological and pathological stress is a rapidly evolving area of science. Studies showed that ERBB receptor expression in tissues and cells of the cardiovascular system varies both spatially (in subcellular localization and tissue distribution) and temporally (e.g., developmental stage, physiological and pathological stress) [68]. Expression of all ERBB receptors, including ERBB1, ERBB2, ERBB3, and ERBB4, was detected on cardiac endothelial cells, fibroblasts, and highly proliferative cells. ERBB2 and ERBB4 receptors are expressed on adult cardiomyocytes. ERBB3 and ERBB4 are expressed on monocytes and cardiac macrophages [68].
NRG4, as an autocrine, paracrine, or endocrine factor, can activate the tyrosine kinases ERBB4 and presumably ERBB3 in the cardiovascular system, which are important transcriptional regulators of cardiomyocyte proliferation and both glucose and lipid metabolism and thermogenesis. NRG4 levels are inversely related to the severity of ischemic heart disease (IHD) [69]. Decreased levels of NRG4 correlated with the presence of IHD and were negatively associated with the SYNTAX score. Adjustment for age, sex, BMI, and HbA1C failed to attenuate this association. In another study, a negative association was found between NRG4 serum levels and the risk of developing acute coronary syndrome (including acute myocardial infarction and unstable angina), whereas no such association was found for stable angina. A negative association between NRG4 levels and the extent of vascular damage was also demonstrated [70]. A decrease in NRG4 levels was associated with an increase in carotid intima thickness [71], an increase in high-sensitivity C-reactive protein concentration [72], an increase in angiographic severity of IHD [69] and the acute coronary syndrome [70]. These results suggest that NRG4 may be a novel adipokine with protective properties against the progression of IHD.
Wang H. et al. [60] demonstrated a protective effect of NRG4 in diabetic cardiomyopathy. They found that autophagy mediates myocardial improvement through NRG4 intervention and that NRG4 treatment alleviates diabetic cardiomyopathy by promoting autophagy [73]. AMPK is a key autophagy activator that suppresses mTOR, a negative regulator of autophagy. NRG4 can activate AMPK, inhibit mTOR, and thereby induce autophagy both in vivo and in vitro [60]. Deletion of NRG4 (NRG4 -/-) in mice has been shown to exacerbate endothelial dysfunction and promote inflammation. Wang H. et al. [62] demonstrated that the anti-atherosclerotic activity of NRG4 is mediated via ERBB4–AKT–NF-kB signaling. NRG4 represses the transcriptional activity of NF-kB by prior activation of AKT [74].
The cardioprotective effects of NRG4 may be associated with a number of direct and indirect mechanisms. Animal studies showed a direct relationship between NRG4, ERBB4, and activation of downstream signaling pathways involved in cardiomyocyte proliferation. The NRG4/ERBB4 signaling pathway not only controls cardiomyocyte formation, differentiation, and migration [75], but also plays a critical role in the repair of cardiac injuries [36]. NRG4 has anti-apoptotic effects on cardiomyocytes through activation of PI3K/Akt (Fig. 2) and prevents apoptosis of endothelial cells, smooth muscle cell senescence, and also reduces pro-inflammatory activation of tissue macrophages and their differentiation into foam cells, thereby inhibiting the progression of atherosclerosis [68]. NRG4 is thus able to regulate the response of the cardiovascular system to inflammation or injury. This adipokine has beneficial effects in both diastolic and systolic heart failure and also prevents atherosclerosis. Through its direct action on cardiomyocytes, endothelial cells, immune cells and fibroblasts, NRG4 is able to exert a therapeutic effect in various forms of cardiopathology.
Mature miRNAs bind to mRNA according to the complementarity principle. Depending on the completeness of binding, the mRNA is either translationally repressed by deadenylation and decapping (if complementarity is only partial) or cleaved and degraded by target hydrolysis (if complementarity is complete) [76]. In most cases, microRNAs only partially bind to the target and only repress protein translation, which is why they are also referred to as “transcriptome sculptors”. Incomplete complementarity leads to the phenomenon of “multifunctionality”—each microRNA can regulate hundreds of target genes (multiplicativity), and conversely, each target gene can be regulated by numerous microRNAs (cooperativity), suggesting a complex genetic network [77, 78]. MicroRNAs regulate almost all biological processes in metabolically active tissues. Therefore, their deregulation may serve as an important indicator of pathology in these organs.
As previously noted, ERBB4 is able to activate the phosphorylation of MAPK42/44
(or ERK1/2), which is a negative regulator of adipogenesis [26, 27, 28]. In the study
by Wang et al. (2022) [79], ERBB4 was the target gene for
miR-146a-5p. miR-146a-5p interacts with ERBB4 and thereby promotes
pre-adipocyte differentiation via the ERK1/2/PPAR-
Huang et al. (2022) [80] demonstrated that miRNa-205-5p inhibits ERBB4 and AKT phosphorylation in aortic vascular smooth muscle cells induced by oxidized low-density lipoprotein. Proliferation and migration of aortic smooth muscle cells are closely related to the development of atherosclerosis. miRNa-205-5p decreased the cell viability of smooth muscle cells induced by oxidized low-density lipoprotein by increasing the expression of Bax/Bcl-2 and cleaved caspase-3, which are required to stimulate cell apoptosis. This suggests that miR-205-5p plays an important role in the initiation and development of atherosclerosis and represents a novel target for alleviating the symptoms of atherosclerosis.
In cardiac myocytes from STAT-deficient mice, knockdown of miR-199a leads to upregulation of ERBB4 protein. Conversely, Erbb4 is downregulated while miR-199a is overexpressed, and miR-199a has been shown to bind directly to Erbb4. Knockdown of Erbb4 in cardiomyocytes by a different mechanism (siRNA) leads to a decrease in pyruvate content and metabolic activity [81]. Upregulation of miR-146a expression in mouse cardiomyocytes after anthracycline treatment with doxorubicin enhanced myocyte damage via targeting the ERBB4 gene [82]. Peripartum cardiomyopathy develops in females before or after child birth. Studies have shown an increase in nine microRNAs (miR-199a-5p and miR-199a-3p, miR-145a-5p, miR-130a-3p, miR-135a-5p, miR-221-3p, miR-222-3p, miR-23a-3p, and miR-19b-3p) in plasma from patients with PPCM, all targeting the ERBB4 gene. Overexpression of these microRNAs reduces the expression of ERBB4 in cardiomyocytes. In a mouse model with cardiomyocyte-specific suppression of Erbb4, it was shown that deficiency of Erbb4 during pregnancy can lead to left ventricular dilatation and enlargement of cardiomyocytes without evidence of myocardial apoptosis or inflammation. These findings suggest that ERBB4 is required to protect the maternal heart from perinatal stress [83].
Recent studies have shown that miR-193b-3p regulates keratinocyte activity, in part by targeting ERBB4. An association between hyperactivation of ERBB4 protein and miR-193b-3p deficiency was found in psoriasis [84]. In addition, miR-204-5p, miR-320b, miR-323a-3p, and miR-331-3p were upregulated in the anterior cingulate cortex and habenula of individuals with major depressive disorders. ERBB4 gene expression was downregulated in both regions and affected by miR-323a-3p in vitro [85].
In our previous study of microRNA profiling in blood serum, we showed that 23 microRNAs were differentially expressed in patients with steatohepatitis and in patients with steatosis [86]. ERBB signaling was significantly enriched in the NASH group but not in patients with steatosis, suggesting a significant impact of ERBB signaling on metabolism. The ERBB4 gene was thought to be regulated by miR-423-5p, miR-200c-5p, miR-93-5p, miR-27a-3p, miR-26a-5p, miR-23a-3p, miR-15b-5p, miR-1-3p, let-7c-5p, and let-7a-5p. The NRG4 gene was thought to be regulated by let-7a-5p, let-7c-5p, miR-423-5p, miR-93-5p, miR-23a-3p, and miR-15b-5p (Fig. 5A, Ref. [86, 87, 88]).
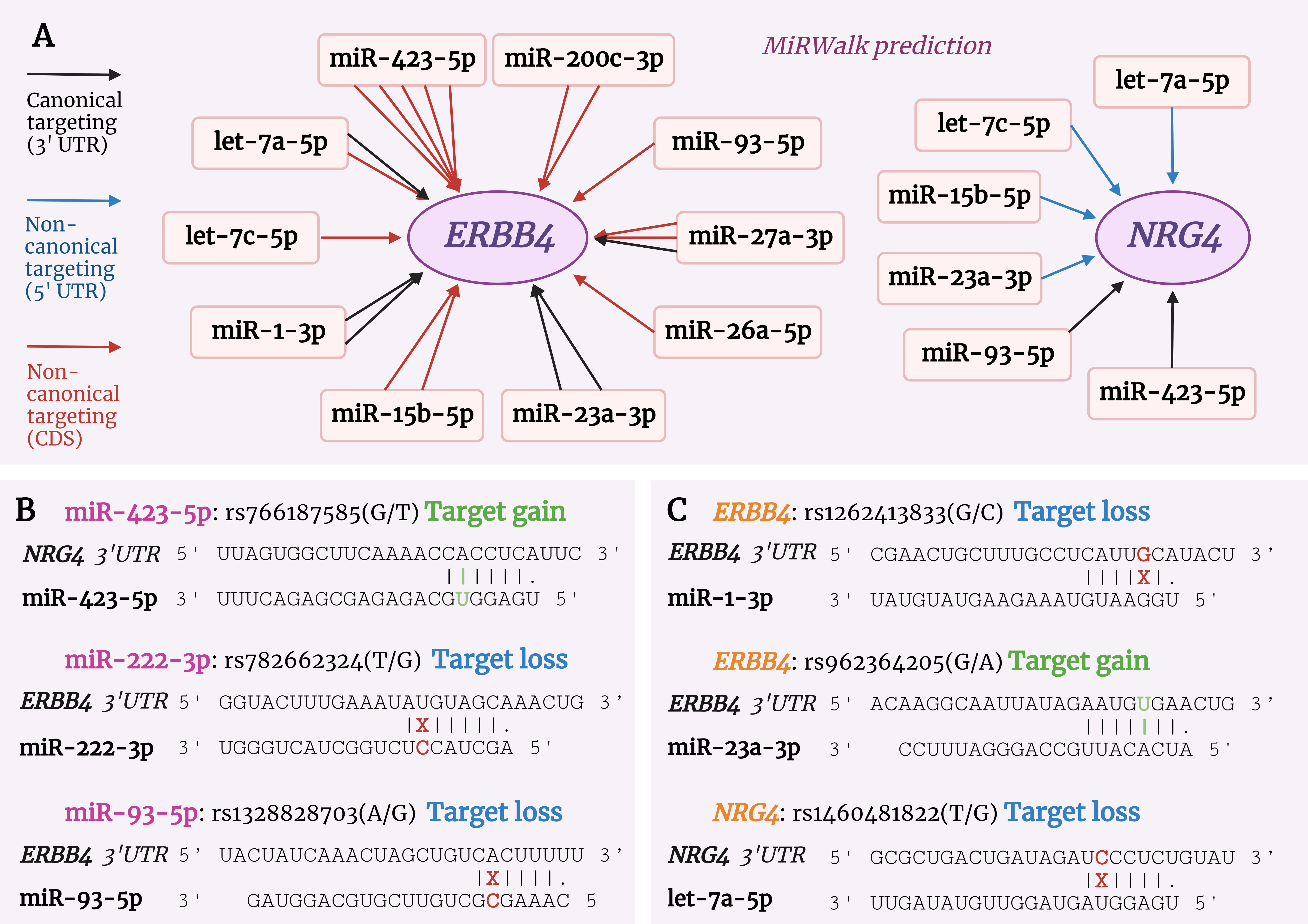 Fig. 5.
Fig. 5.MicroRNA-dependent regulation of the ERBB4 and NRG4 genes. (A) Relationships between microRNAs and genes of the ERBB signaling pathway. In our study [86], microRNAs were differentially expressed in patients with NASH, and their interaction with the ERBB4 and NRG4 genes was bioinformatically predicted by the MirWalk platform [87]. Multiple arrows of a single microRNA indicate that this microRNA is likely to have multiple targeting sites on the mRNA sequence of the gene. The color of the arrow indicates the type of targeting, as shown in the legend. (B,C) Single nucleotide polymorphism (SNP) in miRNAs and the ERBB4 and NRG4 genes, targeting predicted by miRNASNP-v3 [88]. (B) Presence of SNPs in the seed region of miR-423-5p, miR-222-3p and miR-93-5p and the effects of these SNPs on targeting of the NRG4 and ERBB4 genes. (C) Presence of SNPs in the ERBB4 and NRG4 genes affecting the binding strength of these genes to miR-1-3p, miR-23a-3p and let-7a-5p miRNAs.
Recently, the study of SNPs in miRNAs and their target genes has attracted increasing interest. Therefore, we decided to investigate how the interaction between ERBB4 and NRG4 and the microRNAs we identified in our previous study can be modulated by the presence of SNPs. Using the miRNASNP-v3 database [88], we found a SNP in miR-423-5p (rs766187585(G/T)) in its seed region that enhances the binding of this miRNA to the NRG4 gene. The presence of such a substitution in the miRNA is likely to reduce the expression of NRG4 to a greater extent (Fig. 5B). In contrast, the presence of rs782662324(T/G) in hsa-miR-222-3p and rs1328828703(A/G) in miR-93-5p in their seed regions may abolish full complementarity to the ERBB4 gene and lead to downregulation of miRNA function (Fig. 5B). Due to different target prediction algorithms, MirWalk does not predict ERBB4 as a target for hsa-miR-222-3p, whereas miRNASNP-v3 does, which requires further study. A further search in the database also revealed several SNPs in ERBB4 and NRG4 that affect binding to the miRNAs identified in our study. For example, rs1262413833(G/C) and rs962364205(G/A) in the ERBB4 gene affect the interaction with miR-1-3p and miR-23a-3p by enhancing and attenuating the interaction with the miRNA. For NRG4, the SNP mutation rs1460481822(T/G) was found to affect binding to let-7a-5p (Fig. 5C).
An analysis of the literature revealed that data on miRNA-dependent regulation of the NRG4-ERBB4 pathway in diseases associated with metabolic disorders is limited and further research is needed. It has been shown that miR-146a-5p, miRNa-205-5p, miR-199a, miR-193b-3p, miR-145a-5p, miR-130a-3p, miR-135a-5p, miR-221-3p, miR-222-3p, miR-23a-3p, and miR-19b-3p directly or indirectly affect the level of ERBB4 in various pathologies. In our literature search, we did not find any work analyzing the interaction of microRNAs with NRG4. Therefore, to the best of our knowledge, this review is the first to mention the concept of miRNA-dependent epigenetic regulation of NRG4. We hypothesize that the presence of SNPs in miR-222-3p and miR-423-5p, miR-93-5p in their seed regions and SNPs in the ERBB4 and NRG4 genes, may affect the interaction between miRNAs and target genes.
NRG4 is capable of acting as an autocrine, paracrine, and endocrine adipokine. ERBB4 is a transmembrane tyrosine kinase receptor and its gene is alternatively spliced in two specific regions of the primary transcript: the C-terminal cytoplasmic domain (CYT1 and CYT2 variants) and the juxtamembrane ectodomain (JM-A, JM-B, JM-C, and JM-D variants). The CYT-2 isoform has a deletion of exon 26, which causes it to lose the ability to directly activate the downstream signaling proteins PI3KR1-3 and CBL. Alternative splicing of exons 16 and 15b of the ERBB4 gene results in the JM-A and JM-B isoforms. Isoform JM-D contains both exons 16 and 15b, whereas JM-C contains none. The soluble intracellular domain of ERBB4—4ICD—is able to enter the mitochondrial matrix and nucleus and enhance the translocation of transcription factors and act as a transcription cofactor.
In humans, the NRG4 protein is much shorter than other members of the neuregulin family. Five splice isoforms of NRG4 have been discovered: A1-2 and B1-3; only the first two isoforms have the ability to activate ERBB4. Activation of ERBB receptors leads to mobilization of a variety of metabolic pathways involved in angiogenesis, lipid and carbohydrate metabolism, growth, proliferation, regulation of cell death, apoptosis, and autophagy. The PI3K/AKT pathway can be directly activated by dimers containing ERBB3/4. In M1 macrophages, NRG4 is able to induce the mitochondrial apoptosis pathway. In the presence of metabolic dysfunction and the development of chronic inflammation, NRG4 expression decreases in adult and pediatric adipose tissue and in animal models. In contrast, levels of NRG4 production are inconsistent in studies of patients with type 2 DM, which may be related to the experimental design, the specific analytical methods that were utilized, and to a greater extent, the demographics of the population that was studied. Further studies are needed to determine the effects of drug treatment of type 2 DM and its effects on serum NRG4 levels and mRNA expression in metabolically active tissues. In the liver, NRG4 is able to reduce lipid accumulation in liver cells, activate autophagy, and suppress apoptosis and necroptosis, all of which are protective mechanisms that inhibit the progression of NAFLD. However, the role of NRG4 in the pathogenesis of NAFLD in humans is still uncertain and needs to be investigated. NRG4 has direct effects on cardiomyocytes, endothelial cells, immune cells, and fibroblasts, and may have important therapeutic effects in various forms of cardiac pathology.
There is very little data on miRNA-dependent regulation of the ERBB pathway in diseases associated with metabolic disorders. An analysis of the literature revealed that the ERBB4 gene is regulated by miR-146a-5p, miR-205-5p, miR-199a, miR-193b-3p, miR-145a-5p, miR-130a-3p, miR-135a-5p, miR-221-3p, miR-222-3p, miR-23a-3p, and miR-19b-3p. It was shown for the first time that let-7a-5p, let-7c-5p, miR-423-5p, miR-93-5p, miR-23a-3p, and miR-15b-5p most likely regulate NRG4 gene expression. In addition, the presence of SNPs in the seed regions of miR-222-3p, miR-93-5p, and miR-423-5p may affect the interaction with ERBB4 and NRG4. Thus, the presence of mutations can lead to both gain and loss of function, which holds great potential for understanding the regulation of biological processes. This information will need to be verified in further experimental and clinical studies.
MV, MB, AK, OK, KY, VM—original draft preparation; MV, MB, AK, OK, VM, KY, AS, AM, NG—gathered information and performed material systematization; MV, MB, AS, AM—visualization; MV, MB, LL, EK—writing - review & editing; MV, LL—funding acquisition. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We acknowledge the esteemed Pavel Ivanov for his help and support.
This research was funded by the Grant of the President of the Russian Federation number MK-2072.2022.3, the State Assignment Grant number FZWM-2020-0010.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





